Towards Multi-Functional SiO2@YAG:Ce Core–Shell Optical Nanoparticles for Solid State Lighting Applications
Abstract
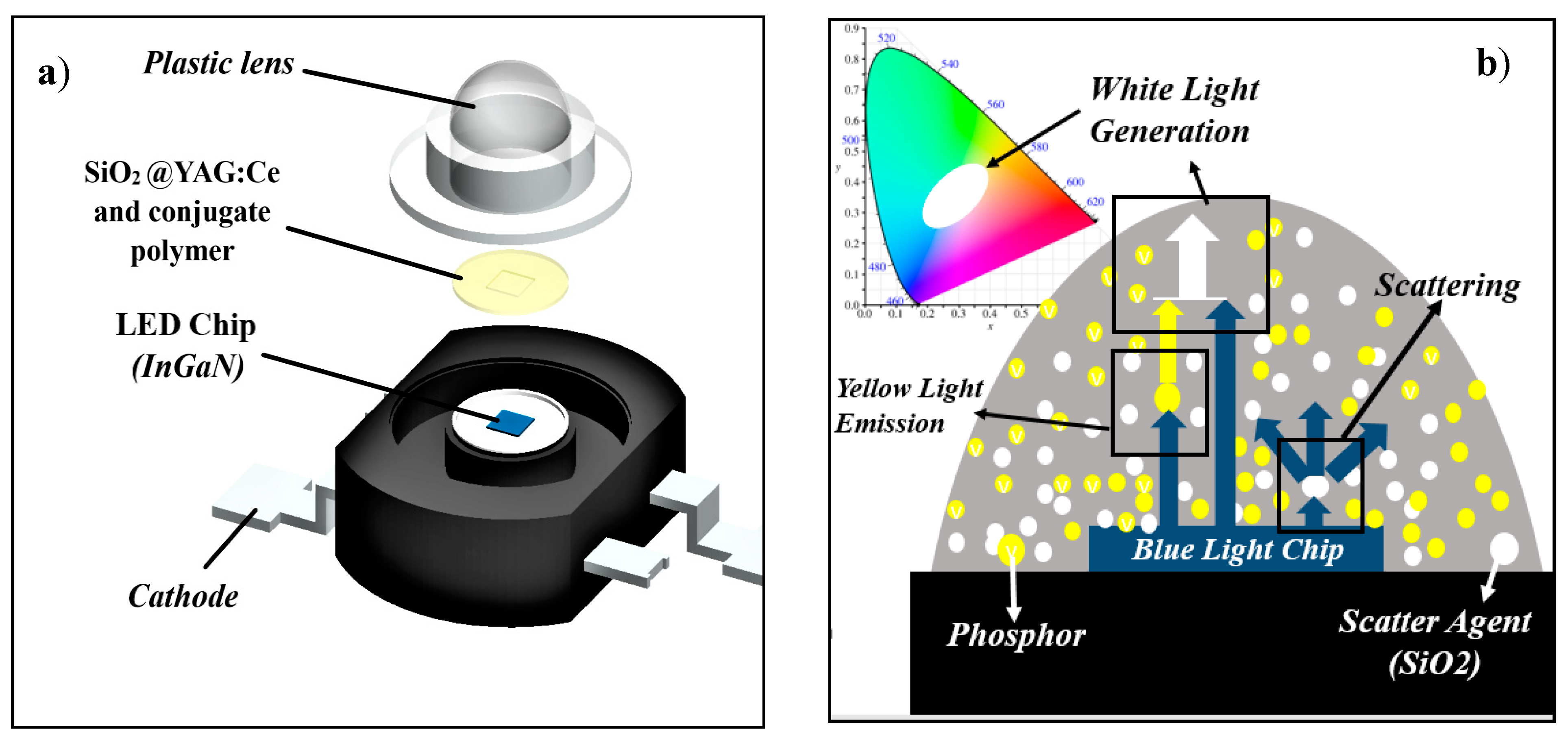
1. Introduction
2. Materials and Methods
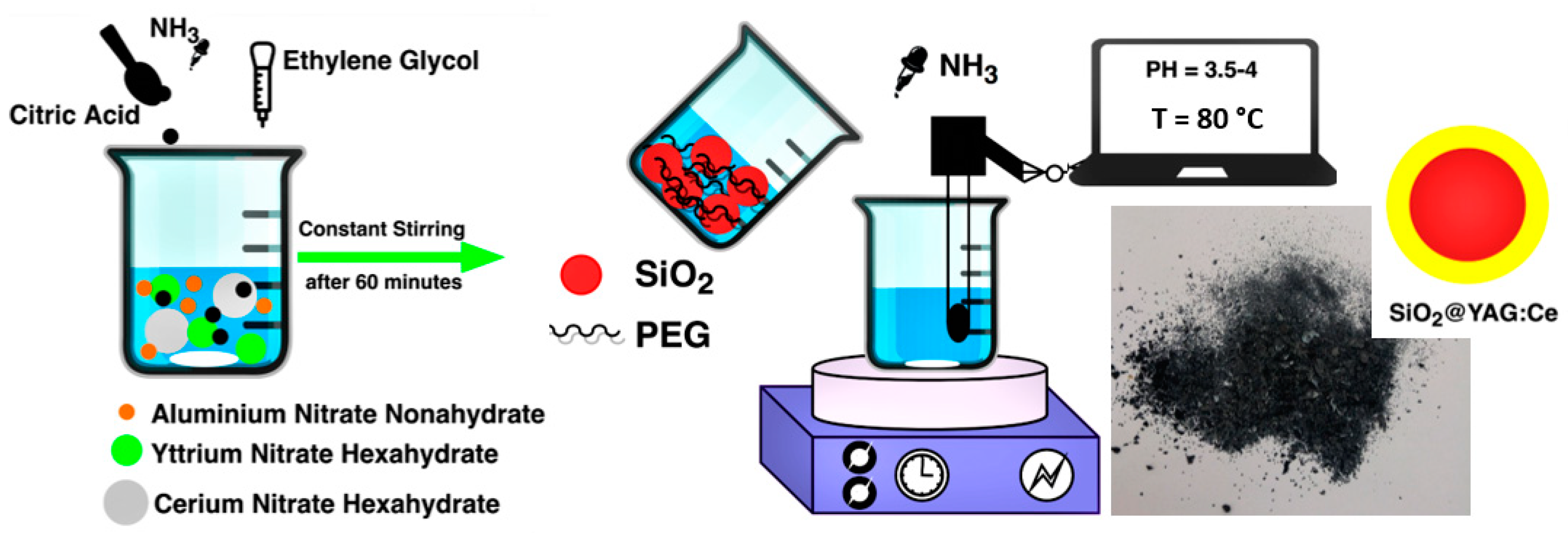
2.1. Synthesis of Core–Shell SiO2@YAG:Ce Nanoparticles
2.2. Characterization of Core–Shell Nanoparticles
3. Results and Discussion
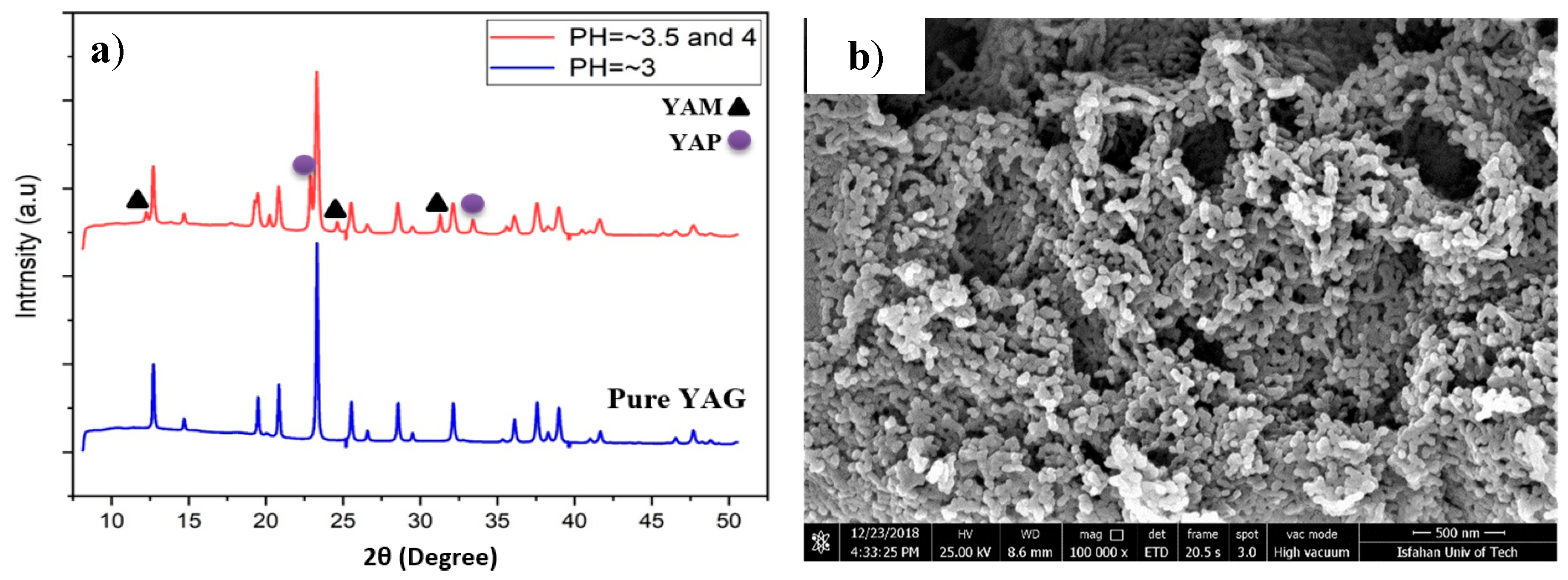
3.1. Analyses of Synthesized Phosphor Particles
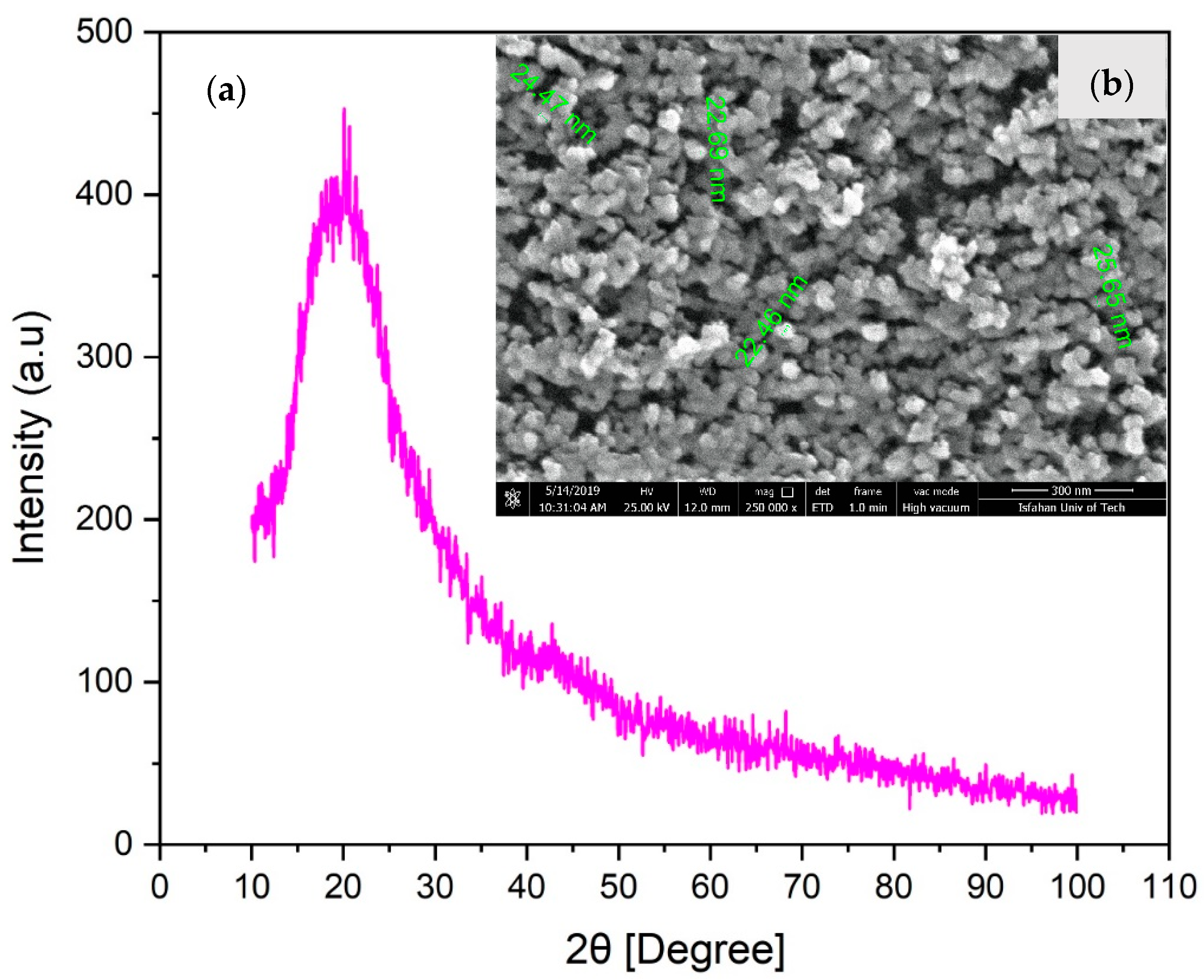
3.2. SAXS Analyses
3.3. HRTEM/TEM Observations
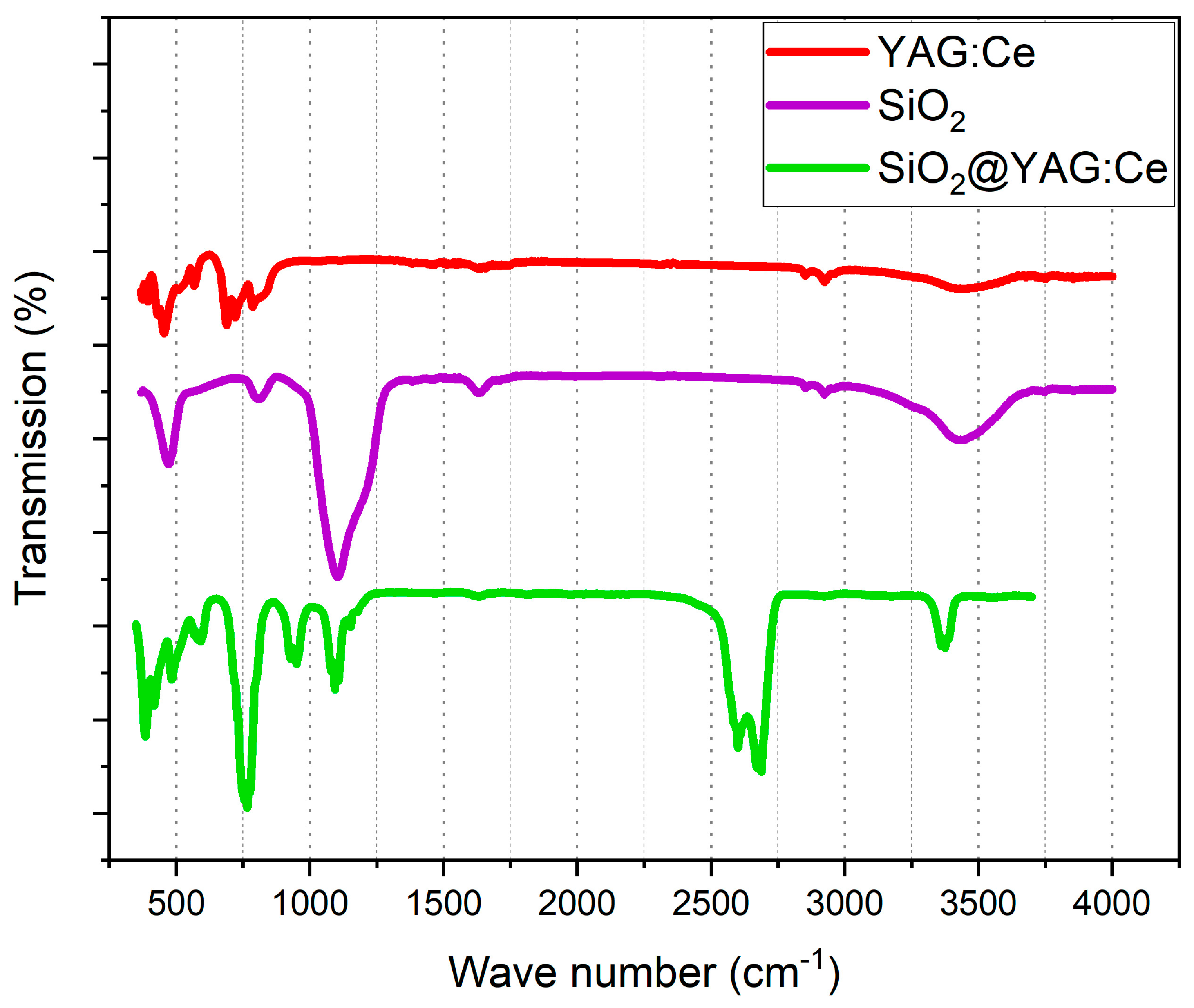
3.4. FTIR Analyses
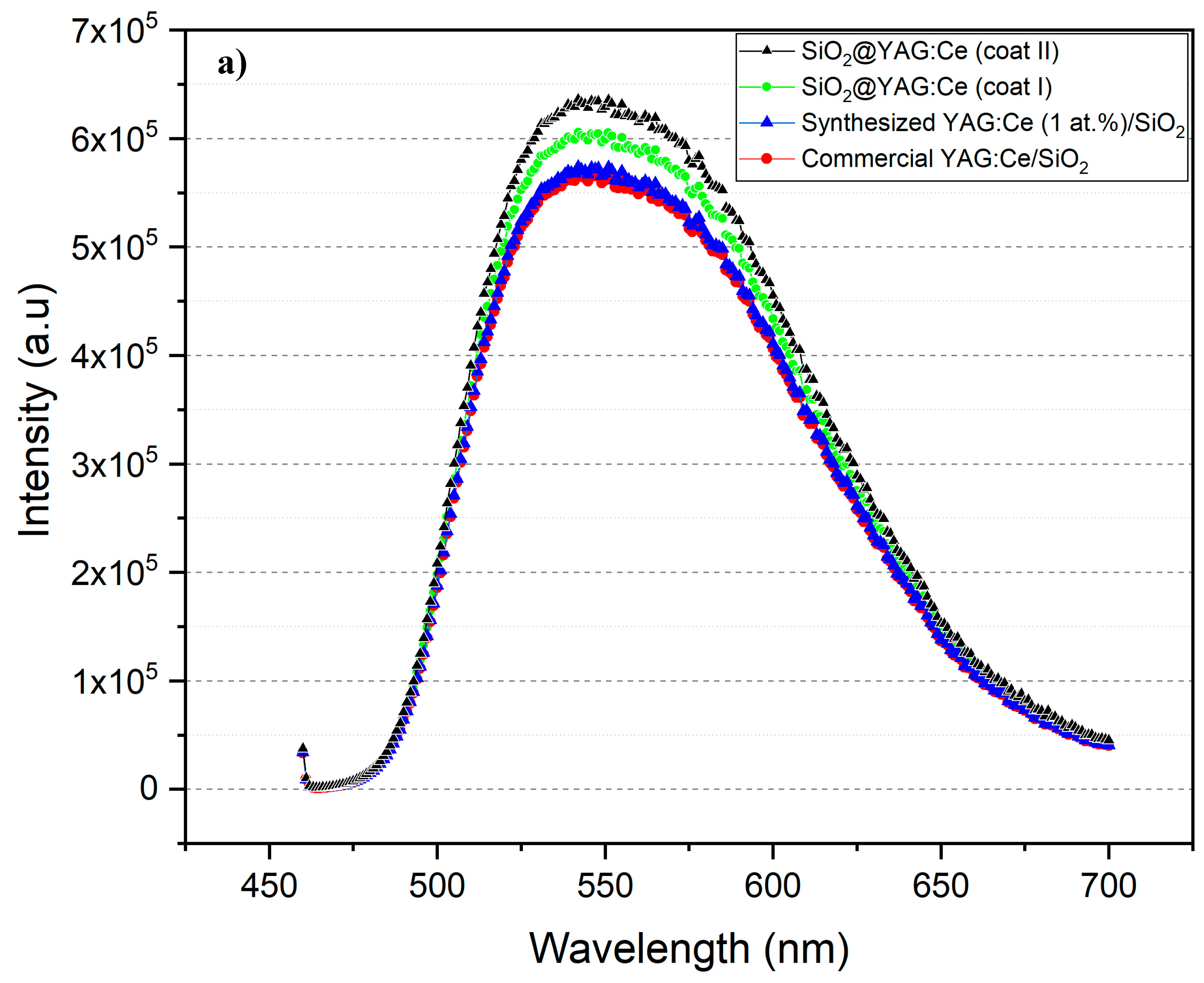
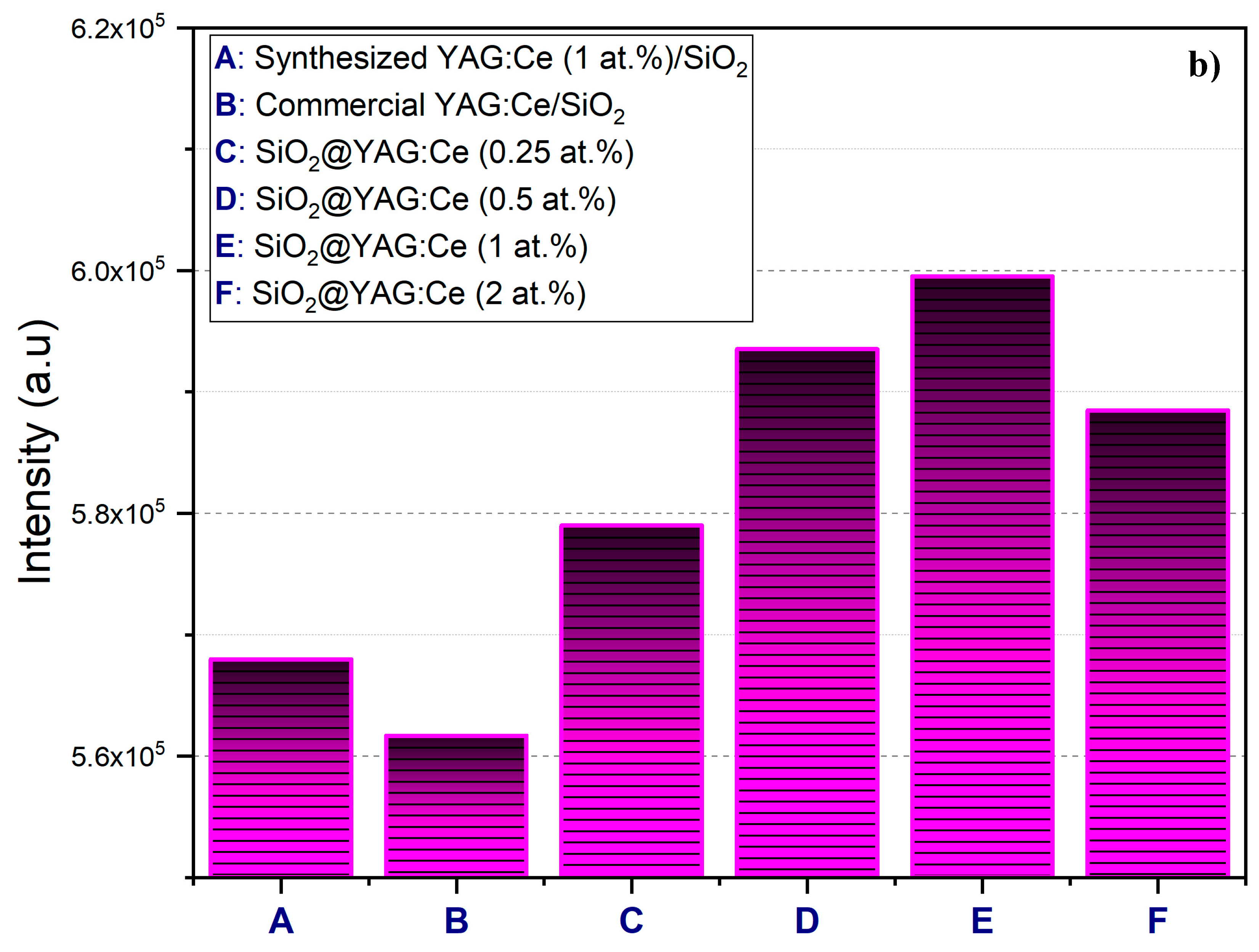
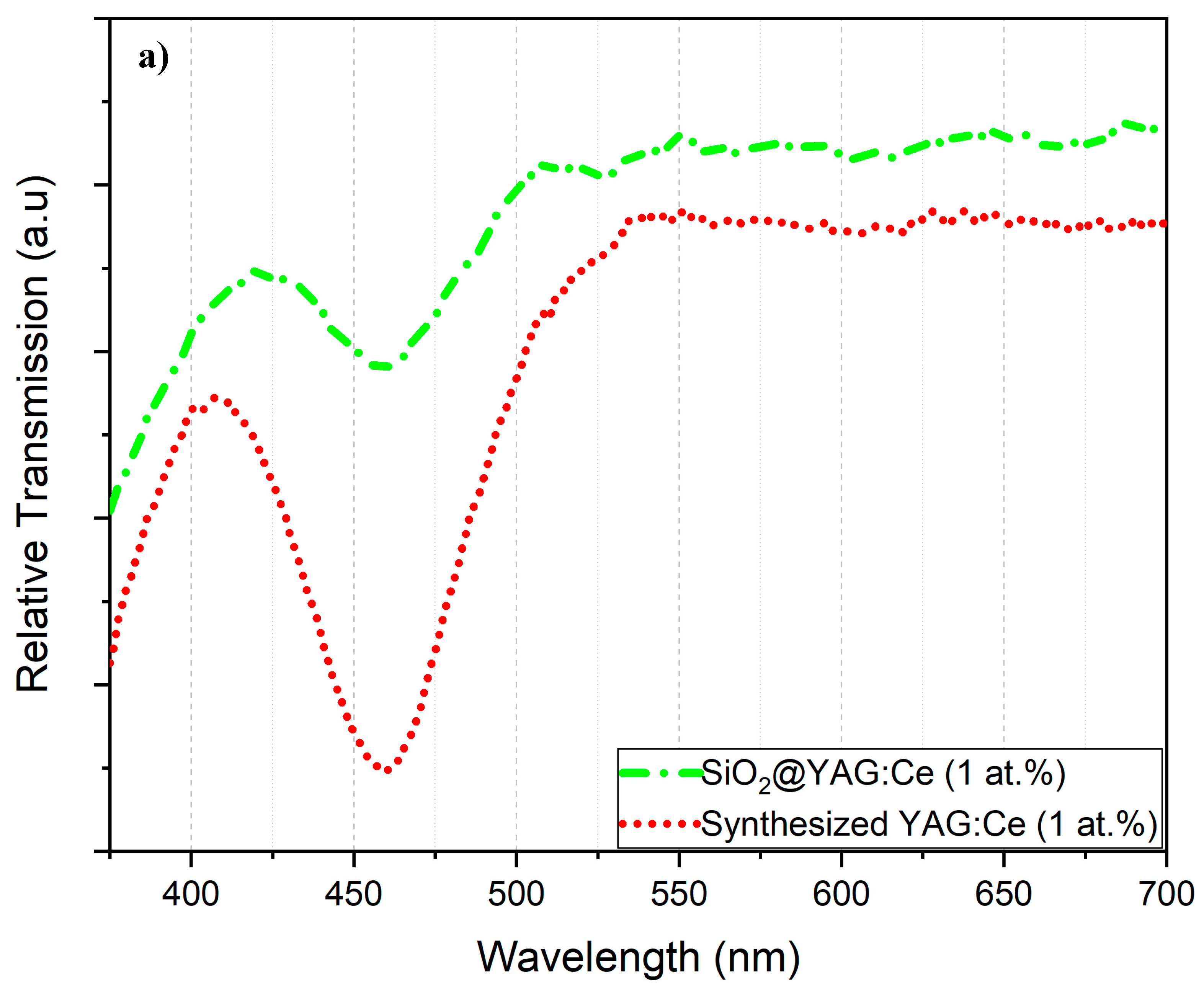
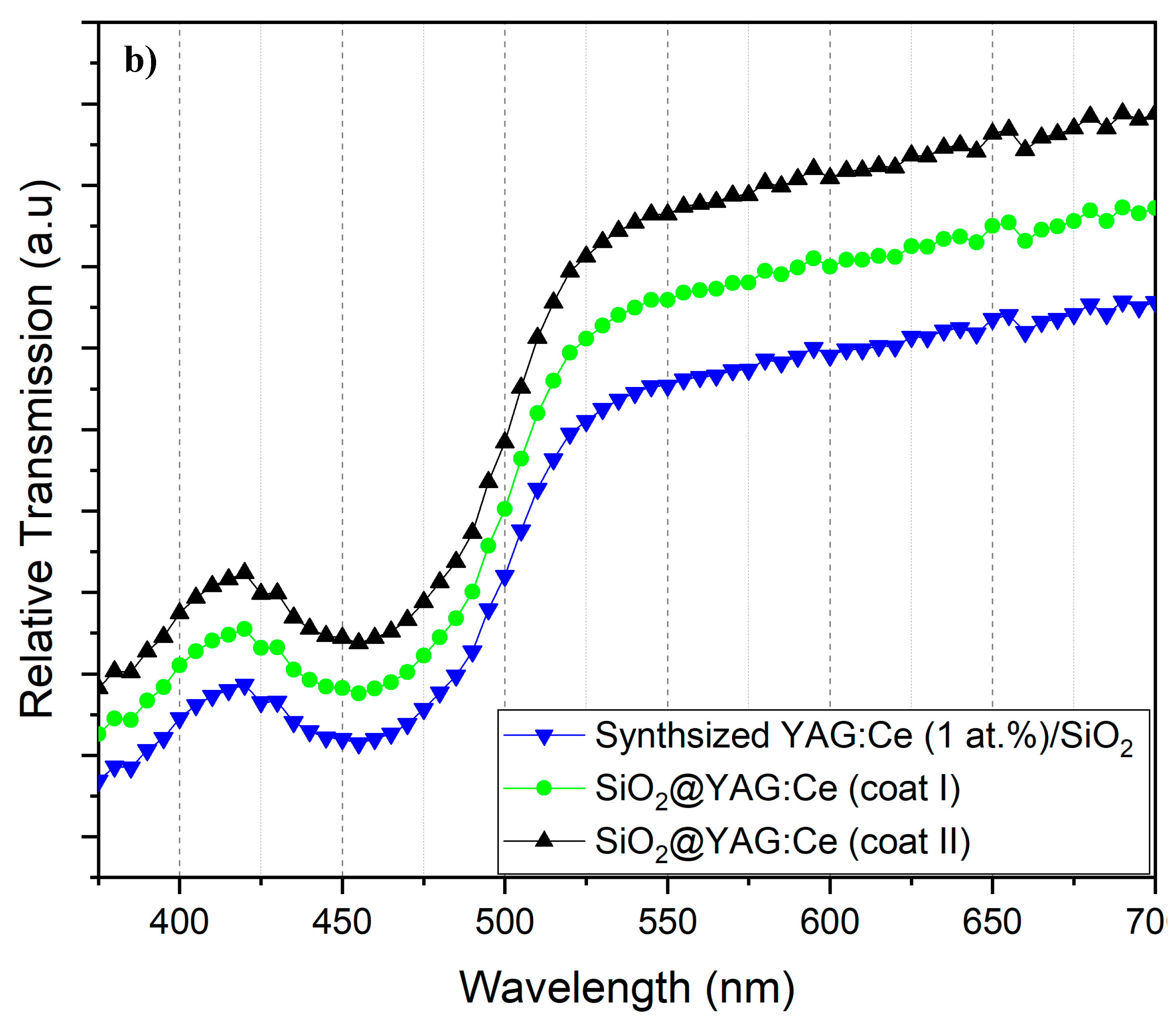
3.5. Photoluminescence (PL) and Diffuse Transmission Spectra (DTS) Analyses
4. Conclusions
- -
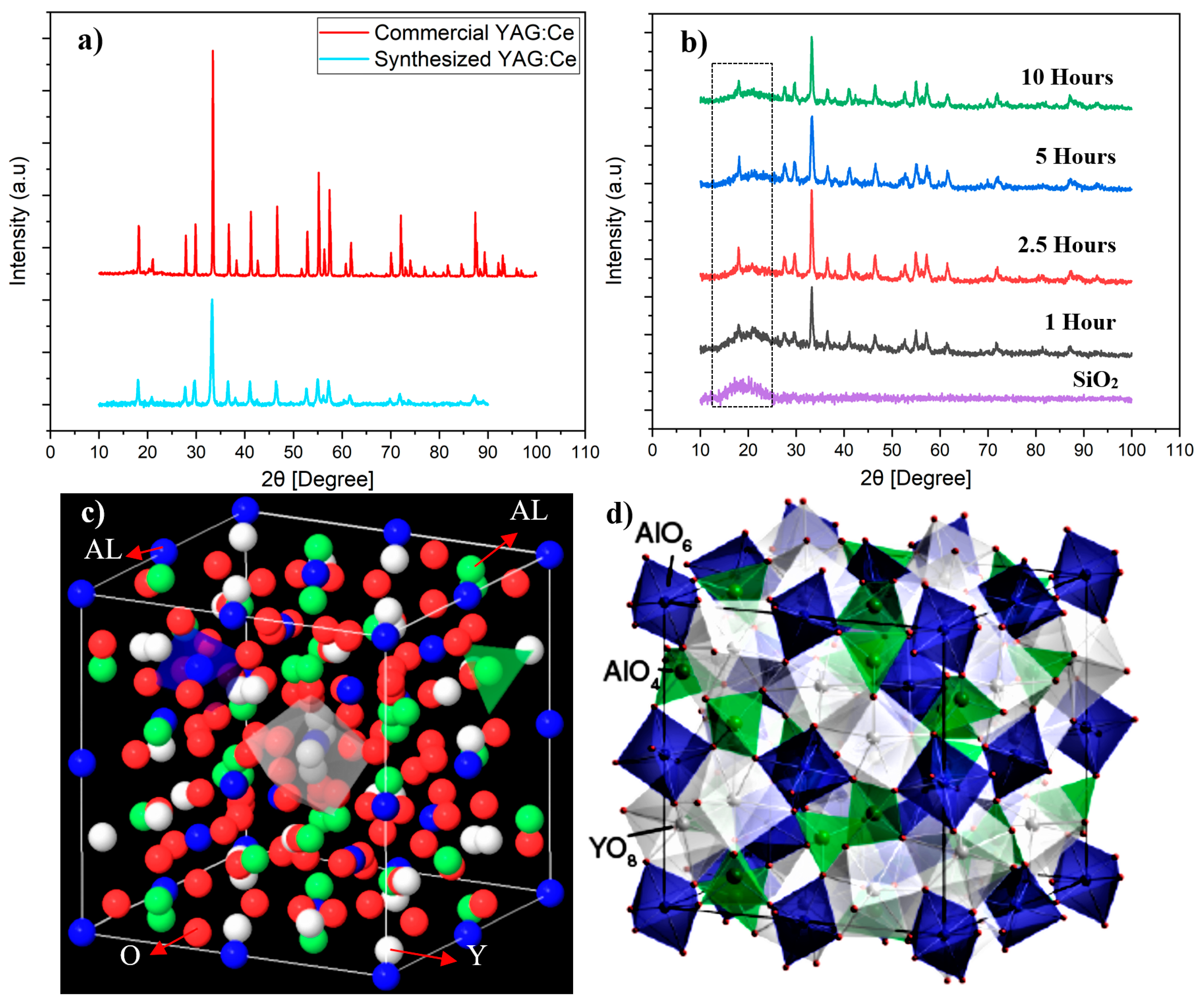
- Results showed that the final phase composition of synthesized powders largely depended on pH values. The optimum condition was achieved when pH was strictly controlled at pH = 3. Any deviation from pH = 3 towards higher values, and more specifically towards the 3.5–4.0 pH range resulted in the appearance of YAM (Y4Al2O9) and YAP (YAlO3) transitional phases in as-synthesized powders. Both phases are known to have adverse attribution to the optical characteristics of YAG:Ce powders.
- -
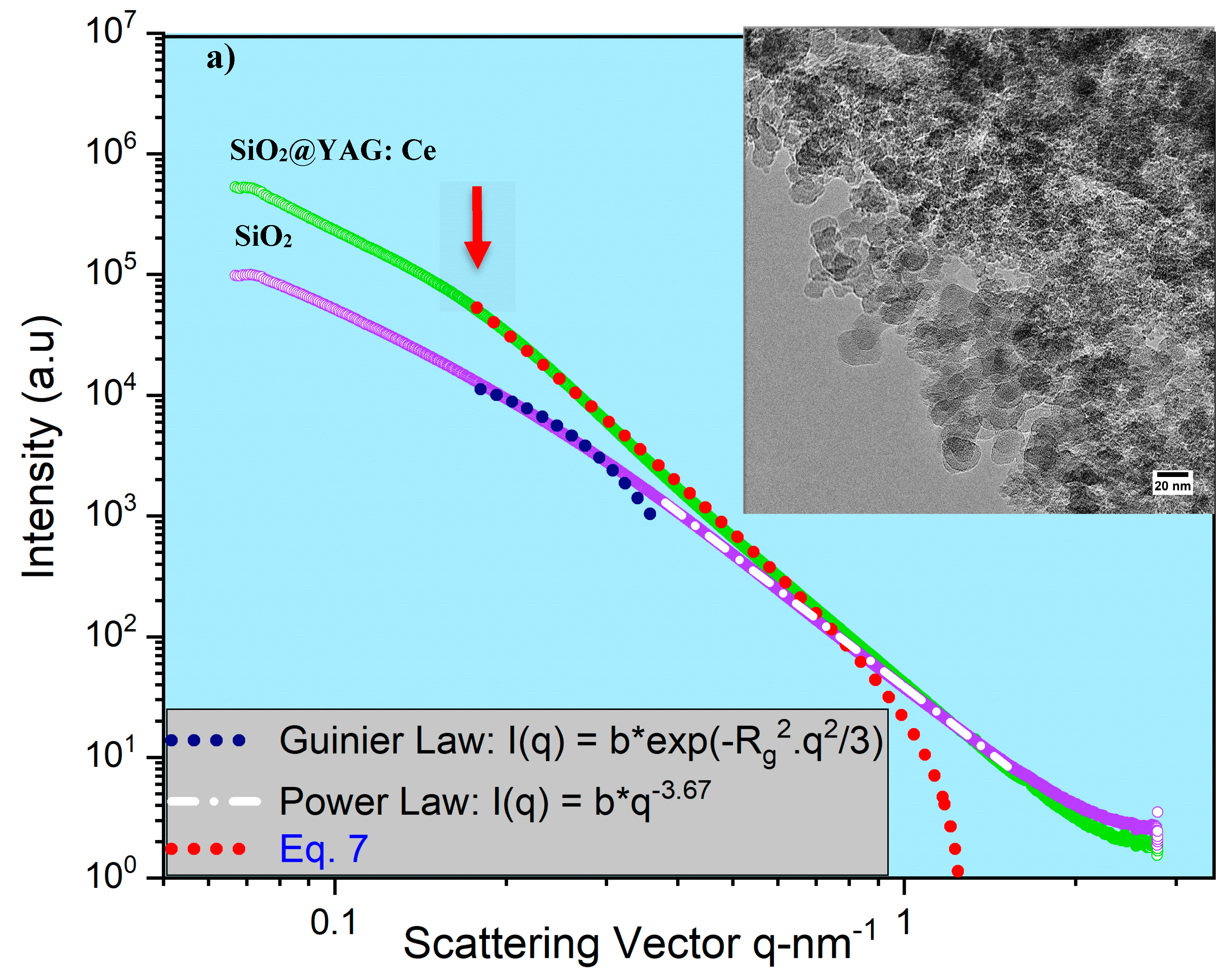
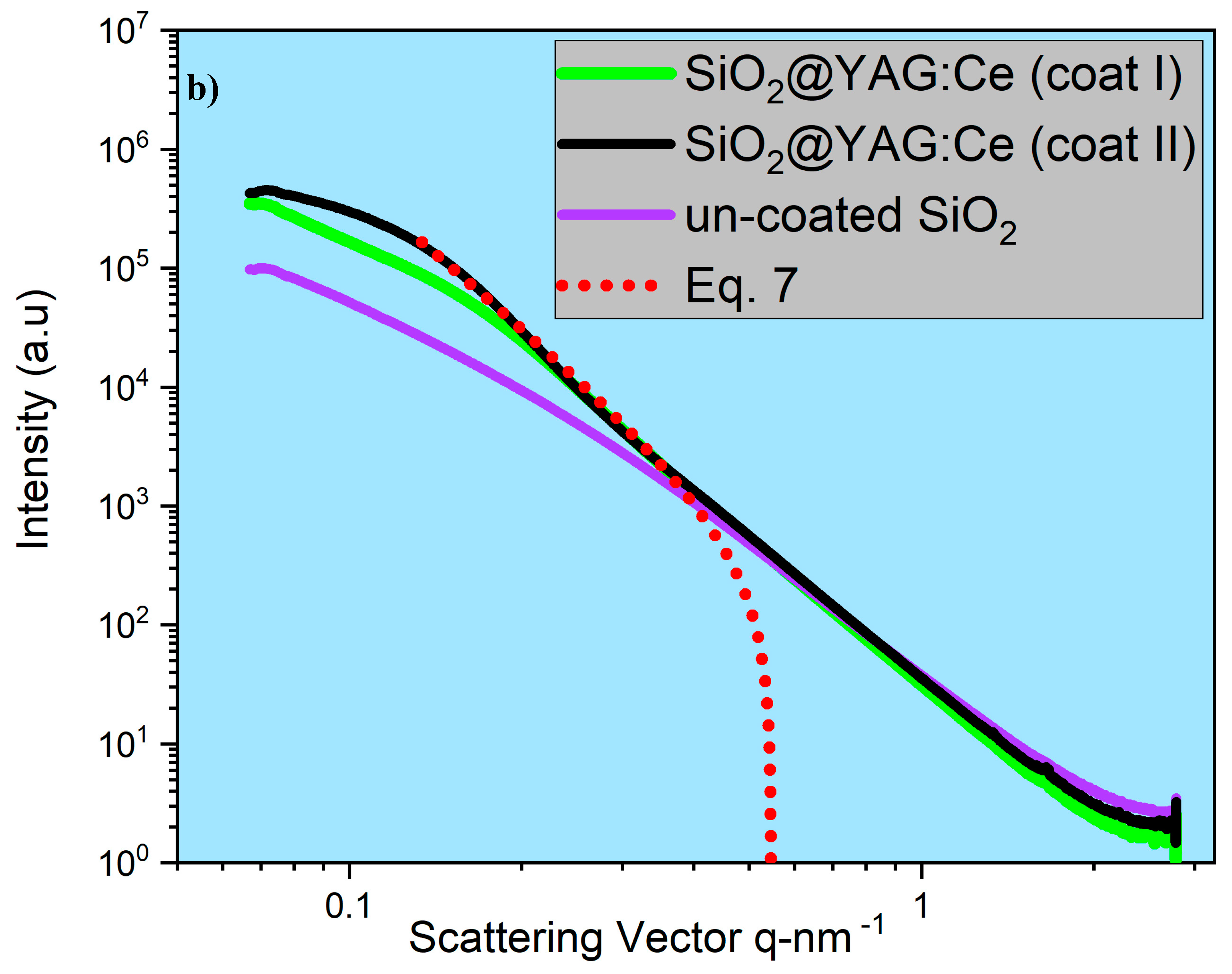
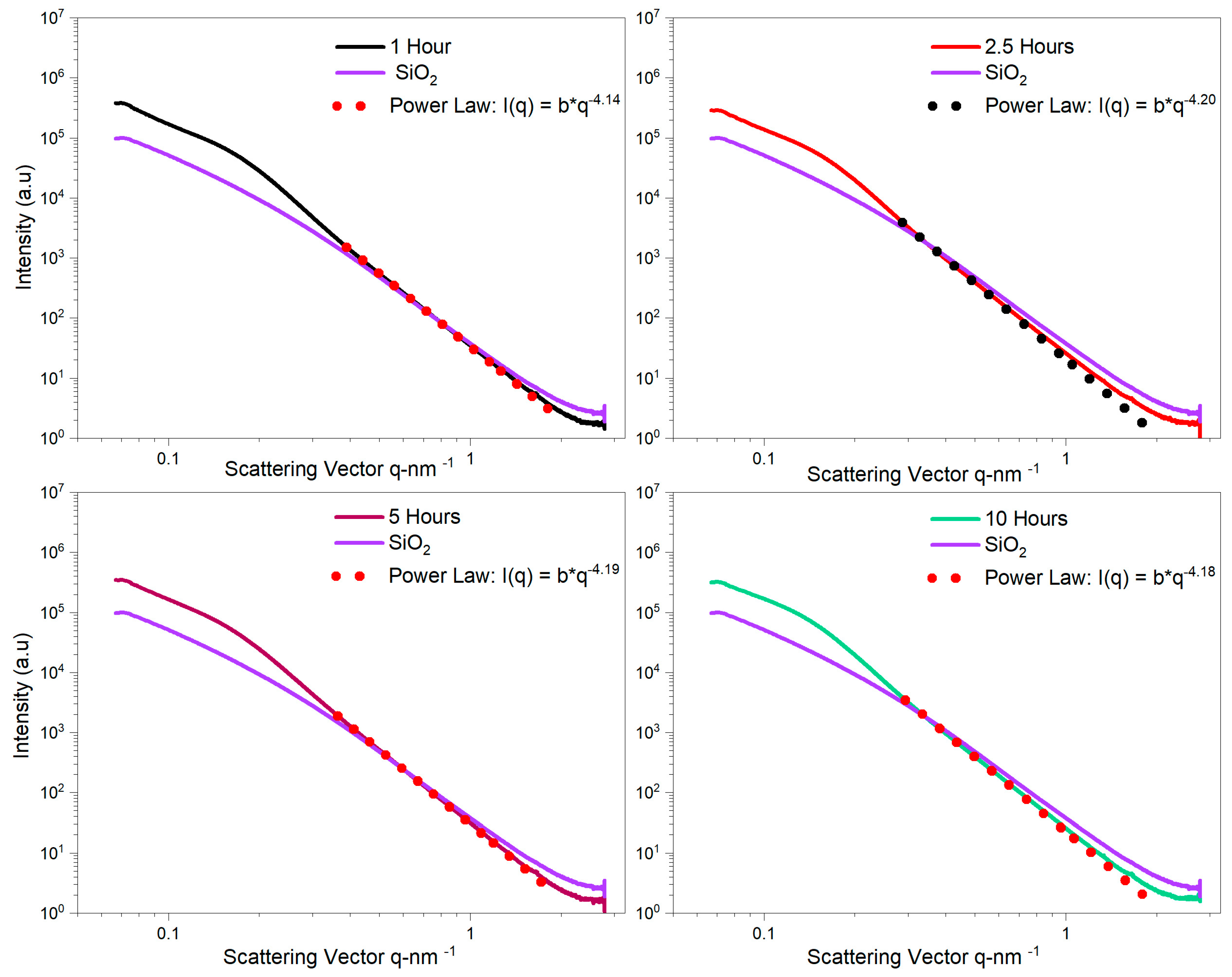
- SAXS analysis showed that the mean thickness of YAG:Ce shell after one and two coating cycles were 2.8 nm and 7.7 nm. This was in agreement in HRTEM observations.
- -
- Heating the sample up to 10 h hardly resulted in any change in the morphology of nanoparticles, inferring that SiO2 cores were still encapsulated by YAG:Ce shell, obviously showing perfect thermal stability of SiO2@YAG:Ce core–shell nanoparticles.
- -
- SiO2@YAG:Ce (1.0 at.% Ce) core–shell nanoparticles show the highest emission, compared to commercial and synthesized SiO2/YAG:Ce mixture composite.
- -
- The number of coating cycles played a vital role in enhancing the PL intensity of the core–shell particles. The increase in the PL intensity with double coating cycle was obviously attributable to the increase of the shell thickness (YAG:Ce) on the SiO2 cores, which in turn increased emitting ions (Ce3+) per core–shell particle.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Rajendran, M.; Samal, S.K.; Vaidyanathan, S. A novel self-activated (bluish-green) and Eu3+ doped (red) phosphors for warm white LEDs. J. Alloys Compd. 2020, 815, 152631. [Google Scholar] [CrossRef]
- Adam, J.; Metzger, W.; Koch, M.; Rogin, P.; Coenen, T.; Atchison, J.S.; König, P. Light emission intensities of luminescent Y2O3:Eu and Gd2O3:Eu particles of various sizes. Nanomaterials 2017, 7, 26. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.C.; Wang, D.Y.; Chen, T.M.; Lee, C.S.; Chen, K.J.; Kuo, H.C. A novel tunable green- to yellow-emitting β-YFS:Ce3+ phosphor for solid-state lighting. ACS Appl. Mater. Interfaces 2011, 3, 3195–3199. [Google Scholar] [CrossRef]
- Du, J.; Poelman, D. Facile synthesis of mn4+-activated double perovskite germanate phosphors with near-infrared persistent luminescence. Nanomaterials 2019, 9, 1759. [Google Scholar] [CrossRef]
- Ho, W.-J.; Liu, J.-J.; Lin, Z.-X.; Shiao, H.-P. Enhancing photovoltaic performance of GaAs single-junction solar cells by applying a spectral conversion layer containing Eu-doped and Yb/Er-doped phosphors. Nanomaterials 2019, 9, 1518. [Google Scholar] [CrossRef]
- Yazdan Mehr, M.; Bahrami, A.; van Driel, W.D.; Fan, X.J.; Davis, J.L.; Zhang, G.Q. Degradation of optical materials in solid-state lighting systems. Int. Mater. Rev. 2019, 65, 102–128. [Google Scholar] [CrossRef]
- Yazdan Mehr, M.; Toroghinejad, M.R.; Karimzadeh, F.; van Driel, W.D.; Zhang, G.Q. A review on discoloration and high accelerated testing of optical materials in LED based-products. Microelectron. Reliab. 2018, 81, 136–142. [Google Scholar] [CrossRef]
- Yazdan Mehr, M.; Volgbert, S.; van Driel, W.D.; Zhang, G.Q. Effects of graphene monolayer coating on the optical performance of remote phosphors. J. Electron. Mater. 2017, 46, 5866–5872. [Google Scholar] [CrossRef]
- Mehr, M.Y.; van Driel, W.D.; Zhang, G.Q. Reliability and lifetime prediction of remote phosphor plates in solid-state lighting applications using accelerated degradation testing. J. Electron. Mater. 2016, 45, 444–452. [Google Scholar] [CrossRef]
- Kiani Khouzani, M.; Bahrami, A.; Yazdan Mehr, M. Spark plasma sintering of Stellite®-6 superalloy. J. Alloys Compd. 2019, 782, 461–468. [Google Scholar] [CrossRef]
- Yazdan Mehr, M.; van Driel, W.D.; Zhang, G.Q. Progress in Understanding color maintenance in solid-state lighting systems. Engineering 2015, 1, 170–178. [Google Scholar] [CrossRef]
- Consulting, N. Energy Savings Forecast of Solid-State Lighting in General Illumination Applications; Navigant Consulting Inc.: Washington, DC, USA, 2014; pp. 2013–2014. [Google Scholar]
- Nhan, N.H.K.; Minh, T.H.Q.; Anh, N.D.Q. Influence of scattering enhancement particles CaCO3, CaF2, SiO2 and TiO2 on color uniformity of white LEDs. Appl. Phys. 2016, 14, 615–620. [Google Scholar] [CrossRef]
- Lai, M.F.; Quoc Anh, N.D.; Ma, H.Y.; Lee, H.Y. Scattering effect of SiO2 particles on correlated color temperature uniformity of multi-chip white light LEDs. J. Chin. Inst. Eng. 2016, 39, 468–472. [Google Scholar] [CrossRef]
- Hsu, C.H.; Chan, Y.C.; Chen, W.C.; Chang, C.H.; Liou, J.K.; Cheng, S.Y.; Guo, D.F.; Liu, W.C. Study of GaN-based LEDs with hybrid SiO 2 microsphere/nanosphere antireflection coating as a passivation layer by a rapid convection deposition. IEEE Trans. Electron Devices 2017, 64, 1134–1139. [Google Scholar] [CrossRef]
- Chung, Y.S.; Jeon, M.Y.; Kim, C.K. Performance changes of surface coated red phosphors with silica nanoparticles and silica nanocomposites. Ind. Eng. Chem. Res. 2009, 48, 740–748. [Google Scholar] [CrossRef]
- Yom, H.S.; Yang, J.K.; Polyakov, A.Y.; Lee, I.H. Performance of InGaN/GaN light emitting diodes with n-GaN layer embedded with SiO2 nano-particles. Appl. Sci. 2018, 8, 1574. [Google Scholar] [CrossRef]
- Nguyen, A.Q.D.; Nguyen, V.H. Application of CaCO3, CaF2, SiO2, and TiO2 particles to silicone lens for enhancing angular color uniformity of white LED lamp. In Journal of Physics: Conference Series, Proceedings of the 6th International Conference on Manufacturing Engineering and Process, Lisbon, Portugal, 27–29 May 2017; Institute of Physics Publishing: Bristol, UK, 2017; Volume 885. [Google Scholar]
- Lee, J.; Kwon, Y.Y.; Choi, E.-H.; Park, J.; Yoon, H.; Kim, H. Enhancement of light-extraction efficiency of organic light-emitting diodes using silica nanoparticles embedded in TiO2 matrices. Opt. Express 2014, 22, A705–A714. [Google Scholar] [CrossRef]
- Suresh, C.; Nagabhushana, H.; Basavaraj, R.B.; Darshan, G.P.; Kavyashree, D.; Daruka Prasad, B.; Sharma, S.C.; Vanithamani, R. SiO2@LaOF:Eu3+ core-shell functional nanomaterials for sensitive visualization of latent fingerprints and WLED applications. J. Colloid Interface Sci. 2018, 518, 200–215. [Google Scholar] [CrossRef]
- Komahal, F.F.; Nagabhushana, H.; Basavaraj, R.B.; Darshan, G.P.; Prasad, B.D.; Sharma, S.C.; Kavyashree, D. Design of Bi-functional composite core–shell SiO2@ZnAl2O4:Eu3+ array as a fluorescent sensors for selective and sensitive latent fingerprints visualization protocol. Adv. Powder Technol. 2018, 29, 1991–2002. [Google Scholar] [CrossRef]
- Sandhyarani, A.; Kokila, M.K.; Darshan, G.P.; Basavaraj, R.B.; Daruka Prasad, B.; Sharma, S.C.; Lakshmi, T.K.S.; Nagabhushana, H. Versatile core—Shell SiO2@SrTiO3:Eu3+, Li + nanopowders as fluorescent label for the visualization of latent fingerprints and anti-counterfeiting applications. Chem. Eng. J. 2017, 327, 1135–1150. [Google Scholar] [CrossRef]
- Rafiaei, S.M.; Isfahani, T.D.; Afshari, H.; Shokouhimehr, M. Improved optical properties of YVO4:Eu3+ nano—Layers on silica spheres. Mater. Chem. Phys. 2018, 203, 274–279. [Google Scholar] [CrossRef]
- Peng, G.H.; Wang, X.; Liang, Z.H.; Wang, Y.C.; Han, X.B.; Wu, J.L. Preparation of spherical core-shell structured SiO2@CaMoO4:Eu3+, Li+ red phosphors. J. Alloys Compd. 2013, 576, 227–231. [Google Scholar] [CrossRef]
- Ju, X.; Li, X.; Yang, Y.; Li, W.; Tao, C.; Feng, W. Luminescence properties of core-shell structured SiO2@CaMoO4:Eu3+ phosphor. J. Solid State Chem. 2012, 187, 109–113. [Google Scholar] [CrossRef]
- Wang, G.; Zou, H.; Gong, L.; Shi, Z.; Xu, X.; Sheng, Y. Synthesis and luminescent properties of monodisperse core-shell structured SiO2@Lu2O3:Eu3+ microspheres. Powder Technol. 2014, 258, 174–179. [Google Scholar] [CrossRef]
- Lu, W.; Guo, X.; Luo, Y.; Li, Q.; Zhu, R.; Pang, H. Core-shell materials for advanced batteries. Chem. Eng. J. 2019, 355, 208–237. [Google Scholar] [CrossRef]
- Feng, H.P.; Tang, L.; Zeng, G.M.; Zhou, Y.; Deng, Y.C.; Ren, X.; Song, B.; Liang, C.; Wei, M.Y.; Yu, J. Core-shell nanomaterials: Applications in energy storage and conversion. Adv. Colloid Interface Sci. 2019, 267, 26–46. [Google Scholar] [CrossRef]
- Xia, G.; Zhou, S.; Zhang, J.; Xu, J. Structural and optical properties of YAG:Ce3+ phosphors by sol-gel combustion method. J. Cryst. Growth 2005, 279, 357–362. [Google Scholar] [CrossRef]
- Hassanzadeh-Tabrizi, S.A. Synthesis and luminescence properties of YAG:Ce nanopowder prepared by the Pechini method. Adv. Powder Technol. 2012, 23, 324–327. [Google Scholar] [CrossRef]
- Selim, F.A.; Khamehchi, A.; Winarski, D.; Agarwal, S. Synthesis and characterization of Ce:YAG nano-phosphors and ceramics. Opt. Mater. Express 2016, 6, 3704. [Google Scholar] [CrossRef]
- Boukerika, A.; Guerbous, L.; Brihi, N. Ce-doped YAG phosphors prepared via sol-gel method: Effect of some modular parameters. J. Alloys Compd. 2014, 614, 383–388. [Google Scholar] [CrossRef]
- Garnet-Y3Al5O12—ChemTube3D. Available online: https://www.chemtube3d.com/ss-ygarnet/ (accessed on 8 October 2019).
- Gutsche, A.; Meier, M.; Guo, X.; Ungerer, J.; Nirschl, H. Modification of a SAXS camera to study structures on multiple scales. J. Nanopart. Res. 2017, 19, 321. [Google Scholar] [CrossRef]
- Guo, X.; Wagner, M.; Gutsche, A.; Meyer, J.; Seipenbusch, M.; Nirschl, H. Laboratory SWAXS combined with a low-pressure impactor for quasi-online analysis of nanoparticles generated by spark discharge. J. Aerosol Sci. 2015, 85, 17–29. [Google Scholar] [CrossRef]
- Guo, X.; Gao, K.; Gutsche, A.; Seipenbusch, M.; Nirschl, H. Combined small- and wide-angle X-ray scattering studies on oxide-supported Pt nanoparticles prepared by a CVS and CVD process. Powder Technol. 2015, 272, 23–33. [Google Scholar] [CrossRef]
- Gutsche, A.; Guo, X.; Dingenouts, N.; Nirschl, H. Synthesis and small angle X-ray scattering (SAXS) characterization of silica spheres covered with gel-like particles formed by means of solvent evaporation. Powder Technol. 2015, 278, 257–265. [Google Scholar] [CrossRef]
- Guo, X.; Gutsche, A.; Nirschl, H. SWAXS investigations on diffuse boundary nanostructures of metallic nanoparticles synthesized by electrical discharges. J. Nanopart. Res. 2013, 15, 2058. [Google Scholar] [CrossRef]
- Gutsche, A.; Daikeler, A.; Guo, X.; Dingenouts, N.; Nirschl, H. Time-resolved SAXS characterization of the shell growth of silica-coated magnetite nanocomposites. J. Nanopart. Res. 2014, 16, 2475. [Google Scholar] [CrossRef]
- Fernandes, N.J.; Akbarzadeh, J.; Peterlik, H.; Giannelis, E.P. Synthesis and properties of highly dispersed ionic silica-poly (ethylene oxide) nanohybrids. ACS Nano 2013, 7, 1265–1271. [Google Scholar] [CrossRef]
- Grünewald, T.A.; Lassenberger, A.; van Oostrum, P.D.J.; Rennhofer, H.; Zirbs, R.; Capone, B.; Vonderhaid, I.; Amenitsch, H.; Lichtenegger, H.C.; Reimhult, E. Core-shell structure of monodisperse Poly (ethylene glycol)-grafted iron oxide nanoparticles studied by Small-Angle X-ray Scattering. Chem. Mater. 2015, 27, 4763–4771. [Google Scholar] [CrossRef]
- Nirschl, H.; Guo, X. Characterisation of structured and functionalised particles by small-angle X-ray scattering (SAXS). Chem. Eng. Res. Des. 2018, 136, 431–446. [Google Scholar] [CrossRef]
- Guo, X.; Gutsche, A.; Wagner, M.; Seipenbusch, M.; Nirschl, H. Simultaneous SWAXS study of metallic and oxide nanostructured particles. J. Nanopart. Res. 2013, 15, 1559. [Google Scholar] [CrossRef]
- Joo, S.H.; Park, J.Y.; Tsung, C.K.; Yamada, Y.; Yang, P.; Somorjai, G.A. Thermally stable Pt/mesoporous silica core-shell nanocatalysts for high-temperature reactions. Nat. Mater. 2009, 8, 126–131. [Google Scholar] [CrossRef] [PubMed]
- He, B.; Zhao, Q.; Zeng, Z.; Wang, X.; Han, S. Effect of hydrothermal reaction time and calcination temperature on properties of Au@CeO2 core–shell catalyst for CO oxidation at low temperature. J. Mater. Sci. 2015, 50, 6339–6348. [Google Scholar] [CrossRef]
- Yan, F.; Jiang, J.; Chen, X.; Tian, S.; Li, K. Synthesis and characterization of silica nanoparticles preparing by low-temperature vapor-phase hydrolysis of SiCl4. Ind. Eng. Chem. Res. 2014, 53, 11884–11890. [Google Scholar] [CrossRef]
- He, G.; Mei, L.; Wang, L.; Liu, G.; Li, J. Synthesis and luminescence properties of nano-/microstructured Y3Al5O12:Ce3+ microspheres by controlled glass crystallization. Cryst. Growth Des. 2011, 11, 5355–5361. [Google Scholar] [CrossRef]
- Xu, J.; Liu, B.; Liu, Z.; Gong, Y.; Hu, B.; Wang, J.; Li, H.; Wang, X.; Du, B. Design of laser-driven SiO2-YAG:Ce composite thick film: Facile synthesis, robust thermal performance, and application in solid-state laser lighting. Opt. Mater. 2018, 75, 508–512. [Google Scholar] [CrossRef]
- Ogi, T.; Nandiyanto, A.B.D.; Okino, K.; Iskandar, F.; Wang, W.N.; Tanabe, E.; Okuyama, K. Towards better phosphor design: Effect of SiO2 nanoparticles on photoluminescence enhancement of YAG:Ce. ECS J. Solid State Sci. Technol. 2013, 2, R91–95. [Google Scholar] [CrossRef]
- Pan, G.H.; Wu, H.; He, S.; Zhang, L.; Hao, Z.; Zhang, X.; Zhang, J. Dye-embedded YAG:Ce3+@SiO2 composite phosphors toward warm wLEDs through radiative energy transfer: Preparation, characterization and luminescence properties. Nanoscale 2018, 10, 22237–22251. [Google Scholar] [CrossRef]
- Sun, Y.H.; Yang, Z.M.; Xie, C.N.; Jiang, Z.H. Preparation and characteristics of core-shell structure Y3Al5O12:Yb3+@SiO2 nanoparticles. J. Alloys Compd. 2012, 528, 1–9. [Google Scholar] [CrossRef]
- Potdevin, A.; Chadeyron, G.; Boyer, D.; Mahiou, R. Sol-gel based YAG:Ce3+ powders for applications in LED devices. Phys. Status Solidi C 2007, 4, 65–69. [Google Scholar] [CrossRef]
- Zhang, L.; Lu, Z.; Zhu, J.; Yang, H.; Han, P.; Chen, Y.; Zhang, Q. Citrate sol-gel combustion preparation and photoluminescence properties of YAG:Ce phosphors. J. Rare Earths 2012, 30, 289–296. [Google Scholar] [CrossRef]
- He, X.; Liu, X.; Li, R.; Yang, B.; Yu, K.; Zeng, M.; Yu, R. Effects of local structure of Ce3+ ions on luminescent properties of Y3Al5O12:Ce nanoparticles. Sci. Rep. 2016, 6, 2238. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.J.; Qu, Y.R.; Zhao, Y.L.; Chu, H. Bin Effect of the composition of lanthanide complexes on their luminescence enhancement by Ag@SiO2 core-shell nanoparticles. Nanomaterials 2018, 8, 98. [Google Scholar] [CrossRef] [PubMed]
- Falahatdoost, S.; Ara, M.H.M.; Shaban, Z.; Ghazyani, N. Optical investigation of shell thickness in light scattering SiO2 particle with TiO2 nanoshells and its application in dye sensitized solar cells. Opt. Mater. 2015, 47, 51–55. [Google Scholar] [CrossRef]
- Son, S.; Hwang, S.H.; Kim, C.; Yun, J.Y.; Jang, J. Designed synthesis of SiO2/TiO2 core/shell structure as light scattering material for highly efficient dye-sensitized solar cells. ACS Appl. Mater. Interfaces 2013, 5, 4815–4820. [Google Scholar] [CrossRef] [PubMed]


© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kiani Khouzani, M.; Bahrami, A.; Yazdan Mehr, M.; van Driel, W.D.; Zhang, G. Towards Multi-Functional SiO2@YAG:Ce Core–Shell Optical Nanoparticles for Solid State Lighting Applications. Nanomaterials 2020, 10, 153. https://doi.org/10.3390/nano10010153
Kiani Khouzani M, Bahrami A, Yazdan Mehr M, van Driel WD, Zhang G. Towards Multi-Functional SiO2@YAG:Ce Core–Shell Optical Nanoparticles for Solid State Lighting Applications. Nanomaterials. 2020; 10(1):153. https://doi.org/10.3390/nano10010153
Chicago/Turabian StyleKiani Khouzani, Mahdi, Abbas Bahrami, Maryam Yazdan Mehr, Willem Dirk van Driel, and Guoqi Zhang. 2020. "Towards Multi-Functional SiO2@YAG:Ce Core–Shell Optical Nanoparticles for Solid State Lighting Applications" Nanomaterials 10, no. 1: 153. https://doi.org/10.3390/nano10010153
APA StyleKiani Khouzani, M., Bahrami, A., Yazdan Mehr, M., van Driel, W. D., & Zhang, G. (2020). Towards Multi-Functional SiO2@YAG:Ce Core–Shell Optical Nanoparticles for Solid State Lighting Applications. Nanomaterials, 10(1), 153. https://doi.org/10.3390/nano10010153





