Modulation of Osteoclast Interactions with Orthopaedic Biomaterials
Abstract
1. Bone Biology of Osteoclasts
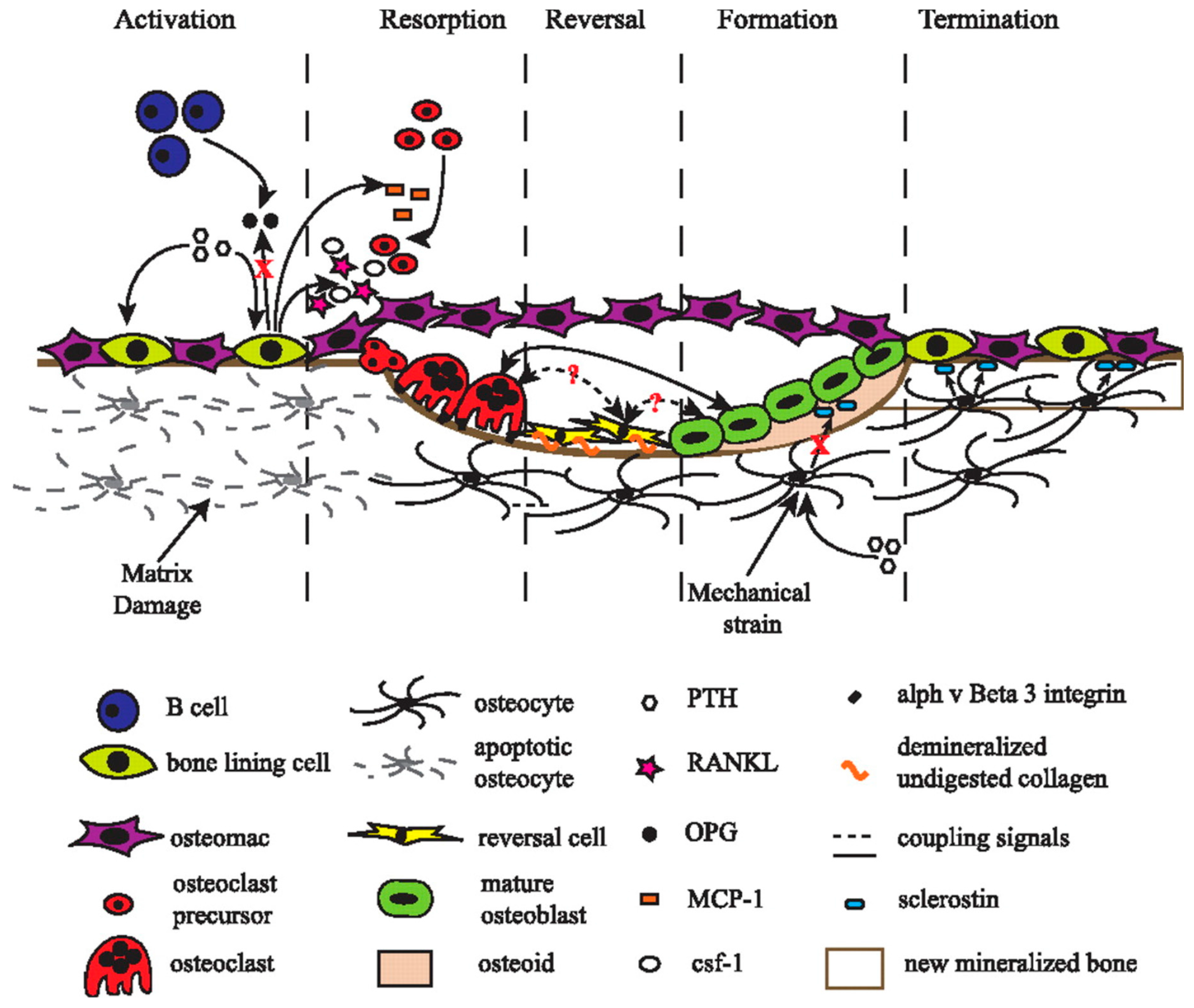
1.1. Bone Remodelling
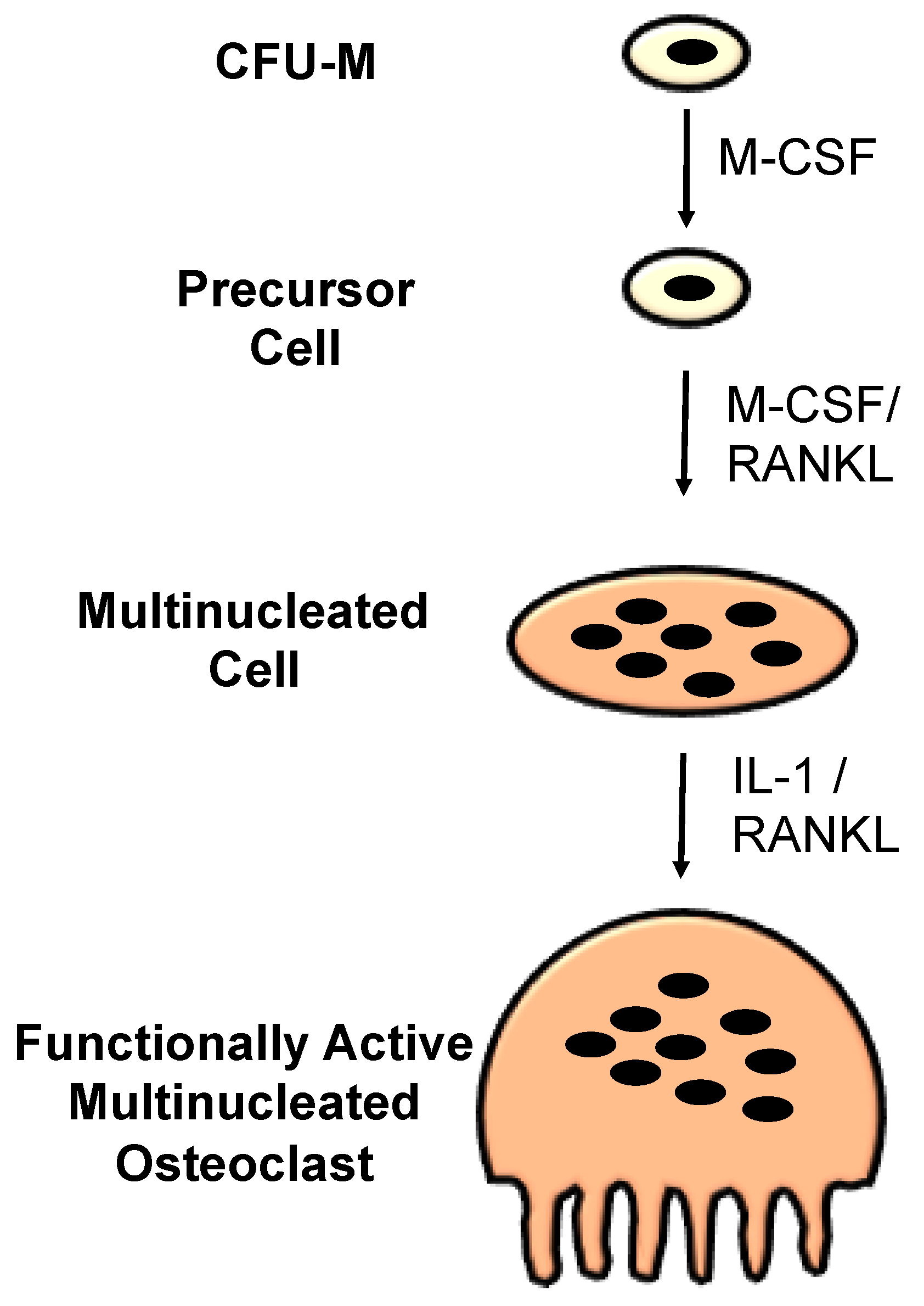
1.2. Osteoclasts
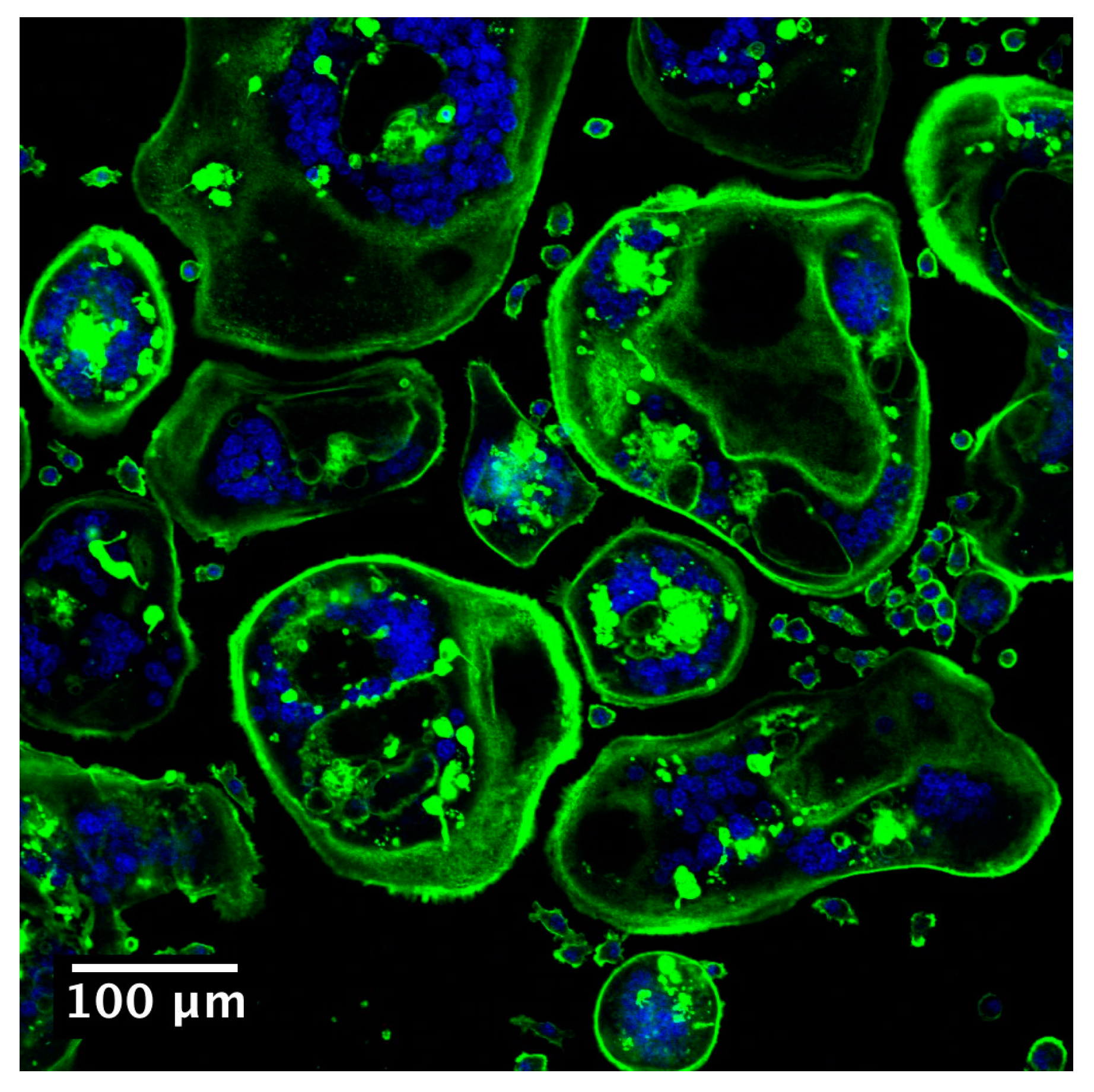
1.3. In Vitro Models of Osteoclasts
2. Biomaterials
3. Metals
3.1. Introduction
3.2. Surface Topography
3.3. Surface Chemistry
4. Bioceramics
4.1. Introduction
4.2. Material Composition
4.3. Surface Topography
5. Polymers
6. Conclusions and Outlook
Acknowledgments
Conflicts of Interest
References
- Lee, T.C.; Taylor, D. Bone remodelling: Should we cry wolff? Ir. J. Med. Sci. 1999, 168, 102. [Google Scholar] [CrossRef] [PubMed]
- Väänänen, H.K. Mechanism of bone turnover. Ann. Med. 1993, 25, 353–359. [Google Scholar] [CrossRef] [PubMed]
- Davies, J.E. Bone bonding at natural and biomaterial surfaces. Biomaterials 2007, 28, 5058–5067. [Google Scholar] [CrossRef] [PubMed]
- Boyle, W.J.; Simonet, W.S.; Lacey, D.L. Osteoclast differentiation and activation. Nature 2003, 423, 337–342. [Google Scholar] [CrossRef] [PubMed]
- Ducy, P.; Schinke, T.; Karsenty, G. The osteoblast: A sophisticated fibroblast under central surveillance. Science 2000, 289, 1501–1504. [Google Scholar] [CrossRef] [PubMed]
- Teitelbaum, S.L. Bone resorption by osteoclasts. Science 2000, 289, 1504–1508. [Google Scholar] [CrossRef] [PubMed]
- Detsch, R.; Boccaccini, A.R. The role of osteoclasts in bone tissue engineering. J. Tissue Eng. Regen. Med. 2015, 9, 1133–1149. [Google Scholar] [CrossRef] [PubMed]
- Takayanagi, H. Osteoimmunology and the effects of the immune system on bone. Nat. Rev. Rheumatol. 2009, 5, 667–676. [Google Scholar] [CrossRef] [PubMed]
- Raggatt, L.J.; Partridge, N.C. Cellular and molecular mechanisms of bone remodeling. J. Biol. Chem. 2010, 285, 25103–25108. [Google Scholar] [CrossRef] [PubMed]
- Delaisse, J.-M. The reversal phase of the bone-remodeling cycle: Cellular prerequisites for coupling resorption and formation. BoneKEy Rep. 2014, 3, 561. [Google Scholar] [CrossRef] [PubMed]
- Johnell, O.; Kanis, J.A. An estimate of the worldwide prevalence and disability associated with osteoporotic fractures. Osteoporos. Int. 2006, 17, 1726–1733. [Google Scholar] [CrossRef] [PubMed]
- Kyllönen, L.; D’Este, M.; Alini, M.; Eglin, D. Local drug delivery for enhancing fracture healing in osteoporotic bone. Acta Biomater. 2015, 11, 412–434. [Google Scholar] [CrossRef] [PubMed]
- Stover, M. Distal femoral fractures: Current treatment, results and problems. Injury 2001, 32, 3–13. [Google Scholar] [CrossRef]
- Syed, A.; Agarwal, M.; Giannoudis, P.; Matthews, S.; Smith, R. Distal femoral fractures: Long-term outcome following stabilisation with the LISS. Injury 2004, 35, 599–607. [Google Scholar] [CrossRef]
- Bolhofner, B.R.; Carmen, B.; Clifford, P. The results of open reduction and internal fixation of distal femur fractures using a biologic (indirect) reduction technique. J. Orthop. Trauma 1996, 10, 372–377. [Google Scholar] [CrossRef] [PubMed]
- Sessa, G.; Evola, F.R.; Costarella, L. Osteosynthesis systems in fragility fracture. Aging Clin. Exp. Res. 2011, 23, 69–70. [Google Scholar] [PubMed]
- Negri, A.L.; Spivacow, F.R. Healing of subtrochanteric atypical fractures after strontium ranelate treatment. Clin. Cases Miner. Bone Metab. Off. J. Ital. Soc. Osteoporos. Miner. Metab. Skelet. Dis. 2012, 9, 166–169. [Google Scholar]
- Larsson, S.; Fazzalari, N.L. Anti-osteoporosis therapy and fracture healing. Arch. Orthop. Trauma Surg. 2014, 134, 291–297. [Google Scholar] [CrossRef] [PubMed]
- Koval, K.J.; Meek, R.; Schemitsch, E.; Liporace, F.; Strauss, E.; Zuckerman, J.D. An AOA critical issue. Geriatric trauma: Young ideas. J. Bone Joint Surg. Am. 2003, 85, 1380–1388. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Xu, X. Stress shielding and fracture healing. Zhonghua Yi Xue Za Zhi 1994, 74, 483–485. [Google Scholar] [PubMed]
- Govender, S.; Csimma, C.; Genant, H.K.; Valentin-Opran, A.; Amit, Y.; Arbel, R.; Aro, H.; Atar, D.; Bishay, M.; Börner, M.G. Recombinant human bone morphogenetic protein-2 for treatment of open tibial fractures. J. Bone Joint Surg. 2002, 84, 2123–2134. [Google Scholar] [CrossRef] [PubMed]
- Giannoudis, P.V.; Tzioupis, C. Clinical applications of BMP-7: The UK perspective. Injury 2005, 36, S47–S50. [Google Scholar] [CrossRef] [PubMed]
- Tejwani, N.C.; Guerado, E. Improving fixation of the osteoporotic fracture: The role of locked plating. J. Orthop. Trauma 2011, 25 (Suppl. S2), S56–S60. [Google Scholar] [CrossRef] [PubMed]
- Wähnert, D.; Hofmann-Fliri, L.; Richards, R.G.; Gueorguiev, B.; Raschke, M.J.; Windolf, M. Implant Augmentation: Adding Bone Cement to Improve the Treatment of Osteoporotic Distal Femur Fractures: A Biomechanical Study Using Human Cadaver Bones. Medicine 2014, 93, e166. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, R.; González-García, C.; Torstrick, B.; Guldberg, R.E.; Salmerón-Sánchez, M.; García, A.J. Simple coating with fibronectin fragment enhances stainless steel screw osseointegration in healthy and osteoporotic rats. Biomaterials 2015, 63, 137–145. [Google Scholar] [CrossRef] [PubMed]
- Li, G.; Zhang, L.; Wang, L.; Yuan, G.; Dai, K.; Pei, J.; Hao, Y. Dual modulation of bone formation and resorption with zoledronic acid-loaded biodegradable magnesium alloy implants improves osteoporotic fracture healing: An in vitro and in vivo study. Acta Biomater. 2018, 65, 486–500. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, N.; Udagawa, N.; Suda, T. A new member of tumor necrosis factor ligand family, ODF/OPGL/TRANCE/RANKL, regulates osteoclast differentiation and function. Biochem. Biophys. Res. Commun. 1999, 256, 449–455. [Google Scholar] [CrossRef] [PubMed]
- Tsurukai, T.; Udagawa, N.; Matsuzaki, K.; Takahashi, N.; Suda, T. Roles of macrophage-colony stimulating factor and osteoclast differentiation factor in osteoclastogenesis. J. Bone Miner. Metab. 2000, 18, 177–184. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, S.; Takahashi, N.; Udagawa, N.; Tamura, T.; Akatsu, T.; Stanley, E.R.; Kurokawa, T.; Suda, T. Macrophage colony-stimulating factor is indispensable for both proliferation and differentiation of osteoclast progenitors. J. Clin. Investig. 1993, 91, 257–263. [Google Scholar] [CrossRef] [PubMed]
- Azuma, Y.; Kaji, K.; Katogi, R.; Takeshita, S.; Kudo, A. Tumor necrosis factor-alpha induces differentiation of and bone resorption by osteoclasts. J. Biol. Chem. 2000, 275, 4858–4864. [Google Scholar] [CrossRef] [PubMed]
- Islam, S.; Hassan, F.; Tumurkhuu, G.; Dagvadorj, J.; Koide, N.; Naiki, Y.; Mori, I.; Yoshida, T.; Yokochi, T. Bacterial lipopolysaccharide induces osteoclast formation in RAW 264.7 macrophage cells. Biochem. Biophys. Res. Commun. 2007, 360, 346–351. [Google Scholar] [CrossRef] [PubMed]
- Lakkakorpi, P.T.; Väänänen, H.K. Cytoskeletal changes in osteoclasts during the resorption cycle. Microsc. Res. Tech. 1996, 33, 171–181. [Google Scholar] [CrossRef]
- Florencio-Silva, R.; Sasso, G.R.; Sasso-Cerri, E.; Simoes, M.J.; Cerri, P.S. Biology of Bone Tissue: Structure, Function, and Factors That Influence Bone Cells. Biomed. Res. Int. 2015, 2015, 421746. [Google Scholar] [CrossRef] [PubMed]
- Blair, H.; Teitelbaum, S.; Ghiselli, R.; Gluck, S. Osteoclastic bone resorption by a polarized vacuolar proton pump. Science 1989, 245, 855–857. [Google Scholar] [CrossRef] [PubMed]
- Silver, I.A.; Murrills, R.J.; Etherington, D.J. Microelectrode studies on the acid microenvironment beneath adherent macrophages and osteoclasts. Exp. Cell Res. 1988, 175, 266–276. [Google Scholar] [CrossRef]
- Yamaza, T.; Goto, T.; Kamiya, T.; Kobayashi, Y.; Sakai, H.; Tanaka, T. Study of immunoelectron microscopic localization of cathepsin K in osteoclasts and other bone cells in the mouse femur. Bone 1998, 23, 499–509. [Google Scholar] [CrossRef]
- Bossard, M.J.; Tomaszek, T.A.; Thompson, S.K.; Amegadzie, B.Y.; Hanning, C.R.; Jones, C.; Kurdyla, J.T.; McNulty, D.E.; Drake, F.H.; Gowen, M.; et al. Proteolytic Activity of Human Osteoclast Cathepsin K: Expression, Purification, Activation, and Substrate Identification. J. Biol. Chem. 1996, 271, 12517–12524. [Google Scholar] [CrossRef] [PubMed]
- Everts, V.; Delaisse, J.M.; Korper, W.; Niehof, A.; Vaes, G.; Beertsen, W. Degradation of collagen in the bone-resorbing compartment underlying the osteoclast involves both cysteine-proteinases and matrix metalloproteinases. J. Cell. Physiol. 1992, 150, 221–231. [Google Scholar] [CrossRef] [PubMed]
- Minkin, C. Bone acid phosphatase: Tartrate-resistant acid phosphatase as a marker of osteoclast function. Calcif. Tissue Int. 1982, 34, 285–290. [Google Scholar] [CrossRef] [PubMed]
- Webster, T.J.; Ergun, C.; Doremus, R.H.; Siegel, R.W.; Bizios, R. Enhanced osteoclast-like cell functions on nanophase ceramics. Biomaterials 2001, 22, 1327–1333. [Google Scholar] [CrossRef]
- Yang, L.; Perez-Amodio, S.; Barrère-de Groot, F.Y.F.; Everts, V.; van Blitterswijk, C.A.; Habibovic, P. The effects of inorganic additives to calcium phosphate on in vitro behavior of osteoblasts and osteoclasts. Biomaterials 2010, 31, 2976–2989. [Google Scholar] [CrossRef] [PubMed]
- Midha, S.; van den Bergh, W.; Kim, T.B.; Lee, P.D.; Jones, J.R.; Mitchell, C.A. Bioactive glass foam scaffolds are remodelled by osteoclasts and support the formation of mineralized matrix and vascular networks in vitro. Adv. Healthc. Mater. 2013, 2, 490–499. [Google Scholar] [CrossRef] [PubMed]
- Hefti, T.; Frischherz, M.; Spencer, N.D.; Hall, H.; Schlottig, F. A comparison of osteoclast resorption pits on bone with titanium and zirconia surfaces. Biomaterials 2010, 31, 7321–7331. [Google Scholar] [CrossRef] [PubMed]
- Kartsogiannis, V.; Ng, K.W. Cell lines and primary cell cultures in the study of bone cell biology. Mol. Cell. Endocrinol. 2004, 228, 79–102. [Google Scholar] [CrossRef] [PubMed]
- Cody, J.J.; Rivera, A.A.; Liu, J.; Liu, J.M.; Douglas, J.T.; Feng, X. A simplified method for the generation of human osteoclasts in vitro. Int. J. Biochem. Mol. Biol. 2011, 2, 183–189. [Google Scholar] [PubMed]
- Hoebertz, A.; Arnett, T.R. Isolated osteoclast cultures. Methods Mol. Med. 2003, 80, 53–64. [Google Scholar] [PubMed]
- Collin-Osdoby, P.; Osdoby, P. RANKL-mediated osteoclast formation from murine RAW 264.7 cells. Methods Mol. Biol. 2012, 816, 187–202. [Google Scholar] [PubMed]
- Miyamoto, A.; Kunisada, T.; Hemmi, H.; Yamane, T.; Yasuda, H.; Miyake, K.; Yamazaki, H.; Hayashi, S.I. Establishment and characterization of an immortal macrophage-like cell line inducible to differentiate to osteoclasts. Biochem. Biophys. Res. Commun. 1998, 242, 703–709. [Google Scholar] [CrossRef] [PubMed]
- Burg, K.J.; Porter, S.; Kellam, J.F. Biomaterial developments for bone tissue engineering. Biomaterials 2000, 21, 2347–2359. [Google Scholar] [CrossRef]
- Currie, L.J.; Sharpe, J.R.; Martin, R. The use of fibrin glue in skin grafts and tissue-engineered skin replacements: A review. Plast. Reconstr. Surg. 2001, 108, 1713–1726. [Google Scholar] [CrossRef] [PubMed]
- Liberski, A.; Ayad, N.; Wojciechowska, D.; Kot, R.; Vo, D.M.P.; Aibibu, D.; Hoffmann, G.; Cherif, C.; Grobelny-Mayer, K.; Snycerski, M.; et al. Weaving for heart valve tissue engineering. Biotechnol. Adv. 2017, 35, 633–656. [Google Scholar] [CrossRef] [PubMed]
- Minkin, C.; Marinho, V.C. Role of the osteoclast at the bone-implant interface. Adv. Dent. Res. 1999, 13, 49–56. [Google Scholar] [CrossRef] [PubMed]
- Geetha, M.; Singh, A.K.; Asokamani, R.; Gogia, A.K. Ti based biomaterials, the ultimate choice for orthopaedic implants—A review. Prog. Mater. Sci. 2009, 54, 397–425. [Google Scholar] [CrossRef]
- Kurtz, S.; Ong, K.; Lau, E.; Mowat, F.; Halpern, M. Projections of primary and revision hip and knee arthroplasty in the United States from 2005 to 2030. J. Bone Joint Surg. Am. 2007, 89, 780–785. [Google Scholar] [PubMed]
- Albrektsson, T.; Brånemark, P.-I.; Hansson, H.-A.; Kasemo, B.; Larsson, K.; Lundström, I.; McQueen, D.H.; Skalak, R. The interface zone of inorganic implantsIn vivo: Titanium implants in bone. Ann. Biomed. Eng. 1983, 11, 1–27. [Google Scholar] [CrossRef]
- Kasemo, B. Biological surface science. Surf. Sci. 2002, 500, 656–677. [Google Scholar] [CrossRef]
- Sommer, B.; Felix, R.; Sprecher, C.; Leunig, M.; Ganz, R.; Hofstetter, W. Wear particles and surface topographies are modulators of osteoclastogenesis in vitro. J. Biomed. Mater. Res. A 2005, 72, 67–76. [Google Scholar] [CrossRef] [PubMed]
- Brinkmann, J.; Hefti, T.; Schlottig, F.; Spencer, N.D.; Hall, H. Response of osteoclasts to titanium surfaces with increasing surface roughness: An in vitro study. Biointerphases 2012, 7, 34. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Li, F.; Zhang, C.; Gao, B.; Tan, P.; Mi, B.; Zhang, Y.; Cheng, H.; Liao, H.; Huo, K.; et al. The Dimension of Titania Nanotubes Influences Implant Success for Osteoclastogenesis and Osteogenesis Patients. J. Nanosci. Nanotechnol. 2015, 15, 4136–4142. [Google Scholar] [CrossRef] [PubMed]
- Makihira, S.; Mine, Y.; Kosaka, E.; Nikawa, H. Titanium surface roughness accelerates RANKL-dependent differentiation in the osteoclast precursor cell line, RAW264.7. Dent. Mater. J. 2007, 26, 739–745. [Google Scholar] [CrossRef] [PubMed]
- Geblinger, D.; Zink, C.; Spencer, N.D.; Addadi, L.; Geiger, B. Effects of surface microtopography on the assembly of the osteoclast resorption apparatus. J. R. Soc. Interface 2012, 9, 1599–1608. [Google Scholar] [CrossRef] [PubMed]
- Shalabi, M.M.; Gortemaker, A.; Van’t Hof, M.A.; Jansen, J.A.; Creugers, N.H. Implant surface roughness and bone healing: A systematic review. J. Dent. Res. 2006, 85, 496–500. [Google Scholar] [CrossRef] [PubMed]
- Nagasawa, M.; Cooper, L.F.; Ogino, Y.; Mendonca, D.; Liang, R.; Yang, S.; Mendonca, G.; Uoshima, K. Topography Influences Adherent Cell Regulation of Osteoclastogenesis. J. Dent. Res. 2016, 95, 319–326. [Google Scholar] [CrossRef] [PubMed]
- Makihira, S.; Mine, Y.; Nikawa, H.; Shuto, T.; Kosaka, E.; Sugiyama, M.; Hosokawa, R. Immobilized-OPG-Fc on a titanium surface inhibits RANKL-dependent osteoclast differentiation in vitro. J. Mater. Sci. Mater. Med. 2010, 21, 647–653. [Google Scholar] [CrossRef] [PubMed]
- Peter, B.; Gauthier, O.; Laib, S.; Bujoli, B.; Guicheux, J.; Janvier, P.; van Lenthe, G.H.; Muller, R.; Zambelli, P.Y.; Bouler, J.M.; et al. Local delivery of bisphosphonate from coated orthopedic implants increases implants mechanical stability in osteoporotic rats. J. Biomed. Mater. Res. A 2006, 76, 133–143. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.; Luo, Z.; Hu, Y.; Shen, X.; Li, M.; Li, L.; Zhang, Y.; Yang, W.; Liu, P.; Cai, K. Enhancement of local bone remodeling in osteoporotic rabbits by biomimic multilayered structures on Ti6Al4V implants. J. Biomed. Mater. Res. A 2016, 104, 1437–1451. [Google Scholar] [CrossRef] [PubMed]
- Bosco, R.; Iafisco, M.; Tampieri, A.; Jansen, J.A.; Leeuwenburgh, S.C.G.; van den Beucken, J.J.J.P. Hydroxyapatite nanocrystals functionalized with alendronate as bioactive components for bone implant coatings to decrease osteoclastic activity. Appl. Surf. Sci. 2015, 328, 516–524. [Google Scholar] [CrossRef]
- Navarro, M.; Michiardi, A.; Castano, O.; Planell, J.A. Biomaterials in orthopaedics. J. R. Soc. Interface 2008, 5, 1137–1158. [Google Scholar] [CrossRef] [PubMed]
- Hench, L. Bioceramics: From Concept to Clinic. J. Am. Ceram. Soc. 1991, 74, 1487–1510. [Google Scholar] [CrossRef]
- Yamada, S.; Heymann, D.; Bouler, J.M.; Daculsi, G. Osteoclastic resorption of calcium phosphate ceramics with different hydroxyapatite/beta-tricalcium phosphate ratios. Biomaterials 1997, 18, 1037–1041. [Google Scholar] [CrossRef]
- Mayr, H.; Schlufter, S.; Detsch, R.; Ziegler, G. Influence of phase composition on degradation and resorption of biphasic calcium phosphate ceramics. Key Eng. Mater. 2008, 361–363, 1043–1046. [Google Scholar] [CrossRef]
- Gomi, K.; Lowenberg, B.; Shapiro, G.; Davies, J.E. Resorption of sintered synthetic hydroxyapatite by osteoclasts in vitro. Biomaterials 1993, 14, 91–96. [Google Scholar] [CrossRef]
- Detsch, R.; Mayr, H.; Ziegler, G. Formation of osteoclast-like cells on HA and TCP ceramics. Acta Biomater. 2008, 4, 139–148. [Google Scholar] [CrossRef] [PubMed]
- Gentleman, E.; Fredholm, Y.C.; Jell, G.; Lotfibakhshaiesh, N.; O’Donnell, M.D.; Hill, R.G.; Stevens, M.M. The effects of strontium-substituted bioactive glasses on osteoblasts and osteoclasts in vitro. Biomaterials 2010, 31, 3949–3956. [Google Scholar] [CrossRef] [PubMed]
- Roy, M.; Fielding, G.; Bandyopadhyay, A.; Bose, S. Effects of Zinc and Strontium Substitution in Tricalcium Phosphate on Osteoclast Differentiation and Resorption. Biomater. Sci. 2013, 1, 74–82. [Google Scholar] [CrossRef] [PubMed]
- Roy, M.; Bose, S. Osteoclastogenesis and osteoclastic resorption of tricalcium phosphate: Effect of strontium and magnesium doping. J. Biomed. Mater. Res. A 2012, 100, 2450–2461. [Google Scholar] [CrossRef] [PubMed]
- Yamada, Y.; Ito, A.; Kojima, H.; Sakane, M.; Miyakawa, S.; Uemura, T.; LeGeros, R.Z. Inhibitory effect of Zn2+ in zinc-containing beta-tricalcium phosphate on resorbing activity of mature osteoclasts. J. Biomed. Mater. Res. A 2008, 84, 344–352. [Google Scholar] [CrossRef] [PubMed]
- Schumacher, M.; Wagner, A.S.; Kokesch-Himmelreich, J.; Bernhardt, A.; Rohnke, M.; Wenisch, S.; Gelinsky, M. Strontium substitution in apatitic CaP cements effectively attenuates osteoclastic resorption but does not inhibit osteoclastogenesis. Acta Biomater. 2016, 37, 184–194. [Google Scholar] [CrossRef] [PubMed]
- Friederichs, R.J.; Brooks, R.A.; Ueda, M.; Best, S.M. In vitro osteoclast formation and resorption of silicon-substituted hydroxyapatite ceramics. J. Biomed. Mater. Res. A 2015, 103, 3312–3322. [Google Scholar] [CrossRef] [PubMed]
- Tortelli, F.; Pujic, N.; Liu, Y.; Laroche, N.; Vico, L.; Cancedda, R. Osteoblast and osteoclast differentiation in an in vitro three-dimensional model of bone. Tissue Eng. Part A 2009, 15, 2373–2383. [Google Scholar] [CrossRef] [PubMed]
- Botelho, C.M.; Brooks, R.A.; Spence, G.; McFarlane, I.; Lopes, M.A.; Best, S.M.; Santos, J.D.; Rushton, N.; Bonfield, W. Differentiation of mononuclear precursors into osteoclasts on the surface of Si-substituted hydroxyapatite. J. Biomed. Mater. Res. A 2006, 78, 709–720. [Google Scholar] [CrossRef] [PubMed]
- Vahabzadeh, S.; Roy, M.; Bose, S. Effects of Silicon on Osteoclast Cell Mediated Degradation, In Vivo Osteogenesis and Vasculogenesis of Brushite Cement. J. Mater. Chem. B Mater. Biol. Med. 2015, 3, 8973–8982. [Google Scholar] [CrossRef] [PubMed]
- Wilson, T.; Parikka, V.; Holmbom, J.; Ylanen, H.; Penttinen, R. Intact surface of bioactive glass S53P4 is resistant to osteoclastic activity. J. Biomed. Mater. Res. A 2006, 77, 67–74. [Google Scholar] [CrossRef] [PubMed]
- Karpov, M.; Laczka, M.; Leboy, P.S.; Osyczka, A.M. Sol-gel bioactive glasses support both osteoblast and osteoclast formation from human bone marrow cells. J. Biomed. Mater. Res. A 2008, 84, 718–726. [Google Scholar] [CrossRef] [PubMed]
- Cacciotti, I.; Lehmann, G.; Camaioni, A.; Bianco, A. AP40 Bioactive Glass Ceramic by Sol-Gel Synthesis: In Vitro Dissolution and Cell-Mediated Bioresorption. Key Eng. Mater. 2013, 541, 41–50. [Google Scholar] [CrossRef]
- Costa-Rodrigues, J.; Fernandes, A.; Lopes, M.A.; Fernandes, M.H. Hydroxyapatite surface roughness: Complex modulation of the osteoclastogenesis of human precursor cells. Acta Biomater. 2012, 8, 1137–1145. [Google Scholar] [CrossRef] [PubMed]
- Gross, K.A.; Muller, D.; Lucas, H.; Haynes, D.R. Osteoclast resorption of thermal spray hydoxyapatite coatings is influenced by surface topography. Acta Biomater. 2012, 8, 1948–1956. [Google Scholar] [CrossRef] [PubMed]
- Costa, D.O.; Prowse, P.D.; Chrones, T.; Sims, S.M.; Hamilton, D.W.; Rizkalla, A.S.; Dixon, S.J. The differential regulation of osteoblast and osteoclast activity by surface topography of hydroxyapatite coatings. Biomaterials 2013, 34, 7215–7226. [Google Scholar] [CrossRef] [PubMed]
- Davison, N.L.; ten Harkel, B.; Schoenmaker, T.; Luo, X.; Yuan, H.; Everts, V.; Barrere-de Groot, F.; de Bruijn, J.D. Osteoclast resorption of beta-tricalcium phosphate controlled by surface architecture. Biomaterials 2014, 35, 7441–7451. [Google Scholar] [CrossRef] [PubMed]
- Torres, A.L.; Santos, S.G.; Oliveira, M.I.; Barbosa, M.A. Fibrinogen promotes resorption of chitosan by human osteoclasts. Acta Biomater. 2013, 9, 6553–6562. [Google Scholar] [CrossRef] [PubMed]
- Heinemann, C.; Heinemann, S.; Bernhardt, A.; Lode, A.; Worch, H.; Hanke, T. In vitro osteoclastogenesis on textile chitosan scaffold. Eur. Cell Mater. 2010, 19, 96–106. [Google Scholar] [CrossRef] [PubMed]
- Rochet, N.; Balaguer, T.; Boukhechba, F.; Laugier, J.P.; Quincey, D.; Goncalves, S.; Carle, G.F. Differentiation and activity of human preosteoclasts on chitosan enriched calcium phosphate cement. Biomaterials 2009, 30, 4260–4267. [Google Scholar] [CrossRef] [PubMed]
- Shafieyan, Y.; Tiedemann, K.; Goulet, A.; Komarova, S.; Quinn, T.M. Monocyte proliferation and differentiation to osteoclasts is affected by density of collagen covalently bound to a poly(dimethyl siloxane) culture surface. J. Biomed. Mater. Res. A 2012, 100, 1573–1581. [Google Scholar] [CrossRef] [PubMed]
- Cool, S.M.; Kenny, B.; Wu, A.; Nurcombe, V.; Trau, M.; Cassady, A.I.; Grondahl, L. Poly(3-hydroxybutyrate-co-3-hydroxyvalerate) composite biomaterials for bone tissue regeneration: In vitro performance assessed by osteoblast proliferation, osteoclast adhesion and resorption, and macrophage proinflammatory response. J. Biomed. Mater. Res. A 2007, 82, 599–610. [Google Scholar] [CrossRef] [PubMed]
- Jones, G.L.; Motta, A.; Marshall, M.J.; El Haj, A.J.; Cartmell, S.H. Osteoblast: Osteoclast co-cultures on silk fibroin, chitosan and PLLA films. Biomaterials 2009, 30, 5376–5384. [Google Scholar] [CrossRef] [PubMed]
- Jeon, O.H.; Panicker, L.M.; Lu, Q.; Chae, J.J.; Feldman, R.A.; Elisseeff, J.H. Human iPSC-derived osteoblasts and osteoclasts together promote bone regeneration in 3D biomaterials. Sci. Rep. 2016, 6, 26761. [Google Scholar] [CrossRef] [PubMed]
| Biomaterials | Advantages | Disadvantages |
|---|---|---|
| Metals | High strength, fatigue resistance | Metal ion toxicity, wear |
| Bioceramics | High bioactivity (bioactive glasses), biodegradability (TCP), low friction coefficient and wear rate (bioinert ceramics) | Brittleness, low fatigue resistance |
| Polymers | Ease of ease of manufacture and modification | Low strength |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Steffi, C.; Shi, Z.; Kong, C.H.; Wang, W. Modulation of Osteoclast Interactions with Orthopaedic Biomaterials. J. Funct. Biomater. 2018, 9, 18. https://doi.org/10.3390/jfb9010018
Steffi C, Shi Z, Kong CH, Wang W. Modulation of Osteoclast Interactions with Orthopaedic Biomaterials. Journal of Functional Biomaterials. 2018; 9(1):18. https://doi.org/10.3390/jfb9010018
Chicago/Turabian StyleSteffi, Chris, Zhilong Shi, Chee Hoe Kong, and Wilson Wang. 2018. "Modulation of Osteoclast Interactions with Orthopaedic Biomaterials" Journal of Functional Biomaterials 9, no. 1: 18. https://doi.org/10.3390/jfb9010018
APA StyleSteffi, C., Shi, Z., Kong, C. H., & Wang, W. (2018). Modulation of Osteoclast Interactions with Orthopaedic Biomaterials. Journal of Functional Biomaterials, 9(1), 18. https://doi.org/10.3390/jfb9010018







