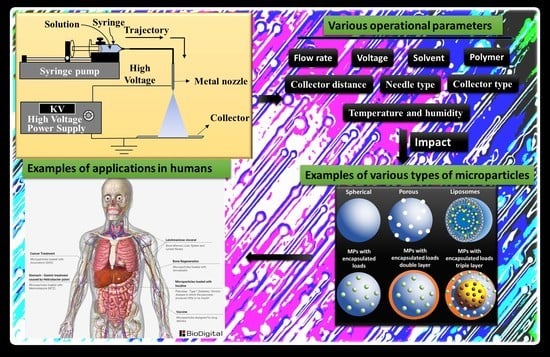
Fabrication of Polymeric Microparticles by Electrospray: The Impact of Experimental Parameters
Abstract
1. Introduction
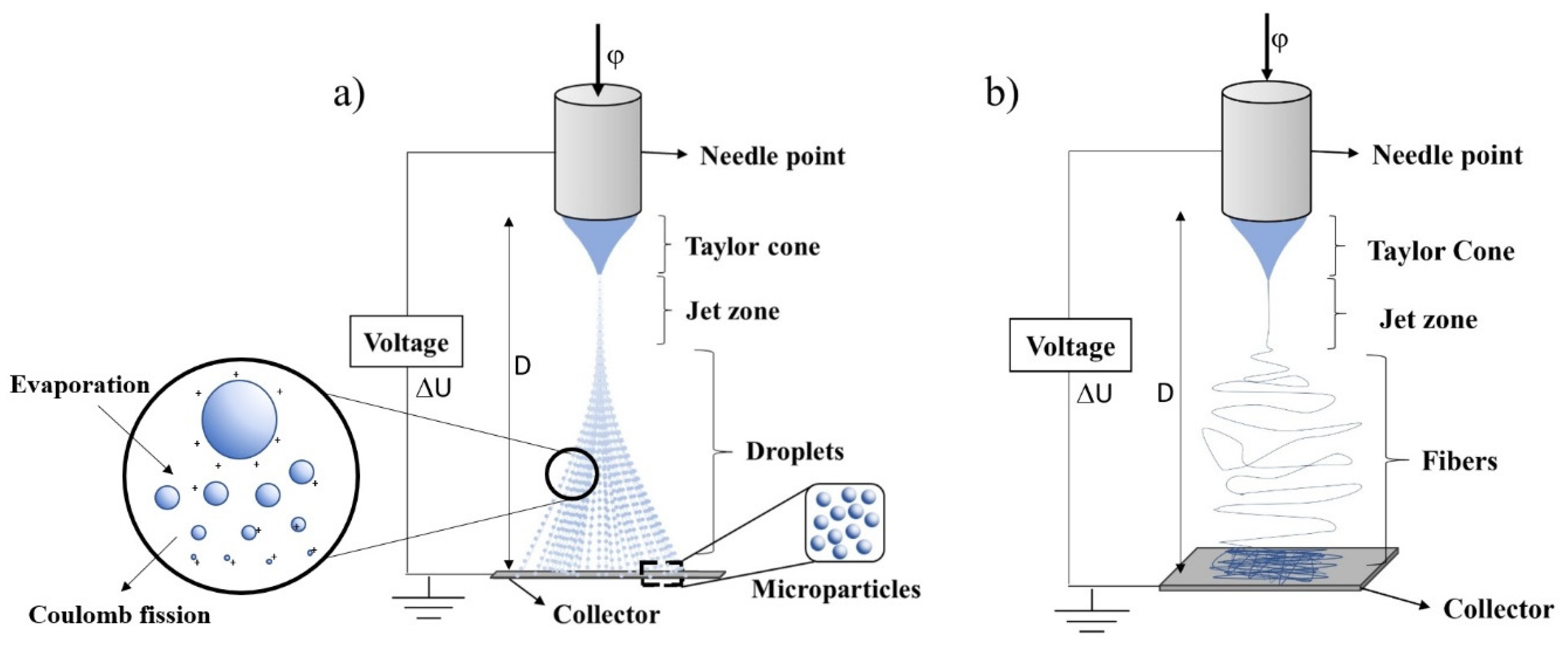
2. Electrospray (ES)
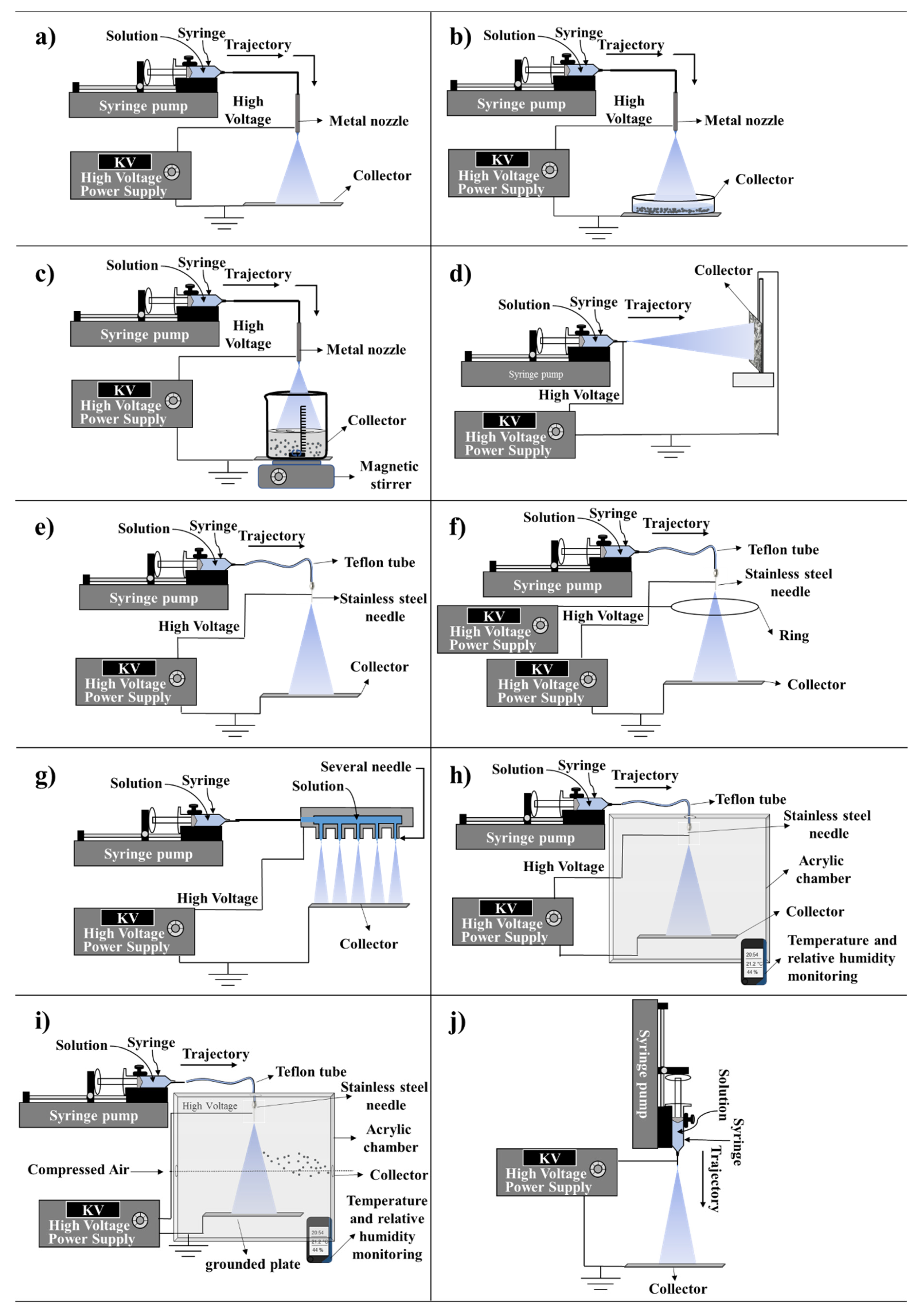
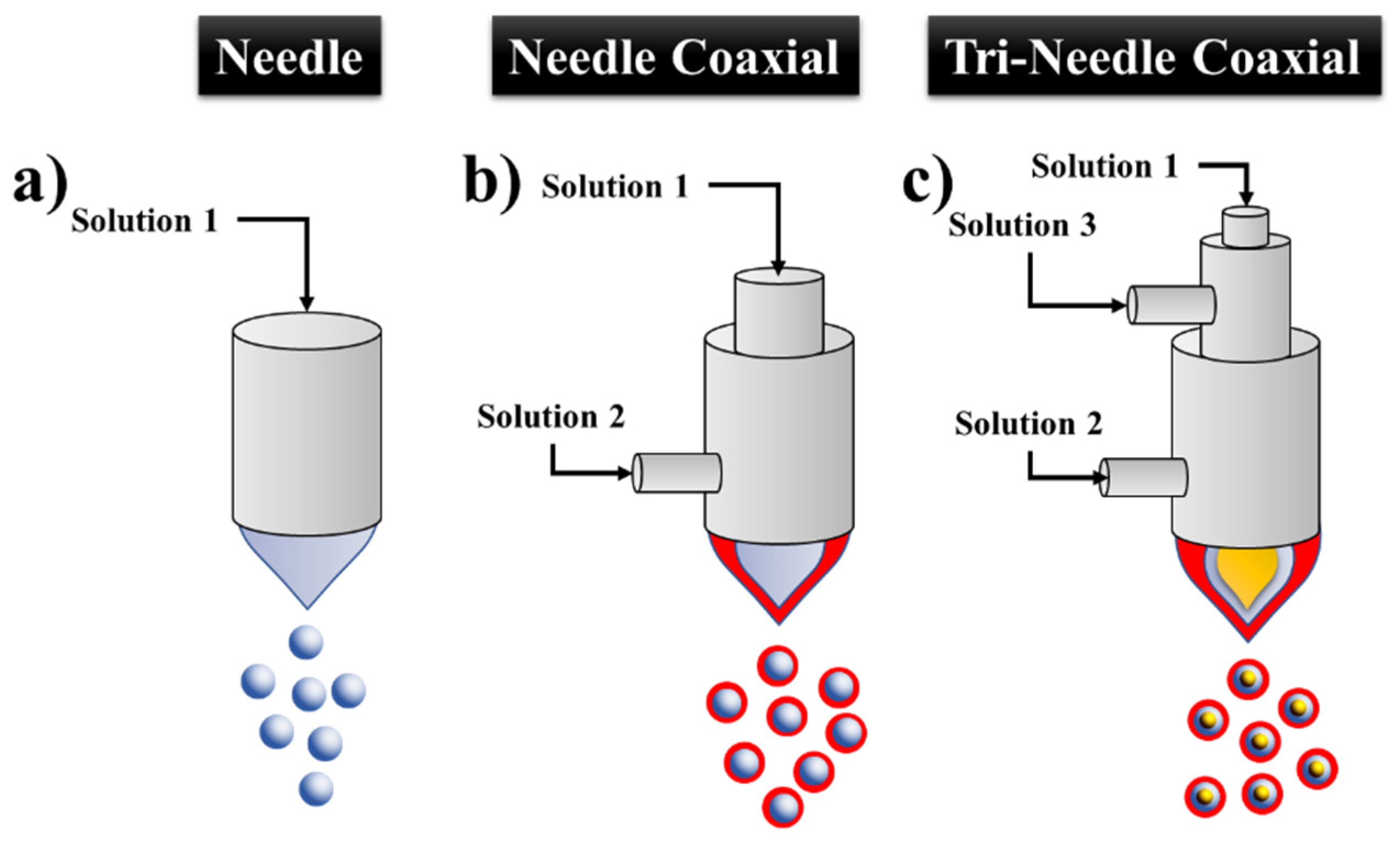
2.1. Types of Configuration
2.2. Type of Needle
2.2.1. Mono-Coaxial ES (MES)
2.2.2. Coaxial ES (CES)
2.2.3. Tri-Axial ES
2.3. Multi-Capillary ES (MCES)
2.4. Experimental Parameters that Influence ES During the Design of Microparticles
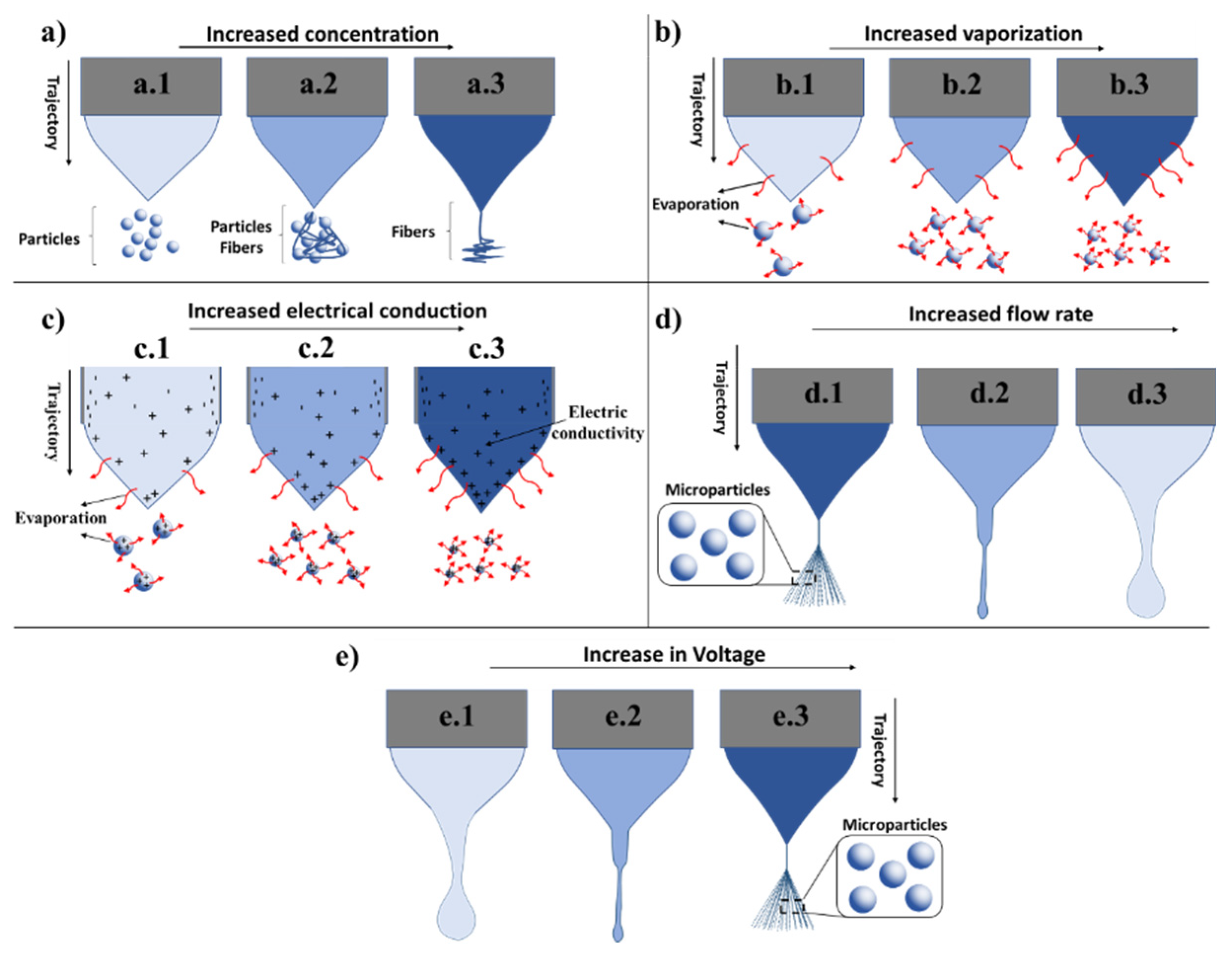
2.4.1. Polymer Concentration
2.4.2. Type of Solvent
2.4.3. Solution Conductivity
2.4.4. Flow Rate (FR)
2.4.5. Electric Field
2.4.6. Temperature and Humidity
2.4.7. Collector Distance
2.4.8. Types of Collectors
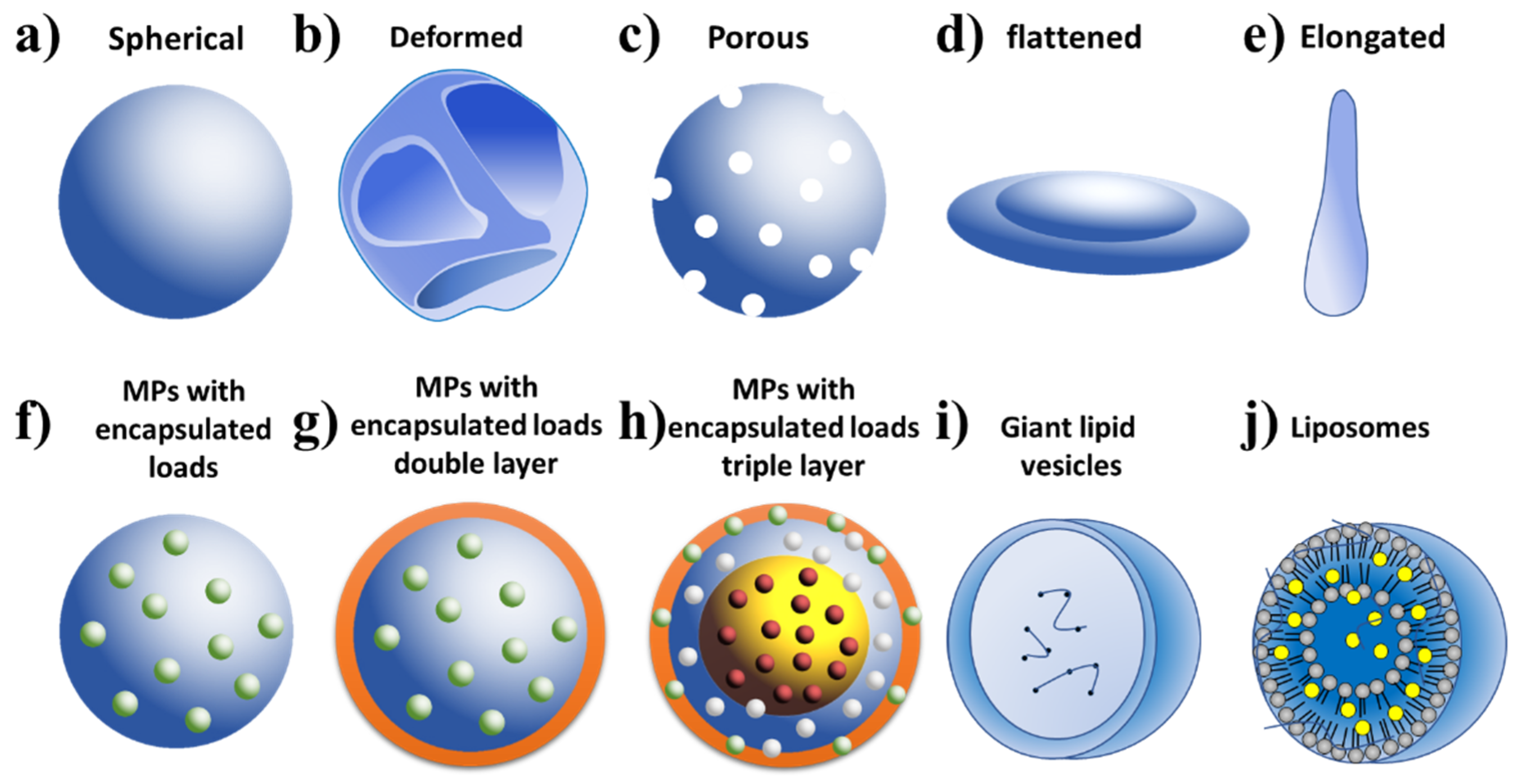
2.5. Types of Microparticles Made by ES and Their Applications
3. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Lee, H.; An, S.; Kim, S.; Jeon, B.; Kim, M.; Kim, I.S. Readily Functionalizable and Stabilizable Polymeric Particles with Controlled Size and Morphology by Electrospray. Sci. Rep. 2018, 8, 15725. [Google Scholar] [CrossRef] [PubMed]
- Bock, N.; Woodruff, M.A.; Hutmacher, D.W.; Dargaville, T.R. Electrospraying, a Reproducible Method for Production of Polymeric Microspheres for Biomedical Applications. Polymers 2011, 3, 131–149. [Google Scholar] [CrossRef]
- Baldino, L.; Cardea, S.; Reverchon, E. A supercritical CO2 assisted electrohydrodynamic process used to produce microparticles and microfibers of a model polymer. J. CO2 Util. 2019, 33, 532–540. [Google Scholar] [CrossRef]
- Nykamp, G.; Carstensen, U.; Müller, B. Jet milling—A new technique for microparticle preparation. Int. J. Pharm. 2002, 242, 79–86. [Google Scholar] [CrossRef]
- Sansone, F.; Mencherini, T.; Picerno, P.; D’Amore, M.; Aquino, R.P.; Lauro, M.R. Maltodextrin/pectin microparticles by spray drying as carrier for nutraceutical extracts. J. Food Eng. 2011, 105, 468–476. [Google Scholar] [CrossRef]
- Brinksmeier, E.; Riemer, O.; Kirchberg, S.; Brandao, C. Injection molded spherical grinding tools: Manufacture and application of a novel tool concept for micro grinding. Prod. Eng. 2013, 7, 383–389. [Google Scholar] [CrossRef]
- Liu, N.; Zhang, S.; Fu, R.; Dresselhaus, M.S.; Dresselhaus, G. Fabrication and structure of carbon aerogel spheres prepared by inverse suspension/emulsion polymerization and ambient pressure drying. J. Appl. Polym. Sci. 2007, 104, 2849–2855. [Google Scholar] [CrossRef]
- O’Donnell, P.B.; McGinity, J.W. Preparation of microspheres by the solvent evaporation technique. Adv. Drug Deliv. Rev. 1997, 28, 25–42. [Google Scholar] [CrossRef]
- Mytnyk, S.; Ziemecka, I.; Olive, A.G.L.; van der Meer, J.W.M.; Totlani, K.A.; Oldenhof, S.; Kreutzer, M.T.; van Steijn, V.; van Esch, J.H. Microcapsules with a permeable hydrogel shell and an aqueous core continuously produced in a 3D microdevice by all-aqueous microfluidics. RSC Adv. 2017, 7, 11331–11337. [Google Scholar] [CrossRef]
- Correia, D.M.; Gonçalves, R.; Ribeiro, C.; Sencadas, V.; Botelho, G.; Ribelles, J.L.G.; Lanceros-Méndez, S. Electrosprayed poly(vinylidene fluoride) microparticles for tissue engineering applications. RSC Adv. 2014, 4, 33013–33021. [Google Scholar] [CrossRef]
- Ye, M.; Kim, S.; Park, K. Issues in long-term protein delivery using biodegradable microparticles. J. Control. Release 2010, 146, 241–260. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.P.; Cui, L.; Yu, D.G.; Zhao, Z.X.; Chen, L. Electrosprayed core–shell solid dispersions of acyclovir fabricated using an epoxy-coated concentric spray head. Int. J. Nanomed. 2014, 9, 1967–1977. [Google Scholar]
- Shams, T.; Illangakoon, U.E.; Parhizkar, M.; Harker, A.H.; Edirisinghe, S.; Orlu, M.; Edirisinghe, M. Electrosprayed microparticles for intestinal delivery of prednisolone. J. R. Soc. Interface 2018, 15. [Google Scholar] [CrossRef] [PubMed]
- Yeo, L.Y.; Gagnon, Z.; Chang, H.C. AC electrospray biomaterials synthesis. Biomaterials 2005, 26, 6122–6128. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.G.; Liu, Q.W.; Yang, Q.B.; Li, Y.C.; Wang, W.; Sun, L.; Zhang, C.Q.; Li, Y.X. Electrospun poly(methyl methacrylate) nanofibers and microparticles. J. Mater. Sci. 2010, 45, 1032–1038. [Google Scholar] [CrossRef]
- Yao, J.; Lim, L.K.; Xie, J.W.; Hua, J.; Wang, C.H. Characterization of electrospraying process for polymeric particle fabrication. J. Aerosol Sci. 2008, 39, 987–1002. [Google Scholar] [CrossRef]
- Dastourani, H.; Jahannama, M.R.; Eslami-Majd, A. A physical insight into electrospray process in cone-jet mode: Role of operating parameters. Int. J. Heat Fluid Flow 2018, 70, 315–335. [Google Scholar] [CrossRef]
- Xie, J.W.; Lim, L.K.; Phua, Y.; Hua, J.; Wang, C.H. Electrohydrodynamic atomization for biodegradable polymeric particle production. J. Colloid Interface Sci. 2006, 302, 103–112. [Google Scholar] [CrossRef]
- Jiang, J.; Zheng, G.; Zhu, P.; Liu, J.; Liu, Y.; Wang, X.; Li, W.; Guo, S. Controlling of Electrospray Deposition for Micropatterns. Micromachines 2018, 9, 72. [Google Scholar] [CrossRef]
- Sander, S.; Tischler, C.; Achelis, L.; Henein, H.; Fritsching, U. Production of polymer microparticles by electrospray atomization. At. Sprays 2017, 27, 457–464. [Google Scholar] [CrossRef]
- Radacsi, N.; Stankiewicz, A.I.; Creyghton, Y.L.M.; van der Heijden, A.E.D.M.; ter Horst, J.H. Electrospray Crystallization for High-Quality Submicron-Sized Crystals. Chem. Eng. Technol. 2011, 34, 624–630. [Google Scholar] [CrossRef]
- Li, X.R.; MacEwan, M.R.; Xie, J.W.; Siewe, D.; Yuan, X.Y.; Xia, Y.N. Fabrication of Density Gradients of Biodegradable Polymer Microparticles and Their Use in Guiding Neurite Outgrowth. Adv. Funct. Mater. 2010, 20, 1632–1637. [Google Scholar] [CrossRef] [PubMed]
- Taylor, G.I. Disintegration of water drops in an electric field. Proc. R. Soc. Lond. Ser. A Math. Phys. Sci. 1964, 280, 383–397. [Google Scholar]
- Kumar, V.; Srivastava, A.; Shanbhogue, K.M.; Ingersol, S.; Sen, A.K. Electrospray performance of interacting multi-capillary emitters in a linear array. J. Micromech. Microeng. 2018, 28. [Google Scholar] [CrossRef]
- Tapia-Hernández, J.A.; Torres-Chávez, P.I.; Ramírez-Wong, B.; Rascón-Chu, A.; Plascencia-Jatomea, M.; Barreras-Urbina, C.G.; Rangel-Vázquez, N.A.; Rodríguez-Félix, F. Micro- and Nanoparticles by Electrospray: Advances and Applications in Foods. J. Agric. Food Chem. 2015, 63, 4699–4707. [Google Scholar] [CrossRef]
- Castillo, J.L.; Martin, S.; Rodriguez-Perez, D.; Higuera, F.J.; Garcia-Ybarra, P.L. Nanostructured porous coatings via electrospray atomization and deposition of nanoparticle suspensions. J. Aerosol Sci. 2018, 125, 148–163. [Google Scholar] [CrossRef]
- Almería, B.; Deng, W.; Fahmy, T.M.; Gomez, A. Controlling the morphology of electrospray-generated PLGA microparticles for drug delivery. J. Colloid Interface Sci. 2010, 343, 125–133. [Google Scholar] [CrossRef]
- Mouthuy, P.A.; Groszkowski, L.; Ye, H. Performances of a portable electrospinning apparatus. Biotechnol. Lett. 2015, 37, 1107–1116. [Google Scholar] [CrossRef]
- Lu, J.; Hou, R.; Yang, Z.; Tang, Z. Development and characterization of drug-loaded biodegradable PLA microcarriers prepared by the electrospraying technique. Int. J. Mol. Med. 2015, 36, 249–254. [Google Scholar] [CrossRef]
- Nath, S.D.; Son, S.; Sadiasa, A.; Min, Y.K.; Lee, B.T. Preparation and characterization of PLGA microspheres by the electrospraying method for delivering simvastatin for bone regeneration. Int. J. Pharm. 2013, 443, 87–94. [Google Scholar] [CrossRef]
- Bussano, R.; Chirio, D.; Costa, L.; Turci, F.; Trotta, M. Preparation and Characterization of Insulin-Loaded Lipid-Based Microspheres Generated by Electrospray. J. Dispers. Sci. Technol. 2011, 32, 1524–1530. [Google Scholar] [CrossRef]
- Jin, C.C.; Li, H.Y.; Williams, G.R.; Wei, R.; Nie, H.L.; Quan, J.; Zhu, L.M. Self-assembled liposomes from electrosprayed polymer-based microparticles. Colloid Polym. Sci. 2014, 292, 2325–2334. [Google Scholar] [CrossRef]
- Mangrio, F.A.; Dwivedi, P.; Han, S.; Zhao, G.; Gao, D.; Si, T.; Xu, R.X. Characteristics of Artemether-Loaded Poly(lactic-co-glycolic) Acid Microparticles Fabricated by Coaxial Electrospray: Validation of Enhanced Encapsulation Efficiency and Bioavailability. Mol. Pharm. 2017, 14, 4725–4733. [Google Scholar] [CrossRef] [PubMed]
- Duong, A.D.; Sharma, S.; Peine, K.J.; Gupta, G.; Satoskar, A.R.; Bachelder, E.M.; Wyslouzil, B.E.; Ainslie, K.M. Electrospray Encapsulation of Toll-Like Receptor Agonist Resiquimod in Polymer Microparticles for the Treatment of Visceral Leishmaniasis. Mol. Pharm. 2013, 10, 1045–1055. [Google Scholar] [CrossRef] [PubMed]
- Funasaki, Y.; Tsuchiya, E.; Maruyama, T. Hollow giant lipid vesicles prepared by coaxially electrospraying solutions of phospholipid and degradable polyelectrolyte. Colloid Polym. Sci. 2014, 292, 3049–3053. [Google Scholar] [CrossRef]
- Suksamran, T.; Opanasopit, P.; Rojanarata, T.; Ngawhirunpat, T.; Ruktanonchai, U.; Supaphol, P. Biodegradable alginate microparticles developed by electrohydrodynamic spraying techniques for oral delivery of protein. J. Microencapsul. 2009, 26, 563–570. [Google Scholar] [CrossRef]
- Suksamran, T.; Ngawhirunpat, T.; Rojanarata, T.; Sajomsang, W.; Pitaksuteepong, T.; Opanasopit, P. Methylated N-(4-N,N-dimethylaminocinnamyl) chitosan-coated electrospray OVA-loaded microparticles for oral vaccination. Int. J. Pharm. 2013, 448, 19–27. [Google Scholar] [CrossRef]
- Duong, A.D.; Ruan, G.; Mahajan, K.; Winter, J.O.; Wyslouzil, B.E. Scalable, Semicontinuous Production of Micelles Encapsulating Nanoparticles via Electrospray. Langmuir 2014, 30, 3939–3948. [Google Scholar] [CrossRef]
- Faramarzi, A.R.; Barzin, J.; Mobedi, H. Effect of solution and apparatus parameters on the morphology and size of electrosprayed PLGA microparticles. Fibers Polym. 2016, 17, 1806–1819. [Google Scholar] [CrossRef]
- Hao, S.; Wang, Y.; Wang, B.; Deng, J.; Zhu, L.; Cao, Y. Formulation of porous poly(lactic-co-glycolic acid) microparticles by electrospray deposition method for controlled drug release. Mater. Sci. Eng. C 2014, 39, 113–119. [Google Scholar] [CrossRef]
- Bock, N.; Dargaville, T.R.; Kirby, G.T.S.; Hutmacher, D.W.; Woodruff, M.A. Growth Factor-Loaded Microparticles for Tissue Engineering: The Discrepancies of In Vitro Characterization Assays. Tissue Eng. Part C Methods 2016, 22, 142–154. [Google Scholar] [CrossRef] [PubMed]
- Bock, N.; Dargaville, T.R.; Woodruff, M.A. Controlling microencapsulation and release of micronized proteins using poly(ethylene glycol) and electrospraying. Eur. J. Pharm. Biopharm. 2014, 87, 366–377. [Google Scholar] [CrossRef]
- Jeyhani, M.; Mak, S.Y.; Sammut, S.; Shum, H.C.; Hwang, D.K.; Tsai, S.S.H. Controlled Electrospray Generation of Nonspherical Alginate Microparticles. ChemPhysChem 2018, 19, 2113–2118. [Google Scholar] [CrossRef] [PubMed]
- Jafari-Nodoushan, M.; Barzin, J.; Mobedi, H. Size and morphology controlling of PLGA microparticles produced by electro hydrodynamic atomization. Polym. Adv. Technol. 2015, 26, 502–513. [Google Scholar] [CrossRef]
- Almería, B.; Gomez, A. Electrospray synthesis of monodisperse polymer particles in a broad (60 nm–2 μm) diameter range: Guiding principles and formulation recipes. J. Colloid Interface Sci. 2014, 417, 121–130. [Google Scholar] [CrossRef] [PubMed]
- Steipel, R.T.; Gallovic, M.D.; Batty, C.J.; Bachelder, E.M.; Ainslie, K.M. Electrospray for generation of drug delivery and vaccine particles applied in vitro and in vivo. Mater. Sci. Eng. C 2019, 105, 110070. [Google Scholar] [CrossRef]
- Matsuura, T.; Maruyama, T. Hollow phosphorylcholine polymer vesicles prepared by a coaxial electrospray technique. Colloid Polym. Sci. 2017, 295, 1251–1256. [Google Scholar] [CrossRef]
- Dai, H.F.; Chen, J.M.; Wang, L.Q. Preparation of stimuli responsive nanoparticles using coaxial electrospray template removal method and its application as drug delivery system. Acta Polym. Sin. 2017, 1947–1954. [Google Scholar]
- Xie, J.; Ng, W.J.; Lee, L.Y.; Wang, C.H. Encapsulation of protein drugs in biodegradable microparticles by co-axial electrospray. J. Colloid Interface Sci. 2008, 317, 469–476. [Google Scholar] [CrossRef]
- Matsuura, T.; Maruyama, T. Calcium phosphate-polymer hybrid microparticles having functionalized surfaces prepared by a coaxially electrospray technique. Colloids Surf. A Physicochem. Eng. Asp. 2017, 526, 64–69. [Google Scholar] [CrossRef]
- Zhang, L.; Huang, J.; Si, T.; Xu, R.X. Coaxial electrospray of microparticles and nanoparticles for biomedical applications. Expert Rev. Med. Devices 2012, 9, 595–612. [Google Scholar] [CrossRef]
- Kavadiya, S.; Biswas, P. Electrospray deposition of biomolecules: Applications, challenges, and recommendations. J. Aerosol Sci. 2018, 125, 182–207. [Google Scholar] [CrossRef]
- Zhang, C.; Yao, Z.C.; Ding, Q.; Choi, J.J.; Ahmad, Z.; Chang, M.W.; Li, J.S. Tri-Needle Coaxial Electrospray Engineering of Magnetic Polymer Yolk–Shell Particles Possessing Dual-Imaging Modality, Multiagent Compartments, and Trigger Release Potential. ACS Appl. Mater. Interfaces 2017, 9, 21485–21495. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.K.; Lee, J.Y.; Oh, H.; Song, D.W.; Kwak, H.W.; Yun, H.; Um, I.C.; Park, Y.H.; Lee, K.H. Effect of shear viscosity on the preparation of sphere-like silk fibroin microparticles by electrospraying. Int. J. Biol. Macromol. 2015, 79, 988–995. [Google Scholar] [CrossRef] [PubMed]
- Hao, S.L.; Wang, Y.Z.; Wang, B.C.; Zou, Q.M.; Zeng, H.; Chen, X.L.; Liu, X.; Liu, J.Y.; Yu, S.K. A novel gastroretentive porous microparticle for anti-Helicobacter pylori therapy: Preparation, in vitro and in vivo evaluation. Int. J. Pharm. 2014, 463, 10–21. [Google Scholar] [CrossRef] [PubMed]
- Gallovic, M.D.; Schully, K.L.; Bell, M.G.; Elberson, M.A.; Palmer, J.R.; Darko, C.A.; Bachelder, E.M.; Wyslouzil, B.E.; Keane-Myers, A.M.; Ainslie, K.M. Acetalated Dextran Microparticulate Vaccine Formulated via Coaxial Electrospray Preserves Toxin Neutralization and Enhances Murine Survival Following Inhalational Bacillus Anthracis Exposure. Adv. Healthc. Mater. 2016, 5, 2617–2627. [Google Scholar] [CrossRef]
- Chen, N.; Johnson, M.M.; Collier, M.A.; Gallovic, M.D.; Bachelder, E.M.; Ainslie, K.M. Tunable degradation of acetalated dextran microparticles enables controlled vaccine adjuvant and antigen delivery to modulate adaptive immune responses. J. Control. Release 2018, 273, 147–159. [Google Scholar] [CrossRef]
- Mutlu, E.C.; Ficai, A.; Ficai, D.; Yildirim, A.B.; Yildirim, M.; Oktar, F.N.; Demir, A. Chitosan/poly(ethylene glycol)/hyaluronic acid biocompatible patches obtained by electrospraying. Biomed. Mater. 2018, 13, 055011. [Google Scholar] [CrossRef]
- Enayati, M.; Chang, M.W.; Bragman, F.; Edirisinghe, M.; Stride, E. Electrohydrodynamic preparation of particles, capsules and bubbles for biomedical engineering applications. Colloids Surf. A Physicochem. Eng. Asp. 2011, 382, 154–164. [Google Scholar] [CrossRef]
- Zhang, X.; Zhao, Y. Programmable patterning of polymeric microparticles by floating electrodes-assisted electrospray. J. Micromech. Microeng. 2012, 22, 047001. [Google Scholar] [CrossRef]
- Pengpong, T.; Sangvanich, P.; Sirilertmukul, K.; Muangsin, N. Design, synthesis and in vitro evaluation of mucoadhesive p-coumarate-thiolated-chitosan as a hydrophobic drug carriers. Eur. J. Pharm. Biopharm. 2014, 86, 487–497. [Google Scholar] [CrossRef] [PubMed]
- Fei, L.; Yoo, S.H.; Villamayor, R.A.R.; Williams, B.P.; Gong, S.Y.; Park, S.; Shin, K.; Joo, Y.L. Graphene Oxide Involved Air-Controlled Electrospray for Uniform, Fast, Instantly Dry, and Binder-Free Electrode Fabrication. ACS Appl. Mater. Interfaces 2017, 9, 9738–9746. [Google Scholar] [CrossRef] [PubMed]
- Tapia-Hernández, J.A.; Rodríguez-Félix, D.E.; Plascencia-Jatomea, M.; Rascón-Chu, A.; López-Ahumada, G.A.; Ruiz-Cruz, S.; Barreras-Urbina, C.G.; Rodríguez-Félix, F. Porous wheat gluten microparticles obtained by electrospray: Preparation and characterization. Adv. Polym. Technol. 2018, 37, 2314–2324. [Google Scholar] [CrossRef]
- Nikoo, A.M.; Kadkhodaee, R.; Ghorani, B.; Razzaq, H.; Tucker, N. Electrospray-assisted encapsulation of caffeine in alginate microhydrogels. Int. J. Biol. Macromol. 2018, 116, 208–216. [Google Scholar] [CrossRef]
- BioDigital Human Studio. Available online: https://www.biodigital.com/ (accessed on 4 November 2019).
- Li, W.; Liu, S.; Yao, H.; Liao, G.; Si, Z.; Gong, X.; Ren, L.; Wang, L. Microparticle templating as a route to nanoscale polymer vesicles with controlled size distribution for anticancer drug delivery. J. Colloid Interface Sci. 2017, 508, 145–153. [Google Scholar] [CrossRef]
- Larsen, G.; Velarde-Ortiz, R.; Minchow, K.; Barrero, A.; Loscertales, I.G. A Method for Making Inorganic and Hybrid (Organic/Inorganic) Fibers and Vesicles with Diameters in the Submicrometer and Micrometer Range via Sol–Gel Chemistry and Electrically Forced Liquid Jets. J. Am. Chem. Soc. 2013, 125, 1154–1155. [Google Scholar] [CrossRef]
- Bielecki, J.; Hantke, M.F.; Daurer, B.J.; Reddy, H.K.N.; Hasse, D.; Larsson, D.S.D.; Gunn, L.H.; Svenda, M.; Munke, A.; Sellberg, J.A.; et al. Electrospray sample injection for single-particle imaging with X-ray lasers. Sci. Adv. 2019, 5. [Google Scholar] [CrossRef]
- Parhizkar, M.; Reardon, P.J.T.; Knowles, J.C.; Browning, R.J.; Stride, E.; Pedley, R.B.; Grego, T.; Edirisinghe, M. Performance of novel high throughput multi electrospray systems for forming of polymeric micro/nanoparticles. Mater. Des. 2017, 126, 73–84. [Google Scholar] [CrossRef]
- Zhang, P.P.; Xia, J.F.; Luo, S.D. Generation of Well-Defined Micro/Nanoparticles via Advanced Manufacturing Techniques for Therapeutic Delivery. Materials 2018, 11, 623. [Google Scholar] [CrossRef]
- Kohane, D.S. Microparticles and Nanoparticles for Drug Delivery. Biotechnol. Bioeng. 2007, 96, 203–209. [Google Scholar] [CrossRef]

| Method | Advantages | Disadvantages |
|---|---|---|
| Jet-milling, spray drying, micro grinding, suspension/emulsion polymerization, solvent evaporation, microfluidics |
|
|
| Electrospray (ES) |
|
|
| Sections | System Parameters | Content Description |
|---|---|---|
| Section 2.4.1. | Polymer concentration | Deals with the influence of variations in polymer concentration in the ES process |
| Section 2.4.2. | Type of solvent | Addresses the variation of solvent types and their mixtures in the ES process |
| Section 2.4.3. | Solution conductivity | Discusses the influence of solution conductivity on the ES process |
| Section 2.4.4. | Flow rate (FR) | Focuses on the discussion of the FR parameter and its influence on ES and the generated MPs morphology |
| Section 2.4.5. | Electric field | Discusses the influence of electric field on ES |
| Section 2.4.6. | Temperature and humidity | Adresses the influence of temperature and humidity on ES |
| Section 2.4.7. | Collector distance | Deals with the influence of collector distance |
| Section 2.4.8. | Types of collectors | Discusses the influence of collector type |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Morais, A.Í.S.; Vieira, E.G.; Afewerki, S.; Sousa, R.B.; Honorio, L.M.C.; Cambrussi, A.N.C.O.; Santos, J.A.; Bezerra, R.D.S.; Furtini, J.A.O.; Silva-Filho, E.C.; et al. Fabrication of Polymeric Microparticles by Electrospray: The Impact of Experimental Parameters. J. Funct. Biomater. 2020, 11, 4. https://doi.org/10.3390/jfb11010004
Morais AÍS, Vieira EG, Afewerki S, Sousa RB, Honorio LMC, Cambrussi ANCO, Santos JA, Bezerra RDS, Furtini JAO, Silva-Filho EC, et al. Fabrication of Polymeric Microparticles by Electrospray: The Impact of Experimental Parameters. Journal of Functional Biomaterials. 2020; 11(1):4. https://doi.org/10.3390/jfb11010004
Chicago/Turabian StyleMorais, Alan Í. S., Ewerton G. Vieira, Samson Afewerki, Ricardo B. Sousa, Luzia M. C. Honorio, Anallyne N. C. O. Cambrussi, Jailson A. Santos, Roosevelt D. S. Bezerra, Josy A. O. Furtini, Edson C. Silva-Filho, and et al. 2020. "Fabrication of Polymeric Microparticles by Electrospray: The Impact of Experimental Parameters" Journal of Functional Biomaterials 11, no. 1: 4. https://doi.org/10.3390/jfb11010004
APA StyleMorais, A. Í. S., Vieira, E. G., Afewerki, S., Sousa, R. B., Honorio, L. M. C., Cambrussi, A. N. C. O., Santos, J. A., Bezerra, R. D. S., Furtini, J. A. O., Silva-Filho, E. C., Webster, T. J., & Lobo, A. O. (2020). Fabrication of Polymeric Microparticles by Electrospray: The Impact of Experimental Parameters. Journal of Functional Biomaterials, 11(1), 4. https://doi.org/10.3390/jfb11010004