Use of Bacteriocinogenic Cultures without Inhibiting Cheese Associated Nonstarter Lactic Acid Bacteria; A Trial with Lactobacillus plantarum
Abstract
:1. Introduction
2. Materials and Methods
2.1. Culture Methods
2.2. Co-Cultures in Milk Based Medium
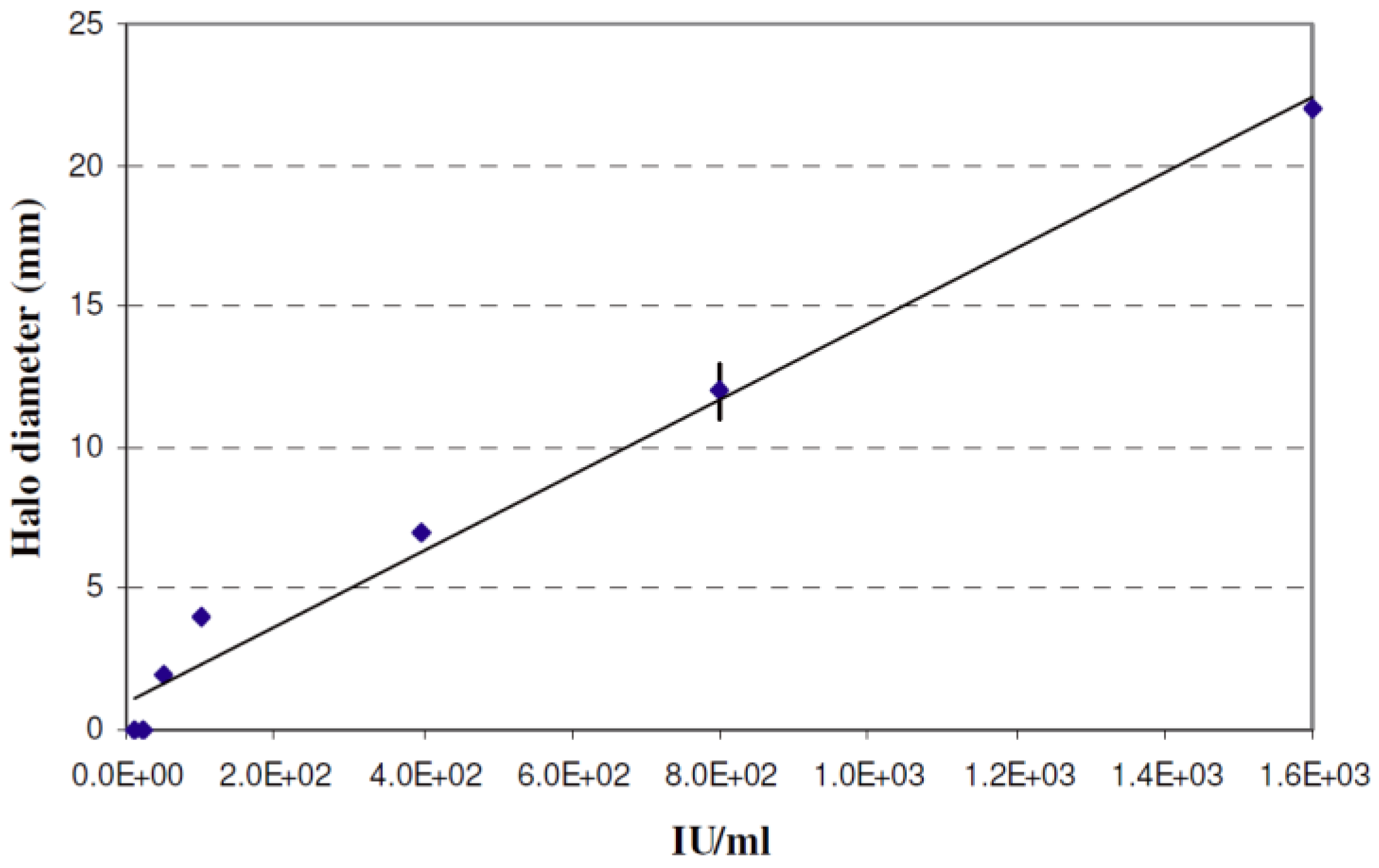
2.3. Quantification of Nisin in the Co-Cultures
2.4. Preparation of Peptide Extracts
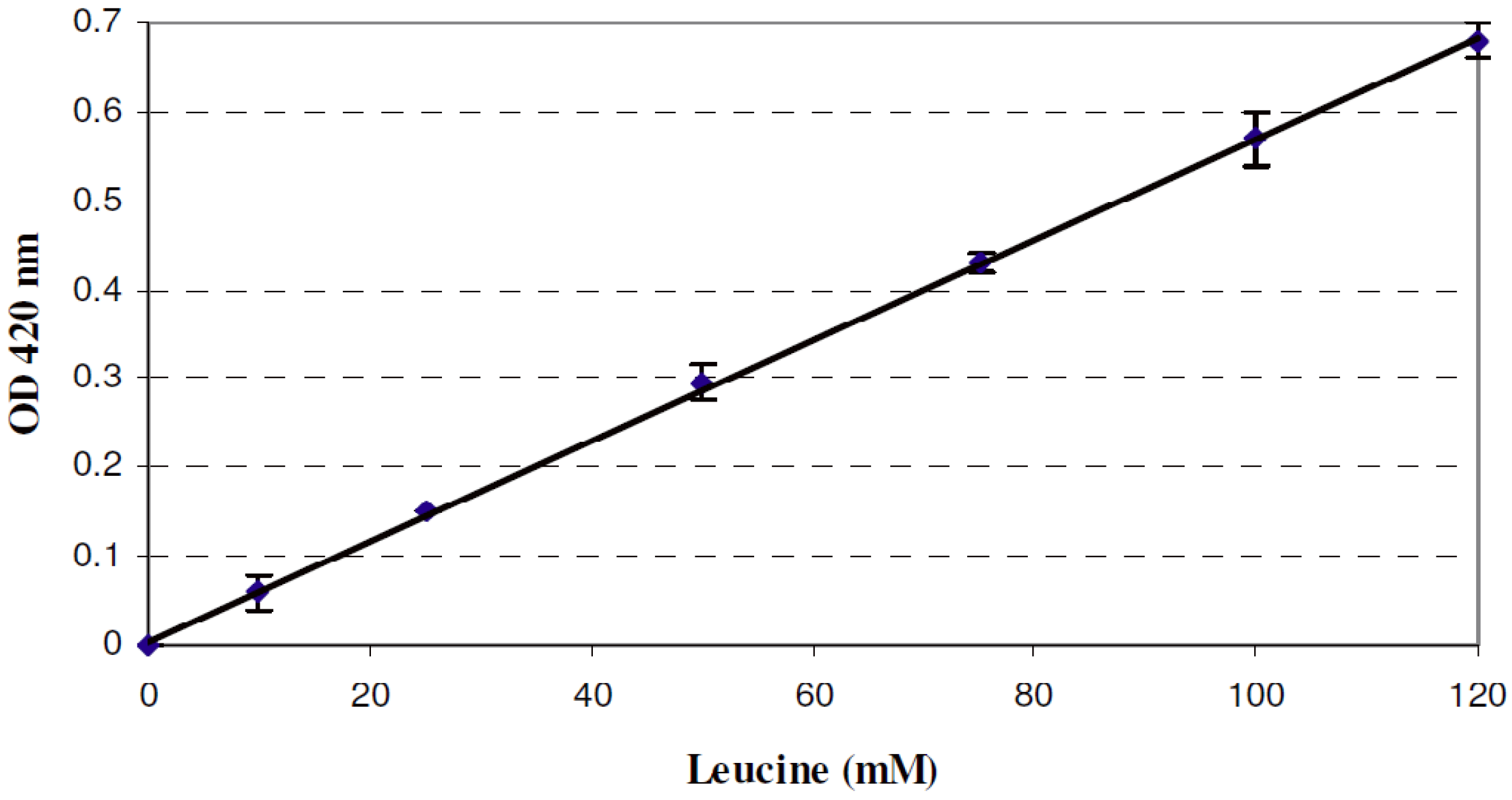
2.5. Quantitative Determination of Free Amino Groups
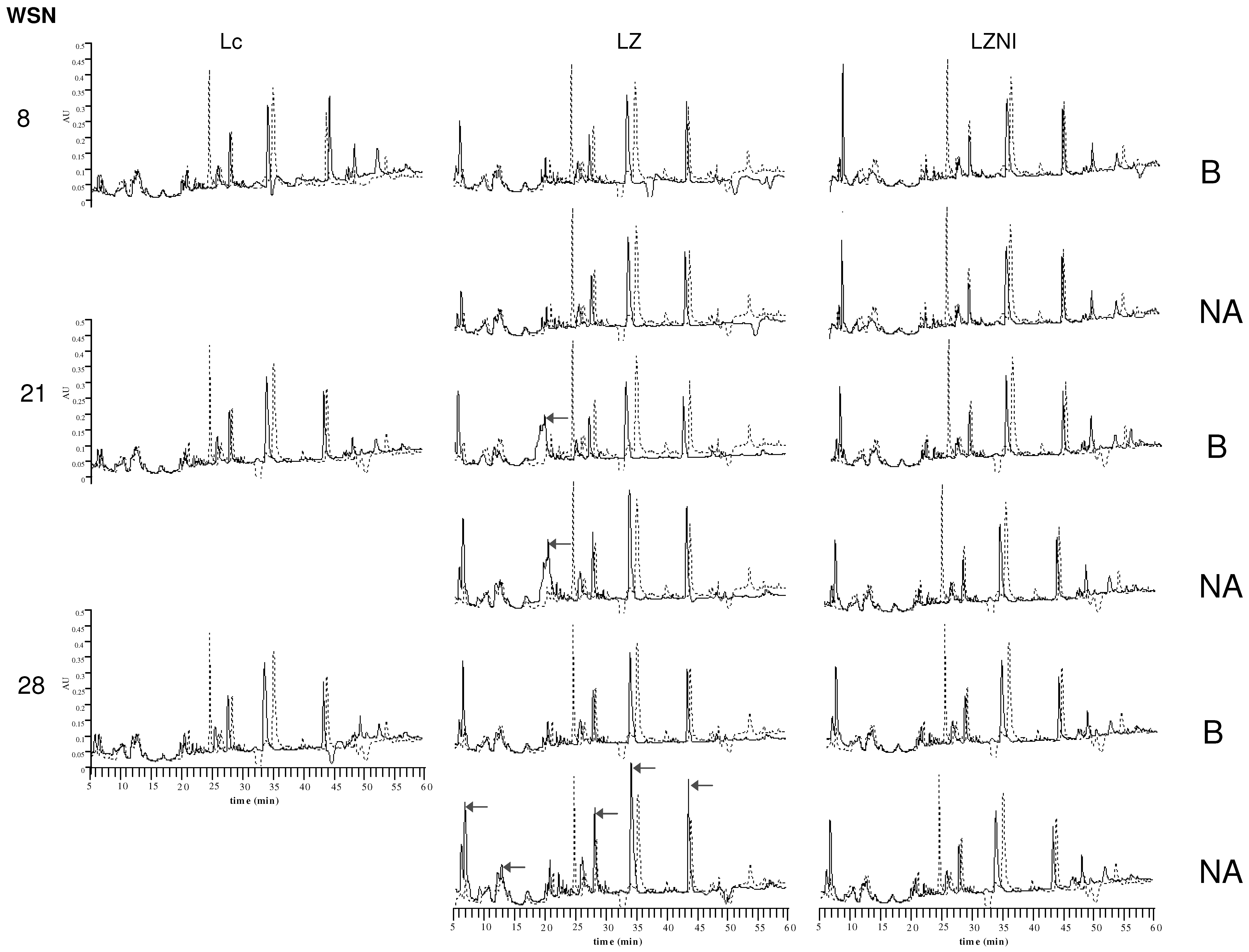
2.6. RP-HPLC Analysis of Peptide Profiles
3. Results
3.1. Sensitivity of NSLAB to Nisin
| Species | Isolation Source | Strain and Halo Size (mm of Diameter) |
|---|---|---|
| L. plantarum | Monte Veronese cheese | LZ (18); R1M2 (16); R2M3 (0); RM2 (10); R12 (9) |
| Reference strains | NCFB 340 (8); ATCC 14917 (10) | |
| L. paracasei | Grana Trentino cheese | RL3 (15); RC4 (14); RL4 (15); RM10 (14); RM133 (16); RM187 (16); RF138 (16); RF102 (15); RF302 (14); RL131 (16); RF135 (15); RM1812 (15); RL188 (15); RR131 (12); RC304 (10); RC303 (18); RC137 (11); RC131 (7); RM1811 (12) |
| Reference strains | ATCC 334 (10); NCFB 151 (11); ATCC 27092 (16); ATCC 25180 (14); DSM 20006 (10); 4114 (11); NCIMB 8001 (8); NCFB 242 (9); NCFB 2743 (11); DSM 20207 (12) |
3.2. Growth and Acidification in Co-Cultures L. plantarum/Nisin Producer

| Day | LZA | LZNA | LZB | LZNB | LZNIA | LZNINA | LZNIB | LZNINB |
|---|---|---|---|---|---|---|---|---|
| 0 | 4.31±0.01 | 4.31±0.01 | 2.31±0.01 | 2.31±0.01 | 4.31±0.01 | 4.31±0.01 | 2.31±0.01 | 2.31±0.01 |
| 1 | 9.6±0.02 | 9.77±0.05 | 9.77±0.01 | 9.77±0.03 | 9.77±0.05 | 9.77±0.02 | 9.3±0.01 | 9.6±0.02 |
3.3. Nisin Activity and Peptide Hydrolysis in Co-Cultures
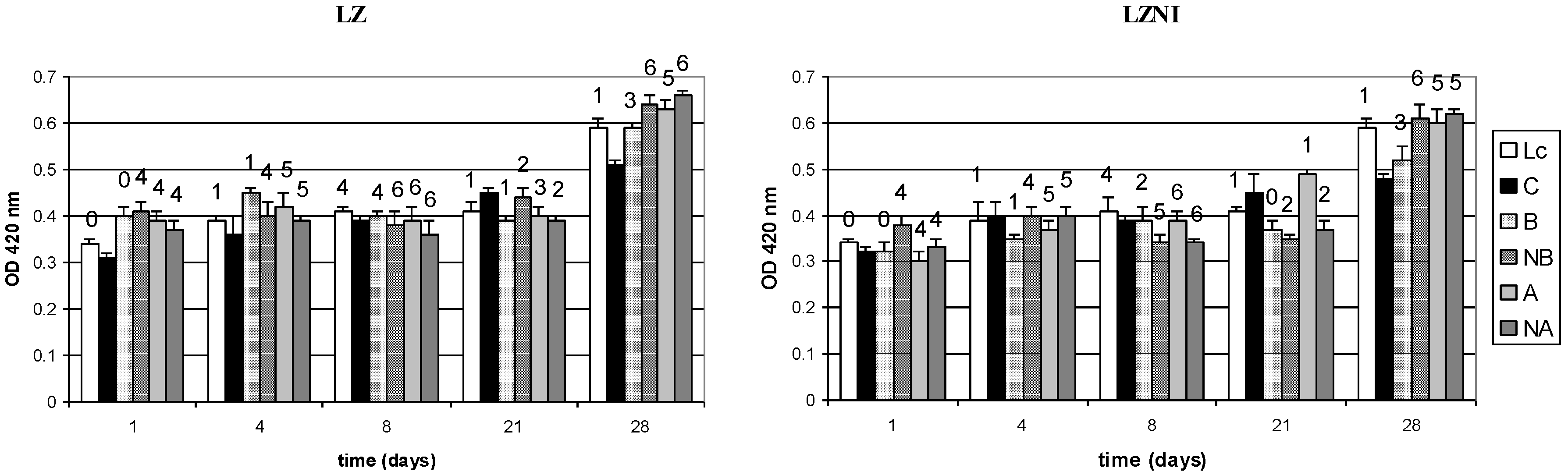
3.4. Reversed-Phase High Performance Liquid Chromatography (RP-HPLC) Analysis
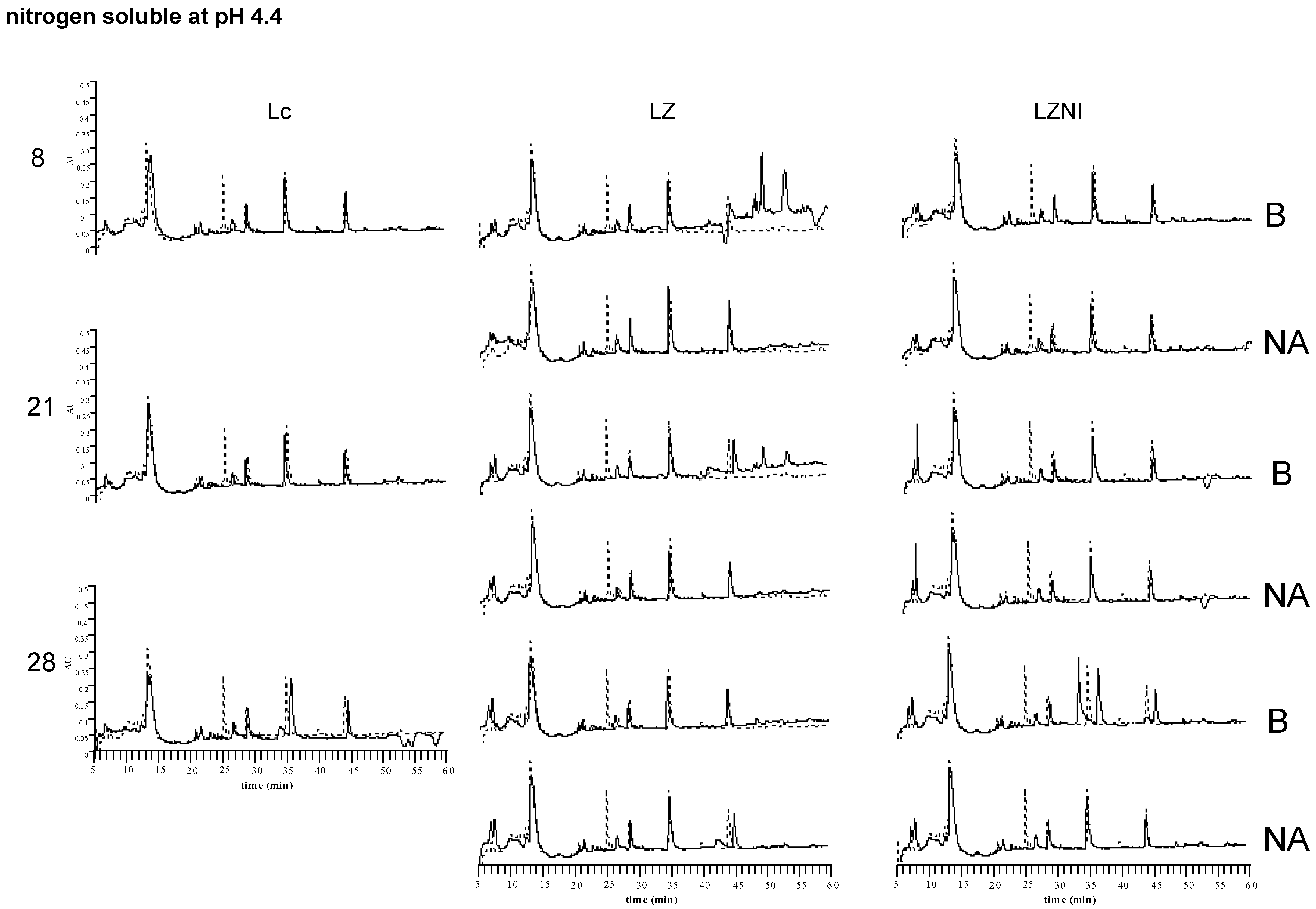
4. Discussion
5. Conclusions
Author Contributions
Conflicts of Interest
References
- Rossi, F.; Gatto, V.; Sabattini, G.; Torriani, S. An assessment of factors characterising the microbiology of Grana Trentino cheese, a Grana-type cheese. Int. J. Dairy Technol. 2012, 65, 401–409. [Google Scholar] [CrossRef]
- Licitra, G.; Carpino, S. The microfloras and sensory profiles of selected protected designation of origin Italian cheeses. Microbiol. Spectr. 2014, 2. [Google Scholar] [CrossRef]
- Aydemir, O.; Harth, H.; Weckx, S.; Dervişoğlu, M.; De Vuyst, L. Microbial communities involved in Kaşar cheese ripening. Food Microbiol. 2015, 46, 587–595. [Google Scholar] [CrossRef] [PubMed]
- De Pasquale, I.; Calasso, M.; Mancini, L.; Ercolini, D.; La Storia, A.; De Angelis, M.; Di Cagno, R.; Gobbetti, M. Causal relationship between microbial ecology dynamics and proteolysis during manufacture and ripening of protected designation of origin (PDO) cheese Canestrato Pugliese. Appl. Environ. Microbiol. 2014, 80, 4085–4094. [Google Scholar] [CrossRef] [PubMed]
- Amarita, F.; Requena, T.; Taborda, G.; Amigo, L.; Pelaez, C. Lactobacillus casei and Lactobacillus plantarum initiate catabolism of methionine by transamination. J. Appl. Microbiol. 2001, 90, 971–978. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Poli, A.; Guglielmini, E.; Sembeni, S.; Spiazzi, M.; Dellaglio, F.; Rossi, F.; Torriani, S. Detection of Staphylococcus aureus and enterotoxin genotype diversity in Monte Veronese, a protected designation of origin Italian cheese. Lett. Appl. Microbiol. 2007, 45, 529–534. [Google Scholar] [CrossRef] [PubMed]
- Garde, S.; Tomillo, J.; Gaya, P.; Medina, M.; Nuñez, M. Proteolysis in Hispánico cheese manufactured using a mesophilic starter, a thermophilic starter and bacteriocin-producing Lactococcus lactis subsp. lactis INIA 415 adjunct culture. J. Agric. Food Chem. 2002, 50, 3479–3485. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Cuesta, M.C.; Requena, T.; Peláez, C. Use of a bacteriocin-producing transconjugant as starter in acceleration of cheese ripening. Int. J. Food Microbiol. 2001, 70, 79–88. [Google Scholar] [CrossRef]
- Ryan, M.P.; Rea, M.C.; Hill, C.; Ross, R.P. An application in Cheddar cheese manufacture for a strain of Lactococcus lactis producing a novel broad-spectrum bacteriocin, lacticin 3147. Appl. Environ. Microbiol. 1996, 62, 612–619. [Google Scholar] [PubMed]
- Ryan, M.P.; Ross, R.P.; Hill, C. Strategy for manipulation of cheese flora using combinations of lacticin-producing and -resistant cultures. Appl. Environ. Microbiol. 2001, 67, 2699–2704. [Google Scholar] [CrossRef] [PubMed][Green Version]
- European Parliament and Council Directive No. 95/2/EC on Food Additives other than Colours and Sweeteners. Available online: http://ec.europa.eu/food/fs/sfp/addit_flavor/flav11_en.pdf (accessed on 14 February 2016).
- EFSA Panel on Biological Hazards (BIOHAZ). Scientific opinion on the maintenance of the list of QPS biological agents intentionally added to food and feed (2013 update). EFSA J. 2013, 11, 3449–3555. [Google Scholar]
- Rilla, N.; Martínez, B.; Delgado, T.; Rodríguez, A. Inhibition of Clostridium tyrobutyricum in Vidiago cheese by Lactococcus lactis ssp. lactis IPLA 729, a nisin Z producer. Int. J. Food Microbiol. 2003, 85, 23–33. [Google Scholar] [CrossRef]
- Perin, L.M.; Dal Bello, B.; Belviso, S.; Zeppa, G.; Carvalho, A.F.; Cocolin, L.; Nero, L.A. Microbiota of Minas cheese as influenced by the nisin producer Lactococcus lactis subsp. lactis GLc05. Int. J. Food Microbiol. 2015, 214, 159–167. [Google Scholar] [CrossRef] [PubMed]
- Rossi, F.; Capodaglio, A.; Dellaglio, F. Genetic modification of Lactobacillus plantarum by heterologous gene integration in a not functional region of the chromosome. Appl. Microbiol. Biotechnol. 2008, 80, 79–86. [Google Scholar] [CrossRef] [PubMed]
- Kuipers, O.P.; Beerthuyzen, M.M.; De Ruyter, P.G.; Luesink, E.J.; De Vos, W.M. Autoregulation of nisin biosynthesis in Lactococcus lactis by signal transduction. J. Biol. Chem. 1995, 270, 27299–27304. [Google Scholar] [CrossRef] [PubMed]
- Ardö, Y.; Polychroniadou, A. Laboratory Manual for Chemical Analysis of Cheese; COST: Brussels, Belgium, 1999; pp. 35–36. [Google Scholar]
- Polychroniadou, A. A simple procedure using trinitrobenzenesulphonic acid for monitoring proteolysis in cheese. J. Dairy Res. 1988, 55, 585–596. [Google Scholar] [CrossRef]
- Ardö, Y.; Gripon, J.C. Comparative study of peptidolysis in some semi-hard round-eyed cheese varieties with different fat contents. J. Dairy Res. 1995, 62, 543–547. [Google Scholar] [CrossRef]
- Benech, R.-O.; Kheadr, E.E.; Lacroix, C.; Fliss, I. Impact of nisin producing culture and liposome-encapsulated nisin on ripening of Lactobacillus added-Cheddar cheese. J. Dairy Sci. 2003, 86, 1895–1909. [Google Scholar] [CrossRef]
- Mota-Meira, M.; LaPointe, G.; Lacroix, C.; Lavoie, M.C. MICs of mutacin B-Ny266, nisin A, vancomycin, and oxacillin against bacterial pathogens. Antimicrob. Agents Chemother. 2000, 44, 24–29. [Google Scholar] [CrossRef] [PubMed]
- Avila, M.; Gómez-Torres, N.; Hernández, M.; Garde, S. Inhibitory activity of reuterin, nisin, lysozyme and nitrite against vegetative cells and spores of dairy-related Clostridium species. Int. J. Food Microbiol. 2014, 172, 70–75. [Google Scholar] [CrossRef] [PubMed]
- Da Silva, M.P.; Daroit, D.J.; Brandelli, A. Inhibition of Listeria monocytogenes in Minas Frescal cheese by free and nanovesicle-encapsulated nisin. Braz. J. Microbiol. 2012, 43, 1414–1418. [Google Scholar]
- Felicio, B.A.; Pinto, M.S.; Oliveira, F.S.; Lempk, M.W.; Pires, A.C.; Lelis, C.A. Effects of nisin on Staphylococcus aureus count and physicochemical properties of Minas Frescal cheese. J. Dairy Sci. 2015, 98, 4364–4369. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.S.; Mantovani, H.C.; Russell, J.B. The binding and degradation of nisin by mixed ruminal bacteria. FEMS Microbiol. Ecol. 2002, 42, 339–345. [Google Scholar] [CrossRef]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rossi, F.; Veneri, G. Use of Bacteriocinogenic Cultures without Inhibiting Cheese Associated Nonstarter Lactic Acid Bacteria; A Trial with Lactobacillus plantarum. Challenges 2016, 7, 4. https://doi.org/10.3390/challe7010004
Rossi F, Veneri G. Use of Bacteriocinogenic Cultures without Inhibiting Cheese Associated Nonstarter Lactic Acid Bacteria; A Trial with Lactobacillus plantarum. Challenges. 2016; 7(1):4. https://doi.org/10.3390/challe7010004
Chicago/Turabian StyleRossi, Franca, and Gianluca Veneri. 2016. "Use of Bacteriocinogenic Cultures without Inhibiting Cheese Associated Nonstarter Lactic Acid Bacteria; A Trial with Lactobacillus plantarum" Challenges 7, no. 1: 4. https://doi.org/10.3390/challe7010004
APA StyleRossi, F., & Veneri, G. (2016). Use of Bacteriocinogenic Cultures without Inhibiting Cheese Associated Nonstarter Lactic Acid Bacteria; A Trial with Lactobacillus plantarum. Challenges, 7(1), 4. https://doi.org/10.3390/challe7010004







