Response of the Durum Wheat Cultivar Um Qais (Triticum turgidum subsp. durum) to Salinity
Abstract
1. Introduction
2. Materials and Methods
2.1. Field Experiment
2.2. Greenhouse Experiment
2.3. Experimental Measurements
2.4. Statistical Analysis
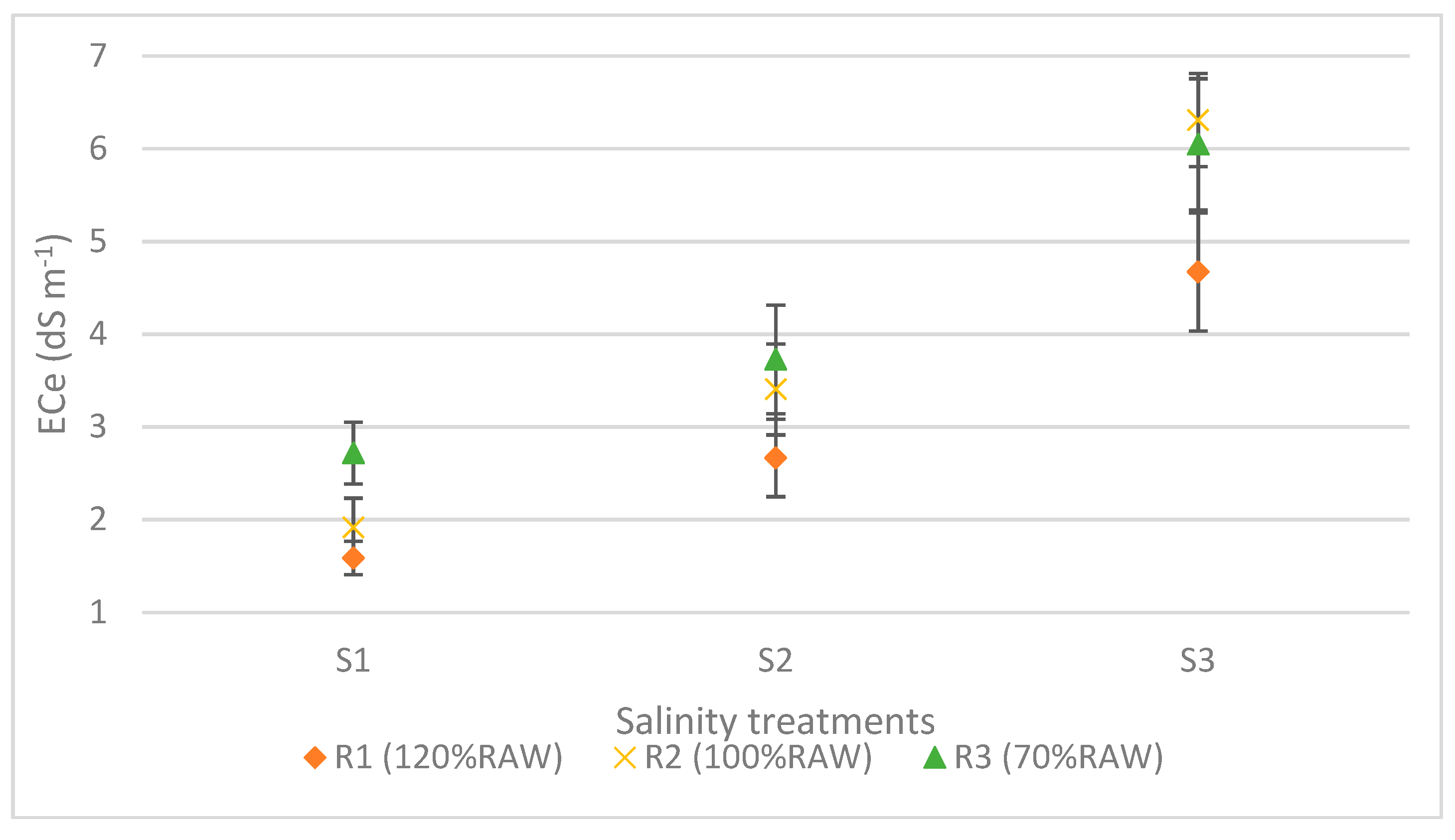
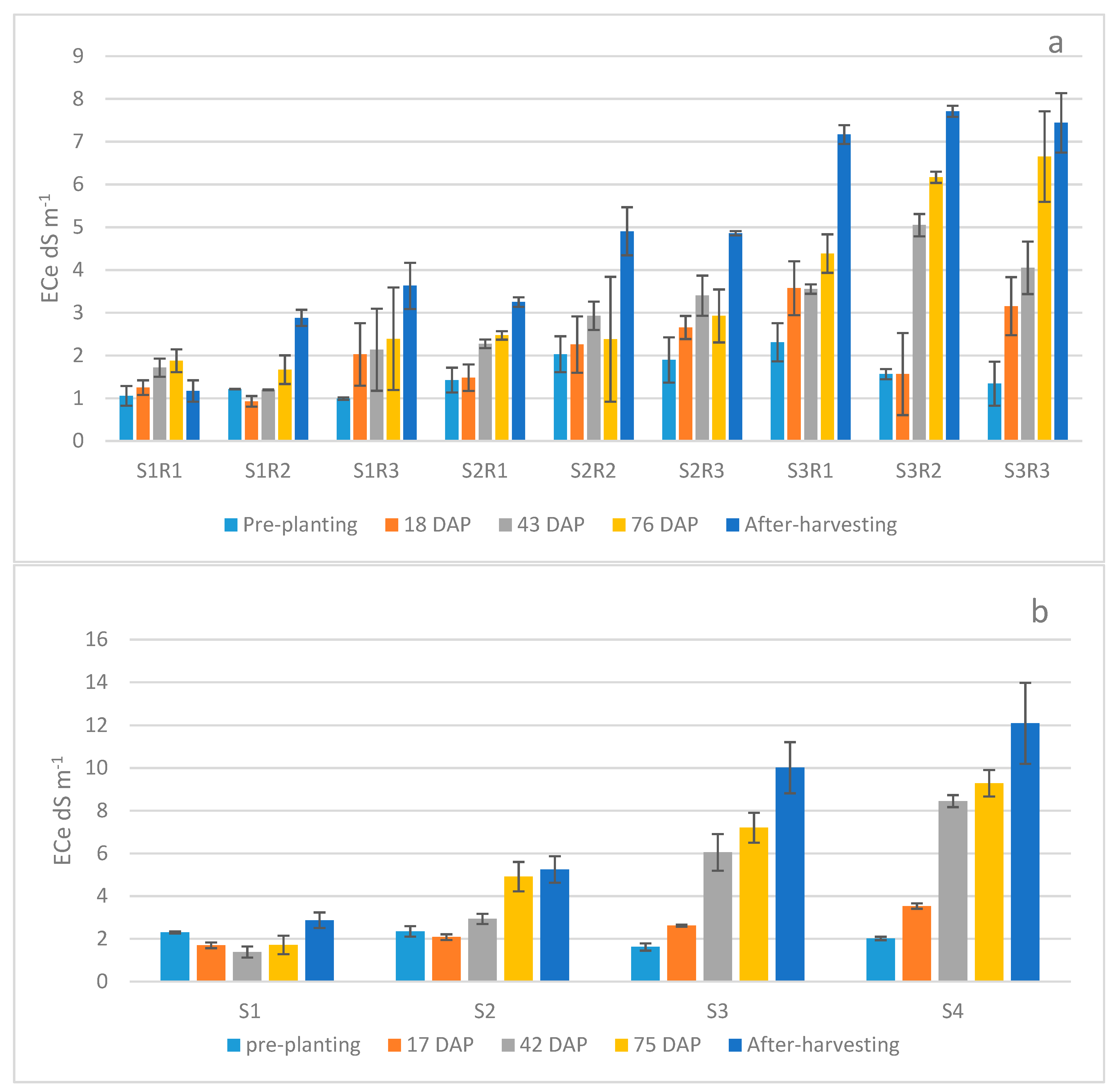
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Flowers, T. Plants and Salinity. J. Exp. Bot. Pref. 2006, 57, IV. [Google Scholar] [CrossRef]
- Läuchli, A.; Grattan, S. Plant Growth and Development Under Salinity Stress. In Advances in Molecular Breeding Toward Drought and Salt Tolerant Crops; Jenks, M.A., Hasegawa, P.M., Jain, S.M., Eds.; Springer: Dordrecht, The Netherlands, 2007; pp. 1–32. [Google Scholar] [CrossRef]
- Hajiboland, R. Role of Arbuscular Mycorrhiza in Amelioration of Salinity. In Salt Stress in Plants; Ahmad, P., Azooz, M.M., Prasad, M.N.V., Eds.; Springer: New York, NY, USA, 2013; pp. 301–354. [Google Scholar] [CrossRef]
- Pimentel, D.; Berger, B.; Filiberto, D.; Newton, M.; Wolfe, B.; Karabinakis, E.; Clark, S.; Poon, E.; Abbett, E.; Nandaopal, S. Water Resources, Agriculture and the Environment. BioScience 2004, 54, 909–918. [Google Scholar] [CrossRef]
- Machado, R.; Serralheiro, R. Soil Salinity: Effect on Vegetable Crop Growth. Management Practices to Prevent and Mitigate Soil Salinization. Horticulturae 2017, 3, 30. [Google Scholar] [CrossRef]
- Jamil, A.; Riaz, S.; Ashraf, M.; Foolad, R. Gene expression profiling of plants under salt stress. Crit. Rev. Plant Sci. 2011, 30, 435–458. [Google Scholar] [CrossRef]
- AbuAisha, E. Permanent Use of Water in Irrigation in Jordan’s Valley. In The Second International Conference of Water; The Jordanian Engineers Syndicate and The Ministry of Water and Irrigating: Amman, Jordan, 2001. [Google Scholar]
- Miyamoto, S.; Chacon, A.; Hossain, M.; Martinez, I. Soil salinity of urban turf areas irrigated with saline water. Landsc. Urban Plan 2005, 71, 233–241. [Google Scholar] [CrossRef]
- Ammari, T.G.; Tahhan, R.; Abubaker, S.; Al-Zu’Bi, Y.; Tahboub, A.; Ta’Any, R.; Abu-Romman, S.; Al-Manaseer, N.; Stietiya, M.H. Soil Salinity Changes in the Jordan Valley Potentially Threaten Sustainable Irrigated Agriculture. Pedosphere 2013, 23, 376–384. [Google Scholar] [CrossRef]
- Ekmekci, B.; Elkoca, O.; Tekkaya, E.; Erden, A. Residual stress state and hardness depth in electric discharge machining: De-ionized water as dielectric liquid. Mach. Sci. Technol. 2005, 9, 39–61. [Google Scholar] [CrossRef]
- Webber, H.A.; Madramootoo, C.A.; Bourgault, M.; Horst, M.G.; Stulina, G.; Smith, D.L. Adapting the CROPGRO Model for Saline Soils: The Case for a Common Bean Crop. Irrig. Sci. 2010, 28, 317–329. [Google Scholar] [CrossRef]
- Al-Zu’bi, Y.; Al-Kharabsheh, A. Multicriteria analysis for water productivity in the Jordan Valley. Water Int. 2003, 28, 501–511. [Google Scholar] [CrossRef]
- Munns, R. Comparative Physiology of Salt and Water Stress. Plant Cell Environ. 2002, 25, 239–250. [Google Scholar] [CrossRef]
- Greenway, H.; Munns, R. Mechanisms of Salt Tolerance in Nonhalophytes. Annu. Rev. Plant Physiol. 1980, 31, 149–190. [Google Scholar] [CrossRef]
- Tavakkoli, E.; Rengasamy, P.; Mcdonald, G.K. The response of barely to salinity stress differs between hydroponics and soil systems. Funct. Plant Biol. 2010, 37, 621–633. [Google Scholar] [CrossRef]
- Borrelli, M.G.; Fragasso, M.; Nigro, F.; Platani, C.; Papa, R.; Beleggia, R.; Trono, D. Analysis of metabolic and mineral changes in response to salt stress in durum wheat (Triticum turgidum ssp. durum) genotypes, which differ in salinity tolerance. Plant Physiol. Biochem. 2018, 133, 57–70. [Google Scholar] [CrossRef] [PubMed]
- Porceddu, E.; Damania, B.; Qualset, O. Genetics and breeding of durum wheat. In Proceedings of the International Symposium on Genetics and Breeding of Durum Wheat, Rome, Italy, 27–30 May 2013. [Google Scholar]
- Elings, A.; Nachit, M. Durum wheat landraces from Syria. I. Agro-ecological and morphological characterization. Euphytica 1991, 53, 211–224. [Google Scholar] [CrossRef]
- Jaradat, A. Phenotypic divergence for morphological and yield-related traits among landrace genotypes of durum wheat from Jordan. Euphytica 1991, 52, 155–164. [Google Scholar] [CrossRef]
- Munns, R.; Passioura, J.B.; Guo, J.; Chazen, O.; Cramer, G.R. water relations and leaf expansoin: Importance of time scale. J. Exp. Bot. 2000, 51, 1495–1504. [Google Scholar] [CrossRef] [PubMed]
- Kabbaj, H.; Sall, A.; Al-Abdallat, A.; Geleta, M.; Amri, A.; Matlouf, A.; Belkadi, B.; Ortiz, R.; Bassi, F. Genetic Diversity within a Global Panel of Durum Wheat (Triticum durum) Landraces and Modern Germplasm Reveals the History of Alleles Exchange. Plant Sci. 2017, 8, 1495–1504. [Google Scholar] [CrossRef]
- Al-Rjoub, F.A.; Al-Samarrai, M.A. Growth and Yield Responses of Three Durum Wheat Cultivars Subjected to Four Levels of Available Soil Moisture. Agric. Sci. 2006, 33, 153–164. [Google Scholar]
- Thalji, T.; Shalaldeh, G. Screening Wheat and Barely Genotypes for Salinity Resistance. J. Agron. 2007, 6, 647–650. [Google Scholar]
- Katerji, N.; Mastrorilli, M.; van Hoon, J.W.; Lahmer, F.Z.; Hamdy, A.; Oweis, T. Durum wheat and barley productivity in saline-drought environments. Eur. J. Agron. 2009, 31, 1–9. [Google Scholar] [CrossRef]
- Dura, S.; Duwayri, M.; Nachit, M. Detection of molecular markers associated with yield and yield components in durum wheat (Triticum turgidum L. var. durum Desf.). Afr. J. Agric. Res. 2013, 8, 2118–2128. [Google Scholar]
- Food and Agriculture Organization of the United Nations (FAO). The State of Food and Agriculture; FAO: Rome, Italy, 1993. [Google Scholar]
- Maas, E.V.; Hoffman, G.J. Crop Salt Tolerance-Current Assessment. J. Irrig. Drain. Div. 1977, 103, 115–134. [Google Scholar]
- El-Hendawy, S.E.; Hu, Y.; Yakout, G.M.; Awad, A.M.; Hafiz, S.E.; Schmidhalter, U. Evaluating Salt Tolerance of Wheat Genotypes Using Multiple Parameters. Eur. J. Agron. 2005, 22, 243–253. [Google Scholar] [CrossRef]
- Munns, R.; Hare, R.A.; James, R.A.; Rebetzke, G.J. Genitic variation for improving the salt tolerance of durum wheat. Aust. J. Res. 2000, 51, 69–74. [Google Scholar] [CrossRef]
- Prathapar, S.A.; Qureshi, A.S. Modelling the Effects of Deficit Irrigation on Soil Salinity, Depth to Water Table and Transpiration in Semi-arid Zones with Monsoonal Rains. Int. J. Water Resour. Dev. 1999, 15, 141–159. [Google Scholar] [CrossRef]
- Allen, R.G.; Pereira, L.S.; Raes, D.; Smith, M. Crop Evapotranspiration, Guidelines for Computing Crop Water Requirements; FAO Irrigation and Drainage Paper 56; FAO: Rome, Italy, 1998. [Google Scholar]
- Day, P.R. Particle Fractionation and Particle-Size Analysis. In Methods of Soil Analysis. Part 1. Physical and Mineralogical Properties, Including Statistics of Measurement and Sampling; Black, C.A., Ed.; Agron. Monogr. 9.1; ASA, SSSA: Madison, WI, USA, 1965; pp. 545–567. [Google Scholar] [CrossRef]
- Gardner, W.H. Water Content. In Methods of Soil Analysis. Part 1. Physical and Mineralogical Properties, Including Statistics of Measurement and Sampling; Black, C.A., Ed.; Agron. Monogr 1965. 9.1; ASA, SSSA: Madison, WI, USA, 1965; pp. 82–127. [Google Scholar] [CrossRef]
- Blake, G.R. Bulk Density1. In Methods of Soil Analysis. Part 1. Physical and Mineralogical Properties, Including Statistics of Measurement and Sampling; Black, C.A., Ed.; Agron. Monogr 1965. 9.1; ASA, SSSA: Madison, WI, USA, 1965; pp. 374–390. [Google Scholar] [CrossRef]
- Rhoades, J.D.; Chanduvi, F.; Lesch, S. Soil Salinity Assessment: Methods of Interpretation of Electrical Conductivity Measurements; FAO: Rome, Italy, 1999. [Google Scholar]
- Chapman, H.D. Cation-Exchange Capacity 1. In Methods of Soil Analysis. Part 2. Chemical and Microbiological Properties, 2nd ed.; Norman, A.G., Ed.; Agron. Monogr 1965. 9.2; ASA, SSSA: Madison, WI, USA, 1965; pp. 891–901. [Google Scholar] [CrossRef]
- Bower, C.A.; Wilcox, L.V. Soluble Salts 1. In Methods of Soil Analysis. Part 2. Chemical and Microbiological Properties; Norman, A.G., Ed.; Agron. Monogr.1965. 9.2; ASA, SSSA: Madison, WI, USA, 1965; pp. 933–951. [Google Scholar] [CrossRef]
- Olsen, S.R.; Dean, L.A. Phosphorus 1. In Methods of Soil Analysis. Part 2. Chemical and Microbiological Properties, 2nd ed.; Norman, A.G., Ed.; Agron. Monogr 1965. 9.2; ASA, SSSA: Madison, WI, USA, 1965; pp. 1035–1049. [Google Scholar] [CrossRef]
- Bremner, J.M. Total Nitrogen 1. In Methods of Soil Analysis. Part 2. Chemical and Microbiological Properties, 2nd ed.; Norman, A.G., Ed.; Agron. Monogr 1965. 9.2; ASA, SSSA: Madison, WI, USA, 1965; pp. 1149–1178. [Google Scholar] [CrossRef]
- Rani, B.; Sharma, V.K. Standarisation of methodology for obtaining the desired salt stress environment for salinity effect observation in rice seedlings. Int. J. Environ. Sci. 2015, 6, 232. [Google Scholar] [CrossRef]
- Ben Ahmed, C.; Magdich, S.; Ben Rouina, B.; Boukhris, M.; Ben Abdullah, F. Saline Water Irrigation Effects on Soil Salinity Distribution and Some Physiological Responses of Field Grown Chemlali Olive. J. Environ. Manag. 2012, 113, 538–544. [Google Scholar] [CrossRef]
- Grieve, C.M.; Grattan, S.R.; Maas, E.V. Plant salt tolerance. Agric. Salin. Assess. Manag. 2012, 2, 405–459. [Google Scholar]
- Barlow, K.M.; Christy, B.P.; O’Leary, G.J.; Riffkin, P.A.; Nuttall, J.G. Simulating the impact of extreme heat and frost events on wheat crop production: A review. Field Crops Res. 2015, 171, 109–119. [Google Scholar] [CrossRef]
- Singh, K.N.; Chatrath, R. Salinity tolerance. In Application of Physiology in Wheat Breeding; Reynolds, M.P., Monasterio, J.I.O., McNab, A., Eds.; CIMMYT: Mexico City, Mexico, 2001; pp. 101–110. [Google Scholar]
- Steppuhn, K.; Volkmar, K.M.; Miller, P.R. Comparing canola foeld pea, dry bean, and durum crops grown in saline media. Crop Sci. 2001, 41, 1827–1833. [Google Scholar] [CrossRef]
- Saqib, M.; Akhtar, J.; Qureshi, R.H. Pot study on wheat growth in saline and waterlogged compacted soil I. Grain yield and yield components. Soil Till. Res. 2004, 77, 169–177. [Google Scholar] [CrossRef]
- Dikgwatlhe, S.B.; Ceronio, G.M.; van Rensburg, L.D. Wheat (Triticum aestivum, L.) Growth and Yield Response to Saline Irrigation Water under Controlled Conditions. S. Afr. J. Plant Soil 2008, 25, 172–177. [Google Scholar] [CrossRef][Green Version]
- Colmer, T.D.; Flowers, T.J.; Munns, R. Use of wild relatives to improve salt tolerance in wheat. J. Exp. Bot. 2006, 57, 1059–1078. [Google Scholar] [CrossRef] [PubMed]


| a. Soil Chemical Properties | |
|---|---|
| Cl (ppm) | 0.33 ± 0.05 |
| Na (ppm) | 41.98 ± 6.52 |
| K (ppm) | 54.55 ± 0.04 |
| Ca (ppm) | 120 ± 0.65 |
| P (ppm) | 42 ± 4.10 |
| Mg (ppm) | 105.60 ± 1.24 |
| HCO3 (ppm) | 231.86 ± 10.01 |
| Organic C (%) | 0.23 ± 0.01 |
| N (%) | 0.50 ± 0.05 |
| CEC (cmolkg−1) | 18.60 ± 0.48 |
| SAR | 0.83 ± 0.02 |
| ECe (dS m−1) | 4.00 ± 0.23 |
| pH | 8.00 ± 0.09 |
| b. Soil Physical Properties | |
| Clay (%) | 16 ± 0.48 |
| Sand (%) | 73 ± 0.65 |
| Upper limit (cm3 cm−3) | 0.19 ± 0.01 |
| Lower limit (cm3 cm−3) | 0.09 ± 0.01 |
| Saturated water content (cm3 cm−3) | 0.38 ± 0.02 |
| Bulk density (g cm−3) | 1.58 ± 0.11 |
| Ca (ppm) | Mg (ppm) | Cl (ppm) | Na (ppm) | SAR | pH | ECiw (dS m−1) | |
|---|---|---|---|---|---|---|---|
| Field exp. | 92 ± 2.10 | 40.8 ± 0.47 | 0.35 ± 0.01 | 240 ± 0.91 | 5.22 ± 0.05 | 7.8 ± 0.04 | 2 ± 0.31 |
| Greenhouse exp. | 28 ± 0.46 | 31.2 ± 2.08 | 0.054 ± 0.01 | 170 ± 0.57 | 5.230 ± 0.05 | 7.53 ± 0.14 | 0.68 ± 0.05 |
| Tillering | Flag Leaf | Maturity | |||||||
|---|---|---|---|---|---|---|---|---|---|
| DAP | LAI | P.HEIGHT | DAP | LAI | P. HEIGHT | DAP | LAI | P. HEIGHT | |
| a. Field Experiment | |||||||||
| S1R1 control | 19 | 0.40 ± 0.11 | 31 ± 0.75 | 53 | 5.03 ± 0.41 | 84 ± 0.75 | 94 | 2.89 ± 0.58 | 103 ± 2.53 |
| S1R2 | 19 | 0.32 ± 0.07 | 31 ± 0.48 | 53 | 5.18 ± 0.34 | 86 ± 0.29 | 94 | 2.53 ± 0.25 | 104 ± 1.32 |
| S1R3 | 19 | 0.38 ± 0.09 | 31 ± 0.63 | 53 | 4.50 ± 0.19 | 85 ± 0.29 | 94 | 2.41 ± 0.46 | 106± 1.81 |
| S2R1 | 19 | 0.47 ± 0.08 | 32 ± 0.48 | 53 | 5.06 ± 0.18 | 84 ± 0.29 | 94 | 2.85 ± 0.18 | 104 ± 1.81 |
| S2R2 | 19 | 0.44 ± 0.03 | 32 ± 0.63 | 53 | 4.36 ± 0.09 | 85 ± 1.04 | 94 | 2.25 ± 0.36 | 103 ± 2.63 |
| S2R3 | 19 | 0.20 ± 0.01 | 32 ± 0.48 | 53 | 4.09 ± 0.10 * | 85 ± 0.63 | 94 | 2.11 ± 0.04 | 103 ± 0.45 |
| S3R1 | 19 | 0.27 ± 0.02 | 30 ± 0.25 | 53 | 3.96 ± 0.16 * | 83 ± 0.29 | 94 | 2.41 ± 0.38 | 103 ± 1.43 |
| S3R2 | 19 | 0.31 ± 0.09 | 30 ± 0.29 | 53 | 3.81 ± 0.20 * | 81 ± 0.41 | 94 | 2.23 ± 0.03 | 101 ± 1.19 |
| S3R3 | 19 | 0.21 ± 0.03 | 31 ± 0.48 | 53 | 3.73 ± 0.18 * | 81 ± 0.41 | 94 | 1.84 ± 0.26 | 098 ± 1.09 |
| b. Greenhouse | |||||||||
| S1 control | 26 | NA | 41 ± 0.48 | 70 | NA | 97 ± 0.35 | 98 | NA | 106 ± 2.39 |
| S2 | 26 | NA | 39 ± 0.48 * | 70 | NA | 84 ± 0.28 * | 98 | NA | 98 ± 1.94 * |
| S3 | 26 | NA | 35 ± 0.25 * | 70 | NA | 79 ± 0.42 * | 98 | NA | 90 ± 2.46 * |
| S4 | 26 | NA | 35 ± 0.25 * | 70 | NA | 73 ± 0.50 * | 96 | NA | 85 ± 0.75 * |
| Treatments | Grains Number m−2 | Grain Yield kg ha−1 | Above-Ground Biomass kg ha−1 |
|---|---|---|---|
| a. Field Experiment | |||
| S1R1 | 6131 ± 150 | 3311 ± 81 | 9353 ± 241 |
| S1R2 | 6124 ± 248 | 3307 ± 134 | 9436 ± 471 |
| S1R3 | 6048 ± 247 | 3265 ± 133 | 9391 ± 407 |
| S2R1 | 6130 ± 181 | 3310 ± 97 | 9930 ± 293 |
| S2R2 | 5773 ± 210 | 3117 ± 11 | 9665 ± 353 |
| S2R3 | 5390 ± 136 | 2911 ± 73 * | 9024 ± 227 |
| S3R1 | 5825 ± 92 | 3145 ± 50 | 9752 ± 155 |
| S3R2 | 5508 ± 105 | 2754 ± 52 * | 8813 ± 169 |
| S3R3 | 4963 ± 80 | 2430 ± 11 * | 8021 ± 38 * |
| b. Greenhouse Experiment | |||
| S1 | 2742 ± 228 | 1448 ± 103 | 4576 ± 355 |
| S2 | 2288 ± 192 | 1148 ± 94 * | 4110 ± 352 |
| S3 | 2568 ± 141 | 1100 ± 52 * | 4129 ± 207 |
| S4 | 2242 ± 73 * | 878 ± 67 * | 3365 ± 109 * |
| Treatments | DF | Mean | CV (%) | SE | F | p-Value | F crit |
|---|---|---|---|---|---|---|---|
| a. Field Experiment | |||||||
| Irrigation water salinity (S) | 2 | NA | NA | NA | 19.82709 * | 9.8 × 10−8 * | 3.109311 |
| Irrigation water amount (R) | 2 | NA | NA | NA | 2.288454 | 0.107945 | 3.109311 |
| Interaction | 4 | NA | NA | NA | 0.127075 | 0.972231 | 2.484441 |
| Number of grains (m−2) | 35 | 5766 | 9 | 82.3 | 5.581253 * | 0.000322 * | 2.305313 |
| Grain yield (kg ha−1) | 35 | 3061 | 11 | 55.8 | 11.13967 * | 8.34 × 10−7 * | 2.305313 |
| Aboveground biomass (kg ha−1) | 35 | 9265 | 8 | 125.9 | 3.981641 * | 0.003164 * | 2.305313 |
| Plant height (cm) | 35 | 102.7 | 4 | 0.60 | 1.552936 | 0.185889 | 2.305313 |
| LAI | 35 | 4.40 | 15 | 0.10 | 6.164087 * | 0.000151 * | 2.305313 |
| b. Greenhouse Experiment | |||||||
| Number of grains (m−2) | 15 | 2460 | 0.00 | 2.3 | 1.958419 | 0.174143 | 3.490295 |
| Grain yield (kg ha−1) | 15 | 1143 | 22 | 64.1 | 8.175895 * | 0.003123 * | 3.490295 |
| Aboveground biomass kg ha−1 | 15 | 4045 | 17 | 167.1 | 3.287114 | 0.058261 | 3.490295 |
| Plant height (cm) | 15 | 94.3 | 10 | 2.3 | 16.5935 * | 0.000853 * | 4.066181 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hamdi, L.; Suleiman, A.; Hoogenboom, G.; Shelia, V. Response of the Durum Wheat Cultivar Um Qais (Triticum turgidum subsp. durum) to Salinity. Agriculture 2019, 9, 135. https://doi.org/10.3390/agriculture9070135
Hamdi L, Suleiman A, Hoogenboom G, Shelia V. Response of the Durum Wheat Cultivar Um Qais (Triticum turgidum subsp. durum) to Salinity. Agriculture. 2019; 9(7):135. https://doi.org/10.3390/agriculture9070135
Chicago/Turabian StyleHamdi, Luma, Ayman Suleiman, Gerrit Hoogenboom, and Vakhtang Shelia. 2019. "Response of the Durum Wheat Cultivar Um Qais (Triticum turgidum subsp. durum) to Salinity" Agriculture 9, no. 7: 135. https://doi.org/10.3390/agriculture9070135
APA StyleHamdi, L., Suleiman, A., Hoogenboom, G., & Shelia, V. (2019). Response of the Durum Wheat Cultivar Um Qais (Triticum turgidum subsp. durum) to Salinity. Agriculture, 9(7), 135. https://doi.org/10.3390/agriculture9070135





