Variability in the Global Proso Millet (Panicum miliaceum L.) Germplasm Collection Conserved at the ICRISAT Genebank
Abstract
1. Introduction
2. Materials and Methods
2.1. Experiment Details
2.2. Data Collection
2.3. Data Analyses
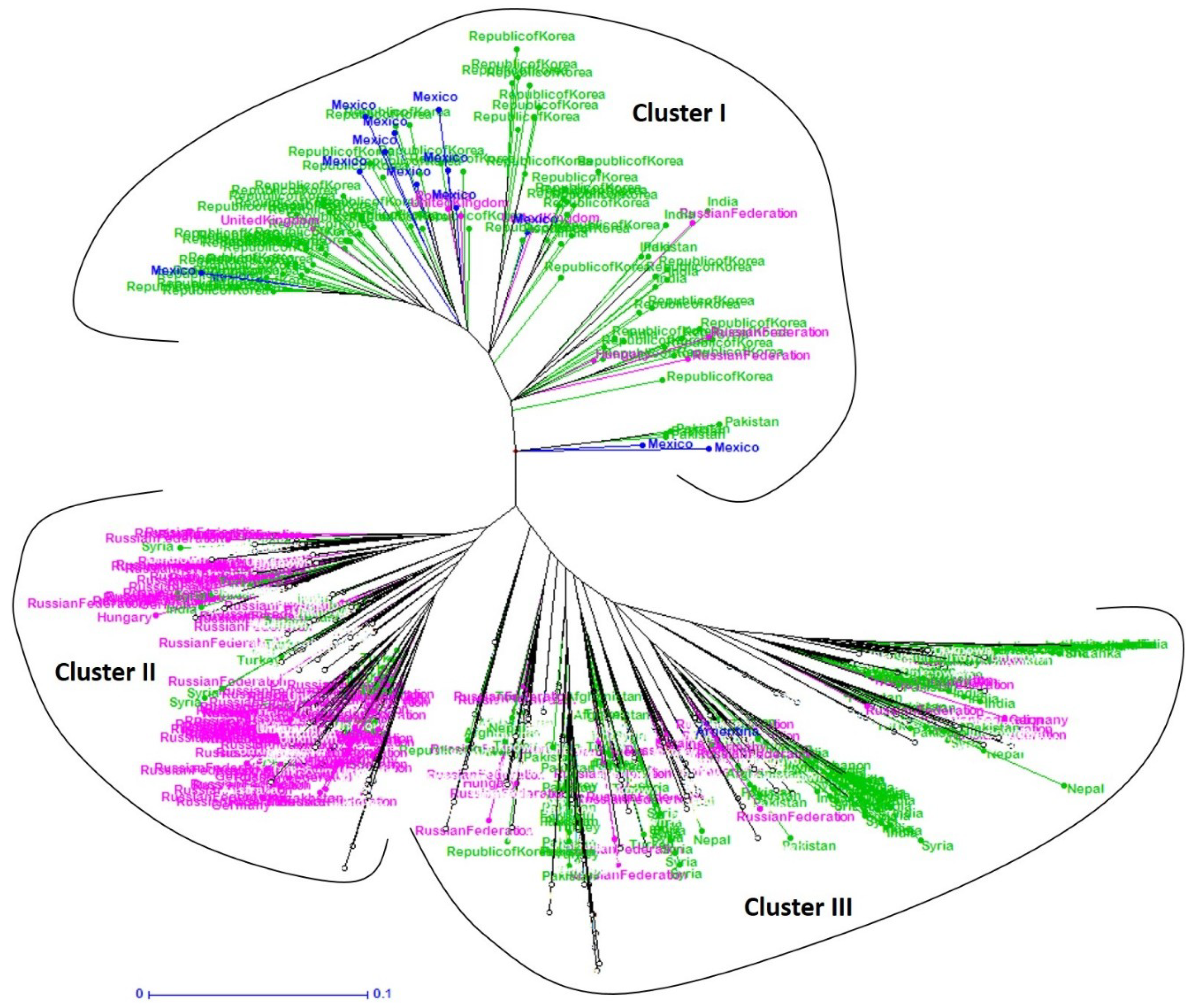
3. Results
3.1. Racial and Geographical Distribution
3.2. Racial and Geographical Diversity
3.2.1. Qualitative Traits
3.2.2. Quantitative Traits
3.2.3. Trait-Specific Sources
3.2.4. Trait Associations
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- De Wet, J.M.J. Origin, evolution and systematics of minor cereals. In Small Millets in Global Agriculture; Seetharam, A., Riley, K.W., Harinarayana, G., Eds.; Oxford & IBH Publishing Co. Pvt. Ltd.: New Delhi, India, 1986; pp. 19–30. [Google Scholar]
- Vavilov, N.I. Centers of origin of cultivated plants. Tr po Prikl Bot Genet Sel [Bull Appl Bot & Genet Sel] 1926, 16, 139–248. (In Russian) [Google Scholar]
- Harlan, J.R. Crops and Man. American Society of Agronomy; Crop Science Society of America, Inc.: Madison, WI, USA, 1975. [Google Scholar]
- Lu, H.; Zhang, J.; Liu, K.-B.; Wu, N.; Li, Y.; Zhou, K.; Ye, M.; Zhang, T.; Zhang, H.; Yang, X.; et al. Earliest domestication of common millet (Panicum miliaceum) in East Asia extended to 10,000 years ago. Proc. Acad. Sci. 2009, 106, 7367–7372. [Google Scholar] [CrossRef] [PubMed]
- Hunt, H.V.; Linden, M.V.; Liu, X.; Motuzaite-Matuzeviciute, G.; Colledge, S.; Jones, M.K. Millets across Eurasia: chronology and context of early records of the genera Panicum and Setaria from archaeological sites in the Old World. Veg. Hist. Archaeobotany 2008, 17, 5–18. [Google Scholar] [CrossRef] [PubMed]
- Hunt, H.V.; Campana, M.G.; Lawes, M.C.; Park, Y.; Bower, M.A.; Howe, C.J.; Jones, M.K. Genetic diversity and phylogeography of broomcorn millet (Panicum miliaceum L.) across Eurasia. Mol. Ecol. 2011, 20, 4756–4771. [Google Scholar] [CrossRef] [PubMed]
- Rajput, S.G.; Plyler-Harveson, T.; Santra, D.K. Development and Characterization of SSR Markers in Proso Millet Based on Switchgrass Genomics. Am. J. Plant Sci. 2014, 5, 175–186. [Google Scholar] [CrossRef]
- Diao, X. Production and genetic improvement of minor cereals in China. Crop. J. 2017, 5, 103–114. [Google Scholar] [CrossRef]
- Habiyaremye, C.; Matanguihan, J.B.; Guedes, J.D.; Ganjyal, G.M.; Whiteman, M.R.; Kidwell, K.K.; Murphy, K.M. Proso Millet (Panicum miliaceum L.) and Its Potential for Cultivation in the Pacific Northwest, U.S.: A Review. Front. Plant Sci. 2017, 7, 501. [Google Scholar] [CrossRef] [PubMed]
- Bhat, B.V.; Tonapi, V.A.; Rao, B.D.; Singode, A.; Santra, D. Production and utilization of millets in India. In Proceedings of the International Millet Symposium and the 3rd International Symposium on Broomcorn Millet (3rd ISBM), Fort Collins, CO, USA, 8–12 August 2018; pp. 24–26. [Google Scholar]
- Park, C.H. Production and Utilization of Broomcorn Millet in Korea. In Proceedings of the International Millet Symposium and the 3rd International Symposium on Broomcorn Millet (3rd ISBM) Program and Abstracts, Fort Collins, CO, USA, 8–12 August 2018; p. 27. [Google Scholar]
- Vetriventhan, M.; Upadhyaya, H.D. Diversity and trait-specific sources for productivity and nutritional traits in the global proso millet (Panicum miliaceum L.) germplasm collection. Crop. J. 2018, 6, 451–463. [Google Scholar] [CrossRef]
- Gomeshe, S.S. Proso millet, Panicum miliaceum (L.): Genetic improvement and research needs. In Millets and Sorghum: Biology and Genetic Improvement; Patil, J.V., Ed.; John Wiley & Sons Ltd.: Chichester, UK, 2017; pp. 150–179. [Google Scholar]
- Kalinová, J.; Moudrý, J. Content and Quality of Protein in Proso Millet (Panicum miliaceum L.) Varieties. Plant Foods Hum. Nutr. 2006, 61, 43–47. [Google Scholar] [CrossRef] [PubMed]
- Saleh, A.S.; Zhang, Q.; Chen, J.; Shen, Q. Millet Grains: Nutritional Quality, Processing, and Potential Health Benefits. Compr. Rev. Food Sci. Food Saf. 2013, 12, 281–295. [Google Scholar] [CrossRef]
- Wang, R.; Hunt, H.V.; Qiao, Z.; Wang, L.; Han, Y. Diversity and Cultivation of Broomcorn Millet (Panicum miliaceum L.) in China: A Review. Econ. Bot. 2016, 70, 332–342. [Google Scholar] [CrossRef]
- IBPGR. Descriptors for Panicum miliaceum and P. sumatrense; IBPGR: Rome, Italy, 1985. [Google Scholar]
- Newman, D. The distribution of range in samples from a normal population, expressed in terms of an independent estimate of standard deviation. Biometrika 1939, 31, 20–30. [Google Scholar] [CrossRef]
- Keuls, M. The use of the “Studentized range” in connection with an analysis of variance. Euphytica 1952, 1, 112–122. [Google Scholar] [CrossRef]
- Levene, H. Robust Tests for Equality of Variances. In Contributions to Probability and Statistics: Essays in Honor of Harold Hotelling; Olkin, I., Ghurye, S.G., Hoeffding, W., Madow, W.G., Mann, H.B., Eds.; Stanford University Press: Palo Alto, CA, USA, 1960; pp. 278–292. [Google Scholar]
- de Mendiburu, F. agricolae: Statistical Procedures for Agricultural Research. R package version 1.2-7. Comprehensive R Arch. Network. Available online: https://cran.r-project.org/package=agricolae (accessed on 21 May 2018).
- Fox, J.; Weisberg, S. An {R} Companion to Applied Regression, 2nd ed.; Sage: Thousand Oaks, CA, USA, 2011; Available online: http://socserv.socsci.mcmaster.ca/jfox/Books/Companion (accessed on 25 October 2018).
- Shannon, C.E.; Weaver, W. The Mathematical Theory of Communication; The University of Illinois Press: Urbana, IL, USA, 1949. [Google Scholar]
- Gower, J.C. A general coefficient of similarity and some of its properties. Biometrics 1971, 27, 857–874. [Google Scholar] [CrossRef]
- Maechler, M.; Rousseeuw, P.; Struyf, A.; Hubert, M.; Hornik, K. Cluster: Cluster Analysis Basics and Extensions, R Package Version 2.0.7-1. 2017.
- Saitou, N.; Nei, M. The neighbour-joining method: A new method for reconstructing phylogenetic trees. Mol. Biol. Evol. 1987, 4, 406–425. [Google Scholar] [PubMed]
- Perrier, X.; Jacquemoud-Collet, J.P. DARwin Software. 2006. Available online: http://darwin.cirad.fr/darwin (accessed on 3 August 2018).

| Country | Race | Unclassified | Total | ||||
|---|---|---|---|---|---|---|---|
| compactum | contractum | miliaceum | ovatum | patentissimum | |||
| Russian Federation | 28 | 13 | 64 | 5 | 11 | 121 | |
| India | 3 | 57 | 8 | 8 | 76 | ||
| Republic of Korea | 2 | 16 | 52 | 3 | 73 | ||
| Turkey | 8 | 6 | 30 | 3 | 2 | 49 | |
| Pakistan | 1 | 1 | 31 | 8 | 41 | ||
| Syria | 1 | 10 | 17 | 2 | 4 | 1 | 35 |
| Afghanistan | 5 | 11 | 16 | ||||
| Mexico | 4 | 3 | 2 | 4 | 13 | ||
| Germany | 1 | 2 | 9 | 12 | |||
| Hungary | 1 | 1 | 6 | 2 | 10 | ||
| Iran | 1 | 8 | 9 | ||||
| Nepal | 4 | 2 | 6 | ||||
| Ukraine | 1 | 3 | 4 | ||||
| United Kingdom | 1 | 2 | 1 | 4 | |||
| Australia | 2 | 2 | |||||
| Bangladesh | 2 | 2 | |||||
| China | 1 | 1 | 2 | ||||
| Iraq | 2 | 2 | |||||
| Kazakhstan | 1 | 1 | 2 | ||||
| Sri Lanka | 2 | 2 | |||||
| Argentina | 1 | 1 | |||||
| Canada | 1 | 1 | |||||
| Japan | 1 | 1 | |||||
| Kenya | 1 | 1 | |||||
| Kyrgyzstan | 1 | 1 | |||||
| Lebanon | 1 | 1 | |||||
| Malawi | 1 | 1 | |||||
| Romania | 1 | 1 | |||||
| Spain | 1 | 1 | |||||
| Socialist Federal Republic of Yugoslavia | 1 | 1 | |||||
| Unknown origin | 45 | 29 | 234 | 35 | 15 | 358 | |
| Total | 98 | 92 | 539 | 48 | 63 | 9 | 849 |
| Trait | Class | Race | Entire Set | ||||
|---|---|---|---|---|---|---|---|
| compactum | contractum | miliaceum | ovatum | patentissimum | |||
| Growth habit | Decumbent | 85 (86.7) † | 53 (58.2) | 407 (75.5) | 43 (89.6) | 38 (60.3) | 627 (74.6) |
| Erect | 11 (11.2) | 24 (26.4) | 93 (17.3) | 3 (6.3) | 18 (28.6) | 149 (17.8) | |
| Erect geniculate | 2 (2.0) | 14 (15.4) | 39 (7.2) | 2 (4.2) | 7 (11.1) | 64 (7.6) | |
| Culm branching | High | 48 (49.0) | 25 (27.5) | 269 (49.9) | 21 (43.8) | 15 (23.8) | 379 (45.0) |
| Low | 14 (14.3) | 31 (34.1) | 93 (17.3) | 1 (2.1) | 26 (41.3) | 165 (19.6) | |
| Medium | 36 (36.7) | 36 (39.6) | 177 (32.8) | 26 (54.2) | 22 (34.9) | 297 (35.4) | |
| Sheath pubescence | Dense | 26 (26.5) | 23 (25.3) | 167 (31.0) | 7 (14.6) | 25 (39.7) | 249 (29.5) |
| Medium | 34 (34.7) | 42 (46.2) | 230 (42.7) | 20 (21.7) | 15 (23.8) | 341 (40.6) | |
| Sparse | 38 (38.8) | 27 (29.7) | 142 (26.3) | 21 (43.8) | 23 (36.5) | 251 (29.9) | |
| Ligule pubescence | Dense | 17 (17.3) | 8 (9.9) | 85 (15.8) | 4 (8.3) | 9 (14.3) | 124 (14.6) |
| Medium | 30 (30.6) | 34 (37.4) | 235 (43.6) | 16 (33.3) | 20 (31.7) | 335 (39.9) | |
| Sparse | 51 (52.0) | 50 (54.9) | 219 (40.6) | 28 (58.3) | 34 (54.0) | 382 (45.5) | |
| Leaf pubescence | Dense | 17 (17.3) | 8 (8.8) | 79 (14.7) | 4 (8.3) | 6 (9.5) | 114 (13.6) |
| Medium | 26 (26.5) | 28 (30.8) | 213 (39.5) | 13 (21.1) | 17 (27.0) | 297 (35.4) | |
| Sparse | 55 (56.1) | 56 (61.5) | 247 (45.8) | 31 (64.6) | 40 (63.5) | 430 (51.1) | |
| Inflorescence shape | Arched dense | 3 (3.1) | 73 (80.2) | 47 (8.7) | - | 1 (1.6) | 124 (14.8) |
| Arched sparse | 2 (2.0) | 5 (5.5) | 13 (2.4) | - | 43 (68.3) | 63 (7.5) | |
| Diffuse dense | 1 (1.0) | 2 (2.2) | 222 (41.2) | - | 8 (12.7) | 233 (27.7) | |
| Diffuse sparse | 4 (4.1) | 3 (3.3) | 234 (43.4) | 2 (4.2) | 11 (17.5) | 254 (30.2) | |
| Elliptic dense | 40 (40.9) | 2 (2.2) | 16 (3.0) | 1 (2.1) | - | 59 (7.0) | |
| Elliptic sparse | 47 (48.0) | 7 (7.7) | 6 (1.1) | 2 (4.2) | - | 62 (7.4) | |
| Globose dense | 1 (1.0) | - | - | 26 (54.2) | - | 27 (3.2) | |
| Globose sparse | - | - | 1 (0.2) | 17 (35.4) | - | 18 (2.1) | |
| Seed color | Black | - | 1 (1.1) | 2 (0.4) | - | - | 3 (0.4) |
| Brown | - | - | 4 (0.7) | - | - | 4 (0.5) | |
| Dark brown | 3 (3.1) | 1 (1.1) | 8 (1.5) | - | - | 12 (1.4) | |
| Dark green | - | 1 (1.1) | 4 (0.7) | - | - | 5 (0.6) | |
| Dark olive green | 1 (1.0) | - | 39 (7.2) | - | 3 (4.8) | 43 (5.1) | |
| Dark red | 12 (12.2) | 2 (2.2) | 8 (1.5) | 2 (4.2) | - | 24 (2.9) | |
| Light brown | 35 (35.7) | 28 (30.8) | 255 (47.3) | 13 (27.1) | 23 (36.5) | 354 (42.1) | |
| Light red | 9 (9.2) | 7 (7.8) | 31 (5.8) | 5 (10.4) | 2 (3.2) | 55 (6.5) | |
| Olive green | - | 2 (2.2) | 13 (2.4) | - | 4 (6.3) | 19 (2.3) | |
| Straw | 17 (17.3) | 28 (30.8) | 93 (17.3) | 22 (45.8) | 20 (31.7) | 180 (21.4) | |
| White | 21 (21) | 22 (24.2) | 82 (15.2) | 6 (12.5) | 11 (17.5) | 142 (16.9) | |
| Apiculus color | Purple | 24 (24.5) | 19 (20.9) | 249 (46.2) | 5 (10.4) | 12 (19.0) | 309 (36.8) |
| Straw | 74 (75.5) | 73 (80.2) | 290 (53.8) | 43 (89.6) | 51 (81.0) | 531 (63.2) | |
| Trait | Entire | Race | Region | ||||||
|---|---|---|---|---|---|---|---|---|---|
| compactum | contractum | miliaceum | ovatum | patentissimum | Americas | Asia | Europe | ||
| Qualitative traits | |||||||||
| Growth habit | 0.313 | 0.195 | 0.415 | 0.306 | 0.176 | 0.394 | 0.171 | 0.436 | 0.293 |
| Culm branching | 0.455 | 0.432 | 0.472 | 0.441 | 0.336 | 0.467 | 0.374 | 0.436 | 0.460 |
| Sheath pubescence | 0.472 | 0.472 | 0.462 | 0.468 | 0.437 | 0.467 | 0.413 | 0.430 | 0.320 |
| Ligule pubescence | 0.437 | 0.437 | 0.396 | 0.443 | 0.386 | 0.424 | 0.371 | 0.458 | 0.293 |
| Leaf pubescence | 0.426 | 0.426 | 0.381 | 0.437 | 0.366 | 0.376 | 0.171 | 0.454 | 0.272 |
| Inflorescence shape | 0.767 | 0.490 | 0.354 | 0.520 | 0.454 | 0.388 | 0.647 | 0.728 | 0.732 |
| Seed color | 0.716 | 0.709 | 0.685 | 0.698 | 0.582 | 0.637 | 0.508 | 0.737 | 0.830 |
| Apiculus color | 0.286 | 0.242 | 0.221 | 0.300 | 0.145 | 0.212 | 0.171 | 0.294 | 0.252 |
| Mean | 0.484 | 0.425 | 0.423 | 0.451 | 0.360 | 0.420 | 0.353 | 0.497 | 0.431 |
| Quantitative traits | |||||||||
| Days to 50% flowering | 0.585 | 0.568 | 0.588 | 0.572 | 0.315 | 0.484 | 0.544 | 0.594 | 0.478 |
| Plant height (cm) | 0.576 | 0.567 | 0.607 | 0.602 | 0.625 | 0.565 | 0.461 | 0.621 | 0.600 |
| Basal tillers number | 0.593 | 0.643 | 0.603 | 0.604 | 0.566 | 0.562 | 0.276 | 0.540 | 0.579 |
| Flag leaf blade length (mm) | 0.618 | 0.629 | 0.624 | 0.600 | 0.568 | 0.519 | 0.541 | 0.622 | 0.609 |
| Flag leaf blade width (mm) | 0.609 | 0.420 | 0.558 | 0.558 | 0.506 | 0.556 | 0.471 | 0.605 | 0.531 |
| Flag leaf sheath length (mm) | 0.635 | 0.508 | 0.591 | 0.626 | 0.565 | 0.556 | 0.544 | 0.585 | 0.569 |
| Peduncle length (mm) | 0.622 | 0.614 | 0.584 | 0.625 | 0.620 | 0.579 | 0.509 | 0.604 | 0.592 |
| Panicle exsertion (mm) | 0.598 | 0.571 | 0.584 | 0.595 | 0.526 | 0.584 | 0.471 | 0.600 | 0.604 |
| Inflorescence length (mm) | 0.628 | 0.491 | 0.561 | 0.626 | 0.598 | 0.561 | 0.471 | 0.602 | 0.598 |
| Inflorescence primary branches number | 0.649 | 0.598 | 0.604 | 0.649 | 0.601 | 0.586 | 0.403 | 0.631 | 0.651 |
| Mean | 0.611 | 0.561 | 0.590 | 0.606 | 0.549 | 0.555 | 0.469 | 0.600 | 0.581 |
| Trait | Mean $ | Range | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| CP # | CT | ML | OV | PA | Entire set (849 Accessions) | CP | CT | ML | OV | PA | Entire set (849 Accessions) | |
| DF ‡ | 33 c (2.5) † | 34 ab (3.0) | 35 a (3.5) | 33 c (1.8) | 34 ab (4.5) | 34 (3.4) | 29–40 | 28–44 | 26–49 | 30–40 | 30–50 | 26–50 |
| PLHT | 52 b (12.9) | 59 a (1.9) | 62 a (18.1) | 46 c (7.5) | 58 a (16.8) | 59 (17.6) | 25–110 | 25–118 | 23–113 | 25–60 | 20–120 | 20–133 |
| BTN | 3 b (1.2) | 3 b (1.3) | 4 a (1.5) | 4 a (1.2) | 3 b (1.3) | 4 (1.4) | 1–7 | 1–8 | 1–9 | 2–7 | 1–9 | 1–9 |
| FLBL | 212 ab (48.9) | 214 ab (59.1) | 229 a (54.2) | 205 b (33.0) | 205 b (47.4) | 222 (53.5) | 90–320 | 80–380 | 90–370 | 140–280 | 130–340 | 80–380 |
| FLBW | 21 b (5.5) | 19 c (6.1) | 19 c (5.8) | 24 a (5.1) | 16 d (5.2) | 19 (6.0) | 9–30 | 6–30 | 7–30 | 14–30 | 6–30 | 6–30 |
| FLSL | 76 b (16.4) | 81 a (17.6) | 84 a (16.5) | 70 c (11.1) | 82 a (13.6) | 82 (16.6) | 50–170 | 40–140 | 30–140 | 55–100 | 50–120 | 30–170 |
| PEDL | 191 ab (71.8) | 197 ab (67.6) | 175 bc (58.8) | 164 c (49.7) | 206 a (78.4) | 181 (63.6) | 50–370 | 60–400 | 15–380 | 55–270 | 70–400 | 15–400 |
| PANEX | 116 ab (68.8) | 117 ab (59.4) | 91 c (53.6) | 99 bc (54.6) | 124 a (78.0) | 100 (59.8) | 0–270 | 20–300 | 0–280 | 0–300 | 0–320 | 0–320 |
| INFL | 145 b (41.9) | 191 a (51.2) | 209 a (53.5) | 108 c (16.6) | 198 a (50.9) | 193 (58.2) | 80–370 | 110–400 | 22–350 | 70–140 | 120–380 | 22–400 |
| INF–PBN | 16 ab (3.5) | 15 ab (4.8) | 17 a (4.1) | 15 ab (3.6) | 14 b (3.4) | 16 (4.2) | 8–24 | 5–26 | 5–29 | 7–25 | 7–23 | 5–29 |
| Top Ten Pairs of Accessions | Gower’s Phenotypic Distance |
|---|---|
| IPm 370 and IPm 2806 | 0.591 |
| IPm 381 and IPm 2734 | 0.586 |
| IPm 381 and IPm 2747 | 0.581 |
| IPm 460 and IPm 2747 | 0.579 |
| IPm 370 and IPm 2734 | 0.578 |
| IPm 2748 and IPm 381 | 0.578 |
| IPm 381 and IPm 2806 | 0.575 |
| IPm 2748 and IPm 370 | 0.575 |
| IPm 381 and IPm 2723 | 0.574 |
| IPm 362 and IPm 2734 | 0.572 |
| Race | compactum | contractum | miliaceum | ovatum |
|---|---|---|---|---|
| contractum | 0.269 | |||
| miliaceum | 0.264 | 0.266 | ||
| ovatum | 0.229 | 0.274 | 0.275 | |
| patentissimum | 0.266 | 0.254 | 0.267 | 0.274 |
| Principal Component (PC) | PC1 | PC2 | PC3 |
|---|---|---|---|
| Eigenvalue | 3.72 | 2.33 | 1.28 |
| Percent variance | 37.19% | 23.3% | 12.83% |
| Trait | |||
| Days to 50% flowering | 0.06 | −0.41 | 0.50 |
| Plant height (cm) | 0.46 | 0.00 | 0.28 |
| Basal tillers number | 0.15 | −0.23 | −0.26 |
| Flag leaf blade length (mm) | 0.44 | −0.13 | −0.18 |
| Flag leaf blade width (mm) | 0.28 | 0.16 | −0.56 |
| Flag leaf sheath length (mm) | 0.40 | 0.05 | 0.04 |
| Peduncle length (mm) | 0.19 | 0.58 | 0.18 |
| Panicle exsertion (mm) | 0.08 | 0.60 | 0.20 |
| Inflorescence length (mm) | 0.42 | −0.15 | 0.32 |
| Inflorescence primary branches number | 0.34 | −0.13 | −0.27 |
| DF # | PLHT | BTN | FLBL | FLBW | FLSL | PEDL | PANEX | INFL | |
|---|---|---|---|---|---|---|---|---|---|
| PLHT | |||||||||
| Entire | 0.259 ** | ||||||||
| compactum | −0.126 | ||||||||
| contractum | 0.008 | ||||||||
| miliaceum | 0.296 ** | ||||||||
| ovatum | −0.566 ** | ||||||||
| patentissimum | 0.187 | ||||||||
| BTN | |||||||||
| Entire | 0.092 ** | 0.183 ** | |||||||
| compactum | 0.065 | 0.200 * | |||||||
| contractum | 0.006 | 0.121 | |||||||
| miliaceum | 0.078 | 0.214 ** | |||||||
| ovatum | 0.094 | −0.024 | |||||||
| patentissimum | 0.211 | −0.02 | |||||||
| FLBL | |||||||||
| Entire | 0.117 ** | 0.656 ** | 0.338 ** | ||||||
| compactum | 0.101 | 0.479 ** | 0.358 ** | ||||||
| contractum | −0.001 | 0.634 ** | 0.371 ** | ||||||
| miliaceum | 0.074 | 0.686 ** | 0.341 ** | ||||||
| ovatum | −0.104 | 0.078 | 0.286 * | ||||||
| patentissimum | 0.377 ** | 0.693 ** | 0.135 | ||||||
| FLBW | |||||||||
| Entire | −0.265 ** | 0.267 ** | 0.118 ** | 0.524 ** | |||||
| compactum | −0.136 | 0.446 ** | 0.289 ** | 0.593 ** | |||||
| contractum | −0.164 | 0.515 ** | 0.287 ** | 0.709 ** | |||||
| miliaceum | −0.273 ** | 0.295 ** | 0.088 * | 0.556 ** | |||||
| ovatum | −0.383 ** | 0.350 * | 0.353 * | 0.539 ** | |||||
| patentissimum | −0.147 | 0.514 ** | −0.347 ** | 0.373 ** | |||||
| FLSL | |||||||||
| Entire | 0.037 | 0.597 ** | 0.101 ** | 0.551 ** | 0.410 ** | ||||
| compactum | −0.171 | 0.417 ** | 0.051 | 0.423 ** | 0.227 * | ||||
| contractum | 0.023 | 0.728 ** | 0.295 ** | 0.637 ** | 0.530 ** | ||||
| miliaceum | −0.006 | 0.574 ** | 0.079 | 0.547 ** | 0.544 ** | ||||
| ovatum | −0.146 | 0.249 | 0.205 | 0.453 ** | 0.332 * | ||||
| patentissimum | 0.107 | 0.601 ** | −0.082 | 0.548 ** | 0.444 ** | ||||
| PEDL | |||||||||
| Entire | −0.354 ** | 0.380 ** | −0.149 ** | 0.089 ** | 0.264 ** | 0.341 ** | |||
| compactum | −0.561 ** | 0.592 ** | −0.105 | −0.011 | 0.279 ** | 0.219 * | |||
| contractum | −0.349 ** | 0.519 ** | 0.044 | 0.270 ** | 0.314 ** | 0.448 ** | |||
| miliaceum | −0.268 ** | 0.381 ** | −0.137 ** | 0.122 ** | 0.297 ** | 0.405 ** | |||
| ovatum | −0.524 ** | 0.825 ** | −0.285 * | 0.028 | 0.076 | 0.277 | |||
| patentissimum | −0.666 ** | 0.297 * | −0.241 | 0.019 | 0.447 ** | 0.214 | |||
| PANEX | |||||||||
| Entire | −0.368 ** | 0.225 ** | −0.193 ** | −0.071 * | 0.153 ** | 0.087 * | 0.937 ** | ||
| compactum | −0.552 ** | 0.500 ** | −0.138 | −0.104 | 0.234 * | 0.075 | 0.983 ** | ||
| contractum | −0.394 ** | 0.346 ** | −0.064 | 0.059 | 0.154 | 0.184 | 0.941 ** | ||
| miliaceum | −0.285 ** | 0.238 ** | −0.175 ** | −0.043 | 0.152 ** | 0.141 ** | 0.951 ** | ||
| ovatum | 0.006 | 0.476 ** | −0.284 * | −0.142 | −0.178 | −0.101 | 0.672 ** | ||
| patentissimum | −0.644 ** | 0.219 | −0.241 | −0.047 | 0.357 ** | 0.044 | 0.920 ** | ||
| INFL | |||||||||
| Entire | 0.327 ** | 0.800 ** | 0.209 ** | 0.638 ** | 0.105 ** | 0.602 ** | 0.148 ** | −0.021 | |
| compactum | 0.086 | 0.709 ** | 0.270 ** | 0.568 ** | 0.264 ** | 0.500 ** | 0.143 | 0.025 | |
| contractum | 0.19 | 0.746 ** | 0.244 * | 0.686 ** | 0.429 ** | 0.712 ** | 0.14 | −0.076 | |
| miliaceum | 0.276 ** | 0.838 ** | 0.237 ** | 0.696 ** | 0.252 ** | 0.582 ** | 0.244 ** | 0.086 * | |
| ovatum | 0.192 | 0.257 | −0.247 | 0.003 | −0.166 | 0.014 | 0.313 * | 0.309 * | |
| patentissimum | 0.442 ** | 0.654 ** | 0.157 | 0.710 ** | 0.189 | 0.542 ** | −0.113 | −0.192 | |
| INF_PBN | |||||||||
| Entire | 0.022 | 0.496 ** | 0.185 ** | 0.554 ** | 0.390 ** | 0.350 ** | 0.003 | −0.115 ** | 0.445 ** |
| compactum | 0.235 * | 0.002 | 0.133 | 0.218 * | 0.077 | −0.022 | −0.399 ** | −0.431 ** | 0.264 ** |
| contractum | −0.047 | 0.588 ** | 0.192 | 0.689 ** | 0.566 ** | 0.456 ** | 0.114 | −0.066 | 0.544 ** |
| miliaceum | −0.035 | 0.534 ** | 0.210 ** | 0.578 ** | 0.440 ** | 0.381 ** | 0.092* | −0.022 | 0.486 * |
| ovatum | −0.061 | −0.008 | −0.031 | 0.25 | 0.342 * | 0.157 | −0.08 | −0.231 | 0.219 |
| patentissimum | 0.104 | 0.601 ** | −0.073 | 0.427 ** | 0.471 ** | 0.297 * | 0.062 | −0.003 | 0.384 ** |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vetriventhan, M.; Azevedo, V.C.R.; Upadhyaya, H.D.; Naresh, D. Variability in the Global Proso Millet (Panicum miliaceum L.) Germplasm Collection Conserved at the ICRISAT Genebank. Agriculture 2019, 9, 112. https://doi.org/10.3390/agriculture9050112
Vetriventhan M, Azevedo VCR, Upadhyaya HD, Naresh D. Variability in the Global Proso Millet (Panicum miliaceum L.) Germplasm Collection Conserved at the ICRISAT Genebank. Agriculture. 2019; 9(5):112. https://doi.org/10.3390/agriculture9050112
Chicago/Turabian StyleVetriventhan, Mani, Vania C.R. Azevedo, Hari D. Upadhyaya, and D. Naresh. 2019. "Variability in the Global Proso Millet (Panicum miliaceum L.) Germplasm Collection Conserved at the ICRISAT Genebank" Agriculture 9, no. 5: 112. https://doi.org/10.3390/agriculture9050112
APA StyleVetriventhan, M., Azevedo, V. C. R., Upadhyaya, H. D., & Naresh, D. (2019). Variability in the Global Proso Millet (Panicum miliaceum L.) Germplasm Collection Conserved at the ICRISAT Genebank. Agriculture, 9(5), 112. https://doi.org/10.3390/agriculture9050112






