Changes in the Nitrogen Budget and Soil Nitrogen in a Field with Paddy–Upland Rotation with Different Histories of Manure Application
Abstract
:1. Introduction
2. Materials and Methods
2.1. Experimental Field
2.2. History of Compost Application
2.3. Plant Cultivation
2.4. Soil Sampling and Analysis
2.5. Plant Growth, Nitrogen Accumulation, and Yield
2.5.1. Soybean
2.5.2. Rice
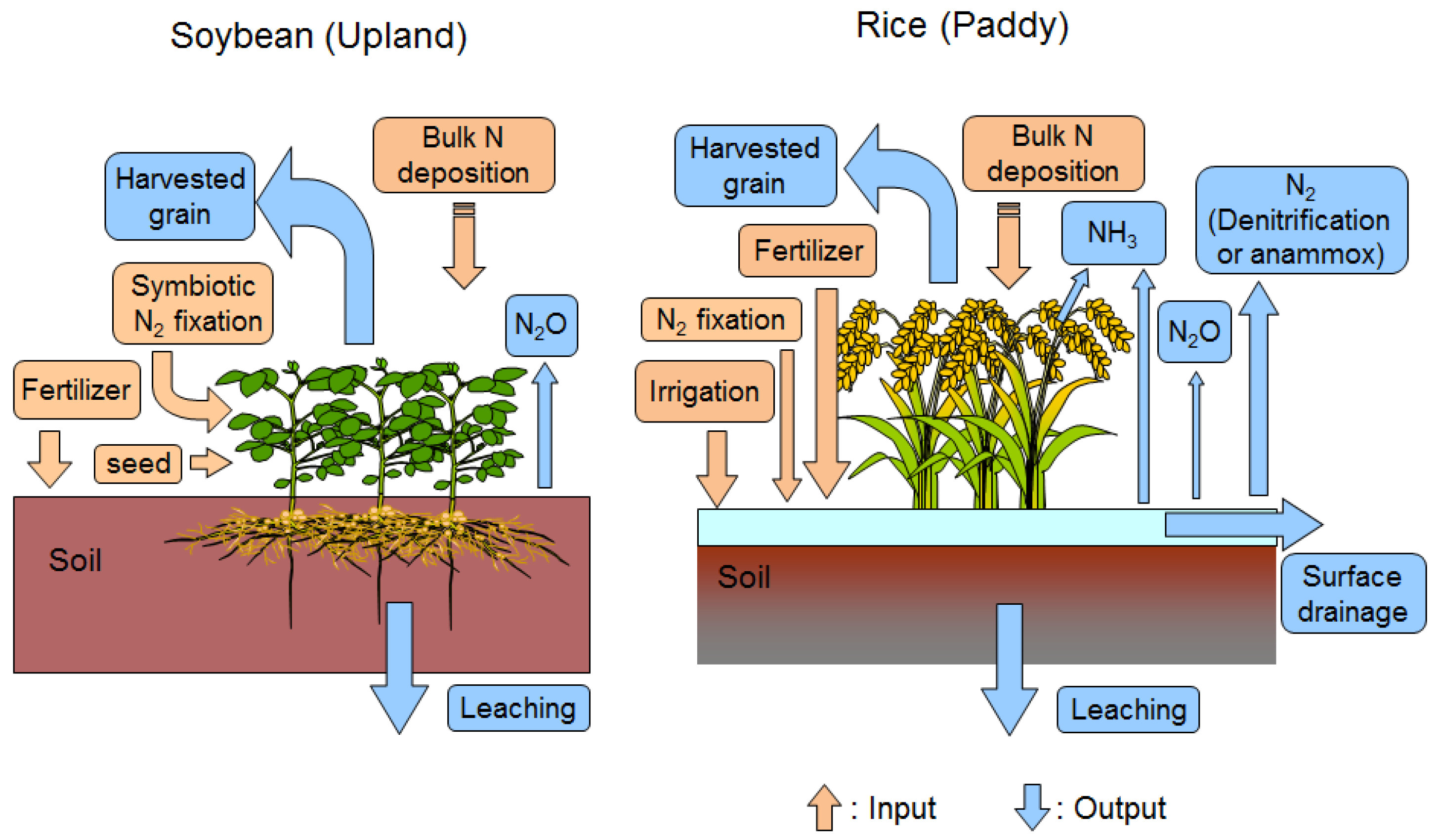
2.6. Measurement of Other Nitrogen Flows and Calculation of Nitrogen Budget
2.6.1. Seeds and Seedlings
2.6.2. Bulk Nitrogen Deposition
2.6.3. Irrigation
2.6.4. Nitrogen Leaching and Surface Drainage
2.6.5. N2O Emission
2.6.6. Other Flows
2.7. Statistical Analyses
3. Results
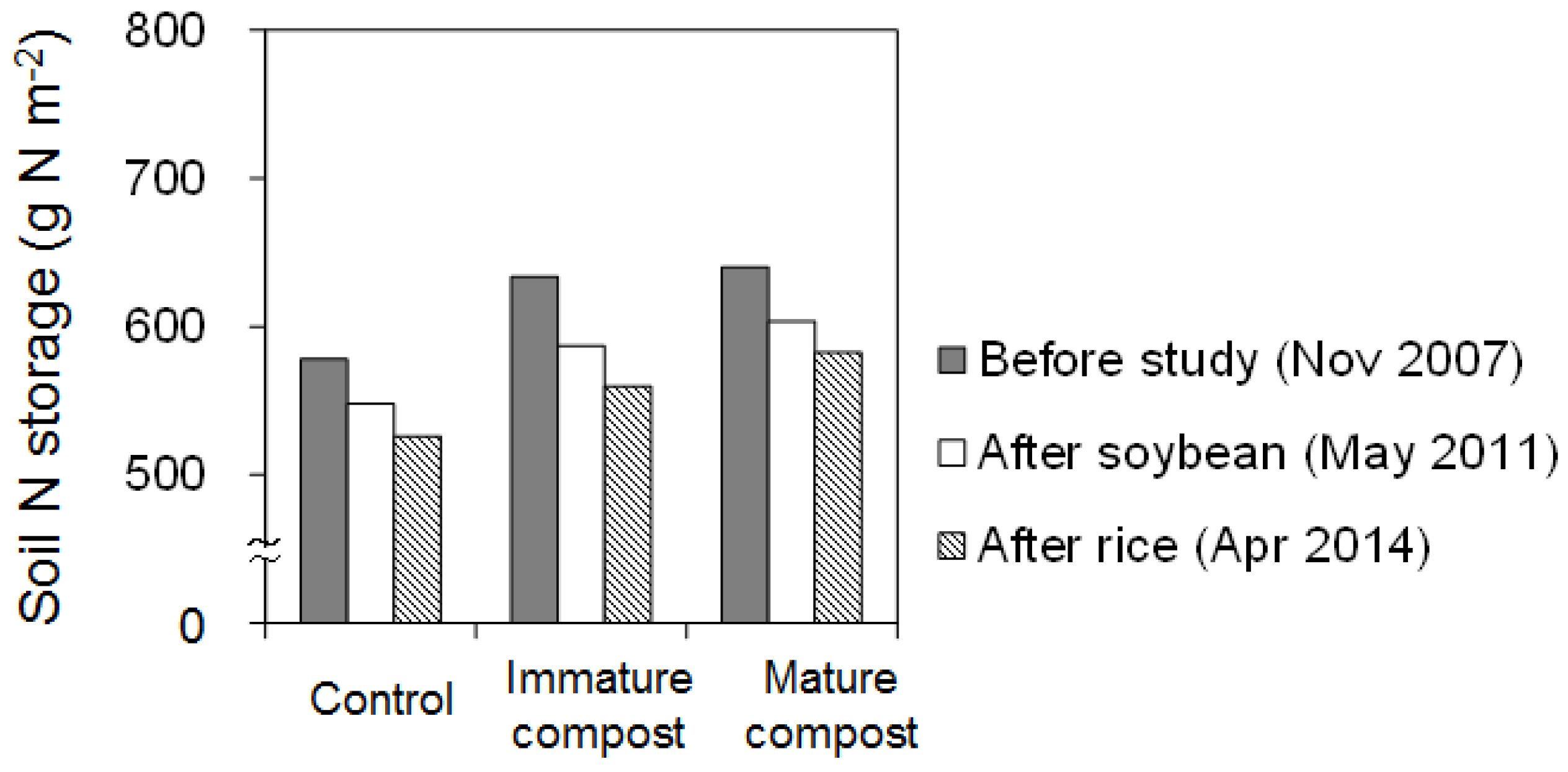
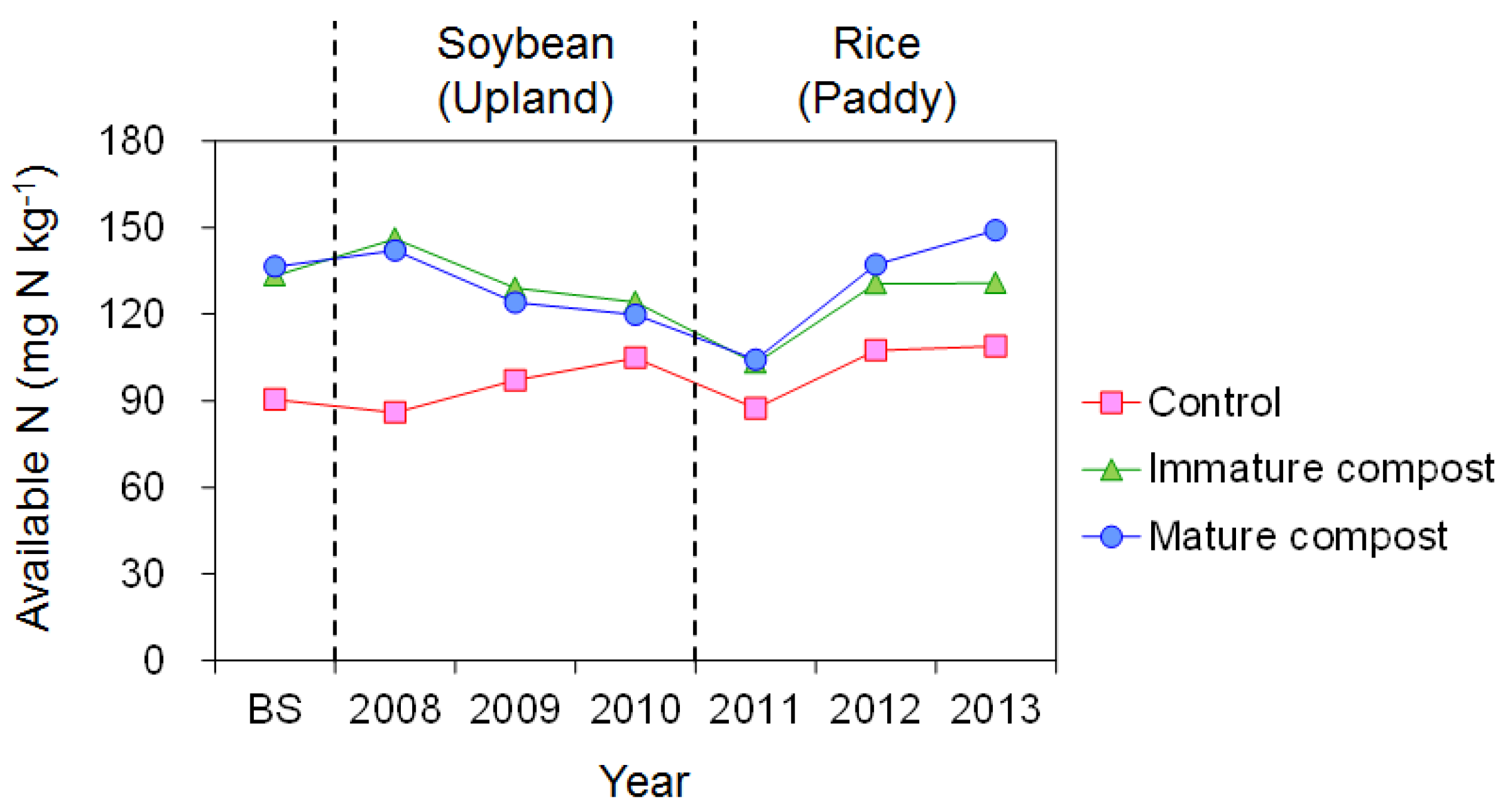
3.1. Changes in Soil Nitrogen
3.2. Plant Growth, Nitrogen Accumulation, and Yield
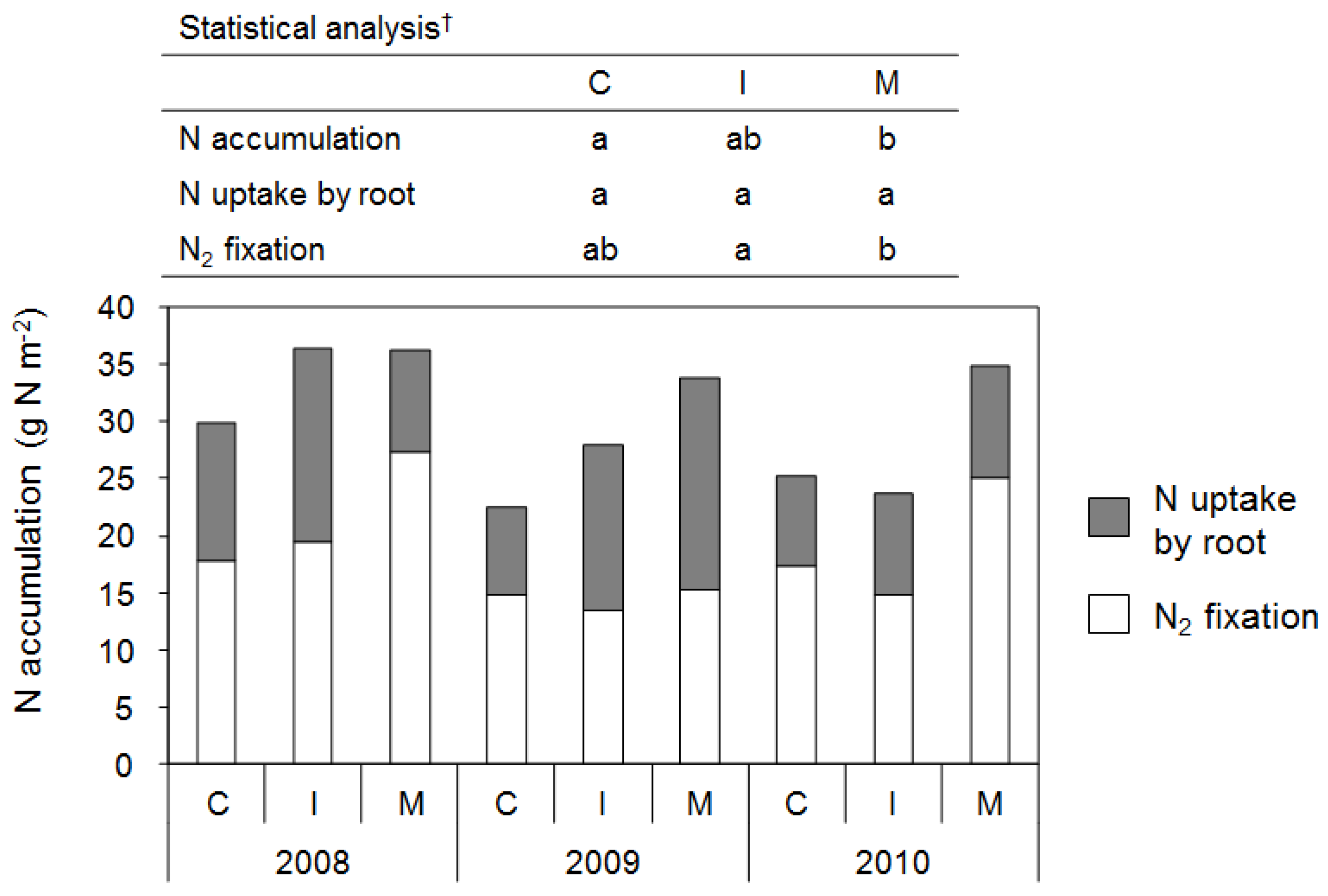
3.2.1. Soybean
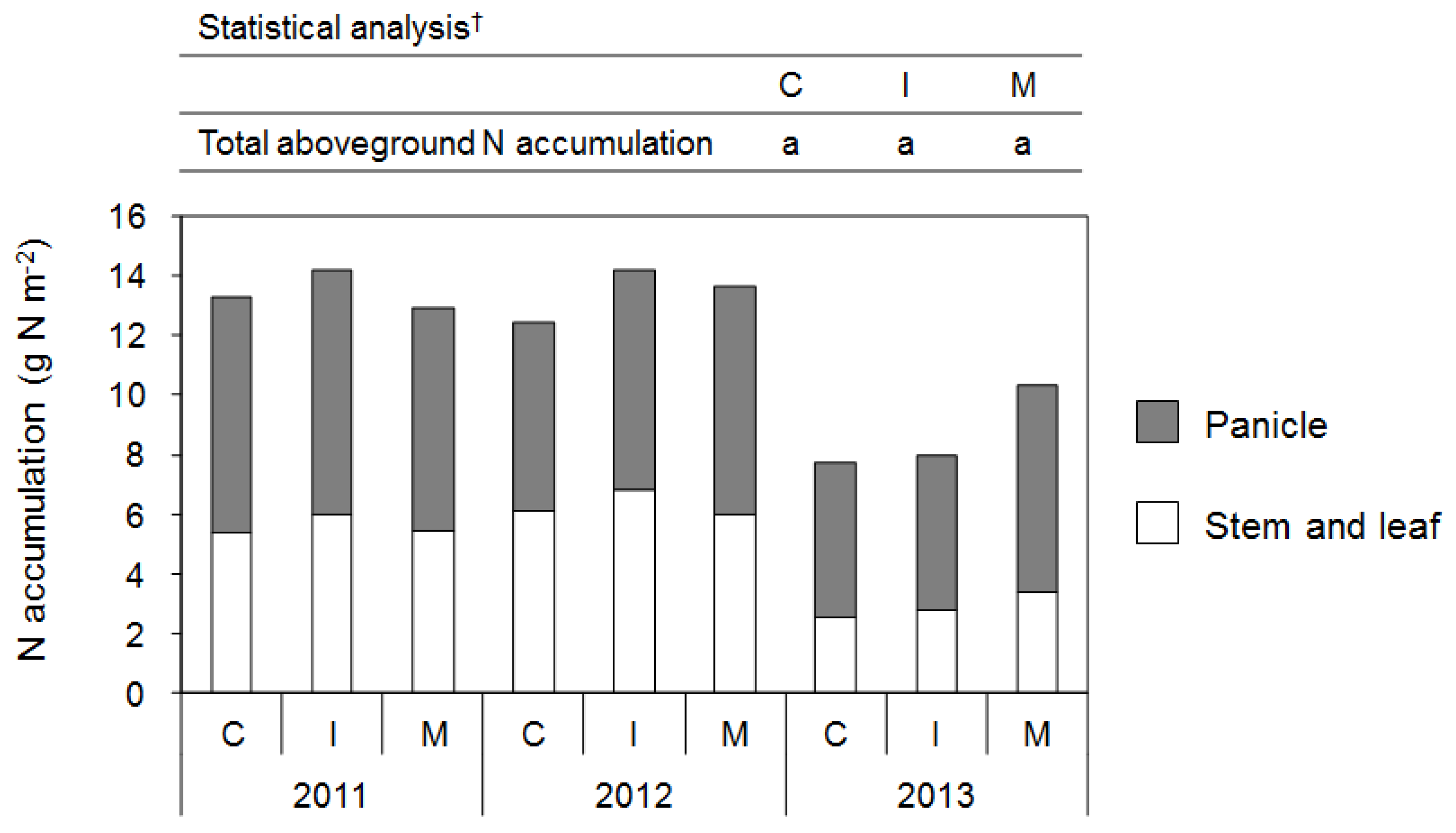
3.2.2. Rice
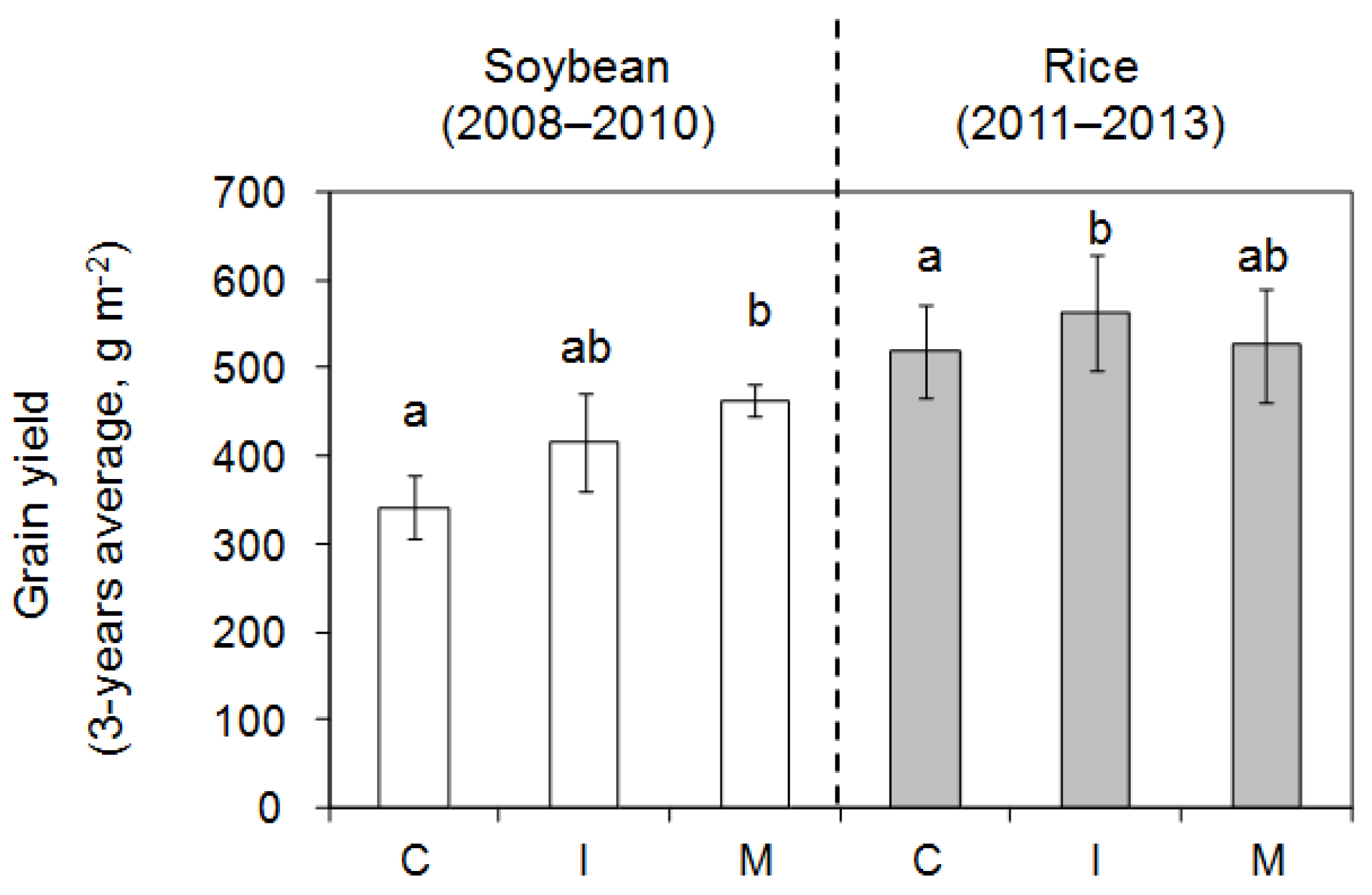
3.2.3. Yield
3.3. Bulk Nitrogen Deposition, Irrigation, Surface Drainage, Leaching, and N2O Emission
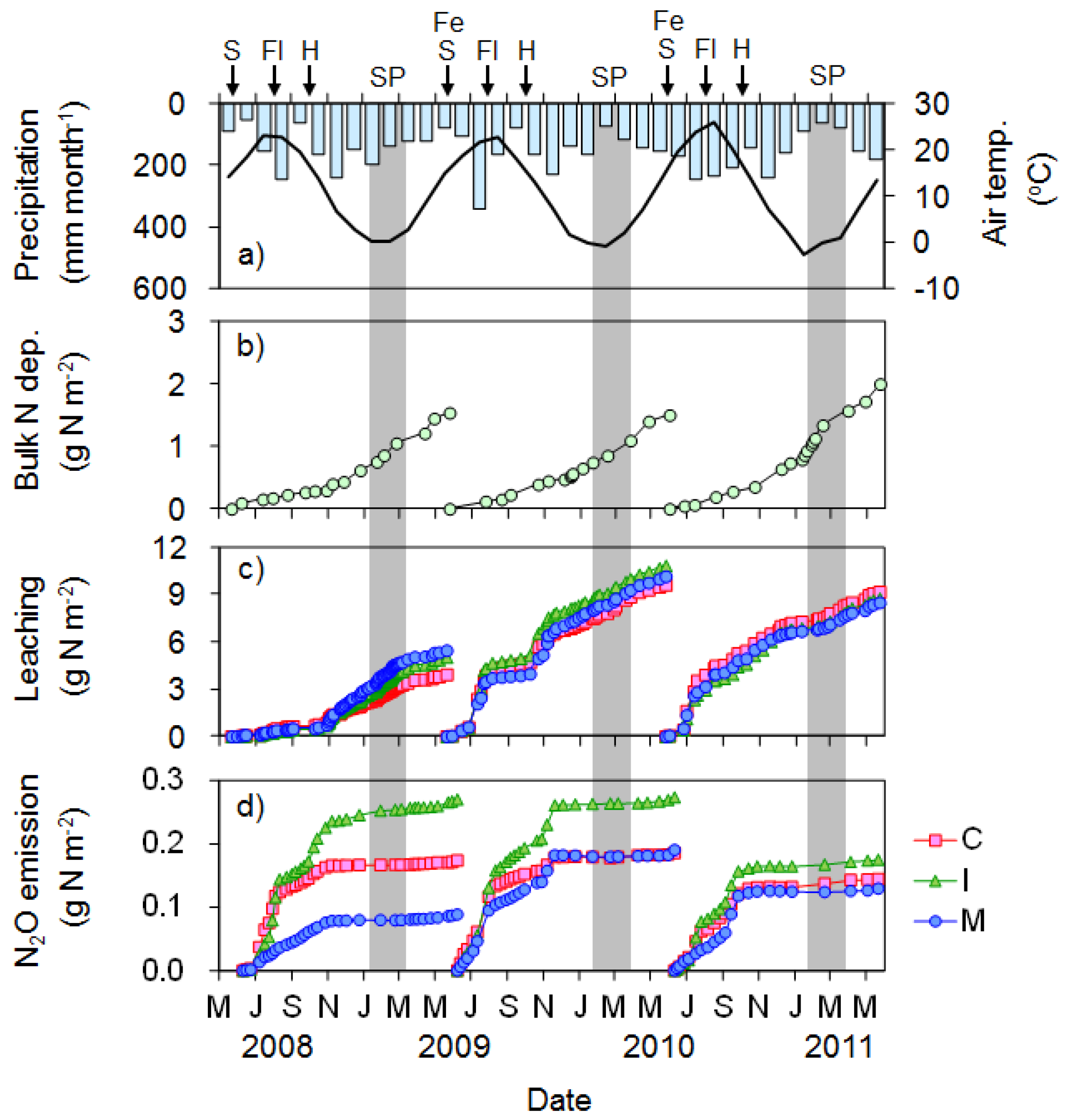
3.3.1. Soybean Cultivation Period
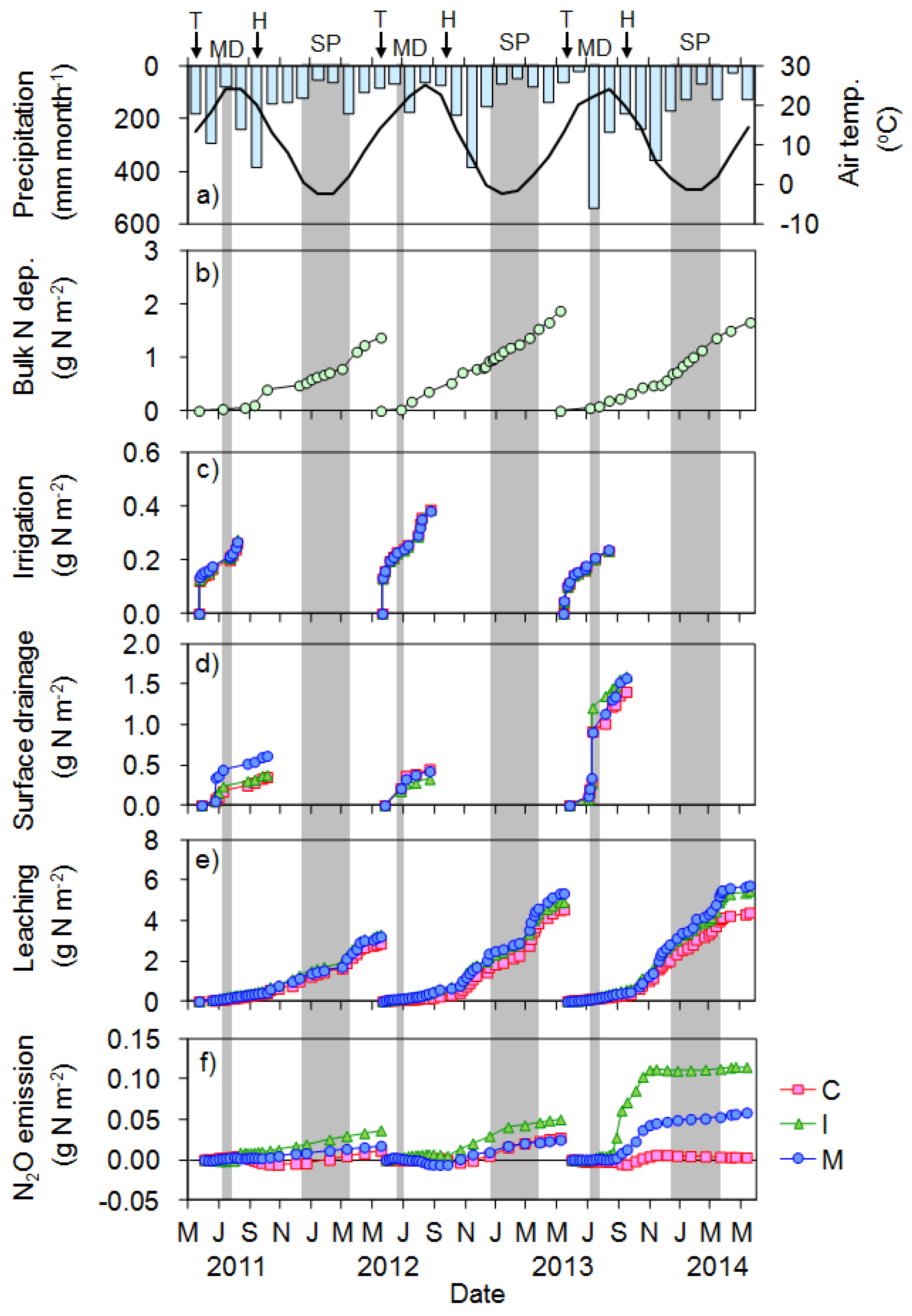
3.3.2. Rice Cultivation Period
3.4. Estimation of NH3 Volatilization and N2 Emission via Denitrification
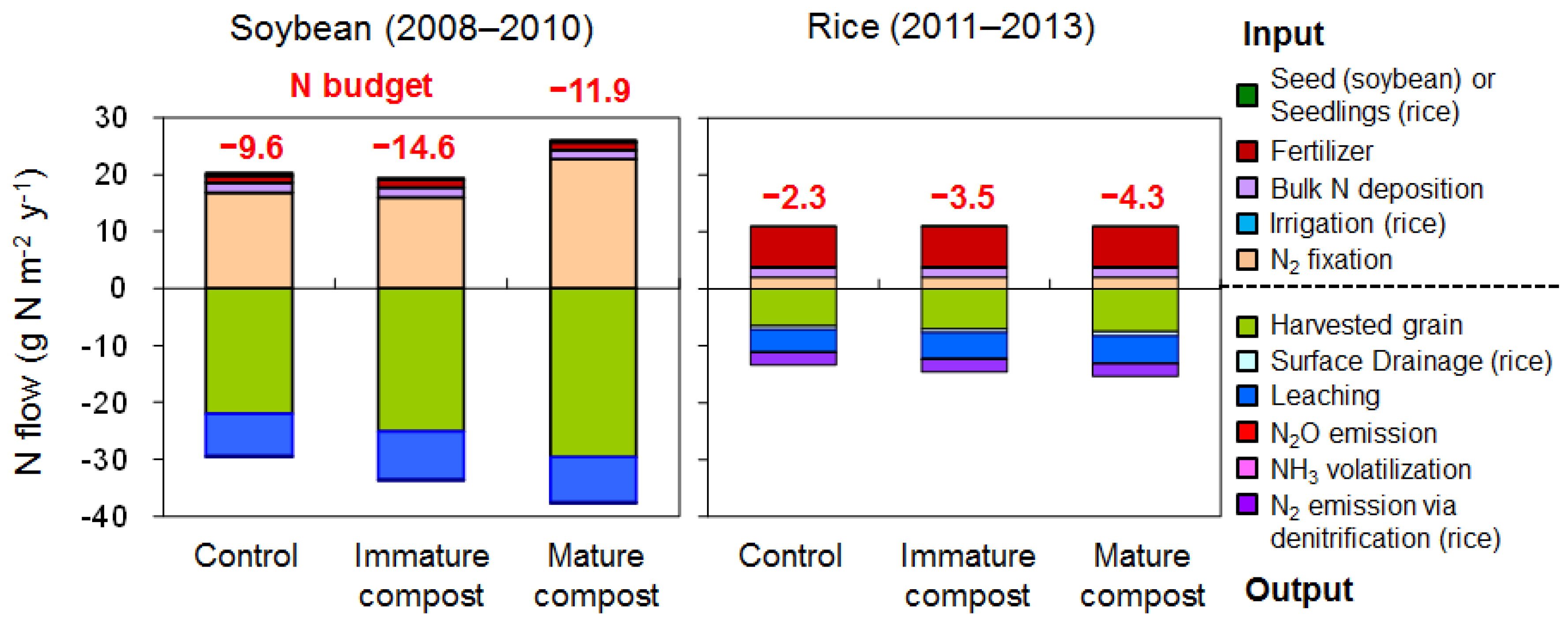
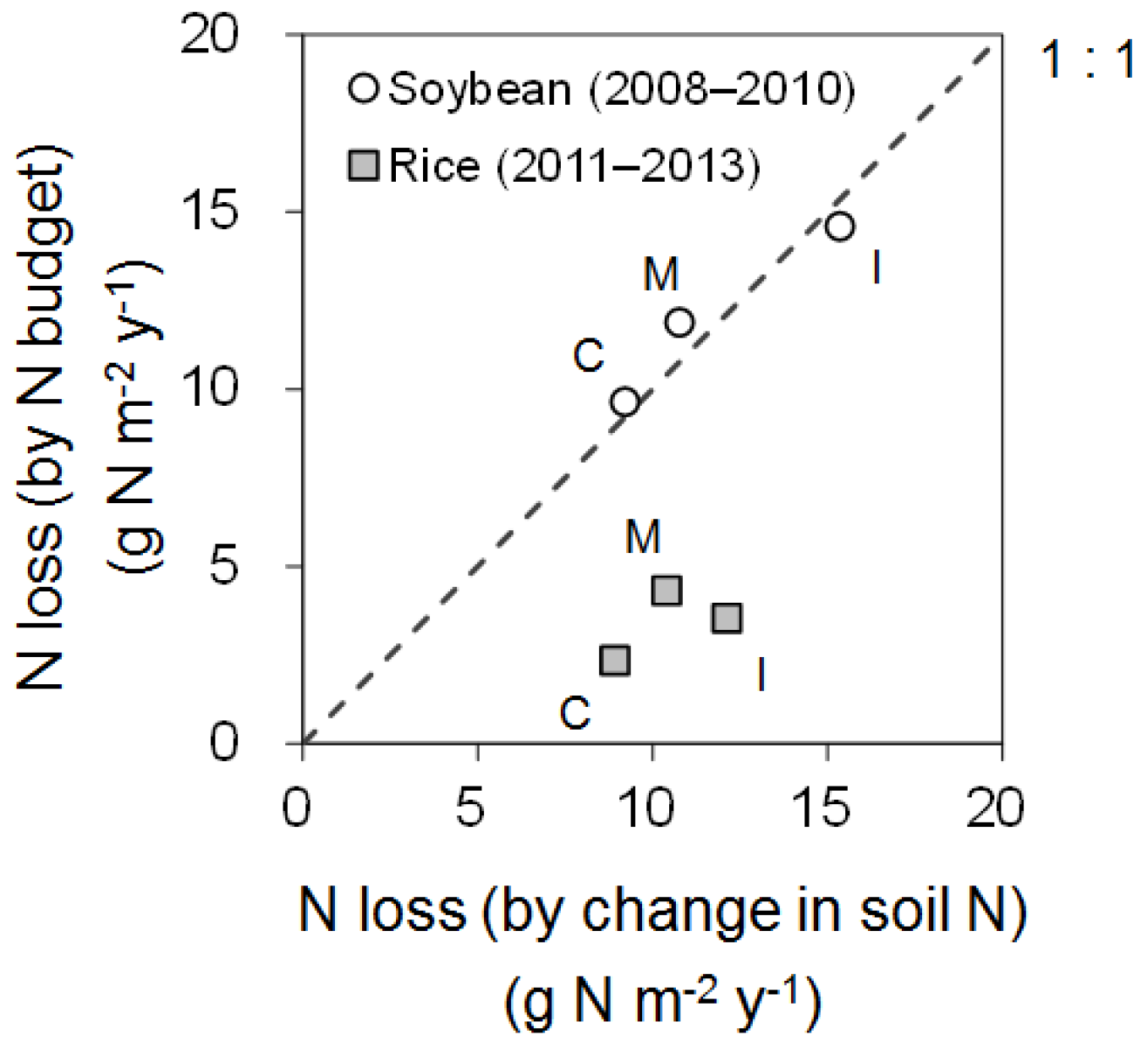
3.5. Nitrogen Budget
4. Discussion
4.1. Soil Nitrogen Fertility
4.2. Plant Growth, Nitrogen Accumulation, and Yield
4.3. Nitrogen Flows
4.4. Nitrogen Budget
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Takahashi, T.; Sumida, H.; Nira, R. A new framework for study of irrigated paddy rice and upland crops rotation farming and its relation to soil and plant nutrition science. 1. Advances and perspectives in irrigated paddy rice and upland crops rotation farming. Jpn. J. Soil Sci. Plant Nutr. 2013, 84, 202–207. (In Japanese) [Google Scholar]
- Sumida, H.; Kato, N.; Nishida, M. Depletion of soil fertility and crop productivity in succession of paddy rice-soybean rotation. Bull. Natl. Agric. Res. Cent. Tohoku Reg. 2005, 103, 39–52, (In Japanese with English summary). [Google Scholar]
- Hirokawa, T.; Inahara, M.; Koike, J. Declines in soil nitrogen fertility after rotation of medium and coarse-textured gray lowland soil and restoration methods with green manure and cattle manure compost. Toyama Pref. Agr. For. Fish. Res. Ctr. 2011, 2, 11–26, (In Japanese with English summary). [Google Scholar]
- Odahara, K.; Fukushima, Y.; Araki, M.; Kaneko, A.; Aramaki, K. The soil fertility status and soybean productivity in paddy-upland rotation fields in Japan’s Chikugo River basin. Jpn. J. Soil Sci. Plant Nutr. 2012, 83, 405–411, (In Japanese with English summary). [Google Scholar]
- Nishida, M.; Sekiya, H.; Yoshida, K. Status of paddy soils as affected by paddy rice and upland soybean rotation in northeast Japan, with special reference to nitrogen fertility. Soil Sci. Plant Nutr. 2013, 59, 208–217. [Google Scholar] [CrossRef]
- Hoshi, S. Nitrogen Fixation, Growth and Yield of Soybean. In Nitrogen Fixation in Root Nodules; Japanese Society of Soil Science and Plant Nutrition, Ed.; Hakuyu-sha: Tokyo, Japan, 1982; pp. 5–33. (In Japanese) [Google Scholar]
- Arihara, J. Innovative Technology for Stable High-Yield Soybean Production; Rural Culture Association: Tokyo, Japan, 2000; pp. 33–59. (In Japanese) [Google Scholar]
- Takakai, F.; Takeda, M.; Kon, K.; Inoue, K.; Nakagawa, S.; Sasaki, K.; Chida, A.; Sekiguchi, K.; Takahashi, T.; Sato, T.; et al. Effects of preceding compost application on the nitrogen budget in an upland soybean field converted from a rice paddy field on gray lowland soil in Akita, Japan. Soil Sci. Plant Nutr. 2010, 56, 760–772. [Google Scholar] [CrossRef]
- Hasukawa, H.; Takahashi, Y.; Toritsuka, S.; Kawamura, H.; Yamada, Y. Field–scale environmental impact assessment and investigation of nitrogen balance for immediate soybean cultivation after construction of new soil water control system. Jpn. J. Soil Sci. Plant Nutr. 2014, 85, 509–514. (In Japanese) [Google Scholar]
- Nishida, M. Changes in Nitrogen Fertility of Soils in Paddy-Upland Rotation Fields and Strategies for Its Fertility Management. In Fertility and Management of Soil in Paddy-Upland Rotation Fields in Japan-Factors of Fertility Change and Approaches for Its Control; Japanese Society of Soil Science and Plant Nutrition, Ed.; Hakuyu-sha: Tokyo, Japan, 2010; pp. 27–52. (In Japanese) [Google Scholar]
- Katayanagi, N.; Ono, K.; Fumoto, T.; Mano, M.; Miyata, A.; Hayashi, K. Validation of the DNDC-Rice model to discover problems in evaluating the nitrogen balance at a paddy-field scale for single-cropping of rice. Nutr. Cycl. Agroecosyst. 2013, 95, 255–268. [Google Scholar] [CrossRef]
- Koyama, T.; App, A. Nitrogen Balance in Flooded Rice Soils. In Nitrogen and Rice; Rockwood, W.G., Ed.; International Rice Research Institute: Los Baños, The Philippines, 1979; pp. 95–104. [Google Scholar]
- Kitada, K.; Shimoda, H.; Kamekawa, K.; Akiyama, Y. Changes in soil nutrients affected by rotation of upland and paddy crop in gray lowland soil and search for the most suitable term of rotation. Jpn. J. Soil Sci. Plant Nutr. 1993, 64, 154–160, (In Japanese with English summary). [Google Scholar]
- Department of Agriculture, Forestry and Fisheries, Akita Prefecture. Guidelines for Soybean Cultivation; Department of Agriculture, Forestry and Fisheries, Akita Prefecture: Akita, Japan, 2015; pp. 26–29. (In Japanese)
- Department of Agriculture, Forestry and Fisheries, Akita Prefecture. Guidelines for Rice Cultivation; Department of Agriculture, Forestry and Fisheries, Akita Prefecture: Akita, Japan, 2014; pp. 43–51. (In Japanese)
- Paustian, K.; Andrén, O.; Janzen, H.; Lal, R.; Smith, P.; Tian, G.; Tiessen, H.; van Noordwijk, M.; Woomer, P. Agricultural soil as a C sink to offset CO2 emissions. Soil Use Manag. 1997, 13, 230–244. [Google Scholar] [CrossRef]
- Koga, N.; Sawamoto, T.; Tsuruta, H. Life cycle inventory-based analysis of greenhouse gas emissions from arable land farming systems in Hokkaido, northern Japan. Soil Sci. Plant Nutr. 2006, 52, 564–574. [Google Scholar] [CrossRef]
- Ogawa, Y. Available Nitrogen. In Analysis Methods of Soil Environment; Konno, T., Ed.; Hakuyu-sha: Tokyo, Japan, 1997; pp. 255–262. (In Japanese) [Google Scholar]
- Takahashi, Y.; Chinushi, T.; Nakano, T.; Ohyama, T. Evaluation of N2 fixation and N absorption activity by relative ureide method in field-grown soybean plants with deep placement of coated urea. Soil Sci. Plant Nutr. 1992, 38, 699–708. [Google Scholar] [CrossRef]
- Takahashi, Y.; Chinushi, T.; Ohyama, T. Quantitative estimation of N2 fixation activity and N absorption rate in field grown soybean plants by relative ureide method. Bull. Fac. Agric. Niigata Univ. 1993, 45, 91–105. [Google Scholar]
- Fehr, W.R.; Canness, C.E.; Burmood, D.T.; Pennington, J.S. Stage development description for soybean Glycine max (L.) Merill. Crop Sci. 1971, 11, 929–931. [Google Scholar] [CrossRef]
- Sato, T.; Yashima, H.; Ohtake, N.; Sueyoshi, K.; Akao, S.; Harper, J.E.; Ohyama, T. Determination of Leghemoglobin components and xylem sap composition by capillary electrophoresis in hypernodulation soybean mutants cultivated in the field. Soil Sci. Plant Nutr. 1998, 44, 635–645. [Google Scholar] [CrossRef]
- Sato, T.; Kaneta, Y.; Furuta, N.; Kobayashi, H.; Shindo, H.; Ota, T.; Sato, A. Effect of soil properties on soybean nodulation and N2 fixation at the early growth stage in heavy soil field in Hachirougata polder, Japan. Soil Sci. Plant Nutr. 2003, 49, 695–702. [Google Scholar] [CrossRef]
- Takakai, F.; Nakagawa, S.; Kon, K.; Sato, T.; Kaneta, Y. Net greenhouse gas budget and soil carbon storage in a field with paddy-upland rotation with different history of preceding manure application. Agriculture 2017. under review. [Google Scholar]
- Sato, N. Effects of Percolation through the Surface Soil Close to the Border on Rice Growth and Yield. Jpn. J. Soil Sci. Plant Nutr. 2001, 72, 104–107. (In Japanese) [Google Scholar]
- Toma, Y.; Hatano, R. Effect of crop residue C:N ratio on N2O emissions from Gray Lowland soil in Mikasa, Hokkaido, Japan. Soil Sci. Plant Nutr. 2007, 53, 198–205. [Google Scholar] [CrossRef]
- Takakai, F.; Ichikawa, J.; Ogawa, M.; Ogaya, S.; Yasuda, K.; Kobayashi, Y.; Sato, T.; Kaneta, Y.; Nagahama, K. Suppression of CH4 emission by rice straw removal and application of bio-ethanol production residue in a paddy field in Akita, Japan. Agriculture 2017, 7, 21. [Google Scholar] [CrossRef]
- Yasuda, M.; Okada, T.; Nozoe, T. Characterristics of nitorgen enrichment by biological fixation (BNF) on different management of paddy soils in the Tohoku distinct of Japan. Jpn. J. Soil Sci. Plant Nutr. 2000, 71, 849–856, (In Japanese with English summary). [Google Scholar]
- Ito., J.; Iimura, K. Direct measurement of N2 gas denitrified from soil and fertilizer nitrogen in paddy field using GC-MS. Jpn. J. Soil Sci. Plant Nutr. 1983, 54, 235–240. (In Japanese) [Google Scholar]
- Hayashi, K.; Nishimura, S.; Yagi, K. Ammonia volatilization from the surface of a Japanese paddy field during rice cultivation. Soil Sci. Plant Nutr. 2006, 52, 545–555. [Google Scholar] [CrossRef]
- Nie, S.-A.; Li, H.; Yang, X.; Zhang, Z.; Weng, B.; Huang, F.; Zhu, G.-B.; Zhu, Y.-G. Nitrogen loss by anaerobic oxidation of ammonium in rice rhizosphere. IMSE J. 2015, 9, 2059–2067. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.-R.; Li, H.; Nie, S.-A.; Su, J.-Q.; Weng, B.-S.; Zhu, G.-B.; Yao, H.-Y.; Gilbert, J.A.; Zhu, Y.-G. Potential contribution of anammox to nitrogen loss from paddy soils in southern China. Appl. Environ. Microbiol. 2015, 81, 938–947. [Google Scholar] [CrossRef] [PubMed]
- Zhou, S.; Borjigin, S.; Riya, S.; Hosomi, M. Denitrification-dependent anammox activity in a permanently flooded fallow ravine paddy field. Ecol. Eng. 2016, 95, 452–456. [Google Scholar] [CrossRef]
- Mitsuchi, M. Characters of humus formed under rice cultivation. Soil Sci. Plant Nutr. 1974, 20, 249–259. [Google Scholar] [CrossRef]
- Wakatsuki, T. Paddy Soils. In Advanced Soil Science; Kyuma, K., Ed.; Asakura Shoten: Tokyo, Japan, 1997; pp. 157–178. (In Japanese) [Google Scholar]
- Wichern, F.; Eberhardt, E.; Mayer, J.; Jorgensen, R.G.; Muller, T. Nitrogen rhizodeposition in agricultural crops: Methods, estimates and future prospects. Soil Biol. Biochem. 2008, 40, 30–48. [Google Scholar] [CrossRef]
- Kaneta, Y.; Kodama, T.; Naganoma, H. Effect of paddy-upland rotation management on the productivity of rice in Hachiroguta reclaimed fields. Part 1: Characteristics of the rice plant’s nitrogen-uptake patterns in rotational paddy fields. Jpn. J. Soil Sci. Plant Nutr. 1989, 60, 127–133, (In Japanese with English summary). [Google Scholar]
- Kaneta, Y.; Kodama, T.; Naganoma, H. Effect of paddy-upland rotation management on the productivity of rice in Hachirogata reclaimed fields. Part 2: Evaluation and estimation of soil nitrogen mineralization and nitrogen uptake by rice plants from subsoil of paddy fields rotated from upland fields. Jpn. J. Soil Sci. Plant Nutr. 1989, 60, 399–405, (In Japanese with English summary). [Google Scholar]
- Toriyama, K. Prospects for Precision Agriculture in Paddy Field by Site-Specific Nitrogen Management. In Evaluation and Management of Soil Ferlity towards Sustainable Food Production; Kosaki, T., Watanabe, K., Yanai, J., Eds.; Hakuyusha: Tokyo, Japan, 2008; pp. 64–92. (In Japanese) [Google Scholar]
- Vanotti, M.B.; Bundy, L.G. Soybean effects on soil nitrogen availability in crop rotations. Agron. J. 1995, 87, 676–680. [Google Scholar] [CrossRef]
- Takahashi, T.; Matsuzaki, N.; Shioya, Y.; Hosokawa, H. Influence of soil properties on the yield of soybean in upland fields converted from rice paddies—A case study in Joetsu region, Niigata, Japan. Bull. Natl. Agric. Res. Cent. 2005, 6, 51–58, (In Japanese with English summary). [Google Scholar]
- Shimada, S.; Kokubun, M.; Matsui, S. Effects of water table on physiological traits and yield of soybean. I. Effects of water table and rain fall on leaf chlorophyll content, root growth and yield. Jpn. J. Crop Sci. 1995, 64, 294–303. [Google Scholar] [CrossRef]
- Streeter, J.G. Nitrate inhibition of legume nodule growth and activity, I. Long term studies with a continuous supply of nitrate. Plant Physiol. 1985, 77, 321–324. [Google Scholar] [CrossRef] [PubMed]
- Streeter, J.G. Nitrate inhibition of legume nodule growth and activity, II. Short term studies with high nitrate supply. Plant Physiol. 1985, 77, 325–328. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, S. Effect of farmyard manure on the nitrogen nutrition of soybean. Jpn. J. Crop Sci. 1979, 48, 17–24, (In Japanese with English summary). [Google Scholar] [CrossRef]
- Israel, D.W. Investigation of the role of phosphorus in symbiotic dinitrogen fixation. Plant Physiol. 1987, 84, 835–840. [Google Scholar] [CrossRef] [PubMed]
- Israel, D.W. Symbiotic dinitrogen fixation and host-plant growth during development of and recovery from phosphorus deficiency. Physiol. Plant. 1993, 88, 294–300. [Google Scholar] [CrossRef]
- Qiao, Y.; Tang, C.; Han, X.; Miao, S. Phosphorus deficiency delays the onset of nodule function in soybean. J. Plant Nutr. 2007, 30, 1341–1353. [Google Scholar] [CrossRef]
- Sa, T.M.; Israel, D.W. Energy status and functioning of phosphorus-deficient soybean nodules. Plant Physiol. 1991, 97, 928–935. [Google Scholar] [CrossRef] [PubMed]
- Oh-e, I.; Uwagoh, R.; Jyo, S.; Kurahashi, T.; Saitoh, K.; Kuroda, T. Effect of rising temperature on flowering, pod set, dry-matter production and seed yield in soybean. Jpn. J. Crop Sci. 2007, 76, 433–444, (In Japanese with English summary). [Google Scholar] [CrossRef]
- Moroyu, H. Changes of physical and chemical properties of paddy soils under the cultivation of upland crops. Jpn. J. Soil Sci. Plant Nutr. 1983, 54, 434–441. (In Japanese) [Google Scholar]
- Kusa, K.; Abe, K.; Ishikawa, T. Effects of Cattle manure application on the leaching of nitrogen, phosphorus, and potassium from forage rice paddies-results from lysimeter experiments. Jpn. J. Grassl. Sci. 2015, 61, 74–82, (In Japanese with English summary). [Google Scholar]
- Minamikawa, K.; Hayakawa, A.; Nishimura, S.; Akiyama, H.; Yagi, K. Comparison of indirect nitrous oxide emission through lysimeter drainage between an Andosol upland field and a Fluvisol paddy field. Soil Sci. Plant Nutr. 2011, 57, 843–854. [Google Scholar] [CrossRef]
- Sato, T. Soil Fertility Management Using Green Manure for Paddy-Upland Rotation Fields. In Fertility and Management of Soil in Paddy-Upland Rotation Fields in Japan—Factors of Fertility Change and Approaches for Its Control; Japanese Society of Soil Science and Plant Nutrition, Ed.; Hakuyu-sha: Tokyo, Japan, 2010; pp. 165–192. (In Japanese) [Google Scholar]
- Sato, T.; Sato, E.; Takakai, F.; Yokoyama, T.; Kaneta, Y. Effects of hairy vetch foliage application on nodulation and nitrogen fixation in soybean cultivated in three soil types. Soil Sci. Plant Nutr. 2011, 57, 313–319. [Google Scholar] [CrossRef]
| Month | Air Temperature (°C) | Precipitation (mm) | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 2008 | 2009 | 2010 | 2011 | 2012 | 2013 | 2014 | Average † | 2008 | 2009 | 2010 | 2011 | 2012 | 2013 | 2014 | Average † | |
| Jan. | −1.7 | 0.0 | −0.3 | −2.5 | −2.4 | −2.3 | −1.4 | −0.7 | 93 | 199 | 165 | 93 | 54 | 71 | 129 | 135 |
| Feb. | −1.3 | 0.0 | −0.9 | −0.1 | −2.5 | −1.8 | −1.4 | −0.4 | 67 | 140 | 73 | 67 | 64 | 51 | 67 | 90 |
| Mar. | 4.2 | 2.8 | 1.8 | 0.9 | 2.0 | 2.1 | 2.0 | 2.4 | 66 | 124 | 119 | 82 | 180 | 80 | 126 | 101 |
| Apr. | 10.4 | 8.8 | 6.8 | 7.4 | 8.4 | 7.0 | 8.4 | 8.3 | 39 | 121 | 142 | 155 | 103 | 141 | 26 | 119 |
| May | 14.2 | 15.0 | 13.4 | 13.5 | 14.2 | 13.5 | 14.5 | 14.2 | 90 | 82 | 154 | 181 | 85 | 64 | 127 | 114 |
| Jun. | 18.3 | 18.8 | 19.6 | 18.3 | 18.3 | 20.2 | 20.1 | 19.0 | 53 | 110 | 174 | 294 | 69 | 22 | 174 | 118 |
| Jul. | 23.0 | 21.7 | 23.8 | 24.0 | 22.4 | 22.3 | 23.0 | 21.9 | 155 | 342 | 246 | 81 | 176 | 544 | 152 | 197 |
| Aug. | 22.6 | 22.6 | 25.8 | 24.2 | 25.3 | 24.0 | 23.3 | 23.7 | 246 | 169 | 234 | 243 | 64 | 250 | 277 | 211 |
| Sep. | 19.6 | 18.1 | 20.1 | 20.3 | 22.6 | 19.7 | 18.2 | 19.6 | 62 | 80 | 207 | 386 | 76 | 184 | 155 | 150 |
| Oct. | 13.8 | 12.9 | 13.6 | 13.2 | 13.8 | 14.3 | 12.1 | 13.1 | 165 | 167 | 143 | 147 | 190 | 241 | 213 | 169 |
| Nov. | 6.6 | 7.5 | 7.1 | 8.1 | 6.5 | 5.6 | 7.5 | 7.3 | 240 | 232 | 243 | 139 | 388 | 358 | 130 | 209 |
| Dec. | 2.6 | 1.7 | 2.5 | 0.4 | −0.1 | 1.5 | 0.0 | 1.9 | 153 | 138 | 161 | 124 | 158 | 174 | 221 | 162 |
| Annual ‡ | 11.0 | 10.8 | 11.1 | 10.6 | 10.7 | 10.5 | 10.5 | 10.9 | 1426 | 1903 | 2059 | 1989 | 1605 | 2178 | 1794 | 1775 |
| May-Sep. ‡ | 19.5 | 19.2 | 20.5 | 20.0 | 20.6 | 20.0 | 19.8 | 19.7 | 605 | 782 | 1015 | 1184 | 470 | 1063 | 884 | 791 |
| Year | Crop | Cultivar | T-N (g m−2) | T-P (g m−2) | T-K (g m−2) | Remarks | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| C | I | M | C | I | M | C | I | M | ||||||||||
| CF | CF | LMC | CF | LMC | CF | CF | LMC | CF | LMC | CF | CF | LMC | CF | LMC | ||||
| 2000 | Soybean | NO | NO | NO | - | NO | - | NO | NO | - | NO | - | NO | NO | - | NO | - | Newly established |
| 2001 | Bare soil (upland) | NO | NO | NO | - | NO | - | NO | NO | - | NO | - | NO | NO | - | NO | - | |
| 2002 | Young soybean | NO | NO | NO | - | NO | - | NO | NO | - | NO | - | NO | NO | - | NO | - | |
| 2003 | Soybean | NO | NO | NO | - | NO | - | NO | NO | - | NO | - | NO | NO | - | NO | - | |
| 2004 | Forage rice | Bekoaoba | 10.0 | 10.0 | 18.6 | 10.0 | 15.6 | 2.6 | 2.6 | 17.9 | 2.6 | 24.5 | 5.0 | 5.0 | NO | 5.0 | NO | 3 kg FM m−2 of immature and mature compost for I and M, respectively every year |
| 2005 | 12.0 | 12.0 | 11.4 | 12.0 | 23.1 | 2.6 | 2.6 | 6.7 | 2.6 | 15.5 | 5.0 | 5.0 | 17.9 | 5.0 | 31.3 | |||
| 2006 | 8.5 | 8.5 | 12.1 | 8.5 | 22.9 | 2.6 | 2.6 | 5.9 | 2.6 | 15.9 | 5.0 | 5.0 | 13.9 | 5.0 | 30.5 | |||
| 2007 | 10.0 | 10.0 | 16.5 | 10.0 | 22.7 | 2.6 | 2.6 | 9.6 | 2.6 | 16.8 | 5.0 | 5.0 | 15.7 | 5.0 | 28.6 | |||
| 2008 | Soybean | Ryuho | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | This study |
| 2009 | 2.0 | 2.0 | - | 2.0 | - | 2.6 | 2.6 | - | 2.6 | - | 5.0 | 5.0 | - | 5.0 | - | |||
| 2010 | 2.0 | 2.0 | - | 2.0 | - | 2.6 | 2.6 | - | 2.6 | - | 5.0 | 5.0 | - | 5.0 | - | |||
| 2011 | Staple rice | Yumeobako | 3.0 | 3.0 | - | 3.0 | - | 0.9 | 0.9 | - | 0.9 | - | 1.7 | 1.7 | - | 1.7 | - | This study |
| 2012 | Akitakomachi | 8.0 | 8.0 | - | 8.0 | - | 3.5 | 3.5 | - | 3.5 | - | 6.6 | 6.6 | - | 6.6 | - | ||
| 2013 | 10.0 | 10.0 | - | 10.0 | - | 4.4 | 4.4 | - | 4.4 | - | 8.3 | 8.3 | - | 8.3 | - | |||
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Takakai, F.; Kikuchi, T.; Sato, T.; Takeda, M.; Sato, K.; Nakagawa, S.; Kon, K.; Sato, T.; Kaneta, Y. Changes in the Nitrogen Budget and Soil Nitrogen in a Field with Paddy–Upland Rotation with Different Histories of Manure Application. Agriculture 2017, 7, 39. https://doi.org/10.3390/agriculture7050039
Takakai F, Kikuchi T, Sato T, Takeda M, Sato K, Nakagawa S, Kon K, Sato T, Kaneta Y. Changes in the Nitrogen Budget and Soil Nitrogen in a Field with Paddy–Upland Rotation with Different Histories of Manure Application. Agriculture. 2017; 7(5):39. https://doi.org/10.3390/agriculture7050039
Chicago/Turabian StyleTakakai, Fumiaki, Takemi Kikuchi, Tomomi Sato, Masato Takeda, Kensuke Sato, Shinpei Nakagawa, Kazuhiro Kon, Takashi Sato, and Yoshihiro Kaneta. 2017. "Changes in the Nitrogen Budget and Soil Nitrogen in a Field with Paddy–Upland Rotation with Different Histories of Manure Application" Agriculture 7, no. 5: 39. https://doi.org/10.3390/agriculture7050039
APA StyleTakakai, F., Kikuchi, T., Sato, T., Takeda, M., Sato, K., Nakagawa, S., Kon, K., Sato, T., & Kaneta, Y. (2017). Changes in the Nitrogen Budget and Soil Nitrogen in a Field with Paddy–Upland Rotation with Different Histories of Manure Application. Agriculture, 7(5), 39. https://doi.org/10.3390/agriculture7050039






