Agroecological Transformation in the Salt Composition of Soil under the Phosphogypsum Influence on Irrigated Lands in Ukraine
Abstract
1. Introduction
- -
- Phosphogypsum serves as a source of macro- and microelements for the development of various ecotrophic groups of microorganisms;
- -
- The acidic reaction of phosphogypsum creates favourable conditions for the breakdown of organic compounds, such as surfactants, hydrocarbons, and other substances, allowing it to be composted with waste containing such substances as sewage sludge, straw, manure, and bird droppings;
- -
- Composting various types of organic waste together with phosphogypsum, as well as its use together with digestate, significantly improves the sanitary-epidemiological situation and can find practical applications in environmental remediation.
2. Materials and Methods
2.1. Materials of Inverstigation
2.1.1. Soil Properties and Composition
2.1.2. Phosphogypsum Composition
2.2. Methodology of Field Research
- -
- 1.4 t/ha—ameliorative dose for displacing exchangeable sodium in saline-sodic soils with malonic reactions;
- -
- 3 t/ha—dose of soil calcium supplementation;
- -
- 6 t/ha—dose calculated by the coagulation-peptisation method.
- (1)
- For HCO31− less than Ca2+,
- Ca1 = Ca2+ − HCO31−
- SO4 tox. = SO42− − Ca1
- Stox. salts = (Na1+ + Mg 2++ Cl1− + SO4 tox.)
- (2)
- For HCO31− more than Ca2+,
- HCO3 tox. = HCO31− − Ca2+
- Stox. salts = (Na1+ + Mg 2++ Cl1−+ SO42− + HCO3 tox.)
- (3)
- For SO42− less than Ca2+,
- Ca1 = Ca2+− HCO31−
- Ca tox. = Ca1 − SO42−
- Stox. salts = (Ca tox. + Na1+ + Mg 2++ Cl1−).
3. Results
4. Discussion
5. Conclusions
6. Patents
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Cherlet, M.; Hutchinson, C.; Reynolds, J.; Hill, J.; Sommer, S.; Von Maltitz, G. World Atlas of Desertification; Publications Office of the European Union: Luxembourg, 2018; ISBN 978-92-79-75349-7. [Google Scholar] [CrossRef]
- Zak, N.A.; Kløve, B.; Haghigh, A.T. Expanding the Irrigated Areas in the MENA and Central Asia: Challenges or Opportunities. Water 2022, 14, 2560. [Google Scholar] [CrossRef]
- Syed, A.; Sarwar, G.; Shah, S.H.; Muhammad, S. Soil Salinity Research in 21st Century in Pakistan: Its Impact on Availability of Plant Nutrients, Growth and Yield of Crops. Commun. Soil Sci. Plant Anal. 2020, 52, 183–200. [Google Scholar] [CrossRef]
- Zajac, Z.; Gomez, O.; Gelati, E.; van der Velde, M.; Bassu, S.; Ceglar, A.; Chukaliev, O.; Panarello, L.; Koeble, R.; van den Berg, M.; et al. Estimation of spatial distribution of irrigated crop areas in Europe for large-scale modelling applications. Agric. Water Manag. 2022, 266, 107527. [Google Scholar] [CrossRef]
- Jiang, Q.; Peng, J.; Biswas, A.; Hu, J.; Zhao, R.; He, K.; Shi, Z. Characterising dryland salinity in three dimensions. Sci. Total Environ. 2019, 682, 190–199. [Google Scholar] [CrossRef] [PubMed]
- Roldán-Cañas, J.; Moreno-Pérez, M.F. Water and irrigation management in arid and semiarid zones. Water 2021, 13, 2446. [Google Scholar] [CrossRef]
- Hassani, A.; Azapagica, A.; Shokri, N. Predicting long-term dynamics of soil salinity and sodicity on a global scale. Proc. Natl. Acad. Sci. USA 2020, 117, 33017–33027. [Google Scholar] [CrossRef] [PubMed]
- Fahad, S.; Sonmez, O.; Saud, S.; Wang, D.; Wu, C.; Adnan, M.; Turan, V. (Eds.) Sustainable Soil and Land Management and Climate Change, 1st ed.; CRC Press: Boca Raton, FL, USA, 2021. [Google Scholar] [CrossRef]
- State Agency of Water Resources of Ukraine. Kyiv. Available online: https://davr.gov.ua/ (accessed on 30 October 2023).
- DSTU 2730:2015; Quality of Natural Water for Irrigation. Agronomic Criteria. Derzhstandart Ukrayiny: Kyiv, Ukraine, 2015; p. 13. (In Ukrainian)
- Balyuk, S.A.; Romaschenko, M.I.; Truskavetskyi, R.S.; Grin, D.S. Soil Reclamation (Systematics, Perspectives, Innovations): Collective Monograph; Government of Ukraine: Kherson, Ukraine, 2015; pp. 363–373.
- Hapich, H.; Andrieiev, V.; Kovalenko, V.; Makarova, T. The analysis of spatial distribution of artificial reservoirs as anthropogenic fragmentation elements of rivers in the Dnipropetrovsk Region, Ukraine. J. Water Land Dev. 2022, 53, 80–85. [Google Scholar] [CrossRef]
- Onopriienko, D.M.; Makarova, T.K.; Hapich, H.V. Assessment of the hydrogeological and ameliorative state of the Kilchen irrigation system territory. IOP Conf. Ser. Earth Environ. Sci. 2023, 1254, 012087. [Google Scholar] [CrossRef]
- Onopriienko, D.M.; Makarova, T.K.; Tkachuk, A.V.; Hapich, H.V.; Roubik, H. The influence of phosphogypsumon the salt composition of salinated soil. Land Reclam. Water Manag. 2023, 301–350. [Google Scholar] [CrossRef]
- Yin, X.; Feng, Q.; Li, Y.; Liu, W.; Zhu, M.; Xu, G.; Zheng, X.; Sindikubwabo, C. Induced soil degradation risks and plant responses by salinity and sodicity in intensive irrigated agro-ecosystems of seasonally-frozen arid regions. J. Hydrol. 2021, 603, 127036. [Google Scholar] [CrossRef]
- Chushkina, I.; Hapich, H.; Matukhno, O.; Pavlychenko, A.; Kovalenko, V.; Sherstiuk, Y. Loss of small rivers across the steppe: Climate change or the hand of man? case study of the Chaplynka River. Int. J. Environ. Stud. 2024, 1–15. [Google Scholar] [CrossRef]
- Eltazarov, S. Soil Salinity Assessment in Syrdarya Province, Uzbekistan; Wageningen University and Research Centre: Wageningen, The Netherlands, 2016; 75p. [Google Scholar] [CrossRef]
- Chornyy, S.G.; Isaeva, V.V. Salinisation of chernozem soils by brackish irrigation water in southern Ukraine. Int. J. Environ. Stud. 2023, 80, 421–432. [Google Scholar] [CrossRef]
- Shevchenko, S.; Derevenets-Shevchenko, K.; Desyatnyk, L.; Shevchenko, M.; Sologub, I.; Shevchenko, O. Tillage effects on soil physical properties and maize phenology. Int. J. Environ. Stud. 2024, 1–10. [Google Scholar] [CrossRef]
- Baliuk, S.; Vorotyntseva, L.; Zakharova, M.; Panarin, R.; Kuts, O.; Mykhailyn, V. Changes in the properties of Chernozem soils under management and strategic approaches to restore their fertility. Int. J. Environ. Stud. 2023, 1–8. [Google Scholar] [CrossRef]
- Nosonenko, O.; Zakharova, M.; Vorotynseva, L.; Afanasiev, Y. Effect of differential of doses of chemical improver on the indicators of halogenesis of dark-chestnut alkaline soil. Bull. Agric. Sci. 2022, 110, 12–19. [Google Scholar] [CrossRef]
- Makarova, T.; Domaratskiy Ye Hapich, G.; Kozlova, O. Agromeliorative efficiency of phosphogypsum application on irrigation saline soils in the Northern Steppe of Ukraine. Indian J. Ecol. 2021, 48, 789–795. [Google Scholar]
- Brillante, L.; Singh, K. Use of Gypsum to Reclaim Salt Problems in Soils. Progressive Crop Consultant. 2022. Available online: https://surl.li/mylmx (accessed on 1 November 2023).
- Bello, S.K.; Alayafi, A.H.; Al-Solaimani, S.G.; Abo-Elyousr, K.A. Mitigating Soil Salinity Stress with Gypsum and Bio-Organic Amendments: A Review. Agronomy 2021, 11, 1735. [Google Scholar] [CrossRef]
- Seo, B.S.; Baek, N.; Park, S.W.; Shin, E.S.; Oh, Y.Y.; Kang, B.H.; Park, H.J.; Choi, W.J. Spatial Variations in Salinity and Sodicity of Reclaimed Tideland Paddy Soils in Coastal Areas of Southwestern Korea. Korean J. Soil 2023, 56, 217–225. [Google Scholar] [CrossRef]
- Baliuk, S.A.; Kucher, A.V.; Maksymenko, N.V. Soil resources of Ukraine: State, problems and strategy of sustainable management. Ukr. Geogr. J. 2021, 2, 3–11. [Google Scholar] [CrossRef]
- Dustnazarova, S.; Khasanov, A.; Khafizova, Z.; Davronov, K. The threat of saline lands, for example, in the Republic of Uzbekistan. E3S Web Conf. 2021, 284, 02002. [Google Scholar] [CrossRef]
- Hopmans, J.W.; Qureshi, A.S.; Kisekka, I.; Munns, R.; Grattan, S.R.; Rengasamy, P.; Ben-Gal, A.; Assouline, S.; Javaux, M.; Minhas, P.S.; et al. Taleisnik Chapter One—Critical knowledge gaps and research priorities in global soil salinity. Adv. Agron. 2021, 169, 1–191. [Google Scholar] [CrossRef]
- Kharytonov, M.; Martynova, N.; Babenko, M.; Rula, I.; Ungureanu, N.; Ștefan, V. Production of sweet sorghum bio-feedstock on technosol using municipal sewage sludge treated with flocculant, in Ukraine. Agriculture 2023, 13, 1129. [Google Scholar] [CrossRef]
- Arienzo, M.; Christen, E.; Jayawardane, N.S.; Quayle, W.C. The relative effects of sodium and potassium on soil hydraulic conductivity and implications for winery wastewater management. Geoderma 2012, 173–174, 303–310. [Google Scholar] [CrossRef]
- Degirmenci, N.; Okucu, A.; Turabi, A. Application of phosphogypsum in soil stabilization. Build. Environ. 2007, 42, 3393–3398. [Google Scholar] [CrossRef]
- Truskavetskyi, R.S.; Baliuk, S.A. Resource-Saving Technologies of Chemical Land Reclamation in the Conditions of Land Reform; Derzhavnyi Instytut Upravlinnya ta Ekonomiky Vodnykh Resursiv: Kyiv, Ukraine, 2000; 70p. [Google Scholar]
- Lyubimova, I.N.; Salpagarova, I.A. Possibility and Feasibility of Returning the Formerly Reclaimed Solonetz Lands to Agricultural Use: A Review. Eurasian Soil Sci. 2020, 53, 1270–1279. [Google Scholar] [CrossRef]
- El Zrelli, R.; Rabaoui, L.; Daghbouj, N.; Abda, H.; Castet, S.; Josse, C.; van Beek, P.; Souhaut, M.; Michel, S.; Bejaoui, N.; et al. Characterization of phosphate rock and phosphogypsum from. Gabes phosphate fertilizer factories (SE Tunisia): High mining potential and implications for environmental protection. Environ. Sci. Pollut. Res. 2018, 25, 14690–14702. [Google Scholar] [CrossRef]
- Manukyan, R.R. Development direction of the soil-formation processes for reclaimed soda solonetz-solonchak soils of the Ararat valley during their cultivation. Ann. Agrar. Sci. 2018, 16, 69–74. [Google Scholar] [CrossRef]
- Wei, Z.; Deng, Z. Research hotspots and trends of comprehensive utilization of phosphogypsum: Bibliometr. analysis. J. Environ. Radioact. 2022, 242, 106778. [Google Scholar] [CrossRef]
- Chernysh, Y.; Yakhnenko, O.; Chubur, V.; Roubík, H. Phosphogypsum Recycling. A Review of Environmental Issues, Current Trends, and Prospects. Appl. Sci. 2021, 11, 15–75. [Google Scholar] [CrossRef]
- Dmytruk, Y.; Cherlinka, V.; Cherlinka, L.; Dent, D. Soils in war and peace. Int. J. Environ. Stud. 2023, 80, 380–393. [Google Scholar] [CrossRef]
- Hapich, H.; Zahrytsenko, A.; Sudakov, A.; Pavlychenko, A.; Yurchenko, S.; Sudakova, D.; Chushkina, I. Prospects of alternative water supply for the population of Ukraine during wartime and post-war reconstruction. Int. J. Environ. Stud. 2024, 81, 1–12. [Google Scholar] [CrossRef]
- Ushkarenko, V.O.; Morozov, V.V.; Zhuzha, V.V.; Zhuzha, A.V. Evolution of the hydrogeological state under the influence of urbanization and land reclamation on the territory of the city of Kherson. Tavriysʹkyy Nauk. Visnyk. 2002, 21, 112–125. (In Ukrainian). Available online: http://surl.li/nelmt (accessed on 1 June 2023).
- Xu, X.; Guo, L.; Wang, S.; Wang, X.; Ren, M.; Zhao, P.; Huang, Z.; Jia, H.; Wang, J.; Lin, A. Effective strategies for reclamation of saline-alkali soil and response mechanisms of the soil-plant system. Sci. Total Environ. 2023, 905, 167179. [Google Scholar] [CrossRef]
- Bouargane, B.; Pérez-Moreno, S.M.; Barba-Lobo, A.; Bakiz, B.; Atbir, A.; Bolívar, J.P. Behavior of heavy metals and natural radionuclides along the Moroccan phosphogypsum carbonation process with several alkaline reagents. Chem. Eng. Sci. 2023, 280, 119013. [Google Scholar] [CrossRef]
- Pohrebennyk, V.; Dzhumelia, E. Methods of Soils Pollution Spread Analysis: Case Study of Mining and Chemical Enterprise in Lviv Region (Ukraine). Ecol. Eng. Environ. Technol. 2021, 4, 39–44. [Google Scholar] [CrossRef] [PubMed]
- Jalali, J.; Gaudin, P.; Capiaux, H.; Ammar, E.; Lebeau, T. Fate and transport of metal trace elements from phosphogypsum piles in Tunisia and their impact on soil bacteria and wild plants. Ecotoxicol. Environ. Saf. 2019, 174, 12–25. [Google Scholar] [CrossRef] [PubMed]
- Haneklaus, N.; Barbossa, S.; Basallote, M.D.; Bertau, M.; Bilal, E.; Chajduk, E.; Chernysh, Y.; Chubur, V.; Cruz, J.; Dziarczykowski, K.; et al. Closing the upcoming EU gypsum gap with phosphogypsum, Resources. Conserv. Recycl. 2022, 182, 106328. [Google Scholar] [CrossRef]
- Onopriienko, D.M.; Makarova, T.K. Analysis of degradation processes of irrigated soils of Dnipropetrovsk region. Taurian Sci. Bull. 2013, 86, 146–151. [Google Scholar]
- Ushkarenko, V.O.; Vozhegova, R.A.; Goloborodko, S.P.; Kokovikhin, S.V. Statistical Analysis of the Results of Field Experiments in Agriculture; Kherson: Ailant, Ukraine, 2013; p. 381. [Google Scholar]
- DSTU 4730:2007; Soil Quality. Determination of the Granulometric Composition by the Pipette Method in the Modification of N.A. Kaczynski. K. Derzhspozhyvstandart Ukrayiny: Kyiv, Ukraine, 2008; p. 18. (In Ukrainian)
- DSTU ISO 11508:2005; Soil Quality. Determination of Particle Density (ISO 11508:1998, IDT). Derzhspozhyvstandart Ukrayiny: Kyiv, Ukraine, 2008; p. 15. (In Ukrainian)
- DSTU ISO 112651; Modern Measures and Technologies of Land Reclamation of Naturally Saline and Secondary Saline Soils of Ukraine: Recommendations/NSC “Institute of Soil Science and Agrochemistry Named After O. N. Sokolovsky. Derzhspozhyvstandart Ukrayiny: Kyiv, Ukraine, 2011; p. 48. (In Ukrainian)
- DSTU 7537:2014; Soil Quality. Determination of Hydrolytic Acidity. Derzhspozhyvstandart Ukrayiny: Kyiv, Ukraine, 2015; p. 12. (In Ukrainian)
- DSTU 8345:2015; Soil Quality. Methods of Determining Cation Exchange Capacity. Derzhspozhyvstandart Ukrayiny: Kyiv, Ukraine, 2017; p. 10. (In Ukrainian)
- Regional Office of Water Resources in Dnipropetrovsk Region (Ukraine). Available online: http://dovr.gov.ua/ (accessed on 1 November 2023).
- Dnipro Mineral Fertilizer Plant. Product Certificates. Available online: https://dzmu.dp.ua/docs.php (accessed on 1 November 2023).
- Instructions for Soil and Salt Survey on Irrigated Lands of Ukraine: DND 33-5.5-11-2002, Valid from 20/08/2002; State Committee of Ukraine for Water Management: Kyiv, Ukraine, 2002; 31p.
- Gharaibeh, M.A.; Albalasmeh, A.A.; Pratt, C.; El Hanandeh, A. Estimation of exchangeable sodium percentage from sodium adsorption ratio of salt-affected soils using traditional and dilution extracts, saturation percentage, electrical conductivity, and generalized regression neural networks. CATENA 2021, 205, 105466. [Google Scholar] [CrossRef]
- Wang, R.; Wan, S.; Sun, J.; Xiao, H. Soil salinity, sodicity and cotton yield parameters under different drip irrigation regimes during saline wasteland reclamation. Agric. Water Manag 2018, 209, 20–31. [Google Scholar] [CrossRef]
- Mau, Y.; Porporato, A. A dynamical system approach to soil salinity and sodicity. Adv. Water Resour. 2015, 83, 68–76. [Google Scholar] [CrossRef]
- Geilfus, C.-M. Chloride in soil: From nutrient to soil pollutant. Environ. Exp. Bot. 2019, 157, 299–309. [Google Scholar] [CrossRef]
- Zakharchenko, E.; Tunguz, V. Effect of ammonium sulfate and phosphogypsum application on nutrients dynamics and acidity of black soil. Bull. Sumy Natl. Agrar. Univ. Ser. Agron. Biol. 2020, 42, 61–69. [Google Scholar] [CrossRef]
- Davydchuk, M.I.; Kisorets, P.F.; Hantsevska, N.A. Influence of calcium-containing chemical ameliorants on physicochemical and agrochemical properties of dark chestnut secondary saline soil. Ecology 2013, 220, 50–54. (In Ukrainian) [Google Scholar]
- Onopriienko, D.; Makarova, T.; Tkachuk, A.; Hapich, H.; Roubik, H. Prevention of degradation processes of soils irrigated with mineralized water through plastering. Ukr. Black Sea Reg. Agrar. Sci. 2023, 27, 9–20. [Google Scholar] [CrossRef]
- Beltrán, J.M. Irrigation with saline water: Benefits and environmental impact Agric. Water Manag. 1999, 98, 0378–3774. [Google Scholar] [CrossRef]
- Bouray, M.; Moir, J.; Condron, L.; Lehto, N. Impacts of Phosphogypsum, Soluble Fertilizer and Lime Amendment of Acid Soils on the Bioavailability of Phosphorus and Sulphur under Lucerne (Medicago sativa). Plants 2020, 9, 883. [Google Scholar] [CrossRef] [PubMed]
- Papastefanou, C.; Stoulos, S.; Ioannidou, A.; Manolopoulou, M. The application of phosphogypsum in agriculture and the radiological impact. J. Environ. Radioact. 2006, 89, 188–198. [Google Scholar] [CrossRef] [PubMed]
- Minhas, P.S.; Ramos, T.B.; Ben-Gal, A.; Pereira, L.S. Coping with salinity in irrigated agriculture: Crop evapotranspiration and water management issues. Agric. Water Manag. 2020, 227, 105832. [Google Scholar] [CrossRef]
- Tariq, A.; Ullah, I.; Sardans, J.; Yaseen, A.; Peñuelas, J. Strigolactones can be a potential tool to fight environmental stresses in arid lands. Environ. Res. 2023, 229, 115966. [Google Scholar] [CrossRef]
- Kong, C.; Camps-Arbestain, M.; Clothier, B.; Bishop, P.; Vázquez, F.M. Reclamation of salt-affected soils using pumice and algal amendments: Impact on soil salinity and the growth of lucerne. Environ. Technol. Innov. 2021, 24, 101867. [Google Scholar] [CrossRef]
- Majumdar, S.; Barman, F.; Paul, A.; Kundu, R. Role of sulfur in protection against major environmental stress in plants. In Biology and Biotechnology of Environmental Stress Tolerance in Plants: Trace Elements in Environmental Stress Tolerance; Apple Academic Press: New York, NY, USA, 2023; pp. 473–529. [Google Scholar] [CrossRef]
- McKenna, B.A.; Kopittke, P.M.; Macfarlane, D.C.; Dalzell, S.A.; Menzies, N.W. Changes in soil chemistry after the application of gypsum and sulfur and irrigation with coal seam water. Geoderma 2019, 337, 782–791. [Google Scholar] [CrossRef]
- Bilal, E.; Bellefqih, H.; Bourgier, V.; Mazouz, H.; Dumitraş, D.G.; Bard, F.; Laborde, M.; Caspar, J.P.; Guilhot, B.; Iatan, L.; et al. Phosphogypsum circular economy considerations: A critical review from more than 65 storage sites worldwide. J. Clean. Prod. 2023, 414, 137561. [Google Scholar] [CrossRef]
- Gabsi, H.; Tallou, A.; Aziz, F.; Boukchina, R.; Karbout, N.; Caceres, L.A.; García-Tenorio, R.; Boudabbous, K.; Moussa, M. Application of Phosphogypsum and Organic Amendment for Bioremediation of Degraded Soil in Tunisia Oasis: Targeting Circular Economy. Sustainability 2023, 15, 47–69. [Google Scholar] [CrossRef]
- Matveeva, V.A.; Smirnov, Y.D.; Suchkov, D.V. Industrial processing of phosphogypsum into organomineral fertilizer. Environ. Geochem. Health 2022, 44, 1605–1618. [Google Scholar] [CrossRef] [PubMed]
- Onopriienko, D.; Shepel, A.; Makarova, T. Influence of phosphogypsum on the chemical composition of aqueous extract from soil. Agrology 2019, 2, 151–155. [Google Scholar] [CrossRef]
- Robinson, M.J.; Dhar, A.; Naeth, M.A.; Nichol, C.K. Phosphogypsum impacts on soil chemical properties and vegetation tissue following reclamation. Environ. Monit. Assess. 2023, 195, 769. [Google Scholar] [CrossRef] [PubMed]
- Laudicina, V.A.; Hurtado, M.D.; Badalucco, L.; Delgado, A.; Palazzolo, E.; Panno, M. Soil chemical and biochemical properties of a salt-marsh alluvial Spanish area after long-term reclamation. Biol. Fertil. Soils 2009, 45, 691–700. [Google Scholar] [CrossRef]
- Mahmoud, E.; Baroudy AEl El-Kader, N.A.; Othman, S.; Khamisy, R.E. Effects of Phosphogypsum and Biochar Addition on Soil Physical Properties and Nutrients Uptake by Maize yield in Vertic Torrifluvents. Int. J. Sci. Eng. Res. 2017, 8, 1–27. [Google Scholar]
- Plyatsuk, L.; Balintova, M.; Chernysh, Y.; Demcak, S.; Holub, M.; Yakhnenko, E. Influence of Phosphogypsum Dump on the Soil Ecosystem in the Sumy region (Ukraine). Appl. Sci 2019, 9, 5559. [Google Scholar] [CrossRef]
- Chernysh, Y.; Plyatsuk, L.; Dychenko, T. The Protective Functions Stimulation of the Soil Complex with the Use of Biogenic Composites Based on The Sewage Sludge and Phosphogypsum. J. Solid Waste Technol. Manag. 2019, 45, 226–233. [Google Scholar] [CrossRef]
- Sengupta, I.; Dhal, P.K. Impact of elevated phosphogypsum on soil fertility and its aerobic biotransformation through indigenous microorganisms (IMO’s) based technology. J. Environ. Manag. 2021, 297, 113195. [Google Scholar] [CrossRef] [PubMed]
- Zielonka, D.; Nerebinski, M.; Kalaji, H.M.; Augustynowicz, J.; Predecka, A.; Russel, S. Efficiency of the photosynthetic apparatus in Cannabis sativa L. fertilized with sludge froma wastewater treatment plant and with phosphogypsum. Ecol. Quest. 2018, 28, 55–61. [Google Scholar] [CrossRef][Green Version]
- Ablieieva, I.; Berezhna, I.; Berezhnyi, D.; Enrich Prast, A.; Geletuha, G.; Lutsenko, S.; Yanchenko, I.; Carraro, G. Technologies for Environmental Safety Application of Digestate as Biofertilizer. Ecol. Eng. Environ. Technol. 2022, 23, 106–119. [Google Scholar] [CrossRef]
- Yelatontsev, D. Utilization of phosphogypsum in phenol removal from coking wastewater. J. Hazard. Mater. Lett. 2023, 4, 100089. [Google Scholar] [CrossRef]
- Tymoshchuk, V.; Rudakov, L.; Pikarenia, D.; Orlinska, O.; Hapich, H. Analyzing stability of protective structures as the elements of geotechnical tailing pond safety. Min. Miner. Depos. 2023, 17, 116–122. [Google Scholar] [CrossRef]
- Qi, J.; Zhu, H.; Zhou, P.; Wang, X.; Wang, Z.; Yang, S.; Yang, D.; Li, B. Application of phosphogypsum in soilization: A review. Int. J. Environ. Sci. Technol. 2023, 20, 10449–10464. [Google Scholar] [CrossRef]

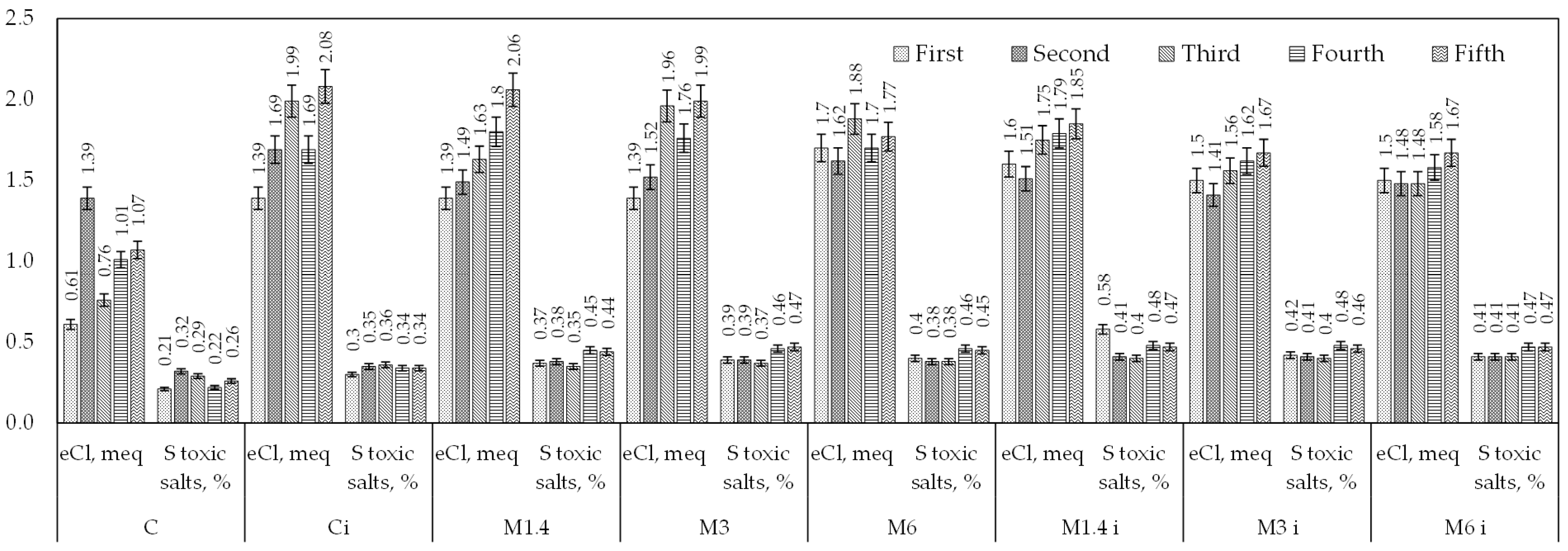
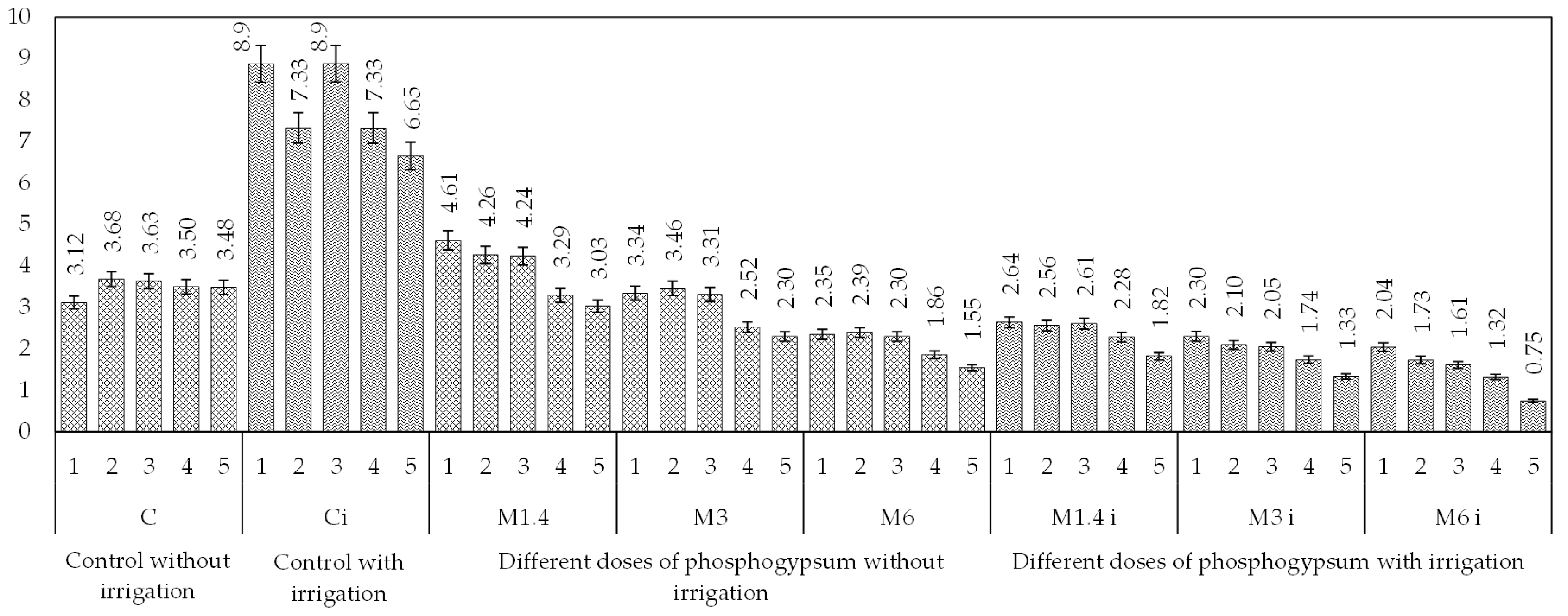
| No. | Control Indicator | Results of Water Analysis by Year | ||||
|---|---|---|---|---|---|---|
| 1st Year 2014 | 2nd Year 2015 | 3rd Year 2016 | 4th Year 2017 | 5th Year 2018 | ||
| 1 | pH | 8.2 | 7.3 | 7.6 | 8.01 | 8.33 |
| SD | 0.13 | 0.2 | 0.34 | 0.1 | 0.24 | |
| 3 | Sulfates, mg/L | 899.5 | 999.43 | 903.5 | 1331.5 | 1154 |
| SD | 3.41 | 4.83 | 4.09 | 5.35 | 5.04 | |
| 4 | Chlorides, mg/L | 453.76 | 564.76 | 466.06 | 490.2 | 520 |
| SD | 2.53 | 2.91 | 2.41 | 2.73 | 3.07 | |
| 7 | Hydrocarbons, mg/L | 306.5 | 393.85 | 310.56 | 380.56 | 551 |
| SD | 2.05 | 2.38 | 2.27 | 2.30 | 3.39 | |
| 8 | Carbonates, mg/L | - | - | - | - | - |
| 9 | Calcium, mg/L | 176.09 | 159.29 | 169.9 | 220.94 | 203 |
| SD | 1.37 | 1.24 | 1.34 | 1.89 | 1.74 | |
| 10 | Magnesium, mg/L | 180.1 | 202 | 179.69 | 200.69 | 201.41 |
| SD | 1.57 | 1.64 | 1.50 | 1.67 | 1.71 | |
| 11 | Potassium + sodium, mg/L | 330 | 423 | 340.85 | 460.54 | 478 |
| SD | 2.15 | 2.44 | 2.27 | 2.56 | 2.84 | |
| 12 | The sum of ions, mg/L | 2345.95 | 2742.33 | 2370.56 | 3084.43 | 3107.41 |
| SD | 15.41 | 16.35 | 15.94 | 16.89 | 17.04 | |
| No. | Indicator | Norm Indicator | Actual Values of the Phosphogypsum Studied |
|---|---|---|---|
| 1 | Aggregate state, appearance, smell | Wet, loose product from white to brown in colour with a specific smell | White, odorless, wet, loose product |
| 2 | Mass fraction of calcium sulfate (CaSO4·2H2O), % not less | 80 | 98.4 |
| 3 | Mass fraction of hygroscopic water, % no more | 28 | 4.5 |
| 4 | Mass fraction of water-soluble fluorine compounds, % no more | 0.6 | 0.1 |
| 5 | Mass fraction of total phosphates (P2O5), % no more | 10 | 1 |
| 6 | Hydrogen index | 6 | 5 |
| Type of Salinity | Ratio of Anions, meq | Additional Conditions | ||
|---|---|---|---|---|
| Cl1−/SO42− | HCO31−/Cl1− | HCO31−/SO42− | ||
| Hydrocarbonate | - | >2.5 | >2.5 | - |
| Chloride | >2.5 | - | - | - |
| Sulphate–chloride | 2.5–1.0 | - | - | - |
| Chloride-sulphate | 1.0–0.25 | - | - | - |
| Sulphate | <0.25 | - | - | - |
| Soda | - | >2.5 | - | HCO31− > Ca2+ + Mg2+ Na1+ > Mg2+ Na1+ > Ca2+ |
| Sodium chloride | - | 2.5–1.0 | - | |
| Sodium chloride | - | 1.0–0.25 | - | |
| Sulphate–sodium | - | - | 2.5–1.0 | |
| Soda-sulfate | - | - | 1.0–0.25 | |
| Sulphate or chloride-hydrocarbonate | - | >1.0 | >1.0 | Na1+ < Ca2+ Na1+ < Mg2+ HCO31− > Na1+ |
| Type of Salinity | Ratio of Anions, meq | ||
|---|---|---|---|
| Ca2+/Mg2+ | Na1+/Mg2+ | Na1+/Ca2+ | |
| Calcium | >2.5 | - | - |
| Magnesium–calcium | 2.5–1.0 | - | - |
| Calcium–magnesium | 1.0–0.25 | - | - |
| Magnesium | <0.25 | - | - |
| Sodium | - | >2.5 | >2.5 |
| Magnesium–sodium | - | 2.5–1.0 | - |
| Sodium–magnesium | - | 1.0–0.25 | - |
| Calcium–sodium | - | - | 2.5–1.0 |
| Sodium–calcium | - | - | 1.0–0.25 |
| Ions | CO32− | HCO31− | SO42− | Cl1− |
|---|---|---|---|---|
| Ca2+ | — | 2 | 5 | 8 |
| Mg2+ | — | 3 | 6 | 9 |
| Na1+ + K1+ | — | 4 | 7 | 10′ |
| Anions/ Cations | Factors | Year of Research | |||||
|---|---|---|---|---|---|---|---|
| Factor A: Irrigation | Factor B: Meliorant | First, 2014 | Second, 2015 | Third, 2016 | Fourth, 2017 | Fifth, 2018 | |
| HCO31− | without irrigation | C | 0.3243 | 0.5900 | 0.3480 | 0.3879 | 0.3600 |
| M1.4 | 0.1693 | 0.5464 | 0.2800 | 0.3423 | 0.3402 | ||
| M3 | 0.2970 | 0.4862 | 0.2730 | 0.3350 | 0.3395 | ||
| M6 | 0.2892 | 0.2560 | 0.2580 | 0.3300 | 0.4303 | ||
| irrigation | Ci | 0.2200 | 0.5750 | 0.3400 | 0.3596 | 0.3587 | |
| M1.4i | 0.3188 | 0.4800 | 0.3155 | 0.3580 | 0.1894 | ||
| M3i | 0.3700 | 0.4340 | 0.2918 | 0.3639 | 0.3467 | ||
| M6i | 0.3699 | 0.5670 | 0.2490 | 0.3564 | 0.3410 | ||
| A—assessment of the significance of partial differences LSD05 irrigation | 0.0365 | 0.0502 | 0.0393 | 0.0368 | 0.0430 | ||
| LSD05—introduction of meliorants | 0.0346 | 0.0525 | 0.0368 | 0.0355 | 0.0355 | ||
| B—assessment of the significance of average (main) effects LSD05-irrigation | 0.0183 | 0.0251 | 0.0197 | 0.0184 | 0.0215 | ||
| LSD05—introduction of meliorants | 0.0245 | 0.0372 | 0.0260 | 0.0251 | 0.0251 | ||
| Cl1− | without irrigation | C | 0.9900 | 0.7000 | 0.8970 | 0.9324 | 0.9590 |
| M1.4 | 1.1589 | 0.9870 | 1.1987 | 1.1160 | 1.1200 | ||
| M3 | 1.0957 | 1.0000 | 1.0987 | 1.0570 | 1.1000 | ||
| M6 | 1.1220 | 0.8790 | 0.8987 | 0.8670 | 0.9640 | ||
| irrigation | Ci | 1.2150 | 1.2480 | 1.2700 | 1.2560 | 1.3570 | |
| M1.4i | 0.9826 | 0.6270 | 0.8500 | 0.8923 | 0.8753 | ||
| M3i | 0.8980 | 0.6211 | 0.8560 | 0.8760 | 0.8750 | ||
| M6i | 0.8760 | 0.6120 | 0.7970 | 0.8327 | 0.7845 | ||
| A—assessment of the significance of partial differences LSD05 irrigation | 0.1313 | 0.0997 | 0.0900 | 0.0957 | 0.1158 | ||
| LSD05—introduction of meliorants | 0.1056 | 0.0981 | 0.1141 | 0.1020 | 0.1101 | ||
| B—assessment of the significance of average (main) effects LSD05-irrigation | 0.0656 | 0.0499 | 0.0450 | 0.0478 | 0.0579 | ||
| LSD05—introduction of meliorants | 0.0747 | 0.0694 | 0.0807 | 0.0722 | 0.0779 | ||
| SO42− | without irrigation | C | 2.3067 | 2.5070 | 2.4217 | 2.3006 | 2.2530 |
| M1.4 | 3.1700 | 2.7884 | 2.6940 | 3.2000 | 2.8800 | ||
| M3 | 3.2990 | 2.8000 | 2.7768 | 3.3586 | 3.0200 | ||
| M6 | 3.3250 | 3.0000 | 2.8453 | 3.4500 | 3.1466 | ||
| irrigation | Ci | 2.9907 | 2.7500 | 2.6500 | 2.7548 | 2.7800 | |
| M1.4i | 2.7427 | 2.7000 | 2.5215 | 2.8090 | 2.7880 | ||
| M3i | 3.0380 | 3.0195 | 2.7640 | 3.2967 | 3.2600 | ||
| M6i | 3.4248 | 3.3660 | 3.1751 | 3.6034 | 3.5770 | ||
| A—assessment of the significance of partial differences LSD05 irrigation | 0.3062 | 0.2447 | 0.3625 | 0.3370 | 0.3328 | ||
| LSD05—introduction of meliorants | 0.3468 | 0.3174 | 0.2812 | 0.3287 | 0.3000 | ||
| B—assessment of the significance of average (main) effects LSD05-irrigation | 0.1531 | 0.1224 | 0.1812 | 0.1685 | 0.1664 | ||
| LSD05—introduction of meliorants | 0.2452 | 0.2244 | 0.1988 | 0.2324 | 0.2121 | ||
| Ca2+ | without irrigation | C | 0.6210 | 0.5970 | 0.5736 | 0.5800 | 0.6000 |
| M1.4 | 0.7300 | 0.7000 | 0.6780 | 0.9500 | 0.8904 | ||
| M3 | 1.0240 | 1.0000 | 0.9870 | 1.3486 | 1.2430 | ||
| M6 | 1.5600 | 1.4780 | 1.4250 | 2.0240 | 1.8760 | ||
| irrigation | Ci | 0.2537 | 0.3130 | 0.3200 | 0.3000 | 0.3123 | |
| M1.4i | 1.1246 | 1.1000 | 1.0923 | 1.2900 | 1.2794 | ||
| M3i | 1.5790 | 1.5200 | 1.4890 | 2.1526 | 2.1000 | ||
| M6i | 2.1570 | 2.1000 | 2.0000 | 2.7345 | 2.6900 | ||
| A—assessment of the significance of partial differences LSD05 irrigation | 0.1758 | 0.1545 | 0.1353 | 0.1697 | 0.1952 | ||
| LSD05—introduction of meliorants | 0.1434 | 0.1245 | 0.1215 | 0.1907 | 0.1655 | ||
| B—assessment of the significance of average (main) effects LSD05-irrigation | 0.0879 | 0.0772 | 0.0677 | 0.0848 | 0.0976 | ||
| LSD05—introduction of meliorants | 0.1014 | 0.0880 | 0.0859 | 0.1349 | 0.1170 | ||
| Mg2+ | without irrigation | C | 0.5800 | 0.4900 | 0.4700 | 0.4872 | 0.4500 |
| M1.4 | 0.3633 | 0.4242 | 0.3950 | 0.7083 | 0.7280 | ||
| M3 | 0.6280 | 0.3952 | 0.4015 | 0.7920 | 0.8611 | ||
| M6 | 0.6875 | 0.3642 | 0.3890 | 0.5230 | 0.8580 | ||
| irrigation | Ci | 0.1560 | 0.2780 | 0.2840 | 0.2040 | 0.3689 | |
| M1.4i | 0.5208 | 0.4500 | 0.3667 | 0.5693 | 0.7400 | ||
| M3i | 0.4270 | 0.4656 | 0.4200 | 0.4160 | 0.7820 | ||
| M6i | 0.2670 | 0.4770 | 0.4390 | 0.4000 | 1.0000 | ||
| A—assessment of the significance of partial differences LSD05 irrigation | 0.0590 | 0.0477 | 0.0452 | 0.0613 | 0.1149 | ||
| LSD05—introduction of meliorants | 0.0473 | 0.0462 | 0.0410 | 0.0528 | 0.0719 | ||
| B—assessment of the significance of average (main) effects LSD05 irrigation | 0.0295 | 0.0238 | 0.0226 | 0.0307 | 0.0575 | ||
| LSD05—introduction of meliorants | 0.0334 | 0.0327 | 0.0290 | 0.0374 | 0.0509 | ||
| Na1+ | without irrigation | C | 2.4200 | 2.7100 | 2.6231 | 2.5537 | 2.5220 |
| M1.4 | 3.4049 | 3.1976 | 3.0997 | 3.0000 | 2.7218 | ||
| M3 | 3.0397 | 2.8910 | 2.7600 | 2.6100 | 2.3554 | ||
| M6 | 2.4887 | 2.2928 | 2.1880 | 2.1000 | 1.8069 | ||
| irrigation | Ci | 4.0160 | 3.9820 | 3.6560 | 3.8664 | 3.8145 | |
| M1.4i | 2.3987 | 2.2570 | 2.2280 | 2.2000 | 1.8333 | ||
| M3i | 2.3000 | 2.0890 | 2.0028 | 1.9680 | 1.5997 | ||
| M6i | 2.2467 | 1.9680 | 1.7821 | 1.6580 | 1.0125 | ||
| A—assessment of the significance of partial differences LSD05 irrigation | 0.3165 | 0.2883 | 0.3776 | 0.3219 | 0.5010 | ||
| LSD05—introduction of meliorants | 0.3559 | 0.3831 | 0.3302 | 0.3772 | 0.3807 | ||
| B—assessment of the significance of average (main) effects LSD05 irrigation | 0.1583 | 0.1441 | 0.1888 | 0.1609 | 0.2505 | ||
| LSD05—introduction of meliorants | 0.2517 | 0.2709 | 0.2335 | 0.2667 | 0.2692 | ||
| Research Variant | First Year, 2014 | Second Year, 2015 | Third Year, 2016 | Fourth Year, 2017 | Fifth Year, 2018 | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| The Sum of Exchangeable Cations, meq/100 g of Soil | Exchangeable Na1+,% of the Sum of Exchangeable Cations | pH | The Sum of Exchangeable Cations, meq/100 g of Soil | Exchangeable Na1+,% of the Sum of Exchangeable Cations | pH | The Sum of Exchangeable Cations, meq/100 g of Soil | Exchangeable Na1+,% of the Sum of Exchangeable Cations | pH | The Sum of Exchangeable Cations, meq/100 g of Soil | Exchangeable Na1+,% of the Sum of Exchangeable Cations | pH | The Sum of Exchangeable Cations, meq/100 g of Soil | Exchangeable Na1+,% of the Sum of Exchangeable Cations | pH | |
| C | 28.76 | 4.03 | 7.4 | 28.62 | 4.19 | 7.2 | 28.34 | 4.24 | 7.4 | 28.53 | 4.25 | 7.5 | 28.3 | 4.19 | 7.5 |
| Ci | 28.87 | 4.50 | 7.5 | 28.91 | 5.00 | 7.4 | 28.27 | 5.13 | 7.5 | 28.58 | 5.18 | 7.8 | 28.48 | 5.27 | 7.9 |
| M1,4 | 29.43 | 2.65 | 7.37 | 29.67 | 2.70 | 6.97 | 26.79 | 2.58 | 7.34 | 29.29 | 1.67 | 7.34 | 28.45 | 1.58 | 7.25 |
| M3 | 30.01 | 2.37 | 7.29 | 29.35 | 2.30 | 6.89 | 27.28 | 2.27 | 7.26 | 29.59 | 1.52 | 7.27 | 28.97 | 1.48 | 7.18 |
| M6 | 30.63 | 2.12 | 7.17 | 29.37 | 2.10 | 6.7 | 27.88 | 2.08 | 7.2 | 30.37 | 1.25 | 7.24 | 29.57 | 1.01 | 7.12 |
| M1,4 i | 27.21 | 2.13 | 7.43 | 26.94 | 2.00 | 7.0 | 26.76 | 1.72 | 7.38 | 27.65 | 1.23 | 7.41 | 27.43 | 1.09 | 7.34 |
| M3 i | 29.96 | 1.54 | 7.36 | 28.35 | 1.43 | 6.91 | 27.88 | 1.36 | 7.3 | 29.7 | 1.05 | 7.35 | 28.86 | 0.97 | 7.27 |
| M6 i | 28.27 | 1.49 | 7.22 | 27.89 | 1.37 | 6.75 | 27.57 | 1.27 | 7.21 | 28.43 | 0.90 | 7.31 | 27.95 | 0.89 | 7.2 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Onopriienko, D.; Makarova, T.; Hapich, H.; Chernysh, Y.; Roubík, H. Agroecological Transformation in the Salt Composition of Soil under the Phosphogypsum Influence on Irrigated Lands in Ukraine. Agriculture 2024, 14, 408. https://doi.org/10.3390/agriculture14030408
Onopriienko D, Makarova T, Hapich H, Chernysh Y, Roubík H. Agroecological Transformation in the Salt Composition of Soil under the Phosphogypsum Influence on Irrigated Lands in Ukraine. Agriculture. 2024; 14(3):408. https://doi.org/10.3390/agriculture14030408
Chicago/Turabian StyleOnopriienko, Dmytro, Tetiana Makarova, Hennadii Hapich, Yelizaveta Chernysh, and Hynek Roubík. 2024. "Agroecological Transformation in the Salt Composition of Soil under the Phosphogypsum Influence on Irrigated Lands in Ukraine" Agriculture 14, no. 3: 408. https://doi.org/10.3390/agriculture14030408
APA StyleOnopriienko, D., Makarova, T., Hapich, H., Chernysh, Y., & Roubík, H. (2024). Agroecological Transformation in the Salt Composition of Soil under the Phosphogypsum Influence on Irrigated Lands in Ukraine. Agriculture, 14(3), 408. https://doi.org/10.3390/agriculture14030408








