Phylogeny and Pathogenicity of Phytophthora Species Associated with Artichoke Crown and Root Rot and Description of Phytophthora marrasii sp. nov. †
Abstract
:1. Introduction
2. Materials and Methods
2.1. Sites, Sampling Procedure, and Phytophthora Isolations
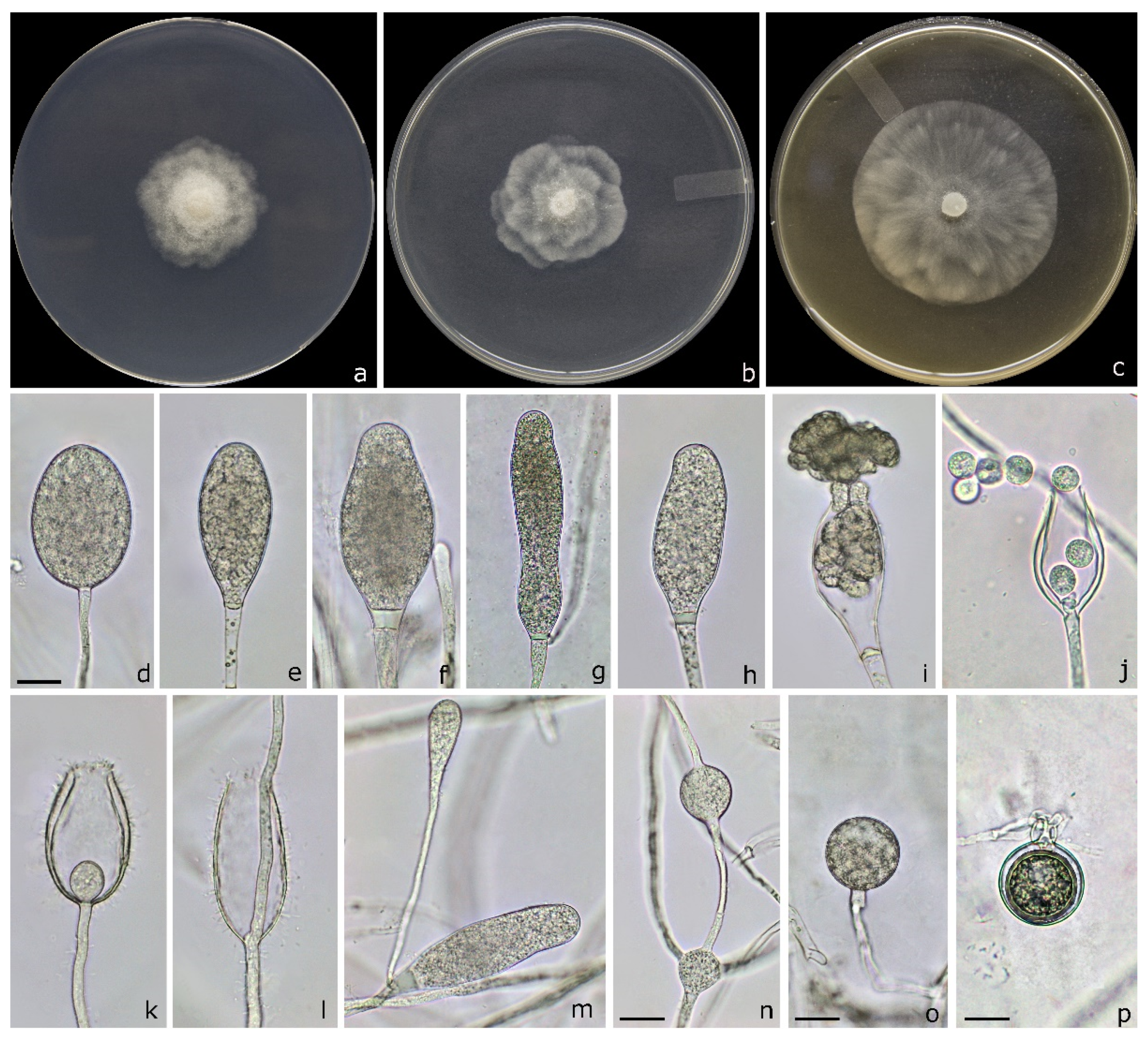
2.2. Morphological Identification and Characterization of Isolates
2.3. DNA Extraction, Polymerase Chain Reaction (PCR) Amplification, and Sequencing
2.4. Phylogenetic Analysis
2.5. Pathogenicity Tests
3. Results
3.1. Symptomatology and Aetiology
3.2. Phylogeny Clade 8
3.3. Taxonomy
3.4. Pathogenicity Tests
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Conflicts of Interest
References
- Mauro, R.; Portis, E.; Acquadro, A.; Lombardo, S.; Mauromicale, G.; Lanteri, S. Genetic diversity of globe artichoke landraces from Sicilian small-holdings: Implications for evolution and domestication of the species. Conserv. Genet. 2009, 10, 431–440. [Google Scholar] [CrossRef] [Green Version]
- Calabrese, N.; Guyon, C.; Jana, C.; Martinez, J.P.; Minisini, S.; Vargas, S. From field to fork, three case studies to understand the globe artichoke supply chain. Acta Hort. 2020, 1284, 181–188. [Google Scholar] [CrossRef]
- Basnizki, J.; Zohary, D. Breeding of seed-planted artichoke. Plant Breed Rev. 1994, 12, 253–269. [Google Scholar]
- Colelli, G.; Calabrese, N. Il Carciofo e il Cardo; Bayer: Bologna, Italy, 2009. [Google Scholar]
- Lattanzio, V.; Kroon, P.A.; Linsalata, V.; Cardinali, A. Globe artichoke: A functional food and source of nutraceutical ingredients. J. Funct. Food 2009, 1, 131–144. [Google Scholar] [CrossRef]
- Ceccarelli, N.; Curadi, M.; Picciarelli, P.; Martelloni, L.; Sbrana, C.; Giovannetti, M. Globe artichoke as a functional food. Mediterr. J. Nutr. Metab. 2010, 3, 197–201. [Google Scholar] [CrossRef]
- Christaki, E.; Bonos, E.; Florou-Paneri, P. Nutritional and functional properties of Cynara crops (globe artichoke and cardoon) and their potential applications: A review. Int. J. Appl. Sci. Technol. 2012, 2, 64–70. [Google Scholar]
- Zayed, A.; Serag, A.; Farag, M.A. Cynara cardunculus L.: Outgoing and potential trends of phytochemical, industrial, nutritive and medicinal merits. J. Funct. Foods 2020, 69, 103937. [Google Scholar] [CrossRef]
- Cardarelli, M.; Rouphael, Y.; Saccardo, F.; Colla, G. An innovative vegetative propagation system for large-scale production of globe artichoke transplants. Part I. Propagation system setup. HortTechnology 2005, 15, 812–816. [Google Scholar] [CrossRef] [Green Version]
- Pandino, G.; Barbagallo, R.N.; Lombardo, S.; Restuccia, C.; Muratore, G.; Licciardello, F.; Mazzaglia, A.; Ricceri, J.; Pesce, G.R.; Mauromicale, G. Quality traits of ready-to-use globe artichoke slices as affected by genotype, harvest time and storage time. Part I: Biochemical and physical aspects. LWT-Food Sci. Technol. 2017, 76, 181–189. [Google Scholar] [CrossRef]
- Bravi, R.; Spataro, G.; Crino, P.; Saccardo, F. L’istituzione del Registro Varietale del carciofo. Seme 2008, 2, 24–29. [Google Scholar]
- Mauro, R.P.; Lombardo, S.; Grazia Longo, A.M.; Pandino, G.; Mauromicale, G. New cropping designs of globe artichoke for industrial use. Ital. J. Agron. 2011, 6, 44–49. [Google Scholar]
- Mauromicale, G.; Ierna, A. Panorama varietale e miglioramento genetico del carciofo. Inf. Agr. 2000, 56, 39–45. [Google Scholar]
- Frau, A.; Mallica, G.; Baghino, L.; Cadinu, M.; Repetto, A. La micropropagazione del carciofo Spinoso sardo: Un valido mezzo per aumentare la produttività degli impianti. Italus Hortus 2004, 11, 38–41. [Google Scholar]
- Rizzo, V.; Lombardo, S.; Pandino, G.; Barbagallo, R.N.; Mazzaglia, A.; Restuccia, C.; Mauromicale, G.; Muratore, G. Shelf-life study of ready-to-cook slices of globe artichoke ‘Spinoso sardo’: Effects of anti-browning solutions and edible coating enriched with Foeniculum vulgare essential oil. J. Sci. Food Agric. 2019, 99, 5219–5228. [Google Scholar] [CrossRef] [PubMed]
- Bedini, L.; Lucchesini, M.; Bertozzi, F.; Graifenberg, A. Plant tissue cultures from four tuscan globe artichoke cultivars. Cent. Eur. J. Biol. 2012, 7, 680–689. [Google Scholar] [CrossRef] [Green Version]
- Ozsan, T.; Onus, A.N. Callogenesis optimization of some globe artichoke [Cynara cardunculus var. scolymus (L.) Fiori] cultivars based on in vivo and in vitro leaf explants. Not. Bot. Horti Agrobo. 2020, 48, 1873–1884. [Google Scholar]
- Ledda, L.; Mameli, M.G.; Milia, M.; Marras, G.F. Influence of plant shading and ovoli typology on globe artichoke development, early production and head atrophy: Preliminary results. Acta Hort. 2003, 660, 365–371. [Google Scholar] [CrossRef]
- Bianco, V.V.; Calabrese, N. Problemi e prospettive della coltivazione, impiego e commercializzazione del carciofo. Atti del Workshop “Orticoltura di pien’aria in Italia: Quali prospettive per il comparto?”. Sassari 2007, 16, 27–58. [Google Scholar]
- Bhat, R.G.; Subbarao, K.V.; Bari, M.A. First Report of Verticillium dahliae causing artichoke wilt in California. Plant Dis. 1999, 83, 782. [Google Scholar] [CrossRef]
- Armengol, J.; Berbegal, M.; Giménez-Jaime, A.; Romero, S.; Beltran, R.; Vicent, A.; Ortega, A.; García-Jiménez, J. Incidence of Verticillium wilt of artichoke in eastern Spain and role of inoculum sources on crop infection. Phytoparasitica 2005, 33, 397–405. [Google Scholar] [CrossRef]
- Cirulli, M.; Bubici, G.; Amenduni, M.; Armengol, J.; Berbegal, M.; Jiménez-Gasco, M.; Jiménez-Díaz, R.M. Verticillium Wilt: A threat to artichoke production. Plant Dis. 2010, 94, 1176–1187. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Marras, F. “I marciumi del colletto” del carciofo causati da Sclerotinia sclerotiorum (Lib.) Massee, dallo Sclerotium rolfsii Sac. e dalla Rhizoctonia solani Kuhn, in Sardegna. Studi Sass. Ann. Fac. Agr. Univ. Sassari 1962, 10, 90–103. [Google Scholar]
- Marras, F.; Foddai, A.; Corda, P. La difesa del carciofo dalle malattie crittogamiche. Inf. Fitopatol. 1985, 9, 19–24. [Google Scholar]
- Serra, S.; Maddau, L.; Franceschini, A.; Marras, F. Chemical and biological control of artichoke crown rot caused by Sclerotium rolfsii (Cynara scolymus L.). Italus Hortus 2004, 11, 74–77. [Google Scholar]
- Larran, S.; Ronco, L.; Carranza, M.R.; Zuccaro, M. Grey mould of the globe artichoke in Argentina. Austral. Plant Pathol. 2008, 33, 461. [Google Scholar] [CrossRef]
- Aydoğdu, M.; Kurbetli, İ.; Ozan, S. First report of Sclerotium rolfsii causing crown rot on globe artichoke in Turkey. Plant Dis. 2016, 100, 2161. [Google Scholar] [CrossRef]
- Corda, P.; Fiori, M.; Carta, C. Osservazioni sul “marciume dei capolini” del carciofo (Cynara scolymus L.) da Botrytis cinerea Pers. in Sardegna. Studi Sass. Ann. Fac. Agr. Univ. Sassari 1982, 16, 193–197. [Google Scholar]
- Ortega, A.; Perez, S. Aggressiveness of Verticillium dahliae isolates from potato to artichoke. Acta Hortic. 2007, 730, 407–412. [Google Scholar] [CrossRef]
- Deligios, P.A.; Tiloca, M.T.; Sulas, L.; Buffa, M.; Caraffini, S.; Doro, L.; Sanna, G.; Spanu, E.; Spissu, E.; Urracci, G.R.; et al. Stable nutrient flows in sustainable and alternative cropping systems of globe artichoke. Agron. Sustain. Dev. 2017, 37, 54. [Google Scholar] [CrossRef] [Green Version]
- Penalver, R.; Duranvila, N.; Lopez, M.M. Characterization and pathogenicity of bacteria from shoot tips of the globe artichoke (Cynara scolymus L). Ann. Appl. Biol. 1994, 125, 501–513. [Google Scholar] [CrossRef]
- Gallitelli, D.; Mascia, T.; Martelli, G.P. Viruses in artichoke. In Advances in Virus Research; Loebenstein, G., Lecoq, H., Eds.; Academic Press: Burlington, MA, USA, 2012; Volume 84, pp. 289–324. [Google Scholar]
- Minutillo, S.A.; Spanò, R.; Gallitelli, D.; Mascia, T. Simultaneous detection of 10 viruses in globe artichoke by a synthetic oligonucleotide-based DNA polyprobe. Eur. J. Plant Pathol. 2021, 160, 991–997. [Google Scholar] [CrossRef]
- Bregant, C.; Sanna, G.P.; Bottos, A.; Maddau, L.; Montecchio, L.; Linaldeddu, B.T. Diversity and pathogenicity of Phytophthora species associated with declining alder trees in Italy and description of Phytophthora alpina sp. nov. Forests 2020, 11, 848. [Google Scholar] [CrossRef]
- Linaldeddu, B.T.; Mulas, A.A.; Bregant, C.; Piras, G.; Montecchio, L. First Report of Phytophthora pistaciae causing root and collar rot on nursery plants of Pistacia lentiscus in Italy. Plant Dis. 2020, 104, 1564. [Google Scholar] [CrossRef]
- Bregant, C.; Mulas, A.A.; Rossetto, G.; Deidda, A.; Maddau, L.; Piras, G.; Linaldeddu, B.T. Phytophthora mediterranea sp. nov., a new species closely related to Phytophthora cinnamomi from nursery plants of Myrtus communis in Italy. Forests 2021, 12, 682. [Google Scholar] [CrossRef]
- Brasier, C.M. Observations on the sexual mechanism in Phytophthora palmivora and related species. Trans. Brit. Mycol. Soc. 1972, 58, 236–251. [Google Scholar] [CrossRef]
- Linaldeddu, B.T.; Maddau, L.; Franceschini, A.; Alves, A.; Phillips, A.J.L. Botryosphaeriaceae species associated with lentisk dieback in Italy and description of Diplodia insularis sp. nov. Mycosphere 2016, 7, 962–977. [Google Scholar] [CrossRef]
- White, T.J.; Bruns, T.; Lee, S.; Taylor, J. Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In PCR Protocols, a Guide to Methods and Applications; Innis, M.A., Gelfand, D.H., Sninsky, J.J., White, T.J., Eds.; Academic Press: San Diego, CA, USA, 1990; pp. 315–322. [Google Scholar]
- Kroon, L.P.N.M.; Bakker, F.T.; Van Den Bosch, G.B.M.; Bonants, P.J.M.; Flier, W.G. Phylogenetic analysis of Phytophthora species based on mitochondrial and nuclear DNA sequences. Fungal Genet. Biol. 2004, 41, 766–782. [Google Scholar] [CrossRef] [PubMed]
- Martin, F.N.; Tooley, P.W. Phylogenetic relationships among Phytophthora species inferred from sequence analysis of mitochondrially encoded cytochrome oxidase I and II genes. Mycologia 2003, 95, 269–284. [Google Scholar] [CrossRef]
- Thompson, J.D.; Gibson, T.J.; Plewniak, F.; Jeanmougin, F.; Higgins, D.G. The CLUSTAL_X windows interface: Flexible strategies for multiple sequence alignment aided by quality analysis tools. Nucleic Acids Res. 1997, 25, 4876–4882. [Google Scholar] [CrossRef] [Green Version]
- Hall, T.A. BioEdit: A user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucleic Acids Symp. Ser. 1999, 41, 95–98. [Google Scholar]
- Kumar, S.; Stecher, G.; Li, M.; Knyaz, C.; Tamura, K. MEGA X: Molecular Evolutionary Genetics Analysis across computing platforms. Mol. Biol. Evol. 2018, 35, 1547–1549. [Google Scholar] [CrossRef] [PubMed]
- Grünwald, N.; Werres, S.; Goss, E.M.; Taylor, C.R.; Fieland, V.J. Phytophthora obscura sp. nov., a new species of the novel Phytophthora subclade 8d. Plant Pathol. 2011, 61, 610–622. [Google Scholar] [CrossRef]
- Erwin, D.C.; Ribeiro, O.K. Phytophthora Diseases Worldwide; American Phytopathological Society: St. Paul, MN, USA, 1996. [Google Scholar]
- Granke, L.; Quesada-Ocampo, L.; Lamour, K.; Hausbeck, M. Advances in Research on Phytophthora capsici on Vegetable Crops in The United States. Plant Dis. 2012, 96, 1588–1600. [Google Scholar] [CrossRef] [Green Version]
- Bertier, L.; Brouwer, H.; de Cock, A.W.; Cooke, D.E.; Olsson, C.H.; Höfte, M. The expansion of Phytophthora clade 8b: Three new species associated with winter grown vegetable crops. Persoonia 2013, 31, 63–76. [Google Scholar] [CrossRef] [Green Version]
- Stanghellini, M.E.; Vilchez, M.; Kim, D.H.; Aguiar, J.L.; Armendariz, J. First report of root rot caused by Pythium aphanidermatum on artichoke. Plant Dis. 2000, 84, 811. [Google Scholar] [CrossRef] [PubMed]
- Grünwald, N.J.; Garbelotto, M.; Goss, E.M.; Heungens, K.; Prospero, S. Emergence of the sudden oak death pathogen Phytophthora ramorum. Trends Microbiol. 2012, 20, 131–138. [Google Scholar] [CrossRef] [PubMed]
- Brasier, C.M. Stimulation of sex organ formation in Phytophthora by antagonistic species of Trichoderma. I. The effect in vitro. New Phytol. 1975, 74, 183. [Google Scholar] [CrossRef]
- Jung, T.; Stukely, M.J.; Hardy, G.E.; White, D.; Paap, T.; Dunstain, W.A.; Burgess, T.I. Multiple new Phytophthora species from ITS Clade 6 associated with natural ecosystems in Australia: Evolutionary and ecological implications. Persoonia 2011, 26, 13–39. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Trionne, E.J.; Roth, L.F. Aerial infection of Chamaecyparis by Phytophthora lateralis. Plant Dis. Rep. 1957, 41, 211–215. [Google Scholar]
- Werres, S.; Marwitz, R.; In’t veld, W.A.M.; De Cock, A.W.A.M.; Bonants, P.J.M.; De Weerdt, M.; Themann, K.; Ilieva, E.; Baayen, R.P. Phytophthora ramorum sp. nov., a new pathogen on Rhododendron and Viburnum. Mycol. Res. 2001, 10, 1155–1165. [Google Scholar] [CrossRef]
- Donahoo, R.; Blomquist, C.L.; Thomas, S.L.; Moulton, J.K.; Cooke, D.E.; Lamour, K.H. Phytophthora foliorum sp. nov., a new species causing leaf blight of azalea. Mycol. Res. 2006, 110, 1309–1322. [Google Scholar] [CrossRef]
- Rizzo, D.M.; Garbelotto, M.; Davidson, J.M.; Slaughter, G.W.; Koike, S.T. Phytophthora ramorum as the cause of extensive mortality of Quercus spp. and Lithocarpus densiflorus in California. Plant Dis. 2002, 86, 205–214. [Google Scholar] [CrossRef] [Green Version]
- Blomquist, C.L.; Irving, T.; Osterbauer, N.; Reeser, P. Phytophthora hibernalis: A New pathogen on Rhododendron and evidence of cross amplification with two PCR detection assays for Phytophthora ramorum. Plant Health Prog. 2005, 6, 27. [Google Scholar] [CrossRef]
- Brasier, C.M.; Vettraino, A.M.; Chang, T.T.; Vannini, A. Phytophthora lateralis discovered in an old growth Chamaecyparis forest in Taiwan. Plant Pathol. 2010, 59, 595–603. [Google Scholar] [CrossRef]
- Brasier, C.M.; Webber, J. Sudden Larch Death. Nature 2010, 466, 824–825. [Google Scholar] [CrossRef]
- Scanu, B.; Linaldeddu, B.T.; Deidda, A.; Jung, T. Diversity of Phytophthora species from declining Mediterranean maquis vegetation, including two new species, Phytophthora crassamura and P. ornamentata sp. nov. PLoS ONE 2015, 10, e0143234. [Google Scholar] [CrossRef] [Green Version]
- Lehtijärvi, A.; Kaya, A.G.A.; Woodward, S.; Jung, T.; Lehtijärvi, H.T.D. Oomycota species associated with deciduous and coniferous seedlings in forest tree nurseries of Western Turkey. For. Pathol. 2017, 47, e12363. [Google Scholar] [CrossRef]
- Sims, L.L.; Chee, C.; Bourret, T.; Hunter, S.; Garbelotto, M. Genetic and phenotypic variation of Phytophthora crassamura isolates from California nurseries and restoration sites. Fungal Biol. 2019, 123, 159–169. [Google Scholar] [CrossRef]
- Brasier, C.M.; Cooke, D.E.L.; Duncan, J.M.; Hansen, E.M. Multiple new phenotypic taxa from trees and riparian ecosystems in Phytophthora gonapodyides–P. megasperma ITS Clade 6, which tend to be high-temperature tolerant and either inbreeding or sterile. Mycol. Res. 2003, 107, 277–290. [Google Scholar] [CrossRef] [PubMed]
- Benson, D.M.; Jones, R.K. Diseases of Woody Ornamentals and Trees in Nurseries; APS Press: St. Paul, MN, USA, 2001. [Google Scholar]
- Pánek, M.; Fér, T.; Mrácek, J.; Tomsovský, M. Evolutionary relationships within the Phytophthora cactorum species complex in Europe. Fungal Biol. 2016, 120, 836–851. [Google Scholar] [CrossRef]
- Burgess, T.I.; White, D.; McDougall, K.M.; Garnas, J.; Dunstan, W.A.; Català, S.; Carnegie, A.J.; Worboys, S.; Cahill, D.; Vettraino, A.M.; et al. Distribution and diversity of Phytophthora across Australia. Pac. Conserv. Biol. 2017, 23, 150–162. [Google Scholar] [CrossRef] [Green Version]
- Ellis, M.A.; Madden, L.V. Leather Rot. Pages 33-35 in: Compendium of Strawberry Diseases, 2nd ed.; Maas, J.L., Ed.; The American Phytopathological Society: St. Paul, MN, USA, 1998. [Google Scholar]
- Hantula, J.; Lilja, A.; Nuorteva, H.; Parikka, P.; Werres, S. Pathogenicity, morphology and genetic variation of Phytophthora cactorum from strawberry, apple, rhododendron, and silver birch. Mycol. Res. 2000, 104, 1062–1068. [Google Scholar] [CrossRef]
- Grünwald, N.J.; Flier, W.G. The biology of Phytophthora infestans at its center of origin. Annu. Rev. Phytopathol. 2005, 43, 171–190. [Google Scholar] [CrossRef] [PubMed]
- Evans, T.A.; Mulrooney, R.P.; Gregory, N.F.; Kee, E. Lima bean downy mildew: Impact, etiology, and management strategies for Delaware and the Mid-Atlantic Region U.S. Plant Dis. 2007, 91, 128–135. [Google Scholar] [CrossRef]
- Linaldeddu, B.T.; Bregant, C.; Montecchio, L.; Favaron, F.; Sella, L. First report of Phytophthora acerina, P. pini and P. plurivora causing root rot and sudden death on olive trees in Italy. Plant Dis. 2020, 104, 996. [Google Scholar] [CrossRef]
- Linaldeddu, B.T.; Bregant, C.; Ruzzon, B.; Montecchio, L. Coniella granati and Phytophthora palmivora the main pathogens involved in pomegranate dieback and mortality in north-eastern Italy. Ital. J. Mycol. 2020, 49, 92–100. [Google Scholar]
- Decloquement, J.; Ramos-Sobrinho, R.; Elias, S.G.; Britto, D.S.; Puig, A.S.; Reis, A.; da Silva, R.A.F.; Honorato-Júnior, J.; Luz, E.D.M.N.; Pinho, D.B.; et al. Phytophthora theobromicola sp. nov.: A new species causing black pod disease on cacao in Brazil. Front. Microbiol. 2021, 12, 486. [Google Scholar] [CrossRef]
- Lawrence, S.A.; Robinson, H.F.; Furkert, D.P.; Brimble, M.A.; Gerth, M.L. Screening a natural product-inspired library for anti-Phytophthora activities. Molecules 2021, 26, 1819. [Google Scholar] [CrossRef] [PubMed]
- Scott, P.; Bader, M.K.F.; Burgess, T.I.; Hardy, G.; Williams, N. Global biogeography and invasion risk of the plant pathogen genus Phytophthora. Environ. Sci. Policy 2019, 101, 175–182. [Google Scholar] [CrossRef]
- Portis, E.; Mauromicale, G.; Barchi, L.; Mauro, R.; Lanteri, S. Population structure and genetic variation in autochthonous globe artichoke germplasm from Sicily Island. Plant Sci. 2005, 168, 1591–1598. [Google Scholar] [CrossRef]
- Cadinu, M.; Baghino, L.; Mallica, G.; Pisanu, A.B.; Repetto, A.; Tatti, A.M.; Muntoni, M. Collection of artichoke germplasm from different Mediterranean regions. Int. Symp. Artichoke Cardoon Wild Relat. 2011, 942, 103–108. [Google Scholar] [CrossRef]



| Farm | Cultivar | Origin of Plants | Planting Period | Sampling Period | Number of Samples |
|---|---|---|---|---|---|
| F1 | Romanesco | Micropropagated | July 2019 | February 2020 | 3 |
| F2 | Romanesco | Micropropagated | July 2019 | February 2020 | 3 |
| F3 | Romanesco | Micropropagated | July 2019 | February 2020 | 1 |
| F4 | Romanesco | Micropropagated | July 2019 | July 2020 | 1 |
| F5 | Romanesco | Micropropagated | July 2019 | July 2020 | 1 |
| F6 | Romanesco | Micropropagated | July 2019 | July 2020 | 1 |
| F7 | Romanesco | Micropropagated | July 2020 | February 2021 | 3 |
| F8 | Romanesco | Micropropagated | July 2020 | February 2021 | 3 |
| F9 | Romanesco | Micropropagated | July 2020 | February 2021 | 3 |
| Species | Code | Host | GenBank Accession Number | ||
|---|---|---|---|---|---|
| ITS | Btub | cox1 | |||
| P. austrocedrae | CBS 122.911 | Austrocedrus chilensis | MG783380 | - | - |
| P. austrocedrae | AG318 | A. chilensis | KU953997 | - | - |
| P. brassicae | CBS 179.87 | Brassica oleracea | NR147859 | - | - |
| P. brassicae | CBS 112277 | B. oleracea | KC478766 | - | - |
| P. cichorii | CBS 115029 | Cychorium intybus | KC478773 | - | - |
| P. cichorii | CBS 115030 | C. intybus | KC478775 | - | - |
| P. cryptogea | CBS 113.19 | n/d | MG865483 | - | - |
| P. cryptogea | SCRP 205 | Solanum tuberosum | AY659423 | - | - |
| P. dauci | CBS 127102 | Daucus carota | MH620160 | - | - |
| P. dauci | CBS 114039 | D. carota | KC478762 | - | - |
| P. drechsleri | CBS 292.35 | Beta vulgaris | MG865484 | - | - |
| P. drechsleri | LEV 6732 | Petunia × hybrida | MH025882 | - | - |
| P. erythroseptica | CBS 129.23 | Solanum tuberosum | MG865486 | - | - |
| P. erythroseptica | MUC 837 | S. tuberosum | MG196242 | - | - |
| P. foliorum | CBS 121655 | Rhododendron sp. | MG865492 | MH493937 | MH136889 |
| P. foliorum | FC160118 | R. ponticon | KX364273 | - | - |
| P. foliorum | P10969 | Rhododendron sp. | HQ261561 | - | HQ261308 |
| P. hibernalis | CBS 114104 | Citrus sp. | MG865506 | MH493948 | MH136900 |
| P. hibernalis | CBS 270.31 | Citrus sinensis | KT183039 | KX252120 | - |
| P. hibernalis | 116-0007 | n/d | MF441623 | - | MF441668 |
| P. lactucae | P19872 | Lactuca sativa | MH620161 | - | - |
| P. lactucae | BPIC1988 | L. sativa | KC478770 | - | - |
| P. lateralis | CBS 168.42 | Chamaecyparis sp. | MG865522 | MH493964 | MH136917 |
| P. lateralis | P2522 | C. lawsoniana | JQ837654 | JQ837665 | JQ837677 |
| P. marrasii | CB150 | Cynara scolymus | MZ569854 | MZ603724 | OK054535 |
| P. marrasii | CB149 | C. scolymus | MZ569855 | MZ603723 | OK054536 |
| P. medicaginis | CBS 119902 | Medicago sativa | MG865532 | - | - |
| P. medicaginis | 23A4 | M. sativa | KF358223 | - | - |
| P. obscura | CBS 129273 | Soil | MG865554 | - | - |
| P. obscura | 6.18 | Acer pseudoplatanus | MN228692 | - | - |
| P. porri | CBS116662 | Allium porrum | KC478749 | - | - |
| P. porri | CBS127099 | A. porrum | KC478750 | - | - |
| P. primulae | CBS 114346 | Primula polyantha | MG865571 | - | - |
| P. primulae | CBS110162 | Primula sp. | HQ643324 | - | - |
| P. pseudocryptogea | CBS 139749 | Isopogon buxifolius | KP288376 | - | - |
| P. pseudocryptogea | SUC 633 | Solanum melongena | KP099906 | - | - |
| P. ramorum | CBS 101553 | Rhododendron sp. | MG865581 | LC595884 | MH136973 |
| P. ramorum | P2058 | Rhododendron sp. | MW300319 | MW323707 | MW323671 |
| P. sansomeana | CBS 117693 | Glycine max | MG865585 | - | - |
| P. sansomeana | KSSO 6-47 | G. max | KU209481 | - | - |
| P. syringae | CBS 110161 | Syringa vulgaris | MG865590 | - | - |
| P. syringae | CBS 364.52 | Prunus armeniaca | HQ643358 | - | - |
| P. trifolii | CBS 117687 | Trifolium sp. | MG865594 | - | - |
| P. trifolii | P1462 | Trifolium sp. | HQ261719 | - | - |
| Scheme | Isolate Code | GenBank Accession Number | Disease Severity * | Re-Isolation Frequency (%) |
|---|---|---|---|---|
| P. cactorum | CB266 | MZ569852 | 2.9 ± 0.3 | 100% |
| P. crassamura | CB267 | MZ569853 | 2.5 ± 0.5 | 100% |
| P. marrasii | CB150 | MZ569854 | 1.9± 0.6 | 100% |
| Control | - | - | 1 (asymptomatic) | - |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bregant, C.; Rossetto, G.; Deidda, A.; Maddau, L.; Franceschini, A.; Ionta, G.; Raiola, A.; Montecchio, L.; Linaldeddu, B.T. Phylogeny and Pathogenicity of Phytophthora Species Associated with Artichoke Crown and Root Rot and Description of Phytophthora marrasii sp. nov. Agriculture 2021, 11, 873. https://doi.org/10.3390/agriculture11090873
Bregant C, Rossetto G, Deidda A, Maddau L, Franceschini A, Ionta G, Raiola A, Montecchio L, Linaldeddu BT. Phylogeny and Pathogenicity of Phytophthora Species Associated with Artichoke Crown and Root Rot and Description of Phytophthora marrasii sp. nov. Agriculture. 2021; 11(9):873. https://doi.org/10.3390/agriculture11090873
Chicago/Turabian StyleBregant, Carlo, Giovanni Rossetto, Antonio Deidda, Lucia Maddau, Antonio Franceschini, Giorgio Ionta, Alessandro Raiola, Lucio Montecchio, and Benedetto T. Linaldeddu. 2021. "Phylogeny and Pathogenicity of Phytophthora Species Associated with Artichoke Crown and Root Rot and Description of Phytophthora marrasii sp. nov." Agriculture 11, no. 9: 873. https://doi.org/10.3390/agriculture11090873
APA StyleBregant, C., Rossetto, G., Deidda, A., Maddau, L., Franceschini, A., Ionta, G., Raiola, A., Montecchio, L., & Linaldeddu, B. T. (2021). Phylogeny and Pathogenicity of Phytophthora Species Associated with Artichoke Crown and Root Rot and Description of Phytophthora marrasii sp. nov. Agriculture, 11(9), 873. https://doi.org/10.3390/agriculture11090873








