Responses of Ammonia-Oxidizing Microorganisms to Intercropping Systems in Different Seasons
Abstract
1. Introduction
2. Materials and Methods
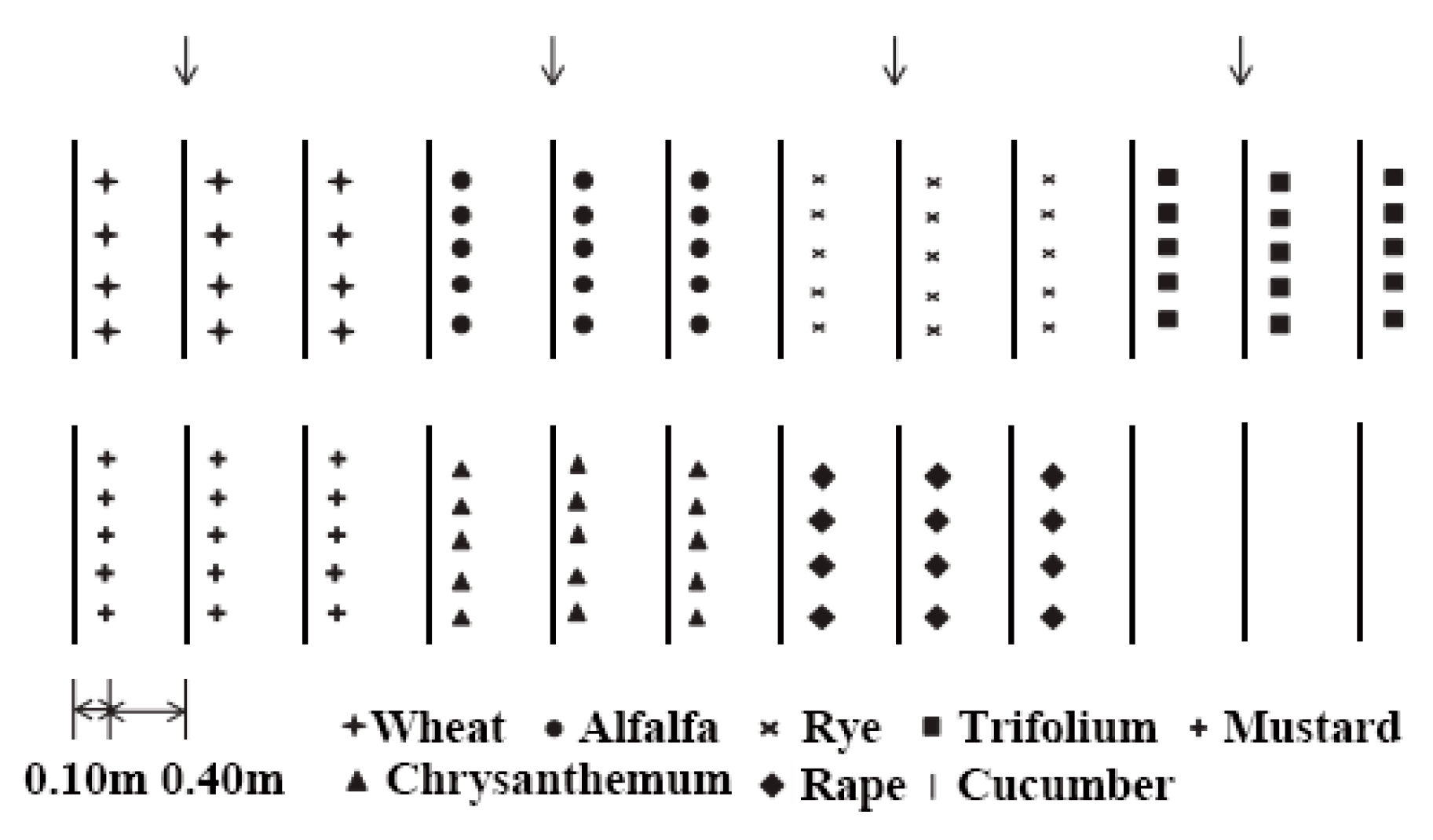
2.1. Experimental Location and Design
2.2. Soil Sampling and Analysis
2.3. Potential Nitrification Rate (PNR)
2.4. DNA Extraction and Quantitative PCR (qPCR)
2.5. Illumina MiSeq Sequencing and Data Processing
2.6. Statistical Analysis
3. Results
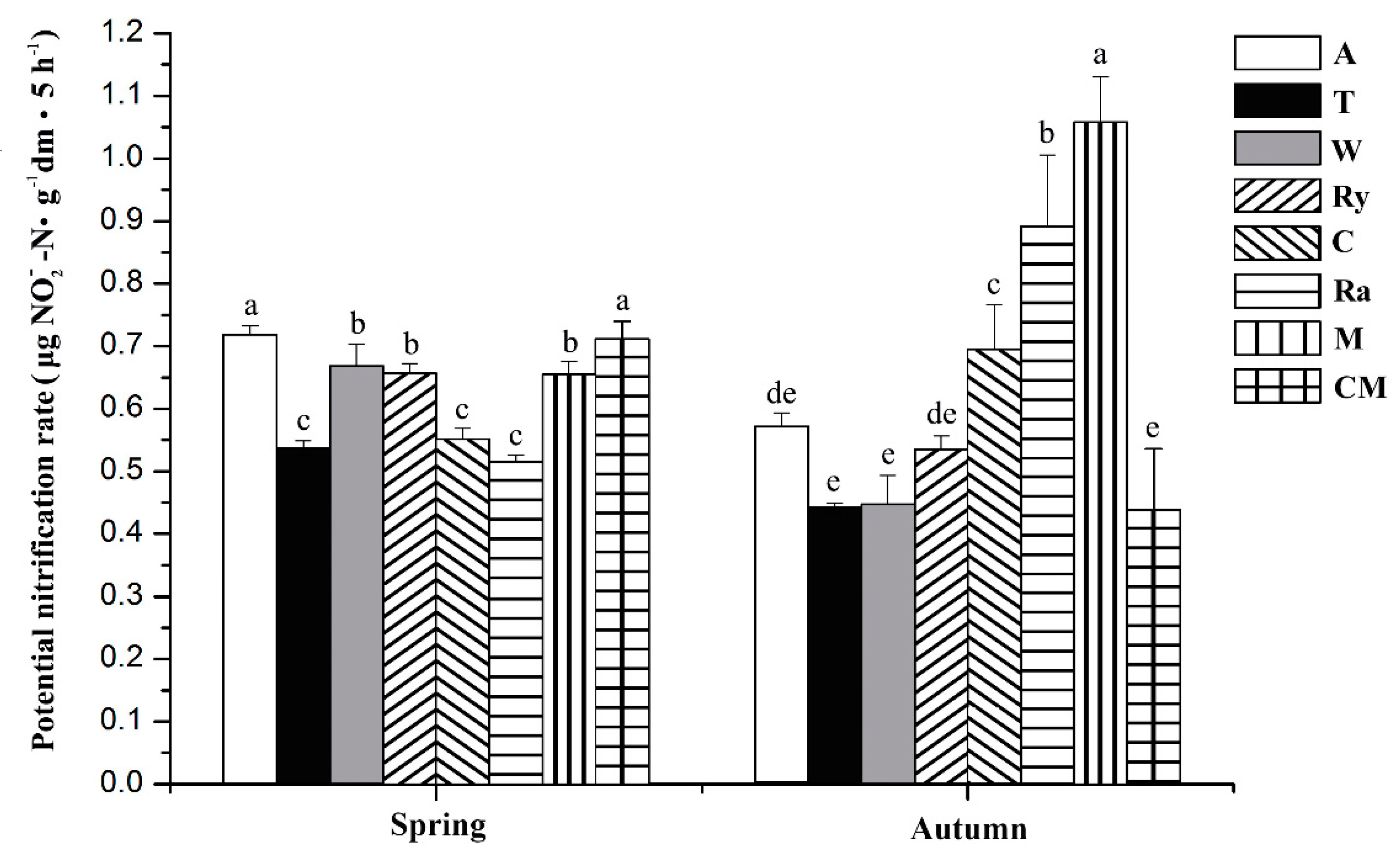
3.1. Soil Physical and Chemical Properties and PNR
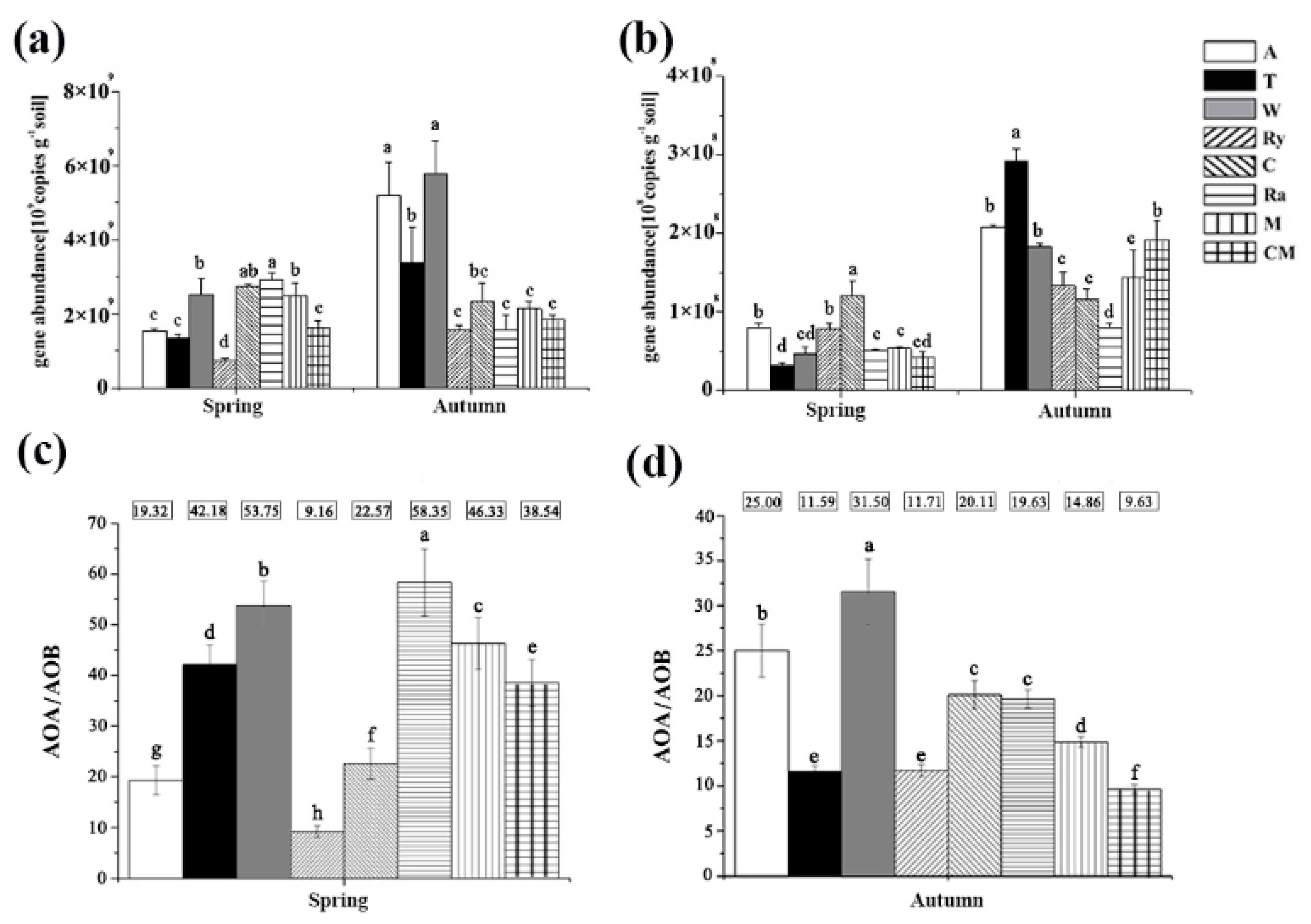
3.2. Soil AOA and AOB Abundances
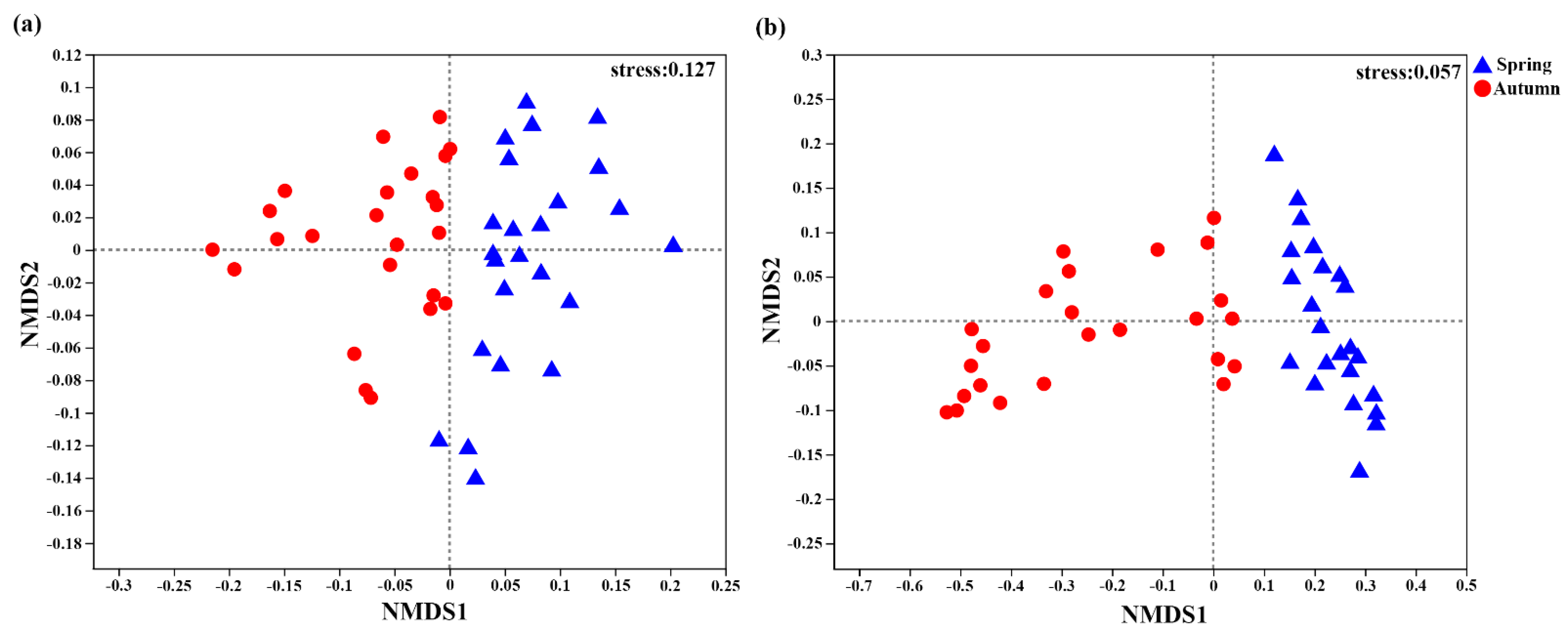
3.3. Soil AOA and AOB Community Alpha and Beta Diversities
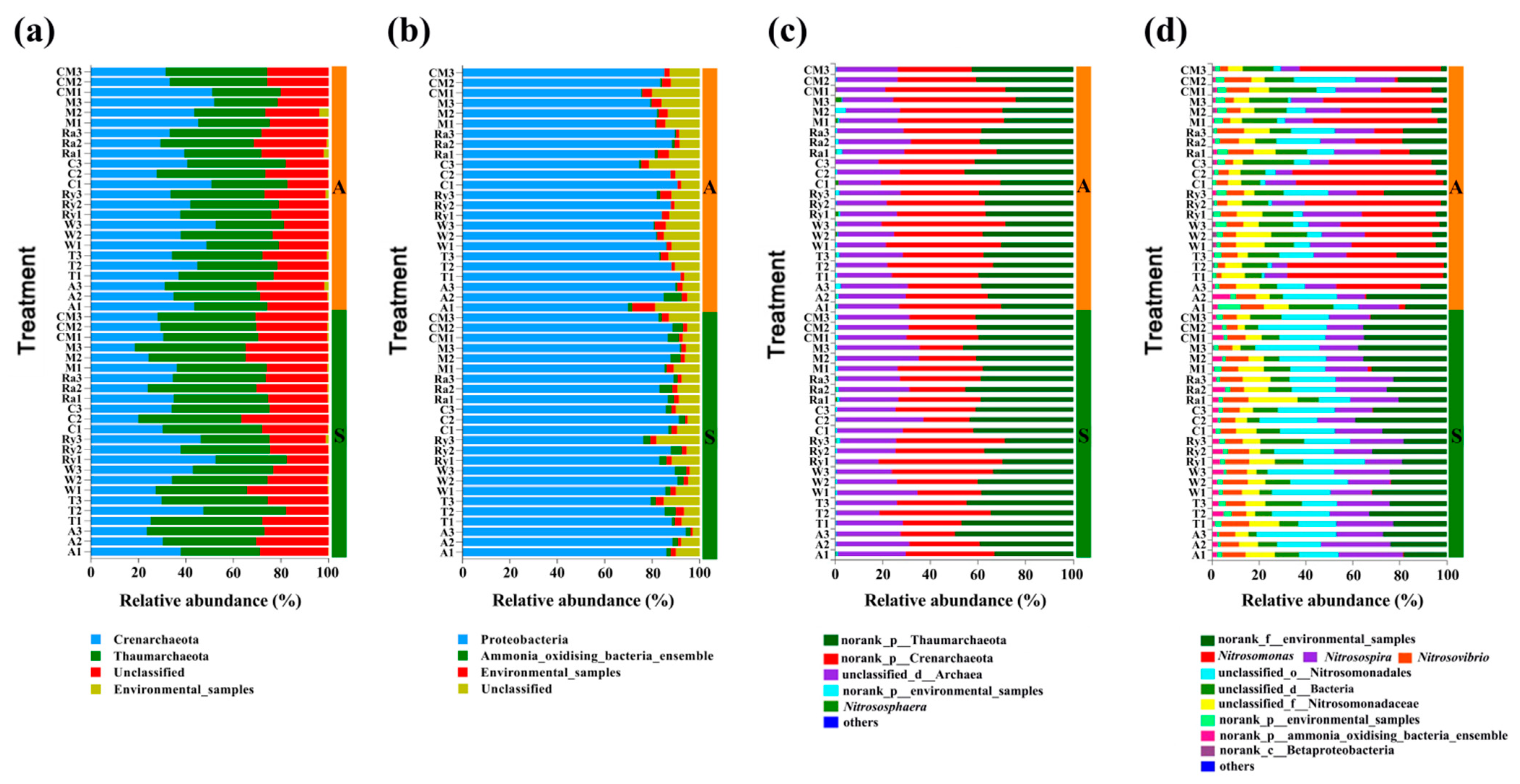
3.4. Soil AOA and AOB Community Composition
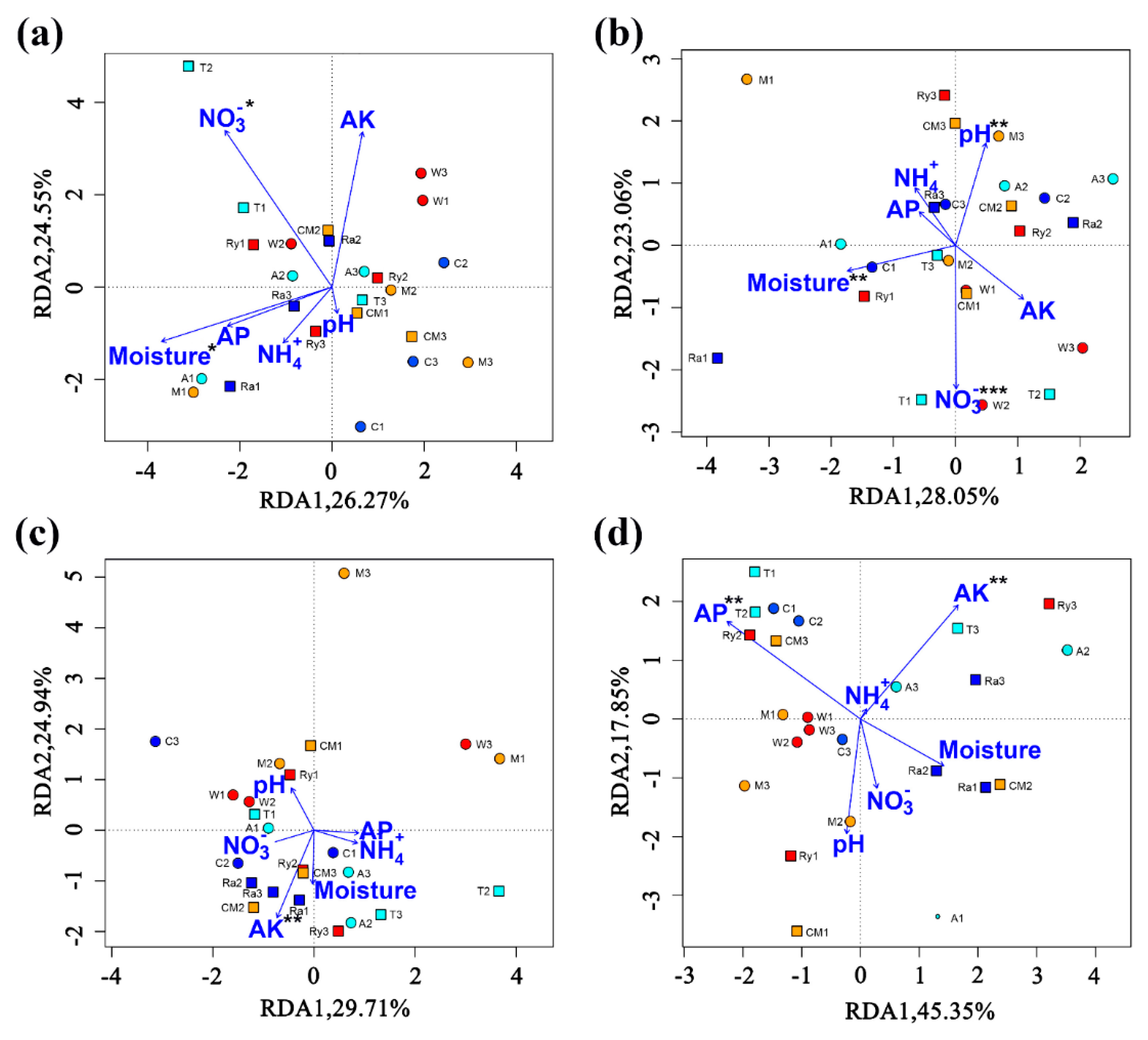
3.5. Relationships between Soil AOA and AOB Communities and Soil Physical and Chemical Properties
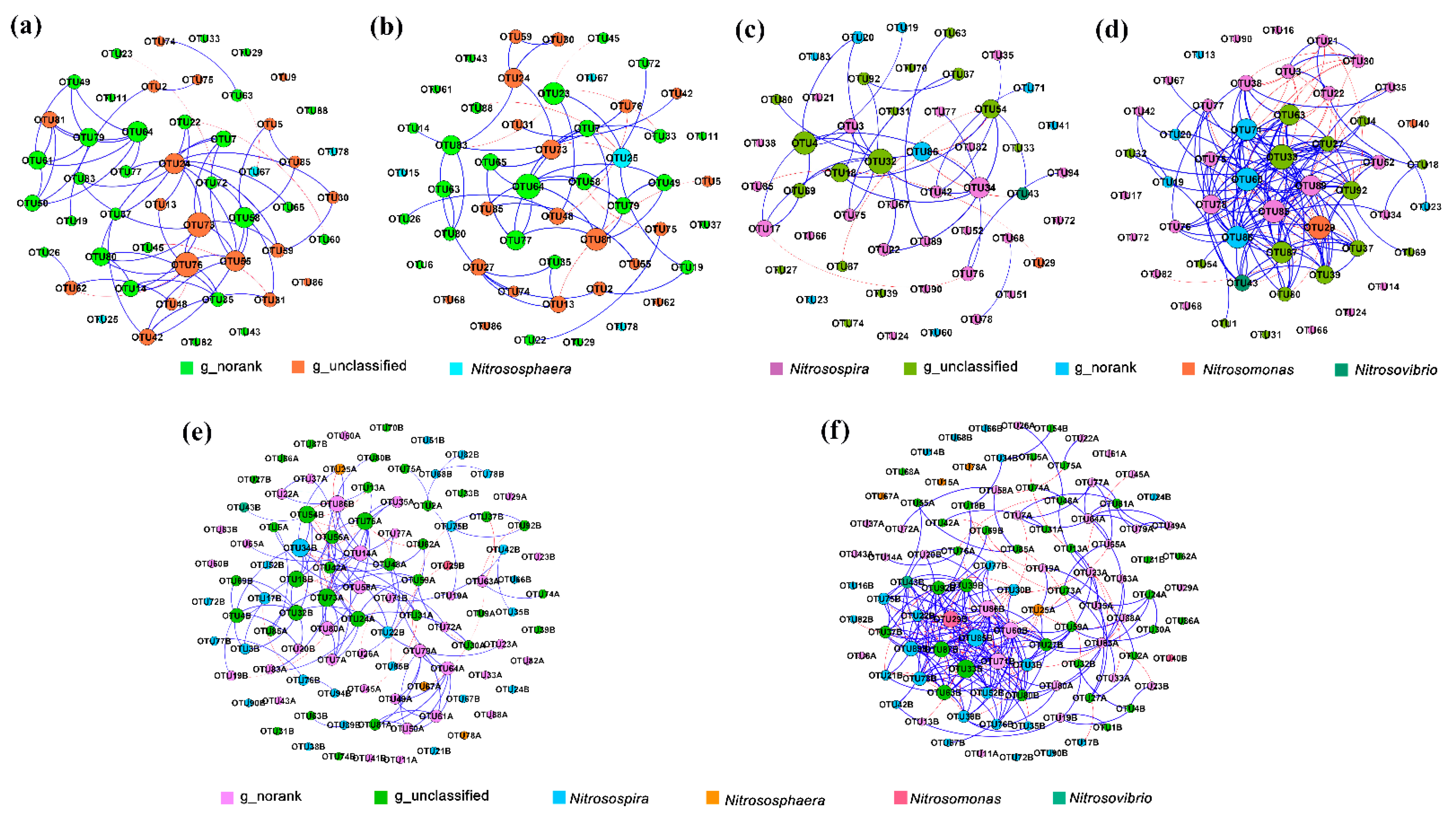
3.6. Co-Occurrence Network Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Fan, F.L.; Yang, Q.B.; Li, Z.J.; Wei, D.; Cui, X.A.; Liang, Y.C. Impacts of organic and inorganic fertilizers on nitrification in a cold climate soil are linked to the bacterial ammonia oxidizer community. Microb. Ecol. 2011, 62, 982–990. [Google Scholar] [CrossRef] [PubMed]
- Li, N.H.; Gao, D.M.; Zhou, X.G.; Chen, S.C.; Li, C.X.; Wu, F.Z. Intercropping with Potato-Onion Enhanced the Soil Microbial Diversity of Tomato. Microorganisms 2020, 8, 834. [Google Scholar] [CrossRef] [PubMed]
- Zhao, M.; Jones, C.M.; Meijer, J.; Lundquista, P.O.; Franssonb, P.; Carlssonc, G.; Hallin, S. Intercropping affects genetic potential for inorganic nitrogen cycling by root -associated microorganisms in Medicago sativa and Dactylis glomerata. Appl. Soil Ecol. 2017, 119, 260–266. [Google Scholar] [CrossRef]
- Chu, H.Y.; Fujii, T.; Morimoto, S.; Lin, X.G.; Yagi, K. Population size and specific nitrification potential of soil ammonia-oxidizing bacteria under long-term fertilizer management. Soil Biol. Biochem. 2008, 40, 1960–1963. [Google Scholar] [CrossRef]
- Philippot, L.; Hallin, S. Towards food, feed and energy crops mitigating climate change. Trends Plant Sci. 2011, 16, 476–480. [Google Scholar] [CrossRef]
- Yao, H.Y.; Gao, Y.M.; Nicol, G.W.; Campbell, C.D.; Prosser, J.I.; Zhang, L.M. Links between ammonia oxidizer community structure, abundance, and nitrification potential in acidic soil. Appl. Environ. Microbiol. 2011, 77, 4618–4625. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y.L.; Liu, X.R.; Zhan, Q.W. Effects of combined biochar and organic fertilizer on nitrous oxide fluxes and the related nitrifier and denitrifier communities in a saline-alkali soil. Sci. Total Environ. 2019, 686, 199–211. [Google Scholar] [CrossRef]
- Zhang, N.N.; Sun, Y.M.; Wang, E.T.; Yang, J.S.; Yuan, H.L.; Scow, K.M. Effects of intercropping and Rhizobial inoculation on the ammonia-oxidizing microorganisms in rhizospheres of maize and faba bean plants. Appl. Soil Ecol. 2015, 85, 76–85. [Google Scholar] [CrossRef]
- Ding, Y.; Huang, X.; Li, Y.; Liu, H.Y.; Zhang, Q.C.; Liu, X.M. Nitrate leaching losses mitigated with intercropping of deep-rooted and shallow-rooted plants. J. Soils Sediments 2021, 21, 364–375. [Google Scholar] [CrossRef]
- Chen, Y.L.; Hu, H.W.; Han, H.Y.; Du, Y.; Wn, S.Q.; Xu, Z.W.; Chen, B.D. Abundance and community structure of ammonia-oxidizing Archaea and Bacteria in response to fertilization and mowing in a temperate steppe in Inner Mongolia. FEMS Microbiol. Ecol. 2014, 89, 67–79. [Google Scholar] [CrossRef]
- Hu, H.; Zhang, L.M.; Yuan, C.; Zheng, Y.; Wang, J.; Chen, D.; He, J. The large-scale distribution of ammonia oxidizers in paddy soils is driven by soil pH, geographic distance, and climatic factors. Front. Microbiol. 2015, 6, 938. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Liu, X.; Li, Y.; Ran, Y.; Liu, Y.; Zhang, Q. High temperatures inhibited the growth of soil bacteria and archaea but not that of fungi and altered nitrous oxide production mechanisms from different nitrogen sources in an acidic soil. Soil Biol. Biochem. 2017, 107, 168–179. [Google Scholar] [CrossRef]
- Ming, H.X.; Zhang, H.Z.; Chen, Q.R.; Wang, Y.T.; Su, J.; Zhao, X.H. Abundance and community structure of ammonium monooxygenase (amoA) genes in the wet season of liaohe estuary sediments. Cont. Shelf Res. 2020, 209, 10425. [Google Scholar] [CrossRef]
- Yang, Y.D.; Ren, Y.F.; Wang, X.Q.; Hu, Y.G.; Wang, Z.M.; Zeng, Z.H. Ammonia-oxidizing archaea and bacteria responding differently to fertilizer type and irrigation frequency as revealed by Illumina Miseq sequencing. J. Soil Sediment 2018, 18, 1029–1040. [Google Scholar] [CrossRef]
- Pereira e Silva, M.C.; Poly, F.; Guillaumaud, N.; van Elsas, J.D.; Salles, J.F. Fluctuations in ammonia oxidizing communities across agricultural soils are driven by soil structure and pH. Front. Microbiol. 2012, 3, 77. [Google Scholar] [CrossRef]
- Zhang, M.M.; Alves, R.J.E.; Zhang, D.D.; Han, L.L.; He, J.Z.; Zhang, L.M. Time-dependent shifts in populations and activity of bacterial and archaeal ammonia oxidizers in response to liming in acidic soils. Soil Biol. Biochem. 2017, 112, 77–89. [Google Scholar] [CrossRef]
- Verhamme, D.T.; Prosser, J.I.; Nicol, G.W. Ammonia concentration determines differential growth of ammonia-oxidising archaea and bacteria in soil microcosms. ISME J. 2011, 5, 1067–1071. [Google Scholar] [CrossRef]
- Li, J.L.; Nedwell, D.B.; Beddow, J.; Dumbrell, A.J.; McKew, B.A.; Thorpe, E.L. amoA gene abundances and nitrification potential rates suggest that benthic ammonia-oxidizing bacteria and not archaea dominate N cycling in the Colne Estuary, United Kingdom. Appl. Environ. Microbiol. 2015, 81, 159–165. [Google Scholar] [CrossRef] [PubMed]
- Di, H.J.; Cameron, K.C.; Shen, J.P.; Winefield, C.S.; O’Callaghan, M.; Bowatte, S. Nitrification driven by bacteria and not archaea in nitrogen-rich grassland soils. Nat. Geosci. 2009, 2, 621–624. [Google Scholar] [CrossRef]
- Shen, J.P.; Zhang, L.M.; Di, H.J.; He, J.Z. A review of ammonia-oxidizing bacteria and archaea in Chinese soils. Front. Microbiol. 2012, 3, 296–303. [Google Scholar] [CrossRef]
- Van Kessel, M.A.H.J.; Speth, D.R.; Albertsen, M.; Nielsen, P.H.; Op den Camp, H.J.M.; Kartal, B. Complete nitrification by a single microorganism. Nature 2015, 528, 555–559. [Google Scholar] [CrossRef]
- Daims, H.; Lebedeva, E.V.; Pjevac, P.; Han, P.; Herbold, C.; Albertsen, M. Complete nitrifi-cation by Nitrospira bacteria. Nature 2015, 528, 504–509. [Google Scholar] [CrossRef]
- Meng, H.; Katayama, Y.; Gu, J.D. More wide occurrence and dominance of ammonia-oxidizing archaea than bacteria at three Angkor sandstone temples of Bayon, Phnom Krom and Wat Athvea in Cambodia. Int. Biodeterior. Biodegrad. 2017, 117, 78–88. [Google Scholar] [CrossRef]
- Tang, H.M.; Xiao, X.P.; Li, C.; Shi, L.H.; Cheng, K.K.; Wen, L. Influences of different manure N input on soil ammonia-oxidizing archaea and bacterial activity and community structure in a double-cropping rice field. J. Appl. Microbiol. 2020. [Google Scholar] [CrossRef]
- Fu, X.; Wu, X.; Zhou, X.; Liu, S.; Shen, Y.; Wu, F. Companion cropping with potato onion enhances the disease resistance of tomato against Verticillium dahliae. Front. Plant Sci. 2015, 6, 726. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Wu, F.; Zhou, X.; Fu, X.; Tao, Y.; Xu, W.; Pan, K.; Liu, S. Effects of Intercropping with Potato Onion on the Growth of Tomato and Rhizosphere Alkaline Phosphatase Genes Diversity. Front. Plant Sci. 2016, 7, 846. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.L.; Fu, X.P.; Zhou, X.G.; Guo, M.Y.; Wu, F.Z. Effects of seven different companion plants on cucumber productivity, soil chemical characteristics and Pseudominas community. J. Integr. Agric. 2017, 16, 2206–2214. [Google Scholar] [CrossRef]
- Li, S.; Wu, F.Z. Diversity and Co-occurrence patterns of soil bacterial and fungal communities in seven intercropping systems. Front. Microbiol. 2018, 9, 1521. [Google Scholar] [CrossRef]
- Schinner, F.; Öhliger, R.; Kandeler, E.; Margesin, R. Methods in Soil Biology; Springer: Berlin/Heidelberg, Germany, 1996. [Google Scholar]
- Francis, C.A.; Roberts, K.J.; Beman, J.M.; Santoro, A.E.; Oakley, B.B. Ubiquity and diversity of ammonia-oxidizing archaea in water columns and sediments of the ocean. Proc. Natl. Acad. Sci. USA 2005, 102, 14683–14688. [Google Scholar] [CrossRef]
- Rotthauwe, J.H.; Witzel, K.P.; Liesack, W. The ammonia monooxygenase structural gene amoA as a functional marker: Molecular fine-scale analysis of natural ammonia-oxidizing populations. Appl. Environ. Microbiol. 1997, 63, 4704–4712. [Google Scholar] [CrossRef]
- Liu, J.N.; Xu, H.S.; Jiang, Y.; Zhang, K.; Hu, Y.G.; Zeng, Z.H. Methane emissions and microbial communities as influenced by dual cropping of Azolla along with early rice. Sci. Rep. 2017, 7, 40635. [Google Scholar] [CrossRef] [PubMed]
- Edgar, R.C. UPARSE: Highly accurate OTU sequences from microbial amplicon reads. Nat. Methods 2013, 10, 996–998. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Ni, L.; Song, Y.; Rhodes, G.; Li, J.; Huang, Q.; Shen, Q. Dynamic response of ammonia-oxidizers to four fertilization regimes across a wheat-rice rotation system. Front. Microbiol. 2017, 8, 630. [Google Scholar] [CrossRef]
- Caporaso, J.G.; Kuczynski, J.; Stombaugh, J.; Bittinger, K.; Bushman, F.D.; Costello, E.K.; Fierer, N.; Pena, A.G.; Goodrich, J.K.; Gordon, J.I. QIIME allows analysis of high-throughput community sequencing data. Nat. Methods 2010, 7, 335–336. [Google Scholar] [CrossRef] [PubMed]
- Cong, W.F.; Hoffland, E.; Li, L.; Six, J.; Sun, J.H.; Bao, X.G. Intercropping enhances soil carbon and nitrogen. Glob. Chang. Biol. 2015, 21, 1715–1726. [Google Scholar] [CrossRef] [PubMed]
- Yu, H.J.; Chen, S.C.; Zhou, X.G.; Wu, F.Z. Root interactions and tomato growth in tomato/potato onion companion-cropping system under different phosphorus levels. J. Plant Interact. 2017, 12, 438–466. [Google Scholar] [CrossRef]
- Bedoussac, L.; Justes, E. The efficiency of a durum wheat-winter pea intercrop to improve yield and wheat grain protein concentration depends on N availability during early growth. Plant Soil 2010, 330, 19–35. [Google Scholar] [CrossRef]
- Sun, R.; Myrold, D.D.; Wang, D.Z.; Guo, X.S.; Chu, H.Y. AOA and AOB communities respond differently to changes of soil pH under long-term fertilization. Soil Ecol. Lett. 2019, 1, 126–135. [Google Scholar] [CrossRef]
- Lawson, A.; Fortuna, A.M.; Cogger, C.; Bary, A.; Stubbs, T. Nitrogen contribution of rye-hairy vetch cover crop mixtures to organically grown sweet corn. Renew. Agric. Food Syst. 2013, 28, 59–69. [Google Scholar] [CrossRef]
- Taylor, A.E.; Zeglin, L.H.; Wanzek, T.A.; Myrold, D.D.; Bottomley, P.J. Dynamics of ammonia-oxidizing archaea and bacteria populations and contributions to soil nitrification potentials. ISME J. 2012, 6, 2024–2032. [Google Scholar] [CrossRef]
- Goldberg, D.E. Competitive ability: Definitions, contingency and correlated traits. Philos. Trans. R. Soc. B 1996, 351, 1377–1385. [Google Scholar]
- Zhang, S.; Lamb, E.G. Plant competitive ability and the transitivity of competitive hierarchies change with plant age. Plant Ecol. 2011, 213, 15–23. [Google Scholar] [CrossRef]
- Könneke, M.; Bernhard, A.E.; Torre, J.R.; Walker, C.B.; Waterbury, J.B.; Stahl, D.A. Isolation of an autotrophic ammonia-oxidizing marine archaeon. Nature 2005, 437, 543–546. [Google Scholar] [CrossRef] [PubMed]
- Leininger, S.; Urich, T.; Schloter, M.; Schwark, L.; Qi, J.; Nicol, G.W. Archaea predominate among ammonia-oxidizing prokaryotes in soils. Nature 2006, 442, 806–809. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Z.F.; Shi, X.J.; Zheng, Y.; Qin, Z.X.; Xie, D.T.; Li, Z.L. Abundance and community composition of ammonia-oxidizing bacteria and archaea in purple soil under long-term fertilization. Eur. J. Soil Biol. 2014, 60, 24–33. [Google Scholar] [CrossRef]
- Wang, Y.F.; Feng, Y.Y.; Ma, X.J.; Gu, J.D. Seasonal dynamics of ammonia/ammonium-oxidizing prokaryotes in oxic and anoxic wetland sediments of subtropical coastal mangrove. Appl. Microbiol. Biotechnol. 2013, 97, 7919–7934. [Google Scholar] [CrossRef]
- O’Sullivan, C.A.; Wakelin, S.A.; Fillery, I.R.P.; Roper, M.M. Factors affecting ammonia-oxidising microorganisms and potential nitrification rates in southern Australian agricultural soils. Soil Res. 2013, 51, 240–252. [Google Scholar] [CrossRef]
- Zhou, G.; Zhang, J.; Zhang, C.; Feng, Y.Z.; Chen, L.; Yu, Z.H. Effects of changes in straw chemical properties and alkaline soils on bacterial communities engaged in straw decomposition at different temperature. Sci. Rep. 2016, 6, 22186–22198. [Google Scholar] [CrossRef]
- Roy, D.; McEvoy, J.; Blonigen, M.; Amundson, M.; Khan, E. Seasonal variation and ex-situ nitrification activity of ammonia oxidizing archaea in biofilm based wastewater treatment processes. Bioresour. Technol. 2017, 244, 850–859. [Google Scholar] [CrossRef]
- Wang, S.Y.; Wang, W.D.; Liu, L.; Zhuang, L.J.; Zhao, S.Y.; Su, Y. Microbial nitrogen cycle hotspots in the plant-bed/ditch system of a constructed wetland with N2O mitigation. Environ. Sci. Technol. 2018, 52, 6226–6236. [Google Scholar] [CrossRef]
- Szukics, U.; Hackl, E.; Boltenstern, S.Z.; Sessitsch, A. Rapid and dissimilar response of ammonia oxidizing archaea and bacteria to nitrogen and water amendment in two temperate forest soils. Microbiol. Res. 2012, 167, 103–109. [Google Scholar] [CrossRef]
- Nicol, G.W.; Leininger, S.; Schleper, C.; Prosser, J.I. The influence of soil pH on the diversity, abundance and transcriptional activity of ammonia oxidizing archaea and bacteria. Environ. Microbiol. 2008, 10, 2966–2978. [Google Scholar] [CrossRef]
- Hayden, H.L.; Drake, J.; Imhof, M.; Oxley, A.P.A.; Norng, S.; Mele, P.M. The abundance of nitrogen cycle genes amoA and nifH depends on land-uses and soil types in South-Eastern Australia. Soil Biol. Biochem. 2010, 42, 1774–1783. [Google Scholar] [CrossRef]
- Jiang, X.; Hou, X.; Zhou, X.; Xin, X.; Wright, A.; Jia, Z. pH regulates key players of nitrification in paddy soils. Soil Biol. Biochem. 2015, 81, 9–16. [Google Scholar] [CrossRef]
- Luo, Y.; Yu, Z.; Zhang, K.; Xu, J.; Brookes, P.C. The properties and functions of biochars in forest ecosystems. J. Soils Sediments 2016, 16, 2005–2020. [Google Scholar] [CrossRef]
- Xiao, H.F.; Schaefer, D.A.; Yang, X.D. pH drives ammonia oxidizing bacteria rather than archaea thereby stimulate nitrification under Ageratina adenophora colonization. Soil Biol. Biochem. 2017, 114, 12–19. [Google Scholar] [CrossRef]
- Xia, W.W.; Zhang, C.X.; Zeng, X.W.; Feng, Y.Z.; Weng, J.H.; Lin, X.G. Autotrophic growth of nitrifying community in an agricultural soil. ISME J. 2011, 5, 1226–1236. [Google Scholar] [CrossRef] [PubMed]
- Allison, S.M.; Prosser, J.I. Ammonia oxidation at low pH by attached populations of nitrifying bacteria. Soil Biol. Biochem. 1993, 25, 935–941. [Google Scholar] [CrossRef]
- Hayatsu, M.; Tago, K.; Uchiyama, I.; Toyoda, A.; Wang, Y.; Shimomura, Y. An acid-tolerant ammonia-oxidizing γ-proteobacterium from soil. ISME J. 2017, 11, 1130–1141. [Google Scholar] [CrossRef]
- Huang, X.; Zhao, J.; Su, J.; Jia, Z.; Shi, X.; Wright, A.L. Neutrophilic bacteria are responsible for autotrophic ammonia oxidation in an acidic forest soil. Soil Biol. Biochem. 2018, 119, 83–89. [Google Scholar] [CrossRef]
- Di, H.J.; Cameron, K.C.; Shen, J.P.; Winefield, C.S.; O’Callaghan, M.; Bowatte, S. Ammonia-oxidizing bacteria and archaea grow under contrasting soil nitrogen conditions. FEMS Microbiol. Ecol. 2010, 72, 386–394. [Google Scholar] [CrossRef]
- Wertz, S.; Goyer, C.; Zebarth, B.J.; Burton, D.L.; Tatti, E.; Chantigny, M.H. Effects of temperatures near the freezing point on N2O emissions, denitrification and on the abundance and structure of nitrifying and denitrifying soil communities. FEMS Microbiol. Ecol. 2013, 83, 242–254. [Google Scholar] [CrossRef]
- Avrahami, S.; Bohannan, B.J.M. Response of Nitrosospira sp. strain AF-like ammonia oxidizers to changes in temperature, soil moisture content, and fertilizer concentration. Appl. Environ. Microbiol. 2007, 73, 1166–1173. [Google Scholar] [CrossRef]
- Chen, X.; Zhang, L.M.; Shen, J.P.; Wei, W.X.; He, J.Z. Abundance and community composition of ammonia-oxidizing archaea and bacteria in an acid paddy soil. Biol. Fert. Soils 2011, 47, 323–331. [Google Scholar] [CrossRef]
- Stopnišek, N.; Rangin, G.C.; Höfferle, S.; Nicol, G.W.; Mulec, I.M.; Prosser, J.I. Thaumarchaeal ammonia oxidation in an acidic forest peat soil is not influenced by ammonium amendment. Appl. Environ. Microbiol. 2010, 76, 7626–7634. [Google Scholar] [CrossRef]
- Zheng, Y.L.; Hou, L.J.; Newell, S.; Liu, M.; Zhou, J.L.; Zhao, H. Community dynamics and activity of ammonia-oxidizing prokaryotes in intertidal sediments of the Yangtze Estuary. Appl. Environ. Microbiol. 2014, 80, 408–419. [Google Scholar] [CrossRef]
- Tang, Y.; Zhang, X.; Li, D.; Wang, H.; Chen, F.; Fu, X. Impacts of nitrogen and phosphorus additions on the abundance and community structure of ammonia oxidizers and denitrifying bacteria in Chinese fir plantations. Soil Biol. Biochem. 2016, 103, 284–293. [Google Scholar] [CrossRef]
- Olesen, J.M.; Bascompte, J.; Dupont, Y.L.; Jordano, P. The modularity of pollination networks. Proc. Natl. Acad. Sci. USA 2007, 104, 19891–19896. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.Q.; Zhao, D.Y.; Zeng, J.; Cao, X.Y.; Jiao, C.C. Network Analysis Reveals Seasonal Patterns of Bacterial Community Networks in Lake Taihu under Aquaculture Conditions. Water 2019, 11, 1868. [Google Scholar] [CrossRef]
- Siles, J.A.; Margesin, R. Seasonal soil microbial responses are limited to changes in functionality at two Alpine forest sites differing in altitude and vegetation. Sci. Rep. 2017, 7, 2204. [Google Scholar] [CrossRef] [PubMed]
| Season | Treatment | Moisture (%) | pH | -N (mg·kg−1) | -N (mg·kg−1) | AP (mg·kg−1) | AK (mg·kg−1) |
|---|---|---|---|---|---|---|---|
| Spring | A | 24.94 ± 5.44 a | 7.32 ± 0.05 a | 27.66 ± 8.17 b | 35.13 ± 9.37 a | 467.14 ± 53.44 b | 533.20 ± 110.66 a |
| T | 23.44 ± 0.41 a | 7.18 ± 0.05 a | 15.33 ± 6.96 b | 63.61 ± 26.32 a | 427.53 ± 51.22 b | 561.11 ± 78.41 a | |
| W | 22.47 ± 0.71 a | 7.16 ± 0.09 a | 16.51 ± 2.76 b | 62.78 ± 24.81 a | 308.54 ± 83.92 c | 566.91 ± 49.71 a | |
| Ry | 22.54 ± 4.01 a | 7.22 ± 0.15 a | 56.31 ± 28.18 a | 54.58 ± 25.28 a | 276.72 ± 66.06 c | 460.27 ± 21.98 a | |
| C | 19.51 ± 1.40 a | 7.22 ± 0.17 a | 30.37 ± 11.53 b | 41.65 ± 14.32 a | 237.93 ± 49.81 c | 589.08 ± 49.52 a | |
| Ra | 21.00 ± 4.73 a | 7.17 ± 0.09 a | 20.39 ± 9.53 b | 37.18 ± 15.57 a | 430.65 ± 50.62 b | 619.80 ± 160.82 a | |
| M | 24.71 ± 2.66 a | 7.20 ± 0.21 a | 35.92 ± 6.91 a b | 33.29 ± 14.66 a | 505.27 ± 111.20 b | 503.53 ± 54.84 a | |
| CM | 23.41 ± 1.31 a | 7.10 ± 0.06 a | 26.77 ± 9.94 b | 44.96 ± 21.57 a | 621.85 ± 41.44 a | 617.54 ± 36.36 a | |
| Autumn | A | 25.50 ± 2.86 a | 6.81 ± 0.14 a | 13.15 ± 3.86 a | 43.64 ± 2.99 a | 388.36 ± 40.70 a | 546.25 ± 37.31 a |
| T | 26.68 ± 1.36 a | 6.78 ± 0.05 a | 12.82 ± 2.92 a | 36.51 ± 3.58 a b | 386.53 ± 37.81 a | 517.75 ± 26.07 b | |
| W | 19.98 ± 8.27 a | 6.91 ± 0.05 a | 22.25 ± 8.56 a | 34.14 ± 3.03 b | 390.63 ± 19.06 a | 431.84 ± 38.31 b | |
| Ry | 26.49 ± 2.73 a | 6.85 ± 0.05 a | 17.63 ± 3.03 a | 34.25 ± 7.82 b | 427.94 ± 12.86 a | 530.36 ± 70.57 a b | |
| C | 25.82 ± 1.76 a | 6.80 ± 0.06 a | 19.60 ± 7.38 a | 43.91 ± 3.41 a | 358.98 ± 26.13 a | 525.23 ± 56.65 a b | |
| Ra | 24.39 ± 0.88 a | 6.84 ± 0.08 a | 19.36 ± 8.49 a | 39.16 ± 8.27 a b | 368.92 ± 59.10 a | 506.17 ± 95.25 a b | |
| M | 25.87 ± 2.33 a | 6.86 ± 0.11 a | 21.33 ± 3.87 a | 32.78 ± 1.70 b | 393.76 ± 34.35 a | 515.80 ± 40.82 a b | |
| CM | 19.11 ± 9.03 a | 6.75 ± 0.08 a | 18.81 ± 4.15 a | 33.66 ± 1.72 b | 425.29 ± 30.81 a | 521.20 ± 22.51 a b |
| Season | Abundance | Moisture | pH | -N | -N | AP | AK |
|---|---|---|---|---|---|---|---|
| Spring | AOA | −0.464 * | −0.069 | −0.201 | 0.475 * | −0.087 | −0.278 |
| AOB | −0.253 | −0.576 ** | 0.331 | 0.598 ** | −0.153 | −0.152 | |
| Autumn | AOA | −0.139 | 0.201 | 0.167 | 0.467 * | −0.459* | −0.464 * |
| AOB | −0.018 | −0.213 | 0.369 | 0.452 * | 0.135 | 0.004 |
| Classified | Season | Treatment | Sequences | Number of OTUs | Shannon | Simpson | Chao 1 | Coverage (%) |
|---|---|---|---|---|---|---|---|---|
| AOA | Spring | A | 11,467 ± 903 | 52 ± 2.12 b | 2.35 ± 0.03 a b | 0.1413 ± 0.0010 c d | 69.25 ± 6.01 a | 99.90 |
| T | 11,860 ± 485 | 53 ± 0.71 a b | 2.34 ± 0.06 a b | 0.1408 ± 0.0003 b c | 64.0 ± 1.41 a b | 99.91 | ||
| W | 13,463 ± 2161 | 54 ± 1.41 a b | 2.36 ± 0.16 a b | 0.1668 ± 0.0157 a | 60.60 ± 4.81 b c | 99.92 | ||
| Ry | 11,599 ± 1216 | 50 ± 2.83 a b | 2.22 ± 0.24 b | 0.1593 ± 0.0085 a b | 54.75 ± 3.89 c d | 99.95 | ||
| C | 11,926 ± 2186 | 57 ± 1.41 a | 2.50 ± 0.05 a | 0.1292 ± 0.0029 d | 60.93 ± 1.04 b c | 99.90 | ||
| Ra | 13,620 ± 1645 | 50 ± 0.71 b | 2.30 ± 0.11 a b | 0.1698 ± 0.0042 a | 50.23 ± 0.89 d | 99.96 | ||
| M | 12,758 ± 1706 | 52 ± 4.24 a b | 2.37 ± 0.03 a b | 0.1345 ± 0.0060 c d | 47.38 ± 3.36 d | 99.95 | ||
| CM | 12,400 ± 571 | 52 ± 0.71 b | 2.48 ± 0.11 a | 0.1386 ± 0.0022 c d | 52.29 ± 0.20 d | 99.94 | ||
| Autumn | A | 12,711 ± 1284 | 52 ± 2.12 a b c | 2.35 ± 0.14 a | 0.1403 ± 0.0014 b | 58.80 ± 0.28 a b c | 99.92 | |
| T | 13,821 ± 2446 | 55 ± 1.41 a | 2.34 ± 0.15 a | 0.1672 ± 0.0185 b | 62.00 ± 1.41 a b | 99.92 | ||
| W | 15,714 ± 2920 | 50 ± 0 b c | 2.06 ± 0.09 b | 0.2498 ± 0.0093 a | 52.00 ± 1.42 d | 99.95 | ||
| Ry | 15,114 ± 2652 | 53 ± 1.41 a b | 2.23 ± 0.07 a b | 0.1878 ± 0.0015 b | 56.27 ± 0.09 c | 99.92 | ||
| C | 12,214 ± 1629 | 52 ± 0 c | 2.06 ± 0.15 b | 0.2401 ± 0.0317 a | 62.17 ± 1.18 a | 99.92 | ||
| Ra | 15,193 ± 1976 | 53 ± 1.41 a b | 2.35 ± 0.09 a | 0.1650 ± 0.0067 b | 58.25 ± 2.47 b c | 99.92 | ||
| M | 16,022 ± 2551 | 52 ± 2.83 c | 2.17 ± 0.21 a b | 0.2405 ± 0.0387 a | 60.47 ± 1.60 a b | 99.93 | ||
| CM | 14,474 ± 608 | 51 ± 0 c | 2.20 ± 0.15 a b | 0.1569 ± 0.0159 b | 52.38 ± 2.30 d | 99.93 | ||
| AOB | Spring | A | 18,500 ± 1023 | 54 ± 2.65 a | 2.63 ± 0.22 a | 0.1236 ± 0.0405 a b | 62.14 ± 9.32 a | 99.92 |
| T | 19,921 ± 1211 | 54 ± 7.51 a | 2.74 ± 0.18 a | 0.1181 ± 0.0364 a b | 55.33 ± 8.81 a | 99.96 | ||
| W | 24,485 ± 991 | 53 ± 1.73 a | 2.62 ± 0.10 a | 0.1302 ± 0.0203 a b | 57.25 ± 4.63 a | 99.94 | ||
| Ry | 21,533 ± 599 | 57 ± 4.04 a | 2.65 ± 0.17 a | 0.1201 ± 0.0323 a b | 62.20 ± 10.05 a | 99.92 | ||
| C | 19,429 ± 1106 | 51 ± 2.08 a | 2.46 ± 0.18 a | 0.1612 ± 0.0394 a b | 54.65 ± 4.79 a | 99.93 | ||
| Ra | 21,797 ± 726 | 53 ± 4.62 a | 2.75 ± 0.03 a | 0.1023 ± 0.0050 b | 56.78 ± 3.89 a | 99.94 | ||
| M | 23,097 ± 1326 | 52 ± 1.0 a | 2.44 ± 0.23 a | 0.1688 ± 0.0355 a | 57.19 ± 3.76 a | 99.92 | ||
| CM | 17,184 ± 878 | 52 ± 4.51 a | 2.42 ± 0.12 a | 0.1699 ± 0.0231 a | 58.23 ± 9.46 a | 99.92 | ||
| Autumn | A | 18,457 ± 917 | 57 ± 1.53 a | 2.58 ± 0.27 a b | 0.1387 ± 0.0478 a b | 60.89 ± 1.46 a | 99.91 | |
| T | 17,755 ± 784 | 52 ± 7.0 c | 1.85 ± 0.62 b | 0.3449 ± 0.1940 a | 44.63 ± 8.29 c | 99.93 | ||
| W | 24,315 ± 1284 | 53 ± 1.15 b c | 2.46 ± 0.15 a b | 0.1623 ± 0.0413 a b | 48.58 ± 2.55 b c | 99.94 | ||
| Ry | 21,624 ± 1157 | 52 ± 8.02 a b | 2.41 ± 0.51 a b | 0.1882 ± 0.1410 a b | 57.05 ± 10.15 a b | 99.91 | ||
| C | 21,039 ± 1437 | 51 ± 1.53 b c | 1.85 ± 0.30 b | 0.3428 ± 0.0994 a | 48.76 ± 4.34 b c | 99.92 | ||
| Ra | 21,426 ± 554 | 52 ± 4.04 a b | 2.72 ± 0.16 a | 0.0991 ± 0.0180 b | 56.07 ± 2.69 a b | 99.93 | ||
| M | 24,196 ± 1451 | 51 ± 1.15 a b | 2.13 ± 0.28 a b | 0.2572 ± 0.0700 a b | 55.38 ± 4.89 a b | 99.92 | ||
| CM | 16,464 ± 343 | 51 ± 4.04 a b c | 2.37 ± 0.54 a b | 0.1995 ± 0.1582 a b | 54.67 ± 3.21 b c | 99.92 |
| Network Indicators | AOA (S) | AOA (A) | AOB (S) | AOB (A) | AOA and AOB (S) | AOA and AOB (A) |
|---|---|---|---|---|---|---|
| Nodes | 50 | 50 | 50 | 50 | 100 | 100 |
| Edges | 112 | 97 | 88 | 217 | 228 | 341 |
| Positive correlation edge numbers | 106 | 89 | 81 | 180 | 197 | 283 |
| Negative correlation edge numbers | 6 | 8 | 7 | 37 | 28 | 58 |
| Average path length (APL) | 2.875 | 4.226 | 2.891 | 2.580 | 3.607 | 4.174 |
| Graph density (GD) | 0.091 | 0.079 | 0.072 | 0.177 | 0.044 | 0.069 |
| Modularity (MD) | 0.707 | 0.897 | 0.722 | 0.466 | 1.141 | 0.676 |
| Clustering coefficient (CC) | 0.627 | 0.567 | 0.592 | 0.678 | 0.631 | 0.656 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
He, X.; Li, S.; Wu, F. Responses of Ammonia-Oxidizing Microorganisms to Intercropping Systems in Different Seasons. Agriculture 2021, 11, 195. https://doi.org/10.3390/agriculture11030195
He X, Li S, Wu F. Responses of Ammonia-Oxidizing Microorganisms to Intercropping Systems in Different Seasons. Agriculture. 2021; 11(3):195. https://doi.org/10.3390/agriculture11030195
Chicago/Turabian StyleHe, Xingjia, Sen Li, and Fengzhi Wu. 2021. "Responses of Ammonia-Oxidizing Microorganisms to Intercropping Systems in Different Seasons" Agriculture 11, no. 3: 195. https://doi.org/10.3390/agriculture11030195
APA StyleHe, X., Li, S., & Wu, F. (2021). Responses of Ammonia-Oxidizing Microorganisms to Intercropping Systems in Different Seasons. Agriculture, 11(3), 195. https://doi.org/10.3390/agriculture11030195





