Abstract
Drought stress affects plant growth and development by altering physiological and biochemical processes resulting in reduced crop productivity. Zinc (Zn) is an essential micronutrient that plays fundamental roles in crop resistance against the drought stress by regulating various physiological and molecular mechanisms. Under drought stress, Zn application improves seed germination, plant water relations, cell membrane stability, osmolyte accumulation, stomatal regulation, water use efficiency and photosynthesis, thus resulting in significantly better plant performance. Moreover, Zn interacts with plant hormones, increases the expression of stress proteins and stimulates the antioxidant enzymes for counteracting drought effects. To better appraise the potential benefits arising from optimum Zn nutrition, in the present review we discuss the role of Zn in plants under drought stress. Our aim is to provide a complete, updated picture in order to orientate future research directions on this topic.
1. Introduction
Abiotic stresses affect all living organisms present on Earth. Among them, drought stress has devastating impacts on agricultural production [1]. Drought stress is becoming more frequent due to unpredictable rainfalls and changes in climate patterns [2]. The rapid increase in atmospheric temperature has increased crop exposure to drought induced stress [3,4]. Its severity is unpredictable, as it depends upon many factors including the distribution and amount of rainfall, evapotranspiration and soil ability to store moisture [5].
Drought stress diminishes crop productivity by reducing water uptake, leaf water status and gas exchange rates [6]. Moreover, it reduces stomatal conductance, which in turn increases leaf temperature and leads to wilting [6,7]. The water deficit conditions have further impacts such as disturbing membrane permeability, nutrient uptake and chlorophyll synthesis, thus negatively affecting plant photosynthetic efficiency [8,9,10,11]. Although water deficit imposes severe impacts on plants, plants modulate drought-induced effects by activating the antioxidant defense system that scavenges the reactive oxygen species (ROS), initiating a wide range of physio-chemical responses [12]. The ROS damage plant cell membranes and macro-molecules including proteins and lipids [13].
Agricultural areas around the globe are already facing a rapid reduction in the supply of irrigation water [14], thus it is urgent to adopt policies in order to protect the crops from extreme weather events including drought. The development of drought tolerant crops is one of the imperative approaches to cope with this adversity; however, this approach is time consuming and requires big investments. Conversely, there are other management approaches that can provide quick solutions to this problem. Among them, the use of the mineral nutrients is considered one of the most promising approaches to diminish the effect of drought. The role of mineral nutrients, specifically Zn, is well understood in plant growth and development under normal conditions. Conversely, Zn role in drought tolerance is still poorly understood.
Zinc is an indispensable micronutrient for crop growth, an important component of carbonic anhydrase and a stimulator of aldolase, which are involved in carbon metabolism [15]. Zn is also an integral component of several biomolecules such as lipids, proteins and co-factor of auxins, and, therefore, it plays an important role in plant nucleic acid metabolism [16]. Zn application has been proved beneficial in improving crop yield and quality [17,18], while its deficiency reduces yield and deteriorates crop quality [19]. Moreover, higher Zn contents have toxic effects on plants and lead to the suppression of cell division and elongation, affecting biomass production [15]. Fewer studies have shown that adequate supply of Zn can modulate drought tolerance in different crops as wheat, sunflower, tomato and red cabbage [20,21,22]. However, there is lack of information regarding Zn-induced mechanisms conferring drought tolerance in plants. Therefore, this review addresses different effects of drought stress on plants and shows the pending research gaps in plant physiological, biochemical and molecular aspects and Zn cross talk with other molecules in Zn-induced drought stress tolerance.
2. Effects of Drought Stress on Plants
Plants are sessile organisms exposed to biotic as well as abiotic stresses during their life cycles. Drought is a most imperative abiotic stress that has deleterious effects on plant growth and development under several viewpoints [23,24]. Additionally, drought stress has negative effects on productivity and quality of crops [25,26]. Plant growth and development depend on cell division, elongation and differentiation, and drought stress affects all these processes owing to reduction in turgor pressure, enzymatic activities and energy supply for photosynthesis [27,28]. Plant water potential (PWP) and turgor are decreased under water limited conditions, thus plants cannot function properly under these conditions [23,27,28].
The decrease in relative water content (RWC) is the initial consequence of drought stress [29]. The lower RWC reflects in substantial reductions of leaf water potential, which lead to closing of stomata. The increase in stomatal resistance reduces the rate of transpiration and, therefore, leads to increase in leaf temperature because the process of transpiration is the key mechanism that controls leaf temperature [30,31]. The higher leaf temperature denatures various proteins and enzymes and reduces membrane permeability and, resultantly, various facets of plant metabolism are affected. These changes are the main reasons for the disturbance in photosynthesis, respiration, mineral nutrition and synthesis of proteins and amino acids [31,32].
The reduction in photosynthesis is the main impact determined by drought [33,34]. Many factors are responsible for this reduction, namely reduced leaf expansion, increased leaf temperature, premature leaf senescence and impaired photosynthetic machinery [35,36]. Moreover, stomatal and non-stomatal factors also inhibit photosynthesis in drought stress [31]. The CO2 limitation due to closing of stomata particularly under light saturation leads to accretion of reduced photo-synthetic electron transport components that negatively affect the photosynthetic efficiency. The accretion of such components results in production of reactive oxygen species (ROS) that cause oxidative damages in the chloroplast [30,37]. The reduction in chlorophyll content due to water stress also decreases the photosynthetic rate [24,38]. The key enzyme involved in carbon metabolism in the Calvin cycle is ribulose-bisphosphate carboxylase/oxygenase (Rubisco) [29]. The activity and amount of Rubisco decrease dramatically under water deficiency, which negatively affects photosynthesis [39]. Drought tolerance is a demanding ability, and the amount of energy required to cope with drought stress is enormous [35]. The major consumers of carbon (C) assimilated through photosynthesis under drought stress are plant roots, which use C for their growth and maintenance [29]. Additionally, environmental conditions affect plant respiration, and changes in C metabolism may occur owing to diminished photosynthesis and active respiration. The growth rates of plants can be precisely determined as the ratio of CO2 assimilation to respiration [36]. Drought also disrupts mineral nutrition and ion homeostasis in plant cells [40]. The reduction in water supply under drought stress reduces soil nutrient availability as well as uptake by plant roots, resulting in reduced nutrient concentration in plant tissues [40]. The change in nutrient uptake and transport to shoots is the main consequence of drought stress in plants.
The hormones play a role of paramount relevance in regulating several plant processes. Abscisic acid (ABA) is the most prominent plant hormone in response to water deficit conditions [35,41]. ABA is primarily synthesized in the vascular system, whence it is transported to the target tissues. The transport occurs in both xylem and phloem, permitting bi-directional transport between roots and shoots. However, under drought stress, pH significantly increases in the apoplast, determining great retention of ABA, which then functions as a root-to-shoot signal resulting in final reduction in transpiration [42]. The ABA induces the accumulation of late embryogenesis abundant (LEA) proteins, which play a significant role in plant adaptation to drought [26]. Moreover, drought rapidly reduces endogenous cytokinin and gibberellin levels. The cytokinins delay leaf senescence, improving plant resilience under drought stress [35].
Plants accumulate various substances including proteins and amino acids in response to drought stress [43]. Water deficiency also alters the amount of proteins [29], whose contents generally decrease under drought stress owing to reduction in their synthesis. The accumulation of osmolytes as proline provides osmotic adaptation that is the main mechanism for plant resistance against drought stress. Water deficiency increases proline content in plants in two different ways: (1) by directly increasing the synthesis of proline; and (2) by reducing the activities of enzymes involved in proline degradation [34]. The accumulation of proline and other osmolytes reduces cell water potential, and therefore increases water uptake under drought stress [34]. Additionally, osmolytes protect delicate cell components from oxidative stress, and the biosynthesis of proline in plants plays a vital role in the energy balance between chloroplasts and mitochondria [44]. In conclusion, drought disrupts various physiological, biochemical and molecular processes determining reductions in growth, yield and quality.
3. General Mechanisms of Plant Response to Drought Stress
Plants cannot move from their place; therefore, they must tolerate the abiotic stress. Plants have developed different mechanisms of resistance against stress conditions. The plants are multicellular organisms whose response against abiotic stresses is very complex [45]. The mechanisms of plant resistance to drought stress rely on three main strategies: drought escape, avoidance and tolerance. The adaptation of plants to drought can be achieved through different physiological, morphological, bio-chemical and molecular responses.
4. Zinc Uptake and Transport in Plants
Plant species have a broad range of Zn requirement and concentration, owing to variations in Zn uptake from soil, transport from roots to shoots and sequestration in plant organs [46]. Plant roots take up Zn in divalent form (Zn2+), although in few cases plant roots also absorb Zn in the form of organic ligand–Zn complexes. Moreover, plants use two different strategies to take up Zn in divalent form depending on the ligands secreted by the roots [46]. The first approach involves the effluxes of reductants as organic acids (OA) and H+ ions that increase the solubility of Zn complexes (Zn phosphates and hydroxides), resulting in release of Zn2+ ions for the absorption the root epidermal cells. The second strategy involves the efflux of phyto-siderophores (lower molecular weight compounds) that make stable complexes with Zn fostering Zn influx into the root epidermal cells. However, this strategy is only found in the roots of cereal crops [46].
The passive uptake of Zn involves the participation of water molecules, and variations in Zn contents across the root cells plasma membranes (RCPM). The major driving energy involved in the uptake of Zn2+ comes from hyper-polarization of RCPM, which is governed by the activities of RCPM H+-ATPase system. The activities of this system help in pumping H+ ions extra-cellularly at the expense of ATP hydrolysis. The release of H+ ions in root rhizosphere results in hyper-polarization of RCPM and decreases soil pH, which in turn increases the rate of cation uptake. However, the charged Zn2+ ions cannot cross the cell membrane freely [47,48], thus specific transporter proteins help in the transport of Zn2+ cations [49,50] (Table 1). Many genes have been recognized that either encode the proteins involved in Zn transport or regulate the expression for optimum Zn transport. The most imperative proteins involved in the transport of Zn are: the ZIP family proteins (Zn, iron-permease family/ZRT-, IRT proteins), the heavy metal ATPase (HMA) family and the metal tolerant proteins (MTP) family. Among these families, the members of the ZIP family are involved in Zn influx into cell cytosol, while HMA are involved in Zn efflux into the apoplast. Moreover, the MTP family is involved in Zn sequestration into the vacuoles and endoplasmic reticulum [46]. The transporter proteins have no close linking with the breakdown of ATP, which supports the passive more than the active Zn uptake mechanism. Moreover, uptake of Zn2+ also occurs with the help of non-selective cation channels linked with passive fluxes of different cations groups [51]. The additional driving force involved in the uptake of metal cations can be due to lower cytoplasmic activities, which is a result of metal sequestration and binding to the intra-cellular sites including, Zn finger proteins, and enzymes [46].

Table 1.
Zn transporter proteins in different plants.
After passing the barriers of casparian strips in roots, Zn2+ enters the living cells of the peri-cycle and xylem parenchyma symplastically. Moreover, another barrier to Zn2+ transport also occurs at this stage known as xylem loading. The continuous activities of H+-ATPase in the xylem parenchyma result in hyper-polarization of membranes that limit the movement of positive ions (Zn2+) out of cell cytosol. Therefore, the Zn2+ loading from the cells of xylem to apo-plastic xylem is an active process [59]. The active efflux of symplastic Zn2+ is governed by particular transporters (HMA family) which are present on the xylem parenchyma. The increased activities and high expression of membrane H+-ATPase in parenchyma of xylem are also accountable for the acidic nature (pH: 5–6.5) of xylem sap. Moreover, in the xylem sap, the metal ions including Zn2+ are transported as forming metal complexes with OA, nicotian-amine histidine and asparagine. After entering the phloem, Zn is further translocated into diverse plant organs and sinks governed by short as well as long distance pathways. Zn mobility is quite high in phloem compared to xylem, owing to greater concentration of solutes including the OA and peptides in phloem sap [46]. Moreover, Zn is transported in phloem tissues in ionic forms or as metal complexes. Generally, xylem has lower solute contents; however, it is fundamental for nutrient transport to different organs [46].
5. Roles of Zn in Plants under Drought Stress
5.1. Effects of Zn on Plant Growth and Development under Drought Stress
Seed germination is the first stage of plant life affected by the drought stress. In turn, seed germination and seedling emergence influence plant density and final productivity [60]. The variation in seedling emergence due to environmental factors is of paramount importance in view of crop management. The interaction among environmental factors and internal seed mechanisms determine the level of germination and emergence under specific conditions [60]. Zn application (seed priming) improves germination and yield of maize, wheat and chickpea under a wide range of environmental conditions [61,62]. The drought stress reduces plumule length and dry weight, owing to reduced remobilization of nutrients from reserves to embryo. The application of Zn (priming) improves the synthesis of IAA and GA3 under drought conditions [63], and the synthesis of these hormones augments plumule length and weight under drought stress.
Zn is a fundamental nutrient for plants as it plays a vital role as metal component and co-factor of many enzymes [64]. The cell membrane is the first target of abiotic stresses [65], and the maintenance of its stability under drought is the core part of drought tolerance [66]. Adequate Zn supply in drought stress maintains membrane permeability, the activity of antioxidant substances, photosynthetic efficiency and WUE [67,68,69]. Moreover, Zn application results in appreciable increase in leaf area, the content of chlorophyll and other photosynthetic pigments, and stomatal conductance, thus resulting in improved growth and yield [70]. Similarly, Sultana et al. [71] noted that Zn countered the adverse impact of drought and remarkably increased wheat productivity. In another study, Chattha et al. [17] noted that Zn application improved maize yield and harvest index in drought stress. Moreover, Hera et al. [65] noted that foliar applied Zn diminished the negative impacts of water deficit and increased growth and yield of wheat. Therefore, Zn application counteracts drought by improving membrane stability, hormone synthesis, the photo-synthetic process and the scavenging of ROS (Table 2).
5.2. Effect of Zn on Plant Water Relations in Drought Stress
Leaf water potential, relative water content (RWC), stomatal conductance and leaf temperature directly affect the plant water relations. The RWC is a main physiological trait which determines plant drought tolerance ability [72,73]. Zn application improves chlorophyll, RWC and carotenoid contents, while it reduces the electrolyte leakage and water loss in drought condition [74]. The application of Zn enhances antioxidant enzyme activities, stomatal conductance and RWC while restraining electrolyte leakage in water deficit situations [75].
Adequate Zn nutrition improves vegetative growth and drought tolerance in alfalfa by enhancing root growth and RWC [76]. The maintenance of good leaf water status is a result of proper control exerted by the guard cells in diverse conditions, which improves the growth and enhances crop survival under stress conditions [77]. Therefore, from this viewpoint, the improvement in RWC mediated by Zn application results in better crop adaptation to water deficit conditions. Zn application supports the RWC owing to the role of Zn in improving tissue formation, osmolyte accumulation and prevention of leaf tissues from destructive effects of water deficient conditions [78,79]. In conclusion, Zn application improves the activities of antioxidants and decreases electrolyte leakage, which in turn increases membrane stability and results in increased RWC under drought stress (Table 2).


Table 2.
Effect of Zn application on growth and physiological characteristics in drought stress.
Table 2.
Effect of Zn application on growth and physiological characteristics in drought stress.
| Crop | Drought Stress | Effects | References |
|---|---|---|---|
| Maize | 40% field capacity | Zn application increases plant biomass, stomatal conductance and quantum yield of photosystem II under drought stress. | [80] |
| Maize | Drought stress at pollination and seed filling | Zn improves grain yield, RWC and chlorophyll content. | [81] |
| Sunflower | Drought stress at seed filling stage | Zn improves seed and oil yield, chlorophyll and proline contents and SOD activities. | [82] |
| Wheat | Drought stress at grain filling stage | Zn application improves yield, chlorophyll content and activities of SOD, POD and CAT. | [83] |
| Triticale | Water stress at heading and booting | Zn application increases the activities of POD, CAT, quantum yield of PS-II, chlorophyll content, stomatal conductance and reduces the electrical conductivity. | [75] |
| Common bean | Irrigation applied at interval of 6, 12 and 18 days | Zn increases plant height, shoot biomass, chlorophyll and carotenoid contents, leaf NPK content and decreases the MDA contents | [84] |
| Tomato | With-held irrigation | Zn supplementation improves the stomatal aperture and chlorophyll content. | [85] |
| Chickpea | 35% field capacity | Zn leads to improvement in PS-II efficiency, CO2 assimilation rate, proline content and activities of SOD, APX, while reduces the MDA contents. | [86] |
| Chickpea | 35% field capacity | Zn application increases chlorophyll and carotenoid contents, seedling vigor and seed yield. | [87] |
| Wheat | Drought stress at heading and an-thesis stage | Zn improves stomatal conductance, WUE, efficiency of PS-II, chlorophyll content and reduces energy dissipation | [88] |
5.3. Effect of Zn on Cell Membrane Stability under Drought Stress
The extent of membrane injury can be estimated by the measurement of electrolyte leakage from the cell. The amount of the leakage can be correlated with different physiological and bio-chemical processes including the synthesis of antioxidant enzymes, WUE, osmotic potential and stomatal regulation [89,90]. Thus, it is not surprising that low electrolyte leakage can be recommended as criteria to identify stress resistance in different crop species and genotypes [91].
Drought induces lipid peroxidation in plants, which results in leakage of essential solutes from the cells and organelles, causing damage to membrane functions and several metabolic processes [92]. Thus, the ability of plants to control the movements of ions in and out of cells can be used as test to check damage to plant tissues. Zn has special properties as it exists in divalent state without redox cycling and, therefore, remains stable in biological mediums [93]. Owing to these properties, Zn protects membrane lipids from ROS, in turn preventing the leakage of ions from the ion channels [90]. Moreover, drought stress enhances malondialdehyde (MDA) content and ion leakage owing to reduction in the activity of antioxidants [91]. The application of Zn improves the activity of antioxidant enzymes such as superoxide dismutase (SOD) and peroxidase (POD) and reduces the effects of oxidative stress, the accumulation of MDA and electrolyte leakage. Therefore, Zn protects the cellular membranes from drought stress damages (Table 2).
5.4. Regulation of Osmolyte Accumulation by Zn under Drought Stress
The osmotic adjustment is the principal mechanism linked with maintenance of higher turgor potential and water retention in plants, in response to drought [94]. The osmotic adjustment is related to accretion of different lower molecular weight substances, such as proline, glycine-betaine, soluble sugars (SS) and soluble proteins (SP). Moreover, as the result of osmolyte accumulation, the activity of organelles and cytoplasm takes place at the normal pace, ensuring sustained growth, photosynthetic efficiency and assimilate partitioning [29].
The SS and SP diminish drought induced damages by favoring leaf turgor and stomatal conductance for efficient CO2 intake and promoting the ability of plant roots to take up more water [95,96]. Besides its role in regulating enzyme activity, Zn also improves protein synthesis and carbohydrate metabolism [97]. The application of Zn improves the expression of Zn finger proteins which in turn stimulate SS and proline accumulation under drought conditions [98,99,100]. Proline accumulation protects not only cells and the enzymes involved in osmo-regulation, but also plants from the osmotic stress [101]. The application of Zn also increases the leaf water potential under drought conditions [20,21].
Moreover, the accumulation of osmolytes augments root water uptake by regulating the leaf water potential, alleviates the oxidative damage and improves drought resistance [13]. Therefore, accumulation of compatible solutes under drought stress owing to Zn supplementation improves drought tolerance. Moreover, solute accumulation owing to Zn application increases water use efficiency (WUE), chlorophyll content and dry matter production [100]. Zn application in drought conditions increases the accumulation of soluble carbohydrates, which in turn protect the plants from the deleterious effects of drought stress [102,103].
Zinc also improves the synthesis of glycine-betaine (GB) in drought stress conditions, which contributes to the increased tolerance [104]. Amino acids are known for their important role of regulating the osmotic homeostasis under stress conditions [105]. Zn application improves the synthesis of the amino acids which protect the plants from drought stress consequences [106,107]. To summarize, Zn application improves the accumulation of compatible solutes which maintain a higher turgor potential and water retention in plants in response to drought. This contributes significantly to crop performance under stress conditions. However, there are still gaps of knowledge concerning the effects of Zn application on the accumulation of proline, GB, polyamines and polyphenols under drought stress conditions. Therefore, future research should focus these gaps providing clues to a better understanding of the comprehensive role of Zn in higher plants.
5.5. Effect of Zn on Stomatal Regulation and Water Use Efficiency under Drought Stress
Water use efficiency is significantly reduced under drought stress, owing to reduction in stomatal conductance. The application of Zn improves the stomatal regulation thanks to the maintenance of membrane integrity. Zn deficiency leads to reduction of K+ in guard cells, and this may be associated with the increase in K+ efflux relative to influx, owing to membrane damage caused by Zn deficiency [108]. Nonetheless, the exact role of Zn in regulating the stomatal opening is still unclear and direly needs deeper investigation. Generally, stomata closing in response to drought improves WUE, due to the fact that a greater reduction in transpiration than in photosynthesis occurs [109]. Zn application in drought conditions improves stomatal conductance and favors accumulation of osmolytes, which in turn improve WUE and drought tolerance [109]. Plants consume less water under drought conditions, but water that is transpired is not efficiently used in case of Zn deficiency [109,110]. Zn deficiency under stress conditions increases leaf temperature owing to closing stomata, and increases the proportion of carbon (C) lost in respiration [111]. Under such circumstance, the application of Zn maintains the stomatal regulation and leaf temperature, thus increasing WUE. In another study, Ahmed et al. [112] noticed that Zn application improved photosynthesis, stomatal conductance and transpiration, and, therefore, it resulted in marked WUE increase under drought stress. In conclusion, Zn application improves the K+ influx in guard cells, which ameliorates the stomatal conductance resulting in significant WUE increase in drought stress (Table 2).
5.6. Effect of Zn on Nutrient Uptake under Drought Stress
The role of mineral nutrients in improving drought tolerance has been studied by many researchers. However, this is still insufficient and somewhat intangible. Macronutrients form the structural components of plants, and their deficiency causes symptoms in plants that are readily observed [113]. Conversely, the micronutrients affect the susceptibility of plants to diverse stress conditions by regulating the enzymatic activities, and modulating signal transductions and accumulation of compatible solutes [114]. There are many reports indicating the noxious impacts of nutrient deficiency on photosynthesis under drought stress. However, the studies related to the effect of drought on mineral nutrient uptake and their effect on plant physiology still have many dark areas [29,113].
Limited information is available about the effects of Zn application on nutrient uptake in drought conditions. In the literature, the exogenously applied Zn markedly increased leaf Zn content [115], as well as the uptake of other nutrients, i.e., iron and manganese [116]. The effect of Zn on nutrient uptake and its interaction with other nutrients are well documented under normal conditions. However, they are still unclear under drought stress and need to be elucidated. Therefore, future research should better focus the effects of Zn application on nutrient uptake and its interaction with other nutrients under drought stress (Table 2).
5.7. Effect of Zn on Photosynthesis under Drought Stress
The abiotic stresses faced by plants, affect several physiological and biochemical mechanisms. Zn has many functions in plants as it is a structural catalytic and co-catalytic component of more than 300 enzymes including carbonic anhydrase, carboxy-peptidase, alcohol dehydrogenase, Cu/Zn superoxide dismutase, fructose 1,6 bisphosphatase and aldolase [113]. Zn deficiency reduces the activity of carbonic anhydrase affecting photosynthesis. The application of Zn repairs the PS-II processes and integrates the Rubisco structure, improving photosynthesis under drought stress [15,117]. Furthermore, Zn supplementation under water deficit conditions improves chlorophyll content and photosynthetic rates, alleviating the effects of drought [69]. Sub-stomatal CO2 concentration (Ci) increases under drought stress owing to low photosynthetic rate. The reduction in photosynthesis rate (Pn) associated with an increase in Ci has been frequently interpreted as a direct effect of drought stress on plant photosynthetic capacity [118]. Zn application leads to reduction in Ci under drought conditions and, therefore, improves Pn. Zn application also improves photosynthesis under drought by its direct effect on stomata opening, detoxification of ROS and activation of enzymes [103]. Drought reduces quantum yield owing to decline in chlorophyll content. However, Zn application under drought increases chlorophyll content and quantum yield, resulting in significant increase in photosynthesis under drought stress [104].
The application of Zn substantially increases chlorophyll content, Fv/Fm and photosynthetic characteristics under drought stress [70,119,120]. Moreover, Zn application improves chlorophyll synthesis owing to the fact that Zn is a structural component of different proteins and enzymes and a co-factor for normal biosynthesis of pigments [121]. In addition, Zn stabilizes membrane permeability and integrity under water stress, which results in increased photosynthesis [120,122]. Moreover, Zn affects the concentration of nutrients involved in the synthesis of chlorophyll and availability of other nutrients including nitrogen and manganese, which serve as constituents in the molecular structure of chlorophyll [104,123]. Zn also triggers the enzymes involved in the chlorophyll biosynthetic pathway, and the activities of antioxidant enzymes which improve photosynthesis in drought stress [124]. Therefore, Zn application improves enzymatic activities, chlorophyll content and Pn; detoxifies ROS, and maintains the Rubisco structure. All this leads to significant improvements in the smoothness of the photosynthesis process under drought stress (Table 2).
5.8. Zn-Mediated Cross Talk with Hormones under Drought Stress
Plant growth regulators (PGR), when applied exogenously, and phyto-hormones, when produced internally, substantially affect plant physiological processes. Both terms are used inter-changeably, when referring to auxins, namely indol acetic acid (IAA), gibberellins (GA), cytokinis (CK), ethylene and abscisic acid (ABA) [125]. Phyto-hormones play an appreciable role in drought tolerance [29]. Zn application substantially influences the IAA level, and Zn is known to be co-enzyme for the synthesis of tryptophane that is a precursor to the formation of IAA [117,126]. The application of Zn under drought stress increases the IAA levels, which in turn improve root growth, facilitate water uptake, and therefore improve drought tolerance [117]. Water deficiency significantly reduces IAA and GA contents, and Zn application significantly improves the levels of IAA and GA reflecting in improved plant performance [127]. The application of Zn highly improves the metabolic pathways of tryptophan in drought conditions. Therefore, being the precursor of IAA and melatonin, higher concentration of tryptophan alleviates the effects of drought by reducing the oxidative damages and regulating the osmotic balance [128]. The initial precursors of melatonin synthesis are tryptophan, which is catalyzed by tryptophan decarboxylase (TDC), and tryptamine 5-hydroxylase (T5H). Together, they synthesize the intermediates of melatonin, i.e., tryptamine and serotonin [129]. Then serotonin is converted into melatonin by serotonin N-acetyltransferase (SNAT) and N-acetylserotonin O-methyltransferase (ASMT)/caffeic acid O-methyltransferase (COMT). Additionally, tryptophan is a precursor for IAA production, which maintains plant growth and osmotic balance by regulating vacuole osmotic pressure under drought [130].
Melatonin and serotonin play a vital role against various abiotic stresses. The higher melatonin content regulates the nitro-oxidative balance, proline metabolism and the antioxidant defense. Hence, it contributes to biomass production and drought tolerance [131,132]. Zn improves melatonin content in drought stress by increasing the upstream substrate contents and the gene expression of enzymes involved in melatonin biosynthesis, which in turn improves the drought tolerance in plants [133]. However, the mechanisms underlying Zn effects on ABA, GA, CK, brassinosteroids, jasmonates and ethylene contents in plants are still unknown. Therefore, future studies should be conducted to answer these questions.
5.9. Detoxification of Reactive Oxygen Species by Zn under Drought Stress
Drought stress leads to over production of ROS resulting in cell membrane damages, MDA accumulation and cell death. Zn application under drought stress reduces MDA content, demonstrating the critical role of Zn in cell membrane protection from ROS in drought stress [100]. Plants have a sophisticated antioxidant defense system containing various enzymes protecting plants from the ROS under drought stress [134]. SOD contains Cu/Zn-SOD, Mn SOD and Fe-SOD, which constitute the first protective systems against O2−, and converts it into H2O2 and O2 [135]. Zn plays a chief role in Cu/Zn-SOD for the elimination of ROS adverse effects [136]. Additionally, Zn binds with cysteine and histidine to restrict ROS accumulation [15].
The application of Zn improves the expression of zinc-finger proteins, increases the expression of these proteins, enhances the activities of the antioxidant enzymes (Figure 1) and decreases the MDA accumulation [100,137]. It was shown that Zn application under drought stress increases the activities of SOD, CAT and APX in cotton and rice, which suggests that application of Zn significantly enhances the activity of the antioxidant system, thus contributing to the alleviation of oxidative stress caused by drought stress [100,138].
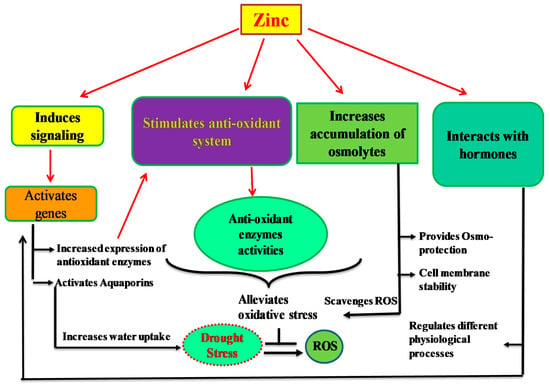
Figure 1.
Mechanisms of drought tolerance induce by Zn. Zinc application improves the antioxidant activities, increases osmolyte accumulation, hormonal cross talk and cell signaling, which in turn improve membrane stability, physiological processes including water uptake and ROS scavenging.
The other lower molecular weight antioxidant substances including ascorbate (ASC), glutathione (GSH) and phenolics can also improve the resistance against drought stress [139]. Zn supplementation increases ASC and GSH contents and protects plants from the oxidative stress caused by drought [122,140]. Total phenolic contents (TPC) and total flavonoid contents (TFC) represent amounts of secondary metabolites protecting plant cells from the attack of ROS. The Zn supplementation under drought stress improves TPC, TFC and ASC in pistachio leaves and vine berries owing to increased antioxidants biosynthesis protecting the cells from ROS [141,142].
The ASC-GHS cycles and biosynthesis of phenolics are involved in the non-enzymatic antioxidant system. MDHAR, DHAR, GS and APX are the imperative enzymes involved in the ASC–GSH cycle. The APX and MDAR enzymes play a prominent role in drought tolerance, and Zn application considerably increases the activity of these enzymes in drought stress [120,143].
5.10. Effect of Zn on Stress Proteins under Drought Stress
The synthesis of stress proteins in plants is a pervasive response against various stresses including the drought stress. Most of the stress proteins are water soluble; thus, they provide significant contribution to stress tolerance mechanism by the hydration of cellular structures [144]. The increase in the expression of Zn finger proteins increases the accumulation of SS and proline and therefore, improves drought tolerance [98].
There is paucity of information available about the effects of Zn on stress proteins. Likewise, C2H2 Zn finger protein is an imperative Zn finger protein that considerably enhances the drought resistance by improving the accumulation of osmotic substances. For instance, in rice the over expression of ZFP252 significantly improves the drought tolerance [99]. Likewise, the over expression of OsMSR15 and ZFP3 in Arabidopsis counters the damaging effects of drought by increasing proline accumulation, reducing the electrolyte leakage and increasing the expression of stress-responsive genes [145,146]. C2H2 Zn finger protein also boosts the drought tolerance by enhancing ROS scavenging. ZFP245 Zn finger protein improves the drought resistance in rice by scavenging the ROS owing to increased activities of SOD and POD [147]. C2H2 Zn finger protein also improves the drought tolerance by means of ABA and other signaling pathways [148]. Therefore, the increase in the expression of Zn finger proteins counters the effects of drought by increasing accretion of compatible solutes, scavenging ROS and affecting the signaling pathways.
There is another group of proteins known as heat shock proteins (HSPs), which are generally produced in plants in response to heat stress. However, some HSPs have been found to be induced under diverse stresses including the drought stress [29,149]. Similarly, drought changes the expression of late embryogenesis abundant (LEA) proteins that prevent the denaturation of plant proteins [150]. However, there is no single study available regarding the effects of Zn application on the expression of HSPs and LEAs under stress. Therefore, future studies should be conducted to quantify the effects of Zn application on the expression of these proteins under dry conditions.
5.11. Effect of Zn on the Activities of Aquaporins under Drought Stress
Aquaporins (AQPs) are channel proteins located in the plasma and intra-cellular membranes. AQPs play a vital role in plant water relations by regulating membrane hydraulic conductivity, osmotic potential, and changes in water permeability [151]. In water limited conditions, the aquaporin gene expression can be regulated to help the plant in maintaining the water balance [152,153,154].
Plant AQPs can be sub-divided into the plasma membrane intrinsic proteins (PIPs), tonoplast intrinsic proteins (TIPs), NOD26-like intrinsic proteins (NIPs), small basic intrinsic proteins (SIPs) and uncharacterized intrinsic proteins (XIPs) [155,156]. AQPs are responsible for 75–95% of water transit in plants [157,158]. Zn application modulates the AQPs PIP2;1 and PIP2;5: the two AQP isoforms play a vital role in plant water dynamics as a part of a quick Zn response and osmotic stress prevention [159,160].
The over-expression these two AQPs isoforms favors water transport to the inner tissues of leaves, and they also prevent the decrease in leaf water potential and xylem tension [161,162]. As for the root AQPs, Zn application improves the expression of root AQPs including the PIP1 and PIP1;4 in Arabidopsis in response to drought stress [155]. After the application of Zn, PIP1;4 promotes water transport from roots to aerial parts in order to maintain the water status and gas exchange characteristics in Brassica rapa [160]. The application of Zn modifies the gene expression of AQPs present in the membranes of leaves and roots, and these proteins are involved in the shoot-to-root hydraulic communications enabling the water transport [163]. The application of Zn induces quick hydraulic signals from plant leaves to roots, resulting in changes in the hydraulic conductance regulating water movements [160]. Moreover, Zn application increases the abundance of isoforms such as PIP1, which in turn improve root hydraulic conductance, photosynthesis and Ca2+ root uptake [160]. Thus, all these changes lead to improvement in growth under drought stress. To summarize, Zn application increases the expression of AQPs, which improve the shoot-to-root hydraulic communications thus favoring the water transport under drought stress. However, there are still many questions that need to be answered, as the particular function of each isoform in response to Zn. Moreover, other mechanisms of AQPs regulation at protein level need to be further elucidated.
6. Methods of Zinc Application to Crops
There are many ways in which Zn can be applied to plants: seed priming, seed coating, soil and foliar application [18,164]. Seed priming with Zn substantially improves the germination and stand establishment [165]. Micronutrient application as seed treatment successfully delivers the nutrients aimed for growth and yield enhancement [165]. However, seed treatments with higher Zn (2 g Zn kg−1 seed) concentration significant inhibit seed germination and subsequent growth [166]. Thus, in view of a suitable scale of Zn application through seed priming, it is necessary to optimize the level of application. Foliar spray of Zn (0.5%) is also considered a quick Zn application method in conditions of low Zn availability to improve crop productivity [18]. Foliar application (1.5 kg ha−1) is a very easy and economical method that markedly increased the yield of wheat and maize [167]. The foliar application is most effective owing to the fact that Zn is directly applied onto plant leaves [168]. The foliar applied Zn (140 g Zn L−1) significantly increased the starch content, grain yield and Zn content in the maize crop, which is prone to deficiency in this micronutrient [169]. In another study, it was noted that foliar applied Zn (300 ppm Zn-EDTA) significantly improved growth and yield traits of mungbean [170]. Foliar applied Zn (3 mg L−1) significantly improved the physiological traits, yield and quality of beans (Phaseolus vulgaris L.) [171]. Soil application is also a common and important method to supply the nutrients needed in larger quantity [172]. Micronutrients including Zn can be easily supplied by soil application. Soil applied Zn (12 kg ha−1) effectively improved the grain and biomass yield of maize [173]. Similarly, in another investigation, applied Zn (15 kg ha−1) resulted in a significant increase of grain yield and Zn content in the wheat crop [174]. The combined soil and foliar application is also useful to improve crop productivity. Soil (50 kg ZnSO4 ha−1) + foliar (0.5%) application of Zn significantly increased the yield and grain Zn content of wheat [17]. In accordance with this, Hassan et al. [18] noted that Zn seed priming (0.3 M ZnSO4), soil (50 kg ZnSO4 ha−1) and foliar (0.5% ZnSO4) applications markedly increased the growth, yield and grain Zn contents of wheat.
7. Conclusions and Future Prospects
Drought is a major abiotic stress considerably limiting crop productivity across the globe. It causes a variety of adverse effects in plants, including reduction in seed germination, seedling establishment, biomass and grain yield and nutrient and water uptake. Additionally, drought stress disturbs plant physiological and molecular processes, which in turn have negative impacts on overall plant performance. Nutrient management plays a pivotal role to develop plant resistance against drought stress. Wise nutrient management improves plant resilience through diverse mechanisms. Several researchers report the role of Zn in support of plant drought tolerance through different mechanisms: Zn alleviates the drought stress by improving the plant water relations, cell membrane stability, osmolytes accumulation, stomatal regulation and water uptake. Additionally, Zn application increases the endogenous hormones (auxins, gibberellins and melatonin) and improves the activities of aquaporins and the antioxidant system, which in turn support the photosynthetic efficiency resulting in significant mitigation of drought stress effects.
The information discussed in this review highlights the important roles of Zn in enhancing drought tolerance in plants. However, there are still several questions which need further investigation. For instance, the role of Zn application on nutrient uptake and its interaction with other nutrients under drought stress is poorly understood. Therefore, future studies should address the interactions between Zn application and other nutrients’ uptake. Moreover, it would be useful to unfold the complexity of Zn signaling in response to drought conditions and to discover if Zn application directly or indirectly enhances the levels of endogenous hormones to counter the effects of drought stress. Meanwhile, genomic and transcriptomic investigations are direly needed to detect the stress responsive proteins and their roles in drought tolerance. The effects of Zn application on accumulations of osmolytes, especially proline, glycin-betaine, polyamines and polyphenols, are still not understood; therefore, future studies are required to quantify the effects of Zn on accumulation of these osmolytes under drought stress. Moreover, there is a paucity of information related to plant ability to adjust osmotically that originates from accumulation of osmolytes, as well as the antioxidative defense resulting from non-enzymatic antioxidants. Similarly, future studies should be undertaken to better understand the effects of Zn on the expression of both heat shock proteins and late embryogenesis abundant proteins under drought conditions. Lastly, more investigations are needed to elucidate the effects of Zn on expression of plant aquaporins and their role in plant water relations under drought stress.
Author Contributions
Conceptualization, M.U.H., M.U.C. and H.G.; resources, H.G.; data curation, Y.L., A.R., A.A.; writing—original draft preparation, M.U.H., M.A., M.U.C. and H.G.; writing—review and editing, L.B., B.S., T.H. and M.N.; supervision, H.G.; and funding acquisition, H.G. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported by the National Key R&D Program of China (2016YFD0300208); National Natural Science Foundation of China (41661070); and Key disciplines (construction) of ecology in the 13th Five-Year Plan of Jiangxi Agricultural University.
Acknowledgments
The National Key R&D Program of China (2016YFD0300208); National Natural Science Foundation of China (41661070) is gratefully acknowledged for providing funds for this work.
Conflicts of Interest
All authors declare no conflict of interest or state.
References
- Qados, A.M.S.A. Effect of salt stress on plant growth and metabolism of bean plant Vicia faba. J. Saudi Soc. Agric. Sci. 2011, 10, 7–15. [Google Scholar]
- Whitmore, T.C. The case of tropical rain forests. The sustainable development of forests: Aspiration and the reality. Naturzale 2000, 15, 13–15. [Google Scholar]
- Fahad, S.; Bajwa, A.A.; Nazir, U.; Anjum, S.A.; Farooq, A.; Zohaib, A.; Sadia, S.; Nasim, W.; Adkins, S.; Saud, S.; et al. Crop production under drought and heat stress: Plant responses and management options. Front. Plant Sci. 2017, 8, 1147. [Google Scholar] [CrossRef]
- Naeem, M.; Muhammad, S.N.; Rashid, A.; Muhammad, Z.I.; Muhammad, Y.A.; Yasir, H.; Fahad, S. Foliar calcium spray confers drought stress tolerance in maize via modulation of plant growth, water relations, proline content and hydrogen peroxide activity. Arch. Agron. Soil Sci. 2018, 64, 116–131. [Google Scholar] [CrossRef]
- Saud, S.; Chen, Y.; Fahad, S.; Hussain, S.; Na, L.; Xin, L.; Alhussien, S. Silicate application increases the photosynthesis and its associated metabolic activities in Kentucky bluegrass under drought stress and post-drought recovery. Environ. Sci. Pollut. Res. 2016, 23, 17647–17655. [Google Scholar] [CrossRef]
- Farooq, M.; Gogoi, N.; Barthakur, S.; Baroowa, B.; Bharadwaj, N.; Alghamdi, S.S.; Siddique, K.H.M. Drought stress in grain legumes during reproduction and grain filling. J. Agron. Crop Sci. 2017, 203, 81–102. [Google Scholar] [CrossRef]
- Sehgal, A.; Sita, K.; Kumar, J.; Kumar, S.; Singh, S.; Siddique, K.H.M.; Nayyar, H. Effects of drought, heat and their interaction on the growth, yield and photosynthetic function of lentil (Lens culinaris Medikus) genotypes varying in heat and drought sensitivity. Front. Plant Sci. 2017, 8, 1776. [Google Scholar] [CrossRef]
- Awasthi, R.; Kaushal, N.; Vadez, V.; Turner, N.C.; Berger, J.; Siddique, K.H.M.; Nayyar, H. Individual and combined effects of transient drought and heat stress on carbon assimilation and seed filling in chickpea. Funct. Plant Biol. 2014, 41, 1148. [Google Scholar] [CrossRef]
- Rahbarian, R.; Khavari-Nejad, R.; Ganjeali, A.; Bagheri, A.; Najafi, F. Drought stress effects on photosynthesis, chlorophyll fluorescence and water relations in tolerant and susceptible chickpea genotypes. Acta Biol. Cracov. Bot. 2011, 53, 47–56. [Google Scholar] [CrossRef]
- Samarah, N.H.; Alqudah, A.M.; Amayreh, J.A.; McAndrews, G.M. The effect of lateterminal drought stress on yield components of four barley cultivars. J. Agron. Crop Sci. 2009, 195, 427–441. [Google Scholar] [CrossRef]
- Gunes, A.; Cicel, N.; Inal, A.; Alpaslan, M.; Eraslan, F.; Guneri, E.; Guzelordu, T. Genotypic response of chickpea (Cicer arietinum L.) cultivars to drought stress implemented at pre- and post-anthesis stages and its relations with nutrient uptake and efficiency. Plant Soil Environ. 2006, 52, 368–376. [Google Scholar] [CrossRef]
- Mar, J.T.; Munn-Bosch, S.; Alegre, L. Redox regulation of water stress responses in field-grown plants. Role of hydrogen peroxide and ascorbate. Plant Physiol. Biochem. 2010, 48, 351–358. [Google Scholar]
- Wu, S.; Hu, C.; Tan, Q.; Nie, Z.; Sun, X. Effects of molybdenum on water utilization, antioxidative defense system and osmotic adjustment ability in winter wheat (Triticum aestivum) under drought stress. Plant Physiol. Biochem. 2014, 83, 365–374. [Google Scholar] [CrossRef] [PubMed]
- Cai, X.; Molden, D.; Mainuddin, M.; Sharma, B.; Mohyddin, A.; Karini, P. Producing more food with less water in the changing world: Assessment of water productivity in 10 major river basins. Water Int. 2013, 36, 421–462. [Google Scholar] [CrossRef]
- Tsonev, T.; Lidon, F.J.C. Zinc in plants—An overview. Emir. J. Food Agric. 2012, 24, 322–333. [Google Scholar]
- Mengel, K.; Kosegarten, H.; Kirkby, E.A.; Appel, T. Principles of Plant Nutrition; Springer: Berlin, Germany, 2001. [Google Scholar]
- Chattha, M.U.; Hassan, M.U.; Khan, I.; Chattha, M.B.; Mahmood, A.; Chattha, M.U.; Nawaz, M.; Subhani, M.N.; Kharal, M.; Khan, S. Biofortification of Wheat Cultivars to Combat Zinc Deficiency. Front. Plant Sci. 2017, 8, 281. [Google Scholar] [CrossRef] [PubMed]
- Hassan, M.U.; Chattha, M.U.; Ullah, A.; Khan, I.; Qadeer, A.; Aamer, M.; Khan, A.U.; Nadeem, F.; Khan, T.A. Agronomic biofortification to improve productivity and grain Zn concentration of bread wheat. Int. J. Agric. Biol. 2019, 21, 615–620. [Google Scholar]
- Mousavi, S.R.; Galavi, M.; Ahmadvand, G. Effect of zinc and manganese foliar application on yield, quality and enrichment on potato (Solanum tuberosum L.). Asian J. Plant Sci. 2007, 6, 1256–1260. [Google Scholar]
- Hajiboland, R.; Amirazad, F. Drought tolerance in Zn-deficient red cabbage (Brassica oleracea L. var. capitata f. rubra) plants. Hortic. Sci. 2010, 37, 88–98. [Google Scholar] [CrossRef]
- Sadoogh, F.S.; Shariatmadari, H.; Khoshgoftarmanesh, A.H.; Mosaddeghi, M.R. Adjusted nutrition of tomato with potassium and zinc in drought stress conditions induced by polyethylene glycol 6000 in hydroponic culture. J. Sci. Technol. Greenh. Cult. 2014, 18, 67–80. [Google Scholar]
- Eslami, M.; Dehghanzadeh, H. The effect of zinc on yield and yield components of sunflower (Helianthus annuus L.) under drought stress. Sci. J. Crop Sci. 2014, 3, 61–65. [Google Scholar]
- Zlatev, Z.; Lidon, F.C. An overview on drought induced changes in plant growth, water relations and photosynthesis. Emir. J. Food Agric. 2012, 24, 57–72. [Google Scholar]
- Mafakheri, A.; Siosemardeh, A.; Bahramnejad, B.; Struik, P.C.; Sohrabi, E. Effect of drought stress on yield, proline and chlorophyll contents in three chickpea cultivars. Aust. J. Crop Sci. 2010, 4, 580–585. [Google Scholar]
- Jaleel, C.A.; Manivannan, P.; Wahid, A.; Farooq, M.; Somasundaram, R.; Panneerselvam, R. Drought stress in plants: A review on morphological characteristics and pigments composition. Int. J. Agric. Biol. 2009, 11, 100–105. [Google Scholar]
- Nezhadahmadi, A.; Prodhan, Z.H.; Faruq, G. Drought tolerance in wheat. Sci. World J. 2013, 2013, 610721. [Google Scholar] [CrossRef] [PubMed]
- Osakabe, Y.; Osakabe, K.; Shinozaki, K.; Tran, L.S.P. Response of plants to water stress. Front. Plant Sci. 2014, 5, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Zare, M.; Azizi, M.H.; Bazrafshan, F. Effect of drought stress on some agronomic traits in ten barley (Hordeum vulgare) cultivars. Tech. J. Eng. Appl. Sci. 2011, 1, 57–62. [Google Scholar]
- Farooq, M.; Wahid, A.; Kobayashi, N.; Fujita, D.; Basra, S.M.A. Plant drought stress: Effects, mechanisms and management. Agron. Sustain. Dev. 2009, 29, 185–212. [Google Scholar] [CrossRef]
- Arbona, V.; Manzi, M.; de Ollas, C.; Gómez-Cadenas, A. Metabolomics as a tool to investigate abiotic stress tolerance in plants. Int. J. Mol. Sci. 2013, 14, 4885–4911. [Google Scholar] [CrossRef]
- Sapeta, H.; Costa, M.; Lourenc, T.; Marocod, J.; Van-der Linde, P.; Oliveiraa, M.M. Drought stress response in Jatropha curcas: Growth and physiology. Environ. Exp. Bot. 2013, 85, 76–84. [Google Scholar] [CrossRef]
- Tiwari, Y.K.; Yadav, S.K. Effect of high-temperature stress on ascorbate–glutathione cycle in maize. Agric. Res. 2020, 9, 179–187. [Google Scholar] [CrossRef]
- Xoconostle-Cazares, B.; Ramirez-Ortega, F.A.; Flores-Elenes, L.; Ruiz-Medrano, R. Drought tolerance in crop plants. Am. J. Plant Physiol. 2010, 5, 241–256. [Google Scholar] [CrossRef]
- Khan, M.A.; Iqbal, M.; Jameel, M.; Nazeer, W.; Shakir, S.; Aslam., M.T.; Iqbal, B. Potentials of molecular based breeding to enhance drought tolerance in wheat (Triticum aestivum L.). Afr. J. Biotechnol. 2011, 10, 11340–11344. [Google Scholar]
- Bhargava, S.; Sawant, K. Drought stress adaptation: Metabolic adjustment and regulation of gene expression. Plant Breed. 2013, 132, 21–32. [Google Scholar] [CrossRef]
- Ding, Y.; Tao, Y.; Zhu, C. Emerging roles of microRNAs in the mediation of srought stress responses in plants. J. Exp. Bot. 2013, 64, 3077–3086. [Google Scholar] [CrossRef]
- Shao, H.B.; Chu, L.Y.; Jaleel, C.A.; Zhao, C.X. Water-deficit stress-induced anatomical changes in higher plants. Comptes Rendus Biol. 2008, 331, 215–225. [Google Scholar] [CrossRef]
- Keyvan, S. The effects of drought stress on yield, relative water content, proline, soluble carbohydrates and chlorophyll of bread wheat cultivars. J. Anim. Plant Sci. 2010, 8, 1051–1060. [Google Scholar]
- Chernyad’ev, I.I. Effect of water stress on the photosynthetic apparatus of plants and the protective role of cytokinins: A review. Appl. Biochem. Microbiol. 2005, 41, 115–128. [Google Scholar] [CrossRef]
- Kheradmand, M.A.; Shahmoradzadeh, F.S.; Fatahi, E.; Raoofi, M.M. Effect of water stress on oil yield and some characteristics of Brassica napus. Int. Res. J. Appl. Basic Sci. 2014, 8, 1447–1453. [Google Scholar]
- Bernacchia, G.; Furini, A. Biochemical and molecular responses to water stress in resurrection plants. Physiol. Plant. 2004, 121, 175–181. [Google Scholar] [CrossRef]
- Finkelstein, R. Abscisic acid synthesis and response. Arab. Book Am. Soc. Plant Biol. 2013, 11, e0166. [Google Scholar] [CrossRef] [PubMed]
- Rahdari, P.; Hoseini, S.M. Drought stress: A review. Int. J. Agron. Plant Prod. 2012, 3, 443–446. [Google Scholar]
- Salehi-Lisar, S.Y.; Bakhshayeshan, A.H. Drought stress in plants: Causes, consequences, and tolerance. In Drought Stress Tolerance in Plants; Springer: Cham, Switzerland, 2016; Volume 1, pp. 1–16. [Google Scholar]
- Rana, R.M.; Rehman, S.U.; Ahmed, J.; Bilal, M. A comprehensive overview of recent advances in drought stress tolerance research in wheat (Triticum aestivum L.). Asian J. Agric. Biol. 2013, 1, 29–37. [Google Scholar]
- Gupta, N.; Ram, H.; Kumar, B. Mechanism of Zinc absorption in plants: Uptake, transport, translocation and accumulation. Rev. Environ. Sci. Biol. Technol. 2016, 15, 89–109. [Google Scholar] [CrossRef]
- Palmer, C.M.; Guerinot, M.L. Facing the challenges of Cu, Fe and Zn homeostasis in plants. Nat. Chem. Biol. 2009, 5, 333–340. [Google Scholar] [CrossRef] [PubMed]
- Alberts, B.; Johnson, A.; Walter, P.; Lewis, J.; Raff, M.; Roberts, K. Molecular Biology of the Cell, 5th ed.; Taylor & Francis, Garland Science: New York, NY, USA, 2007. [Google Scholar]
- Guerinot, M.L. The ZIP family of metal transporters. Biochim. Biophys. Acta (BBA) Biomembr. 2000, 1465, 190–198. [Google Scholar] [CrossRef]
- Clemens, S.; Palmgren, M.G.; Kramer, U. A long way ahead: Understanding and engineering plant metal accumulation. Trends Plant Sci. 2002, 7, 309–315. [Google Scholar] [CrossRef]
- Demidchik, V.; Davenport, R.J.; Tester, M. Non selective cation channels in plants. Annu. Rev. Plant Biol. 2002, 53, 67–107. [Google Scholar] [CrossRef]
- Xu, Y.; Wang, B.; Yu, J.; Ao, G.; Zhao, Q. Cloning and characterisation of ZmZLP1, a gene encoding an endoplasmic reticulum-localised Zn transporter in Zea mays. Funct. Plant Biol. 2010, 37, 194–205. [Google Scholar] [CrossRef]
- Kim, Y.Y.; Choi, H.; Segami, S.; Cho, H.T.; Martinoia, E.; Maeshima, M.; Lee, Y. AtHMA1 contributes to the detoxification of excess Zn (II) in Arabidopsis. Plant J. 2009, 58, 737–753. [Google Scholar] [CrossRef]
- Hanikenne, M.; Talke, I.N.; Haydon, M.J.; Lanz, C.; Nolte, A.; Motte, P.; Kroymann, J.; Weigel, D.; Kramer, U. Evolution of metal hyper accumulation required cis-regulatory changes and triplication of HMA4. Nature 2008, 453, 391–395. [Google Scholar] [CrossRef] [PubMed]
- Le-Jean, M.; Schikora, A.; Mari, S.; Briat, J.F.; Curie, C. A lossof-function mutation in AtYSL1 reveals its role in iron and nicotianamine seed loading. Plant J. 2005, 44, 769–782. [Google Scholar] [CrossRef] [PubMed]
- Desbrosses, F.A.G.; Voigt, K.; Schroder, A.; Arrivault, S.; Thomine, S.; Kramer, U. Arabidopsis thaliana MTP1 is a Zn transporter in the vacuolar membrane which mediates Zn detoxification and drives leaf Zn accumulation. FEBS Lett. 2005, 579, 4165–4174. [Google Scholar] [CrossRef]
- Song, W.Y.; Choi, K.S.; Kim, D.Y.; Geisler, M.; Park, J.; Vincenzetti, V.; Schellenberg, M.; Kim, S.H.; Lim, Y.P.; Noh, E.W.; et al. ArabidopsisPCR2 is a zinc exporter involved in both zinc extrusion and long-distance zinc transport. Plant Cell 2010, 22, 2237–2252. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Xu, Y.H.; Yi, H.Y.; Gong, J.M. Vacuolar membrane transporters OsVIT1 and OsVIT2 modulate iron translocation between flag leaves and seeds in rice. Plant J. 2012, 72, 400–410. [Google Scholar] [CrossRef]
- Sondergaard, T.E.; Schulz, A.; Palmgren, M.G. Energization of transport processes in plants. Roles of the plasma membrane H+-ATPase? Plant Physiol. 2004, 136, 2475–2482. [Google Scholar] [CrossRef]
- Sedghi, M.; Hadi, M.; Toluie, S.G. Effect of nano zinc oxide on the germination parameters of soybean seeds under drought stress. Ann. West Univ. Timis. Ser. Bio. 2013, 16, 73. [Google Scholar]
- Harris, D.; Rashid, A.; Arif, M.; Yunas, M. Alleviating micronutrient deficiencies in alkaline soils of the North-West Frontier Province of Pakistan: On-farm seed priming with zinc in wheat and chickpea. In Micronutrients in South and South East Asia; Andersen, P., Tuladhar, J.K., Karki, K.B., Maskey, S.L., Eds.; International Centre for Integrated Mountain Development International: Kathmandu, Nepal, 2005; pp. 143–151. [Google Scholar]
- Harris, D.; Rashid, A.; Miraj, G.; Arif, M.; Shah, H. On-farm seed priming with zinc sulphate solution—A cost-effective way to increase the maize yields of resource-poor farmers. Field Crops Res. 2007, 10, 119–127. [Google Scholar] [CrossRef]
- Cakmak, I. Enrichment of cereal grains with zinc: Agronomic or genetic biofortification. Plant Soil 2008, 30, 1–17. [Google Scholar] [CrossRef]
- Marschner, H. Mineral Nutrition of Higher Plants, 2nd ed.; Academic Press: New York, NY, USA, 1995. [Google Scholar]
- Hera, M.H.R.; Hossain, M.; Paul, A.K. Effect of foliar zinc spray on growth and yield of heat tolerant wheat under water stress. Int. J. Biol. Environ. Eng. 2018, 1, 10–16. [Google Scholar]
- Bajjii, M.; Kinet, J.M.; Lutts, S. The use of the electrolyte leakage method for assessing cell membrane stability as a water stress tolerance test in durum wheat. Plant Growth Regul. 2002, 36, 61–70. [Google Scholar] [CrossRef]
- Karam, F.; Lahoud, R.; Masaad., R.; Kabalan, R.; Breidi, J.; Chalita, C.; Rouphael, Y. Evaporation, seed yield and water use efficiency of drip irrigated sunflower under full and deficit irrigation conditions. Agric. Water Manag. 2007, 90, 213–223. [Google Scholar] [CrossRef]
- Bagci, S.A.; Ekiz, H.; Yilmaz, A.; Cakmak, I. Effect of zinc deficiency and drought on grain yield of field-grown wheat cultivars in Central Anatolia. J. Agron. Crop Sci. 2007, 193, 198–206. [Google Scholar] [CrossRef]
- Babaeian, M.; Heidari, M.; Ghanbari, A. Effect of water stress and foliar micronutrient application on physiological characterstics and nutrient uptake in sunflower (Helianthus annuus L.). Iran. J. Crop Sci. 2010, 12, 311–391. [Google Scholar]
- Karim, M.; Zhang, Y.Q.; Zhao, R.R.; Chen, X.P.; Zhang, F.S.; Zou, C.Q. Alleviation of drought stress in winter wheat by late foliar application of zinc, boron, and manganese. J. Plant Nutr. Soil Sci. 2012, 175, 142–151. [Google Scholar] [CrossRef]
- Sultana, S.; Naser, H.M.; Shil, N.C.; Akhter, S.; Begum, R.A. Effect of foliar application of zinc on yield of wheat grown by avoiding irrigation at different growth stages. Bangladesh J. Agric. Res. 2016, 41, 323–334. [Google Scholar] [CrossRef]
- Sánchez-Blanco, M.J.; Rodríguez, P.; Morales, M.A.; Ortuño, M.F.; Torrecillas, A. Comparative growth and water relation of Cistusalbidus and Cistusmons peliensis plants during water deficit conditions and recovery. Plant Sci. 2002, 162, 107–113. [Google Scholar] [CrossRef]
- Velu, G.; Palanisami, K. Impact of moisture stress on growth and yield of sunflower. Madras Agric. J. 2002, 88, 660–665. [Google Scholar]
- Khan, R.; Gul, S.; Hamayun, M.; Shah, M.; Sayyed, A.; Ismail, H.; Begum, A.; Gul, H. Effect of foliar application of zinc and manganese on growth and some biochemical constituents of Brassica junceae grown under water stress. Am.-Eurasian J. Agric. Environ. Sci. 2016, 16, 984–997. [Google Scholar]
- Arough, Y.; Seyed-Sharifi, R.; Seyed-Sharifi, R. Bio fertilizers and zinc effects on some physiological parameters of triticale under water-limitation condition. J. Plant Interact. 2016, 11, 167–177. [Google Scholar] [CrossRef]
- Grewal, H.S.; Williams, R. Zinc nutrition affects alfalfa responses to water stress and excessive moisture. J. Plant Nutri. 2000, 23, 949–962. [Google Scholar] [CrossRef]
- Steudle, E. Water uptake by roots: Effects of water deficit. J. Exp. Bot. 2002, 51, 1531–1542. [Google Scholar] [CrossRef] [PubMed]
- Gadallah, M.A.A.; Ramadan, T. Effects of zinc and salinity on growth and anatomical structure of Carthamus tinctorius L. Biolgia Plant. 1997, 39, 411–418. [Google Scholar] [CrossRef]
- Gadallah, M.A.A. Effects of indole-3-acetic acid and zinc on growth, osmotic potential and soluble carbon and nitrogen components of soybean plants growing under water deficit. J. Arid Environ. 2000, 44, 451–467. [Google Scholar] [CrossRef]
- Wang, H.; Liu, R.L.; Jin, J.Y. Effects of zinc and soil moisture on photosynthetic rate and chlorophyll fluorescence parameters of maize. Biol. Plant. 2009, 53, 191–194. [Google Scholar] [CrossRef]
- Vazin, F. Effect of zinc sulfate on quantitative and qualitative characteristics of corn (Zea mays) in drought stress. Cercet. Agron. Mold. 2012, 45, 15–24. [Google Scholar] [CrossRef]
- Zafar, S.; Nasri, M.; Moghadam, H.R.T.; Zahedi, H. Effect of zinc and sulfur foliar applications on physiological characteristics of sunflower (Helianthus annuus L.) under water deficit stress. Int. J. Biosci. 2014, 5, 87–96. [Google Scholar]
- Yavas, I.; Unay, A. Effects of zinc and salicylic acid on wheat under drought stress. J. Anim. Plant Sci. 2016, 26, 1012–1101. [Google Scholar]
- Ibrahim, S.A.; Desoky, E.; Elrys, A.S. Influencing of water stress and micronutrients on physio-chemical attributes, yield and anatomical features of Common Bean plants (Phaseolus vulgaris L.). Egypt. J. Agron. 2017, 39, 251–265. [Google Scholar] [CrossRef]
- Sakya, A.T.; Sulistyaningsih, E.; Indradewa, D.; Purwanto, B.H. Stomata character and chlorophyll content of tomato in response to Zn application under drought condition. IOP Conf. Ser. Earth Environ. Sci. 2018, 142, 012033. [Google Scholar] [CrossRef]
- Ullah, A.; Romdhane, L.; Rehman, A.; Farooq, M. Adequate zinc nutrition improves the tolerance against drought and heat stresses in chickpea. Plant Physiol. Biochem. 2019, 143, 11–18. [Google Scholar] [CrossRef] [PubMed]
- Mahmood, A.; Kanwal, H.; Kausar, A.; Ilyas, A.; Akhter, N.; Ilyas, M.; Nisa, Z.; Khalid, H. Seed priming with zinc modulate growth, pigments and yield of chickpea (Cicer Arietinum L.) under water deficit conditions. Appl. Ecol. Environ. Res. 2019, 17, 147–160. [Google Scholar] [CrossRef]
- Pavia, I.; Roque, J.; Rocha, L.; Ferreira, H.; Castro, C.; Carvalho, A.; Silva, E.; Brito, C.; Gonçalves, A.; Lima-Brito, J.; et al. Zinc priming and foliar application enhances photoprotection mechanisms in drought-stressed wheat plants during anthesis. Plant Physiol. Biochem. 2019, 140, 27–42. [Google Scholar] [CrossRef]
- Liu, X.Z.; Huang, B.R. Heat stress injury in relation to membrane lipid peroxidation in creeping bent grass. Crop Sci. 2000, 40, 503–510. [Google Scholar] [CrossRef]
- Sreenivasulu, N.; Grimm, B.; Wobus, U.; Weshke, W. Differential response of antioxidant compounds to salinity stress in salt-tolerant and salt-sensitive seedlings of foxtail millet (Setaria italica). Physiolgia Plant. 2000, 109, 435–442. [Google Scholar] [CrossRef]
- Ghanepour, S.; Shakiba, M.R.; Toorchi, M.; Oustan, S. Role of Zn nutrition in membrane stability, leaf hydration status, and growth of common bean grown under soil moisture stress. J. Biodivers. Environ. Sci. 2015, 6, 9–20. [Google Scholar]
- Blokhina, O.; Virolainen, E.; Fagersttedt, K.V. Antioxidants, oxidative damage, and oxygen deprivation stress. Ann. Bot. 2003, 91, 179–194. [Google Scholar] [CrossRef]
- Vallee, B.L.; Falchuk, K.H. The biochemical basis of zinc physiology. Physiol. Rev. 1993, 73, 79–118. [Google Scholar] [CrossRef]
- DaCosta, M.; Huang, B.R. Osmotic adjustment associated with variation in bent grass tolerance to drought stress. J. Am. Soc. Hortic. Sci. 2006, 131, 338–344. [Google Scholar] [CrossRef]
- Blum, A. Drought resistance, water-use efficiency, and yield potential, are they compatible, dissonant, or mutually exclusive? Crop Pasture Sci. 2005, 56, 1159–1168. [Google Scholar] [CrossRef]
- Kiani, S.P.; Talia, P.; Maury, P.; Grieu, P.; Heinz, R.; Perrault, A.; Nishinakamasu, V.; Hopp, E.; Gentzbittel, L.; Paniego, N. Genetic analysis of plant water status and osmotic adjustment in recombinant inbred lines of sunflower under two water treatments. Plant Sci. 2007, 172, 773–787. [Google Scholar] [CrossRef]
- Mousavi, S.R. Zinc in crop production and interaction with phosphorus. Aust. J. Basic Appl. Sci. 2011, 9, 1503–1509. [Google Scholar]
- Xu, D.Q.; Huang, J.; Guo, S.Q.; Yang, X.; Bao, Y.M.; Tang, H.J.; Zhang, H.S. Overexpression of a TFIIIA-type zinc finger protein gene ZFP252 enhances drought and salt tolerance in rice (Oryza sativa L.). FEBS Lett. 2008, 582, 1037–1043. [Google Scholar] [CrossRef] [PubMed]
- Luo, X.; Bai, X.; Zhu, D.; Li, Y.; Ji, W.; Cai, H.; Zhu, Y. GsZFP1, a new Cys2/His2-type zinc-finger protein, is a positive regulator of plant tolerance to cold and drought stress. Planta 2012, 235, 1141–1155. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.; Hu, C.; Tan, Q.; Li, L.; Shi, K.; Zheng, Y.; Sun, X. Drought stress tolerance mediated by zinc-induced antioxidative defense and osmotic adjustment in cotton (Gossypium hirsutum). Acta Physiol. Plant. 2015, 37, 167. [Google Scholar] [CrossRef]
- Hasegawa, P.M.; Bressan, R.A.; Zhu, J.K.; Bohnert, H.J. Plant cellular and molecular responses to high salinity. Annu. Rev. Plant Physiol. Mol. Biol. 2000, 51, 463–499. [Google Scholar] [CrossRef] [PubMed]
- Aziz, E.; Ezz El-Din, A.; Omer, E.A. Influence of zinc and iron on plant growth and chemical constituents of Cymbopogon citrates L. grown in newly reclaimed land. Int. J. Acad. Res. 2010, 2, 278–283. [Google Scholar]
- Dehnavi, M.M.; Sheshbahre, M.J. Soybean leaf physiological responses to drought stress improved via enhanced seed zinc and iron concentrations. J. Plant Process. Funct. 2017, 5, 13–21. [Google Scholar]
- Dehnavi, M.M.; Misagh, M.; Yadavi, A.; Merajipoor, M. Physiological responses of sesame (Sesamum indicum L.) to foliar application of boron and zincunder drought stress. J. Plant Process Funct. 2017, 6, 27–36. [Google Scholar]
- Zushi, K. Comparison of chemical composition contents of tomato fruit grown under water and salinity stresses. J. SHITA 2005, 17, 128–136. [Google Scholar] [CrossRef]
- Mishra, L.K.; Abidi, A.B. Phosphorus-zinc interaction: Effects on yield components, biochemical composition and bread making qualities of wheat. World Appl. Sci. 2010, 10, 568–573. [Google Scholar]
- Bharti, K.; Pandey, N.; Shankhdhar, D.; Srivastava, P.C.; Shankhdhar, S.C. Improving nutritional quality of wheat through soil and foliar zinc application. Plant Soil Environ. 2013, 59, 348–352. [Google Scholar] [CrossRef]
- Sharma, P.N.; Tripathi, A.; Bisht, S.S. Zinc requirement for stomatal opening in cauliflower. Plant Physiol. 1995, 107, 751–756. [Google Scholar] [CrossRef] [PubMed]
- Khan, H.R.; McDonald, G.K.; Rengel, Z. Zinc fertilization and water stress affects plant water relations, stomatal conductance and osmotic adjustment in chickpea (Cicer arientinum L.). Plant Soil 2004, 267, 271–284. [Google Scholar] [CrossRef]
- Khan, H.R.; McDonald, G.K.; Rengel, Z. Zn fertilization improves water use efficiency, grain yield and seed Zn content in chickpea. Plant Soil 2003, 249, 389–400. [Google Scholar] [CrossRef]
- Hall, A.E.; Richards, R.A.; Condon, A.G.; Wright, G.C.; Farquhar, G.D. Carbon isotope discrimination and plant breeding. Plant Breed. Rev. 1994, 12, 81–113. [Google Scholar]
- Ahmed, N.; Ahmad, F.; Abid, M.; Ullah, M.A. Impact of zinc fertilization on gas exchange characteristics and water use efficiency of cotton crop under arid environment. Pak. J. Bot. 2009, 41, 2189–2197. [Google Scholar]
- Ahanger, M.A.; MoradTalab, N.; Fathi Abd-Allah, E.; Ahmad, P.; Hajiboland, R. Plant growth under drought stress: Significance of mineral nutrients. In Water Stress and Crop Plants: A Sustainable Approach; John Wiley & Sons, Ltd.: West Sussex, UK, 2016; Volume 2. [Google Scholar]
- Hajiboland, R. Effect of Micronutrient Deficiencies on Plants Stress Responses. In Abiotic Stress Responses in Plants; Ahmad, P., Prasad, M.N.V., Eds.; Springer: New York, NY, USA, 2012; pp. 281–330. [Google Scholar]
- Hong, W.; Ji-Yun, J. Effects of zinc deficiency and drought on plant growth and metabolism of reactive oxygen species in maize (Zea mays L.). Agric. Sci. China 2007, 6, 988–995. [Google Scholar]
- Babaeian, M.; Piri, I.; Tavassoli, A.; Esmaeilian, Y.; Gholami, H. Effect of water stress and micronutrients (Fe, Zn and Mn) on chlorophyll fluorescence, leaf chlorophyll content and sunflower nutrient uptake in Sistan region. Afr. J. Agric. Res. 2011, 6, 3526–3531. [Google Scholar]
- Waraich, E.A.; Ahmad, R.; Ashraf, M.Y. Role of mineral nutrition in alleviation of drought stress in plants. Aust. J. Crop Sci. 2011, 5, 764. [Google Scholar]
- Moradi, L.; Ehsanzadeh, P. Effects of Cd on photosynthesis and growth of safflower (Carhamus tinctorius L.) genotypes. Photosynthetica 2015, 53, 506–518. [Google Scholar] [CrossRef]
- Peleg, Z.; Saranga, Y.; Yazici, A.; Fahima, T.; Ozturk, L.; Cakmak, I. Grain zinc, iron and protein concentrations and zinc-efficiency in wild emmer wheat under contrasting irrigation regimes. Plant Soil 2008, 306, 57–67. [Google Scholar] [CrossRef]
- Ma, D.; Sun, D.; Wang, C.; Ding, H.; Qin, H.; Hou, J.; Huang, X.; Xie, Y.; Guo, T. Physiological responses and yield of wheat plants in zinc-mediated alleviation of drought stress. Front. Plant Sci. 2017, 8, 860. [Google Scholar] [CrossRef] [PubMed]
- Balashouri, P. Effect of zinc on germination, growth, pigment content and phytomass of Vigna radiata and Sorghum bicolor. J. Ecobiol. 1995, 7, 109–114. [Google Scholar]
- Cakmak, I. Tansley Review No. 111–possible roles of zinc in protecting plant cells from damage by reactive oxygen species. New Phytol. 2000, 146, 185–205. [Google Scholar] [CrossRef]
- Kaya, C.; Higgs, D. Response of tomato (Lycopersicon esculentum L.) cultivars to application of zinc when grown in sand culture at low zinc. Sci. Hortic. 2002, 93, 53–64. [Google Scholar] [CrossRef]
- Ayad, H.S.; Reda, F.; Abdalla, M.S.A. Effect of putrescine and zinc on vegetative growth, photosynthetic pigments, lipid peroxidation and essential oil content of geranium (Pelargonium graveolens L.). World J. Agri. Sci. 2010, 6, 601–608. [Google Scholar]
- Taiz, L.; Zeiger, E. Plant Physiology, 4th ed.; Sinauer Associates Inc. Publishers: Sunderland, MA, USA, 2006. [Google Scholar]
- Bennett, J.P.; Skoog, F. Preliminary Experiments on the Relation of Growth-promoting Substances to the Rest Period in Fruit Trees. Plant Physiol. 2002, 13, 219–225. [Google Scholar] [CrossRef]
- Moghadam, H.R.T.; Zahedi, H.; Ashkiani, A. Effect of zinc foliar application on auxin and gibberellin hormones and catalase and superoxide dismutase enzyme activity of corn (Zea mays L) under water stress. Maydica 2013, 58, 218–223. [Google Scholar]
- Jamil, M.; Ahamd, M.; Anwar, F.; Zahir, Z.A.; Kharal, M.A.; Nazli, F. Inducing drought tolerance in wheat through combined use of L-tryptophan and pseudomonas fluorescens. Pak. J. Agric. Sci. 2018, 55, 331–337. [Google Scholar]
- Tan, D.X.; Manchester, L.C.; Esteban-Zubero, E.; Zhou, Z.; Reiter, R.J. Melatonin as a potent and inducible endogenous antioxidant: Synthesis and metabolism. Molecules 2015, 20, 18886–18906. [Google Scholar] [CrossRef] [PubMed]
- Awan, I.U.; Baloch, M.S.; Sadozai, N.S.; Sulemani, M.Z. Stimulatory effect of GA3 and IAA on ripening process, kernel development and quality of rice. Pak. J. Biol. Sci. 1999, 2, 410–412. [Google Scholar]
- Fleta-Soriano, E.; Diaz, L.; Bonet, E.; Munne-Bosch, S. Melatonin may exert a protective role against drought stress in maize. J. Agron. Crop Sci. 2017, 203, 286–294. [Google Scholar] [CrossRef]
- Sharif, R.; Xie, C.; Zhang, H.; Arnao, M.; Ali, M.; Ali, Q.; Muhammad, I.; Shalmani, A.; Nawaz, M.A.; Chen, P. Melatonin and its effects on plant systems. Molecules 2018, 23, 2352. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.; Song, F.; Guo, J.; Zhu, X.; Liu, S.; Liu, F.; Li, X. Nano-ZnO-Induced Drought Tolerance Is Associated with Melatonin Synthesis and Metabolism in Maize. Int. J. Mol. Sci. 2020, 21, 782. [Google Scholar] [CrossRef] [PubMed]
- Reddy, A.R.; Chaitanya, K.V.; Vivekanandan, M. Drought-induced responses of photosynthesis and antioxidant metabolism in higher plants. J. Plant Physiol. 2004, 161, 1189–1202. [Google Scholar] [CrossRef]
- Gratao, P.L.; Monteiro, C.C.; Antunes, A.M.; Peres, L.; Azevedo, R.A. Acquired tolerance of tomato (Lycopersicon esculentum cv. Micro-Tom) plants to cadmium-induced stress. Ann. Appl. Biol. 2008, 153, 321–333. [Google Scholar] [CrossRef]
- Mousavi, S.R.; Galavi, M.; Rezaei, M. Zinc importance for crop production—A review. Int. J. Agron. Plant Prod. 2013, 4, 64–68. [Google Scholar]
- Li, G.; Tai, F.; Zheng, Y.; Luo, J.; Gong, S.; Zhang, Z.; Li, X. Two cotton Cys2/His2-type zinc-finger proteins, GhDi19-1 and GhDi19-2, are involved in plant response to salt/drought stress and abscisic acid signaling. Plant Mol. Biol. 2010, 74, 437–452. [Google Scholar] [CrossRef]
- Thounaojam, T.C.; Panda, P.; Choudhury, S.; Patra, H.K.; Panda, S.K. Zinc ameliorates copper-induced oxidative stress in developing rice (Oryza sativa L.) seedlings. Protoplasma 2014, 251, 61–69. [Google Scholar] [CrossRef]
- Chen, Z.; Young, T.E.; Ling, J.; Chang, S.C.; Gallie, D.R. Increasing vitamin C content of plants through enhanced ascorbate recycling. Proc. Natl. Acad. Sci. USA 2003, 100, 3525–3530. [Google Scholar] [CrossRef] [PubMed]
- Jiang, L.; Tang, Y.Z.; Zhang, R.X.; Wang, W. Effect of fertilizer activating agent on the ascorbate-glutathione cycle and potassium content in tobacco leaves. J. Nanjing Agric. Univ. 2000, 23, 13–16. [Google Scholar]
- Tavallali, V.; Rahemi, M.; Eshghi, S.; Kholdebarin, B.; Ramezanian, A. Zinc alleviates salt stress and increase antioxidant enzyme activity in the leaves of pistachio (Pistacia vera L. ‘Badami’) seedings. Turk. J. Agric. For. 2010, 34, 349–359. [Google Scholar]
- Song, C.Z.; Liu, M.Y.; Meng, J.F.; Chi, M.; Xi, Z.M.; Zhang, Z.W. Promoting effect of foliage sprayed zinc sulfate on accumulation of sugar and phenolics in berries of Vitis vinifera cv. Merlot growing on zinc deficient soil. Molecules 2015, 20, 2536–2554. [Google Scholar] [CrossRef]
- Liu, Y.J.; Yuan, Y.; Liu, Y.Y.; Liu, Y.; Fu, J.J.; Zheng, J.; Wang, G.Y. Gene families of maize glutathione-ascorbate redox cycle respond differently to abiotic stresses. J. Plant Physiol. 2012, 169, 183–192. [Google Scholar] [CrossRef]
- Wahid, A.; Gelani, S.; Ashraf, M.; Foolad, M.R. Heat tolerance in plants: An overview. Environ. Exp. Bot. 2007, 61, 199–223. [Google Scholar] [CrossRef]
- Zhang, X.; Zhang, B.; Li, M.J.; Yin, X.M.; Huang, L.F.; Cui, Y.C.; Wang, M.L.; Xia, X. OsMSR15 encoding a rice C2H2-type zinc finger protein confers enhanced drought tolerance in transgenic Arabidopsis. J. Plant Biol. 2016, 59, 271–281. [Google Scholar] [CrossRef]
- Han, G.; Lu, C.; Guo, J.; Qiao, Z.; Sui, N.; Qiu, N.; Wang, B. C2H2 Zinc Finger Proteins: Master Regulators of Abiotic Stress Responses in Plants. Front. Plant Sci. 2020, 11, 115. [Google Scholar] [CrossRef]
- Huang, X.Y.; Chao, D.Y.; Gao, J.P.; Zhu, M.Z.; Shi, M.; Lin, H.X. A previously unknown zinc finger protein, DST, regulates drought and salt tolerance in rice via stomatal aperture control. Genes Dev. 2009, 23, 1805–1817. [Google Scholar] [CrossRef]
- Sakamoto, H.; Maruyama, K.; Sakuma, Y.; Meshi, T.; Iwabuchi, M.; Shinozaki, K.; Yamaguchi-Shinozaki, K. Arabidopsis Cys2/His2-type zinc-finger proteins function as transcription repressors under drought, cold, and high-salinity stress conditions. Plant Physiol. 2004, 136, 2734–2746. [Google Scholar] [CrossRef]
- Coca, M.A.; Almoguera, C.; Jordano, J. Expression of sunflower low molecular weight heat shock proteins during embryogenesis and persistence after germination: Localization and possible functional implications. Plant Mol. Biol. 1994, 25, 479–492. [Google Scholar] [CrossRef] [PubMed]
- Mahajan, S.; Tuteja, N. Cold, salinity and drought stresses: An overview. Arch. Biochem. Biophys. 2005, 444, 139–158. [Google Scholar] [CrossRef] [PubMed]
- Heinen, R.B.; Ye, Q.; Chaumont, F. Role of aquaporins in leaf physiology. J. Exp. Bot. 2009, 60, 2971–2985. [Google Scholar] [CrossRef] [PubMed]
- Lian, H.L.; Yu, X.; Ye, Q.; Ding, X.S.; Kitagawa, Y.; Kwak, S.S.; Su, W.A.; Tang, Z.C. The role of aquaporin RWC3 in drought avoidance in rice. Plant Cell Physiol. 2004, 45, 481–489. [Google Scholar] [CrossRef] [PubMed]
- Galmes, J.; Pou, A.; Alsina, M.M.; Tomas, M.; Medrano, H.; Flexas, J. Aquaporin expression in response to different water stress intensities and recovery in Richter-110 (Vitis sp.): Relationship with Eco physiological status. Planta 2007, 226, 671–681. [Google Scholar] [CrossRef] [PubMed]
- Kaldenhoff, R.; Ribas-Carbo, M.; Flexas, J.; Lovisolo, C.; Heckwolf, M.; Uehlein, N. Aquaporins and plant water balance. Plant Cell Environ. 2008, 31, 658–666. [Google Scholar] [CrossRef]
- Danielson, J.Å.H.; Johanson, U. Unexpected complexity of the aquaporin gene family in the moss Physcomitrella patens. BMC Plant Biol. 2008, 8, 45. [Google Scholar] [CrossRef]
- Ishibashi, K.; Hara, S.; Kondo, S. Aquaporin water channels in mammals. Clin. Exp. Nephrol. 2009, 13, 107–117. [Google Scholar] [CrossRef]
- Henzler, T.; Ye, Q.; Steudle, E. Oxidative gating of water channels (aquaporins) in Chara by hydroxyl radicals. Plant Cell Environ. 2004, 27, 1184–1195. [Google Scholar] [CrossRef]
- Tyerman, S.D.; Bohnert, H.J.; Maurel, C.; Steudle, E.; Smith, J.A.C. Plant aquaporins: Their molecular biology, biophysics and significance for plant water relations. J. Exp. Bot. 1999, 50, 1055–1071. [Google Scholar] [CrossRef]
- Rucinska, S.R. Water relations in plants subjected to heavy metal. Acta Physiol. Plant. 2016, 38, 257. [Google Scholar] [CrossRef]
- Fatemi, H.; Zaghdoud, C.; Nortes, P.A.; Carvajal, M.; Martínez-Ballesta, M.D.C. Differential Aquaporin Response to Distinct Effects of Two Zn Concentrations after Foliar Application in Pak Choi (Brassica rapa L.) Plants. Agronomy 2020, 10, 450. [Google Scholar] [CrossRef]
- Aroca, R.; Ferrante, A.; Vernieri, P.; Chrispeels, M.J. Drought, abscisic acid and transpiration rate effects on the regulation of PIP aquaporin gene expression and abundance in Phaseolus vulgaris plants. Ann. Bot. 2006, 98, 1301–1310. [Google Scholar] [CrossRef] [PubMed]
- Maurel, C.; Verdoucq, L.; Luu, D.T.; Santoni, V. Plant aquaporins: Membrane channels with multiple integrated functions. Annu. Rev. Plant Biol. 2008, 59, 595–624. [Google Scholar] [CrossRef] [PubMed]
- Vandeleur, R.K.; Sullivan, W.; Athman, A.; Jordans, C.; Gilliham, M.; Kaiser, B.N.; Tyerman, S.D. Rapid shoot-to-root signalling regulates root hydraulic conductance via aquaporins. Plant Cell Environ. 2014, 37, 520–538. [Google Scholar] [CrossRef]
- Farooq, M.; Ullah, A.; Rehman, A.; Nawaz, A.; Nadeem, A.; Wakeel, A.; Nadeem, F.; Siddique, K.H.M. Application of zinc improves the productivity and biofortification of fine grain aromatic rice grown in dry seeded and puddled transplanted production systems. Field Crop Res. 2018, 216, 53–62. [Google Scholar] [CrossRef]
- Farooq, M.; Wahid, A.; Kadambot, H.; Siddique, M. Micronutrient application through seed treatment. J. Soil Sci. Plant Nutr. 2012, 12, 125–142. [Google Scholar] [CrossRef]
- Rehman, A.; Farooq, M. Zinc seed coating improves the growth, grain yield and grain biofortification of bread wheat. Acta Physiol. Plant. 2016, 3, 238. [Google Scholar] [CrossRef]
- Grzebisz, W.; Wronska, M.; Diatta, J.B.; Dullin, P. Effect of zinc foliar application at early stages of maize growth on patterns of nutrients and dry matter accumulation by the canopy. Part I. Zinc uptake patterns and its redistribution among maize organs. J. Elem. 2008, 13, 17–28. [Google Scholar]
- Baloch, Q.B.; Chachar, Q.I.; Tareen, M.N. Effect of foliar application of macro and micro nutrients on production of green chillies (Capsicum annuum L.). J. Agric. Technol. 2008, 4, 177–184. [Google Scholar]
- Leach, K.; Hameleers, A. The effects of a foliar spray containing phosphorus and zinc on the development, composition and yield of forage maize. Grass For Sci. 2008, 56, 3113–3115. [Google Scholar] [CrossRef]
- Thalooth, A.T.; Tawfik, M.M.; Mohamed, H.M. A comparative study on the effect of foliar application of zinc, potassium and magnesium on growth, yield and some chemical constituents of mungbean plants grown under water stress conditions. World J. Agric. Sci. 2006, 2, 37–46. [Google Scholar]
- Yadavi, A.; Aboueshaghi, R.S.; Dehnavi, M.M.; Balouchi, H. Effect of micronutrients foliar application on grain qualitative characteristics and some physiological traits of bean (Phaseolus vulgaris L.) under drought stress. Indian J. Fundam. Appl. Life Sci. 2014, 4, 124–131. [Google Scholar]
- Cakmak, I.; Pfeiffer, W.H.; Mcclafferty, B. Biofortification of durum wheat with zinc and iron. Cereal Chem. 2010, 87, 10–20. [Google Scholar] [CrossRef]
- Tariq, A.; Randhawa, M.A.; Anjum, S.A.; Nadeem, M.; Naeem, M. Exploring the role of zinc in maize (Zea mays L.) through soil and foliar application. Univers. J. Agric. Res. 2015, 3, 69–75. [Google Scholar]
- Khan, H.; Hassan, Z.U.; Maitlo, A.A. Yield and micronutrients content of bread wheat (Triticum aestivum L.) under a multi-nutrient fertilizer-Hal-tonic. Int. J. Agric. Biol. 2006, 8, 366–370. [Google Scholar]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).