Key Strategies to Optimize Outcomes in Mild-to-Moderate Ulcerative Colitis
Abstract
1. Introduction
2. Current Recommendations in Mild-to-Moderate UC
3. Drawbacks and Challenges in Mild-to-Moderate UC Management
4. Key Strategies to Improve the Management of Mild-to-Moderate UC
4.1. 5-ASA Optimization
4.1.1. Once-Daily (OD) Dosing
4.1.2. Combination of Oral and Rectal 5-ASA
4.1.3. Increasing Dose of 5-ASA
4.2. Budesonide MMX Integration in the Therapeutic Armamentarium
4.3. Patient Stratification for Earlier Intervention
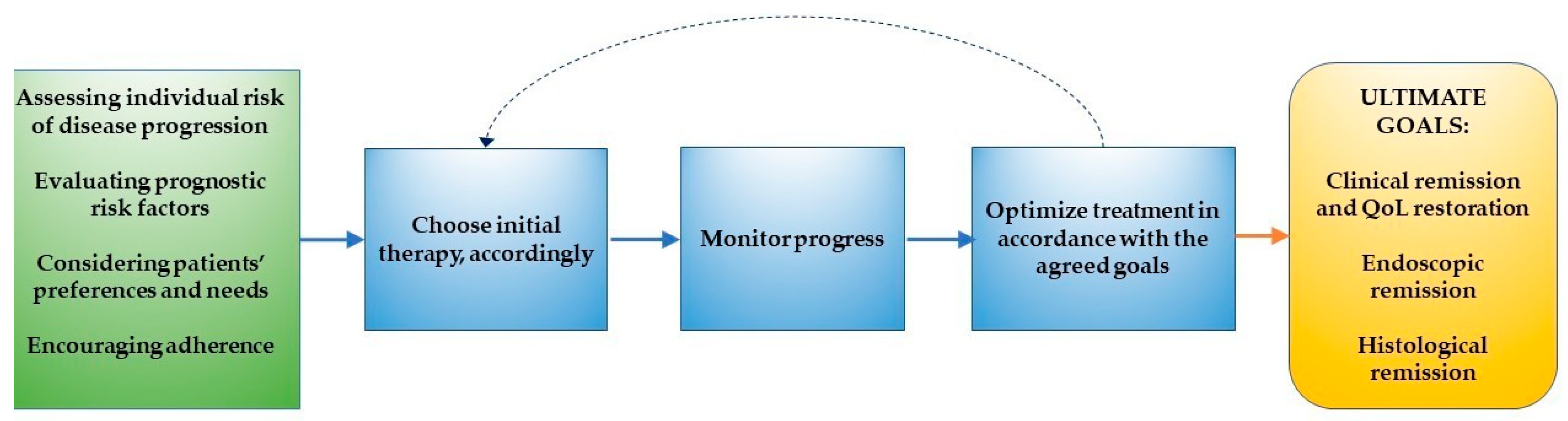
4.4. Shared Decision-Making and Patient Involvement
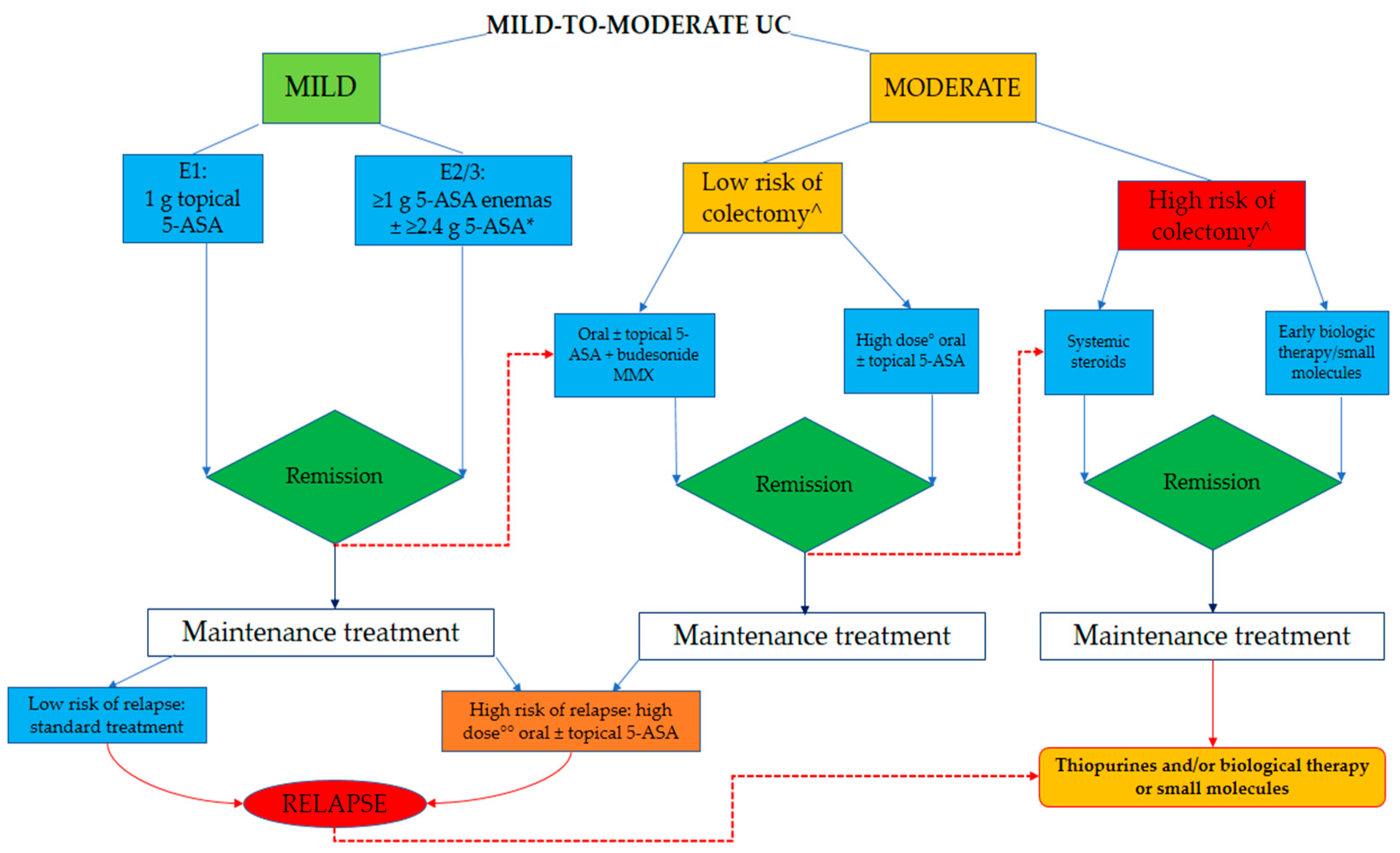
5. A Proposed Algorithm for Practical Guidance
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Ungaro, R.; Mehandru, S.; Allen, P.B.; Peyrin-Biroulet, L.; Colombel, J.F. Ulcerative colitis. Lancet 2017, 389, 1756–1770. [Google Scholar] [CrossRef]
- Roda, G.; Narula, N.; Pinotti, R.; Skamnelos, A.; Katsanos, K.H.; Ungaro, R.; Burisch, J.; Torres, J.; Colombel, J.F. Systematic review with meta-analysis: Proximal disease extension in limited ulcerative colitis. Aliment. Pharmacol. Ther. 2017, 45, 1481–1492. [Google Scholar] [CrossRef] [PubMed]
- Reinisch, W.; Sandborn, W.J.; Bala, M.; Yan, S.; Feagan, B.G.; Rutgeerts, P.; Radford-Smith, G.; Xu, S.; Eisenberg, D.; Olson, A.; et al. Response and remission are associated with improved quality of life, employment and disability status, hours worked, and productivity of patients with ulcerative colitis. Inflamm. Bowel Dis. 2007, 13, 1135–1140. [Google Scholar] [CrossRef] [PubMed]
- Ramos, A.; Calvet, X.; Sicilia, B.; Vergara, M.; Figuerola, A.; Motos, J.; Sastre, A.; Villoria, A.; Gomollón, F. IBD-related work disability in the community: Prevalence, severity and predictive factors. A cross-sectional study. United Eur. Gastroenterol. J. 2015, 3, 335–342. [Google Scholar] [CrossRef] [PubMed]
- Yashiro, M. Ulcerative colitis-associated colorectal cancer. World J. Gastroenterol. 2014, 20, 16389–16397. [Google Scholar] [CrossRef] [PubMed]
- Magro, F.; Gionchetti, P.; Eliakim, R.; Ardizzone, S.; Armuzzi, A.; Barreiro-de Acosta, M.; Burisch, J.; Gecse, K.B.; Hart, A.L.; Hindryckx, P.; et al. Third European evidence-based consensus on diagnosis and management of ulcerative colitis. Part 1: Definitions, diagnosis, extra-intestinal manifestations, pregnancy, cancer surveillance, surgery, and ileo-anal pouch disorders. J. Crohn’s Colitis 2017, 11, 649–670. [Google Scholar] [CrossRef]
- Silverberg, M.S.; Satsangi, J.; Ahmad, T.; Arnott, I.D.R.; Bernstein, C.N.; Brant, S.R.; Caprilli, R.; Colombel, J.F.; Gasche, C.; Geboes, K.; et al. Toward an integrated clinical, molecular and serological classification of inflammatory bowel disease: Report of a Working Party of the 2005 Montreal World Congress of Gastroenterology. Can. J. Gastroenterol. 2005, 19 (Suppl. A), 5A–36A. [Google Scholar] [CrossRef]
- Satsangi, J.; Silverberg, M.S.; Vermeire, S.; Colombel, J.F. The Montreal classification of inflammatory bowel disease: Controversies, consensus, and implications. Gut 2006, 55, 749–753. [Google Scholar] [CrossRef]
- Truelove, S.C.; Witts, L.J. Cortisone in ulcerative colitis. BMJ 1955. [Google Scholar] [CrossRef]
- Fumery, M.; Singh, S.; Dulai, P.S.; Gower-Rousseau, C.; Peyrin-Biroulet, L.; Sandborn, W.J. Natural history of adult ulcerative colitis in population-based cohorts: A systematic review. Clin. Gastroenterol. Hepatol. 2018, 16, 343–356.e3. [Google Scholar] [CrossRef]
- Harbord, M.; Eliakim, R.; Bettenworth, D.; Karmiris, K.; Katsanos, K.; Kopylov, U.; Kucharzik, T.; Molnár, T.; Raine, T.; Sebastian, S.; et al. Third European evidence-based consensus on diagnosis and management of ulcerative colitis. Part 2: Current management. J. Crohn’s Colitis 2017, 11, 769–784. [Google Scholar] [CrossRef] [PubMed]
- De Cassan, C.; Fiorino, G.; Danese, S. Second-generation corticosteroids for the treatment of Crohn’s disease and ulcerative colitis: More effective and less side effects? Dig. Dis. 2012, 30, 368–375. [Google Scholar] [CrossRef] [PubMed]
- Danese, S.; Roda, G.; Peyrin-Biroulet, L. Evolving therapeutic goals in ulcerative colitis: Towards disease clearance. Nat. Rev. Gastroenterol. Hepatol. 2020, 17, 1–2. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, N.H.; Fumery, M.; Dulai, P.S.; Prokop, L.J.; Sandborn, W.J.; Murad, M.H.; Singh, S. Comparative efficacy and tolerability of pharmacological agents for management of mild to moderate ulcerative colitis: A systematic review and network meta-analyses. Lancet Gastroenterol. Hepatol. 2018, 3, 742–753. [Google Scholar] [CrossRef]
- Salice, M.; Rizzello, F.; Calabrese, C.; Calandrini, L.; Gionchetti, P. A current overview of corticosteroid use in active ulcerative colitis. Exp. Rev. Gastroenterol. Hepatol. 2019, 13, 557–561. [Google Scholar] [CrossRef] [PubMed]
- Lichtenstein, G.R.; Feagan, B.G.; Cohen, R.D.; Salzberg, B.A.; Diamond, R.H.; Price, S.; Langholff, W.; Londhe, A.; Sandborn, W.J. Serious infection and mortality in patients with Crohn’s disease: More than 5 years of follow-up in the TREAT registry. Am. J. Gastroenterol. 2012, 107, 1409–1422. [Google Scholar] [CrossRef] [PubMed]
- Hu, M.Y.; Peppercorn, M.A. MMX mesalamine: A novel high-dose, once-daily 5-aminosalicylate formulation for the treatment of ulcerative colitis. Exp. Opin. Pharmacother. 2008, 9, 1049–1058. [Google Scholar] [CrossRef]
- Le Berre, C.; Ananthakrishnan, A.N.; Danese, S.; Singh, S.; Peyrin-Biroulet, L. Ulcerative colitis and Crohn’s disease have similar burden and goals for treatment. Clin. Gastroenterol. Hepatol. 2020, 18, 14–22. [Google Scholar] [CrossRef]
- Feuerstein, J.D.; Moss, A.C.; Farraye, F.A. Ulcerative colitis. Mayo Clin. Proc. 2019, 94, 1357–1373. [Google Scholar] [CrossRef]
- Rosenberg, L.; Nanda, K.S.; Zenlea, T.; Gifford, A.; Lawlor, G.O.; Falchuk, K.R.; Wolf, J.L.; Cheifetz, A.S.; Goldsmith, J.D.; Moss, A.C. Histologic markers of inflammation in patients with ulcerative colitis in clinical remission: Correlates of histological inflammation. Clin. Gastroenterol. Hepatol. 2013, 11, 991–996. [Google Scholar] [CrossRef]
- Giugliano, F.P.; Strisciuglio, C.; Martinelli, M.; Andreozzi, M.; Cenni, S.; Campione, S.; D’Armiento, M.; Staiano, A.; Miele, E. Does Azathioprine induce endoscopic and histologic healing in pediatric inflammatory bowel disease? A prospective, observational study. Dig. Liver Dis. 2018, 50, 240–246. [Google Scholar] [CrossRef] [PubMed]
- Paoluzi, P.; D’Albasio, G.; Pera, A.; Bianchi Porro, G.; Paoluzi, O.A.; Pica, R.; Cottone, M.; Miglioli, M.; Prantera, C.; Sturniolo, G.; et al. Oral and topical 5-aminosalicylic acid (mesalazine) in inducing and maintaining remission in mild-moderate relapse of ulcerative colitis: One-year randomised multicentre trial. Dig. Liver Dis. 2002, 34, 787–793. [Google Scholar] [CrossRef]
- Colombel, J.F.; Ordás, I.; Ullman, T.; Rutgeerts, P.; Chai, A.; O’Byrne, S.; Lu, T.T.; Panés, J. Agreement between rectosigmoidoscopy and colonoscopy analyses of disease activity and healing in patients with ulcerative colitis. Gastroenterology 2016, 150, 389–395.e3. [Google Scholar] [CrossRef] [PubMed]
- D’Amico, F.; Bonovas, S.; Danese, S.; Peyrin-Biroulet, L. Review article: Faecal calprotectin and histologic remission in ulcerative colitis. Aliment. Pharmacol. Ther. 2020, 51, 689–698. [Google Scholar] [CrossRef] [PubMed]
- Lasson, A.; Ohman, L.; Stotzer, P.O.; Isaksson, S.; Uberbacher, O.; Ung, K.A.; Strid, H. Pharmacological intervention based on fecal calprotectin levels in patients with ulcerative colitis at high risk of a relapse: A prospective, randomized, controlled study. United Eur. Gastroenterol. J. 2015, 3, 72–79. [Google Scholar] [CrossRef]
- Osterman, M.T.; Aberra, F.N.; Cross, R.; Liakos, S.; McCabe, R.; Shafran, I.; Wolf, D.; Hardi, R.; Nessel, L.; Brensinger, C.; et al. Mesalamine dose escalation reduces fecal calprotectin in patients with quiescent ulcerative colitis. Clin. Gastroenterol. Hepatol. 2014, 12, 1887–1893.e3. [Google Scholar] [CrossRef]
- D’Amico, F.; Nancey, S.; Danese, S.; Peyrin-Biroulet, L. A Practical guide for faecal calprotectin measurement: Myths and realities. J. Crohn’s Colitis 2020, jjaa093, Published online ahead of print. [Google Scholar]
- Allocca, M.; Fiorino, G.; Bonovas, S.; Furfaro, F.; Gilardi, D.; Argollo, M.; Magnoni, P.; Peyrin-Biroulet, L.; Danese, S. Accuracy of humanitas ultrasound criteria in assessing disease activity and severity in ulcerative colitis: A prospective study. J. Crohn’s Colitis 2018, 12, 1385–1391. [Google Scholar] [CrossRef]
- Bezzio, C.; Fascì-Spurio, F.; Viganò, C.; Meucci, G.; Papi, C.; Saibeni, S. The problem of adherence to therapy in ulcerative colitis and the potential utility of multi-matrix system (MMX) technology. Exp. Rev. Gastroenterol. Hepatol. 2017, 11, 33–41. [Google Scholar] [CrossRef]
- Kane, S.V.; Cohen, R.D.; Aikens, J.E.; Hanauer, S.B. Prevalence of nonadherence with maintenance mesalamine in quiescent ulcerative colitis. Am. J. Gastroenterol. 2001, 96, 2929–2933. [Google Scholar] [CrossRef]
- Kane, S.; Huo, D.; Aikens, J.; Hanauer, S. Medication nonadherence and the outcomes of patients with quiescent ulcerative colitis. Am. J. Med. 2003, 114, 39–43. [Google Scholar] [CrossRef]
- Moody, G.A.; Jayanthi, V.; Probert, C.S.J.; Mac Kay, H.; Mayberry, J.F. Long-term therapy with sulphasalazine protects against colorectal cancer in ulcerative colitis: A retrospective study of colorectal cancer risk and compliance with treatment in Leicestershire. Eur. J. Gastroenterol. Hepatol. 1996, 8, 1179–1183. [Google Scholar] [CrossRef] [PubMed]
- Mitra, D.; Hodgkins, P.; Yen, L.; Davis, K.L.; Cohen, R.D. Association between oral 5-ASA adherence and health care utilization and costs among patients with active ulcerative colitis. BMC Gastroenterol. 2012, 12, 132. [Google Scholar] [CrossRef] [PubMed]
- Goodhand, J.R.; Kamperidis, N.; Sirwan, B.; Macken, L.; Tshuma, N.; Koodun, Y.; Chowdhury, F.A.; Croft, N.M.; Direkze, N.; Langmead, L.; et al. Factors associated with thiopurine non-adherence in patients with inflammatory bowel disease. Aliment. Pharmacol. Ther. 2013, 38, 1097–1108. [Google Scholar] [CrossRef] [PubMed]
- Lopez, A.; Billioud, V.; Peyrin-Biroulet, C.; Peyrin-Biroulet, L. Adherence to anti-TNF therapy in inflammatory bowel diseases: A systematic review. Inflamm. Bowel Dis. 2013, 19, 1528–1533. [Google Scholar] [CrossRef]
- Shale, M.J.; Riley, S.A. Studies of compliance with delayed-release mesalazine therapy in patients with inflammatory bowel disease. Aliment. Pharmacol. Ther. 2003, 15, 191–198. [Google Scholar] [CrossRef]
- Kane, S.V. Systematic review: Adherence issues in the treatment of ulcerative colitis. Aliment. Pharmacol. Ther. 2006, 23, 577–585. [Google Scholar] [CrossRef]
- D’Incà, R.; Bertomoro, P.; Mazzocco, K.; Vettorato, M.G.; Rumiati, R.; Sturniolo, G.C. Risk factors for non-adherence to medication in inflammatory bowel disease patients. Aliment. Pharmacol. Ther. 2008, 27, 166–172. [Google Scholar] [CrossRef]
- Singh, S.; Feuerstein, J.D.; Binion, D.G.; Tremaine, W.J. AGA Technical review on the management of mild-to-moderate ulcerative colitis. Gastroenterology 2019, 156, 769–808.e29. [Google Scholar] [CrossRef]
- Gisbert, J.P.; Gomollón, F.; Hinojosa, J.; Román, A.L.S. Adherence of gastroenterologists to European Crohn’s and Colitis Organisation consensus on ulcerative colitis: A real-life survey in Spain. J. Crohn’s Colitis 2010, 4, 567–574. [Google Scholar] [CrossRef]
- Dignass, A.U.; Bokemeyer, B.; Adamek, H.; Mross, M.; Vinter-Jensen, L.; Börner, N.; Silvennoinen, J.; Tan, G.; Pool, M.O.; Stijnen, T.; et al. Mesalamine once daily is more effective than twice daily in patients with quiescent ulcerative colitis. Clin. Gastroenterol. Hepatol. 2009, 7, 762–769. [Google Scholar] [CrossRef]
- Feagan, B.G.; MacDonald, J.K. Once daily oral mesalamine compared to conventional dosing for induction and maintenance of remission in ulcerative colitis: A systematic review and meta-analysis. Inflamm. Bowel Dis. 2012, 18, 1785–1794. [Google Scholar] [CrossRef] [PubMed]
- Flourié, B.; Hagège, H.; Tucat, G.; Maetz, D.; Hébuterne, X.; Kuyvenhoven, J.P.; Tan, T.G.; Pierik, M.J.; Masclee, A.A.M.; Dewit, O.; et al. Randomised clinical trial: Once-vs. twice-daily prolonged-release mesalazine for active ulcerative colitis. Aliment. Pharmacol. Ther. 2013, 37, 767–775. [Google Scholar] [CrossRef] [PubMed]
- D’Haens, G.R.; Sandborn, W.J.; Zou, G.; Stitt, L.W.; Rutgeerts, P.J.; Gilgen, D.; Jairath, V.; Hindryckx, P.; Shackelton, L.M.; Vandervoort, M.K.; et al. Randomised non-inferiority trial: 1600 mg versus 400 mg tablets of mesalazine for the treatment of mild-to-moderate ulcerative colitis. Aliment. Pharmacol. Ther. 2017, 46, 292–302. [Google Scholar] [CrossRef] [PubMed]
- Sandborn, W.J.; Korzenik, J.; Lashner, B.; Leighton, J.A.; Mahadevan, U.; Marion, J.F.; Safdi, M.; Sninsky, C.A.; Patel, R.M.; Friedenberg, K.A.; et al. Once-daily dosing of delayed-release oral mesalamine (400-mg tablet) is as effective as twice-daily dosing for maintenance of remission of ulcerative colitis. Gastroenterology 2010, 138, 1286–1296.e3. [Google Scholar] [CrossRef]
- Kamm, M.A.; Sandborn, W.J.; Gassull, M.; Schreiber, S.; Jackowski, L.; Butler, T.; Lyne, A.; Stephenson, D.; Palmen, M.; Joseph, R.E. Once-daily, high-concentration MMX mesalamine in active ulcerative colitis. Gastroenterology 2007, 132, 66–75. [Google Scholar] [CrossRef]
- Lichtenstein, G.R.; Kamm, M.A.; Boddu, P.; Gubergrits, N.; Lyne, A.; Butler, T.; Lees, K.; Joseph, R.E.; Sandborn, W.J. Effect of once- or twice-daily MMX mesalamine (Spd476) For the induction of remission of mild to moderately active ulcerative colitis. Clin. Gastroenterol. Hepatol. 2007, 5, 95–102. [Google Scholar] [CrossRef]
- Kane, S.; Katz, S.; Jamal, M.M.; Safdi, M.; Dolin, B.; Solomon, D.; Palmen, M.; Barrett, K. Strategies in maintenance for patients receiving long-term therapy (SIMPLE): A study of MMX mesalamine for the long-term maintenance of quiescent ulcerative colitis. Inflamm. Bowel Dis. 2012, 18, 1026–1033. [Google Scholar] [CrossRef]
- D’Albasio, G.; Pacini, F.; Camarri, E.; Messori, A.; Trallori, G.; Bonanomi, A.G.; Bardazzi, G.; Milla, M.; Ferrero, S.; Biagini, M.; et al. Combined therapy with 5-aminosalicylic acid tablets and enemas for maintaining remission in ulcerative colitis: A randomized double-blind study. Am. J. Gastroenterol. 1997, 92, 1143–1147. [Google Scholar]
- Yokoyama, H.; Takagi, S.; Kuriyama, S.; Takahashi, S.; Takahashi, H.; Iwabuchi, M.; Takahashi, S.; Kinouchi, Y.; Hiwatashi, N.; Tsuji, I.; et al. Effect of weekend 5-aminosalicylic acid (mesalazine) enema as maintenance therapy for ulcerative colitis: Results from a randomized controlled study. Inflamm. Bowel Dis. 2007, 13, 1115–1120. [Google Scholar] [CrossRef]
- Hanauer, S.B.; Sandborn, W.J.; Dallaire, C.; Archambault, A.; Yacyshyn, B.; Yeh, C.; Smith-Hall, N. Delayed-release oral mesalamine 4.8 g/day (800 mg tablets) compared with 2.4 g/day (400 mg tablets) for the treatment of mildly to moderately active ulcerative colitis: The ASCENDI trial. Can. J. Gastroenterol. 2007, 21, 827–834. [Google Scholar] [CrossRef]
- Hanauer, S.B.; Sandborn, W.J.; Kornbluth, A.; Katz, S.; Safdi, M.; Woogen, S.; Regalli, G.; Yeh, C.; Smith-Hall, N.; Ajayi, F. Delayed-release oral mesalamine at 4.8 g/day (800 mg tablet) for the treatment of moderately active ulcerative colitis: The ASCEND II trial. Am. J. Gastroenterol. 2005, 100, 2478–2485. [Google Scholar] [CrossRef] [PubMed]
- Hiwatashi, N.; Suzuki, Y.; Mitsuyama, K.; Munakata, A.; Hibi, T. Clinical trial: Effects of an oral preparation of mesalazine at 4 g/day on moderately active ulcerative colitis. A phase III parallel-dosing study. J. Gastroenterol. 2011, 46, 46–56. [Google Scholar] [CrossRef]
- Pica, R.; Cassieri, C.; Cocco, A.; Zippi, M.; Marcheggiano, A.; De Nitto, D.; Avallone, E.V.; Crispino, P.; Occhigrossi, G.; Paoluzi, P. A randomized trial comparing 4.8 vs. 2.4 g/day of oral mesalazine for maintenance of remission in ulcerative colitis. Dig. Liver Dis. 2015, 47, 933–937. [Google Scholar] [CrossRef] [PubMed]
- Fiorino, G.; Fries, W.; De La Rue, S.A.; Malesci, A.C.; Repici, A.; Danese, S. New drug delivery systems in inflammatory bowel disease: MMX and tailored delivery to the gut. Curr. Med. Chem. 2010, 17, 1851–1857. [Google Scholar] [CrossRef] [PubMed]
- Willian, M.K.; D’haens, G.; Yarlas, A.; Joshi, A.V. Changes in health-related quality of life and work-related outcomes for patients with mild-to-moderate ulcerative colitis receiving short-term and long-term treatment with multimatrix mesalamine: A prospective, open-label study. J. Patient Rep. Outcomes 2018, 2, 22. [Google Scholar] [CrossRef]
- Hinojosa, J.; Gisbert, J.P.; Gomollón, F.; López San Román, A. Adherence of gastroenterologists to European Crohn’s and Colitis Organisation Consensus on Crohn’s disease: A real-life survey in Spain. J. Crohn’s Colitis 2012, 6, 763–770. [Google Scholar] [CrossRef][Green Version]
- Buckland, A.; Bodger, K. The cost-utility of high dose oral mesalazine for moderately active ulcerative colitis. Aliment. Pharmacol. Ther. 2008, 28, 1287–1296. [Google Scholar] [CrossRef]
- Feagan, B.G.; Macdonald, J.K. Oral 5-aminosalicylic acid for maintenance of remission in ulcerative colitis. Cochrane Database Syst. Rev. 2012, 10, CD000544. [Google Scholar]
- Ko, C.W.; Singh, S.; Feuerstein, J.D.; Falck-Ytter, C.; Falck-Ytter, Y.; Cross, R.K.; Crockett, S.; Feuerstein, J.; Flamm, S.; Inadomi, J.; et al. AGA clinical practice guidelines on the management of mild-to-moderate ulcerative colitis. Gastroenterology 2019, 156, 748–764. [Google Scholar] [CrossRef]
- Angelucci, E.; Malesci, A.; Danese, S. Budesonide: Teaching an old dog new tricks for inflammatory Bowel disease treatment. Curr. Med. Chem. 2008, 15, 2527–2535. [Google Scholar] [CrossRef]
- Hoy, S.M. Budesonide MMX®: A review of its use in patients with mild to moderate ulcerative colitis. Drugs 2015, 75, 879–886. [Google Scholar] [CrossRef] [PubMed]
- Sandborn, W.J.; Travis, S.; Moro, L.; Jones, R.; Gautille, T.; Bagin, R.; Huang, M.; Yeung, P.; Ballard, E.D. Once-daily budesonide MMX® extended-release tablets induce remission in patients with mild to moderate ulcerative colitis: Results from the CORE i study. Gastroenterology 2012, 143, 1218–1226.e2. [Google Scholar] [CrossRef] [PubMed]
- Travis, S.P.L.; Danese, S.; Kupcinskas, L.; Alexeeva, O.; D’Haens, G.; Gibson, P.R.; Moro, L.; Jones, R.; Ballard, E.D.; Masure, J.; et al. Once-daily budesonide MMX in active, mild-to-moderate ulcerative colitis: Results from the randomised CORE II study. Gut 2014, 63, 433–441. [Google Scholar] [CrossRef]
- Sandborn, W.J.; Danese, S.; D’Haens, G.; Moro, L.; Jones, R.; Bagin, R.; Huang, M.; David Ballard, E.; Masure, J.; Travis, S. Induction of clinical and colonoscopic remission of mild-to-moderate ulcerative colitis with budesonide MMX 9 mg: Pooled analysis of two phase 3 studies. Aliment. Pharmacol. Ther. 2015, 41, 409–418. [Google Scholar] [CrossRef] [PubMed]
- Lichtenstein, G.R.; Travis, S.; Danese, S.; D’Haens, G.; Moro, L.; Jones, R.; Huang, M.; Ballard, E.D.; Bagin, R.; Hardiman, Y.; et al. Budesonide MMX for the induction of remission of mild to moderate ulcerative colitis: A pooled safety analysis. J. Crohn’s. Colitis 2015, 9, 738–746. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Rubin, D.T.; Cohen, R.D.; Sandborn, W.J.; Lichtenstein, G.R.; Axler, J.; Riddell, R.H.; Zhu, C.; Barrett, A.C.; Bortey, E.; Forbes, W.P. Budesonide multimatrix is efficacious for mesalamine-refractory, mild to moderate ulcerative colitis: A randomised, placebo-controlled trial. J. Crohn’s Colitis 2017, 11, 785–791. [Google Scholar] [CrossRef]
- Bonovas, S.; Nikolopoulos, G.K.; Piovani, D.; González-Lorenzo, M.; Pantavou, K.; Lytras, T.; Peyrin-Biroulet, L.; Danese, S. Comparative assessment of budesonide-MMX and mesalamine in active, mild-to-moderate ulcerative colitis: A systematic review and network meta-analysis. Br. J. Clin. Pharmacol. 2019, 85, 2244–2254. [Google Scholar] [CrossRef] [PubMed]
- Maconi, G.; Mezzina, N.; Landi, S.; Grillo, S.; Bezzio, C.; Bosani, M.; Pastorelli, L.; Dell’Era, A.; Chibbar, R.; Carmagnola, S.; et al. Use, effectiveness and tolerability of budesonide-MMX in ulcerative colitis: A real-life experience. United Eur. Gastroenterol. J. 2019, 7, 1164–1170. [Google Scholar] [CrossRef]
- Danese, S.; Hart, A.; Dignass, A.; Fiorino, G.; Louis, E.; Bonovas, S.; D’Haens, G.; Dotan, I.; Rogler, G.; Paridaens, K.; et al. A multicentre prospective cohort study assessing the effectiveness of budesonide MMX® (Cortiment®MMX®) for active, mild-to-moderate ulcerative colitis. United Eur. Gastroenterol. J. 2019, 7, 1171–1182. [Google Scholar] [CrossRef]
- Magro, F.; Estevinho, M.M. Moving from efficacy to effectiveness: Budesonide multimatrix in ulcerative colitis. United Eur. Gastroenterol. J. 2019, 7, 1153–1155. [Google Scholar] [CrossRef]
- Peyrin-Biroulet, L.; Sandborn, W.; Sands, B.E.; Reinisch, W.; Bemelman, W.; Bryant, R.V.; D’Haens, G.; Dotan, I.; Dubinsky, M.; Feagan, B.; et al. Selecting therapeutic targets in inflammatory bowel disease (Stride): Determining therapeutic goals for treat-to-target. Am. J. Gastroenterol. 2015, 110, 1324–1338. [Google Scholar] [CrossRef]
- Colombel, J.F.; D’Haens, G.; Lee, W.J.; Petersson, J.; Panaccione, R. Outcomes and strategies to support a treat-to-target approach in inflammatory bowel disease: A systematic review. J. Crohn’s Colitis 2020, 14, 254–266. [Google Scholar] [CrossRef]
- Im, J.P.; Ye, B.D.; Kim, Y.S.; Kim, J.S. Changing treatment paradigms for the management of inflammatory bowel disease. Korean J. Intern. Med. 2018, 33, 28–35. [Google Scholar] [CrossRef] [PubMed]
- Solitano, V.; D’Amico, F.; Zacharopoulou, E.; Peyrin-Biroulet, L.; Danese, S. Early Intervention in Ulcerative Colitis: Ready for Prime Time? J. Clin. Med. 2020, 9, 2646. [Google Scholar] [CrossRef] [PubMed]
- Ochsenkühn, T.; D’Haens, G. Current misunderstandings in the management of ulcerative colitis. Gut 2011, 60, 1294–1299. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Siegel, C.A.; Bernstein, C.N. Identifying patients with inflammatory bowel diseases at high vs. low risk of complications. Clin. Gastroenterol. Hepatol. 2020, 18, 1261–1267. [Google Scholar] [CrossRef] [PubMed]
- Le Berre, C.; Roda, G.; Nedeljkovic Protic, M.; Danese, S.; Peyrin-Biroulet, L. Modern use of 5-aminosalicylic acid compounds for ulcerative colitis. Exp. Opin. Biol. Ther. 2020, 20, 363–378. [Google Scholar] [CrossRef]
- Dias, C.C.; Rodrigues, P.P.; da Costa-Pereira, A.; Magro, F. Clinical predictors of colectomy in patients with ulcerative colitis: Systematic review and meta-analysis of cohort studies. J. Crohn’s Colitis 2015, 9, 156–163. [Google Scholar] [CrossRef]
- Dassopoulos, T.; Cohen, R.D.; Scherl, E.J.; Schwartz, R.M.; Kosinski, L.; Regueiro, M.D. Ulcerative colitis care pathway. Gastroenterology 2015, 149, 238–245. [Google Scholar] [CrossRef]
- Jackson, B.; De Cruz, P. Algorithms to facilitate shared decision-making for the management of mild-to-moderate ulcerative colitis. Exp. Rev. Gastroenterol. Hepatol. 2018, 12, 1079–1100. [Google Scholar] [CrossRef]
- Hawthorne, A.B.; Rubin, G.; Ghosh, S. Review article: Medication non-adherence in ulcerative colitis—Strategies to improve adherence with mesalazine and other maintenance therapies. Aliment. Pharmacol. Ther. 2008, 27, 1157–1166. [Google Scholar] [CrossRef] [PubMed]
- Pedersen, N.; Thielsen, P.; Martinsen, L.; Bennedsen, M.; Haaber, A.; Langholz, E.; Végh, Z.; Duricova, D.; Jess, T.; Bell, S.; et al. EHealth: Individualization of mesalazine treatment through a self-managed web-based solution in mild-to-moderate ulcerative colitis. Inflamm. Bowel Dis. 2014, 20, 2276–2285. [Google Scholar] [CrossRef] [PubMed]
- Moshkovska, T.; Stone, M.A.; Smith, R.M.; Bankart, J.; Baker, R.; Mayberry, J.F. Impact of a tailored patient preference intervention in adherence to 5-aminosalicylic acid medication in ulcerative colitis: Results from an exploratory randomized controlled trial. Inflamm. Bowel Dis. 2011, 17, 1874–1881. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, A.P.; Nelson, E.; Reeves, D.; Richardson, G.; Roberts, C.; Robinson, A.; Rogers, A.E.; Sculpher, M.; Thompson, D.G. A randomised controlled trial to assess the effectiveness and cost of a patient orientated self management approach to chronic inflammatory bowel disease. Gut 2004, 53, 1639–1645. [Google Scholar] [CrossRef] [PubMed]
- Danese, S.; Banerjee, R.; Cummings, J.R.F.; Dotan, I.; Kotze, P.G.; Leong, R.W.L.; Paridaens, K.; Peyrin-Biroulet, L.; Scott, G.; Van Assche, G.; et al. Consensus recommendations for patient-centered therapy in mild-to-moderate ulcerative colitis: The i Support Therapy-Access to Rapid Treatment (iSTART) approach. Intest. Res. 2018, 16, 522–528. [Google Scholar] [CrossRef] [PubMed]
- Bonovas, S.; Nikolopoulos, G.K.; Lytras, T.; Fiorino, G.; Peyrin-Biroulet, L.; Danese, S. Comparative safety of systemic and low-bioavailability steroids in inflammatory bowel disease: Systematic review and network meta-analysis. Br. J. Clin. Pharmacol. 2018, 84, 239–251. [Google Scholar] [CrossRef]
- Berg, D.R.; Colombel, J.F.; Ungaro, R. The role of early biologic therapy in inflammatory bowel disease. Inflamm. Bowel Dis. 2019, 25, 1896–1905. [Google Scholar] [CrossRef]
- Danese, S.; Fiorino, G.; Peyrin-Biroulet, L. Positioning therapies in ulcerative colitis. Clin. Gastroenterol. Hepatol. 2020, 18, 1280–1290.e1. [Google Scholar] [CrossRef]
- De Boer, N.K.H.; Peyrin-Biroulet, L.; Jharap, B.; Sanderson, J.D.; Meijer, B.; Atreya, I.; Barclay, M.L.; Colombel, J.F.; Lopez, A.; Beaugerie, L.; et al. Thiopurines in inflammatory bowel disease: New findings and perspectives. J. Crohn’s Colitis 2018, 12, 610–620. [Google Scholar] [CrossRef]
| Author (Year) | Study Design | Number of Patients | Study Arms | Primary Outcome | Results | Conclusions |
|---|---|---|---|---|---|---|
| Dignass et al. (2009) [41] | Randomized non-inferiority trial | 362 | 5-ASA (2 g) OD 5-ASA (1 g) BD | 1-yr. remission rates (UCDAI score <2) | 70.9% 58.9% (p = 0.024) | Prolonged-release oral 5-ASA 2 g once daily is associated with better remission rates |
| Flourie et al. (2013) [43] | Randomized non-inferiority trial | 206 | 5-ASA (4 g/day) OD + enema 1 g/day 5-ASA (4 g/day) BD + enema 1 g/day | Clinical and endoscopic remission at w 8 (UCDAI score <1) | 52.1% 41.8% (p = 0.14) | Combined with 5-ASA enema, prolonged-release 5-ASA OD 4 g is as effective as 2 g twice daily for inducing remission |
| D’Haens et al. (2017) [44] | Randomized non-inferiority trial | 817 | 5-ASA (3.2 g) OD 5-ASA (3.2 g) BD | Clinical and endoscopic remission at w 8 (MCS ≤ 2 with no individual score >1) | 22.4% 24.6% (p = 0.005) | 3.2 mg 5-ASA OD is non-inferior to a BD regimen |
| Sandborn et al. (2010) [45] | Randomized non-inferiority trial | 1023 | 5-ASA (1.6–2.4 g/day) OD 5-ASA (1.6–2.4 g/day) BD | Clinical remission (SCCAI score ≤2 points) at mo. 6 | 90.5% 91.8% (p = 0.05) | OD dosing of delayed-release 5-ASA is as effective as BD dosing for maintenance of clinical remission |
| Kamm et al. (2007) [46] | RCT | 343 | MMX 5-ASA 2.4 g/day OD MMX 5-ASA 4.8 g/day OD Delayed-release oral 5-ASA 2.4 g/day (3 divided doses) Placebo | Proportion of patients in clinical and endoscopic remission (modified UCDAI <1 with rectal bleeding and stool frequency scores of 0, no mucosal friability, and a >1-point reduction in sigmoidoscopy score from baseline) at w 8 | 40.5% (p = 0.01) 41.2% (p = 0.007) 32.6% (p = 0.124) 22.1% | OD MMX 5-ASA 2.4 or 4.8 g/day are both superior to placebo in the induction of clinical and endoscopic remission |
| Lichtenstein et al. (2007) [47] | RCT | 280 | MMX 5-ASA 2.4 g/day BD MMX 5-ASA 4.8 g/day OD Placebo | Clinical and endoscopic remission (modified UCDAI score <1, with a score of 0 for rectal bleeding and stool frequency, and at least a 1-point reduction in sigmoidoscopy score) at w 8 | 34.1% (p < 0.01) 29.2% 12.9% | BD and OD MMX 5-ASA are efficacious for the induction of clinical and endoscopic remission |
| Kane et al. (2012) [48] | Phase IV multicentre open label | 290 | MMX 5-ASA 2.4 g/day OD | Clinical recurrence (defined as ≥4 bowel movements per day above the patient’s normal frequency and which were associated with any of the following symptoms: urgency, abdominal pain, or rectal bleeding) at mo. 6 | 23.5% | MMX 5-ASA 2.4 g/day OD is effective for maintaining quiescence |
| D’Albasio et al. (1997) [49] | RCT | 69 | 5-ASA tablets (1.6 g/day) and 5-ASA enemas (4 g/100 mL) twice weekly 5-ASA (1.6 g/day) and placebo enemas/twice weekly | Maintenance of remission (mild symptoms and normal endoscopic appearance of mucosa) at mo. 12 | 39% 69% (p = 0.036) | 5-ASA given daily by oral route and intermittently by topical route can be more effective than oral therapy alone. |
| Yokoyama et al. (2007) [50] | RCT | 24 | Weekend 5-ASA enema group (1 g 5-ASA enemas in the weekend plus oral 5-ASA 3 g/day for 7 days) Daily oral 5-ASA use only group (only oral 5-ASA 3 g/day for 7 days) | Incidence of relapse (as a score of ≥6 in clinical activity index and ≥3 in the endoscopic index) | 18.2% 76.9% (multivariate HR: 0.19, 95% CI, 0.04–0.94) | Adding weekend 1 g 5-ASA enema to daily 3 g oral 5-ASA as maintenance therapy |
| Hanauer et al. (2007) [51] | RCT | 301 | 5-ASA 2.4 g/day 5-ASA 4.8 g/day | Overall improvement (defined as complete remission or response to therapy) from baseline to w 6 | 57% 72% (p = 0.0384) | 4.8 g/day dose may enhance treatment success rates in patients with moderate disease compared with mesalamine 2.4 g/day |
| Hanauer et al. (2005) [52] | RCT | 386 | 5-ASA 2.4 g/day 5-ASA 4.8 g/day | Overall improvement (defined as either complete remission or a clinical response to therapy) from baseline to w 6 | 59% 72% (p = 0.036) | 4.8 g/day dose results in significantly higher rates of overall improvement in patients with moderate disease compared with 2.4 g/day |
| Hiwatashi et al. (2011) [53] | RCT | 123 | 5-ASA 4 g/day (2 divided doses) 5-ASA 2.25 g/day (3 divided doses) | UCDAI score before and after 8 weeks of treatment | 3.0 (95% CI −3.8 to −2.3) 0.8 (95% CI −1.8 to 0.1) | 4 g/day results in a significantly superior change in UCDAI score compared with 2.25 g/day |
| Pica et al. (2015) [54] | RCT | 112 | 5-ASA 4.8 g 5-ASA 2.4 g | Maintenance of remission (defined as the absence of symptoms and the endoscopically documented absence of the inflammatory changes typical of active UC) at mo. 12 | 75% 64.2% (p = 0.3) | A daily dose of 4.8 g oral mesalamine results in increased rates and duration of remission compared to 2.4 g, in patients younger than 40 years and/or with extensive disease |
| Author (Year) | Study Design | Number of Patients | Study Arms | Primary Outcome | Results | Conclusions |
|---|---|---|---|---|---|---|
| Sandborn et al. (2012) [63] | RCT | 509 | Budesonide MMX 9 mg Budesonide 6 mg Mesalamine 2.4 g Placebo | Combined clinical and endoscopic remission (UCDAI score ≤1 point, with sub-scores of 0 for both rectal bleeding and stool frequency, no mucosal friability on colonoscopy, and a ≥1-point reduction from baseline in the endoscopic index score) at w 8 | 17.9% (p = 0.0143) 13.2% (p = 0.1393) 12.1% (p = 0.2200) 7.4% | Budesonide MMX 9 mg is safe and more effective than placebo in inducing remission |
| Travis et al. (2014) [64] | RCT | 410 | Budesonide MMX 9 mg Budesonide MMX 6 mg Budesonide 9 mg Placebo | Combined clinical and endoscopic remission (UCDAI score ≤1, with a rectal bleeding score of 0, stool frequency score of 0, mucosal appearance score of 0 and a ≥1-point reduction in baseline endoscopic index score) at w 8 | 17.4% (p = 0.0047) 8.3% (p > 0.05) 12.6% (p = 0.0481) 4.5% | Budesonide MMX 9 mg is safe and more effective than placebo in inducing combined clinical and endoscopic remission |
| Rubin et al. (2017) [67] | RCT | 510 | Budesonide MMX 9 mg Placebo | Combined clinical and endoscopic remission (UCDAI score of ≤1, with subscale scores of 0 for rectal bleeding, stool frequency, and mucosal appearance) at w 8 | 13.0% (p = 0.049) 7.5% | Budesonide MMX is safe and efficacious for inducing clinical and endoscopic remission for mild-to-moderate UC refractory to oral mesalamine therapy |
| Maconi et al. (2019) [69] | Retrospective cohort study | 82 | Budesonide MMX | Clinical remission (pMayo of 0–1 with a rectal bleeding sub-score = 0) at mo. 2 | 50% | Budesonide MMX is safe and effective in patients with mild disease activity |
| Danese et al. (2019) [70] | Prospective cohort study | 326 | Cohort 1: budesonide MMX + 5-ASA at least 14 days after increased/optimized 5-ASA dose Cohort 2: budesonide MMX + 5-ASA within 14 days since 5-ASA increased/optimized or without 5-ASA dose modification Cohort 3: budesonide MMX as monotherapy | Clinical benefit (≥3 point reduction UCDAI clinical sub-score) at the end of induction treatment | 64.3% (p = 0.0096) 62.1% 33.3% | Budesonide is safe and well tolerated in about 60% of mild-to-moderate UC patients, in a real-life setting |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Solitano, V.; D’Amico, F.; Fiorino, G.; Paridaens, K.; Peyrin-Biroulet, L.; Danese, S. Key Strategies to Optimize Outcomes in Mild-to-Moderate Ulcerative Colitis. J. Clin. Med. 2020, 9, 2905. https://doi.org/10.3390/jcm9092905
Solitano V, D’Amico F, Fiorino G, Paridaens K, Peyrin-Biroulet L, Danese S. Key Strategies to Optimize Outcomes in Mild-to-Moderate Ulcerative Colitis. Journal of Clinical Medicine. 2020; 9(9):2905. https://doi.org/10.3390/jcm9092905
Chicago/Turabian StyleSolitano, Virginia, Ferdinando D’Amico, Gionata Fiorino, Kristine Paridaens, Laurent Peyrin-Biroulet, and Silvio Danese. 2020. "Key Strategies to Optimize Outcomes in Mild-to-Moderate Ulcerative Colitis" Journal of Clinical Medicine 9, no. 9: 2905. https://doi.org/10.3390/jcm9092905
APA StyleSolitano, V., D’Amico, F., Fiorino, G., Paridaens, K., Peyrin-Biroulet, L., & Danese, S. (2020). Key Strategies to Optimize Outcomes in Mild-to-Moderate Ulcerative Colitis. Journal of Clinical Medicine, 9(9), 2905. https://doi.org/10.3390/jcm9092905







