Upper Respiratory Tract Microbiome and Otitis Media Intertalk: Lessons from the Literature
Abstract
:1. Introduction
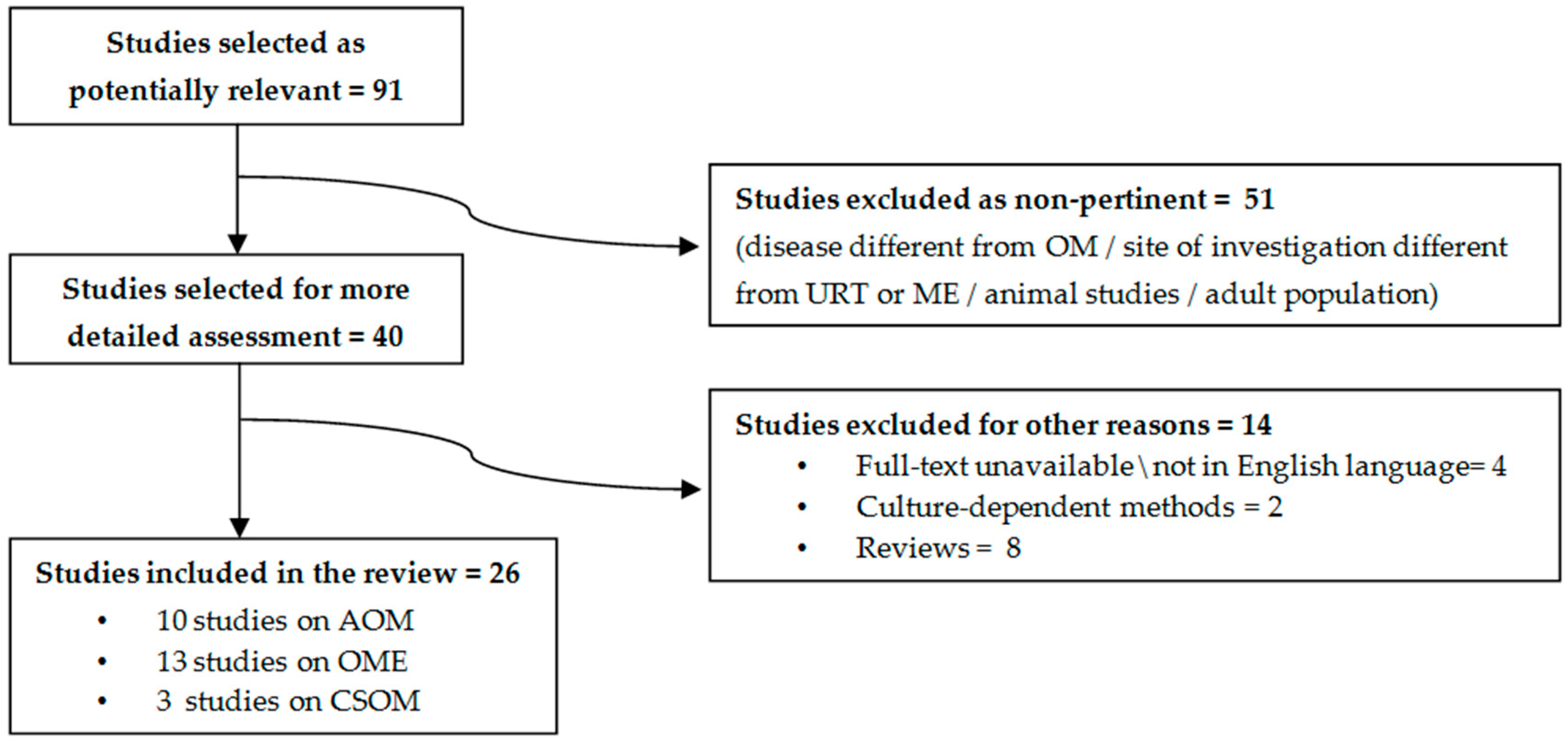
2. Methods
3. Environmental Factors and Microbiota Development in the First Years of Life
3.1. Delivery Route
3.2. Breastfeeding
3.3. Antibiotic Therapy
3.4. Pneumococcal Vaccination
3.5. Smoking
4. Acute Otitis Media
5. Otitis Media with Effusion
6. Chronic Suppurative Otitis Media
7. Probiotic Therapy
8. Conclusions
- Defining standard criteria of specimen collection, analysis, and data reporting, in order to facilitate data comparison across studies;
- Deepening our knowledge on the impact of various exogenous factors that have been less explored, such as active/passive smoking, vaccines, and viral infections;
- Confirming the role of Corynebacterium and/or Dolosigranulum as keystone taxa, in order to evaluate their possible use as probiotics;
- Understanding the development of URT and ME microbiota at different ages, in order to identify a potential “window of opportunity” in which therapeutic interventions as probiotic administration could be more effective, before the establishment of a stable microbial community that could be modulated with difficulty;
- Investigating the concordance between NP and ME microbiota, in order to better define the role of adenoid pad as a proxy for ME;
- Providing data on microbial communities in ME, which is no longer considered a sterile site;
- Defining with major detail the features of NP and ME microbial communities in different OM phenotypes, in particular in children with recurrent STMP.
Author Contributions
Funding
Conflicts of Interest
References
- The NIH HMP Working Group; Peterson, J.; Garges, S.; Giovanni, M.; McInnes, P.; Wang, L.; Schloss, J.A.; Bonazzi, V.; McEwen, J.E.; Wetterstrand, K.A.; et al. The NIH Human Microbiome Project. Genome Res. 2009, 19, 2317–2323. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Patel, A.H.; Harris, K.A.; Fitzgerald, F. What is broad-range 16S rDNA PCR? Arch. Dis. Child. Educ. Pract. Ed. 2017, 102, 261–264. [Google Scholar] [CrossRef] [PubMed]
- Samuelson, D.R.; Welsh, D.A.; Shellito, J.E. Regulation of lung immunity and host defense by the intestinal microbiota. Front. Microbiol. 2015, 6, 1085. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Buffie, C.G.; Pamer, E.G. Microbiota-mediated colonization resistance against intestinal pathogens. Nat. Rev. Immunol. 2013, 13, 790–801. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Honda, K.; Littman, D.R. The microbiome in infectious disease and inflammation. Annu. Rev. Immunol. 2012, 30, 759–795. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schuster, S.C. Next-generation sequencing transforms today’s biology. Nat. Methods 2007, 5, 16–18. [Google Scholar] [CrossRef] [PubMed]
- Knight, R.; Vrbanac, A.; Taylor, B.C.; Aksenov, A.; Callewaert, C.; Debelius, J.; González, A.; Kosciolek, T.; McCall, L.-I.; McDonald, D.; et al. Best practices for analysing microbiomes. Nat. Rev. Genet. 2018, 16, 410–422. [Google Scholar] [CrossRef] [Green Version]
- Paradise, J.L.; Rockette, H.E.; Colborn, D.K.; Bernard, B.S.; Smith, C.G.; Kurs-Lasky, M.; Janosky, J.E. Otitis Media in 2253 Pittsburgh-Area Infants: Prevalence and Risk Factors During the First Two Years of Life. Pediatrics 1997, 99, 318–333. [Google Scholar] [CrossRef]
- Marom, T.; Marchisio, P.G.; Tamir, S.O.; Torretta, S.; Gavriel, H.; Esposito, S. Complementary and Alternative Medicine Treatment Options for Otitis Media. Medicine 2016, 95, e2695. [Google Scholar] [CrossRef]
- Schilder, A.G.; Chonmaitree, T.; Cripps, A.W.; Rosenfeld, R.M.; Casselbrant, M.L.; Haggard, M.P.; Venekamp, R.P. Otitis media. Nat. Rev. Dis. Prim. 2016, 2, 16063. [Google Scholar] [CrossRef]
- Bernstein, J.M.; Reddy, M.S.; Scannapieco, F.A.; Faden, H.S.; Ballow, M. The Microbial Ecology and Immunology of the Adenoid: Implications for Otitis Media. Ann. N.Y. Acad. Sci. 1997, 830, 19–31. [Google Scholar] [CrossRef] [PubMed]
- Nistico, L.; Kreft, R.; Gieseke, A.; Coticchia, J.M.; Burrows, A.; Khampang, P.; Liu, Y.; Kerschner, J.E.; Post, J.C.; Lonergan, S.; et al. Adenoid Reservoir for Pathogenic Biofilm Bacteria. J. Clin. Microbiol. 2011, 49, 1411–1420. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hoa, M.; Tomovic, S.; Nistico, L.; Hall-Stoodley, L.; Stoodley, P.; Sachdeva, L.; Berk, R.; Coticchia, J.M. Identification of adenoid biofilms with middle ear pathogens in otitis-prone children utilizing SEM and FISH. Int. J. Pediatr. Otorhinolaryngol. 2009, 73, 1242–1248. [Google Scholar] [CrossRef] [PubMed]
- Bogaert, D.; De Groot, R.; Hermans, P. Streptococcus pneumoniae colonisation: The key to pneumococcal disease. Lancet Infect. Dis. 2004, 4, 144–154. [Google Scholar] [CrossRef]
- Man, W.H.; Piters, W.A.A.D.S.; Bogaert, D. The microbiota of the respiratory tract: Gatekeeper to respiratory health. Nat. Rev. Genet. 2017, 15, 259–270. [Google Scholar] [CrossRef] [PubMed]
- Piters, W.A.A.D.S.; Sanders, E.A.M.; Bogaert, D. The role of the local microbial ecosystem in respiratory health and disease. Philos. Trans. R. Soc. B Biol. Sci. 2015, 370, 20140294. [Google Scholar] [CrossRef] [Green Version]
- Tano, K.; Grahn-Håkansson, E.; Holm, S.E.; Hellström, S. Inhibition of OM pathogens by alpha-hemolytic streptococci from healthy children, children with SOM and children with rAOM. Int. J. Pediatr. Otorhinolaryngol. 2000, 56, 185–190. [Google Scholar] [CrossRef]
- Tano, K.; Olofsson, C.; Grahn-Håkansson, E.; Holm, S.E. In vitro inhibition of S. pneumoniae, nontypable H. influenzae and M. catharralis by alpha-hemolytic streptococci from healthy children. Int. J. Pediatr. Otorhinolaryngol. 1999, 47, 49–56. [Google Scholar] [CrossRef]
- Pettigrew, M.M.; Laufer, A.S.; Gent, J.F.; Kong, Y.; Fennie, K.; Metlay, J.P. Upper Respiratory Tract Microbial Communities, Acute Otitis Media Pathogens, and Antibiotic Use in Healthy and Sick Children. Appl. Environ. Microbiol. 2012, 78, 6262–6270. [Google Scholar] [CrossRef] [Green Version]
- Laufer, A.S.; Metlay, J.P.; Gent, J.F.; Fennie, K.; Kong, Y.; Pettigrew, M.M. Microbial Communities of the Upper Respiratory Tract and Otitis Media in Children. mBio 2011, 2, e00245–e00310. [Google Scholar] [CrossRef] [Green Version]
- Bomar, L.; Brugger, S.D.; Yost, B.H.; Davies, S.S.; Lemon, K.P. Corynebacterium accolensReleases Antipneumococcal Free Fatty Acids from Human Nostril and Skin Surface Triacylglycerols. mBio 2016, 7, 01725. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lappan, R.; Imbrogno, K.; Sikazwe, C.; Anderson, D.; Mok, D.; Coates, H.; Vijayasekaran, S.; Bumbak, P.; Blyth, C.; Jamieson, S.E.; et al. A microbiome case-control study of recurrent acute otitis media identified potentially protective bacterial genera. BMC Microbiol. 2018, 18, 13. [Google Scholar] [CrossRef] [PubMed]
- Tano, K.; Håkansson, E.G.; Holm, S.E.; Hellström, S. A nasal spray with alpha-haemolytic streptococci as long term prophylaxis against recurrent otitis media. Int. J. Pediatr. Otorhinolaryngol. 2002, 62, 17–23. [Google Scholar] [CrossRef]
- Marchisio, P.G.; Santagati, M.C.; Scillato, M.; Baggi, E.; Fattizzo, M.; Rosazza, C.; Stefani, S.; Esposito, S.; Principi, N. Streptococcus salivarius 24SMB administered by nasal spray for the prevention of acute otitis media in otitis-prone children. Eur. J. Clin. Microbiol. Infect. Dis. 2015, 34, 2377–2383. [Google Scholar] [CrossRef] [PubMed]
- Costello, E.K.; Lauber, C.L.; Hamady, M.; Fierer, N.; Gordon, J.I.; Knight, R. Bacterial Community Variation in Human Body Habitats Across Space and Time. Science 2009, 326, 1694–1697. [Google Scholar] [CrossRef] [Green Version]
- Thomas, S.; Izard, J.; Walsh, E.; Batich, K.; Chongsathidkiet, P.; Clarke, G.; Sela, D.A.; Muller, A.J.; Mullin, J.M.; Albert, K.; et al. The Host Microbiome Regulates and Maintains Human Health: A Primer and Perspective for Non-Microbiologists. Cancer Res. 2017, 77, 1783–1812. [Google Scholar] [CrossRef] [Green Version]
- Charbonneau, M.R.; Blanton, L.V.; DiGiulio, D.B.; Relman, D.A.; Lebrilla, C.B.; Mills, D.A.; Gordon, J.I. A microbial perspective of human developmental biology. Nature 2016, 535, 48–55. [Google Scholar] [CrossRef] [Green Version]
- Dominguez-Bello, M.G.; Costello, E.K.; Contreras, M.; Magris, M.; Hidalgo, G.; Fierer, N.; Knight, R. Delivery mode shapes the acquisition and structure of the initial microbiota across multiple body habitats in newborns. Proc. Natl. Acad. Sci. USA 2010, 107, 11971–11975. [Google Scholar] [CrossRef] [Green Version]
- Bosch, A.A.; Levin, E.; Van Houten, M.A.; Hasrat, R.; Kalkman, G.; Biesbroek, G.; Piters, W.A.A.D.S.; De Groot, P.-K.C.; Pernet, P.; Keijser, B.J.; et al. Development of Upper Respiratory Tract Microbiota in Infancy is Affected by Mode of Delivery. EBioMedicine 2016, 9, 336–345. [Google Scholar] [CrossRef] [Green Version]
- Biesbroek, G.; Tsivtsivadze, E.; Sanders, E.A.M.; Montijn, R.; Veenhoven, R.H.; Keijser, B.J.F.; Bogaert, D. Early Respiratory Microbiota Composition Determines Bacterial Succession Patterns and Respiratory Health in Children. Am. J. Respir. Crit. Care Med. 2014, 190, 1283–1292. [Google Scholar] [CrossRef]
- Teo, S.M.; Mok, D.; Pham, K.; Kusel, M.; Serralha, M.; Troy, N.; Holt, B.J.; Hales, B.J.; Walker, M.L.; Hollams, E.; et al. The infant nasopharyngeal microbiome impacts severity of lower respiratory infection and risk of asthma development. Cell Host Microbe 2015, 17, 704–715. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Guibas, G.V.; Moschonis, G.; Xepapadaki, P.; Roumpedaki, E.; Androutsos, O.; Manios, Y.; Papadopoulos, N.G. Conception viain vitrofertilization and delivery by Caesarean section are associated with paediatric asthma incidence. Clin. Exp. Allergy 2013, 43, 1058–1066. [Google Scholar] [CrossRef] [PubMed]
- Kristensen, K.; Fisker, N.; Haerskjold, A.; Ravn, H.; Simões, E.A.F.; Stensballe, L. Caesarean Section and Hospitalization for Respiratory Syncytial Virus Infection: A population-based study. Pediatr. Infect. Dis. J. 2015, 34, 145–148. [Google Scholar] [CrossRef] [PubMed]
- Grice, E.A.; Segre, J.A. The skin microbiome. Nat. Rev. Genet. 2011, 9, 244–253. [Google Scholar] [CrossRef]
- Mendling, W. Vaginal Microbiota. Adv. Exp. Med. Biol. 2016, 902, 83–93. [Google Scholar] [CrossRef]
- Chu, D.M.; Ma, J.; Prince, A.L.; Antony, K.M.; Seferovic, M.D.; Aagaard, K.M. Maturation of the infant microbiome community structure and function across multiple body sites and in relation to mode of delivery. Nat. Med. 2017, 23, 314–326. [Google Scholar] [CrossRef] [Green Version]
- Tarrant, M.; Kwok, M.K.; Lam, T.-H.; Leung, G.M.; Schooling, C.M. Breast-feeding and Childhood Hospitalizations for Infections. Epidemiology 2010, 21, 847–854. [Google Scholar] [CrossRef] [Green Version]
- Duijts, L.; Jaddoe, V.W.V.; Hofman, A.; Moll, H.A. Prolonged and Exclusive Breastfeeding Reduces the Risk of Infectious Diseases in Infancy. Pediatrics 2010, 126, e18–e25. [Google Scholar] [CrossRef] [Green Version]
- Labbok, M.; Clark, D.; Goldman, A.S. Breastfeeding: Maintaining an irreplaceable immunological resource. Nat. Rev. Immunol. 2004, 4, 565–572. [Google Scholar] [CrossRef]
- Biesbroek, G.; Bosch, A.A.; Wang, X.; Keijser, B.J.F.; Veenhoven, R.H.; Sanders, E.A.; Bogaert, D. The Impact of Breastfeeding on Nasopharyngeal Microbial Communities in Infants. Am. J. Respir. Crit. Care Med. 2014, 190, 298–308. [Google Scholar] [CrossRef]
- Bosch, A.A.T.M.; Wouter, A.A.d.S.P.; Van Houten, M.A.; Chu, M.L.J.N.; Biesbroek, G.; Kool, J.; Pernet, P.; De Groot, P.-K.C.M.; Eijkemans, M.J.C.; Keijser, B.J.F.; et al. Maturation of the Infant Respiratory Microbiota, Environmental Drivers, and Health Consequences. A Prospective Cohort Study. Am. J. Respir. Crit. Care Med. 2017, 196, 1582–1590. [Google Scholar] [CrossRef] [PubMed]
- Sullivan, Å.; Edlund, C.; Nord, C.E. Effect of antimicrobial agents on the ecological balance of human microflora. Lancet Infect. Dis. 2001, 1, 101–114. [Google Scholar] [CrossRef]
- Hicks, L.A.; Taylor, T.H.; Hunkler, R.J. U.S. Outpatient Antibiotic Prescribing, 2010. N. Engl. J. Med. 2013, 368, 1461–1462. [Google Scholar] [CrossRef] [PubMed]
- Hilty, M.; Qi, W.; Brugger, S.D.; Frei, L.; Agyeman, P.; Frey, P.M.; Aebi, S.; Mühlemann, K. Nasopharyngeal Microbiota in Infants with Acute Otitis Media. J. Infect. Dis. 2012, 205, 1048–1055. [Google Scholar] [CrossRef]
- Chonmaitree, T.; Jennings, K.; Golovko, G.; Khanipov, K.; Pimenova, M.; Patel, J.A.; McCormick, D.P.; Loeffelholz, M.J.; Fofanov, Y. Nasopharyngeal microbiota in infants and changes during viral upper respiratory tract infection and acute otitis media. PLoS ONE 2017, 12, e0180630. [Google Scholar] [CrossRef]
- Pettigrew, M.M.; Alderson, M.R.; Bakaletz, L.O.; Barenkamp, S.J.; Hakansson, A.P.; Mason, K.M.; Nokso-Koivisto, J.; Patel, J.; Pelton, S.I.; Murphy, T.F. Panel 6: Vaccines. Otolaryngol. Neck Surg. 2017, 156 (Suppl. 4), S76–S87. [Google Scholar] [CrossRef]
- Ngo, C.C.; Massa, H.M.; Thornton, R.B.; Cripps, A.W. Predominant Bacteria Detected from the Middle Ear Fluid of Children Experiencing Otitis Media: A Systematic Review. PLoS ONE 2016, 11, e0150949. [Google Scholar] [CrossRef] [Green Version]
- Gladstone, R.A.; Jefferies, J.M.; Tocheva, A.S.; Beard, K.R.; Garley, D.; Chong, W.W.; Bentley, S.D.; Faust, S.N.; Clarke, S.C. Five winters of pneumococcal serotype replacement in UK carriage following PCV introduction. Vaccine 2015, 33, 2015–2021. [Google Scholar] [CrossRef] [Green Version]
- Biesbroek, G.; Wang, X.; Keijser, B.J.; Eijkemans, R.M.; Trzcinski, K.; Rots, N.Y.; Veenhoven, R.H.; Sanders, E.A.; Bogaert, D. Seven-Valent Pneumococcal Conjugate Vaccine and Nasopharyngeal Microbiota in Healthy Children. Emerg. Infect. Dis. 2014, 20, 201–210. [Google Scholar] [CrossRef]
- Mika, M.; Maurer, J.; Korten, I.; Allemann, A.; Aebi, S.; Brugger, S.D.; Qi, W.; Frey, U.; Latzin, P.; Hilty, M. Influence of the pneumococcal conjugate vaccines on the temporal variation of pneumococcal carriage and the nasal microbiota in healthy infants: A longitudinal analysis of a case–control study. Microbiome 2017, 5, 85. [Google Scholar] [CrossRef]
- Feazel, L.M.; Santorico, S.A.; Robertson, C.E.; Bashraheil, M.; Scott, J.A.G.; Frank, D.; Hammitt, L.L. Effects of Vaccination with 10-Valent Pneumococcal Non-Typeable Haemophilus influenza Protein D Conjugate Vaccine (PHiD-CV) on the Nasopharyngeal Microbiome of Kenyan Toddlers. PLoS ONE 2015, 10, e0128064. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kwambana-Adams, B.A.; Hanson, B.; Worwui, A.; Agbla, S.; Foster-Nyarko, E.; Ceesay, F.; Ebruke, C.; Egere, U.; Zhou, Y.; Ndukum, M.; et al. Rapid replacement by non-vaccine pneumococcal serotypes may mitigate the impact of the pneumococcal conjugate vaccine on nasopharyngeal bacterial ecology. Sci. Rep. 2017, 7, 1–11. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Andrade, D.C.; Borges, I.C.; Bouzas, M.L.; Oliveira, J.R.; Fukutani, K.F.; Queiroz, A.T.; De Oliveira, C.I.; Barral, A.; Van Weyenbergh, J.; Nascimento-Carvalho, C. 10-valent pneumococcal conjugate vaccine (PCV10) decreases metabolic activity but not nasopharyngeal carriage of Streptococcus pneumoniae and Haemophilus influenzae. Vaccine 2017, 35, 4105–4111. [Google Scholar] [CrossRef]
- Adegbola, R.A.; DeAntonio, R.; Hill, P.C.; Roca, A.; Usuf, E.; Hoet, B.; Greenwood, B.M. Carriage of Streptococcus pneumoniae and Other Respiratory Bacterial Pathogens in Low and Lower-Middle Income Countries: A Systematic Review and Meta-Analysis. PLoS ONE 2014, 9, e103293. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Boelsen, L.K.; Dunne, E.M.; Mika, M.; Eggers, S.; Nguyen, C.D.; Ratu, F.T.; Russell, F.M.; Mulholland, E.K.; Hilty, M.; Satzke, C. The association between pneumococcal vaccination, ethnicity, and the nasopharyngeal microbiota of children in Fiji. Microbiome 2019, 7, 106. [Google Scholar] [CrossRef]
- Charlson, E.S.; Chen, J.; Custers-Allen, R.; Bittinger, K.; Li, H.; Sinha, R.; Hwang, J.; Bushman, F.D.; Collman, R.G. Disordered Microbial Communities in the Upper Respiratory Tract of Cigarette Smokers. PLoS ONE 2010, 5, e15216. [Google Scholar] [CrossRef] [Green Version]
- Teele, D.W.; Klein, J.O.; Rosner, B. Epidemiology of Otitis Media During the First Seven Years of Life in Children in Greater Boston: A Prospective, Cohort Study. J. Infect. Dis. 1989, 160, 83–94. [Google Scholar] [CrossRef]
- Dirain, C.O.; Silva, R.C.; Collins, W.O.; Antonelli, P.J. The Adenoid Microbiome in Recurrent Acute Otitis Media and Obstructive Sleep Apnea. J. Int. Adv. Otol. 2017, 13, 333–339. [Google Scholar] [CrossRef] [Green Version]
- Sillanpää, S.; Kramna, L.; Oikarinen, S.; Sipilä, M.; Rautiainen, M.; Aittoniemi, J.; Laranne, J.; Hyöty, H.; Cinek, O. Next-Generation Sequencing Combined with Specific PCR Assays To Determine the Bacterial 16S rRNA Gene Profiles of Middle Ear Fluid Collected from Children with Acute Otitis Media. mSphere 2017, 2, 00006–00017. [Google Scholar] [CrossRef] [Green Version]
- Xu, Q.; Gill, S.; Xu, L.; Gonzalez, E.; Pichichero, M.E. Comparative Analysis of Microbiome in Nasopharynx and Middle Ear in Young Children With Acute Otitis Media. Front. Genet. 2019, 10, 1–7. [Google Scholar] [CrossRef] [Green Version]
- Brugger, S.D.; Kraemer, J.G.; Qi, W.; Bomar, L.; Oppliger, A.; Hilty, M. Age-Dependent Dissimilarity of the Nasopharyngeal and Middle Ear Microbiota in Children With Acute Otitis Media. Front. Genet. 2019, 10, 555. [Google Scholar] [CrossRef] [PubMed]
- Berger, G. Nature of spontaneous tympanic membrane perforation in acute otitis media in children. J. Laryngol. Otol. 1989, 103, 1150–1153. [Google Scholar] [CrossRef] [PubMed]
- Torretta, S.; Marchisio, P. Otitis media in children: A proposal for a new nosological classification. Int. J. Pediatr. Otorhinolaryngol. 2017, 93, 174–175. [Google Scholar] [CrossRef] [PubMed]
- Marchisio, P.G.; Nazzari, E.; Torretta, S.; Esposito, S.; Principi, N. Medical prevention of recurrent acute otitis media: An updated overview. Expert Rev. Anti Infect. Ther. 2014, 12, 611–620. [Google Scholar] [CrossRef]
- Marchisio, P.G.; Esposito, S.; Bianchini, S.; Dusi, E.; Fusi, M.; Nazzari, E.; Picchi, R.; Galeone, C.; Principi, N. Efficacy of Injectable Trivalent Virosomal-Adjuvanted Inactivated Influenza Vaccine in Preventing Acute Otitis Media in Children With Recurrent Complicated or Noncomplicated Acute Otitis Media. Pediatr. Infect. Dis. J. 2009, 28, 855–859. [Google Scholar] [CrossRef]
- Marchisio, P.G.; Consonni, D.; Baggi, E.; Zampiero, A.; Bianchini, S.; Terranova, L.; Tirelli, S.; Esposito, S.; Principi, N. Vitamin D Supplementation Reduces the Risk of Acute Otitis Media in Otitis-prone Children. Pediatr. Infect. Dis. J. 2013, 32, 1055–1060. [Google Scholar] [CrossRef]
- Man, W.H.; Van Dongen, T.M.; Venekamp, R.P.; Pluimakers, V.G.; Chu, M.L.J.; Van Houten, M.A.; Sanders, E.A.; Schilder, A.G.M.; Bogaert, D. Respiratory Microbiota Predicts Clinical Disease Course of Acute Otorrhea in Children With Tympanostomy Tubes. Pediatr. Infect. Dis. J. 2019, 38, e116–e125. [Google Scholar] [CrossRef] [Green Version]
- Liu, C.M.; Cosetti, M.K.; Aziz, M.; Buchhagen, J.L.; Contente-Cuomo, T.L.; Price, L.B.; Keim, P.; Lalwani, A.K. The Otologic MicrobiomeA Study of the Bacterial Microbiota in a Pediatric Patient With Chronic Serous Otitis Media Using 16SrRNA Gene-Based Pyrosequencing. Arch. Otolaryngol. Head Neck Surg. 2011, 137, 664–668. [Google Scholar] [CrossRef] [Green Version]
- Frank, D.N.; Spiegelman, G.B.; Davis, W.; Wagner, E.; Lyons, E.; Pace, N.R. Culture-independent molecular analysis of microbial constituents of the healthy human outer ear. J. Clin. Microbiol. 2003, 41, 295–303. [Google Scholar] [CrossRef] [Green Version]
- Jervis-Bardy, J.; Rogers, G.B.; Morris, P.S.; Smith-Vaughan, H.C.; Nosworthy, E.; Leong, L.E.X.; Smith, R.J.; Weyrich, L.S.; De Haan, J.; Carney, A.S.; et al. The microbiome of otitis media with effusion in Indigenous Australian children. Int. J. Pediatr. Otorhinolaryngol. 2015, 79, 1548–1555. [Google Scholar] [CrossRef]
- Fagö-Olsen, H.; Dines, L.M.; Sørensen, C.H.; Jensen, A. The Adenoids but Not the Palatine Tonsils Serve as a Reservoir for Bacteria Associated with Secretory Otitis Media in Small Children. mSystems 2019, 4, e00169–e00218. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Johnston, J.; Hoggard, M.; Biswas, K.; Astudillo-García, C.; Radcliff, F.J.; Mahadevan, M.; Douglas, R.G. Pathogen reservoir hypothesis investigated by analyses of the adenotonsillar and middle ear microbiota. Int. J. Pediatr. Otorhinolaryngol. 2019, 118, 103–109. [Google Scholar] [CrossRef]
- Ari, O.; Karabudak, S.; Kalcioglu, M.T.; Gunduz, A.Y.; Durmaz, R. The bacteriome of otitis media with effusion: Does it originate from the adenoid? Int. J. Pediatr. Otorhinolaryngol. 2019, 126, 109624. [Google Scholar] [CrossRef] [PubMed]
- Chan, C.L.; Wabnitz, D.; Bardy, J.J.; Bassiouni, A.; Wormald, P.-J.; Vreugde, S.; Psaltis, A.J. The microbiome of otitis media with effusion. Laryngoscope 2016, 126, 2844–2851. [Google Scholar] [CrossRef] [PubMed]
- Boers, S.A.; De Zeeuw, M.; Jansen, R.; Van Der Schroeff, M.P.; Van Rossum, A.M.C.; Hays, J.P.; Verhaegh, S.J.C. Characterization of the nasopharyngeal and middle ear microbiota in gastroesophageal reflux-prone versus gastroesophageal reflux non-prone children. Eur. J. Clin. Microbiol. Infect. Dis. 2018, 37, 851–857. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xu, J.; Dai, W.; Liang, Q.; Ren, D. The microbiomes of adenoid and middle ear in children with otitis media with effusion and hypertrophy from a tertiary hospital in China. Int. J. Pediatr. Otorhinolaryngol. 2020, 134, 110058. [Google Scholar] [CrossRef]
- Chan, C.L.; Wabnitz, D.; Bassiouni, A.; Wormald, P.-J.; Vreugde, S.; Psaltis, A.J. Identification of the Bacterial Reservoirs for the Middle Ear Using Phylogenic Analysis. JAMA Otolaryngol. Neck Surg. 2017, 143, 155–161. [Google Scholar] [CrossRef] [Green Version]
- Krueger, A.; Val, S.; Pérez-Losada, M.; Panchapakesan, K.; Devaney, J.; Duah, V.; DeMason, C.; Poley, M.; Rose, M.; Preciado, D.; et al. Relationship of the Middle Ear Effusion Microbiome to Secretory Mucin Production in Pediatric Patients With Chronic Otitis Media. Pediatr. Infect. Dis. J. 2017, 36, 635–640. [Google Scholar] [CrossRef] [PubMed]
- Kolbe, A.R.; Castro-Nallar, E.; Preciado, D.; Pérez-Losada, M. Altered Middle Ear Microbiome in Children With Chronic Otitis Media With Effusion and Respiratory Illnesses. Front. Microbiol. 2019, 9, 1–10. [Google Scholar] [CrossRef]
- Kim, S.K.; Hong, S.J.; Pak, K.H.; Hong, S.M. Analysis of the Microbiome in the Adenoids of Korean Children with Otitis Media with Effusion. J. Int. Adv. Otol. 2019, 15, 379–385. [Google Scholar] [CrossRef]
- Walker, R.E.; Walker, C.G.; Camargo, C.A., Jr.; Bartley, J.; Flint, D.; Thompson, J.M.D.; Mitchell, E.A. Nasal microbial composition and chronic otitis media with effusion: A case-control study. PLoS ONE 2019, 14, e0212473. [Google Scholar] [CrossRef] [PubMed]
- Neeff, M.; Biswas, K.; Hoggard, M.; Taylor, M.W.; Douglas, R.G. Molecular Microbiological Profile of Chronic Suppurative Otitis Media. J. Clin. Microbiol. 2016, 54, 2538–2546. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Minami, S.B.; Mutai, H.; Suzuki, T.; Horii, A.; Oishi, N.; Wasano, K.; Katsura, M.; Tanaka, F.; Takiguchi, T.; Fujii, M.; et al. Microbiomes of the normal middle ear and ears with chronic otitis media. Laryngoscope 2017, 127, E371–E377. [Google Scholar] [CrossRef] [PubMed]
- Santos-Cortez, R.L.P.; University of Washington Center for Mendelian Genomics; Chiong, C.M.; Reyes-Quintos, M.R.T.; Tantoco, M.L.C.; Wang, X.; Acharya, A.; Abbe, I.; Giese, A.P.J.; Smith, J.D.; et al. Rare A2ML1 variants confer susceptibility to otitis media. Nat. Genet. 2015, 47, 917–920. [Google Scholar] [CrossRef] [Green Version]
- Santos-Cortez, R.L.P.; Hutchinson, D.S.; Ajami, N.J.; Reyes-Quintos, M.R.T.; Tantoco, M.L.C.; Labra, P.J.; Lagrana, S.M.; Pedro, M.; Llanes, E.G.D.V.; Gloria-Cruz, T.L.; et al. Middle ear microbiome differences in indigenous Filipinos with chronic otitis media due to a duplication in the A2ML1 gene. Infect. Dis. Poverty 2016, 5, 97. [Google Scholar] [CrossRef] [Green Version]
- Hill, C.; Guarner, F.; Reid, G.; Gibson, G.R.; Merenstein, D.J.; Pot, B.; Morelli, L.; Canani, R.B.; Flint, H.J.; Salminen, S.; et al. The International Scientific Association for Probiotics and Prebiotics consensus statement on the scope and appropriate use of the term probiotic. Nat. Rev. Gastroenterol. Hepatol. 2014, 11, 506–514. [Google Scholar] [CrossRef] [Green Version]
- Broek, M.F.L.V.D.; De Boeck, I.; Kiekens, F.; Boudewyns, A.; Vanderveken, O.M.; Lebeer, S. Translating Recent Microbiome Insights in Otitis Media into Probiotic Strategies. Clin. Microbiol. Rev. 2019, 32, 1–33. [Google Scholar] [CrossRef]
- Chen, T.Y.; Hendrickx, A.; Stevenson, D.S.; Bird, P.; Walls, T. No evidence from a systematic review for the use of probiotics to prevent otitis media. Acta Paediatr. 2020. [Google Scholar] [CrossRef] [PubMed]
- Lappan, R.; Jamieson, S.E.; Peacock, C.S. Reviewing the Pathogenic Potential of the Otitis-Associated Bacteria Alloiococcus otitidis and Turicella otitidis. Front. Cell Infect. Microbiol. 2020, 10, 51. [Google Scholar] [CrossRef]
| Microbiota | Ecological communities of commensal, symbiotic and pathogenic microorganisms that colonize several body sites, as the gastrointestinal tract, respiratory system, oral cavity, skin, and female reproductive system |
| Microbiome | Genetic material of the microorganisms of a community |
| Keystone Species | Microorganisms with a great impact on an ecological community, considered important in maintaining its organization and function |
| Biodiversity | Number of OTUs in a community and their relative abundance. It is determined by richness (how many OTUs in a sample?) and evenness (how equally distributed relative abundances are in a sample?) |
| Alpha-Diversity | Diversity within sample: how abundant OTUs are in relation to others in the same sample? |
| Beta-Diversity | Measure that compares different microbial communities |
| Operational Taxonomic Unit (OTU) | Cluster of related sequences (usually with 97% or more similarity) that represent a taxonomic unit of a microorganism |
| Title (Year of Publication) [Ref] | Study Design | N. of Subjects | Age | Site of Investigation | Main Findings |
|---|---|---|---|---|---|
| Microbial Communities of the Upper Respiratory Tract and Otitis Media in Children (2011) [20] | Comparison of NP microbial communities in children with and without OM | 108 (25 with AOM; 83 without AOM) | 6–78 m | NP |
|
| Nasopharyngeal Microbiota in Infants with Acute Otitis Media (2012) [44] | Comparison of NP microbial communities in children with and without OM | 163 (153 with AOM; 10 without AOM) | <2 y | NP |
|
| Upper Respiratory Tract Microbial Communities, Acute Otitis Media Pathogens, and Antibiotic Use in Healthy and Sick Children (2012) [19] | Comparison of NP microbial communities in healthy children vs. children with URTI with and without concurrent AOM | 240 (73 healthy subjects; 95 subjects with URTI without concurrent AOM; 72 subjects with URTI with concurrent AOM) | 6 m–3 y | NP |
|
| Nasopharyngeal microbiota in infants and changes during viral upper respiratory tract infection and acute otitis media (2017) [45] | NP microbiota analysis of children followed from near birth for the first 12 months of life or until the occurrence of the first AOM episode. NP swabs collected monthly or during each URTI or AOM episode. | 139 patients (971 samples) | <1 y | NP |
|
| The Adenoid Microbiome in Recurrent Acute Otitis Media and Obstructive Sleep Apnea (2017) [58] | Comparison of adenoid microbiota in subjects undergoing surgery for RAOM or OSA | 10 (5 AOM; 5 OSA) | 2–11 y | Adenoid |
|
| Next-Generation Sequencing Combined with Specific PCR Assays To Determine the Bacterial 16S rRNA Gene Profiles of Middle Ear Fluid Collected from Children with Acute Otitis Media (2017) [59] | ME microbiota analysis during AOM episodes | 79 subjects (90 samples) | 5–42 m | ME |
|
| A microbiome case-control study of recurrent acute otitis media identified potentially protective bacterial genera (2018) [22] | Comparison of NP microbiota between children undergoing grommet insertion for RAOM (cases) vs. healthy children (controls); analysis of ME and EAC microbiota in cases | 196 (93 cases; 103 controls) | <5 y | NP ME EAC |
|
| Comparative Analysis of Microbiome in Nasopharynx and Middle Ear in Young Children with Acute Otitis Media (2019) [60] | Comparison of NP microbiota 1 to 3 weeks prior to onset of AOM vs. at onset of AOM; comparison of NP and ME microbiome during AOM | 6 | 6–24 m | NP ME |
|
| Age-Dependent Dissimilarity of the Nasopharyngeal and Middle Ear Microbiota in Children with Acute Otitis Media (2019) [61] | NP microbiota analysis during AOM; Paired NP and ME microbiota analysis in children with STMP | 286 (42/286 MEF from STMP) | 0–6 y | NP ME |
|
| Respiratory Microbiota Predicts Clinical Disease Course of Acute Otorrhea in Children with Tympanostomy Tubes (2019) [67] | Paired analysis of NP and ME microbiota in children with otorrhea on tympanostomy tubes | 94 | <5 y | NP ME |
|
| Title (Year of Publication) [Ref] | Study Design | N. of Subjects | Age | Site of Investigation | Main Findings |
|---|---|---|---|---|---|
| The Otologic Microbiome: A Study of the Bacterial Microbiota in a Pediatric Patient with Chronic Serous Otitis Media Using 16SrRNA Gene-Based Pyrosequencing (2011) [68] | Microbiota analysis in ME, adenoid, and tonsil specimens from one pediatric patient with chronic serous otitis media undergoing adenotonsillectomy and bilateral tympanic tube insertion | 1 | 8 y | Adenoid ME Tonsil |
|
| The microbiome of otitis media with effusion in Indigenous Australian children (2015) [70] | MEF, NP, and adenoid microbiota analysis in children undergoing surgical treatment for OME | 11 | 3–9 y | NP Adenoid ME |
|
| The Microbiome of Otitis Media with Effusion (2016) [74] | ME and adenoid microbiota analysis in children undergoing adenoidectomy with ventilation tube insertion for chronic OME. Adenoid microbiota analysis from healthy subjects enrolled as controls | 33 (23 subjects with OME; 10 healthy controls) | OME group: 1–8 y Control group: 1–12 y | ME Adenoid |
|
| The Relationship of the Middle Ear Effusion Microbiome to Secretory Mucin Production in Pediatric Patients with Chronic Otitis Media (2016) [78] | Microbiota analysis and mucin detection in MEF collected from children undergoing myringotomy with tympanostomy tube placement for chronic OME | 55 | 3–176 m | ME |
|
| Identification of the Bacterial Reservoirs for the Middle Ear Using Phylogenic Analysis (2017) [77] | ME and EAC microbiota analysis in children undergoing surgery for OME. Adenoid pad and ME microbiota analysis data were included from a previous study | 18 | 1–14 y | ME EAC |
|
| Pathogen reservoir hypothesis investigated by analyses of the adenotonsillar and middle ear microbiota (2018) [72] | Adenoid, middle ear, and tonsil microbiota analysis in children undergoing surgical treatment for OME | 10 | 5–10 y | Adenoid ME Tonsil |
|
| Characterization of the nasopharyngeal and middle ear microbiota in gastroesophageal reflux-prone versus gastroesophageal reflux non-prone children (2018) [75] | Analysis of NP and ME microbiota in children suffering from GER-associated OM vs. OM only undergoing surgical treatment for RAOM, COME, or both | 30 (9 subjects with GER-associated OM; 21 subjects with OM without GER) | GER group 1.3–6 y No GER group 0.8–12–8 y | NP ME |
|
| The Adenoids but Not the Palatine Tonsils Serve as a Reservoir for Bacteria Associated with Secretory Otitis Media in Small Children (2019) [76] | Adenoid and tonsillar microbiota analysis in children undergoing surgical treatment for hyperplasia of adenoids/tonsils without infection (HP group) vs. children undergoing surgery for SOM | 28 (112 samples) (14 subjects in HP group; 14 subjects in SOM group) | HP group 24–65 m SOM group 15–59 m | Adenoid Tonsils |
|
| Nasal microbial composition and chronic otitis media with effusion: A case-control study (2019) [81] | Comparison of nasal microbiota between children undergoing surgery for COME vs. healthy subjects | 178 (73 cases; 105 controls) | Case group: mean age 47.5 m Control group: mean age 49.6 m | Nasal (anterior nares) |
|
| Altered Middle Ear Microbiome in Children with Chronic Otitis Media with Effusion and Respiratory Illnesses (2019) [79] | Comparison of ME microbiota children with chronic OME and history of lower airways disease (asthma or bronchiolitis) vs. children with chronic OME without history of lower airways disease | 50 (13 with history of lower airway disease) | 3–176 m | ME |
|
| Analysis of the Microbiome in the Adenoids of Korean Children with Otitis Media with Effusion (2019) [80] | Adenoid microbiota comparison between children undergoing surgery for OME vs. children without undergoing surgery for obstructive symptoms | 32 (16 subjects with OME; 16 subjects without OME) | 19 m–15 y | Adenoid |
|
| The bacteriome of otitis media with effusion: does it originate from the adenoid? (2019) [73] | Adenoid and ME microbiota analysis in children undergoing surgery for OME | 25 | 1.5–9 y | Adenoid ME |
|
| The microbiomes of adenoid and middle ear in children with otitis media with effusion and hypertrophy from a tertiary hospital in China (2020) [76] | Adenoid and ME microbiota analysis in children undergoing surgical treatment for OME vs. adenoid microbiota analysis in children undergoing surgery for OSA without ear disease | 30 (15 in OME group; 15 in OSA group) | OME group 60–108 m OSA group 8–96 m | Adenoid ME |
|
| Title (Year of Publication) [Ref] | Study Design | N. of Subjects | Age | Site of Investigation | Main Findings |
|---|---|---|---|---|---|
| Molecular Microbiological Profile of Chronic Suppurative Otitis Media (2016) [82] | Comparison of ME and mastoid microbiota in patients with CSOM undergoing surgery vs. healthy controls | 46 (24 subjects with CSOM; 22 healthy subjects) | 6 m–85 y | ME Mastoid cavity |
|
| Microbiomes of the Normal Middle Ear and Ears with Chronic Otitis Media (2017) [83] | ME microbiota analysis in patients undergoing tympanoplasty for wet or dry COM vs. subjects undergoing surgery from other conditions than otitis media | 155 (67 healthy subjects; 44 subjects with COM without active infection; 44 subjects with COM with active infection) | 1–84 y | ME |
|
| Middle ear microbiome differences in indigenous Filipinos with chronic otitis media due to a duplication in the A2ML1 gene (2016) [85] | ME and EAC microbiota analysis in indigenous Filipinos with chronic otitis media; comparison of microbial communities in subjects carriers of A2ML1 variant vs. non carrier subjects | 16 (11 subjects carriers of A2ML1 variant) | 4–24 y | ME EAC |
|
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Folino, F.; Ruggiero, L.; Capaccio, P.; Coro, I.; Aliberti, S.; Drago, L.; Marchisio, P.; Torretta, S. Upper Respiratory Tract Microbiome and Otitis Media Intertalk: Lessons from the Literature. J. Clin. Med. 2020, 9, 2845. https://doi.org/10.3390/jcm9092845
Folino F, Ruggiero L, Capaccio P, Coro I, Aliberti S, Drago L, Marchisio P, Torretta S. Upper Respiratory Tract Microbiome and Otitis Media Intertalk: Lessons from the Literature. Journal of Clinical Medicine. 2020; 9(9):2845. https://doi.org/10.3390/jcm9092845
Chicago/Turabian StyleFolino, Francesco, Luca Ruggiero, Pasquale Capaccio, Ilaria Coro, Stefano Aliberti, Lorenzo Drago, Paola Marchisio, and Sara Torretta. 2020. "Upper Respiratory Tract Microbiome and Otitis Media Intertalk: Lessons from the Literature" Journal of Clinical Medicine 9, no. 9: 2845. https://doi.org/10.3390/jcm9092845
APA StyleFolino, F., Ruggiero, L., Capaccio, P., Coro, I., Aliberti, S., Drago, L., Marchisio, P., & Torretta, S. (2020). Upper Respiratory Tract Microbiome and Otitis Media Intertalk: Lessons from the Literature. Journal of Clinical Medicine, 9(9), 2845. https://doi.org/10.3390/jcm9092845






