Prevention of Type 1 Diabetes: Past Experiences and Future Opportunities
Abstract
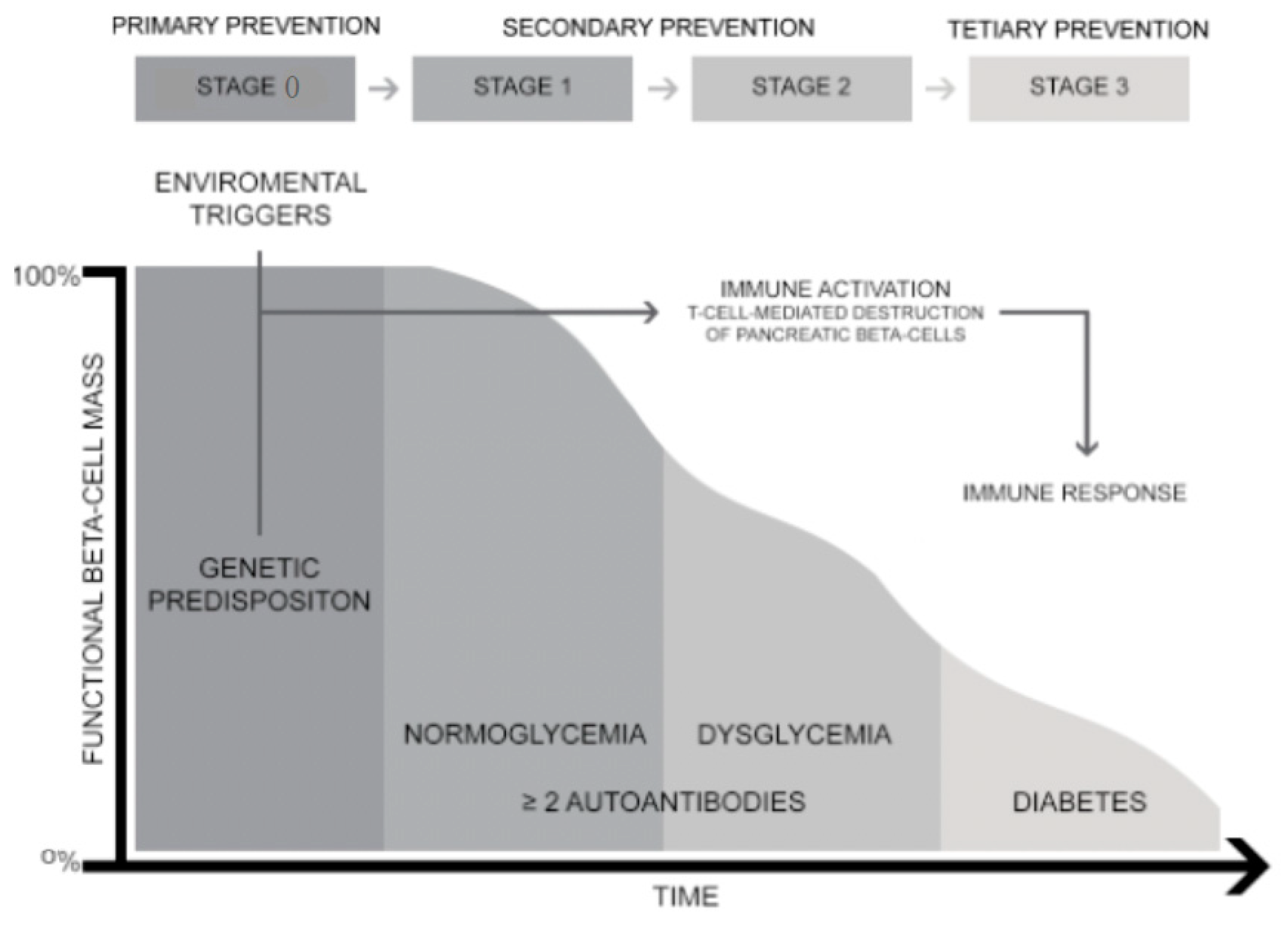
1. Introduction
2. Primary Prevention
3. Secondary Prevention
4. Tertiary Prevention
5. Discussion
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Nam, H.C. IDF Diabetes Atlas, 9th ed.; International Diabetes Federation: Brussels, Belgium, 2019; ISBN 9782930229874. [Google Scholar]
- Noble, J.A.; Valdes, A.M. Genetics of the HLA Region in the Prediction of Type 1 Diabetes. Curr. Diabetes Rep. 2011, 11, 533–542. [Google Scholar] [CrossRef]
- Rewers, M.; Ludvigsson, J. Environmental risk factors for type 1 diabetes. Lancet 2016, 387, 2340–2348. [Google Scholar] [CrossRef]
- Pellegrino, M.; Crinò, A.; Rosado, M.M.; Fierabracci, A. Identification and functional characterization of CD8+ T regulatory cells in type 1 diabetes patients. PLoS ONE 2019, 14, e0210839. [Google Scholar] [CrossRef] [PubMed]
- Taplin, C.E.; Barker, J.M. Autoantibodies in type 1 diabetes. Autoimmunity 2008, 41, 11–18. [Google Scholar] [CrossRef] [PubMed]
- Niechciał, E.; Rogowicz-Frontczak, A.; Piłaciński, S.; Fichna, M.; Skowronska, B.; Fichna, P.; Zozulińska-Ziółkiewicz, D. Autoantibodies against zinc transporter 8 are related to age and metabolic state in patients with newly diagnosed autoimmune diabetes. Acta Diabetol. 2018, 55, 287–294. [Google Scholar] [CrossRef] [PubMed]
- Redondo, M.J.; Steck, A.K.; Pugliese, A. Genetics of type 1 diabetes. Pediatr. Diabetes 2017, 19, 346–353. [Google Scholar] [CrossRef] [PubMed]
- Hanscombe, K.B.; Morris, D.L.; Noble, J.A.; Dilthey, A.T.; Tombleson, P.; Kaufman, K.M.; Comeau, M.; Langefeld, C.D.; Alarcon-Riquelme, M.E.; Gaffney, P.M.; et al. Genetic fine mapping of systemic lupus erythematosus MHC associations in Europeans and African Americans. Hum. Mol. Genet. 2018, 27, 3813–3824. [Google Scholar] [CrossRef]
- Kawabata, Y.; Ikegami, H.; Kawaguchi, Y.; Fujisawa, T.; Shintani, M.; Ono, M.; Nishino, M.; Uchigata, Y.; Lee, I.; Ogihara, T. Asian-Specific HLA Haplotypes Reveal Heterogeneity of the Contribution of HLA-DR and -DQ Haplotypes to Susceptibility to Type 1 Diabetes. Diabetes 2002, 51, 545–551. [Google Scholar] [CrossRef]
- Rawshani, A.; Sattar, N.; Franzén, S.; Rawshani, A.; Hattersley, A.T.; Svensson, A.M.; Eliasson, B.; Gudbjörnsdottir, S. Excess mortality and cardiovascular disease in young adults with type 1 diabetes in relation to age at onset: A nationwide, register-based cohort study. Lancet 2018, 392, 477–486. [Google Scholar] [CrossRef]
- Couper, J.; Haller, M.J.; Greenbaum, C.; Ziegler, A.G.; Wherrett, D.K.; Knip, M.; Craig, M.; Couper, J.J. ISPAD Clinical Practice Consensus Guidelines 2018: Stages of type 1 diabetes in children and adolescents. Pediatr. Diabetes 2018, 19, 20–27. [Google Scholar] [CrossRef]
- Eisenbarth, G.; Nayak, R.; Rabinowe, S. Type I diabetes as a chronic autoimmune disease. J. Diabet. Complicat. 1988, 2, 54–58. [Google Scholar] [CrossRef]
- Insel, R.; Dunne, J.L.; Atkinson, M.A.; Chiang, J.L.; Dabelea, D.; Gottlieb, P.A.; Greenbaum, C.J.; Herold, K.C.; Krischer, J.P.; Lernmark, A.; et al. Staging Presymptomatic Type 1 Diabetes: A Scientific Statement of JDRF, the Endocrine Society, and the American Diabetes Association. Diabetes Care 2015, 38, 1964–1974. [Google Scholar] [CrossRef] [PubMed]
- Writing Group for the TRIGR Study Group; Knip, M.; Åkerblom, H.K.; Al Taji, E.; Becker, D.; Bruining, J.; Castaño, L.; Danne, T.; De Beaufort, C.; Dosch, H.M.; et al. Effect of Hydrolyzed Infant Formula vs Conventional Formula on Risk of Type 1 Diabetes. JAMA 2018, 319, 38–48. [Google Scholar] [CrossRef] [PubMed]
- Vaarala, O.; Ilonen, J.; Ruohtula, T.; Pesola, J.; Virtanen, S.M.; Härkönen, T.; Koski, M.; Kallioinen, H.; Tossavainen, O.; Poussa, T.; et al. Removal of Bovine Insulin From Cow’s Milk Formula and Early Initiation of Beta-Cell Autoimmunity in the FINDIA Pilot Study. Arch. Pediatr. Adolesc. Med. 2012, 166, 608–614. [Google Scholar] [CrossRef]
- Hummel, S.; Pflüger, M.; Hummel, M.; Bonifacio, E.; Ziegler, A.-G. Primary Dietary Intervention Study to Reduce the Risk of Islet Autoimmunity in Children at Increased Risk for Type 1 Diabetes. Diabetes Care 2011, 34, 1301–1305. [Google Scholar] [CrossRef]
- Chase, H.P.; Lescheck, E.; Rafkin-Mervis, L.; Krause-Steinrauf, H.; Chritton, S.; Asare, S.M.; Adams, S.; Skyler, J.S.; Clare-Salzler, M. Nutritional Intervention to Prevent (NIP) Type 1 Diabetes A Pilot Trial. ICAN Infant Child Adolesc. Nutr. 2009, 1, 98–107. [Google Scholar] [CrossRef][Green Version]
- Simpson, M.; Brady, H.; Yin, X.; Seifert, J.; Barriga, K.; Hoffman, M.; Bugawan, T.; Baron, A.E.; Sokol, R.J.; Eisenbarth, G.; et al. No association of vitamin D intake or 25-hydroxyvitamin D levels in childhood with risk of islet autoimmunity and type 1 diabetes: The Diabetes Autoimmunity Study in the Young (DAISY). Diabetologia 2011, 54, 2779–2788. [Google Scholar] [CrossRef]
- Bonifacio, E.; Ziegler, A.G.; Klingensmith, G.; Schober, E.; Bingley, P.J.; Rottenkolber, M.; Theil, A.; Eugster, A.; Puff, R.; Peplow, C.; et al. Effects of High-Dose Oral Insulin on Immune Responses in Children at High Risk for Type 1 Diabetes. JAMA 2015, 313, 1541–1549. [Google Scholar] [CrossRef]
- Ziegler, A.-G.; Achenbach, P.; Berner, R.; Casteels, K.; Danne, T.; Gündert, M.; Hasford, J.; Hoffmann, V.S.; Kordonouri, O.; Lange, K.; et al. Oral insulin therapy for primary prevention of type 1 diabetes in infants with high genetic risk: The GPPAD-POInT (global platform for the prevention of autoimmune diabetes primary oral insulin trial) study protocol. BMJ Open 2019, 9, e028578. [Google Scholar] [CrossRef]
- Kroger, C.J.; Clark, M.; Ke, Q.; Tisch, R.M. Therapies to Suppress β Cell Autoimmunity in Type 1 Diabetes. Front. Immunol. 2018, 9, 1891. [Google Scholar] [CrossRef]
- Babad, J.; Geliebter, A.; DiLorenzo, T.P. T-cell autoantigens in the non-obese diabetic mouse model of autoimmune diabetes. Immunology 2010, 131, 459–465. [Google Scholar] [CrossRef] [PubMed]
- Gundersen E Is Diabetes of Infectious Origin J Infect Dis 41197 202 1927 15 | Course Hero. Available online: https://www.coursehero.com/file/p50tinl/Gundersen-E-Is-diabetes-of-infectious-origin-J-Infect-Dis-41197-202-1927-15/ (accessed on 11 August 2020).
- Smatti, M.K.; Cyprian, F.S.; Nasrallah, G.K.; Al Thani, A.A.; Almishal, R.O.; Yassine, H.M. Viruses and Autoimmunity: A Review on the Potential Interaction and Molecular Mechanisms. Viruses 2019, 11, 762. [Google Scholar] [CrossRef] [PubMed]
- Oikarinen, M.; Tauriainen, S.; Oikarinen, S.; Honkanen, T.; Collin, P.; Rantala, I.; Mäki, M.; Kaukinen, K.; Hyöty, H. Type 1 Diabetes Is Associated With Enterovirus Infection in Gut Mucosa. Diabetes 2012, 61, 687–691. [Google Scholar] [CrossRef] [PubMed]
- Filippi, C.M.; Von Herrath, M.G. Viral Trigger for Type 1 Diabetes: Pros and Cons. Diabetes 2008, 57, 2863–2871. [Google Scholar] [CrossRef]
- Sarmiento, L.; Cubas-Dueñas, I.; Cabrera-Rode, E. Evidence of association between type 1 diabetes and exposure to enterovirus in Cuban children and adolescents. MEDICC Rev. 2013, 15, 29–32. [Google Scholar] [CrossRef]
- Stone, V.M.; Hankaniemi, M.M.; Laitinen, O.H.; Sioofy-Khojine, A.; Lin, A.; Lozano, I.M.D.; Mazur, M.A.; Marjomäki, V.; Loré, K.; Hyöty, H.; et al. A hexavalent Coxsackievirus B vaccine is highly immunogenic and has a strong protective capacity in mice and nonhuman primates. Sci. Adv. 2020, 6, eaaz2433. [Google Scholar] [CrossRef]
- Lampeter, E.F.; Klinghammer, A.; Scherbaum, W.A.; Heinze, E.; Haastert, B.; Giani, G.; Kolb, H. The Deutsche Nicotinamide Intervention Study: An attempt to prevent type 1 diabetes. DENIS Group. Diabetes 1998, 47, 980–984. [Google Scholar] [CrossRef]
- Gale, E.A. European Nicotinamide Diabetes Intervention Trial (ENDIT): A randomised controlled trial of intervention before the onset of type 1 diabetes. Lancet 2004, 363, 925–931. [Google Scholar] [CrossRef]
- Skyler, J.S.; Brown, D.; Chase, H.P.; Collier, E.; Cowie, C.; Eisenbarth, G.S.; Fradkin, J.; Grave, G.; Greenbaum, C.; Jackson, R.A.; et al. Effects of Insulin in Relatives of Patients with Type 1 Diabetes Mellitus. N. Engl. J. Med. 2002, 346, 1685–1691. [Google Scholar] [CrossRef]
- American Diabetes Association. Effects of Oral Insulin in Relatives of Patients With Type 1 Diabetes: The Diabetes Prevention Trial-Type 1. Diabetes Care 2005, 28, 1068–1076. [Google Scholar] [CrossRef]
- Writing Committee for the Type 1 Diabetes TrialNet Oral Insulin Study Group; Krischer, J.P.; Schatz, D.A.; Bundy, B.; Skyler, J.S.; Greenbaum, C.J. Effect of Oral Insulin on Prevention of Diabetes in Relatives of Patients With Type 1 Diabetes: A Randomized Clinical Trial. JAMA 2017, 318, 1891–1902. [Google Scholar] [CrossRef] [PubMed]
- Harrison, L.C.; Honeyman, M.C.; Steele, C.E.; Stone, N.L.; Sarugeri, E.; Bonifacio, E.; Couper, J.J.; Colman, P.G. Pancreatic-Cell Function and Immune Responses to Insulin After Administration of Intranasal Insulin to Humans At Risk for Type 1 Diabetes. Diabetes Care 2004, 27, 2348–2355. [Google Scholar] [CrossRef] [PubMed]
- Trial of Intranasal Insulin in Children and Young Adults at Risk of Type 1 Diabetes—Full Text View—ClinicalTrials.gov. Available online: https://clinicaltrials.gov/ct2/show/NCT00336674 (accessed on 13 July 2020).
- Kick, K.; Assfalg, R.; Aydin, S.; Pozza, S.B.D.; Böcker, D.; Braig, S.; Bunk, M.; Dunstheimer, D.; Durmashkina, A.; Ermer, U.; et al. Recruiting young pre-symptomatic children for a clinical trial in type 1 diabetes: Insights from the Fr1da insulin intervention study. Contemp. Clin. Trials Commun. 2018, 11, 170–173. [Google Scholar] [CrossRef] [PubMed]
- Näntö-Salonen, K.; Kupila, A.; Simell, S.; Siljander, H.; Salonsaari, T.; Hekkala, A.; Korhonen, S.; Erkkola, R.; Sipilä, J.I.; Haavisto, L.; et al. Nasal insulin to prevent type 1 diabetes in children with HLA genotypes and autoantibodies conferring increased risk of disease: A double-blind, randomised controlled trial. Lancet 2008, 372, 1746–1755. [Google Scholar] [CrossRef]
- Elding Larsson, H.; Lundgren, M.; Jonsdottir, B.; Cuthbertson, D.; Krischer, J. Safety and efficacy of autoantigen-specific therapy with 2 doses of alum-formulated glutamate decarboxylase in children with multiple islet autoantibodies and risk for type 1 diabetes: A randomized clinical trial. Pediatr. Diabetes 2018, 19, 410–419. [Google Scholar] [CrossRef]
- Prevention Trial: Immune-tolerance With Alum-GAD (Diamyd) and Vitamin D3 to Children With Multiple Islet Autoantibodies—Full Text View—ClinicalTrials.gov. Available online: https://clinicaltrials.gov/ct2/show/NCT02387164 (accessed on 13 July 2020).
- Herold, K.C.; Bundy, B.N.; Long, S.A.; Bluestone, J.A.; DiMeglio, L.A.; Dufort, M.J.; Gitelman, S.E.; Gottlieb, P.A.; Krischer, J.P.; Linsley, P.S.; et al. An Anti-CD3 Antibody, Teplizumab, in Relatives at Risk for Type 1 Diabetes. N. Engl. J. Med. 2019, 381, 603–613. [Google Scholar] [CrossRef]
- CTLA4-Ig (Abatacept) for Prevention of Abnormal Glucose Tolerance and Diabetes in Relatives At -Risk for Type 1—Full Text View—ClinicalTrials.gov. Available online: https://www.clinicaltrials.gov/ct2/show/NCT01773707 (accessed on 13 July 2020).
- Hydroxychloroquine in Individuals At-risk for Type 1 Diabetes Mellitus—Full Text View—ClinicalTrials.gov. Available online: https://www.clinicaltrials.gov/ct2/show/NCT03428945 (accessed on 13 July 2020).
- A Study to Evaluate SIMPONI (Golimumab) Therapy in Children, Adolescents and Young Adults With Pre-Symptomatic Type 1 Diabetes—Full Text View—ClinicalTrials.gov. Available online: https://clinicaltrials.gov/ct2/show/NCT03298542 (accessed on 13 July 2020).
- Yamada, K.; Nonaka, K.; Hanafusa, T.; Miyazaki, A.; Toyoshima, H.; Tarui, S. Preventive and Therapeutic Effects of Large-Dose Nicotinamide Injections on Diabetes Associated with Insulitis: An Observation in Nonobese Diabetic (NOD) Mice. Diabetes 1982, 31, 749–753. [Google Scholar] [CrossRef]
- Vehik, K.; Cuthbertson, D.; Ruhlig, H.; Schatz, D.A.; Peakman, M.; Krischer, J.P. Long-Term Outcome of Individuals Treated With Oral Insulin. Diabetes Care 2011, 34, 1585–1590. [Google Scholar] [CrossRef]
- Ziegler, A.G.; Kick, K.; Bonifacio, E.; Haupt, F.; Hippich, M.; Dunstheimer, D.; Lang, M.; Laub, O.; Warncke, K.; Lange, K.; et al. Yield of a Public Health Screening of Children for Islet Autoantibodies in Bavaria, Germany. JAMA 2020, 323, 339–351. [Google Scholar] [CrossRef]
- Kanaani, J.; Cianciaruso, C.; Phelps, E.; Pasquier, M.; Brioudes, E.; Billestrup, N.; Baekkeskov, S. Compartmentalization of GABA Synthesis by GAD67 Differs between Pancreatic Beta Cells and Neurons. PLoS ONE 2015, 10, e0117130. [Google Scholar] [CrossRef]
- Tisch, R.; Yang, X.D.; Singer, S.M.; Liblau, R.S.; Fugger, L.; McDevitt, H.O.; Cooke, A.; Mandel, T.E. Immune response to glutamic acid decarboxylase correlates with insulitis in non-obese diabetic mice. J. Endocrinol. Investig. 1994, 17, 586–593. [Google Scholar] [CrossRef] [PubMed]
- Arif, S.; Gomez-Tourino, I.; Kamra, Y.; Pujol-Autonell, I.; Hanton, E.; Tree, T.; Melandri, D.; Hull, C.; Wherrett, D.K.; Beam, C.; et al. GAD-alum immunotherapy in type 1 diabetes expands bifunctional Th1/Th2 autoreactive CD4 T cells. Diabetologia 2020, 63, 1186–1198. [Google Scholar] [CrossRef] [PubMed]
- Rabinovitch, A.; Suarez-Pinzon, W.L. Role of cytokines in the pathogenesis of autoimmune diabetes mellitus. Rev. Endocr. Metab. Disord. 2003, 4, 291–299. [Google Scholar] [CrossRef]
- Kany, S.; Vollrath, J.T.; Relja, B. Cytokines in Inflammatory Disease. Int. J. Mol. Sci. 2019, 20, 6008. [Google Scholar] [CrossRef] [PubMed]
- Silverstein, J.; MacLaren, N.; Riley, W.; Spillar, R.; Radjenovic, D.; Johnson, S. Immunosuppression with Azathioprine and Prednisone in Recent-Onset Insulin-Dependent Diabetes Mellitus. N. Engl. J. Med. 1988, 319, 599–604. [Google Scholar] [CrossRef]
- Bougnères, P.F.; Landais, P.; Boisson, C.; Carel, J.C.; Frament, N.; Boitard, C.; Chaussain, J.L.; Bach, J.F. Limited Duration of Remission of Insulin Dependency in Children With Recent Overt Type I Diabetes Treated With Low-Dose Cyclosporin. Diabetes 1990, 39, 1264–1272. [Google Scholar] [CrossRef]
- Keymeulen, B.; Vandemeulebroucke, E.; Ziegler, A.G.; Mathieu, C.; Kaufman, L.; Hale, G.; Gorus, F.; Goldman, M.; Walter, M.; Candon, S.; et al. Insulin Needs after CD3-Antibody Therapy in New-Onset Type 1 Diabetes. N. Engl. J. Med. 2005, 352, 2598–2608. [Google Scholar] [CrossRef]
- Keymeulen, B.; Walter, M.; Mathieu, C.; Kaufman, L.; Gorus, F.; Hilbrands, R.; Vandemeulebroucke, E.; Van De Velde, U.; Crenier, L.; De Block, C.; et al. Four-year metabolic outcome of a randomised controlled CD3-antibody trial in recent-onset type 1 diabetic patients depends on their age and baseline residual beta cell mass. Diabetologia 2010, 53, 614–623. [Google Scholar] [CrossRef]
- Trial of Otelixizumab for Adolescents and Adults with Newly Diagnosed Type 1 Diabetes Mellitus (Autoimmune): DEFEND-2—Full Text View—ClinicalTrials.gov. Available online: https://clinicaltrials.gov/ct2/show/NCT01123083 (accessed on 13 July 2020).
- Sherry, N.; Hagopian, W.; Ludvigsson, J.; Jain, S.M.; Wahlen, J.; Ferry, R.J.; Bode, B.; Aronoff, S.; Holland, C.; Carlin, D.; et al. Teplizumab for treatment of type 1 diabetes (Protégé study): 1-year results from a randomised, placebo-controlled trial. Lancet 2011, 378, 487–497. [Google Scholar] [CrossRef]
- Herold, K.C.; Gitelman, S.E.; Ehlers, M.R.; Gottlieb, P.A.; Greenbaum, C.J.; Hagopian, W.; Boyle, K.D.; Keyes-Elstein, L.; Aggarwal, S.; Phippard, D.; et al. Teplizumab (Anti-CD3 mAb) Treatment Preserves C-Peptide Responses in Patients With New-Onset Type 1 Diabetes in a Randomized Controlled Trial. Diabetes 2013, 62, 3766–3774. [Google Scholar] [CrossRef]
- Orban, T.; Bundy, B.; Becker, D.J.; DiMeglio, L.A.; Gitelman, S.E.; Goland, R.; Gottlieb, P.A.; Greenbaum, C.J.; Marks, J.B.; Monzavi, R.; et al. Co-stimulation modulation with abatacept in patients with recent-onset type 1 diabetes: A randomised, double-blind, placebo-controlled trial. Lancet 2011, 378, 412–419. [Google Scholar] [CrossRef]
- Rigby, M.R.; Harris, K.M.; Pinckney, A.; DiMeglio, L.A.; Rendell, M.S.; Felner, E.I.; Dostou, J.M.; Gitelman, S.E.; Griffin, K.J.; Tsalikian, E.; et al. Alefacept provides sustained clinical and immunological effects in new-onset type 1 diabetes patients. J. Clin. Investig. 2015, 125, 3285–3296. [Google Scholar] [CrossRef] [PubMed]
- Pescovitz, M.D.; Greenbaum, C.J.; Krause-Steinrauf, H.; Becker, D.J.; Gitelman, S.E.; Goland, R.; Gottlieb, P.A.; Marks, J.B.; McGee, P.F.; Moran, A.M.; et al. Rituximab, B-Lymphocyte Depletion, and Preservation of Beta-Cell Function. N. Engl. J. Med. 2009, 361, 2143–2152. [Google Scholar] [CrossRef] [PubMed]
- Gitelman, S.E.; Gottlieb, P.A.; Rigby, M.R.; Felner, E.I.; Willi, S.M.; Fisher, L.K.; Moran, A.; Gottschalk, M.; Moore, W.V.; Pinckney, A.; et al. Antithymocyte globulin treatment for patients with recent-onset type 1 diabetes: 12-month results of a randomised, placebo-controlled, phase 2 trial. Lancet Diabetes Endocrinol. 2013, 1, 306–316. [Google Scholar] [CrossRef]
- Gitelman, S.E.; Gottlieb, P.A.; Felner, E.I.; Willi, S.M.; Fisher, L.K.; Moran, A.; Gottschalk, M.; Moore, W.V.; Pinckney, A.; Keyes-Elstein, L.; et al. Antithymocyte globulin therapy for patients with recent-onset type 1 diabetes: 2 year results of a randomised trial. Diabetologia 2016, 59, 1153–1161. [Google Scholar] [CrossRef]
- Haller, M.J.; Gitelman, S.E.; Gottlieb, P.A.; Michels, A.W.; Perry, D.J.; Schultz, A.R.; Hulme, M.A.; Shuster, J.J.; Zou, B.; Wasserfall, C.H.; et al. Antithymocyte Globulin Plus G-CSF Combination Therapy Leads to Sustained Immunomodulatory and Metabolic Effects in a Subset of Responders With Established Type 1 Diabetes. Diabetes 2016, 65, 3765–3775. [Google Scholar] [CrossRef]
- Haller, M.J.; Long, S.A.; Blanchfield, J.L.; Schatz, D.; Skyler, J.S.; Krischer, J.P.; Bundy, B.N.; Geyer, S.M.; Warnock, M.V.; Miller, J.L.; et al. Low-Dose Anti-Thymocyte Globulin Preserves C-Peptide, Reduces HbA1c, and Increases Regulatory to Conventional T-Cell Ratios in New-Onset Type 1 Diabetes: Two-Year Clinical Trial Data. Diabetes 2019, 68, 1267–1276. [Google Scholar] [CrossRef]
- Ludvigsson, J.; Faresjö, M.; Hjorth, M.; Axelsson, S.; Chéramy, M.; Pihl, M.; Vaarala, O.; Forsander, G.; Ivarsson, S.; Johansson, C.; et al. GAD Treatment and Insulin Secretion in Recent-Onset Type 1 Diabetes. N. Engl. J. Med. 2008, 359, 1909–1920. [Google Scholar] [CrossRef]
- Ludvigsson, J.; Krisky, D.; Casas, R.; Battelino, T.; Castaño, L.; Greening, J.; Kordonouri, O.; Otonkoski, T.; Pozzilli, P.; Robert, J.J.; et al. GAD65 Antigen Therapy in Recently Diagnosed Type 1 Diabetes Mellitus. N. Engl. J. Med. 2012, 366, 433–442. [Google Scholar] [CrossRef]
- Marek-Trzonkowska, N.; Myśliwiec, M.; Dobyszuk, A.; Grabowska, M.; Derkowska, I.; Juścińska, J.; Owczuk, R.; Szadkowska, A.; Witkowski, P.; Młynarski, W.; et al. Therapy of type 1 diabetes with CD4 + CD25highCD127-regulatory T cells prolongs survival of pancreatic islets—Results of one year follow-up. Clin. Immunol. 2014, 153, 23–30. [Google Scholar] [CrossRef]
- Bluestone, J.A.; Buckner, J.H.; Fitch, M.; Gitelman, S.E.; Gupta, S.; Hellerstein, M.K.; Herold, K.C.; Lares, A.; Lee, M.R.; Li, K.; et al. Type 1 diabetes immunotherapy using polyclonal regulatory T cells. Sci. Transl. Med. 2015, 7, 315ra189. [Google Scholar] [CrossRef]
- Hartemann, A.; Bensimon, G.; Payan, C.A.; Jacqueminet, S.; Bourron, O.; Nicolas, N.; Fonfrede, M.; Rosenzwajg, M.; Bernard, C.; Klatzmann, D. Low-dose interleukin 2 in patients with type 1 diabetes: A phase 1/2 randomised, double-blind, placebo-controlled trial. Lancet Diabetes Endocrinol. 2013, 1, 295–305. [Google Scholar] [CrossRef]
- Marcovecchio, M.L.; Wicker, L.S.; Dunger, D.B.; Dutton, S.J.; Kopijasz, S.; Scudder, C.; Todd, J.A.; Johnson, P.R.V. Interleukin-2 Therapy of Autoimmunity in Diabetes (ITAD): A phase 2, multicentre, double-blind, randomized, placebo-controlled trial. Wellcome Open Res. 2020, 5, 49. [Google Scholar] [CrossRef] [PubMed]
- Gottlieb, P.; Quinlan, S.; Krause-Steinrauf, H.; Greenbaum, C.J.; Wilson, D.M.; Rodriguez, H.; Schatz, D.A.; Moran, A.M.; Lachin, J.M.; Skyler, J.S. Failure to Preserve β-Cell Function With Mycophenolate Mofetil and Daclizumab Combined Therapy in Patients With New-Onset Type 1 Diabetes. Diabetes Care 2010, 33, 826–832. [Google Scholar] [CrossRef] [PubMed]
- Krogvold, L.; Edwin, B.; Buanes, T.A.; Frisk, G.; Skog, O.; Anagandula, M.; Korsgren, O.; Undlien, D.; Eike, M.C.; Richardson, S.J.; et al. Detection of a Low-Grade Enteroviral Infection in the Islets of Langerhans of Living Patients Newly Diagnosed With Type 1 Diabetes. Diabetes 2014, 64, 1682–1687. [Google Scholar] [CrossRef]
- Griffin, K.J.; Thompson, P.A.; Gottschalk, M.; Kyllo, J.H.; Rabinovitch, A. Combination therapy with sitagliptin and lansoprazole in patients with recent-onset type 1 diabetes (REPAIR-T1D): 12-month results of a multicentre, randomised, placebo-controlled, phase 2 trial. Lancet Diabetes Endocrinol. 2014, 2, 710–718. [Google Scholar] [CrossRef]
- Snaith, J.R.; Samocha-Bonet, D.; Holmes-Walker, D.J.; Greenfield, J.R. MON-LB113 Insulin Resistance in Type 1 Diabetes Managed With Metformin (INTIMET): Rationale and Study Design of a Randomised Placebo-Controlled Trial. J. Endocr. Soc. 2020, 4. [Google Scholar] [CrossRef]
- Christie, M.; Mølvig, J.; Hawkes, C.J.; Carstensen, B.; Mandrup-Poulsen, T. IA-2 antibody-negative status predicts remission and recovery of C-peptide levels in type 1 diabetic patients treated with cyclosporin. Diabetes Care 2002, 25, 1192–1197. [Google Scholar] [CrossRef][Green Version]
- Hagopian, W.; Ferry, R.J.; Sherry, N.; Carlin, D.; Bonvini, E.; Johnson, S.; Stein, K.E.; Koenig, S.; Daifotis, A.G.; Herold, K.C.; et al. Teplizumab Preserves C-Peptide in Recent-Onset Type 1 Diabetes. Diabetes 2013, 62, 3901–3908. [Google Scholar] [CrossRef]
- Pescovitz, M.D.; Greenbaum, C.J.; Bundy, B.; Becker, D.J.; Gitelman, S.E.; Goland, R.; Gottlieb, P.A.; Marks, J.B.; Moran, A.; Raskin, P.; et al. B-Lymphocyte Depletion With Rituximab and β-Cell Function: Two-Year Results. Diabetes Care 2014, 37, 453–459. [Google Scholar] [CrossRef]
- Wherrett, D.K.; Bundy, B.; Becker, D.J.; DiMeglio, L.A.; Gitelman, S.E.; Goland, R.; Gottlieb, P.A.; Greenbaum, C.J.; Herold, K.C.; Marks, J.B.; et al. Antigen-based therapy with glutamic acid decarboxylase (GAD) vaccine in patients with recent-onset type 1 diabetes: A randomised double-blind trial. Lancet 2011, 378, 319–327. [Google Scholar] [CrossRef]
- Ludvigsson, J.; Wahlberg, J.; Casas, R. Intralymphatic Injection of Autoantigen in Type 1 Diabetes. N. Engl. J. Med. 2017, 376, 697–699. [Google Scholar] [CrossRef] [PubMed]
- Tavira, B.; Barcenilla, H.; Wahlberg, J.; Achenbach, P.; Ludvigsson, J.; Casas, R. Intralymphatic Glutamic Acid Decarboxylase-Alum Administration Induced Th2-Like-Specific Immunomodulation in Responder Patients: A Pilot Clinical Trial in Type 1 Diabetes. J. Diabetes Res. 2018, 2018, 9391845. [Google Scholar] [CrossRef] [PubMed]
- Long, S.A.; Buckner, J.H. CD4 + FOXP3 + T regulatory cells in human autoimmunity: More than a numbers game. J. Immunol. 2011, 187, 2061–2066. [Google Scholar] [CrossRef]
- Tang, Q.; Adams, J.Y.; Penaranda, C.; Melli, K.; Piaggio, E.; Sgouroudis, E.; Piccirillo, C.A.; Salomon, B.; Bluestone, J.A. Central Role of Defective Interleukin-2 Production in the Triggering of Islet Autoimmune Destruction. Immunity 2008, 28, 687–697. [Google Scholar] [CrossRef] [PubMed]
- Grinberg-Bleyer, Y.; Baeyens, A.; You, S.; Elhage, R.; Fourcade, G.; Gregoire, S.; Cagnard, N.; Carpentier, W.; Tang, Q.; Bluestone, J.; et al. IL-2 reverses established type 1 diabetes in NOD mice by a local effect on pancreatic regulatory T cells. J. Exp. Med. 2010, 207, 1871–1878. [Google Scholar] [CrossRef] [PubMed]
- Clinical Trials Register—Search for 2015-003350-41. Available online: https://www.clinicaltrialsregister.eu/ctr-search/search?query=2015-003350-41 (accessed on 11 August 2020).
- Takeda, Y.; Fujita, Y.; Honjo, J.; Yanagimachi, T.; Sakagami, H.; Takiyama, Y.; Makino, Y.; Abiko, A.; Kieffer, T.J.; Haneda, M. Reduction of both beta cell death and alpha cell proliferation by dipeptidyl peptidase-4 inhibition in a streptozotocin-induced model of diabetes in mice. Diabetologia 2011, 55, 404–412. [Google Scholar] [CrossRef]
- D’Alessio, D.A.; Denney, A.M.; Hermiller, L.M.; Prigeon, R.L.; Martin, J.M.; Tharp, W.G.; Saylan, M.L.; He, Y.; Dunning, B.E.; Foley, J.E.; et al. Treatment with the dipeptidyl peptidase-4 inhibitor vildagliptin improves fasting islet-cell function in subjects with type 2 diabetes. J. Clin. Endocrinol. Metab. 2008, 94, 81–88. [Google Scholar] [CrossRef] [PubMed]
- Foley, J.E.; Bunck, M.C.; Möller-Goede, D.L.; Poelma, M.; Nijpels, G.; Eekhoff, E.; Schweizer, A.; Heine, R.J.; Diamant, M. Beta cell function following 1 year vildagliptin or placebo treatment and after 12 week washout in drug-naive patients with type 2 diabetes and mild hyperglycaemia: A randomised controlled trial. Diabetologia 2011, 54, 1985–1991. [Google Scholar] [CrossRef] [PubMed]
- Pinheiro, M.M.; Stoppa, C.L.; Valduga, C.J.; Okuyama, C.E.; Gorjão, R.; Pereira, R.M.S.; Diniz, S.N. Sitagliptin inhibit human lymphocytes proliferation and Th1/Th17 differentiation in vitro. Eur. J. Pharm. Sci. 2017, 100, 17–24. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Yang, L.; Xiang, Y.; Liu, L.; Huang, G.; Long, Z.; Li, X.; Leslie, R.D.; Wang, X.; Zhou, Z. Dipeptidyl Peptidase 4 Inhibitor Sitagliptin Maintains β-Cell Function in Patients With Recent-Onset Latent Autoimmune Diabetes in Adults: One Year Prospective Study. J. Clin. Endocrinol. Metab. 2014, 99, E876–E880. [Google Scholar] [CrossRef]
- Wang, X.; Yang, L.; Cheng, Y.; Zheng, P.; Hu, J.; Huang, G.; Zhou, Z. Altered T-cell subsets and transcription factors in latent autoimmune diabetes in adults taking sitagliptin, a dipeptidyl peptidase-4 inhibitor: A 1-year open-label randomized controlled trial. J. Diabetes Investig. 2018, 10, 375–382. [Google Scholar] [CrossRef] [PubMed]
- The Effect of Saxagliptin on Glucose Fluctuation and Immune Regulation in Patients with Type 1 Diabetes—Full Text View—ClinicalTrials.gov. Available online: https://clinicaltrials.gov/ct2/show/NCT02307695 (accessed on 12 August 2020).
- Protective Effects of Saxagliptin (And Vitamin D3) on β Cell Function in Adult-onset Latent Autoimmune Diabetes—Full Text View—ClinicalTrials.gov. Available online: https://clinicaltrials.gov/ct2/show/NCT02407899 (accessed on 12 August 2020).
- Treatment of Latent Autoimmune Diabetes of the Adult—Full Text View—ClinicalTrials.gov. Available online: https://clinicaltrials.gov/ct2/show/NCT01140438 (accessed on 12 August 2020).
- Kibirige, M.; Metcalf, B.; Renuka, R.; Wilkin, T.J. Testing the accelerator hypothesis: The relationship between body mass and age at diagnosis of type 1 diabetes. Diabetes Care 2003, 26, 2865–2870. [Google Scholar] [CrossRef] [PubMed]
- Knip, M.; Simell, O. Environmental Triggers of Type 1 Diabetes. Cold Spring Harb. Perspect. Med. 2012, 2, a007690. [Google Scholar] [CrossRef] [PubMed]
- Ziegler, A.G.; Babydiab-Babydiet Study Group; Bonifacio, E. Age-related islet autoantibody incidence in offspring of patients with type 1 diabetes. Diabetologia 2012, 55, 1937–1943. [Google Scholar] [CrossRef] [PubMed]
- Ziegler, A.G.; Rewers, M.; Simell, O.; Simell, T.; Lempainen, J.; Steck, A.; Winkler, C.; Ilonen, J.; Veijola, R.; Knip, M.; et al. Seroconversion to Multiple Islet Autoantibodies and Risk of Progression to Diabetes in Children. JAMA 2013, 309, 2473. [Google Scholar] [CrossRef]
| DRB1 | DQB1 | DQA1 | Ethnic Backgrounds |
|---|---|---|---|
| High-risk haplotypes | |||
| 03:01 | 02:01 | 05:01 | Caucasians, Koreans |
| 04:01 | 03:02 | 03:01 | Caucasians |
| 04:02 | 04:02 | 04:01 | Caucasians |
| 04:05 | 03:02 | 03:01 | Caucasians |
| 04:01 | 03:03 | 04:05 | Japanese, Koreans |
| 03:02 | 03:01 | 08:02 | Japanese |
| 03:03 | 03:00 | 09:01 | Japanese, Koreans |
| 06:04 | 01:02 | 13:02 | Japanese |
| Moderate risk haplotypes | |||
| 01 | 05:01 | 01:01 | Caucasians |
| 08:01 | 04:02 | 04:01 | Caucasians |
| 09:01 | 03:03 | 03:01 | Caucasians |
| Study [Ref.] | Inclusion Criteria | Age | Intervention | Follow-Up/Primary Endpoint | Outcome | Status |
|---|---|---|---|---|---|---|
| TRIGR [14] | Newborns with risk-associated HLA genotypes | 0–7 days | Hydrolyzed infant formula | 10 years/T1D | Failed to delay or prevent the development of T1D | Completed |
| FINDIA [15] | Newborns with high-risk HLA | Infants | Insulin-free whey-based formula | 2 years/islet autoantibodies, T1D | Reduced the incidence of autoantibodies by age 3 years | Completed |
| BABYDIET [16] | First degree relatives with high-risk HLA | <3 months | Gluten-free diet | 3 years/islet autoantibodies | No evidence of reducing the risk for autoantibodies development | Completed |
| NIP [17] | Pregnant mothers and newborns with genetic risk for T1D | >24 weeks gestation/ newborns | Docosahexaenoic acid (DHA) | 2 years/20% higher plasma levels of DHA | No effect on autoimmunity | Completed |
| DAISY [18] | First-degree relatives of patients with T1D and newborns with genetic risk | <8 years/ newborns | Vitamin D | 2 years/islet autoantibodies, T1D | Failed to reduce the risk of islet autoantibodies/T1D development | Completed |
| Pre-POINT [19] | Individuals with familial risk | 1.5–7 years | Oral insulin | 3–18 months/islet autoantibodies | A high dose of daily oral insulin is safe and appears to change the immune response to insulin. | Completed |
| GPPAD-POInT [20] | Children with genetic risk for T1D | 4–7 months | Oral insulin | 7.5 years/islet autoantibodies, T1D | Not yet reported | Ongoing |
| Study | Inclusion Criteria | Age | Intervention | Follow-Up/Primary Endpoint | Outcome | Status |
|---|---|---|---|---|---|---|
| DENIS [29] | First-degree relatives of patients with T1D | 3–12 year | Nicotinamide | 3.8 years/T1D | Failed to delay of T1D development | Completed |
| ENDIT [30] | Family members with ICA positive but OGTT negative | <40 year | Nicotinamide | 5 years/T1D | No effect on halting or preventing T1D | Completed |
| DPT-1 (first arm) [31] | ICA-positive T1D siblings with decreased first-phase insulin secretion | 3–45 year | Parenteral insulin | 5 years/T1D | The incidence of T1D in the intervention group was virtually the same as in the observation group | Completed |
| DPT-1 (second arm) [32] | ICA-positive T1D siblings with normal first-phase insulin secretion | 3–45 year | Oral insulin | 5 years/T1D | Failed to delay or prevent T1D development | Completed |
| TrialNet Oral Insulin [33] | Relatives with at least 2 autoantibodies | 1–45 year | Oral insulin | 7–8 years/T1D | No effect on reducing the risk of T1D onset | Completed |
| INIT [34] | Individuals with one or more T1D-related autoantibodies | 4–33 year | Nasal insulin | 1 year/T1D | Improved immune tolerance, but no significant effects on beta-cell function | Completed |
| INIT-II [35] | Relatives with at least 2 autoantibodies, HLA | 4–30 year | Nasal insulin | 5 years/T1D | Intranasal insulin-induced immune response, but has no effect on the prevention of T1D | Active, not recruiting |
| Fr1da Insulin Intervention [36] | General population with at least two islet autoantibodies | 2–12 year | Oral insulin | 1–7 years/T1D | Not yet reported | Ongoing |
| DIPP [37] | General population with high-risk genotypes for T1D | Newborn | Nasal insulin | 10 years/T1D | Failed to find an effect of nasal insulin administration on T1D progression | Completed |
| DiAPREV-IT [38] | Individuals with multiple islet autoantibodies | 4–18 year | GAD-Alum | 5 years/T1D | Failed to halt the progression of the autoimmune process | Completed |
| DiAPREV-IT2 [39] | Individuals with multiple islet autoantibodies | 10–18 year | GAD-Alum and Vitamin D | 5 years/T1D | Not yet reported | Ongoing |
| TrialNet Teplizumab [40] | Relatives with at least 2 autoantibodies, with impaired OGTT | 8–45 year | Anti-CD3 (teplizumab) | 4–6 years/T1D | Teplizumab can delay T1D diagnosis a median of 2 years | Completed |
| TrialNet Abatacept [41] | Relatives with at least 2 autoantibodies | 6–45 year | Anti–CTLA-4 (abatacept) | 5–6 years/AGT, T1D | Not yet reported | Ongoing |
| TrialNet HCQ [42] | Individuals with multiple islet autoantibodies | 3–18 year | HCQ | 5–6 years/AGT, T1D | Not yet reported | Ongoing |
| SIMPONI [43] | Individuals with at least 2 autoantibodies, with impaired OGTT | 6–21 year | TNF-a (golimumab) | 5–6years/AEs | Not yet reported | Ongoing |
| Study | Time from Diagnosis/ Eligibility | Age | Intervention | Follow-Up/Primary End Point | Outcome | Status |
|---|---|---|---|---|---|---|
| Silverstein et al. [52] | <2 weeks | 4–32 year | Azathioprine and Prednisone | 2.5 years/MMTT C-peptide | Short-lived remission in the treatment group | Completed |
| Bougneres et al. [53] | Newly diagnosed | 7–15 year | Cyclosporine A | 2 years/glucagon stimulation test C-peptide | Cyclosporin A transiently maintained a residual insulin secretion | Completed |
| Keymeulen et al. [54,55] | <4 weeks/> C-peptide 0.2 pmol/mL | 12–39 year | Anti-CD3 (otelixizumab) | 48 months/glucagon stimulation test C-peptide | Anti-CD3 maintained a residual beta-cell function | Completed |
| DEFEND-1 and -2 [56] | ≤12 weeks/C-peptide 0.2–3.5 pmol/mL | 12–45 year | Anti-CD3 (otelixizumab) | 1 year/MMTT C-peptide | No preservation of beta-cell function | Completed |
| Protégé study [57] | ≤12 weeks/C-peptide detectable | 8–35 year | Anti-CD3 (teplizumab) | 2 years/insulin dose + HbA1c MMTT C-peptide | Improved C-peptide responses in the 14-day high dose subgroup | Completed |
| AbATE [58] | <8 weeks | 8–30 year | Anti-CD3 (teplizumab) | 2 years/MMTT C-peptide | Lower insulin requirement in the treatment group | Completed |
| Orban et al. [59] | <14 weeks/> C-peptide 0.2 pmol/mL | 6–36 year | CTLA4-Ig (abatacept) | 2 years/MMTT C-peptide | Abatacept slowed the decline of beta-cell function over two years. | Completed |
| TIDAL [60] | <14 weeks/> C-peptide 0.2 pmol/mL * | 12–35 year | Alafacept | 2 years/MMTT C-peptide | C-peptide in the treatment group was significantly higher compared to placebo | Completed |
| TrialNet Rituximab [61] | ≤12 weeks/C-peptide 0.2 pmol/mL * | 8–45 year | Anti-CD20 (rituximab) | 2 years/MMTT C-peptide | C-peptide levels were significantly higher in the rituximab versus the placebo group | Completed |
| START [62,63] | 6 weeks/C-peptide * 0.4 pmol/mL | 12–35 year | ATG | 2 years/MMTT C-peptide | Failed to show that ATG preserves beta-cell function in new-onset T1D | Completed |
| TrialNet ATG-GCSF [64,65] | <14 weeks/C-peptide 0.2 pmol/mL * | 12–45 year | ATG/GCSF or ATG alone | 2 year/MMTT C-peptide | Low-dose ATG preserved beta-cell function and improved insulin production | Completed |
| Ludvigsson et al. [66] | <12 weeks/C-peptide 0.1 pmol/mL, GAD autoantibody positive | 10–20 year | rhGAD65-alum | 15 months/MMTT C-peptide | No difference in C-peptide concentrations or insulin requirements | Completed |
| TrialNet GAD Study [67] | <12 weeks/C-peptide 0.2 pmol/mL, GAD autoantibody positive | 3–45 year | rhGAD65-alum | 2 years/MMTT C-peptide | No change in the course of loss of insulin secretion | Completed |
| Marek-Trzonkowska et al. [68] | <8 weeks/C-peptide 0.1 pmol/mL | 5–18 year | Infusion of ex vivo expanded Tregs | 2 years/MMTT C-peptide | An increase in Tregs number in peripheral blood and c-peptide levels | Completed |
| Bluestone et al. [69] | >3 weeks and <24 months/C-peptide 0.1 pmol/mL * | 18–45 year | Infusion of expanded polyclonal Tregs | 2.5 years/MMTT C-peptide | C-peptide levels persisted out to 2+ years after transfer in several individuals | Completed |
| Hartemann et al. [70] | ≤ 2 years | 18–50 year | Aldesleukin (IL-2) | Kinetic parameters of Treg proportions variation within CD4+ T cells in peripheral blood | IL-2 induced a dose-dependent increase in the proportion of Treg cells | Completed |
| ITAD [71] | <6 weeks/C-peptide 0.2 pmol/mL | 6–18 year | Aldesleukin (IL-2) | Differences in C-peptide over the 6 month-treatment periods between the active and placebo groups | Not yet available | Recruiting |
| Gottlieb et al. [72] | ≤12 weeks/C-peptide 0.2 pmol/mL * | 8–45 year | Mycophenolate mofetil alone or plus daclizumab | 2 years/MMTT C-peptide | No effect on residual beta-cell function | Completed |
| DiViDInt [73] | <3 weeks | 6–15 year | Pleconaril and ribavirin | 3 years/MMTT C-peptide | Not yet available | Ongoing |
| REPAIR-T1D [74] | <6 months | 11–36 year | Sitagliptin plus lansoprazole | 1 year/MMTT C-peptide | No effect on residual beta-cell function | Completed |
| DPP-IV LADA [75] | <3 years/diagnosed with LADA | 25–70 year | Sitagliptin | 2 years/MMTT C-peptide | Beneficial effect on beta-cell function | Completed |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Beik, P.; Ciesielska, M.; Kucza, M.; Kurczewska, A.; Kuźmińska, J.; Maćkowiak, B.; Niechciał, E. Prevention of Type 1 Diabetes: Past Experiences and Future Opportunities. J. Clin. Med. 2020, 9, 2805. https://doi.org/10.3390/jcm9092805
Beik P, Ciesielska M, Kucza M, Kurczewska A, Kuźmińska J, Maćkowiak B, Niechciał E. Prevention of Type 1 Diabetes: Past Experiences and Future Opportunities. Journal of Clinical Medicine. 2020; 9(9):2805. https://doi.org/10.3390/jcm9092805
Chicago/Turabian StyleBeik, Przemysław, Martyna Ciesielska, Maria Kucza, Alicja Kurczewska, Joanna Kuźmińska, Bartosz Maćkowiak, and Elżbieta Niechciał. 2020. "Prevention of Type 1 Diabetes: Past Experiences and Future Opportunities" Journal of Clinical Medicine 9, no. 9: 2805. https://doi.org/10.3390/jcm9092805
APA StyleBeik, P., Ciesielska, M., Kucza, M., Kurczewska, A., Kuźmińska, J., Maćkowiak, B., & Niechciał, E. (2020). Prevention of Type 1 Diabetes: Past Experiences and Future Opportunities. Journal of Clinical Medicine, 9(9), 2805. https://doi.org/10.3390/jcm9092805





