Respiratory Muscle Strengths and Their Association with Lean Mass and Handgrip Strengths in Older Institutionalized Individuals
Abstract
1. Introduction
2. Materials and Methods
2.1. Design and Study Population
2.2. Sociodemographic and Clinical Variables
2.3. Measurement of Respiratory Muscle Function
2.4. Measurement of Sarcopenia
2.5. Haemogram and Analytical Parameters
2.6. Statistical Analysis
3. Results
3.1. Sociodemographic and Clinical Parameters of the Study Sample
3.2. Evaluation of Skeletal Muscle Mass and Function
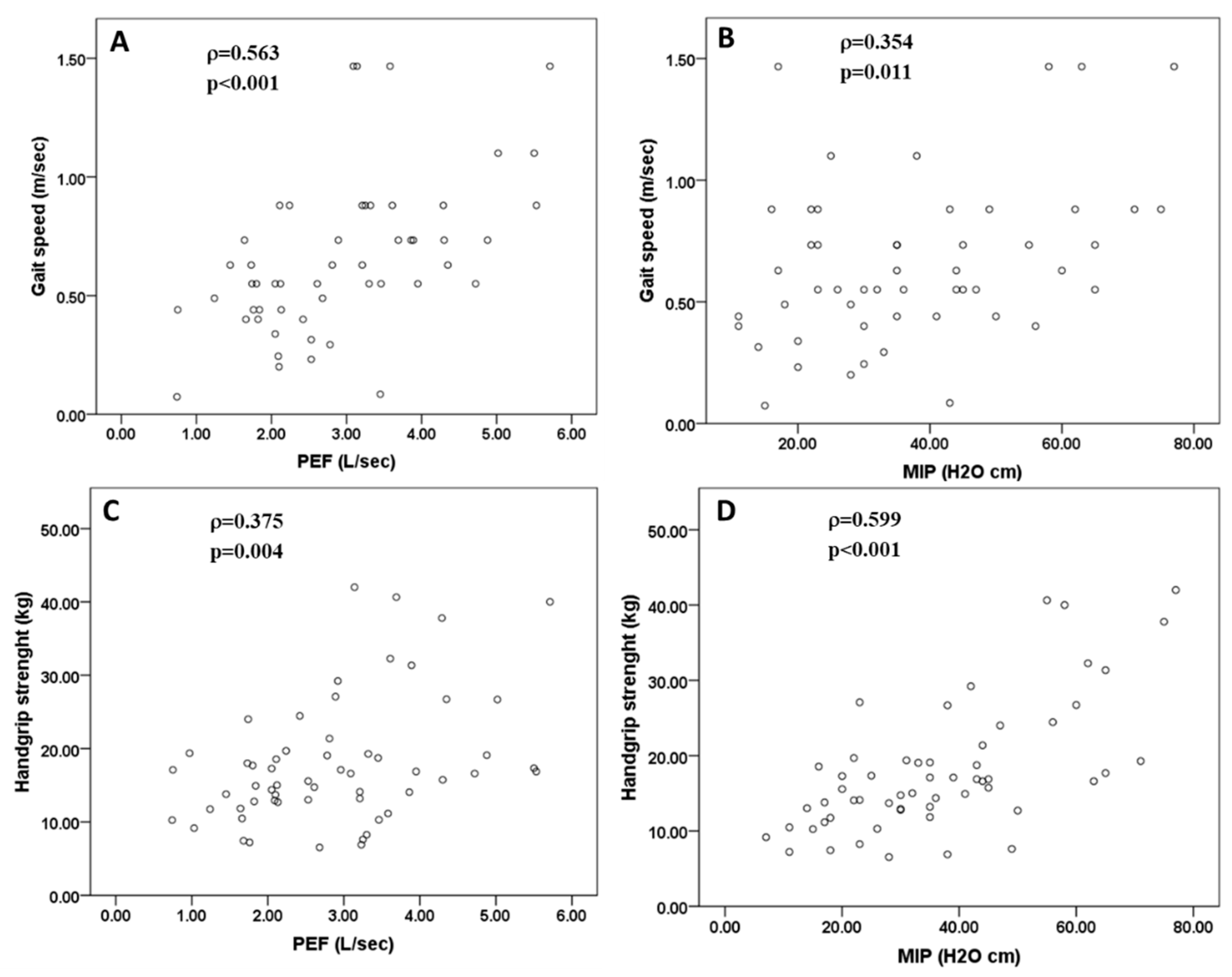
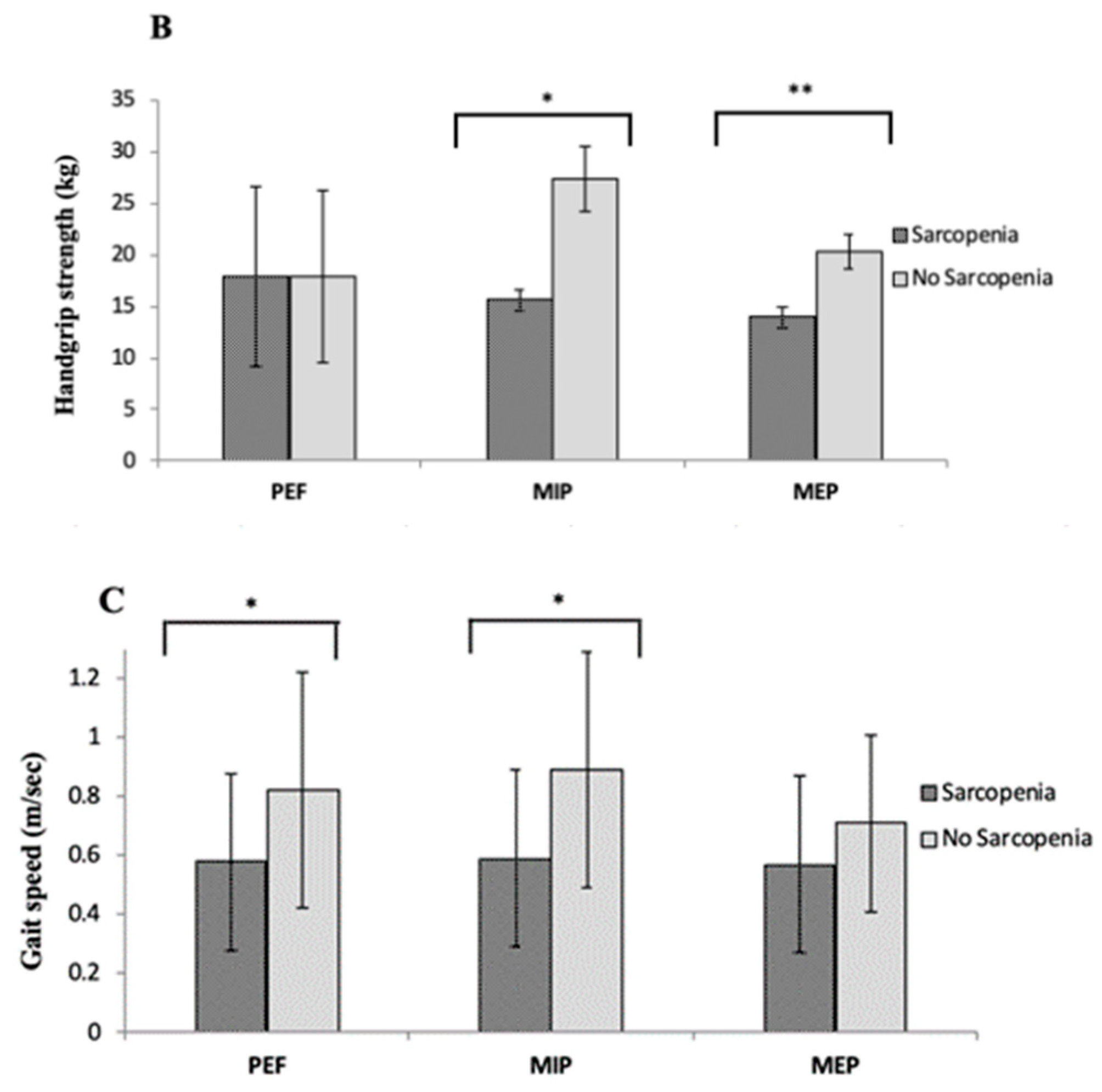
3.3. Evaluation of the Relationship between Muscle Skeleñata Parameters (Mass and Function) and Muscle Respiratory Function
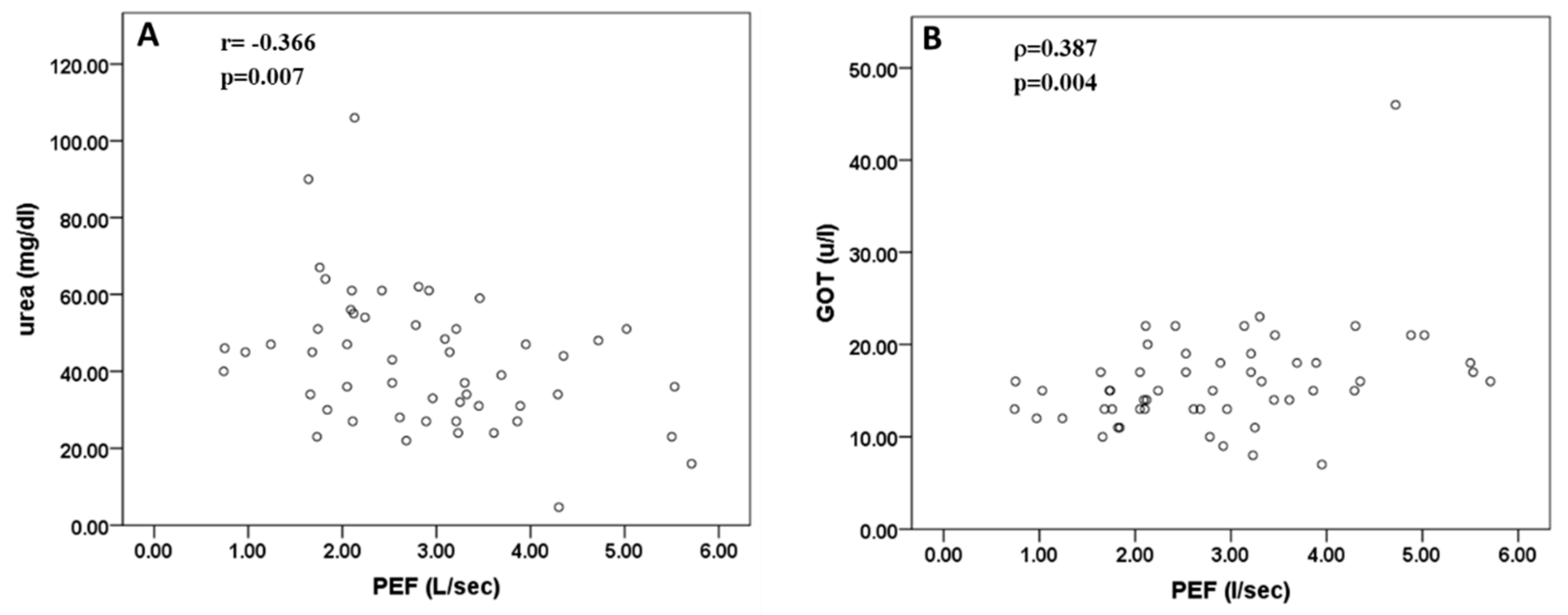
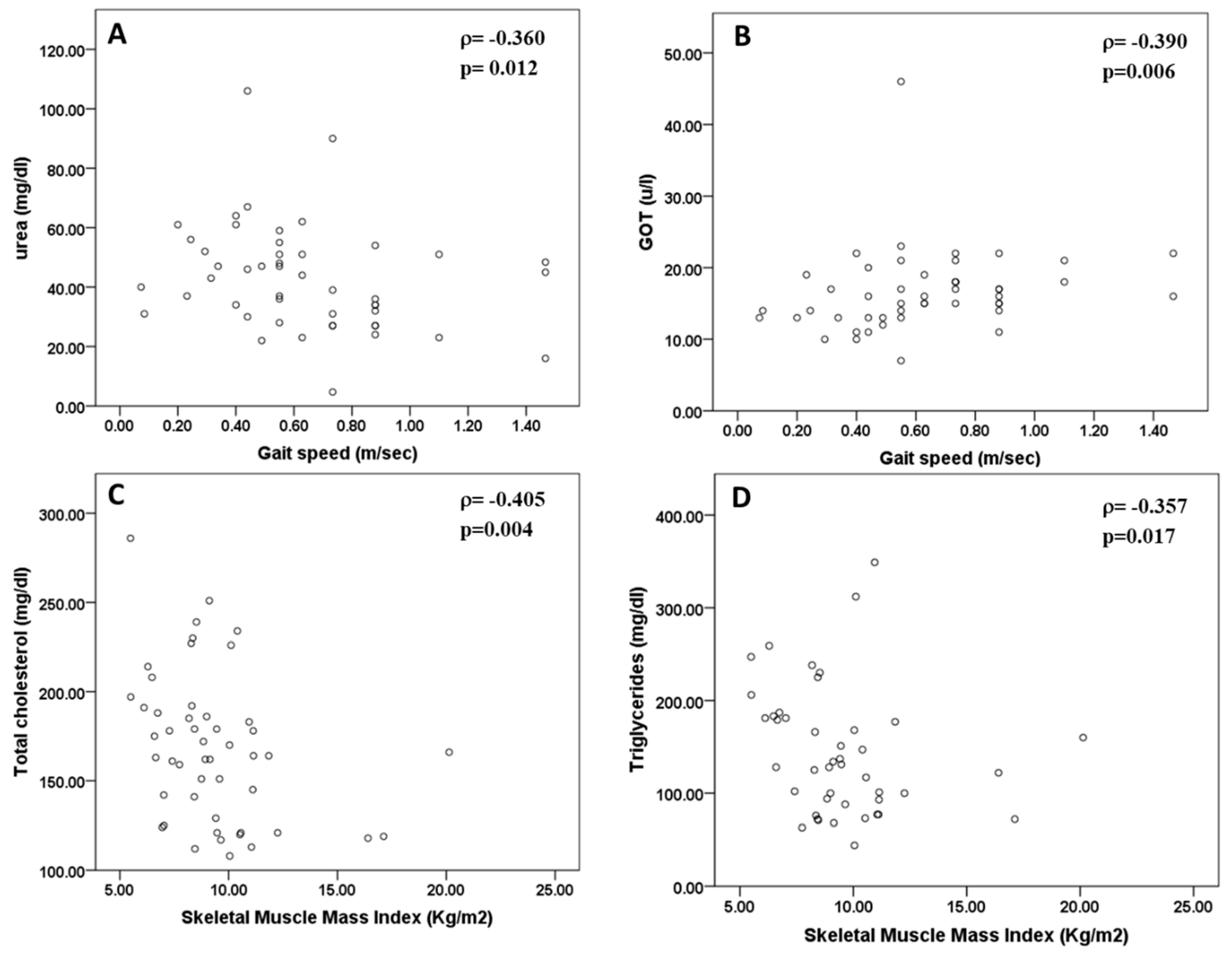
3.4. Evaluation of the Relationship between Sarcopenia Parameters and Blood Analytical Markers
4. Discussion
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| 95% CI | 95% Confidence interval |
| EDTA | Ethylenediaminetetraacetic acid |
| EWGSOP | European Working Group for Sarcopenia in Older People |
| FEV1 | Forced Expiratory Volume in first second |
| FEV2575 | Mesoexpiratory volume |
| FVC | Forced vital capacity |
| GOT | Glutamic oxaloacetic transaminase |
| GPT | Glutamic pyruvic transaminase |
| MEP | Maximal expiratory pressure |
| MIP | Maximal inspiratory pressure |
| NS | Non-sarcopenic individuals |
| PEF | Peak expiratory flow |
| RMS | Respiratory muscle strength |
| S | Sarcopenic individuals |
| Sat O2 | Oxyhemoglobinic saturation |
| SD | Standard deviation |
| VC | Vital capacity |
References
- Cruz-Jentoft, A.J.; Bahat, G.; Bauer, J.; Boirie, Y.; Bruyère, O.; Cederholm, T.; Cooper, C.; Landi, F.; Rolland, Y.; Sayer, A.A.; et al. Sarcopenia: Revised {European} consensus on definition and diagnosis. Age Ageing 2019, 48, 16–31. [Google Scholar] [CrossRef] [PubMed]
- Cruz-Jentoft, A.J.; Baeyens, J.P.; Bauer, J.M.; Boirie, Y.; Cederholm, T.; Landi, F.; Martin, F.C.; Michel, J.P.; Rolland, Y.; Schneider, S.M.; et al. Sarcopenia: European consensus on definition and diagnosis. Age Ageing 2010, 39, 412–423. [Google Scholar] [CrossRef] [PubMed]
- Elliott, J.E.; Greising, S.M.; Mantilla, C.B.; Sieck, G.C. Functional impact of sarcopenia in respiratory muscles. Respir. Physiol. Neurobiol. 2016, 226, 137–146. [Google Scholar] [CrossRef] [PubMed]
- Ohara, D.G.; Pegorari, M.S.; Oliveira dos Santos, N.L.; de Fátima Ribeiro Silva, C.; Monteiro, R.L.; Matos, A.P.; Jamami, M. Respiratory Muscle Strength as a Discriminator of Sarcopenia in Community-Dwelling Elderly: A Cross-Sectional Study. J. Nutr. Health Aging 2018, 22, 952–958. [Google Scholar] [CrossRef] [PubMed]
- Papadopoulou, S.K.; Tsintavis, P.; Potsaki, G.; Papandreou, D. Differences in the Prevalence of Sarcopenia in Community-Dwelling, Nursing Home and Hospitalized Individuals. A Systematic Review and Meta-Analysis. J. Nutr. Heal. Aging 2020, 24, 83–90. [Google Scholar] [CrossRef]
- Rodríguez-Rejón, A.I.; Ruiz-López, M.D.; Wanden-Berghe, C.; Artacho, R. Prevalence and Diagnosis of Sarcopenia in Residential Facilities: A Systematic Review. Adv. Nutr. 2019, 10, 51–58. [Google Scholar] [CrossRef]
- Shen, Y.; Chen, J.; Chen, X.; Hou, L.S.; Lin, X.; Yang, M. Prevalence and Associated Factors of Sarcopenia in Nursing Home Residents: A Systematic Review and Meta-analysis. J. Am. Med. Dir. Assoc. 2019, 20, 5–13. [Google Scholar] [CrossRef]
- Bravo-José, P.; Moreno, E.; Espert, M.; Romeu, M.; Martínez, P.; Navarro, C. Prevalence of sarcopenia and associated factors in institutionalised older adult patients. Clin. Nutr. ESPEN 2018, 27, 113–119. [Google Scholar] [CrossRef]
- Kovacs, E.; Lowery, E.; Kuhlmann, E.; Brubaker, A. The aging lung. Clin. Interv. Aging 2013, 8, 1489. [Google Scholar] [CrossRef]
- Skloot, G.S. The Effects of Aging on Lung Structure and Function. Clin. Geriatr. Med. 2017, 33, 447–457. [Google Scholar] [CrossRef]
- Conn, P.M. Handbook of Models for Human Aging; Elsevier Academic Press: Cambridge, MA, USA, 2006; ISBN 9780080460062. [Google Scholar]
- Sharma, G.; Goodwin, J. Effect of aging on respiratory system physiology and immunology. Clin. Interv. Aging 2006, 1, 253–260. [Google Scholar] [CrossRef]
- Vaz Fragoso, C.A.; Gahbauer, E.A.; Van Ness, P.H.; Concato, J.; Gill, T.M. Peak Expiratory Flow as a Predictor of Subsequent Disability and Death in Community-Living Older Persons. J. Am. Geriatr. Soc. 2008, 56, 1014–1020. [Google Scholar] [CrossRef]
- Roberts, M.H.; Mapel, D.W. Limited Lung Function: Impact of Reduced Peak Expiratory Flow on Health Status, Health-Care Utilization, and Expected Survival in Older Adults. Am. J. Epidemiol. 2012, 176, 127–134. [Google Scholar] [CrossRef] [PubMed]
- Ida, S.; Watanabe, M.; Yoshida, N.; Baba, Y.; Umezaki, N.; Harada, K.; Karashima, R.; Imamura, Y.; Iwagami, S.; Baba, H. Sarcopenia is a Predictor of Postoperative Respiratory Complications in Patients with Esophageal Cancer. Ann. Surg. Oncol. 2015, 22, 4432–4437. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Tan, S.; Jiang, Y.; Xi, Q.; Meng, Q.; Zhuang, Q.; Han, Y.; Sui, X.; Wu, G. Sarcopenia as a predictor of poor surgical and oncologic outcomes after abdominal surgery for digestive tract cancer: A prospective cohort study. Clin. Nutr. 2018. [Google Scholar] [CrossRef] [PubMed]
- Nishigori, T.; Okabe, H.; Tanaka, E.; Tsunoda, S.; Hisamori, S.; Sakai, Y. Sarcopenia as a predictor of pulmonary complications after esophagectomy for thoracic esophageal cancer. J. Surg. Oncol. 2016, 113, 678–684. [Google Scholar] [CrossRef]
- Maeda, K.; Akagi, J. Muscle Mass Loss Is a Potential Predictor of 90-Day Mortality in Older Adults with Aspiration Pneumonia. J. Am. Geriatr. Soc. 2017, 65, e18–e22. [Google Scholar] [CrossRef]
- Izawa, K.P.; Watanabe, S.; Oka, K.; Kasahara, Y.; Morio, Y.; Hiraki, K.; Hirano, Y.; Omori, Y.; Suzuki, N.; Kida, K.; et al. Respiratory muscle strength in relation to sarcopenia in elderly cardiac patients. Aging Clin. Exp. Res. 2016, 28, 1143–1148. [Google Scholar] [CrossRef]
- Omran, M.L.; Morley, J.E. Assessment of protein energy malnutrition in older persons, part II: Laboratory evaluation. Nutrition 2000, 16, 131–140. [Google Scholar] [CrossRef]
- Zhang, Z.; Pereira, S.L.; Luo, M.; Matheson, E.M. Evaluation of blood biomarkers associated with risk of malnutrition in older adults: A systematic review and meta-analysis. Nutrients 2017, 9, 829. [Google Scholar] [CrossRef]
- Li, S.; Zhang, J.; Zheng, H.; Wang, X.; Liu, Z.; Sun, T. Prognostic Role of Serum Albumin, Total Lymphocyte Count, and Mini Nutritional Assessment on Outcomes After Geriatric Hip Fracture Surgery: A Meta-Analysis and Systematic Review. J. Arthroplast. 2019, 34, 1287–1296. [Google Scholar] [CrossRef] [PubMed]
- Shakersain, B.; Santoni, G.; Faxén-Irving, G.; Rizzuto, D.; Fratiglioni, L.; Xu, W. Nutritional status and survival among old adults: An 11-year population-based longitudinal study. Eur. J. Clin. Nutr. 2016, 70, 320–325. [Google Scholar] [CrossRef] [PubMed]
- Zelada Rodríguez, M.A.; Gómez-Pavón, J.; Sorando Fernández, P.; Franco Salinas, A.; Mercedes Guzmán, L.; Baztán, J.J. Fiabilidad interobservador de los 4 índices de comorbilidad más utilizados en pacientes ancianos. Rev. Esp. Geriatr. Gerontol. 2012, 47, 67–70. [Google Scholar] [CrossRef]
- Charlson, M.E.; Pompei, P.; Ales, K.L.; MacKenzie, C.R. A new method of classifying prognostic comorbidity in longitudinal studies: Development and validation. J. Chronic Dis. 1987, 40, 373–383. [Google Scholar] [CrossRef]
- Miller, M.R.; Hankinson, J.; Brusasco, V.; Burgos, F.; Casaburi, R.; Coates, A.; Crapo, R.; Enright, P.; van der Grinten, C.P.M.; Gustafsson, P.; et al. Standardisation of spirometry. Eur. Respir. J. 2005, 26, 319–338. [Google Scholar] [CrossRef] [PubMed]
- American Thoracic Society. ATS/ERS Statement on respiratory muscle testing. Am. J. Respir. Crit. Care Med. 2002, 166, 518–624. [Google Scholar] [CrossRef]
- Laveneziana, P.; Albuquerque, A.; Aliverti, A.; Babb, T.; Barreiro, E.; Dres, M.; Dubé, B.P.; Fauroux, B.; Gea, J.; Guenette, J.A.; et al. ERS statement on respiratory muscle testing at rest and during exercise. Eur. Respir. J. 2019, 53. [Google Scholar] [CrossRef]
- Roberts, H.C.; Denison, H.J.; Martin, H.J.; Patel, H.P.; Syddall, H.; Cooper, C.; Sayer, A.A. A review of the measurement of grip strength in clinical and epidemiological studies: Towards a standardised approach. Age Ageing 2011, 40, 423–429. [Google Scholar] [CrossRef]
- Studenski, S.; Perera, S.; Patel, K.; Rosano, C.; Faulkner, K.; Inzitari, M.; Brach, J.; Chandler, J.; Cawthon, P.; Connor, E.B.; et al. Gait speed and survival in older adults. JAMA J. Am. Med. Assoc. 2011, 305, 50–58. [Google Scholar] [CrossRef]
- Janssen, I.; Heymsfield, S.B.; Baumgartner, R.N.; Ross, R. Estimation of skeletal muscle mass by bioelectrical impedance analysis. J. Appl. Physiol. 2000, 89, 465–471. [Google Scholar] [CrossRef]
- Martínez-Arnau, F.M.; Fonfría-Vivas, R.; Buigues, C.; Castillo, Y.; Molina, P.; Hoogland, A.J.; van Doesburg, F.; Pruimboom, L.; Fernández-Garrido, J.; Cauli, O. Effects of leucine administration in sarcopenia: A randomized and placebo-controlled clinical trial. Nutrients 2020, 12, 932. [Google Scholar] [CrossRef] [PubMed]
- Masanés, F.; Rojano i Luque, X.; Salvà, A.; Serra-Rexach, J.A.; Artaza, I.; Formiga, F.; Cuesta, F.; López Soto, A.; Ruiz, D.; Cruz-Jentoft, A.J. Cut-off points for muscle mass—not grip strength or gait speed—determine variations in sarcopenia prevalence. J. Nutr. Heal. Aging 2017, 21, 825–829. [Google Scholar] [CrossRef]
- Liu, X.; Hou, L.; Xia, X.; Liu, Y.; Zuo, Z.; Zhang, Y.; Zhao, W.; Hao, Q.; Yue, J.; Dong, B. Prevalence of sarcopenia in multi ethnics adults and the association with cognitive impairment: Findings from West-China health and aging trend study. BMC Geriatr. 2020, 20. [Google Scholar] [CrossRef] [PubMed]
- Kera, T.; Kawai, H.; Hirano, H.; Kojima, M.; Watanabe, Y.; Motokawa, K.; Fujiwara, Y.; Ihara, K.; Kim, H.; Obuchi, S. Definition of Respiratory Sarcopenia With Peak Expiratory Flow Rate. J. Am. Med. Dir. Assoc. 2019, 20, 1021–1025. [Google Scholar] [CrossRef] [PubMed]
- Bahat, G.; Tufan, A.; Ozkaya, H.; Tufan, F.; Akpinar, T.S.; Akin, S.; Bahat, Z.; Kaya, Z.; Kiyan, E.; Erten, N.; et al. Relation between hand grip strength, respiratory muscle strength and spirometric measures in male nursing home residents. Aging Male 2014, 17, 136–140. [Google Scholar] [CrossRef]
- Schramm, C.M. Current concepts of respiratory complications of neuromuscular disease in children. Curr. Opin. Pediatr. 2000, 12, 203–207. [Google Scholar] [CrossRef] [PubMed]
- Kang, S.W.; Bach, J.R. Maximum insufflation capacity: Vital capacity and cough flows in neuromuscular disease. Am. J. Phys. Med. Rehabil. 2000, 79, 222–227. [Google Scholar] [CrossRef] [PubMed]
- Lowery, E.M.; Brubaker, A.L.; Kuhlmann, E.; Kovacs, E.J. The aging lung. Clin. Interv. Aging 2013, 8, 1489–1496. [Google Scholar] [PubMed]
- Lalley, P.M. The aging respiratory system-Pulmonary structure, function and neural control. Respir. Physiol. Neurobiol. 2013, 187, 199–210. [Google Scholar] [CrossRef]
- Schmidt-Nowara, W.W.; Altman, A.R. Atelectasis and neuromuscular respiratory failure. Chest 1984, 85, 792–795. [Google Scholar] [CrossRef]
- Kera, T.; Kawai, H.; Hirano, H.; Kojima, M.; Fujiwara, Y.; Ihara, K.; Obuchi, S. Relationships among peak expiratory flow rate, body composition, physical function, and sarcopenia in community-dwelling older adults. Aging Clin. Exp. Res. 2018, 30, 331–340. [Google Scholar] [CrossRef] [PubMed]
- Janssens, J.P.; Pache, J.C.; Nicod, L.P. Physiological changes in respiratory function associated with ageing. Eur. Respir. J. 1999, 13, 197–205. [Google Scholar] [CrossRef] [PubMed]
- Elagizi, A.; Kachur, S.; Lavie, C.J.; Carbone, S.; Pandey, A.; Ortega, F.B.; Milani, R.V. An Overview and Update on Obesity and the Obesity Paradox in Cardiovascular Diseases. Prog. Cardiovasc. Dis. 2018, 61, 142–150. [Google Scholar] [CrossRef] [PubMed]
- Do, J.G.; Park, C.H.; Lee, Y.T.; Yoon, K.J. Association between underweight and pulmonary function in 282,135 healthy adults: A cross-sectional study in Korean population. Sci. Rep. 2019, 9, 14308. [Google Scholar] [CrossRef] [PubMed]
- Jeon, Y.K.; Shin, M.J.; Kim, M.H.; Mok, J.H.; Kim, S.S.; Kim, B.H.; Kim, S.J.; Kim, Y.K.; Chang, J.H.; Shin, Y.B.; et al. Low pulmonary function is related with a high risk of sarcopenia in community-dwelling older adults: The Korea National Health and Nutrition Examination Survey (KNHANES) 2008–2011. Osteoporos. Int. 2015, 26, 2423–2429. [Google Scholar] [CrossRef] [PubMed]
- Park, C.H.; Yi, Y.; Do, J.G.; Lee, Y.T.; Yoon, K.J. Relationship between skeletal muscle mass and lung function in Korean adults without clinically apparent lung disease. Medicine 2018, 97. [Google Scholar] [CrossRef]
- Yanek, L.R.; Vaidya, D.; Kral, B.G.; Dobrosielski, D.A.; Moy, T.F.; Stewart, K.J.; Becker, D.M. Lean Mass and Fat Mass as Contributors to Physical Fitness in an Overweight and Obese African American Population. Ethn Dis. 2015, 25, 214–219. [Google Scholar]
- Carbone, S.; Billingsley, H.E.; Rodriguez-Miguelez, P.; Kirkman, D.L.; Garten, R.; Franco, R.L.; Lee, D.-C.; Lavie, C.J. Lean Mass Abnormalities in Heart Failure: The Role of Sarcopenia, Sarcopenic Obesity, and Cachexia. Curr. Probl. Cardiol. 2019, 100417. [Google Scholar] [CrossRef]
- Magnani, K.L.; Cataneo, A.J.M. Respiratory muscle strength in obese individuals and influence of upper-body fat distribution. Sao Paulo Med. J. 2007, 125, 215–219. [Google Scholar] [CrossRef]
- Lin, C.K.; Lin, C.C. Work of breathing and respiratory drive in obesity. Respirology 2012, 17, 402–411. [Google Scholar] [CrossRef]
- Lima, T.R.L.; Almeida, V.P.; Ferreira, A.S.; Guimarães, F.S.; Lopes, A.J. Handgrip strength and pulmonary disease in the elderly: What is the link? Aging Dis. 2019, 10, 1109–1129. [Google Scholar] [CrossRef] [PubMed]
- iee Shin, H.; Kim, D.K.; Seo, K.M.; Kang, S.H.; Lee, S.Y.; Son, S. Relation between respiratory muscle strength and skeletal muscle mass and hand grip strength in the healthy elderly. Ann. Rehabil. Med. 2017, 41, 686–692. [Google Scholar] [CrossRef] [PubMed]
- Sawaya, Y.; Ishizaka, M.; Kubo, A.; Sadakiyo, K.; Yakabi, A.; Sato, T.; Shiba, T.; Onoda, K.; Maruyama, H. Correlation between skeletal muscle mass index and parameters of respiratory function and muscle strength in young healthy adults according to gender. J. Phys. Ther. Sci. 2018, 30, 1424–1427. [Google Scholar] [CrossRef]
- Peterson, S.J.; Park, J.; Zellner, H.K.; Moss, O.A.; Welch, A.; Sclamberg, J.; Moran, E.; Hicks-McGarry, S.; Becker, E.A.; Foley, S. Relationship Between Respiratory Muscle Strength, Handgrip Strength, and Muscle Mass in Hospitalized Patients. J. Parenter. Enter. Nutr. 2019. [Google Scholar] [CrossRef] [PubMed]
- Chowdhury, M.S.I.; Rahman, A.Z.; Haque, M.; Nahar, N.; Taher, A. Serum Aspartate Aminotransferase (AST) and Alanine Aminotransferase (ALT) Levels in Different Grades of Protein Energy Malnutrition. J. Bangladesh Soc. Physiol. 1970, 2, 17–19. [Google Scholar] [CrossRef][Green Version]
- Karaphillis, E.; Goldstein, R.; Murphy, S.; Qayyum, R. Serum alanine aminotransferase levels and all-cause mortality. Eur. J. Gastroenterol. Hepatol. 2017, 29, 284–288. [Google Scholar] [CrossRef]
- Nathwani, R.A.; Pais, S.; Reynolds, T.B.; Kaplowitz, N. Serum alanine aminotransferase in skeletal muscle diseases. Hepatology 2005, 41, 380–382. [Google Scholar] [CrossRef]
- Malakouti, M.; Kataria, A.; Ali, S.K.; Schenker, S. Elevated Liver Enzymes in Asymptomatic Patients—What Should I Do? J. Clin. Transl. Hepatol. 2017, 5, 1–10. [Google Scholar] [CrossRef]
- Shibata, M.; Nakajima, K.; Higuchi, R.; Iwane, T.; Sugiyama, M.; Nakamura , T. Nakamura High Concentration of Serum Aspartate Aminotransferase in Older Underweight People: Results of the Kanagawa Investigation of the Total Check-Up Data from the National Database-2 (KITCHEN-2). J. Clin. Med. 2019, 8, 1282. [Google Scholar] [CrossRef]
- Zamboni, M.; Mazzali, G.; Fantin, F.; Rossi, A.; Di Francesco, V. Sarcopenic obesity: A new category of obesity in the elderly. Nutr. Metab. Cardiovasc. Dis. 2008, 18, 388–395. [Google Scholar] [CrossRef]
- Stenholm, S.; Harris, T.B.; Rantanen, T.; Visser, M.; Kritchevsky, S.B.; Ferrucci, L. Sarcopenic obesity: Definition, cause and consequences. Curr. Opin. Clin. Nutr. Metab. Care 2008, 11, 693–700. [Google Scholar] [CrossRef] [PubMed]
- Haines, R.W.; Zolfaghari, P.; Wan, Y.; Pearse, R.M.; Puthucheary, Z.; Prowle, J.R. Elevated urea-to-creatinine ratio provides a biochemical signature of muscle catabolism and persistent critical illness after major trauma. Intensive Care Med. 2019, 45, 1718–1731. [Google Scholar] [CrossRef] [PubMed]
- Lattanzi, B.; D’Ambrosio, D.; Merli, M. Hepatic Encephalopathy and Sarcopenia: Two Faces of the Same Metabolic Alteration. J. Clin. Exp. Hepatol. 2019, 9, 125–130. [Google Scholar] [CrossRef] [PubMed]
- Kang, Y.J.; Yoo, J.I.; Ha, Y.C. Sarcopenia feature selection and risk prediction using machine learning: A cross-sectional study. Medicine 2019, 98, e17699. [Google Scholar] [CrossRef]
- Macchi, C.; Molino-Lova, R.; Polcaro, P.; Guarducci, L.; Lauretani, F.; Cecchi, F.; Bandinelli, S.; Guralnik, J.M.; Ferrucci, L. Higher circulating levels of uric acid are prospectively associated with better muscle function in older persons. Mech. Ageing Dev. 2008, 129, 522–527. [Google Scholar] [CrossRef]
- Can, B.; Kara, O.; Kizilarslanoglu, M.C.; Arik, G.; Aycicek, G.S.; Sumer, F.; Civelek, R.; Demirtas, C.; Ulger, Z. Serum markers of inflammation and oxidative stress in sarcopenia. Aging Clin. Exp. Res. 2017, 29, 745–752. [Google Scholar] [CrossRef]
- Molino-Lova, R.; Sofi, F.; Pasquini, G.; Vannetti, F.; Del Ry, S.; Vassalle, C.; Clerici, M.; Sorbi, S.; Macchi, C. Higher uric acid serum levels are associated with better muscle function in the oldest old: Results from the Mugello Study. Eur. J. Intern. Med. 2017, 41, 39–43. [Google Scholar] [CrossRef]
- Batsis, J.A.; Villareal, D.T. Sarcopenic obesity in older adults: Aetiology, epidemiology and treatment strategies. Nat. Rev. Endocrinol. 2018, 14, 513–537. [Google Scholar] [CrossRef]
- Carnio, S.; LoVerso, F.; Baraibar, M.A.; Longa, E.; Khan, M.M.; Maffei, M.; Reischl, M.; Canepari, M.; Loefler, S.; Kern, H.; et al. Autophagy Impairment in Muscle Induces Neuromuscular Junction Degeneration and Precocious Aging. Cell Rep. 2014, 8, 1509–1521. [Google Scholar] [CrossRef]
- Baek, S.J.; Nam, G.E.; Han, K.D.; Choi, S.W.; Jung, S.W.; Bok, A.R.; Kim, Y.H.; Lee, K.S.; Han, B.D.; Kim, D.H. Sarcopenia and sarcopenic obesity and their association with dyslipidemia in Korean elderly men: The 2008-2010 Korea National Health and Nutrition Examination Survey. J. Endocrinol. Investig. 2014, 37, 247–260. [Google Scholar] [CrossRef]
- Mesinovic, J.; McMillan, L.; Shore-Lorenti, C.; De Courten, B.; Ebeling, P.; Scott, D. Metabolic Syndrome and Its Associations with Components of Sarcopenia in Overweight and Obese Older Adults. J. Clin. Med. 2019, 8, 145. [Google Scholar] [CrossRef] [PubMed]
- Lu, C.W.; Yang, K.C.; Chang, H.H.; Lee, L.T.; Chen, C.Y.; Huang, K.C. Sarcopenic obesity is closely associated with metabolic syndrome. Obes. Res. Clin. Pract. 2013, 7. [Google Scholar] [CrossRef] [PubMed]
- Buchman, A.S.; Boyle, P.A.; Wilson, R.S.; Leurgans, S.; Shah, R.C.; Bennett, D.A. Respiratory muscle strength predicts decline in mobility in older persons. Neuroepidemiology 2008, 31, 174–180. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Davenport, P.; Sapienza, C. Effect of expiratory muscle strength training on elderly cough function. Arch. Gerontol. Geriatr. 2009, 48, 361–366. [Google Scholar] [CrossRef]
- Kim, J.; Sapienza, C.M. Implications of expiratory muscle strength training for rehabilitation of the elderly: Tutorial. J. Rehabil. Res. Dev. 2005, 42, 211–223. [Google Scholar] [CrossRef]
- Laghi, F.; Tobin, M.J. Disorders of the respiratory muscles. Am. J. Respir. Crit. Care Med. 2003, 168, 10–48. [Google Scholar] [CrossRef]
- Meek, P.M.; Schwartzstein, R.M.; Adams, L.; Altose, M.D.; Breslin, E.H.; Carrieri-Kohlman, V.; Gift, A.; Hanley, M.V.; Harver, A.; Jones, P.W.; et al. Dyspnea: Mechanisms, assessment, and management: A consensus statement. Am. J. Respir. Crit. Care Med. 1999, 159, 321–340. [Google Scholar]
- Caruso, P.; De Albuquerque, A.L.P.; Santana, P.V.; Cardenas, L.Z.; Ferreira, J.G.; Prina, E.; Trevizan, P.F.; Pereira, M.C.; Iamonti, V.; Pletsch, R.; et al. Métodos diagnósticos para avaliação da força muscular inspiratória e expiratória. J. Bras. Pneumol. 2015, 41, 110–123. [Google Scholar] [CrossRef]


| Clinical and Demographic Characteristics of Participants | Mean Value ± SD (Range) or Percentage |
|---|---|
| Age (years) | 78.6 ± 8.9 (55–93) |
| Sex | Male 32.8%Female 67.2% |
| IBM (kg/m2) | 28.9 ± 6.1 (18.7–50.2) |
| Smokers | 15.5% |
| Use of bronchodilators as a usual treatment | 21.1% |
| Walking ability | Independent 63.8%Can 3.4%Walker 32.8% |
| Comorbidities (Charlson index) | 5.4 ± 1.9 (1–11) |
| Diabetes | 31.0% |
| Chronic obstructive pulmonary disease | 17.2% |
| Hypertension | 32.8% |
| Hypercholesterolemia | 37.9% |
| Congestive heart failure | 10.3% |
| Renal failure | 12.1% |
| Osteoporosis | 20.7% |
| Depression | 19.0% |
| Respiratory Parameters | Mean Value (± SD) | Range |
|---|---|---|
| SatO2 (%) | 95.9 ± 1.9 | 91.0–99.0 |
| Heart rate (bpm) | 77.1 ± 14.2 | 49.0–114.0 |
| FVC (L/s) | 1.8 ± 0.7 | 0.3–4.4 |
| FEV1(L/s) | 1.3 ± 0.5 | 0.3–2.9 |
| FEV1/FVC (%) | 76.5 ± 12.1 | 45.9–100.0 |
| FEV25-75 (L/s) | 1.2 ± 0.5 | 0.3–3.1 |
| PEF (L/s) | 2.8 ± 1.2 | 0.7–5.7 |
| MIP (H2O cm) | 36.5 ± 17.4 | 7.0–77.0 |
| MEP (H2O cm) | 58.9 ± 23.7 | 10.0–99.0 |
| Anthropometric Analysis | Mean Value (± SD) | Range | % of Individuals Fulfilling the EWGSOP Criterion for Sarcopenia |
|---|---|---|---|
| Muscle mass (Janssen) | 22.8 ± 8.2 | 13.2–49.5 | Reduced lean mass: 17.6% |
| Skeletal muscle mass index (Janssen) | 9.2 ± 2.8 | 5.5–20.1 | Reduced lean mass: 17.6% |
| Hand grip in dominant hand (Kg) | 17.9 ± 8.5 | 6.5–42.0 | Muscle strength (dominant hand): 84.5% |
| Hand grip in non-dominant hand (Kg) | 16.5 ± 7.6 | 3.3–36.7 | Muscle strength (non-dominant hand): 84.5% |
| Walking speed (m/s) | 0.6 ± 0.3 | 0.1–1.5 | Physical performance: 65.4% |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Martínez-Arnau, F.M.; Buigues, C.; Fonfría-Vivas, R.; Cauli, O. Respiratory Muscle Strengths and Their Association with Lean Mass and Handgrip Strengths in Older Institutionalized Individuals. J. Clin. Med. 2020, 9, 2727. https://doi.org/10.3390/jcm9092727
Martínez-Arnau FM, Buigues C, Fonfría-Vivas R, Cauli O. Respiratory Muscle Strengths and Their Association with Lean Mass and Handgrip Strengths in Older Institutionalized Individuals. Journal of Clinical Medicine. 2020; 9(9):2727. https://doi.org/10.3390/jcm9092727
Chicago/Turabian StyleMartínez-Arnau, Francisco Miguel, Cristina Buigues, Rosa Fonfría-Vivas, and Omar Cauli. 2020. "Respiratory Muscle Strengths and Their Association with Lean Mass and Handgrip Strengths in Older Institutionalized Individuals" Journal of Clinical Medicine 9, no. 9: 2727. https://doi.org/10.3390/jcm9092727
APA StyleMartínez-Arnau, F. M., Buigues, C., Fonfría-Vivas, R., & Cauli, O. (2020). Respiratory Muscle Strengths and Their Association with Lean Mass and Handgrip Strengths in Older Institutionalized Individuals. Journal of Clinical Medicine, 9(9), 2727. https://doi.org/10.3390/jcm9092727






