Implications of Hereditary Origin on the Immune Phenotype of Mismatch Repair-Deficient Cancers: Systematic Literature Review
Abstract
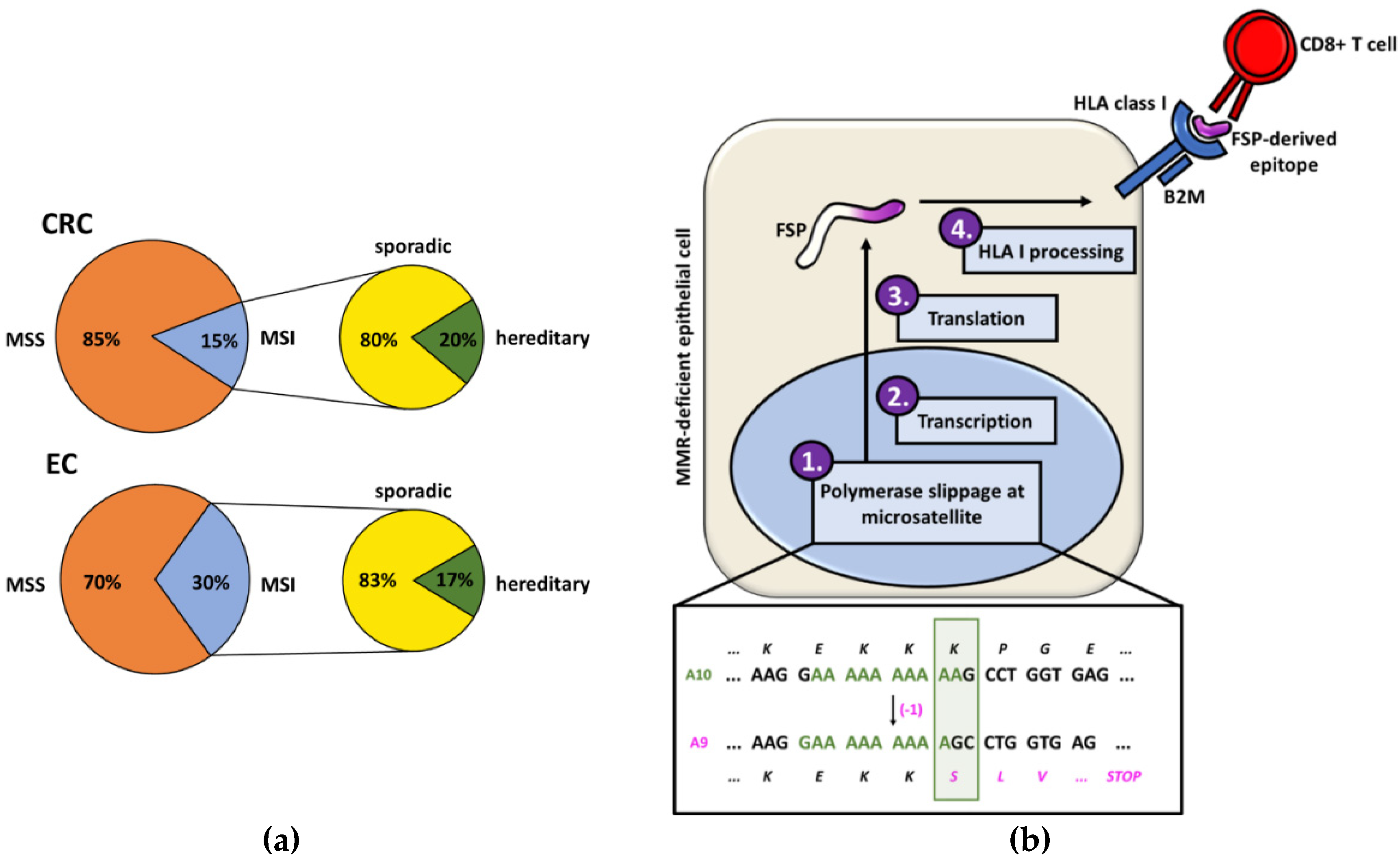
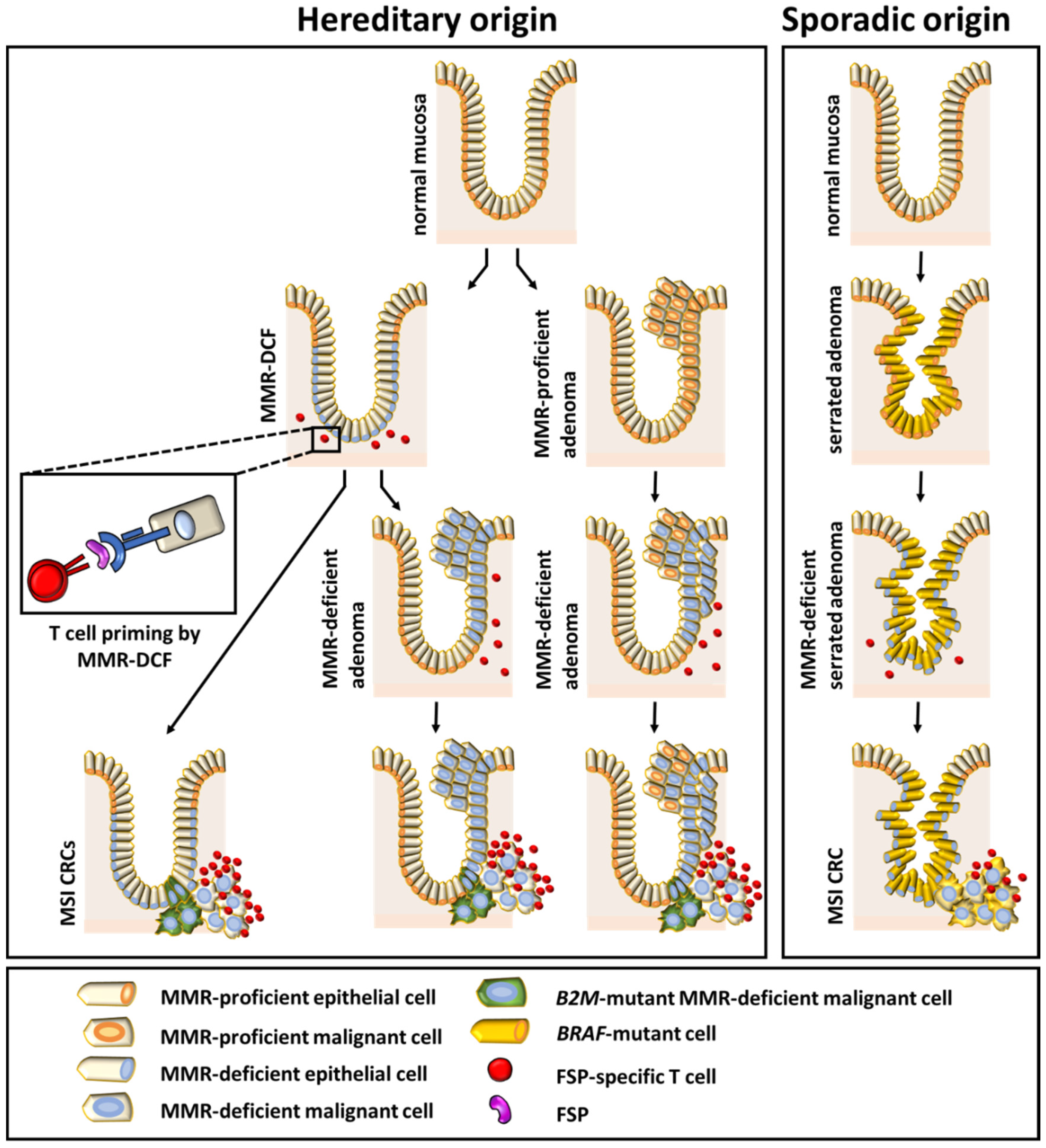
1. Introduction
2. Methods
2.1. Systematic Literature Search
2.2. Statistical Analysis
3. Results
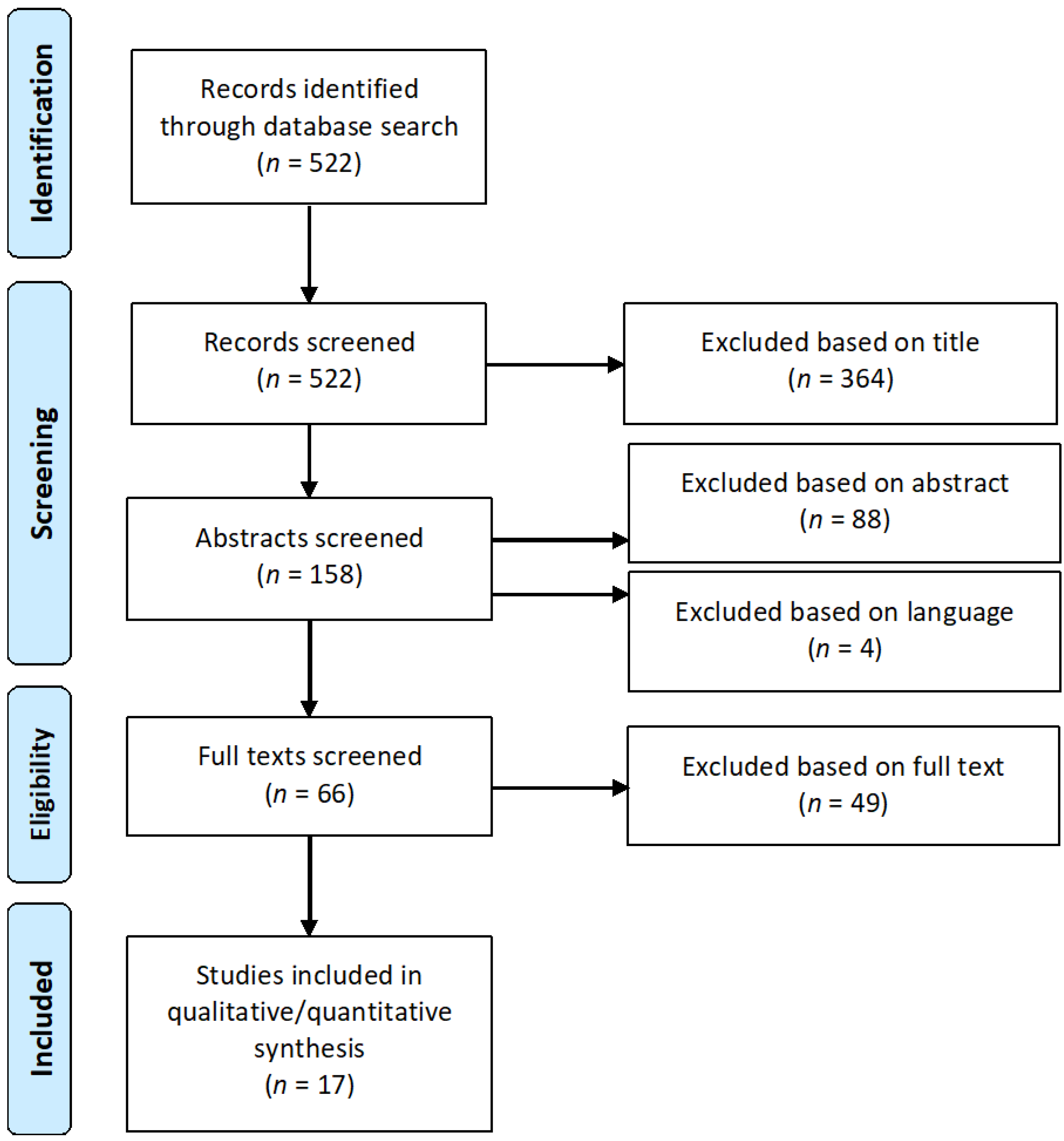
3.1. Outcome of the Systematic Literature Search
3.2. Immune Infiltration in Hereditary and Sporadic MSI CRCs
3.3. Immune Infiltration in Hereditary and Sporadic Pre-malignant Colorectal Lesions
3.4. Immune Infiltration in Hereditary and Sporadic ECs
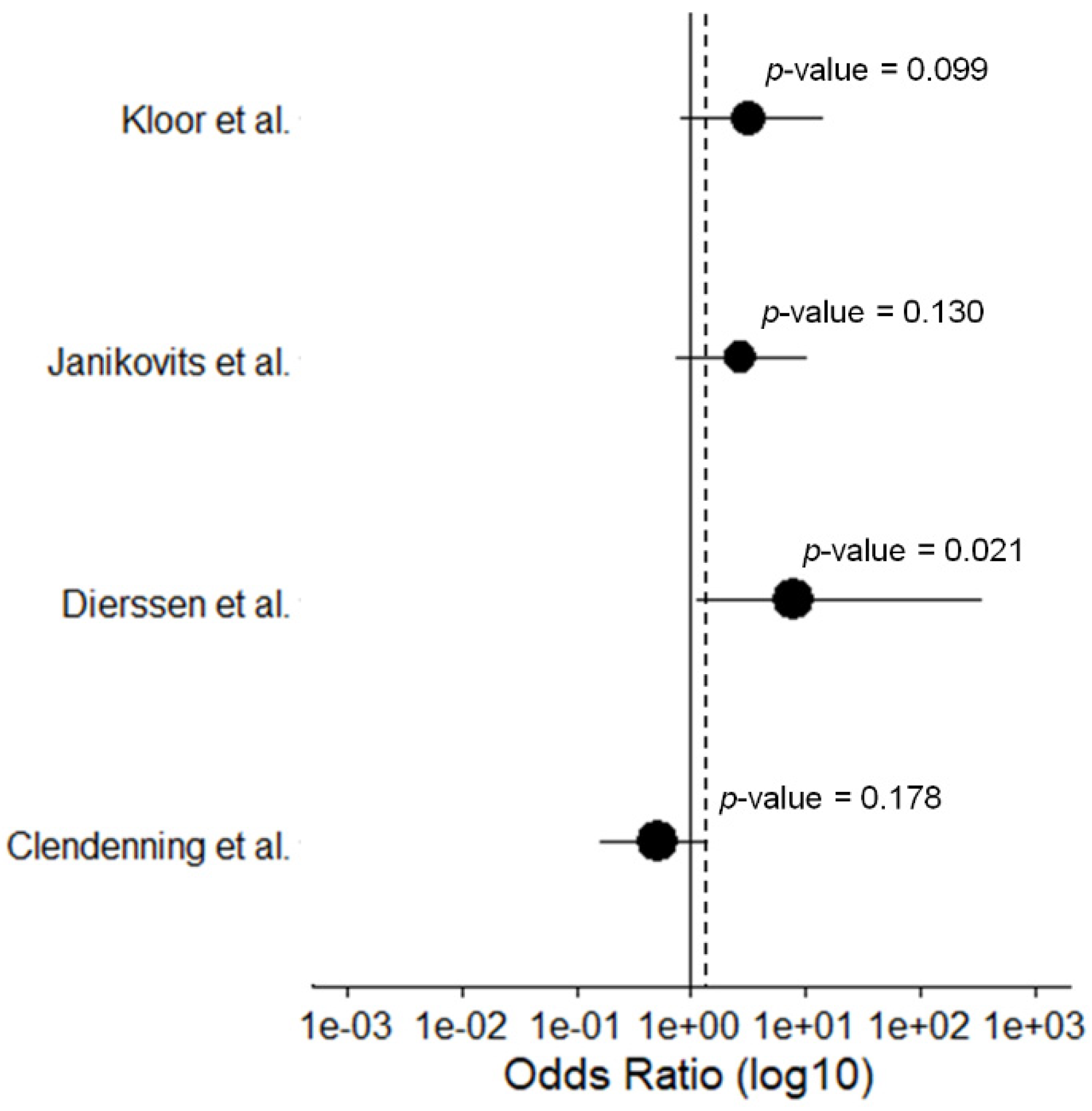
3.5. Immune Evasion Mechanisms in Hereditary and Sporadic MSI CRCs
3.6. Response to Therapy in Hereditary and Sporadic MSI CRCs
3.7. Alternative Explanations for Differing Immune Phenotypes
4. Summary
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Hause, R.J.; Pritchard, C.C.; Shendure, J.; Salipante, S.J. Classification and characterization of microsatellite instability across 18 cancer types. Nat. Med. 2016, 22, 1342–1350. [Google Scholar] [CrossRef]
- Kloor, M.; Von Knebel-Doeberitz, M. The immune biology of microsatellite unstable cancer. Trends Cancer 2016, 2, 121–133. [Google Scholar] [CrossRef]
- Linnebacher, M.; Gebert, J.; Rudy, W.; Woerner, S.; Yuan, Y.P.; Bork, P.; Von Knebel Doeberitz, M. Frameshift peptide-derived T-cell epitopes: A source of novel tumor-specific antigens. Int. J. Cancer 2001, 93, 6–11. [Google Scholar] [CrossRef]
- Saeterdal, I.; Gjertsen, M.K.; Straten, P.; Eriksen, J.A.; Gaudernack, G. A TFGβRII frameshift-mutation-derived CTL epitope recognised by HLA-A2-restricted CD8+T cells. Cancer Immunol. Immunother. 2001, 50, 469–476. [Google Scholar] [CrossRef] [PubMed]
- Ripberger, E.; Linnebacher, M.; Schwitalle, Y.; Gebert, J.; Von Knebel Doeberitz, M. Identification of an HLA-A0201-restricted CTL epitope generated by a tumor-specific frameshift mutation in a coding microsatellite of the OGT gene. J. Clin. Immunol. 2003, 23, 415–423. [Google Scholar] [CrossRef] [PubMed]
- Kloor, M.; Staffa, L.; Ahadova, A. Clinical significance of microsatellite instability in colorectal cancer. Langenbeck’s Arch. Surg. 2014, 399, 23–31. [Google Scholar] [CrossRef] [PubMed]
- Shia, J.; Holck, S.; DePetris, G.; Greenson, J.K.; Klimstra, D.S. Lynch syndrome-associated neoplasms: A discussion on histopathology and immunohistochemistry. Fam. Cancer 2013, 12, 241–260. [Google Scholar] [CrossRef]
- Buckowitz, A.; Knaebel, H.-P.; Benner, A.; Bläker, H.; Gebert, J.; Kienle, P.; Von Knebel Doeberitz, M.; Kloor, M. Microsatellite instability in colorectal cancer is associated with local lymphocyte infiltration and low frequency of distant metastases. Br. J. Cancer 2005, 92, 1746–1753. [Google Scholar] [CrossRef]
- Smyrk, T.C.; Watson, P.; Kaul, K.; Lynch, H.T. Tumor-Infiltrating Lymphocytes Are a Marker for Microsatellite Instability in Colorectal Carcinoma. Cancer 2001, 91, 2417–2422. [Google Scholar] [CrossRef]
- Dolcetti, R.; Viel, A.; Doglioni, C.; Russo, A.; Guidoboni, M.; Capozzi, E.; Vecchiato, N.; Macrì, E.; Fornasarig, M.; Boiocchi, M. High prevalence of activated intraepithelial cytotoxic T lymphocytes and increased neoplastic cell apoptosis in colorectal carcinomas with microsatellite instability. Am. J. Pathol. 1999, 154, 1805–1813. [Google Scholar] [CrossRef]
- Michael-Robinson, J.M. Tumour infiltrating lymphocytes and apoptosis are independent features in colorectal cancer stratified according to microsatellite instability status. Gut 2001, 48, 360–366. [Google Scholar] [CrossRef]
- Kloor, M.; Michel, S.; Buckowitz, B.; Rüschoff, J.; Büttner, R.; Holinski-Feder, E.; Dippold, W.; Wagner, R.; Tariverdian, M.; Benner, A.; et al. Beta2-microglobulin mutations in microsatellite unstable colorectal tumors. Int. J. Cancer 2007, 121, 454–458. [Google Scholar] [CrossRef] [PubMed]
- Ozcan, M.; Janikovits, J.; von Knebel Doeberitz, M.; Kloor, M. Complex pattern of immune evasion in MSI colorectal cancer. Oncoimmunology 2018, 7, e1445453. [Google Scholar] [CrossRef] [PubMed]
- Benatti, P.; Gafà, R.; Barana, D.; Marino, M.; Scarselli, A.; Pedroni, M.; Maestri, I.; Guerzoni, L.; Roncucci, L.; Menigatti, M.; et al. Microsatellite instability and colorectal cancer prognosis. Clin. Cancer Res. 2005, 11, 8332–8340. [Google Scholar] [CrossRef]
- Popat, S.; Hubner, R.; Houlston, R.S. Systematic Review of Microsatellite Instability and Colorectal Cancer Prognosis. J. Clin. Oncol. 2005, 23, 609–618. [Google Scholar] [CrossRef] [PubMed]
- Le, D.T.; Uram, J.N.; Wang, H.; Bartlett, B.R.; Kemberling, H.; Eyring, A.D.; Skora, A.D.; Luber, B.S.; Azad, N.S.; Laheru, D.; et al. PD-1 Blockade in Tumors with Mismatch-Repair Deficiency. N. Engl. J. Med. 2015, 372, 2509–2520. [Google Scholar] [CrossRef] [PubMed]
- Le, D.T.; Durham, J.N.; Smith, K.N.; Wang, H.; Bjarne, R.; Aulakh, L.K.; Lu, S.; Kemberling, H.; Wilt, C.; Brandon, S.; et al. Mismatch-repair deficiency predicts response of solid tumors to PD-1. Science 2017, 357, 409–413. [Google Scholar] [CrossRef]
- Kane, M.F.; Loda, M.; Gaida, G.M.; Lipman, J.; Mishra, R.; Goldman, H.; Jessup, J.M.; Kolodner, R. Methylation of the hMLH1 promoter correlates with lack of expression of hMLH1 in sporadic colon tumors and mismatch repair-defective human tumor cell lines. Cancer Res. 1997, 57, 808–811. [Google Scholar] [CrossRef]
- Cunningham, J.M.; Christensen, E.R.; Tester, D.J.; Burgart, L.J.; Thibodeau, S.N.; Roche, P.C. Hypermethylation of the hMLHl Promoter in Colon Cancer with Microsatellite Instability. Cancer Res. 1998, 58, 3455–3460. [Google Scholar]
- Veigl, M.L.; Kasturi, L.; Olechnowicz, J.; Ma, A.H.; Lutterbaugh, J.D.; Periyasamy, S.; Li, G.M.; Drummond, J.; Modrich, P.L.; Sedwick, W.D.; et al. Biallelic inactivation of hMLH1 by epigenetic gene silencing, a novel mechanism causing human MSI cancers. Proc. Natl. Acad. Sci. USA 1998, 95, 8698–8702. [Google Scholar] [CrossRef]
- Thibodeau, S.N.; French, A.J.; Cunningham, J.M.; Tester, D.; Burgart, L.J.; Roche, P.C.; McDonnell, S.K.; Schaid, D.J.; Vockley, C.W.; Michels, V.V.; et al. Microsatellite instability in colorectal cancer: Different mutator phenotypes and the principal involvement of hMLH1. Cancer Res. 1998, 58, 1713–1718. [Google Scholar] [PubMed]
- Herman, J.G.; Umar, A.; Polyak, K.; Graff, J.R.; Ahuja, N.; Issa, J.-P.J.; Markowitz, S.; Willson, J.K.V.; Hamilton, S.R.; Kinzler, K.W.; et al. Incidence and functional consequences of hMLH1 promoter hypermethylation in colorectal carcinoma. Proc. Natl. Acad. Sci. USA 1998, 95, 6870–6875. [Google Scholar] [CrossRef] [PubMed]
- Boland, C.R.; Goel, A. Microsatellite Instability in Colorectal Cancer. Gastroenterology 2010, 138, 2073–2087. [Google Scholar] [CrossRef] [PubMed]
- Knudson, A.G. Mutation and cancer: Statistical study of retinoblastoma. Proc. Natl. Acad. Sci. USA 1971, 68, 820–823. [Google Scholar] [CrossRef] [PubMed]
- Hampel, H.; Frankel, W.L.; Martin, E.; Arnold, M.; Khanduja, K.; Kuebler, P.; Nakagawa, H.; Sotamaa, K.; Prior, T.W.; Westman, J.; et al. Screening for the Lynch Syndrome (Hereditary Nonpolyposis Colorectal Cancer). N. Engl. J. Med. 2005, 352, 1851–1860. [Google Scholar] [CrossRef]
- ten Broeke, S.W.; Brohet, R.M.; Tops, C.M.; van der Klift, H.M.; Velthuizen, M.E.; Bernstein, I.; Capellá Munar, G.; Gomez Garcia, E.; Hoogerbrugge, N.; Letteboer, T.G.W.; et al. Lynch Syndrome Caused by Germline PMS2 Mutations: Delineating the Cancer Risk. J. Clin. Oncol. 2015, 33, 319–325. [Google Scholar] [CrossRef]
- Jasperson, K.W.; Tuohy, T.M.; Neklason, D.W.; Burt, R.W. Hereditary and Familial Colon Cancer. Gastroenterology 2010, 138, 2044–2058. [Google Scholar] [CrossRef]
- Vilar, E.; Gruber, S.B. Microsatellite Instability in Colorectal Cancer. Nat. Rev. Clin. Oncol. 2010, 7, 153–162. [Google Scholar] [CrossRef]
- Liccardo, R.; De Rosa, M.; Izzo, P.; Duraturo, F. Novel implications in molecular diagnosis of lynch syndrome. Gastroenterol. Res. Pract. 2017, 2017. [Google Scholar] [CrossRef]
- Giardiello, F.M.; Allen, J.I.; Axilbund, J.E.; Boland, C.R.; Burke, C.A.; Burt, R.W.; Church, J.M.; Dominitz, J.A.; Johnson, D.A.; Kaltenbach, T.; et al. Guidelines on genetic evaluation and management of lynch syndrome: A consensus statement by the us multi-society task force on colorectal cancer. Gastroenterology 2014, 147, 502–526. [Google Scholar] [CrossRef]
- Dominguez-Valentin, M.; Sampson, J.R.; Seppälä, T.T.; ten Broeke, S.W.; Plazzer, J.P.; Nakken, S.; Engel, C.; Aretz, S.; Jenkins, M.A.; Sunde, L.; et al. Cancer risks by gene, age, and gender in 6350 carriers of pathogenic mismatch repair variants: Findings from the Prospective Lynch Syndrome Database. Genet. Med. 2020, 22, 15–25. [Google Scholar] [CrossRef] [PubMed]
- Barrow, E.; Hill, J.; Evans, D.G. Cancer risk in Lynch Syndrome. Fam. Cancer 2013, 12, 229–240. [Google Scholar] [CrossRef] [PubMed]
- Boland, C.R.; Thibodeau, S.N.; Hamilton, S.R.; Sidransky, D.; Eshleman, J.R.; Burt, R.W.; Meltzer, S.J.; Rodriguez-Bigas, M.A.; Fodde, R.; Ranzani, G.N.; et al. A National Cancer Institute Workshop on Microsatellite Instability for cancer detection and familial predisposition: Development of international criteria for the determination of microsatellite instability in colorectal cancer. Cancer Res. 1998, 58, 5248–5257. [Google Scholar] [CrossRef] [PubMed]
- Umar, A.; Boland, C.R.; Terdiman, J.P.; Syngal, S.; De La Chapelle, A.; Rüschoff, J.; Fishel, R.; Lindor, N.M.; Burgart, L.J.; Hamelin, R.; et al. Revised Bethesda Guidelines for Hereditary Nonpolyposis Colorectal Cancer (Lynch Syndrome) and Microsatellite Instability. J. Natl. Cancer Inst. 2004, 96, 261–268. [Google Scholar] [CrossRef]
- Bettington, M.; Walker, N.; Clouston, A.; Brown, I.; Leggett, B.; Whitehall, V. The serrated pathway to colorectal carcinoma: Current concepts and challenges. Histopathology 2013, 62, 367–386. [Google Scholar] [CrossRef]
- Brenner, H.; Kloor, M.; Pox, C.P. Colorectal cancer. Lancet 2014, 383, 1490–1502. [Google Scholar] [CrossRef]
- Jass, J.R. Pathology of Hereditary Nonpolyposis. Ann. N. Y. Acad. Sci. 1994, 14, 1631–1634. [Google Scholar]
- Lynch, H.T.; Smyrk, T.; Jass, J.R. Hereditary nonpolyposis colorectal cancer and colonic adenomas: Aggressive adenomas? Semin. Surg. Oncol. 1995, 11, 406–410. [Google Scholar] [CrossRef]
- Love, R.R. Adenomas are precursor lesions for malignant growth in nonpolyposis hereditary carcinoma of the colon and rectum. Surg. Gynecol. Obstet. 1986, 162, 8–12. [Google Scholar]
- Pai, R.K.; Dudley, B.; Karloski, E.; Brand, R.E.; O’callaghan, N.; Rosty, C.; Buchanan, D.D.; Jenkins, M.A.; Thibodeau, S.N.; French, A.J.; et al. DNA Mismatch Repair Protein Deficient Non-Neoplastic Colonic Crypts: A Novel Indicator of Lynch Syndrome. Mod. Pathol. 2018, 31, 1608–1618. [Google Scholar] [CrossRef]
- Kloor, M.; Huth, C.; Voigt, A.Y.; Benner, A.; Schirmacher, P.; von Knebel Doeberitz, M.; Bläker, H. Prevalence of mismatch repair-deficient crypt foci in Lynch syndrome: A pathological study. Lancet Oncol. 2012, 13, 598–606. [Google Scholar] [CrossRef]
- Staffa, L.; Echterdiek, F.; Nelius, N.; Benner, A.; Werft, W.; Lahrmann, B.; Grabe, N.; Schneider, M.; Tariverdian, M.; Von Knebel Doeberitz, M.; et al. Mismatch repair-deficient crypt foci in lynch syndrome-molecular alterations and association with clinical parameters. PLoS ONE 2015, 10, e0121980. [Google Scholar] [CrossRef] [PubMed]
- Ahadova, A.; Gallon, R.; Gebert, J.; Ballhausen, A.; Endris, V.; Kirchner, M.; Stenzinger, A.; Burn, J.; von Knebel Doeberitz, M.; Bläker, H.; et al. Three molecular pathways model colorectal carcinogenesis in Lynch syndrome. Int. J. Cancer 2018, 143, 139–150. [Google Scholar] [CrossRef] [PubMed]
- RStudio Team. RStudio: Integrated Development for R; RStudio Inc.: Boston, MA, USA, 2019. [Google Scholar]
- Aragon, T.J. Epitools: Epidemiology Tools. Available online: https://CRAN.R-project.org/package=epitools (accessed on 8 May 2020).
- Wickham, H. ggplot2: Elegant Graphics for Data Analysis; Springer: New York, NY, USA, 2016; Available online: https://CRAN.Rproject.org/package=epitools (accessed on 8 May 2020).
- Janikovits, J.; Müller, M.; Krzykalla, J.; Körner, S.; Echterdiek, F.; Lahrmann, B.; Grabe, N.; Schneider, M.; Benner, A.; Von Knebel Doeberitz, M.; et al. High numbers of PDCD1 (PD-1)-positive T cells and B2M mutations in microsatellite-unstable colorectal cancer. Oncoimmunology 2018, 7, e1390640. [Google Scholar] [CrossRef] [PubMed]
- Shia, J.; Ellis, N.A.; Paty, P.B.; Nash, G.M.; Qin, J.; Offit, K.; Zhang, X.-M.; Markowitz, A.J.; Nafa, K.; Guillem, J.G.; et al. Value of histopathology in predicting microsatellite instability in hereditary nonpolyposis colorectal cancer and sporadic colorectal cancer. Am. J. Surg. Pathol. 2003, 27, 1407–1417. [Google Scholar] [CrossRef] [PubMed]
- Takemoto, N.; Konishi, F.; Yamashita, K.; Kojima, M.; Furukawa, T.; Miyakura, Y.; Shitoh, K.; Nagai, H. The Correlation of Microsatellite Instability and Tumor-infiltrating Lymphocytes in Hereditary Non-polyposis Colorectal Cancer (HNPCC) and Sporadic Colorectal Cancers: The Significance of Different Types of Lymphocyte Infiltration. Jpn. J. Clin. Oncol. 2004, 34, 90–98. [Google Scholar] [CrossRef]
- Koornstra, J.J.; de Jong, S.; Boersma-van Eck, W.; Zwart, N.; Hollema, H.; de Vries, E.G.E.; Kleibeuker, J.H. Fas ligand expression in lynch syndrome-associated colorectal tumours. Pathol. Oncol. Res. 2009, 15, 399–406. [Google Scholar] [CrossRef][Green Version]
- Surmann, E.-M.; Voigt, A.Y.; Michel, S.; Bauer, K.; Reuschenbach, M.; Ferrone, S.; von Knebel Doeberitz, M.; Kloor, M. Association of high CD4-positive T cell infiltration with mutations in HLA class II-regulatory genes in microsatellite-unstable colorectal cancer. Cancer Immunol. Immunother. 2015, 64, 357–366. [Google Scholar] [CrossRef]
- Pakish, J.B.; Zhang, Q.; Chen, Z.; Liang, H.; Chisholm, G.B.; Yuan, Y.; Mok, S.C.; Broaddus, R.R.; Lu, K.H.; Yates, M.S. Immune Microenvironment in Microsatellite-Instable Endometrial Cancers: Hereditary or Sporadic Origin Matters. Clin. Cancer Res. 2017, 23, 4473–4481. [Google Scholar] [CrossRef]
- Ramchander, N.C.; Ryan, N.A.J.; Bolton, J.; Bosse, T.; Walker, T.D.J.; Harries, L.; Evans, D.G.; Crosbie, E.J. Distinct Immunological Landscapes Characterize Inherited and Sporadic Mismatch Repair Deficient Endometrial Cancer. Front. Immunol. 2020, 10, 3023. [Google Scholar] [CrossRef]
- Young, J.; Simms, L.A.; Biden, K.G.; Wynter, C.; Whitehall, V.; Karamatic, R.; George, J.; Goldblatt, J.; Walpole, I.; Robin, S.-A.; et al. Features of Colorectal Cancers with High-Level Microsatellite Instability Occurring in Familial and Sporadic Settings Parallel Pathways of Tumorigenesis. Am. J. Pathol. 2001, 159, 2107–2116. [Google Scholar] [CrossRef]
- Jass, J.R.; Walsh, M.D.; Barker, M.; Simms, L.A.; Young, J.; Leggett, B.A. Distinction between familial and sporadic forms of colorectal cancer showing DNA microsatellite instability. Eur. J. Cancer 2002, 38, 858–866. [Google Scholar] [CrossRef]
- Wang, T.; Lee, L.H.; Vyas, M.; Zhang, L.; Ganesh, K.; Firat, C.; Segal, N.H.; Desai, A.; Hechtman, J.F. Colorectal carcinoma with double somatic mismatch repair gene inactivation: Clinical and pathological characteristics and response to immune checkpoint blockade. Mod. Pathol. 2019, 32, 1551–1562. [Google Scholar] [CrossRef] [PubMed]
- Tougeron, D.; Maby, P.; Elie, N.; Fauquembergue, É.; Le Pessot, F.; Cornic, M.; Sabourin, J.-C.; Michel, P.; Frébourg, T.; Latouche, J.-B. Regulatory T Lymphocytes Are Associated with Less Aggressive Histologic Features in Microsatellite-Unstable Colorectal Cancers. PLoS ONE 2013, 8, e61001. [Google Scholar] [CrossRef]
- Meijer, T.W.H.; Hoogerbrugge, N.; Nagengast, F.M.; Ligtenberg, M.J.L.; Van Krieken, J.H.J.M. In Lynch syndrome adenomas, loss of mismatch repair proteins is related to an enhanced lymphocytic response. Histopathology 2009, 55, 414–422. [Google Scholar] [CrossRef]
- Chang, K.; Taggart, M.W.; Reyes-Uribe, L.; Borras, E.; Riquelme, E.; Barnett, R.M.; Leoni, G.; San Lucas, F.A.; Catanese, M.T.; Mori, F.; et al. Immune Profiling of Premalignant Lesions in Patients With Lynch Syndrome. JAMA Oncol. 2018, 4, 1085–1092. [Google Scholar] [CrossRef]
- Ahtiainen, M.; Wirta, E.V.; Kuopio, T.; Seppälä, T.; Rantala, J.; Mecklin, J.P.; Böhm, J. Combined prognostic value of CD274 (PD-L1)/PDCDI (PD-1) expression and immune cell infiltration in colorectal cancer as per mismatch repair status. Mod. Pathol. 2019, 32, 866–883. [Google Scholar] [CrossRef]
- Lee, K.S.; Kwak, Y.; Ahn, S.; Shin, E.; Oh, H.-K.; Kim, D.-W.; Kang, S.-B.; Choe, G.; Kim, W.H.; Lee, H.S. Prognostic implication of CD274 (PD-L1) protein expression in tumor-infiltrating immune cells for microsatellite unstable and stable colorectal cancer. Cancer Immunol. Immunother. 2017, 66, 927–939. [Google Scholar] [CrossRef]
- Pfuderer, P.L.; Ballhausen, A.; Seidler, F.; Stark, H.-J.; Grabe, N.; Frayling, I.M.; Ager, A.; Von Knebel Doeberitz, M.; Kloor, M.; Ahadova, A. High endothelial venules are associated with microsatellite instability, hereditary background and immune evasion in colorectal cancer. Br. J. Cancer 2019, 121, 395–404. [Google Scholar] [CrossRef]
- Girard, J.-P.; Springer, T.A. High endothelial venules (HEVs): Specialized endothelium for lymphocyte migration. Immunol. Today 1995, 16, 449–457. [Google Scholar] [CrossRef]
- Schwitalle, Y.; Kloor, M.; Eiermann, S.; Linnebacher, M.; Kienle, P.; Knaebel, H.P.; Tariverdian, M.; Benner, A.; von Knebel Doeberitz, M. Immune Response Against Frameshift-Induced Neopeptides in HNPCC Patients and Healthy HNPCC Mutation Carriers. Gastroenterology 2008, 134, 988–997. [Google Scholar] [CrossRef] [PubMed]
- Sloan, E.A.; Ring, K.L.; Willis, B.C.; Modesitt, S.C.; Mills, A.M. PD-L1 Expression in Mismatch Repair-deficient Endometrial Carcinomas, Including Lynch Syndrome-associated and MLH1 Promoter Hypermethylated Tumors. Am. J. Surg. Pathol. 2017, 41, 326–333. [Google Scholar] [CrossRef] [PubMed]
- McKenzie, R.; Scott, R.J.; Otton, G.; Scurry, J. Early changes of endometrial neoplasia revealed by loss of mismatch repair gene protein expression in a patient diagnosed with Lynch syndrome. Pathology 2016, 48, 78–80. [Google Scholar] [CrossRef]
- Dunn, G.P.; Bruce, A.T.; Ikeda, H.; Old, L.J.; Schreiber, R.D. Cancer immunoediting: From immunosurveillance to tumor escape. Nat. Immunol. 2002, 3, 991–998. [Google Scholar] [CrossRef] [PubMed]
- Kloor, M.; Michel, S.; Von Knebel Doeberitz, M. Immune evasion of microsatellite unstable colorectal cancers. Int. J. Cancer 2010, 127, 1001–1010. [Google Scholar] [CrossRef] [PubMed]
- Tikidzhieva, A.; Benner, A.; Michel, S.; Formentini, A.; Link, K.-H.; Dippold, W.; von Knebel Doeberitz, M.; Kornmann, M.; Kloor, M. Microsatellite instability and Beta2-Microglobulin mutations as prognostic markers in colon cancer: Results of the FOGT-4 trial. Br. J. Cancer 2012, 106, 1239–1245. [Google Scholar] [CrossRef] [PubMed]
- Woerner, S.M.; Yuan, Y.P.; Benner, A.; Korff, S.; Von Knebel Doeberitz, M.; Bork, P. SelTarbase, a database of human mononucleotide-microsatellite mutations and their potential impact to tumorigenesis and immunology. Nucleic Acids Res. 2010, 38, D682–D689. [Google Scholar] [CrossRef]
- Echterdiek, F.; Janikovits, J.; Staffa, L.; Müller, M.; Lahrmann, B.; Frühschütz, M.; Hartog, B.; Nelius, N.; Benner, A.; Tariverdian, M.; et al. Low density of FOXP3-positive T cells in normal colonic mucosa is related to the presence of beta2-microglobulin mutations in Lynch syndrome-associated colorectal cancer. Oncoimmunology 2016, 5, e1075692. [Google Scholar] [CrossRef]
- Clendenning, M.; Huang, A.; Jayasekara, H.; Lorans, M.; Preston, S.; O’Callaghan, N.; Pope, B.J.; Macrae, F.A.; Winship, I.M.; Milne, R.L.; et al. Somatic mutations of the coding microsatellites within the beta-2-microglobulin gene in mismatch repair-deficient colorectal cancers and adenomas. Fam. Cancer 2018, 17, 91–100. [Google Scholar] [CrossRef]
- Dierssen, J.W.F.; de Miranda, N.F.C.C.; Mulder, A.; van Puijenbroek, M.; Verduyn, W.; Claas, F.H.J.; van de Velde, C.J.H.; Fleuren, G.J.; Cornelisse, C.J.; Corver, W.E.; et al. High-resolution analysis of HLA class I alterations in colorectal cancer. BMC Cancer 2006, 6. [Google Scholar] [CrossRef]
- Dabir, P.D.; Bruggeling, C.E.; van der Post, R.S.; Dutilh, B.E.; Hoogerbrugge, N.; Ligtenberg, M.J.L.; Boleij, A.; Nagtegaal, I.D. Microsatellite instability screening in colorectal adenomas to detect Lynch syndrome patients? A systematic review and meta-analysis. Eur. J. Hum. Genet. 2020, 28, 277–286. [Google Scholar] [CrossRef] [PubMed]
- Binder, H.; Hopp, L.; Schweiger, M.R.; Hoffmann, S.; Jühling, F.; Kerick, M.; Timmermann, B.; Siebert, S.; Grimm, C.; Nersisyan, L.; et al. Genomic and transcriptomic heterogeneity of colorectal tumours arising in Lynch syndrome. J. Pathol. 2017, 243, 242–254. [Google Scholar] [CrossRef] [PubMed]
- Seliger, B.; Kloor, M.; Ferrone, S. HLA class II antigen-processing pathway in tumors: Molecular defects and clinical relevance. Oncoimmunology 2017, 6, e1171447. [Google Scholar] [CrossRef] [PubMed]
- Ribic, C.M.; Sargent, D.J.; Moore, M.J.; Thibodeau, S.N.; French, A.J.; Goldberg, R.M.; Hamilton, S.R.; Laurent-Puig, P.; Gryfe, R.; Shepherd, L.E.; et al. Tumor microsatellite-instability status as a predictor of benefit from fluorouracil-based adjuvant chemotherapy for colon cancer. N. Engl. J. Med. 2003, 349, 247–257. [Google Scholar] [CrossRef] [PubMed]
- Benson, A.B.; Schrag, D.; Somerfield, M.R.; Cohen, A.M.; Figueredo, A.T.; Flynn, P.J.; Krzyzanowska, M.K.; Maroun, J.; McAllister, P.; Van Cutsem, E.; et al. American Society of Clinical Oncology recommendations on adjuvant chemotherapy for stage II colon cancer. J. Clin. Oncol. 2004, 22, 3408–3419. [Google Scholar] [CrossRef]
- Alwers, E.; Jansen, L.; Bläker, H.; Kloor, M.; Tagscherer, K.E.; Roth, W.; Boakye, D.; Herpel, E.; Grüllich, C.; Chang-Claude, J.; et al. Microsatellite instability and survival after adjuvant chemotherapy among stage II and III colon cancer patients: Results from a population-based study. Mol. Oncol. 2019, 14, 363–372. [Google Scholar] [CrossRef]
- Jo, W.-S.; Carethers, J.M. Chemotherapeutic implications in microsatellite unstable colorectal cancer. Cancer Biomark. 2006, 2, 51–60. [Google Scholar] [CrossRef]
- Sargent, D.J.; Marsoni, S.; Monges, G.; Thibodeau, S.N.; Labianca, R.; Hamilton, S.R.; French, A.J.; Kabat, B.; Foster, N.R.; Torri, V.; et al. Defective mismatch repair as a predictive marker for lack of efficacy of fluorouracil-based adjuvant therapy in colon cancer. J. Clin. Oncol. 2010, 28, 3219–3226. [Google Scholar] [CrossRef]
- Sinicrope, F.A.; Foster, N.R.; Thibodeau, S.N.; Marsoni, S.; Monges, G.; Labianca, R.; Yothers, G.; Allegra, C.; Moore, M.J.; Gallinger, S.; et al. DNA mismatch repair status and colon cancer recurrence and survival in clinical trials of 5-fluorouracil-based adjuvant therapy. J. Natl. Cancer Inst. 2011, 103, 863–875. [Google Scholar] [CrossRef]
- Zaanan, A.; Hunriques, J.; Cohen, R.; Sefrioui, D.; Evrard, C.; de la Fouchardiere, C.; Lecomte, T.; Aparicio, T.; Svrcek, M.; Taieb, J.; et al. Efficacy of Anti-EGFR in MSI Metastatic Colorectal Cancer Depending on Sporadic or Familial Orgin. Available online: https://academic.oup.com/jnci/advance-article/doi/10.1093/jnci/djaa072/5838189 (accessed on 8 May 2020).
- Overman, M.J.; Lonardi, S.; Wong, K.Y.M.; Lenz, H.J.; Gelsomino, F.; Aglietta, M.; Morse, M.A.; Van Cutsem, E.; McDermott, R.; Hill, A.; et al. Durable clinical benefit with nivolumab plus ipilimumab in DNA mismatch repair-deficient/microsatellite instability-high metastatic colorectal cancer. J. Clin. Oncol. 2018, 36, 773–779. [Google Scholar] [CrossRef]
- Yaghoubi, N.; Soltani, A.; Ghazvini, K.; Hassanian, S.M.; Hashemy, S.I. PD-1/PD-L1 blockade as a novel treatment for colorectal cancer. Biomed. Pharmacother. 2019, 110, 312–318. [Google Scholar] [CrossRef] [PubMed]
- Jiang, P.; Gu, S.; Pan, D.; Fu, J.; Sahu, A.; Hu, X.; Li, Z.; Traugh, N.; Bu, X.; Li, B.; et al. Signatures of T cell dysfunction and exclusion predict cancer immunotherapy response. Nat. Med. 2018, 24, 1550–1558. [Google Scholar] [CrossRef] [PubMed]
- Tumeh, P.C.; Harview, C.L.; Yearley, J.H.; Shintaku, I.P.; Taylor, E.J.M.; Robert, L.; Chmielowski, B.; Spasic, M.; Henry, G.; Ciobanu, V.; et al. PD-1 blockade induces responses by inhibiting adaptive immune resistance HHS Public Access. Nature 2014, 515, 568–571. [Google Scholar] [CrossRef] [PubMed]
- Liao, N.S.; Bix, M.; Zijlstra, M.; Jaenisch, R.; Raulet, D. MHC class I deficiency: Susceptibility to natural killer (NK) cells and impaired NK activity. Science 1991, 253, 199–202. [Google Scholar] [CrossRef] [PubMed]
- Jager, M.J.; Hurks, H.M.H.; Levitskaya, J.; Kiessling, R. HLA expression in uveal melanoma: There is no rule without some exception. Hum. Immunol. 2002, 63, 444–451. [Google Scholar] [CrossRef]
- Reimers, M.S.; Bastiaannet, E.; Langley, R.E.; Van Eijk, R.; Van Vlierberghe, R.L.P.; Lemmens, V.E.P.; Van Herk-Sukel, M.P.P.; Van Wezel, T.; Fodde, R.; Kuppen, P.J.K.; et al. Expression of HLA class i antigen, aspirin use, and survival after a diagnosis of colon cancer. JAMA Intern. Med. 2014, 174, 732–739. [Google Scholar] [CrossRef]
- Bari, S.; Wang, X.; Ajose, T.; Muzaffar, J. Characterization of Lynch syndrome (LS) associated cancers in patients with immune dysfunction. J. Clin. Oncol. 2019, 37, 1532. [Google Scholar] [CrossRef]
- Sinicrope, F.A.; Sargent, D.J. Clinical implications of microsatellite instability in sporadic colon cancers. Curr. Opin. Oncol. 2009, 21, 369–373. [Google Scholar] [CrossRef]
- Weng, N. Aging of the Immune System: How Much Can the Adaptive Immune System Adapt? Immunity 2006, 24, 495–499. [Google Scholar] [CrossRef]
- Goronzy, J.J.; Weyand, C.M. Immune aging and autoimmunity. Cell. Mol. Life Sci. 2012, 69, 1615–1623. [Google Scholar] [CrossRef]
- Weyand, C.M.; Goronzy, J.J. Aging of the Immune System. Mechanisms and Therapeutic Targets. Ann. Am. Thorac. Soc. 2016, 13, S422–S428. [Google Scholar] [CrossRef] [PubMed]
- Tsai, Y.-J.; Huang, S.-C.; Lin, H.-H.; Lin, C.-C.; Lan, Y.-T.; Wang, H.-S.; Yang, S.-H.; Jiang, J.-K.; Chen, W.-S.; Lin, T.; et al. Differences in gene mutations according to gender among patients with colorectal cancer. World J. Surg. Oncol. 2018, 16, 128. [Google Scholar] [CrossRef] [PubMed]
- Benedix, F.; Kube, R.; Meyer, F.; Schmidt, U.; Gastinger, I.; Lippert, H. Comparison of 17,641 Patients With Right- and Left-Sided Colon Cancer: Differences in Epidemiology, Perioperative Course, Histology, and Survival. Dis. Colon Rectum 2010, 53, 57–64. [Google Scholar] [CrossRef] [PubMed]
- Writing Group for the Women’s Health Initiative Investigators WG for the WHI. Risks and Benefits of Estrogen Plus Progestin in Healthy Postmenopausal Women: Principal Results From the Women’s Health Initiative Randomized Controlled Trial. JAMA J. Am. Med. Assoc. 2002, 288, 321–333. [Google Scholar] [CrossRef] [PubMed]
- Slattery, M.L.; Potter, J.; Caan, B.; Edwards, S.; Coates, A.; Ma, K.N.; Berry, T.D.; Samowitz, W.S. Estrogens Reduce and Withdrawal of Estrogens Increase Risk of Microsatellite-Instability-positive Colon Cancer. Cancer Res. 2001, 61, 126–130. [Google Scholar] [PubMed]
- Johnson, A.A.; Akman, K.; Calimport, S.R.G.; Wuttke, D.; Stolzing, A.; De Magalhães, J.P. The role of DNA methylation in aging, rejuvenation, and age-related disease. Rejuvenation Res. 2012, 15, 483–494. [Google Scholar] [CrossRef]
- Davies, H.; Bignell, G.R.; Cox, C.; Stephens, P.; Edkins, S.; Clegg, S.; Teague, J.; Woffendin, H.; Garnett, M.J.; Bottomley, W.; et al. Mutations of the BRAF gene in human cancer. Nature 2002, 417, 949–954. [Google Scholar] [CrossRef]
- Bond, C.E.; Whitehall, V.L.J. How the BRAF V600E Mutation Defines a Distinct Subgroup of Colorectal Cancer: Molecular and Clinical Implications. Gastroenterol. Res. Pract. 2018, 2018, 9250757. [Google Scholar] [CrossRef]
- Petermann, K.B.; Rozenberg, G.I.; Zedek, D.; Groben, P.; McKinnon, K.; Buehler, C.; Kim, W.Y.; Shields, J.M.; Penland, S.; Bear, J.E.; et al. CD200 is induced by ERK and is a potential therapeutic target in melanoma. J. Clin. Investig. 2007, 117, 3922–3929. [Google Scholar] [CrossRef]
- Sumimoto, H.; Imabayashi, F.; Iwata, T.; Kawakami, Y. The BRAF-MAPK signaling pathway is essential for cancer-immune evasion in human melanoma cells. J. Exp. Med. 2006, 203, 1651–1656. [Google Scholar] [CrossRef]
- Parsons, M.T.; Buchanan, D.D.; Thompson, B.; Young, J.P.; Spurdle, A.B. Correlation of tumour BRAF mutations and MLH1 methylation with germline mismatch repair (MMR) gene mutation status: A literature review assessing utility of tumour features for MMR variant classification. J. Med. Genet. 2012, 49, 151–157. [Google Scholar] [CrossRef] [PubMed]
- Li, W.Q.; Kawakami, K.; Ruszkiewicz, A.; Bennett, G.; Moore, J.; Iacopetta, B. BRAF mutations are associated with distinctive clinical, pathological and molecular features of colorectal cancer independently of microsatellite instability status. Mol. Cancer 2006, 5, 2. [Google Scholar] [CrossRef] [PubMed]
- Phipps, A.I.; Buchanan, D.D.; Makar, K.W.; Burnett-Hartman, A.N.; Coghill, A.E.; Passarelli, M.N.; Baron, J.A.; Ahnen, D.J.; Win, A.K.; Potter, J.D.; et al. BRAF mutation status and survival after colorectal cancer diagnosis according to patient and tumor characteristics. Cancer Epidemiol. Prev. Biomark. 2012, 21, 1792–1798. [Google Scholar] [CrossRef] [PubMed]
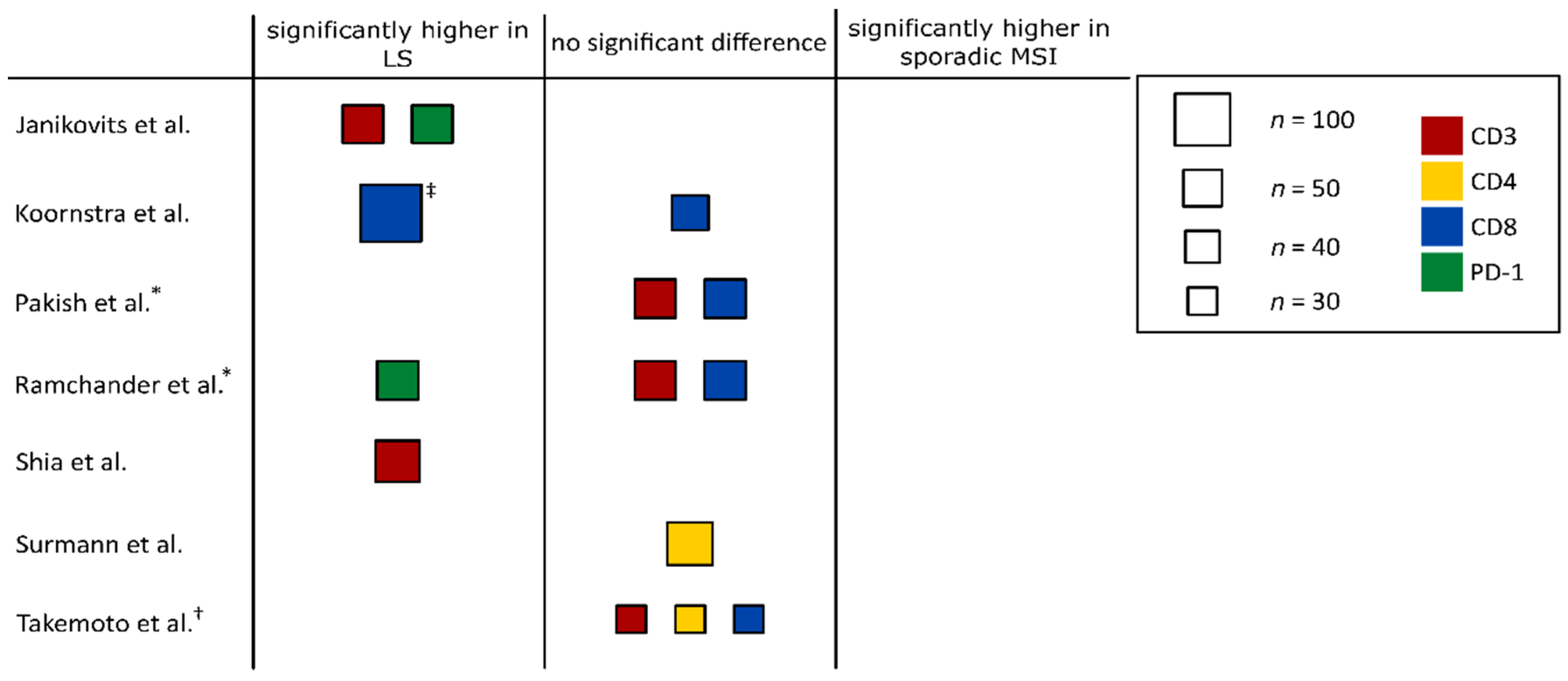
| Sample Number | LS Status | Marker (Clone) | Positive Cell Counts | Analyzed Area | Counting Method | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| Proven | Suspected | |||||||||
| colorectal cancer | Hereditary MSI CRCs | Sporadic MSI CRCs | Hereditary MSI CRCs | Sporadic MSI CRCs | ||||||
| Janikovits et al., 2018 [47] | n = 18 | n = 38 | x | CD3 (PS1) | 143.1 * | 92.5 | tumor epithelial | median of positive cells per 0.25 mm2 | ||
| PD-1 (NAT105) | 31 * | 2.7 | ||||||||
| Koornstra et al., 2009 [50] | n = 20 | n = 26 | x | x | CD8 (na) | 18.6 | 23.9 | tumor epithelial | mean of positive cells in 10 hpf (400 × magnification) | |
| Shia et al., 2003 [48] | n = 30 | n = 35 | x | CD3 (na) | 65 * | 19 | tumor epithelial | median of positive cells in 10 hpf (=1.96 mm2) | ||
| Surmann et al., 2015 [51] | n = 35 | n = 34 | n.a. | CD4 (IF6) | 53.4 | 68.9 | tumor epithelial | median of positive cells per 0.25 mm2 | ||
| CD4 (IF6) | 119.4 | 141.2 | stromal | |||||||
| Takemoto et al., 2004 [49] | n = 19 | n = 12 | x | CD3 (×100) | 165.4 | 153.2 | stromal | mean/median of positive cells per 250 µm2 | ||
| CD4 (×20) | 91.2 | 84.7 | ||||||||
| CD8 (×20) | 51.5 | 47.6 | ||||||||
| no numbers for epithelial cells | ||||||||||
| colorectal adenomas | Koornstra et al., 2009 [50] | Hereditary Colorectal Adenomas | Sporadic Colorectal Adenomas | Hereditary Colorectal Adenomas | Sporadic Colorectal Adenomas | |||||
| n = 50 | n = 69 | x | CD8 (na) | 6.6 * | 3.9 | epithelial | mean of positive cells in 10 hpf (400 × magnification) | |||
| endometrial cancer | hereditary MSI ECs | sporadic MSI ECs | hereditary MSI ECs | sporadic MSI ECs | ||||||
| Pakish et al., 2017 [52] | n = 20 | n = 38 | x | CD3 (SP7) | 84.3 | 84.6 | stromal | median of positive cells per 1 mm2 | ||
| CD4 (4B12) | 16.3 | 22.1 | ||||||||
| CD8 (4B11) | 82.8 * | 34.2 | ||||||||
| PD-L1 (E1L3N) | 289.3 | 297.9 | ||||||||
| CD3 (SP7) | 18.7 | 30 | tumor epithelial | |||||||
| CD4 (4B12) | 2.8 | 8.6 | ||||||||
| CD8 (4B11) | 8.2 | 4.2 | ||||||||
| PD-L1 (E1L3N) | 4.8 | 8.2 | ||||||||
| Ramchander et al., 2020 [53] | n = 25 | n = 33 | x | CD3 (F7.2.38) | 590 | 617 | tumor center | mean of positive cells per 200 µm2 | ||
| CD8 (C8/144B) | 291 | 233 | ||||||||
| CD45RO (UCLH1) | 597 | 653 | ||||||||
| FoxP3 (236A/E7) | 72 | 58 | ||||||||
| PD-1 (NAT105) | 156 * | 108 | ||||||||
| CD3 (F7.2.38) | 386 | 241 | invasive margin | |||||||
| CD8 (C8/144B) | 287 * | 116 | ||||||||
| CD45RO (UCLH1) | 548 * | 296 | ||||||||
| FoxP3 (236A/E7) | 61 | 28 | ||||||||
| PD-1 (NAT105) | 118 * | 49 | ||||||||
| HLA I-B2M Mutations | Colorectal MSI Adenomas | LS Status | ||||
|---|---|---|---|---|---|---|
| Hereditary MSI CRCs | Sporadic MSI CRCs | Proven | Suspected | |||
| Clendenning et al., 2018 [72] | 17.1% (7/41) | 29% (20/69) | 0% (0/42) | x | x | |
| Dierssen et al., 2006 [73] | 20% (15/75) | 3% (1/33) | x | x | ||
| Janikovits et al., 2018 [47] | 50% (9/18) | 26.3% (10/38) | x | x | ||
| Kloor et al., 2007 [12] | 36.4% (16/44) | 15.4% (4/26) | 15.8% (6/38) | x | ||
| Total | 35.9% (47/131) | 26.7% (35/131) | 7.5% (6/80) | |||
| 95% CI | 0.2816 to 0.4440 | 0.1985 to 0.3491 | ||||
| p-value | 0.1425 | |||||
| HLA II-Mutations in CIITA and RFX5 | ||||||
| CIITA | RFX5 | |||||
| Hereditary MSI CRCs | Sporadic MSI CRCs | Hereditary MSI CRCs | Sporadic MSI CRCs | |||
| Surmann et al., 2015 [51] | 8.9% (3/35) | 0% (0/34) | 14.3% (5/35) | 26.5% (9/34) | n.a. | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bohaumilitzky, L.; von Knebel Doeberitz, M.; Kloor, M.; Ahadova, A. Implications of Hereditary Origin on the Immune Phenotype of Mismatch Repair-Deficient Cancers: Systematic Literature Review. J. Clin. Med. 2020, 9, 1741. https://doi.org/10.3390/jcm9061741
Bohaumilitzky L, von Knebel Doeberitz M, Kloor M, Ahadova A. Implications of Hereditary Origin on the Immune Phenotype of Mismatch Repair-Deficient Cancers: Systematic Literature Review. Journal of Clinical Medicine. 2020; 9(6):1741. https://doi.org/10.3390/jcm9061741
Chicago/Turabian StyleBohaumilitzky, Lena, Magnus von Knebel Doeberitz, Matthias Kloor, and Aysel Ahadova. 2020. "Implications of Hereditary Origin on the Immune Phenotype of Mismatch Repair-Deficient Cancers: Systematic Literature Review" Journal of Clinical Medicine 9, no. 6: 1741. https://doi.org/10.3390/jcm9061741
APA StyleBohaumilitzky, L., von Knebel Doeberitz, M., Kloor, M., & Ahadova, A. (2020). Implications of Hereditary Origin on the Immune Phenotype of Mismatch Repair-Deficient Cancers: Systematic Literature Review. Journal of Clinical Medicine, 9(6), 1741. https://doi.org/10.3390/jcm9061741





