Inflammation-Induced Mucosal KYNU Expression Identifies Human Ileal Crohn’s Disease
Abstract
1. Introduction
2. Methods
2.1. Ethical Approval
2.2. Patient and Tissue Sample Characteristics
2.3. Immunohistochemistry
2.4. Primary Cell Isolation of Antigen-Presenting Cells and CD3+ T Cells
2.5. Cultivation and Stimulation of Antigen-Presenting Cells and CD3+ T Cells
2.6. RNA Isolation, cDNA Synthesis and Real-Time Quantitative PCR
2.7. Statistical Analysis
3. Results
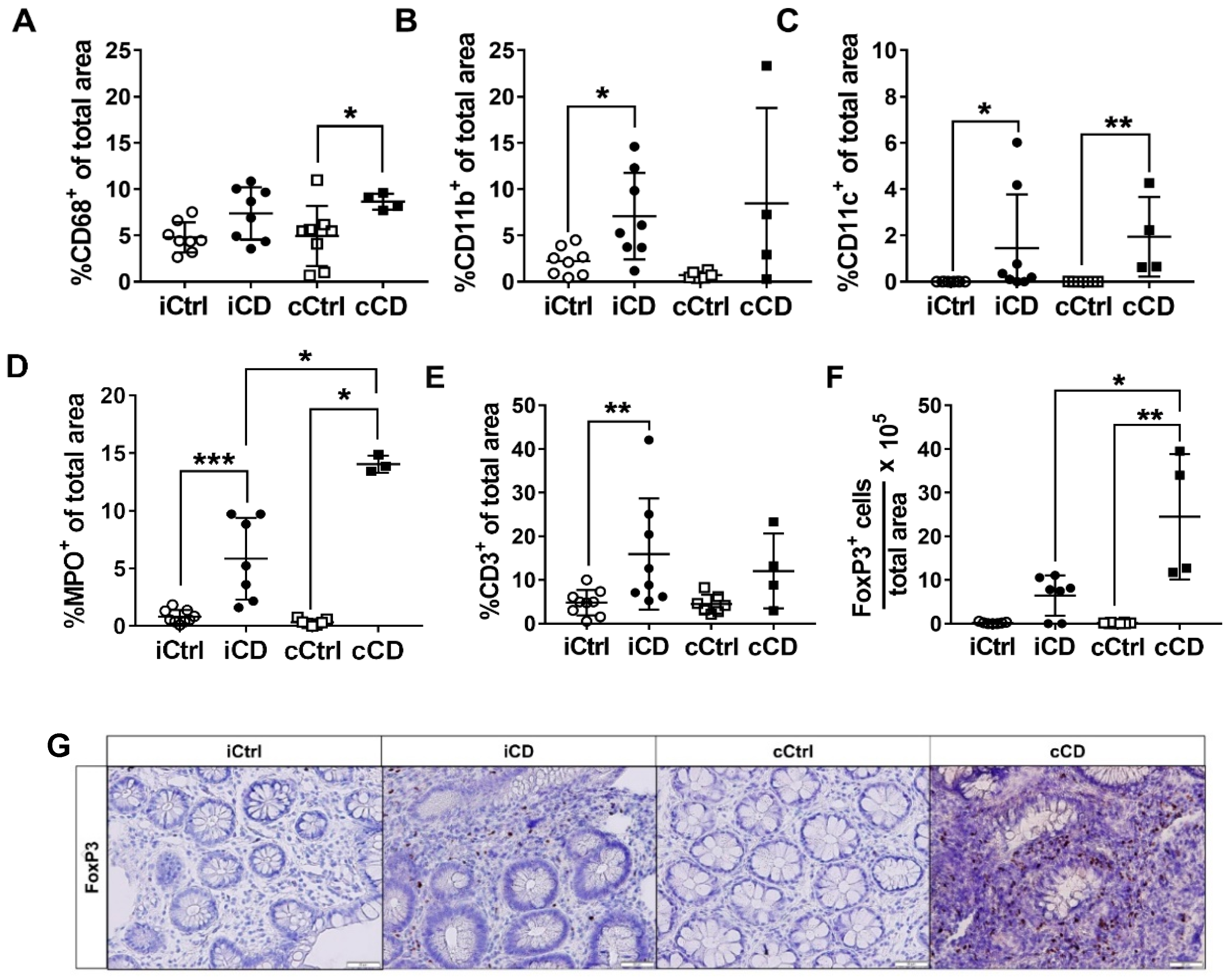
3.1. Local Intestinal Tissue Inflammation in Patients with CD Is Characterized by Immune Cell Infiltrates and Varies According to Anatomic Localization
3.2. Inflammation-Induced IDO1 Expression Is More Pronounced in iCD Compared to cCD
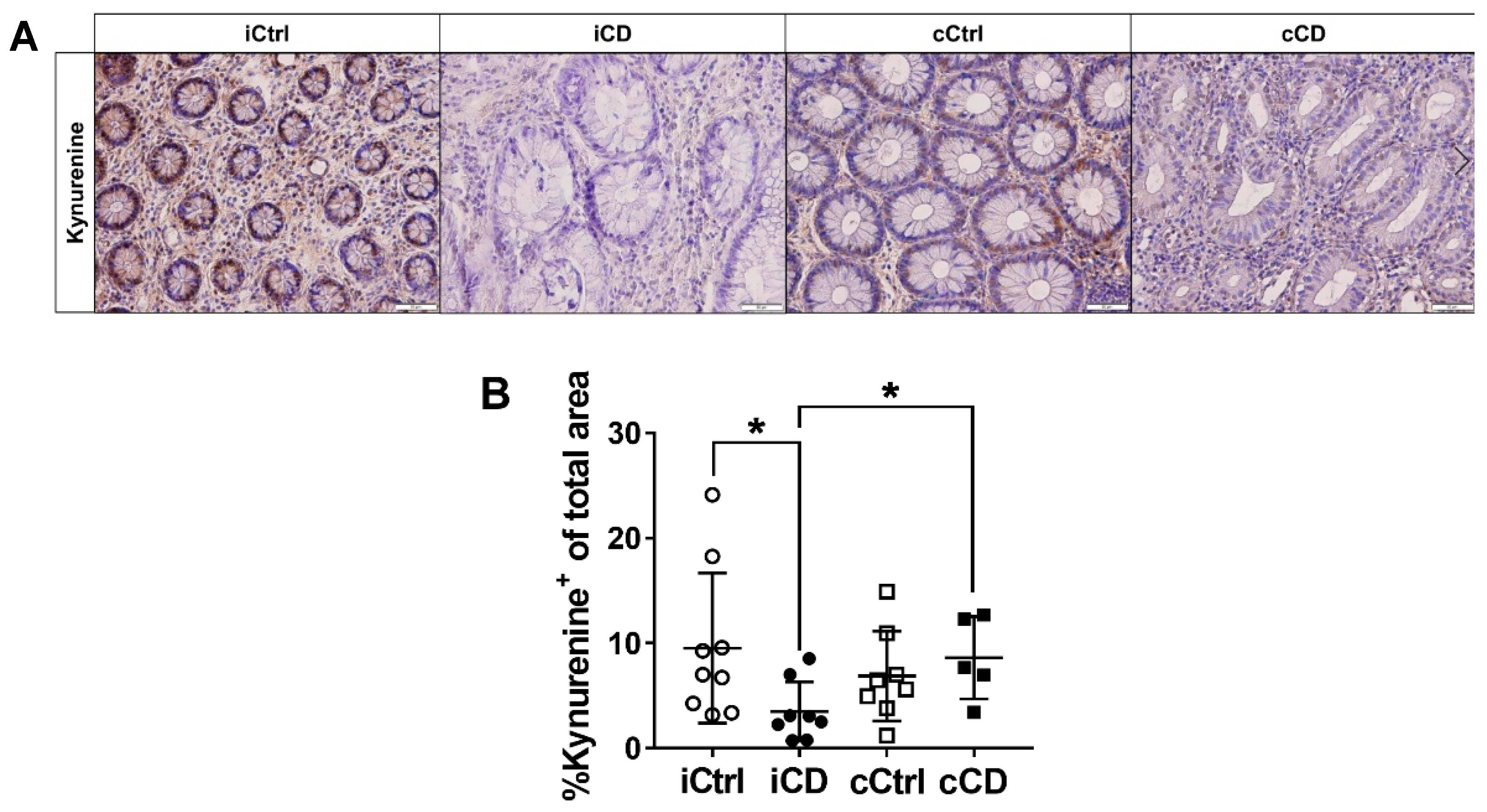
3.3. Despite High Expression of IDO1, Local Kynurenine Levels Are Reduced in Inflamed Ileal Tissue of Patients with CD
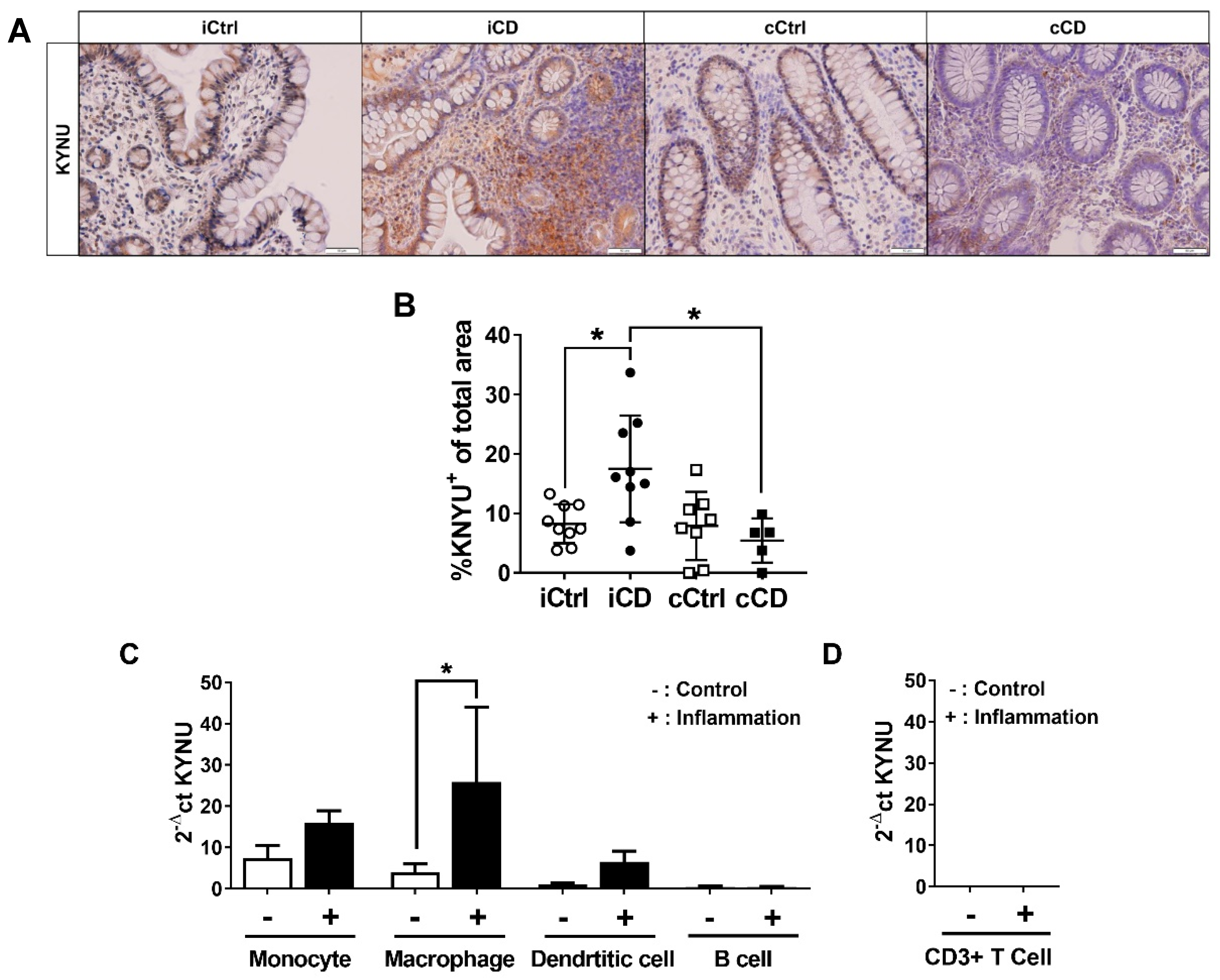
3.4. Expression of Kynureninase Differs between iCD and cCD
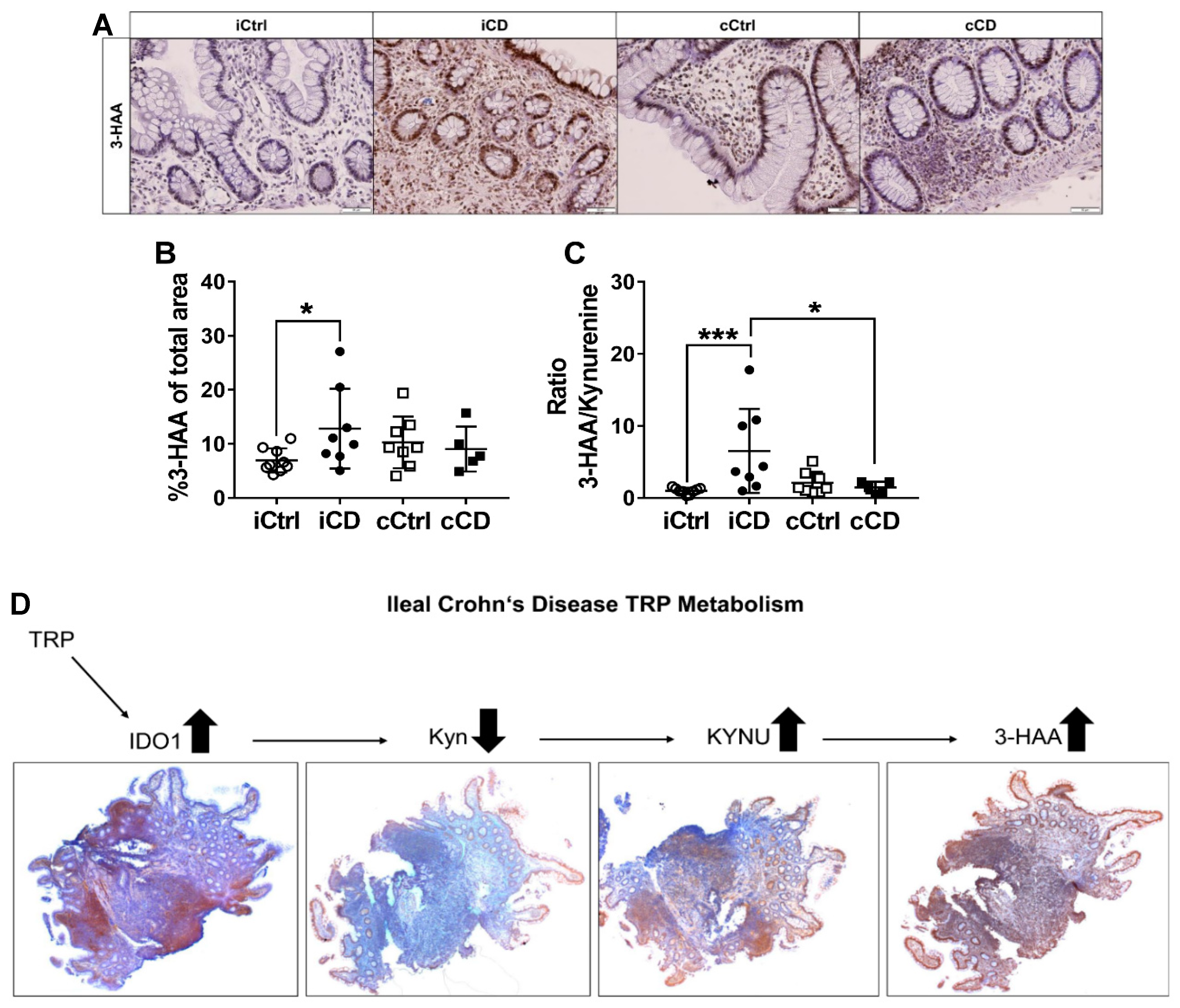
3.5. Kynurenine Downstream Metabolite 3-Hydoxyanthranilic Acid Is Increased in iCD
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Dulai, P.S.; Singh, S.; Vande Casteele, N.; Boland, B.S.; Rivera-Nieves, J.; Ernst, P.B.; Eckmann, L.; Barrett, K.E.; Chang, J.T.; Sandborn, W.J. Should We Divide Crohn’s Disease Into Ileum-Dominant and Isolated Colonic Diseases? Clin. Gastroenterol. Hepatol. 2019, 17, 2634–2643. [Google Scholar] [CrossRef]
- Arijs, I.; Quintens, R.; Van Lommel, L.; Van Steen, K.; De Hertogh, G.; Lemaire, K.; Schraenen, A.; Perrier, C.; Van Assche, G.; Vermeire, S.; et al. Predictive Value of Epithelial Gene Expression Profiles for Response to Infliximab in Crohn’s Disease. Inflamm. Bowel Dis. 2010, 16, 2090–2098. [Google Scholar] [CrossRef]
- Digby-Bell, J.L.; Atreya, R.; Monteleone, G.; Powell, N. Interrogating Host Immunity to Predict Treatment Response in Inflammatory Bowel Disease. Nat. Rev. Gastroenterol. Hepatol. 2020, 17, 9–20. [Google Scholar] [CrossRef]
- Ferrante, M.; Henckaerts, L.; Joossens, M.; Pierik, M.; Joossens, S.; Dotan, N.; Norman, G.L.; Altstock, R.T.; Van Steen, K.; Rutgeerts, P.; et al. New Serological Markers in Inflammatory Bowel Disease Are Associated with Complicated Disease Behaviour. Gut 2007, 56, 1394–1403. [Google Scholar] [CrossRef]
- Hancock, L.; Beckly, J.; Geremia, A.; Cooney, R.; Cummings, F.; Pathan, S.; Guo, C.; Warren, B.F.; Mortensen, N.; Ahmad, T.; et al. Clinical and Molecular Characteristics of Isolated Colonic Crohnʼs Disease. Inflamm. Bowel Dis. 2008, 14, 1667–1677. [Google Scholar] [CrossRef][Green Version]
- Naftali, T.; Reshef, L.; Kovacs, A.; Porat, R.; Amir, I.; Konikoff, F.M.; Gophna, U. Distinct Microbiotas Are Associated with Ileum-Restricted and Colon-Involving Crohn’s Disease. Inflamm. Bowel Dis. 2016, 22, 293–302. [Google Scholar] [CrossRef]
- Ahmad, T.; Armuzzi, A.; Bunce, M.; Mulcahy–Hawes, K.; Marshall, S.E.; Orchard, T.R.; Crawshaw, J.; Large, O.; De Silva, A.; Cook, J.T.; et al. The Molecular Classification of the Clinical Manifestations of Crohn’s Disease. Gastroenterology 2002, 122, 854–866. [Google Scholar] [CrossRef] [PubMed]
- Silverberg, M.S.; Mirea, L.; Bull, S.B.; Murphy, J.E.; Steinhart, A.H.; Greenberg, G.R.; McLeod, R.S.; Cohen, Z.; Wade, J.A.; Siminovitch, K.A. A Population- and Family-Based Study of Canadian Families Reveals Association of HLA DRB1*0103 with Colonic Involvement in Inflammatory Bowel Disease. Inflamm. Bowel Dis. 2003, 9, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Fernandez, L.; Mendoza, J.L.; Martinez, A.; Urcelay, E.; Fernandez-Arquero, M.; Garcia-Paredes, J.; Peña, A.S.; Diaz-Rubio, M.; de la Concha, E.G. IBD1 and IBD3 Determine Location of Crohn’s Disease in the Spanish Population. Inflamm. Bowel Dis. 2004, 10, 715–722. [Google Scholar] [CrossRef]
- Nikolaus, S.; Schulte, B.; Al-Massad, N.; Thieme, F.; Schulte, D.M.; Bethge, J.; Rehman, A.; Tran, F.; Aden, K.; Häsler, R.; et al. Increased Tryptophan Metabolism Is Associated With Activity of Inflammatory Bowel Diseases. Gastroenterology 2017, 153, 1504–1516.e2. [Google Scholar] [CrossRef] [PubMed]
- Gupta, N.K.; Thaker, A.I.; Kanuri, N.; Riehl, T.E.; Rowley, C.W.; Stenson, W.F.; Ciorba, M.A. Serum Analysis of Tryptophan Catabolism Pathway: Correlation with Crohn’s Disease Activity. Inflamm. Bowel Dis. 2012, 18, 1214–1220. [Google Scholar] [CrossRef] [PubMed]
- Matteoli, G.; Mazzini, E.; Iliev, I.D.; Mileti, E.; Fallarino, F.; Puccetti, P.; Chieppa, M.; Rescigno, M. Gut CD103+ Dendritic Cells Express Indoleamine 2,3-Dioxygenase Which Influences T Regulatory/T Effector Cell Balance and Oral Tolerance Induction. Gut 2010, 59, 595–604. [Google Scholar] [CrossRef] [PubMed]
- Wolf, A.M.; Wolf, D.; Rumpold, H.; Moschen, A.R.; Kaser, A.; Obrist, P.; Fuchs, D.; Brandacher, G.; Winkler, C.; Geboes, K.; et al. Overexpression of Indoleamine 2,3-Dioxygenase in Human Inflammatory Bowel Disease. Clin. Immunol. 2004, 113, 47–55. [Google Scholar] [CrossRef] [PubMed]
- Hubbard, T.D.; Murray, I.A.; Perdew, G.H. Indole and Tryptophan Metabolism: Endogenous and Dietary Routes to Ah Receptor Activation. Drug Metab. Dispos. 2015, 43, 1522–1535. [Google Scholar] [CrossRef]
- Hao, N.; Whitelaw, M.L. The Emerging Roles of AhR in Physiology and Immunity. Biochem. Pharmacol. 2013, 86, 561–570. [Google Scholar] [CrossRef]
- Grohmann, U.; Fallarino, F.; Puccetti, P. Tolerance, DCs and tryptophan: Much ado about IDO. Trends Immunol. 2003, 24, 242–248. [Google Scholar] [CrossRef]
- Platten, M.; von Knebel Doeberitz, N.; Oezen, I.; Wick, W.; Ochs, K. Cancer Immunotherapy by Targeting IDO1/TDO and Their Downstream Effectors. Front. Immunol. 2015, 5, 673. [Google Scholar] [CrossRef]
- Liu, Y.; Liang, X.; Dong, W.; Fang, Y.; Lv, J.; Zhang, T.; Fiskesund, R.; Xie, J.; Liu, J.; Yin, X.; et al. Tumor-Repopulating Cells Induce PD-1 Expression in CD8+ T Cells by Transferring Kynurenine and AhR Activation. Cancer Cell 2018, 33, 480–494.e7. [Google Scholar] [CrossRef]
- Fallarino, F.; Grohmann, U.; Hwang, K.W.; Orabona, C.; Vacca, C.; Bianchi, R.; Belladonna, M.L.; Fioretti, M.C.; Alegre, M.-L.; Puccetti, P. Modulation of Tryptophan Catabolism by Regulatory T Cells. Nat. Immunol. 2003, 4, 1206–1212. [Google Scholar] [CrossRef]
- Mezrich, J.D.; Fechner, J.H.; Zhang, X.; Johnson, B.P.; Burlingham, W.J.; Bradfield, C.A. An Interaction between Kynurenine and the Aryl Hydrocarbon Receptor Can Generate Regulatory T Cells. J. Immunol. 2010, 185, 3190–3198. [Google Scholar] [CrossRef]
- Harden, J.L.; Lewis, S.M.; Lish, S.R.; Suárez-Fariñas, M.; Gareau, D.; Lentini, T.; Johnson-Huang, L.M.; Krueger, J.G.; Lowes, M.A. The Tryptophan Metabolism Enzyme L-Kynureninase Is a Novel Inflammatory Factor in Psoriasis and Other Inflammatory Diseases. J. Allergy Clin. Immunol. 2016, 137, 1830–1840. [Google Scholar] [CrossRef] [PubMed]
- Badawy, A.A.B. Kynurenine Pathway and Human Systems. Exp. Gerontol. 2020, 129, 110770. [Google Scholar] [CrossRef]
- Wang, Q.; Ding, Y.; Song, P.; Zhu, H.; Okon, I.; Ding, Y.-N.; Chen, H.-Z.; Liu, D.-P.; Zou, M.-H. Tryptophan-Derived 3-Hydroxyanthranilic Acid Contributes to Angiotensin II-Induced Abdominal Aortic Aneurysm Formation in Mice In Vivo. Circulation 2017, 136, 2271–2283. [Google Scholar] [CrossRef] [PubMed]
- Riley, S.A.; Mani, V.; Goodman, M.J.; Dutt, S.; Herd, M.E. Microscopic Activity in Ulcerative Colitis: What Does It Mean? Gut 1991, 32, 174–178. [Google Scholar] [CrossRef] [PubMed]
- von Knethen, A.; Brüne, B. Delayed Activation of PPARγ by LPS and IFN-γ Attenuates the Oxidative Burst in Macrophages. FASEB J. 2001, 15, 535–544. [Google Scholar]
- Nair, S.; Archer, G.E.; Tedder, T.F. Isolation and Generation of Human Dendritic Cells. Curr. Protoc. Immunol. 2012, 99, 1–23. [Google Scholar] [CrossRef]
- Munn, D.H.; Sharma, M.D.; Lee, J.R.; Jhaver, K.G.; Johnson, T.S.; Keskin, D.B.; Marshall, B.; Chandler, P.; Antonia, S.J.; Burgess, R.; et al. Potential Regulatory Function of Human Dendritic Cells Expressing Indoleamine 2,3-Dioxygenase. Science 2002, 297, 1867–1870. [Google Scholar] [CrossRef]
- Heyes, M.P.; Chen, C.Y.; Major, E.O.; Saito, K. Different Kynurenine Pathway Enzymes Limit Quinolinic Acid Formation by Various Human Cell Types. Biochem. J. 1997, 326, 351–356. [Google Scholar] [CrossRef]
- Murakami, Y.; Saito, K. Species and cell types difference in tryptophan metabolism. Int. J. Tryptophan Res. 2013, 6 (Suppl. 1), 47–54. [Google Scholar] [CrossRef]
- Pfefferkorn, E.R.; Rebhun, S.; Eckel, M. Characterization of an Indoleamine 2,3-Dioxygenase Induced by Gamma-Interferon in Cultured Human Fibroblasts. J. Interferon Res. 1986, 6, 267–279. [Google Scholar] [CrossRef]
- Badawy, A.A.-B. Tryptophan: The Key to Boosting Brain Serotonin Synthesis in Depressive Illness. J. Psychopharmacol. 2013, 27, 878–893. [Google Scholar] [CrossRef] [PubMed]
- Lanis, J.M.; Alexeev, E.E.; Curtis, V.F.; Kitzenberg, D.A.; Kao, D.J.; Battista, K.D.; Gerich, M.E.; Glover, L.E.; Kominsky, D.J.; Colgan, S.P. Tryptophan Metabolite Activation of the Aryl Hydrocarbon Receptor Regulates IL-10 Receptor Expression on Intestinal Epithelia. Mucosal Immunol. 2017, 10, 1133–1144. [Google Scholar] [CrossRef] [PubMed]
- Giorgini, F.; Huang, S.Y.; Sathyasaikumar, K.V.; Notarangelo, F.M.; Thomas, M.A.R.; Tararina, M.; Wu, H.Q.; Schwarcz, R.; Muchowski, P.J. Targeted Deletion of Kynurenine 3-Monooxygenase in Mice a New Tool for Studying Kynurenine Pathway Metabolism in Periphery and Brain. J. Biol. Chem. 2013, 288, 36554–36566. [Google Scholar] [CrossRef] [PubMed]
- Weber, W.P.; Feder-Mengus, C.; Chiarugi, A.; Rosenthal, R.; Reschner, A.; Schumacher, R.; Zajac, P.; Misteli, H.; Frey, D.M.; Oertli, D.; et al. Differential Effects of the Trytophan Metabolite 3-Hydroxyanthranilic Acid on the Proliferation of Human CD8 T Cells Induced by TCR Triggering or Homeostatic Cytokines. Eur. J. Immunol. 2006, 36, 296–304. [Google Scholar] [CrossRef] [PubMed]
- Lee, W.-S.; Lee, S.-M.; Kim, M.-K.; Park, S.-G.; Choi, I.-W.; Choi, I.; Joo, Y.-D.; Park, S.-J.; Kang, S.-W.; Seo, S.-K. The Tryptophan Metabolite 3-Hydroxyanthranilic Acid Suppresses T Cell Responses by Inhibiting Dendritic Cell Activation. Int. Immunopharmacol. 2013, 17, 721–726. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.-M.; Lee, Y.-S.; Choi, J.-H.; Park, S.-G.; Choi, I.-W.; Joo, Y.-D.; Lee, W.-S.; Lee, J.-N.; Choi, I.; Seo, S.-K. Tryptophan Metabolite 3-Hydroxyanthranilic Acid Selectively Induces Activated T Cell Death via Intracellular GSH Depletion. Immunol. Lett. 2010, 132, 53–60. [Google Scholar] [CrossRef]
- Pawlak, K.; Kowalewska, A.; Mysliwiec, M.; Pawlak, D. 3-Hydroxyanthranilic Acid Is Independently Associated with Monocyte Chemoattractant Protein-1 (CCL2) and Macrophage Inflammatory Protein-1beta (CCL4) in Patients with Chronic Kidney Disease. Clin. Biochem. 2010, 43, 1101–1106. [Google Scholar] [CrossRef]

| Control (Ctrl) | Crohn’s Disease (CD) | Ulcerative Colitis (UC) | |
|---|---|---|---|
| n | 12 | 12 | 11 |
| Age (year, average ± SD) | 31 ± 11 | 41 ± 17 | 45 ± 15 |
| Gender (f/m) | 8/4 | 6/6 | 7/4 |
| Year of diagnosis | |||
| 2015 | - | 11 | 9 |
| 2016 | - | 1 | 2 |
| Localization (number of samples) | |||
| small intestine | 9 | 9 | - |
| large intestine | 12 | 5 | 11 |
| Histological Score | |||
| Riley score [22] (mean, range) | |||
| total | 0 | 3 (2–4) | 2.3 (2–4) |
| small intestine | 0 | 3.2 (2–4) | - |
| large intestine | 0 | 2.8 (2–4) | 2.3 (2–4) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Huhn, M.; Herrero San Juan, M.; Melcher, B.; Dreis, C.; Schmidt, K.G.; Schwiebs, A.; Collins, J.; Pfeilschifter, J.M.; Vieth, M.; Stein, J.; et al. Inflammation-Induced Mucosal KYNU Expression Identifies Human Ileal Crohn’s Disease. J. Clin. Med. 2020, 9, 1360. https://doi.org/10.3390/jcm9051360
Huhn M, Herrero San Juan M, Melcher B, Dreis C, Schmidt KG, Schwiebs A, Collins J, Pfeilschifter JM, Vieth M, Stein J, et al. Inflammation-Induced Mucosal KYNU Expression Identifies Human Ileal Crohn’s Disease. Journal of Clinical Medicine. 2020; 9(5):1360. https://doi.org/10.3390/jcm9051360
Chicago/Turabian StyleHuhn, Meik, Martina Herrero San Juan, Balint Melcher, Caroline Dreis, Katrin G. Schmidt, Anja Schwiebs, Janet Collins, Josef M. Pfeilschifter, Michael Vieth, Jürgen Stein, and et al. 2020. "Inflammation-Induced Mucosal KYNU Expression Identifies Human Ileal Crohn’s Disease" Journal of Clinical Medicine 9, no. 5: 1360. https://doi.org/10.3390/jcm9051360
APA StyleHuhn, M., Herrero San Juan, M., Melcher, B., Dreis, C., Schmidt, K. G., Schwiebs, A., Collins, J., Pfeilschifter, J. M., Vieth, M., Stein, J., & Radeke, H. H. (2020). Inflammation-Induced Mucosal KYNU Expression Identifies Human Ileal Crohn’s Disease. Journal of Clinical Medicine, 9(5), 1360. https://doi.org/10.3390/jcm9051360





