Physical Activity for the Treatment of Chronic Low Back Pain in Elderly Patients: A Systematic Review
Abstract
1. Introduction
2. Materials and Methods
2.1. Eligibility Criteria
2.1.1. Study Inclusion Criteria
- Peer-reviewed studies of each level of evidence according to Oxford Classification. We included randomized clinical trials (RCT) and non-randomized controlled studies (NRCT) designs such as observational studies (OS), pre-post interventional studies (PPIS), and case-series studies (CS). We excluded case reports, technical notes, letters to editors, instructional courses, in vitro studies, cadaver investigation, systematic reviews, and meta-analyses.
- Studies including elderly patients (mean age > 65 years) suffering by CLBP (at least > 3 months).
- Clinical outcomes (disability and pain) of patients treated with PA (cardiovascular or aerobic) or exercise programs that included loaded (against gravity or resistance) as a component. To define a study as eligible, it had to include at least one pain assessment or one disability assessment. The disability outcome needed to be evaluated by one or more of the following scales: 36-Item Short Form Health Survey (SF-36) Version 1.0 and 2.0 (SF-36); Roland Morris Disability Questionnaire (RMDQ); Oswestry Disability Index (ODI); and Back function (FFBH-R) [27]. The pain outcome had to be evaluated by one or more of the following scales: Numerical pain rating scale (NRS); Global Rating Change (GRC); Patient Pain Questionnaire (PPQ); and Visual rating scale (VRS).
- Only articles written in English and Italian languages were included.
2.1.2. Study Exclusion Criteria
- Studies with a mean age of patients < 65 years old;
- Studies in which PA was a part of a multidisciplinary program;
- Studies including participants who had physical problems that did not allow them to perform PA (diabetes untreated, muscle-skeletal problems, postural problems, neurological diseases, cardiovascular conditions).
2.2. Search Protocol
2.3. Study Selection
2.4. Data Extraction
2.5. Quality of Evidence
- High Quality of Evidence: among 75% of articles included are considered with low risk bias. Further researches are useful to change either the estimate or confidence in results.
- Moderate Quality of Evidence: one of the GRADE domains is not met. Further studies are required to improve the quality of the study and the evidence.
- Low Quality of Evidence: two of the GRADE domains are not met. Further research is very important.
- Very Low Quality of Evidence: three of the GRADE domains are not met. The results of the study are very uncertain. In the case of studies with a sample size inferior to 300 subjects the quality of the study is considered very low if there was also a high risk of bias (assessed with different tools. In our study we used Rob2 and ROBINS-I).
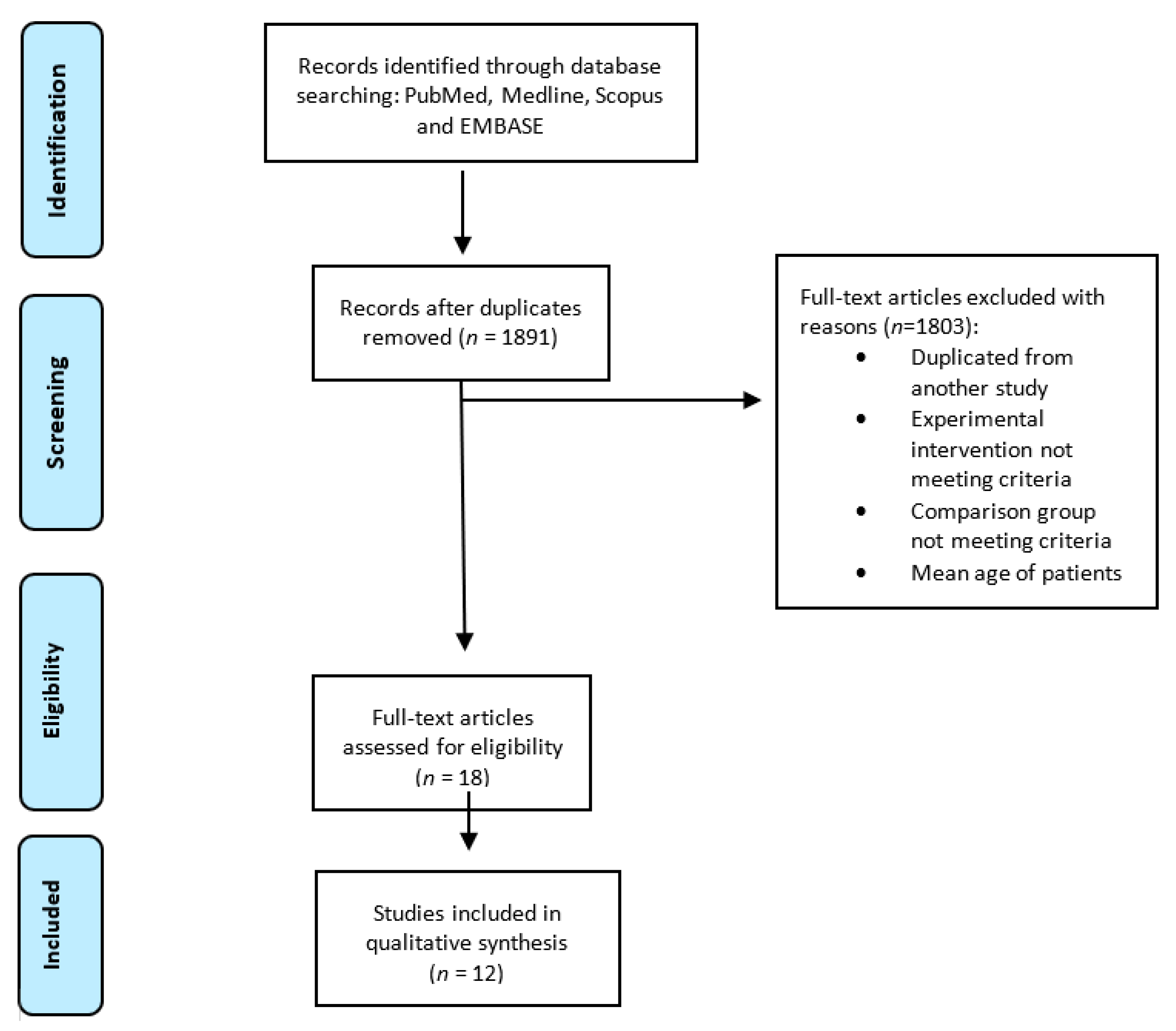
3. Results
3.1. Study Selection
3.2. Study Characteristics
3.3. Methodological Quality
3.4. Results of Individual Studies
3.4.1. Randomized Controlled Trials (RCTs)
Outcome: Pain
Outcome: Disability
3.4.2. Non-Randomized Controlled Trials (NRCT)
Outcome: Pain
Outcome: Disability
3.4.3. Other Studies (Pre-Post Intervention and Case Series)
Outcome: Disability
4. Discussion
Limitations
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Burton, A.K.; Balagué, F.; Cardon, G.; Eriksen, H.R.; Henrotin, Y.; Lahad, A.; Leclerc, A.; Müller, G.; van der Beek, A.J. Chapter 2 European guidelines for prevention in low back pain. Eur. Spine J. 2006, 15, s136–s168. [Google Scholar] [CrossRef] [PubMed]
- Wong, A.Y.; Karppinen, J.; Samartzis, D. Low back pain in older adults: Risk factors, management options and future directions. Scoliosis Spinal Disord. 2017, 12, 14. [Google Scholar] [CrossRef] [PubMed]
- Dunn, K.M.; Hestbaek, L.; Cassidy, J.D. Low back pain across the life course. Best Pract. Res. Clin. Rheumatol. 2013, 27, 591–600. [Google Scholar] [CrossRef] [PubMed]
- Cannata, F.; Vadalà, G.; Ambrosio, L.; Fallucca, S.; Napoli, N.; Papalia, R.; Pozzilli, P.; Denaro, V. Intervertebral disc degeneration: A focus on obesity and type 2 diabetes. Diabetes Metab. Res. Rev. 2020, 36, e3224. [Google Scholar] [CrossRef]
- Hoy, D.; Bain, C.; Williams, G.; March, L.; Brooks, P.; Blyth, F.; Woolf, A.; Vos, T.; Buchbinder, R.A. Systematic review of the global prevalence of low back pain. Arthritis Rheum. 2012, 64, 2028–2037. [Google Scholar] [CrossRef]
- Hartvigsen, J.; Hancock, M.J.; Kongsted, A.; Louw, Q.; Ferreira, M.L.; Genevay, S.; Hoy, D.; Karppinen, J.; Pransky, G.; Sieper, J.; et al. What low back pain is and why we need to pay attention. Lancet 2018, 391, 2356–2367. [Google Scholar] [CrossRef]
- Meucci, R.D.; Fassa, A.G.; Faria, N.M.X. Prevalence of chronic low back pain: Systematic review. Rev. Saúde Pública 2015, 49, 73. [Google Scholar] [CrossRef]
- Vadalà, G.; Russo, F.; Battisti, S.; Stellato, L.; Martina, F.; Del Vescovo, R.; Giacalone, A.; Borthakur, A.; Zobel, B.B.; Denaro, V. Early intervertebral disc degeneration changes in asymptomatic weightlifters assessed by t1ρ-magnetic resonance imaging. Spine 2014, 39, 1881–1886. [Google Scholar] [CrossRef]
- Gallagher, R.M. Low back pain, health status, and quality of life in older adults: Challenge and opportunity. Pain. Med. 2003, 4, 305–307. [Google Scholar] [CrossRef][Green Version]
- Deyo, R.A.; Weinstein, J.N. Low back pain. N. Engl. J. Med. 2001, 344, 363–370. [Google Scholar] [CrossRef]
- Vadalà, G.; Russo, F.; Musumeci, M.; D’Este, M.; Cattani, C.; Catanzaro, G.; Tirindelli, M.C.; Lazzari, L.; Alini, M.; Giordano, R.; et al. Clinically relevant hydrogel-based on hyaluronic acid and platelet rich plasma as a carrier for mesenchymal stem cells: Rheological and biological characterization. J. Orthop. Res. 2017, 35, 2109–2116. [Google Scholar] [CrossRef] [PubMed]
- Hubert, M.G.; Vadala, G.; Sowa, G.; Studer, R.K.; Kang, J.D. Gene therapy for the treatment of degenerative disk disease. J. Am. Acad. Orthop. Surg. 2008, 16, 312–319. [Google Scholar] [CrossRef] [PubMed]
- Maher, C.; Underwood, M.; Buchbinder, R. Non-specific low back pain. Lancet 2017, 389, 736–747. [Google Scholar] [CrossRef]
- Cayea, D.; Perera, S.; Weiner, D.K. Chronic low back pain in older adults: What physicians know, what they think they know, and what they should be taught. J. Am. Geriatr. Soc. 2006, 54, 1772–1777. [Google Scholar] [CrossRef]
- Macedo, L.G.; Maher, C.G.; Latimer, J.; McAuley, J.H. Motor control exercise for persistent, nonspecific low back pain: A systematic review. Phys. Ther. 2009, 89, 9–25. [Google Scholar] [CrossRef]
- Scheele, J.; Enthoven, W.T.M.; Bierma-Zeinstra, S.M.A.; Peul, W.C.; van Tulder, M.W.; Bohnen, A.M.; Berger, M.Y.; Koes, B.W.; Luijsterburg, P.A.J. Course and prognosis of older back pain patients in general practice: A prospective cohort study. Pain 2013, 154, 951–957. [Google Scholar] [CrossRef]
- Weiner, D.K.; Rudy, T.E.; Kim, Y.S.; Golla, S. Do medical factors predict disability in older adults with persistent low back pain? Pain 2004, 112, 214–220. [Google Scholar] [CrossRef]
- Paeck, T.; Ferreira, M.L.; Sun, C.; Lin, C.W.C.; Tiedemann, A.; Maher, C.G. Are older adults missing from low back pain clinical trials? A systematic review and meta-analysis. Arthritis Care Res. 2014, 66, 1220–1226. [Google Scholar] [CrossRef]
- Richardson, C.A.; Jull, G.A. Muscle control-pain control. What exercises would you prescribe? Man. Ther. 1995, 1, 2–10. [Google Scholar]
- Airaksinen, O.; Brox, J.I.; Cedraschi, C.; Hildebrandt, J.; Klaber-Moffett, J.; Kovacs, F.; Mannion, A.F.; Reis, S.; Staal, J.B.; Ursin, H.; et al. European guidelines for the management of chronic nonspecific low back pain. Eur. Spine J. 2006, 15, S192–S300. [Google Scholar] [CrossRef]
- Chou, R.; Qaseem, A.; Snow, V.; Casey, D.; Cross, J.T.; Shekelle, P.; Owens, D.K.; Clinical Efficacy Assessment Subcommittee of the American College of Physicians; American College of Physicians; American Pain Society Low Back Pain Guidelines Panel. Diagnosis and treatment of low back pain: A joint clinical practice guideline from the American College of Physicians and the American Pain Society. Ann. Intern. Med. 2007, 147, 478–491. [Google Scholar] [PubMed]
- Hayden, J.A.; van Tulder, M.W.; Malmivaara, A.V.; Koes, B.W. Meta-analysis: Exercise therapy for nonspecific low back pain. Ann. Intern. Med. 2005, 142, 765–775. [Google Scholar] [CrossRef] [PubMed]
- Henschke, N.; Ostelo, R.W.; van Tulder, M.W.; Vlaeyen, J.W.; Morley, S.; Assendelft, W.J.; Main, C.J. Behavioural treatment for chronic low-back pain. Cochrane Database Syst. Rev. 2010, CD002014. [Google Scholar] [CrossRef] [PubMed]
- Ruegsegger, G.N.; Booth, F.W. Health Benefits of Exercise. Cold Spring Harb. Perspect. Med. 2018, 8, a029694. [Google Scholar] [CrossRef]
- van Middelkoop, M.; Rubinstein, S.M.; Verhagen, A.P.; Ostelo, R.W.; Koes, B.W.; van Tulder, M.W. Exercise therapy for chronic nonspecific low-back pain. Best Pract. Res. Clin. Rheumatol. 2010, 24, 193–204. [Google Scholar] [CrossRef]
- Ryan, R.; Hill, S. How to GRADE the quality of the evidence. Cochrane Consum. Commun. Gr 2019, 1–24. [Google Scholar] [CrossRef]
- Longo, U.G.; Loppini, M.; Denaro, L.; Maffulli, N.; Denaro, V. Rating scales for low back pain. Br. Med. Bull. 2010, 94, 81–144. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. Preferred Reporting Items for Systematic Reviews and Meta-Analyses: The PRISMA Statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef]
- Sterne, J.A.C.; Savović, J.; Page, M.J.; Elbers, R.G.; Blencowe, N.S.; Boutron, I.; Cates, C.J.; Cheng, H.Y.; Corbett, M.S.; Eldridge, S.M.; et al. RoB 2: A revised tool for assessing risk of bias in randomised trials. BMJ 2019, 366. [Google Scholar] [CrossRef]
- Sterne, J.A.; Hernán, M.A.; Reeves, B.C.; Savović, J.; Berkman, N.D.; Viswanathan, M.; Henry, D.; Altman, D.G.; Ansari, M.T.; Boutron, I.; et al. ROBINS-I: A tool for assessing risk of bias in non-randomised studies of interventions. BMJ 2016, 355. [Google Scholar] [CrossRef]
- Teut, M.; Knilli, J.; Daus, D.; Roll, S.; Witt, C.M. Qigong or Yoga Versus No Intervention in Older Adults with Chronic Low Back Pain—A Randomized Controlled Trial. J. Pain 2016, 17, 796–805. [Google Scholar] [CrossRef] [PubMed]
- Ferrell, B.A.; Josephson, K.R.; Pollan, A.M.; Loy, S.; Ferrell, B.R. A randomized trial of walking versus physical methods for chronic pain management. Aging Clin. Exp. Res. 1997, 9, 99–105. [Google Scholar] [CrossRef] [PubMed]
- Vincent, H.K.; George, S.Z.; Seay, A.N.; Vincent, K.R.; Hurley, R.W. Resistance Exercise, Disability, and Pain Catastrophizing in Obese Adults with Back Pain. Med. Sci. Sports Exerc. 2014, 46, 1693–1701. [Google Scholar] [CrossRef] [PubMed]
- Hicks, G.E.; Benvenuti, F.; Fiaschi, V.; Lombardi, B.; Segenni, L.; Stuart, M.; Pretzer-Aboff, I.; Gianfranco, G.; Macchi, C. Adherence to a community-based exercise program is a strong predictor of improved back pain status in older adults: An observational study. Clin. J. Pain 2012, 28, 195–203. [Google Scholar] [CrossRef] [PubMed]
- Tsatsakos, G.; Kouli, O.; Michalopoulou, M.; Malliou, P.; Godolias, G. Effect of Physical Activity on Functional Status in Elderly with Chronic Low Back Pain. J. Yoga Phys. Ther. 2014, 4, 2. [Google Scholar]
- Vincent, H.K.; Vincent, K.R.; Seay, A.N.; Conrad, B.P.; Hurley, R.W.; George, S.Z. Back strength predicts walking improvement in obese, older adults with chronic low back pain. PMR 2014, 6, 418–426. [Google Scholar] [CrossRef]
- Khalil, T.M.; Abdel-Moty, E.; Diaz, E.L.; Steele-Rosomoff, R.; Rosomoff, H.L. Efficacy of physical restoration in the elderly. Exp. Aging Res. 1994, 20, 189–199. [Google Scholar] [CrossRef]
- Mailloux, J.; Finno, M.; Rainville, J. Long-term exercise adherence in the elderly with chronic low back pain. Am. J. Phys. Med. Rehabil. 2006, 85, 120–126. [Google Scholar] [CrossRef]
- Beissner, K.; Parker, S.; Henderson, C.R., Jr.; Pal, A.; Papaleontiou, M.; Reid, M.C. Implementing a Combined Cognitive-Behavioral + Exercise Therapy Protocol for Use by Older Adults with Chronic Back Pain: Evidence for a Possible Race/Ethnicity Effect. J. Aging Phys. Act. 2012, 16, 246–265. [Google Scholar] [CrossRef]
- Iversen, M.D.; Fossel, A.H.; Katz, J.N. Enhancing function in older adults with chronic low back pain: A pilot study of endurance training. Arch. Phys. Med. Rehabil. 2003, 84, 1324–1331. [Google Scholar] [CrossRef]
- Costantino, C.; Romiti, D. Effectiveness of Back School program versus hydrotherapy in elderly patients with chronic non-specific low back pain: A randomized clinical trial. Acta Biomedica 2014, 85, 52–61. [Google Scholar] [PubMed]
- Holmes, B.; Leggett, S.; Mooney, V.; Nichols, J.; Negri, S.; Hoeyberghs, A. Comparison of female geriatric lumbar-extension strength: Asymptotic versus chronic low back pain patients and their response to active rehabilitation. J. Spinal Disord. 1996, 9, 17–22. [Google Scholar] [CrossRef] [PubMed]
- Di Martino, A.; Russo, F.; Denaro, L.; Denaro, V. How to treat lumbar disc herniation in pregnancy? A systematic review on current standards. Eur. Spine J. 2017, 26, 496–504. [Google Scholar] [CrossRef] [PubMed]
- Di Martino, A.; Papapietro, N.; Lanotte, A.; Russo, F.; Vadalà, G.; Denaro, V. Spondylodiscitis: Standards of current treatment. Curr. Med. Res. Opin. 2012, 28, 689–699. [Google Scholar] [CrossRef]
- Vitoula, K.; Venneri, A.; Varrassi, G.; Paladini, A.; Sykioti, P.; Adewusi, J.; Zis, P. Behavioral Therapy Approaches for the Management of Low Back Pain: An Up-To-Date Systematic Review. Pain Ther. 2018, 7, 1–12. [Google Scholar] [CrossRef]
- Vadalà, G.; Russo, F.; Ambrosio, L.; Papalia, R.; Denaro, V. Mesenchymal stem cells for intervertebral disc regeneration. J. Biol. Regul. Homeost. Agents 2016, 30, 173–179. [Google Scholar]
- Russo, F.; Hartman, R.A.; Bell, K.M.; Vo, N.; Sowa, G.A.; Kang, J.D.; Vadalà, G.; Denaro, V. Biomechanical Evaluation of Transpedicular Nucleotomy With Intact Annulus Fibrosus. Spine 2017, 42, E193–E201. [Google Scholar] [CrossRef]
- Papalia, R.; Zampogna, B.; Torre, G.; Lanotte, A.; Vasta, S.; Albo, E.; Tecame, A.; Denaro, V. Sarcopenia and its relationship with osteoarthritis: Risk factor or direct consequence? Musculoskelet. Surg. 2014, 98, 9–14. [Google Scholar] [CrossRef]
- Bijlsma, A.Y.; Meskers, C.G.M.; van den Eshof, N.; Westendorp, R.G.; Sipilä, S.; Stenroth, L.; Sillanpää, E.; McPhee, J.S.; Jones, D.A.; Narici, M.V.; et al. Diagnostic criteria for sarcopenia and physical performance. Age 2014, 36, 275–285. [Google Scholar] [CrossRef]
- Morley, J.E. Frailty and sarcopenia in elderly. Wien. Klin. Wochenschr. 2016, 128, 439–445. [Google Scholar] [CrossRef]
- Landi, F.; Marzetti, E.; Martone, A.M.; Bernabei, R.; Onder, G. Exercise as a remedy for sarcopenia. Curr. Opin. Clin. Nutr. Metab. Care 2014, 17, 25–31. [Google Scholar] [CrossRef] [PubMed]
- Colaianni, G.; Cinti, S.; Colucci, S.; Grano, M. Irisin and musculoskeletal health. Ann. NY Acad. Sci. 2017, 1402, 5–9. [Google Scholar] [CrossRef] [PubMed]
- Greenhill, C. Exercise: Osteocalcin in the adaptation to exercise. Nat. Rev. Endocrinol. 2016, 12, 434. [Google Scholar] [CrossRef] [PubMed]
- Briganti, S.I.; Gaspa, G.; Tabacco, G.; Naciu, A.M.; Cesareo, R.; Manfrini, S.; Palermo, A. Irisin as a regulator of bone and glucose metabolism. Minerva Endocrinol. 2018, 43, 489–500. [Google Scholar]
- Korta, P.; Pocheć, E.; Mazur-Biały, A. Irisin as a Multifunctional Protein: Implications for Health and Certain Diseases. Medicina 2019, 55, 485. [Google Scholar] [CrossRef]
- Diaz-Franco, M.C.; Franco-Diaz de Leon, R.; Villafan-Bernal, J.R. Osteocalcin-GPRC6A: An update of its clinical and biological multi-organic interactions (Review). Mol. Med. Rep. 2019, 19, 15–22. [Google Scholar] [CrossRef]
- Karsenty, G. Update on the biology of osteocalcin. Endocr. Pract. 2017, 23, 1270–1274. [Google Scholar] [CrossRef]
- Mera, P.; Laue, K.; Ferron, M.; Confavreux, C.; Wei, J.; Galán-Díez, M.; Lacampagne, A.; Mitchell, S.J.; Mattison, J.A.; Chen, Y.; et al. Osteocalcin Signaling in Myofibers Is Necessary and Sufficient for Optimum Adaptation to Exercise. Cell Metab. 2016, 23, 1078–1092. [Google Scholar] [CrossRef]
- Russo, F.; Ambrosio, L.; Ngo, K.; Vadalà, G.; Denaro, V.; Fan, Y.; Sowa, G.; Kang, J.D.; Vo, N. The Role of Type I Diabetes in Intervertebral Disc Degeneration. Spine 2019, 44, 1177–1185. [Google Scholar] [CrossRef]
- Chahla, S.E.; Frohnert, B.I.; Thomas, W.; Kelly, A.S.; Nathan, B.M.; Polgreen, L.E. Higher daily physical activity is associated with higher osteocalcin levels in adolescents. Prev. Med. Rep. 2015, 2, 568–571. [Google Scholar] [CrossRef][Green Version]
- Castrogiovanni, P.; Trovato, F.M.; Szychlinska, M.A.; Nsir, H.; Imbesi, R.; Musumeci, G. The importance of physical activity in osteoporosis. From the molecular pathways to the clinical evidence. Histol. Histopathol. 2016, 31, 1183–1194. [Google Scholar] [PubMed]
- McMillan, L.B.; Zengin, A.; Ebeling, P.R.; Scott, D. Prescribing Physical Activity for the Prevention and Treatment of Osteoporosis in Older Adults. Healthcare 2017, 5, 85. [Google Scholar] [CrossRef] [PubMed]
- Warburton, D.E.R.; Nicol, C.W.; Bredin, S.S.D. Health benefits of physical activity: The evidence. Can. Med. Assoc. J. 2006, 174, 801–809. [Google Scholar] [CrossRef] [PubMed]
| Unique ID | Randomization process | Deviations From Intended Interventions | Missing Outcome Data | Measurement of the Outcome | Selection of the Reported Result | Overall |
|---|---|---|---|---|---|---|
| Vincent et al. 2014 |  |  |  |  |  |  |
| Vincent et al. 2014 II study |  |  |  |  |  |  |
| Tsatsako et al. 2016 |  |  |  |  |  |  |
| Costantino et al. 2014 |  |  |  |  |  |  |
| Ferrel et al. 1996 |  |  |  |  |  |  |
| Teut et al. 2016 |  |  |  |  |  |  |
| Holmes et al. 1996 |  |  |  |  |  |  |
 : low risk;
: low risk;  : some concern;
: some concern;  : high risk.
: high risk.| Unique ID | D1 | D2 | D3 | D4 | D5 | D6 | D7 | Overall |
|---|---|---|---|---|---|---|---|---|
| Iversen et al. |  |  |  |  |  |  |  |  |
| Beissner et al. |  |  |  |  |  |  |  |  |
| Khalil et al. |  |  |  |  |  |  |  |  |
| Mailloux et al. |  |  |  |  |  |  |  |  |
| Hicks et al. |  |  |  |  |  |  |  |  |
 : Critical;
: Critical;  : Serious;
: Serious;  : Moderate;
: Moderate;  : Low.
: Low.| Certainty Assessment | № of Patients | Effect | Certainty | Comments | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| № of Studies | Study Design | Risk of Bias | Inconsistency | Indirectness | Imprecision | Other Considerations | Physical Activity | NO Intervention | Absolute (95% C.I.) | ||
| Disability RCTs (assessed with: ODI; Scale from: 0% to 100%) | |||||||||||
| 2 [33,35] | randomized trials | not serious | serious | not serious | serious | none | 52 | 46 | MD 1.24% lower (1.94 lower to 0.54 lower), (p = 0.0005 *) | ⊕⊕◯◯ LOW | PA group shows a lower ODI mean value after treatment. It represents a possible positive influence of PA in improving disability |
| Disability RCTs (assessed with: SF-36; Scale from: 0 to 100) | |||||||||||
| 2 [31,32] | randomised trials | not serious | serious | not serious | serious | none | 77 | 67 | MD 2.88 point higher (−3.30 lower to 9.6 higher), (p = 0.36) | ⊕⊕◯◯ LOW | PA group shows a higher SF-36 mean value after treatment. It represents a possible positive influence of PA in improving disability |
| Pain RCT (assessed with: NRS; Scale from: 0 to 10) | |||||||||||
| 1 [36] | randomized trials | not serious | not serious | not serious | serious | none | 35 | 17 | MD 1.73 points lower (3.11 lower to 0.35 lower), (p = 0.01 *) | ⊕⊕⊕◯ MODERATE | PA group shows a lower mean NRS after treatment. It represents a possible positive influence of PA in improving pain |
| Pain NRCT (assessed with: Global Rating Change; Scale from: 1 to 10) | |||||||||||
| 1 [34] | observational studies | serious | not serious | not serious | serious | none | 238 | 154 | MD 1 points lower (1.53 lower to 0.47 lower), (p < 0.001 *) | ⊕⊕◯◯ LOW | PA group shows a lower mean pain value after treatment. It represents a possible positive influence of PA in improving pain |
| Outcomes | Anticipated Absolute Effects * (95% C.I.) | № of Participants (Studies) | Certainty of the Evidence (GRADE) |
|---|---|---|---|
| Risk with PA | |||
| Disability RCTs assessed with: ODI Scale from: 0% to 100% | MD 1.24% lower (1.94 lower to 0.54 lower), (p = 0.0005 *) | 98 (2 RCTs) [33,35] | ⊕⊕◯◯ LOW |
| Disability RCTs assessed with: SF-36 Scale from: 0 to 100 | MD 2.88 point higher (−3.30 lower to 9.6 higher), (p = 0.36) | 144 (2 RCTs) [31,32] | ⊕⊕◯◯ LOW |
| Pain RCT assessed with: NRS Scale from: 0 to 10 | MD 1.73 points lower (3.11 lower to 0.35 lower), (p = 0.01*) | 52 (1 RCT) [36] | ⊕⊕⊕◯ MODERATE |
| Pain NRCT assessed with: Global Rating Change Scale from: 1 to 10 | MD 1 points lower (1.53 lower to 0.47 lower), (p < 0.001 *) | 392 (1 observational study) [34] | ⊕⊕◯◯ LOW |
| Study | Type of Study (LOE) | n. of Patients/Mean Age (y)/ | Exclusion | Inclusion | Type of Intervention | Control Group | Frequency | Outcome Summary | Outcome Measure/Difference Between Groups | Conclusions |
|---|---|---|---|---|---|---|---|---|---|---|
| Mailloux et al. [38] 2006 | CS (IV) | 126/76/48 | Compression fracture within the last 6 months, and lack of cognitive or language skills necessary to complete paper-and-pencil measures. | CLBP | Stretching and endurance | No | 6 weeks 2 session/week 2 hour/session | Disability Pain | ODI T0–T1 value Physical activity group: 28 (17) to 16 (13) (p = 0.01) Control: 38 (17) to 25 (17) (p = 0.01) | The exercise behaviours of older adults with CLBP can increase after an exercise-oriented spine physical therapy. |
| Beissner et al. [39] 2012 | OS (II) | 59/75.57/2 | Not reported | Patients >60 years old; ability to speak English or Spanish; LBP in the past three month, cognitively intact. | Overall fitness: warmup, stretching, endurance exercises, walking, and a cool down | No | 9 weeks 2 session/week | Disability | RMDQ T0–T1 values Physical activity group: 3.12 (0.72) to 7.83 (0.77), (p < 0.001) | The race/ethnicity could have a role in the improvement of CLBP with a conservative treatment |
| Iversen et al. [40] 2003 | PPIS (III) | 26/72/3 | Pain with lumbar flexion; low back surgery in the last year; epidural steroid injection during the last 6 months; currently receiving physical therapy or participating in an exercise training program; other medical problems that limited their function more than LBP | Patients >65 years old; low back, buttock, and/or leg pain exacerbated by passive lumbar extension in standing; and symptoms that last for at least 6 months. | Indoor cycling | No | 3 months 3 session/week | Disability | SF-36 T0–T1 Physical activity group: 7.2 (p = 0.6) | The bicycle program was safe and effective for improving functional status and well-being. |
| Costantino et al. [41] 2014 | RCT (I) | 56/73.46/3 | Musculoskeletal disorders, cardiac diseases; fever or infectious disease; previous spinal surgery, trauma; previous physical therapies in the last three months | Patients >65 and <80 years old; Diagnosis of chronic non-specific low back pain; Chronic low back pain recurrence in the last three months. | Back school Program | Yes: Hydrotherapy | 3 months 2 session/week 1 hour/session | Disability | RMDQ T0–T1 difference Back school: 3.26 ± 1.02, (p < 0.001); Hydrotherapy: 4.96 ± 0.71 (p < 0.001) SF-36 (Version 2.0) T0–T1 difference Back school: 13.30 ± 1.44 (p < 0.001); Hydrotherapy: 14.19 ± 1.98 (p < 0.001) | Back School program and Hydrotherapy could be valid treatment options in the rehabilitation of non-specific CLBP in older people. |
| Ferrel et al. [32] 1997 | RCT (I) | 33/73/1.5 | Unstable cardiovascular or pulmonary diseases, inflammatory arthritis or nerve root compression; psychiatric disease, or alcohol abuse | Age >65 years CLBP, use of analgesic medication; ability to walk independently and able to understand and read English. | Three groups: Group 1: low intensity walking.Group 2: pain education program. Group 3: usual care | Yes: Education programme (one 90- minute session + weekly telephone reinforcement) | 6 weeks 4 session/week 1 hour/session | Pain Disability | SF-36 (Version 1.0) T0–T1 difference Intervention: 58.5 (27.7), (p < 0.001) Control: 43 (16.7), (p < 0.001) PPQ T0–T1 difference Intervention: 28.9 (18.5), (p < 0.001) Control: 57.8 (24,9), (p < 0.001) | Patient education and fitness walking can improve overall pain management and related functional limitations |
| Hicks et al. [34] 2012 | OS (II) | 392/66.8/12 | Unstable angina, hypertension, pulmonary disease, dementia, aphasia, back pain attributable to organic causes, back, presence of 2 or more of the following sign: lower-extremity strength, sensation, or reflexes | LBP > 4 months, capability to rise from a chair and walk, capability to travel to the exercise facility, and limited participation in physical activity at the initiation of the exercise program | Strengthening: abdominal strengthening, thoracolumbar, and scapula retraction in lying or standing position or sitting Stretching: hamstring and calf Endurance: 5–10 minutes walking | No | 12 months 2 session/week 1 hour/session-20–30 steps | Pain Adherence to exercise Performance | GRC T0–T1 difference Physical activity group: 4.6 (2.5) Control: 4.9 (2.7) (p = 0.246) | Patients were able to safely participate in exercise program and back pain improved 12 months later. |
| Holmes et al. [42] 1996 | RCT (I) | 38/68.3/3 | Not reported | CLBP | Flexion and extension cycles of isotonic resistance exercises | Yes: No exercises | 4 weeks, 2 session/week | Pain | VRS T0–T1 values Physical activity group: from 5.3 to 2.1 (p < 0.05) Control: data not reported (p > 0.05) | In many patients lumbar exercises and resistance exercises could improve CLBP |
| Khalil et al. [37] 1992 | OS (II) | 59/68/1 | Not reported | In the active restoration program: Low back pain and a diagnosis of myofascial pain syndrome. In the passive restoration program: weakness of quadriceps and/or tibialis anterior. | Mixed isotonic and isokinetic progressive resistive exercise of muscles | No control group The passive approach was based on the use of functional electric stimulation (FES) as an adjunct treatment to strengthen lower extremity muscles weakened by disuse | 4 weeks 1 session/day | Pain | Pain level 1–10 T0–T1 values Physical activity group: 5.5 to 3.3 (p < 0.01) Control: data not reported | Physical activity can improve symptoms and functional ability of older people that suffer of low back pain. Moreover, FES could be a helpful device in the rehabilitation of weak muscles |
| Teut et al. [31] 2016 | RCT (I) | 176/73/3 | Acute neurological symptoms within the last 3 months, severe organic or psychiatric disease, metastatic bone disease | Adults ≥65 years old, chronic low back pain for at least 6 months | Yoga group:Viniyoga methodQuijong group:"Dantian“ and Nei Yang Gong exercises from the Training System Liu Ya Fei | Yes: No intervention group | Yoga group: 3 months 24 classes 45 minutes/class Quijong group: 3 months 12 classes 90 minutes/class | Disability | SF-36 T0–T1 value Yoga: 36.3 ± 8.7 to 59.47 (C.I. 54.73; 64.21) Qijong: 37.5 ± 7.8 to 61.01 (C.I. 55.88; 66.14) Control: 36.5 ± 9.3 to 61.17 (C.I. 56.32; 66.02), (p = 0.50) FFBH-R T0–T1 value Yoga: 68.7 ± 15.4 to 66.55 (C.I. 62.89; 70.21) Qijong: 70.4 ± 18.7 to 69.23 (C.I. 65.9) | High satisfaction of patients with the yoga and qigong classes, but participation in a 3- or 6-month period of yoga or qigong program did not improve chronic back pain, back function and quality of life. |
| Tsatsakos [35] et al. 2014 | RCT (I) | 80/67.7/1 | Back surgery, Cauda equina syndrome, spondylolisthesis, rheumatoid conditions | Patients >60 years old, of both sexes and with pain in the lumbar region for a period over 12 weeks | 10.000 steps/day performed on a treadmill and during the common life. | Yes: recommendation relaxation, and ergonomic | 1 month 8000 steps/day | Disability | ODI T0–T1 value Physical activity group: 7.56 (3.22) to 8.06 (4.94) Control: 11.77 (5.27) to 10.00 (5.03), p = 0.46) | Walking shows that it has no effect in the functional status of the elderly with CLBP. |
| Vincent et al. [33] 2014 | RCT (I) | 49/67.5/4 | Being wheelchair bound | In men and women, BMI ≥30 kg/m2, LBP for ≥6 months | Resistance exercise intervention (TOTRX) Lumbar extension intervention (LEXT) | Yes: No intervention | TOTRX: 4 months, 3 session/week 15 exercise/session LEXT: 4 months, 3 session/week 2 sets of lumbar | Disability | ODI T0–T1 values TOTXR: 29.4 (11.2) to 18.0 (12.6) LEXT: 28.6 (15.2) to 22.6 (14.2) Control: 24.4 (12.1) to 22.9 (12.4), (p = 0.015) RMDQ | Resistance exercise show improvement in patients walking endurance. Lumbar extension strength in obese older adults with CLBP |
| Vincent et al. [36] 2014 | RCT (I) | 49/68.5/4 | Wheelchair bound, ability to participate in resistance exercise, acute back pain back surgery within the previous two years | CLBP> 6 months and abdominal obesity and free of abnormal cardiovascular responses during electrocardiogram (ECG) screening tests | TOTRX LEXT | Yes: Behavioural advices: strengthening exercise and nutritional choices | TOTRX: 4 months, 3 session/week 15 sets/exercise/session LEXT: 4 months, 3 session/week 2 sets of lumbar exercises- 15 reps/exercise/session | Pain | NRS T1 value TOTXR: 4.3 (1.8) to 2.0 (1.7) LEXTR: 5.0 (1.8) to 3.7 (2.6) Control: 5.2 (2.4) to 4.6 (2.4), (p < 0.006) | Total body resistance exercise (including lumbar extension exercise) was more effective than lumbar extension exercise alone in reducing self-reported disability scores due to back pain |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vadalà, G.; Russo, F.; De Salvatore, S.; Cortina, G.; Albo, E.; Papalia, R.; Denaro, V. Physical Activity for the Treatment of Chronic Low Back Pain in Elderly Patients: A Systematic Review. J. Clin. Med. 2020, 9, 1023. https://doi.org/10.3390/jcm9041023
Vadalà G, Russo F, De Salvatore S, Cortina G, Albo E, Papalia R, Denaro V. Physical Activity for the Treatment of Chronic Low Back Pain in Elderly Patients: A Systematic Review. Journal of Clinical Medicine. 2020; 9(4):1023. https://doi.org/10.3390/jcm9041023
Chicago/Turabian StyleVadalà, Gianluca, Fabrizio Russo, Sergio De Salvatore, Gabriele Cortina, Erika Albo, Rocco Papalia, and Vincenzo Denaro. 2020. "Physical Activity for the Treatment of Chronic Low Back Pain in Elderly Patients: A Systematic Review" Journal of Clinical Medicine 9, no. 4: 1023. https://doi.org/10.3390/jcm9041023
APA StyleVadalà, G., Russo, F., De Salvatore, S., Cortina, G., Albo, E., Papalia, R., & Denaro, V. (2020). Physical Activity for the Treatment of Chronic Low Back Pain in Elderly Patients: A Systematic Review. Journal of Clinical Medicine, 9(4), 1023. https://doi.org/10.3390/jcm9041023








