Immunohistochemical Results of Soft Tissues around a New Implant Healing-Abutment Surface: A Human Study
Abstract
1. Introduction
2. Experimental Section
2.1. Materials and Methods
2.1.1. Study Design and Patient Selection
- -
- Patients between 18 and 75 years
- -
- Patients with no systemic and/or oral health disease
- -
- At least six months of healing after tooth extraction
- -
- Adequate dimension of the attached gingiva (>2 mm) or keratinized tissue at the site selected and adequate residual bone crest
- -
- Patients with poor oral hygiene
- -
- Plaque score (PS) and bleeding on probing (BOP) more than 25% [26]
- -
- Patients with active periodontal disease
- -
- Insufficient bone thickness for implant insertion and bone augmentation procedures
- -
- patients who necessitate immediate implant loading protocols
- -
- Uncontrolled diabetes mellitus
- -
- Immune diseases
- -
- Patients who smoke more than 10 cig/die
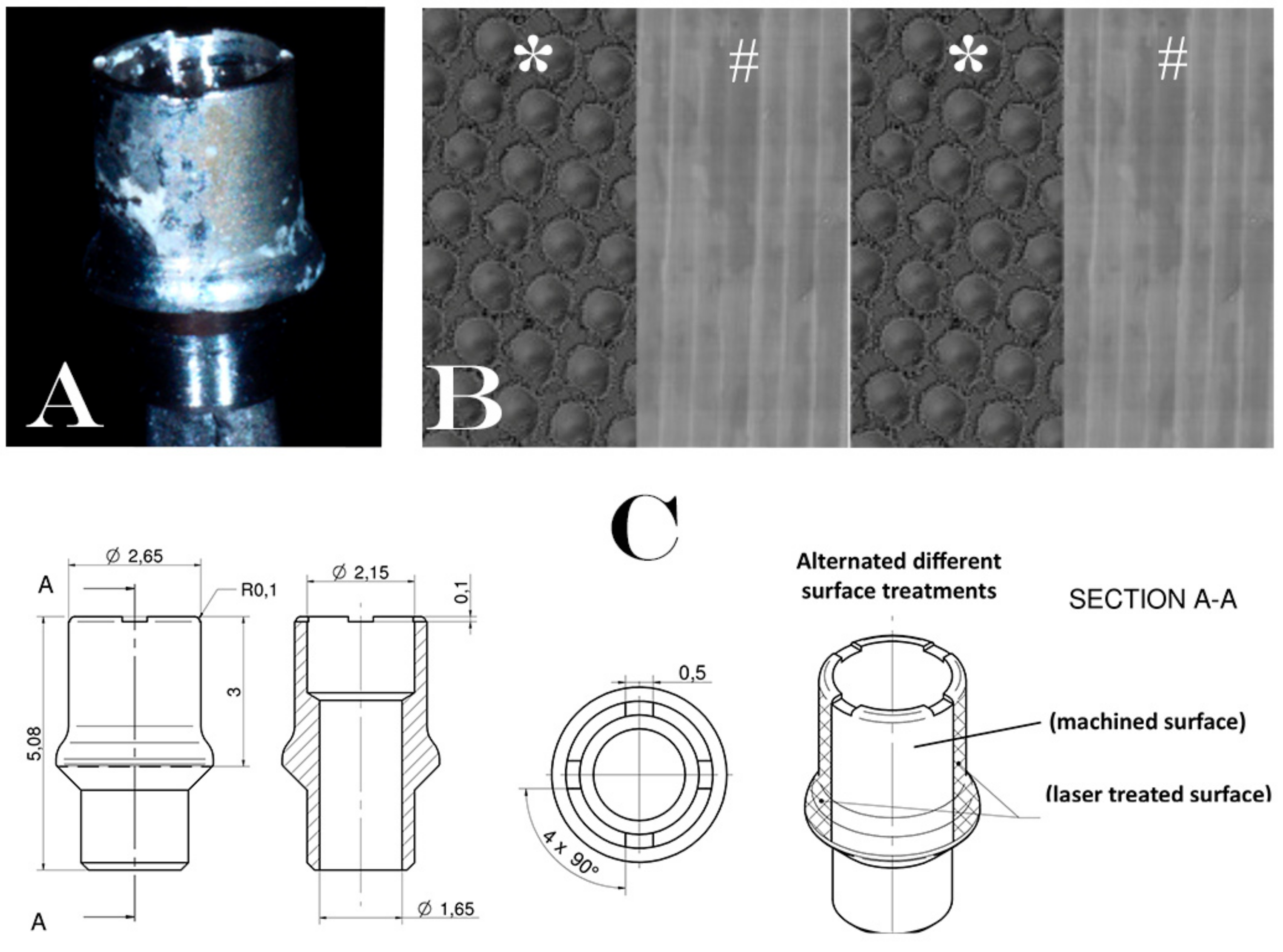
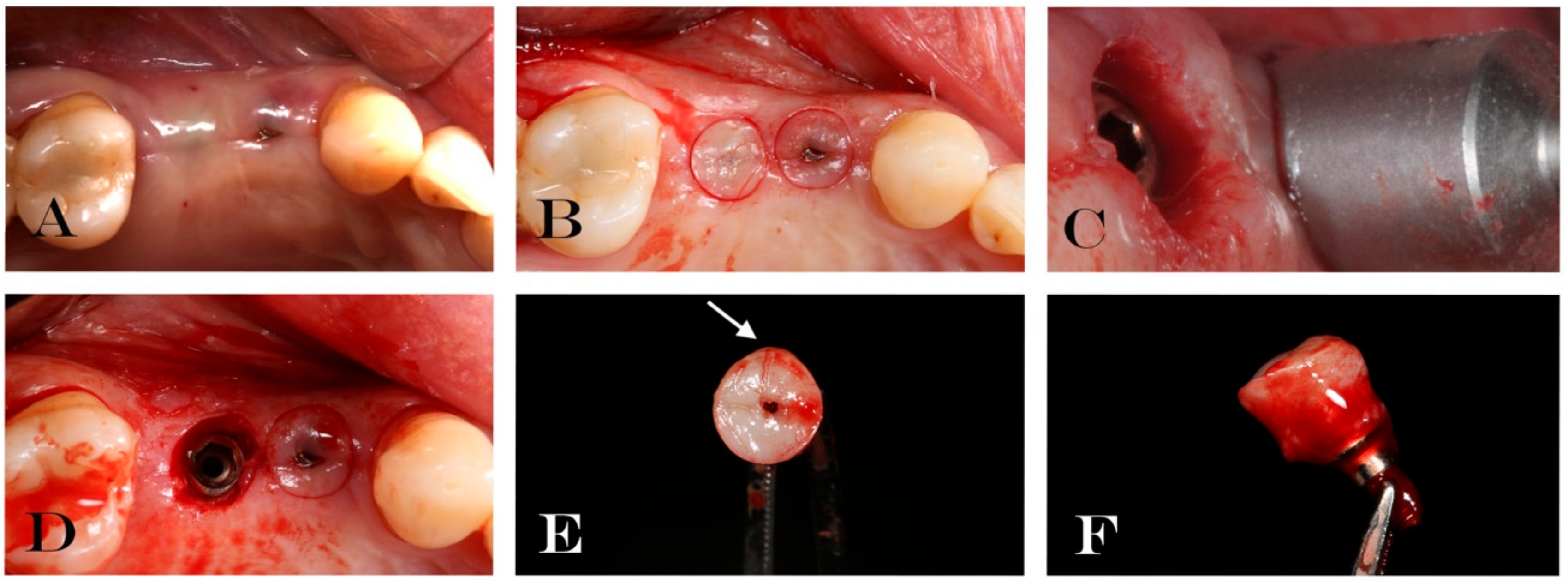
2.1.2. Surgical Treatment
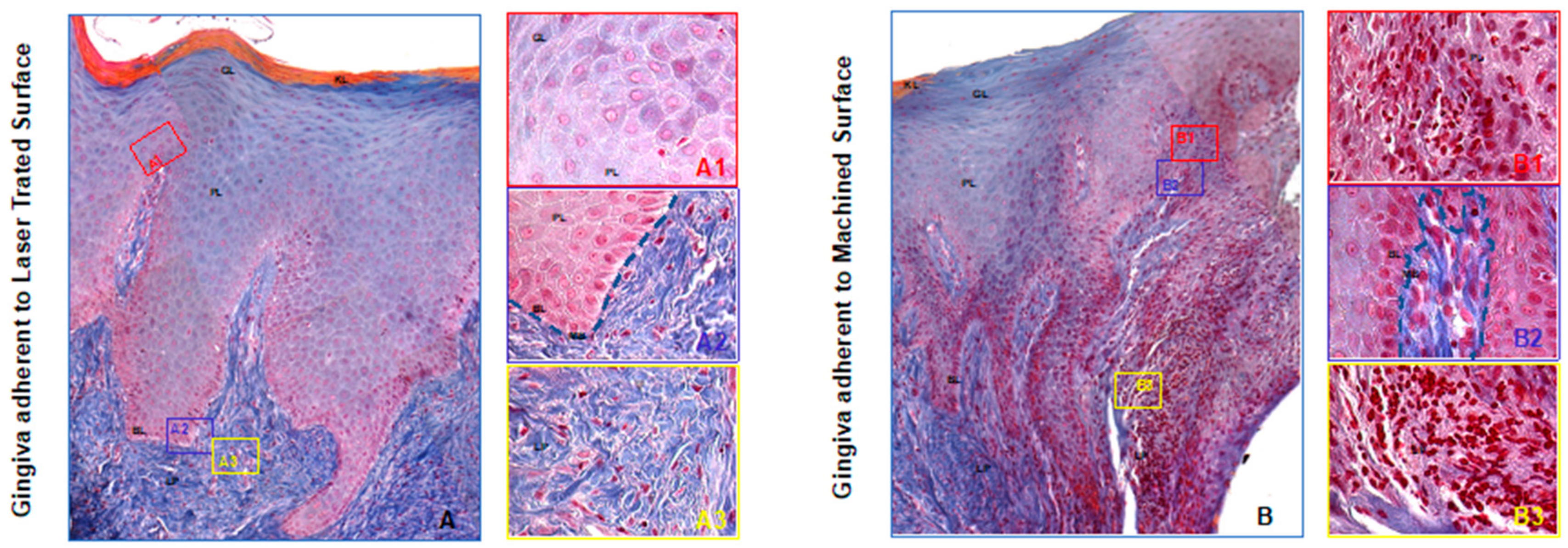
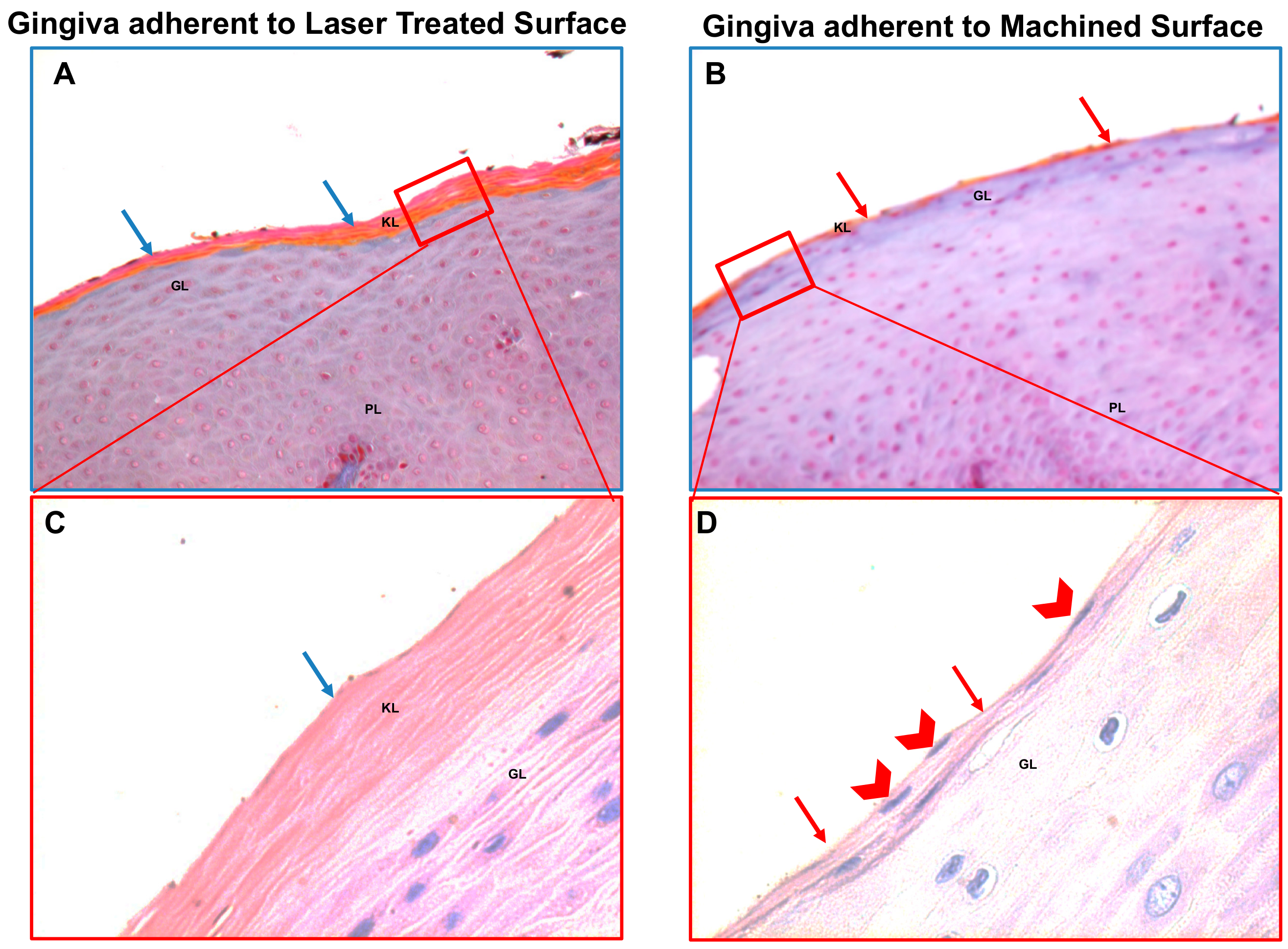
2.1.3. Specimen Retrieval and Analyses
2.1.4. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Simonis, P.; Dufour, T.; Tenenbaum, H. Long-term implant survival and success: A 10–16-year follow-up of non-submerged dental implants. Clin. Oral Implant. Res. 2010, 21, 772–777. [Google Scholar] [CrossRef] [PubMed]
- Moraschini, V.; Poubel, L.A.; Ferreira, V.F.; Barboza Edos, S. Evaluation of survival and success rates of dental implants reported in longitudinal studies with a follow-up period of at least 10 years: A systematic review. Int. J. Oral Maxillofac. Surg. 2015, 44, 377–388. [Google Scholar] [CrossRef] [PubMed]
- Misch, C.E.; Perel, M.L.; Wang, H.L.; Sammartino, G.; Galindo-Moreno, P.; Trisi, P.; Steigmann, M.; Rebaudi, A.; Palti, A.; Pikos, M.A.; et al. Implant success, survival, and failure: The International Congress of Oral Implantologists (ICOI) Pisa Consensus Conference. Implant Dent. 2008, 17, 5–15. [Google Scholar] [CrossRef] [PubMed]
- Berardini, M.; Trisi, P.; Sinjari, B.; Rutjes, A.W.; Caputi, S. The Effects of High Insertion Torque Versus Low Insertion Torque on Marginal Bone Resorption and Implant Failure Rates: A Systematic Review with Meta-Analyses. Implant Dent. 2016, 25, 532–540. [Google Scholar] [CrossRef]
- Caricasulo, R.; Malchiodi, L.; Ghensi, P.; Fantozzi, G.; Cucchi, A. The influence of implant-abutment connection to peri-implant bone loss: A systematic review and meta-analysis. Clin. Implant Dent. Relat. Res. 2018, 20, 653–664. [Google Scholar] [CrossRef]
- Sinjari, B.; D’Addazio, G.; De Tullio, I.; Traini, T.; Caputi, S. Peri-Implant Bone Resorption during Healing Abutment Placement: The Effect of a 0.20% Chlorhexidine Gel vs. Placebo-A Randomized Double Blind Controlled Human Study. Biomed. Res. Int. 2018, 2018. [Google Scholar] [CrossRef]
- D’Ercole, S.; D’Addazio, G.; Di Lodovico, S.; Traini, T.; Di Giulio, M.; Sinjari, B. Porphyromonas Gingivalis Load is Balanced by 0.20% Chlorhexidine Gel. A Randomized, Double-Blind, Controlled, Microbiological and Immunohistochemical Human Study. J. Clin. Med. 2020, 9, 284. [Google Scholar] [CrossRef]
- Levin, L. Dealing with dental implant failures. J. Appl. Oral Sci. 2008, 16, 171–175. [Google Scholar] [CrossRef]
- Schwarz, F.; Derks, J.; Monje, A.; Wang, H.-L. Peri-implantitis. J. Clin. Periodontol. 2018, 45, S246–S266. [Google Scholar] [CrossRef]
- Koldsland, O.C.; Scheie, A.; Aass, A.M. Prevalence of peri-implantitis related to severity of the disease with different degrees of bone loss. J. Periodontol. 2010, 81, 231–238. [Google Scholar] [CrossRef]
- De Avila, E.D.; de Molon, R.S.; Vergani, C.E.; de Assis Mollo, F., Jr.; Salih, V. The Relationship between Biofilm and Physical-Chemical Properties of Implant Abutment Materials for Successful Dental Implants. Materials 2014, 7, 3651–3662. [Google Scholar] [CrossRef] [PubMed]
- Sinjari, B.; D’Addazio, G.; Traini, T.; Varvara, G.; Scarano, A.; Murmura, G.; Caputi, S. A 10-year retrospective comparative human study on screw-retained versus cemented dental implant abutments. J. Biol. Regul. Homeost. Agents 2019, 33, 787–797. [Google Scholar] [PubMed]
- Rompen, E.; Domken, O.; Degidi, M.; Pontes, A.E.; Piattelli, A. The effect of material characteristics, of surface topography and of implant components and connections on soft tissue integration: A literature review. Clin. Oral Implant. Res. 2006, 17, 55–67. [Google Scholar] [CrossRef] [PubMed]
- Gursoy, U.K.; Könönen, E.; Luukkonen, N.; Uitto, V.J. Human neutrophil defensins and their effect on epithelial cells. J. Periodontol. 2013, 84, 126–133. [Google Scholar] [CrossRef]
- Fujita, T.; Yoshimoto, T.; Kajiya, M.; Ouhara, K.; Matsuda, S.; Takemura, T.; Akutagawa, K.; Takeda, K.; Mizuno, N.; Kurihara, H. Regulation of defensive function on gingival epithelial cells can prevent periodontal disease. Jpn. Dent. Sci. Rev. 2018, 54, 66–75. [Google Scholar] [CrossRef]
- Nagarakanti, S.; Ramya, S.; Babu, P.; Arun, K.V.; Sudarsan, S. Differential expression of E-cadherin and cytokeratin 19 and net proliferative rate of gingival keratinocytes in oral epithelium in periodontal health and disease. J. Periodontol. 2007, 78, 2197–2202. [Google Scholar] [CrossRef]
- Jin, C.; Lee, G.; Oh, C.; Kim, H.J.; Kim, H.M. Substrate roughness induces the development of defective E-cadherin junctions in human gingival keratinocytes. J. Periodontal Implant Sci. 2017, 47, 116–131. [Google Scholar] [CrossRef]
- Hartsock, A.; Nelson, W.J. Adherens and tight junctions: Structure, function and connections to the actin cytoskeleton. Biochim. Biophys. Acta 2008, 1778, 660–669. [Google Scholar] [CrossRef]
- Räisänen, L.; Könönen, M.; Juhanoja, J.; Varpavaara, P.; Hautaniemi, J.; Kivilahti, J.; Hormia, M. Expression of cell adhesion complexes in epithelial cells seeded on biomaterial surfaces. J. Biomed. Mater. Res. 2000, 49, 79–87. [Google Scholar] [CrossRef]
- Säuberlich, S.; Klee, D.; Richter, E.J.; Höcker, H.; Spiekermann, H. Cell culture tests for assessing the tolerance of soft tissue to variously modified titanium surfaces. Clin. Oral Implant. Res. 1999, 10, 379–393. [Google Scholar] [CrossRef]
- Artese, L.; Scarano, A.; Perrotti, V.; Gehrke, P.; Piattelli, A. Inflammatory infiltrate, microvessel density, nitric oxide synthase expression, vascular endothelial growth factor expression, and proliferative activity in peri-implant soft tissues around titanium and zirconium oxide healing caps. J. Periodontol. 2006, 77, 73–80. [Google Scholar]
- Mangano, C.; Mangano, F.G.; Shibli, J.A.; Roth, L.A.; d’Addazio, G.; Piattelli, A.; Iezzi, G. Immunohistochemical Evaluation of Peri-Implant Soft Tissues around Machined and Direct Metal Laser Sintered (DMLS) Healing Abutments in Humans. Int. J. Environ. Res. Public Health 2018, 30, 1611. [Google Scholar] [CrossRef] [PubMed]
- Sinjari, B.; Traini, T.; Caputi, S.; Mortellaro, C.; Scarano, A. Evaluation of Fibrin Clot Attachment on Titanium Laser-Conditioned Surface Using Scanning Electron Microscopy. J. Craniofacial Surg. 2018, 29, 2277–2281. [Google Scholar] [CrossRef] [PubMed]
- Di Giulio, M.; Traini, T.; Sinjari, B.; Nostro, A.; Caputi, S.; Cellini, L. Porphyromonas gingivalis biofilm formation in different titanium surfaces, an in vitro study. Clin. Oral Implant. Res. 2016, 27, 918–925. [Google Scholar] [CrossRef]
- Sinjari, B.; Guarnieri, S.; Diomede, F.; Merciaro, I.; Mariggio, M.A.; Caputi, S.; Trubiani, O. Influence of titanium laser surface geometry on proliferation and on morphological features of human mandibular primary osteoblasts. J. Biol. Regul. Homeost. Agents 2012, 26, 505–513. [Google Scholar]
- Löe, H. The Gingival Index, the Plaque Index and the Retention Index Systems. J. Periodontol. 1967, 38, 610–616. [Google Scholar] [CrossRef]
- Femminella, B.; Iaconi, M.C.; Di Tullio, M.; Romano, L.; Sinjari, B.; D’Arcangelo, C.; De Ninis, P.; Paolantonio, M. Clinical Comparison of Platelet-Rich Fibrin and a Gelatin Sponge in the Management of Palatal Wounds after Epithelialized Free Gingival Graft Harvest: A Randomized Clinical Trial. J. Periodontol. 2016, 87, 103–113. [Google Scholar] [CrossRef]
- Migliaccio, A.R.; Martelli, F.; Verrucci, M.; Sanchez, M.; Valeri, M.; Migliaccio, G.; Vannucchi, A.M.; Zingariello, M.; Di Baldassarre, A.; Ghinassi, B.; et al. Gata1 expression driven by the alternative HS2 enhancer in the spleen rescues the hematopoietic failure induced by the hypomorphic Gata1low mutation. Blood 2009, 3, 2107–2120. [Google Scholar] [CrossRef]
- D’Amico, M.A.; Ghinassi, B.; Izzicupo, P.; Di Ruscio, A.; Di Baldassarre, A. IL-6 Activates PI3K and PKCζ Signaling and Determines Cardiac Differentiation in Rat Embryonic H9c2 Cells. J. Cell. Physiol. 2016, 231, 576–586. [Google Scholar] [CrossRef]
- Martelli, F.; Ghinassi, B.; Lorenzini, R.; Vannucchi, A.M.; Rana, R.A.; Nishikawa, M.; Partamian, S.; Migliaccio, G.; Migliaccio, A.R. Thrombopoietin inhibits murine mast cell differentiation. Stem Cells 2008, 26, 912–919. [Google Scholar] [CrossRef][Green Version]
- Scarano, A.; Murmura, G.; Sinjari, B.; Sollazzo, V.; Spinelli, G.; Carinci, F. Analysis and structural examination of screw loosening in oral implants. Int. J. Immunopathol. Pharmacol. 2011, 24, 77–81. [Google Scholar] [CrossRef] [PubMed]
- D’Ercole, S.; Tripodi, D.; Marzo, G.; Bernardi, S.; Continenza, M.A.; Piattelli, A.; Iaculli, F.; Mummolo, S. Micro leakage of bacteria in different implant-abutment assemblies: An in vitro study. J. Appl. Biomater. Funct. Mater. 2015, 13, e174–e180. [Google Scholar] [PubMed]
- Fröjd, V.; Chávez de Paz, L.; Andersson, M.; Wennerberg, A.; Davies, J.R.; Svensäter, G. In situ analysis of multispecies biofilm formation on customized titanium surfaces. Mol. Oral Microbiol. 2011, 26, 241–252. [Google Scholar] [CrossRef] [PubMed]
- Kasten, F.H.; Soileau, K.; Meffert, R.M. Quantitative evaluation of human gingival epithelial cell attachment to implant surfaces in vitro. Int. J. Periodontics Restor. Dent. 1990, 10, 68–79. [Google Scholar]
- Guy, S.C.; McQuade, M.J.; Scheidt, M.J.; McPherson, J.C.; Rossmann, J.A.; Van Dyke, T.E. In vitro attachment of human gingival fibroblasts to endosseous implant materials. J. Periodontol. 1993, 64, 542–546. [Google Scholar] [CrossRef] [PubMed]
- Chai, W.L.; Moharamzadeh, K.; Brook, I.M.; Van Noort, R. A review of histomorphometric analysis techniques for assessing implant-soft tissue interface. Biotech. Histochem. 2011, 86, 242–254. [Google Scholar] [CrossRef] [PubMed]
- Abrahamsson, I.; Zitzmann, N.U.; Berglundh, T.; Linder, E.; Wennerberg, A.; Lindhe, J. The mucosal attachment to titanium implants with different surface characteristics: An experimental study in dogs. J. Clin. Periodontol. 2002, 29, 448–455. [Google Scholar] [CrossRef]
- Kloss, F.R.; Steinmüller-Nethl, D.; Stigler, R.G.; Ennemoser, T.; Rasse, M.; Hächl, O. In vivo investigation on connective tissue healing to polished surfaces with different surface wettability. Clin. Oral Implant. Res. 2011, 22, 699–705. [Google Scholar] [CrossRef]
- Leong, A.; De Kok, I.; Mendonça, D.; Cooper, L.F. Molecular Assessment of Human Peri-implant Mucosal Healing at Laser-Modified and Machined Titanium Abutments. Int. J. Oral Maxillofac. Implant. 2018, 33, 895–904. [Google Scholar] [CrossRef]
- Dellavia, C.; Canullo, L.; Allievi, C.; Lang, N.P.; Pellegrini, G. Soft tissue surrounding switched platform implants: An immunohistochemical evaluation. Clin. Oral Implant. Res. 2013, 24, 63–70. [Google Scholar] [CrossRef]
- Canullo, L.; Pellegrini, G.; Allievi, C.; Trombelli, L.; Annibali, S.; Dellavia, C. Soft tissues around long-term platform switching implant restorations: A histological human evaluation. Preliminary results. J. Clin. Periodontol. 2011, 38, 86–94. [Google Scholar] [CrossRef] [PubMed]
- Perrotti, V.; Zhang, D.; Liang, A.; Wong, J.; Quaranta, A. The Effect of One-Abutment at One-Time on Marginal Bone Loss Around Implants Placed in Healed Bone: A Systematic Review of Human Studies. Implant Dent. 2019, 28, 603–612. [Google Scholar] [CrossRef] [PubMed]
- Canullo, L.; Tallarico, M.; Gracis, S.; Vela, X.; Rodriguez, X.; Covani, U. Clinical Considerations on Strategies That Avoid Multiple Connections and Disconnections of Implant Abutments. Int. J. Periodontics Restor. Dent. 2020, 40, 9–17. [Google Scholar] [CrossRef] [PubMed]
- Nothdurft, F.P.; Fontana, D.; Ruppenthal, S.; May, A.; Aktas, C.; Mehraein, Y.; Lipp, P.; Kaestner, L. Differential Behavior of Fibroblasts and Epithelial Cells on Structured Implant Abutment Materials: A Comparison of Materials and Surface Topographies. Clin. Implant Dent. Relat. Res. 2015, 17, 1237–1249. [Google Scholar] [CrossRef] [PubMed]
- Furuhashi, A.; Ayukawa, Y.; Atsuta, I.; Okawachi, H.; Koyano, K. The difference of fibroblast behavior on titanium substrata with different surface characteristics. Odontology 2012, 100, 199–205. [Google Scholar] [CrossRef] [PubMed]
- Chehroudi, B.; Gould, T.R.; Brunette, D.M. Effects of a grooved titanium-coated implant surface on epithelial cell behavior in vitro and in vivo. J. Biomed. Mater. Res. 1989, 23, 1067–1085. [Google Scholar] [CrossRef] [PubMed]
- Chehroudi, B.; Gould, T.R.; Brunette, D.M. A light and electron microscopic study of the effects of surface topography on the behavior of cells attached to titanium-coated percutaneous implants. J. Biomed. Mater. Res. 1991, 25, 387–405. [Google Scholar] [CrossRef]
- Nevins, M.; Kim, D.M.; Jun, S.H.; Guze, K.; Schupbach, P.; Nevins, M.L. Histologic evidence of a connective tissue attachment to laser microgrooved abutments: A canine study. Int. J. Periodontics Restor. Dent. 2010, 30, 245–255. [Google Scholar]
- Neiva, R.; Tovar, N.; Jimbo, R.; Gil, L.F.; Goldberg, P.; Barbosa, J.P.; Lilin, T.; Coelho, P.G. The Effect of Laser-Etched Surface Design on Soft Tissue Healing of Two Different Implant Abutment Systems: An Experimental Study in Dogs. Int. J. Periodontics Restor. Dent. 2016, 36, 673–679. [Google Scholar] [CrossRef]
- Olson, A.; Le, V.; Aldahl, J.; Yu, E.J.; Hooker, E.; He, Y.; Lee, D.H.; Kim, W.K.; Cardiff, R.D.; Geradts, J.; et al. The comprehensive role of E-cadherin in maintaining prostatic epithelial integrity during oncogenic transformation and tumor progression. PLoS Genet. 2019, 28, e1008451. [Google Scholar] [CrossRef]
- Kim, W.K.; Kwon, Y.; Jang, M.; Park, M.; Kim, J.; Cho, S.; Jang, D.G.; Lee, W.B.; Jung, S.H.; Choi, H.J.; et al. β-catenin activation down-regulates cell-cell junction-related genes and induces epithelial-to-mesenchymal transition in colorectal cancers. Sci. Rep. 2019, 9, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Lakschevitz, F.S.; Hassanpour, S.; Rubin, A.; Fine, N.; Sun, C.; Glogauer, M. Identification of neutrophil surface marker changes in health and inflammation using high-throughput screening flow cytometry. Exp. Cell Res. 2016, 15, 200–209. [Google Scholar] [CrossRef] [PubMed]
- Gibbs, S.; Roffel, S.; Meyer, M.; Gasser, A. Biology of soft tissue repair: Gingival epithelium in wound healing and attachment to the tooth and abutment surface. Eur. Cells Mater. 2019, 14, 63–78. [Google Scholar] [CrossRef] [PubMed]
- Könönen, E.; Gursoy, M.; Gursoy, U.K. Periodontitis: A Multifaceted Disease of Tooth-Supporting Tissues. J. Clin. Med. 2019, 31, 1135. [Google Scholar] [CrossRef] [PubMed]
| Subject Characteristics | n = 38 |
|---|---|
| Age Mean | 56.55 ± 9.95 years |
| Gender (Male) | 25 (65.79%) |
| Implant Position (Upper) | 25 (65.79%) |
| Implant Position (Lower) | 13 (34.21%) |
| Final Torque Insertion (NCM) Mean | 49.73 ± 11.05 |
| BOP# Mean at Implant Placement | 7.32% |
| BOP Mean at Second Surgical Stage | 9.68% |
| PS # Mean at Implant placement | 13.87% |
| PS Mean at Second Surgical Stage | 17.76% |
| Implant Success at Second Surgical Stage | 38 (100%) |
| Biopsy Performed and Analyzed | 30 (78.94%) |
| Antibody | Epithelium Facing the - | Lamina Propria Facing the - | ||
|---|---|---|---|---|
| Laser Treated Surface | Machined Surface | Laser treated surface | Machined Surface | |
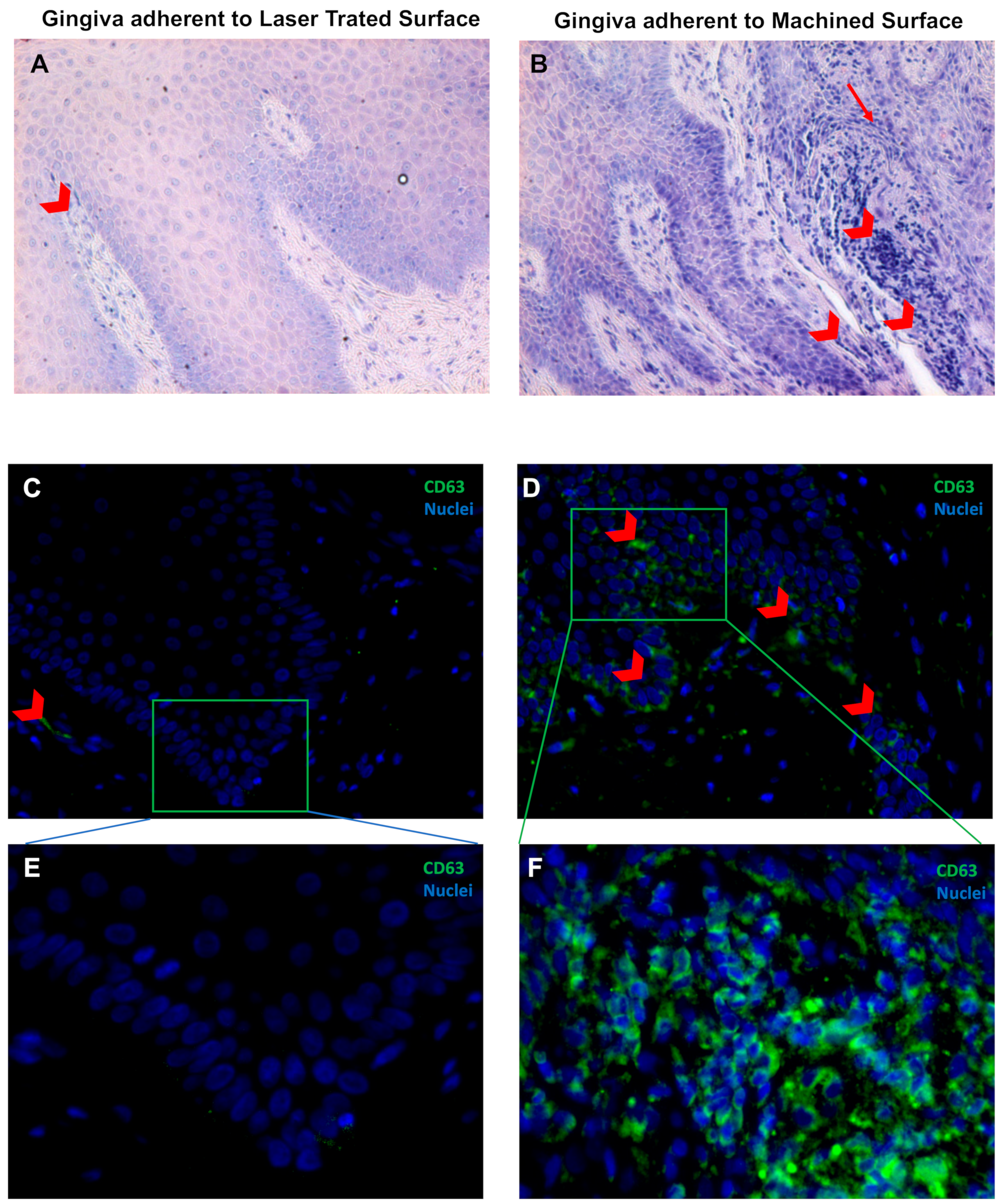
| CD63# | 2.6 ± 1.2 | 9.3 ± 1.6 * | 6.1 ± 3.2 | 30.3 ± 12.5 * |
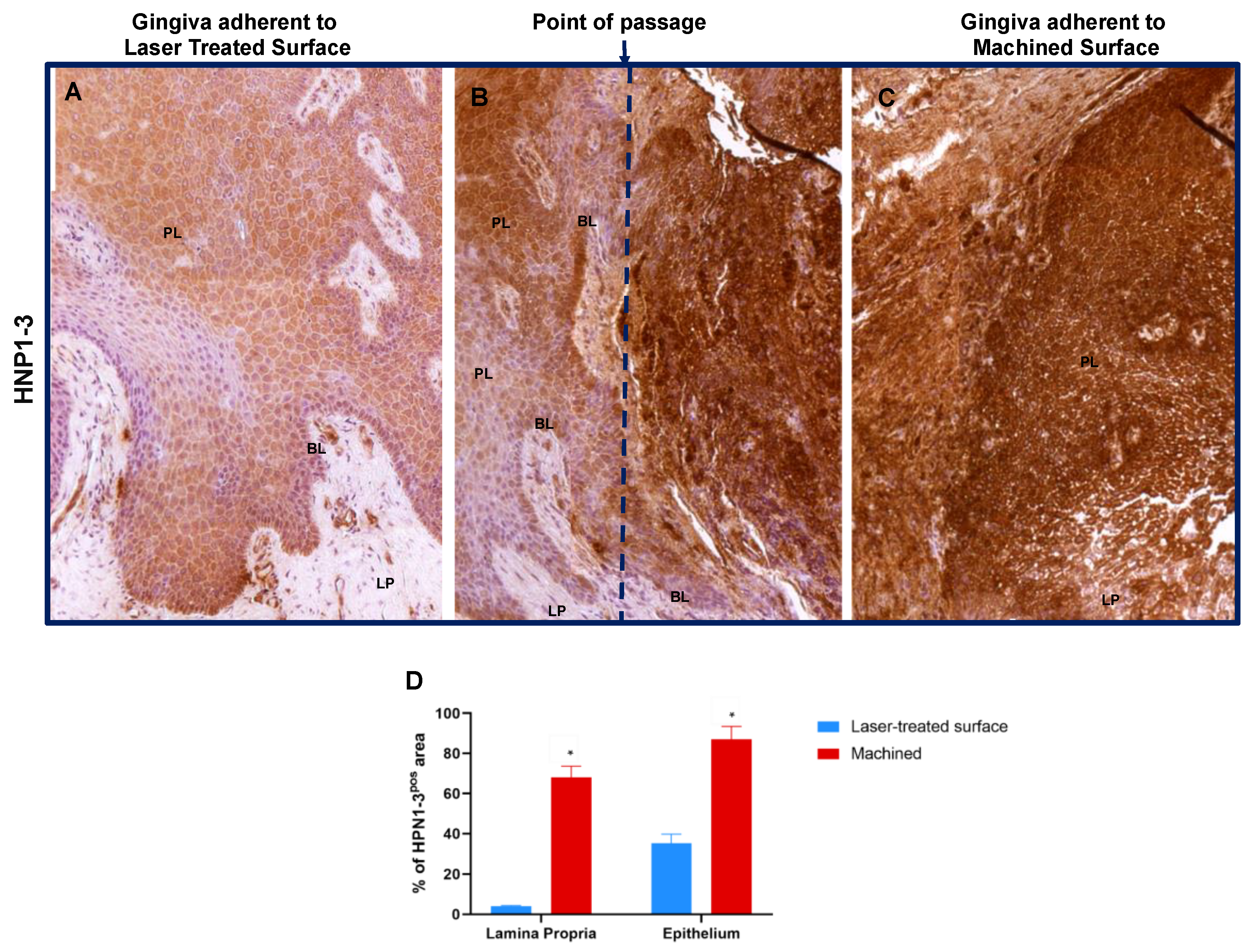
| HNP1–3# | 34.9 ± 4.7 | 87.0 ± 6.6 * | 4.1 ± 0.4 | 68.0 ± 5.4 * |
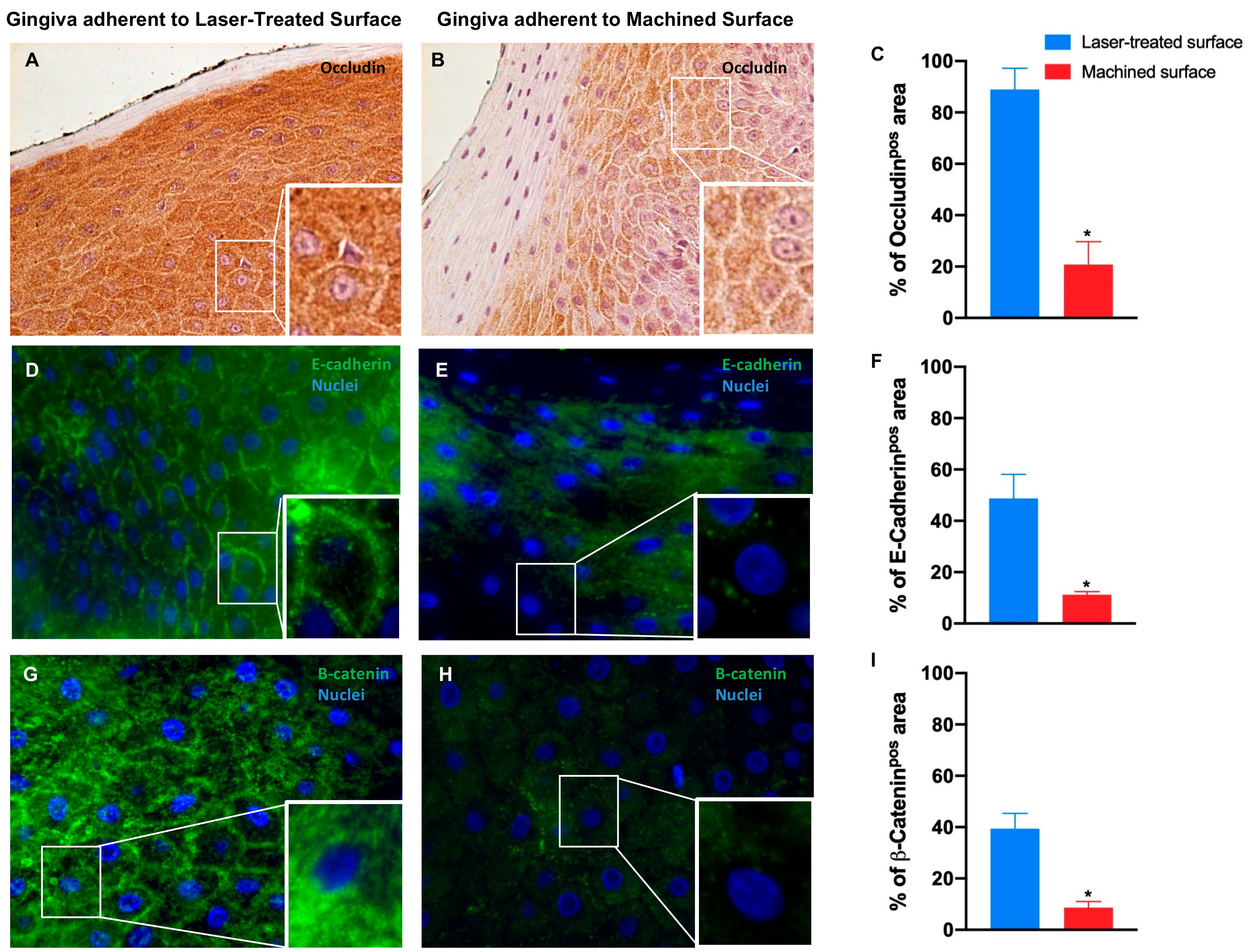
| Occludin | 88.9 ± 8.2 | 20.8 ± 8.9 * | - | - |
| E-Cadherin | 48.7 ± 9.5 | 11.2 ± 1.2 * | - | - |
| β-Catenin | 39.4 ± 6.0 | 8.6 ± 2.5 * | - | - |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ghinassi, B.; D’Addazio, G.; Di Baldassarre, A.; Femminella, B.; Di Vincenzo, G.; Piattelli, M.; Gaggi, G.; Sinjari, B. Immunohistochemical Results of Soft Tissues around a New Implant Healing-Abutment Surface: A Human Study. J. Clin. Med. 2020, 9, 1009. https://doi.org/10.3390/jcm9041009
Ghinassi B, D’Addazio G, Di Baldassarre A, Femminella B, Di Vincenzo G, Piattelli M, Gaggi G, Sinjari B. Immunohistochemical Results of Soft Tissues around a New Implant Healing-Abutment Surface: A Human Study. Journal of Clinical Medicine. 2020; 9(4):1009. https://doi.org/10.3390/jcm9041009
Chicago/Turabian StyleGhinassi, Barbara, Gianmaria D’Addazio, Angela Di Baldassarre, Beatrice Femminella, Giorgio Di Vincenzo, Maurizio Piattelli, Giulia Gaggi, and Bruna Sinjari. 2020. "Immunohistochemical Results of Soft Tissues around a New Implant Healing-Abutment Surface: A Human Study" Journal of Clinical Medicine 9, no. 4: 1009. https://doi.org/10.3390/jcm9041009
APA StyleGhinassi, B., D’Addazio, G., Di Baldassarre, A., Femminella, B., Di Vincenzo, G., Piattelli, M., Gaggi, G., & Sinjari, B. (2020). Immunohistochemical Results of Soft Tissues around a New Implant Healing-Abutment Surface: A Human Study. Journal of Clinical Medicine, 9(4), 1009. https://doi.org/10.3390/jcm9041009









