Incubation Period and Other Epidemiological Characteristics of 2019 Novel Coronavirus Infections with Right Truncation: A Statistical Analysis of Publicly Available Case Data
Abstract
1. Introduction
2. Methods
2.1. Epidemiological Data
2.2. Statistical Model
3. Results
4. Discussion
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- National Health Commission of the People’s Republic of China. Update on Pneumonia of New Coronavirus Infection as of 21:00 on January 31. Available online: http://www.nhc.gov.cn/xcs/yqtb/202002/84faf71e096446fdb1ae44939ba5c528.shtml (accessed on 1 February 2020).
- Li, Q.; Guan, X.; Wu, P.; Wang, X.; Zhou, L.; Tong, Y.; Ren, R.; Leung, K.S.M.; Lau, E.H.Y.; Wong, J.Y.; et al. Early transmission dynamics in Wuhan, China, of novel coronavirus-infected pneumonia. N. Engl. J. Med. 2020. [Google Scholar] [CrossRef] [PubMed]
- European Centre for Disease Prevention and Control data. Geographical Distribution of 2019-nCov Cases. Available online: https://www.ecdc.europa.eu/en/geographical-distribution-2019-ncov-cases (accessed on 24 January 2020).
- Nishiura, H.; Lee, H.W.; Cho, S.H.; Lee, W.G.; In, T.S.; Moon, S.U.; Chung, G.T.; Kim, T.S. Estimates of short- and long-term incubation periods of Plasmodium vivax malaria in the Republic of Korea. Trans. R. Soc. Trop. Med. Hyg. 2007, 101, 338–343. [Google Scholar] [CrossRef] [PubMed]
- Lessler, J.; Reich, N.G.; Cummings, D.A.; New York City Department of Health and Mental Hygiene Swine Influenza Investigation Team. Outbreak of 2009 pandemic influenza A (H1N1) at a New York City school. N. Engl. J. Med. 2009, 361, 2628–2636. [Google Scholar] [CrossRef] [PubMed]
- Nishiura, H. Determination of the appropriate quarantine period following smallpox exposure: An objective approach using the incubation period distribution. Int. J. Hyg. Environ. Health. 2009, 212, 97–104. [Google Scholar] [CrossRef] [PubMed]
- Nishiura, H.; Jung, S.; Linton, N.M.; Kinoshita, R.; Yang, Y.; Hayashi, K.; Kobayashi, T.; Yuan, B.; Akhmetzhanov, A.R. The extent of transmission of novel coronavirus in Wuhan, China, 2020. J. Clin. Med. 2020, 24, 9. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.; Wang, Y.; Li, X.; Ren, L.; Zhao, J.; Hu, Y.; Zhang, L.; Fan, G.; Xu, J.; Gu, X. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 2020. [Google Scholar] [CrossRef]
- Donnelly, C.A.; Ghani, A.C.; Leung, G.M.; Hedley, A.J.; Fraser, C.; Riley, S.; Abu-Raddad, L.J.; Ho, L.M.; Thach, T.Q.; Chau, P. Epidemiological determinants of spread of causal agent of severe acute respiratory syndrome in Hong Kong. Lancet 2003, 361, 1761–1766. [Google Scholar] [CrossRef]
- Reich, N.G.; Lessler, J.; Cummings, D.A.T.; Brookmeyer, R. Estimating incubation period distributions with coarse data. Stat. Med. 2009, 28, 2769–2784. [Google Scholar] [CrossRef] [PubMed]
- Jung, S.; Akhmetzhanov, A.R.; Hayashi, K.; Linton, N.M.; Yang, Y.; Yuan, B.; Kobayashi, T.; Kinoshita, R.; Nishiura, H. Real time estimation of the risk of death from novel coronavirus (2019-nCoV) infection: Inference using exported cases. medrxiv 2020. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing. Available online: https://www.r-project.org (accessed on 24 January 2020).
- Bezanson, J.; Edelman, A.; Karpinski, S.; Shah, V.B. Julia: A fresh approach to numerical computing. SIAM Rev. 2017, 59, 65–98. [Google Scholar] [CrossRef]
- Stan Development Team. Stan Modeling Language Users Guide and Reference Manual, Version 2.22.0. Available online: http://mc-stan.org (accessed on 24 January 2020).
- Lessler, J.; Reich, N.G.; Brookmeyer, R.; Perl, T.M.; Nelson, K.E.; Cummings, D.A. Incubation periods of acute respiratory viral infections: A systematic review. Lancet Infect. Dis. 2009, 9, 291–300. [Google Scholar] [CrossRef]
- Virlogeux, V.; Park, M.; Wu, J.T.; Cowling, B. Association between severity of MERS-CoV infection and incubation period. Emerg. Infect. Dis. 2016, 22, 526–528. [Google Scholar] [CrossRef] [PubMed]
- Backer, J.A.; Klinkenberg, D.; Wallinga, J. The incubation period of 2019-nCoV infections among travellers from Wuhan, China. medRxiv 2020. [Google Scholar] [CrossRef]
- Fraser, C.; Donnelly, C.A.; Cauchemez, S.; Hanage, W.P.; Van Kerkhove, M.D.; Hollingsworth, T.D.; Griffin, J.; Baggaley, R.F.; Jenkins, H.E.; Lyons, E.J.; et al. WHO rapid pandemic assessment collaboration. Pandemic potential of a strain of influenza A (H1N1): Early findings. Science 2009, 324, 1557–1561. [Google Scholar] [CrossRef] [PubMed]
- Ghani, A.C.; Donnelly, C.A.; Cox, D.R.; Griffin, J.T.; Fraser, C.; Lam, T.H.; Ho, L.M.; Chan, W.S.; Anderson, R.M.; Hedley, A.J.; et al. Methods for estimating the case fatality ratio for a novel, emerging infectious disease. Am. J. Epidemiol. 2005, 162, 479–486. [Google Scholar] [CrossRef] [PubMed]
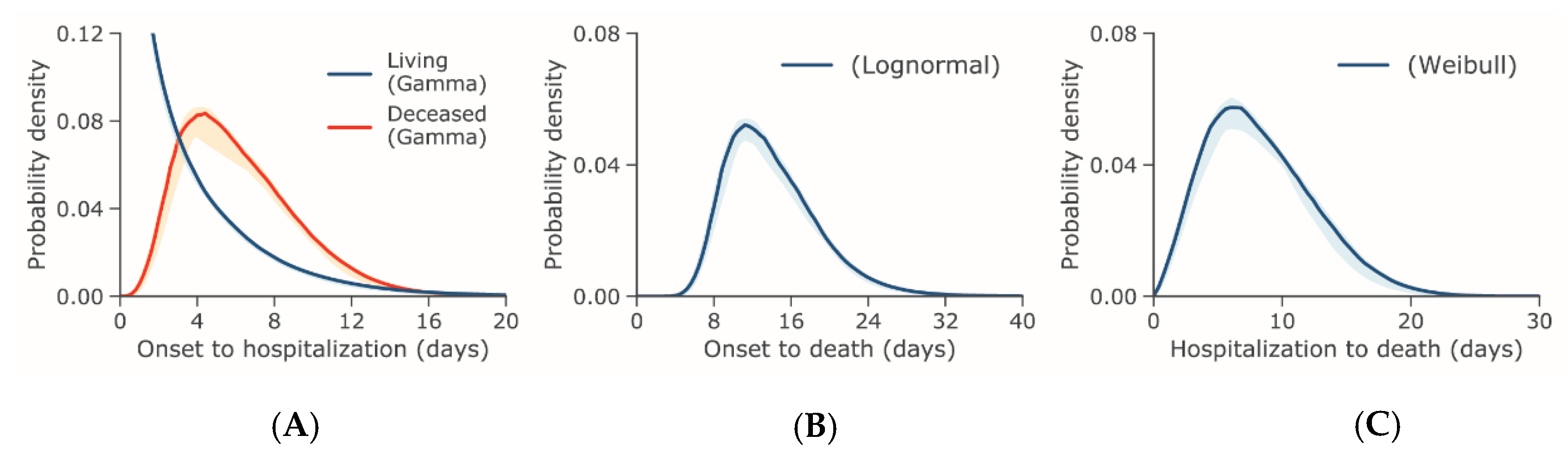
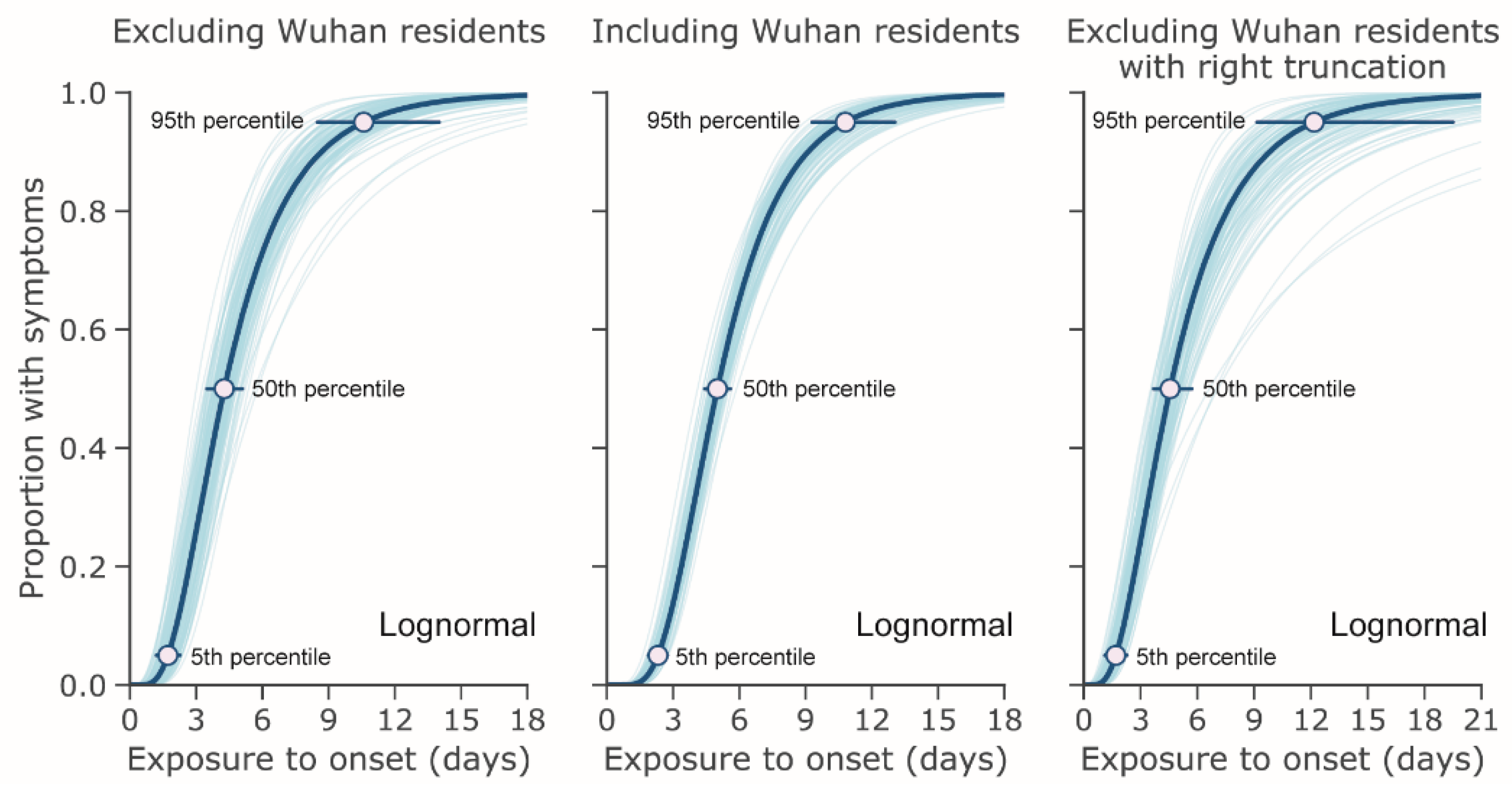
| Incubation Period Excluding WR (Days) | Incubation Period Including WR (Days) | Onset to Hospital Admission, Living (Days) | Onset to Hospital Admission, Deceased (Days) | Onset to Death (Days) | Hospital Admission to Death (Days) | |
|---|---|---|---|---|---|---|
| Number of cases | 52 cases | 158 cases | 155 cases | 34 cases | 34 cases | 39 cases |
| Lognormal | ||||||
| Mean | 5.0 (4.2, 6.0) | 5.6 (5.0, 6.3) | 3.9 (2.9, 5.3) | 6.2 (5.0, 7.8) | 14.5 (12.5, 17.0) | 8.6 (6.8, 10.8) |
| SD | 3.0 (2.1, 4.5) | 2.8 (2.2, 3.6) | 9.1 (5.2, 16.3) | 4.3 (2.9, 6.6) | 6.7 (4.9, 9.4) | 6.7 (4.5, 10.3) |
| 5% | 1.7 (1.2, 2.3) | 2.3 (1.9, 2.7) | 0.2 (0.1, 0.3) | 1.9 (1.2, 2.5) | 6.5 (4.9, 7.9) | 2.2 (1.5, 3.0) |
| Median | 4.3 (3.5, 5.1) | 5.0 (4.4, 5.6) | 1.5 (1.2, 1.9) | 5.1 (4.1, 6.3) | 13.2 (11.3, 15.3) | 6.7 (5.3, 8.3) |
| 95% | 10.6 (8.5, 14.1) | 10.8 (9.3, 12.9) | 14.0 (10.3, 20.1) | 13.9 (10.8, 19.6) | 26.8 (22.3, 34.5) | 20.5 (15.7, 28.7) |
| 99% | 15.4 (11.7, 22.5) | 14.9 (12.3, 18.7) | 35.0 (23.2, 56.9) | 21.1 (15.3, 32.8) | 36.0 (28.6, 49.8) | 32.6 (23.3, 50.1) |
| WAIC | 257.2 | 859.6 | 693.5 | 183.9 | 221.9 | 240.1 |
| Weight | 1.00 | 1.00 | 0.00 | 0.33 | 1.00 | 0.02 |
| Weibull | ||||||
| Mean | 5.4 (4.3, 6.6) | 5.8 (5.2, 6.5) | 3.4 (2.7, 4.2) | 6.5 (5.2, 8.0) | 15.1 (12.7, 17.8) | 8.9 (7.3, 10.4) |
| SD | 3.6 (2.8, 4.7) | 2.8 (2.3, 3.5) | 4.4 (3.3, 6.0) | 4.0 (3.1, 5.4) | 7.6 (6.1, 9.7) | 5.4 (4.2, 7.3) |
| 5% | 0.9 (0.5, 1.4) | 2.1 (1.5, 2.6) | 0.1 (0.0, 0.1) | 1.9 (1.2, 2.5) | 4.2 (2.5, 6.0) | 1.7 (0.9, 2.7) |
| Median | 4.7 (3.6, 5.8) | 5.3 (4.7, 6.0) | 1.8 (1.4, 2.3) | 5.1 (4.1, 6.3) | 14.3 (11.8, 17.1) | 8.0 (6.2, 9.8) |
| 95% | 12.0 (9.8, 15.6) | 11.0 (9.6, 12.9) | 11.7 (9.3, 15.6) | 13.9 (10.8, 19.6) | 28.6 (24.5, 34.9) | 18.8 (15.5, 24.4) |
| 99% | 15.9 (12.8, 21.5) | 14.2 (12.1, 17.0) | 20.3 (15.3, 29.2) | 21.1 (15.3, 32.8) | 35.0 (29.5, 44.4) | 24.2 (19.5, 33.0) |
| WAIC | 273.8 | 871.8 | 662.4 | 185.5 | 231.3 | 236.1 |
| Weight | 0.00 | 0.00 | 0.00 | 0.29 | 0.00 | 0.96 |
| Gamma | ||||||
| Mean | 5.3 (4.3, 6.6) | 6.0 (5.3, 6.7) | 3.3 (2.7, 4.0) | 6.5 (5.2, 8.0) | 15.0 (12.8, 17.5) | 8.8 (7.2, 10.8) |
| SD | 3.2 (2.4, 4.3) | 3.1 (2.7, 3.7) | 4.2 (3.3, 5.4) | 4.0 (2.9, 5.6) | 6.9 (5.2, 9.1) | 5.7 (4.3, 7.8) |
| 5% | 1.4 (0.7, 2.0) | 1.5 (1.1, 2.0) | 0.0 (0.0, 0.1) | 1.6 (0.8, 2.4) | 5.8 (3.9, 7.6) | 1.9 (1.0, 2.9) |
| Median | 4.7 (3.8, 5.7) | 5.6 (4.9, 6.4) | 1.7 (1.2, 2.2) | 5.6 (4.5, 6.9) | 13.9 (11.8, 17.1) | 7.6 (6.0, 9.3) |
| 95% | 11.3 (9.2, 14.5) | 11.7 (10.3, 13.4) | 11.6 (9.4, 14.7) | 13.8 (11.1, 18.6) | 27.4 (23.2, 34.1) | 19.9 (15.7, 25.7) |
| 99% | 15.2 (12.0, 20.2) | 14.5 (12.6, 16.9) | 19.4 (15.4, 25.1) | 18.6 (14.6, 26.2) | 34.9 (28.8, 44.8) | 26.8 (21.1, 36.5) |
| WAIC | 265.8 | 895.9 | 656.6 | 183.4 | 225.1 | 236.3 |
| Weight | 0.00 | 0.00 | 1.00 | 0.38 | 0.00 | 0.02 |
| Lognormal | Incubation Period Excluding WR (Days) | Onset to Hospital Admission, Living (Days) | Onset to Hospital Admission, Deceased (Days) | Onset to Death (Days) | Hospital Admission to Death (Days) |
|---|---|---|---|---|---|
| Mean | 5.6 (4.4, 7.4) | 9.7 (5.4, 17.0) | 6.6 (5.2, 8.8) | 20.2 (15.1, 29.5) | 13.0 (8.7, 20.9) |
| SD | 3.9 (2.4, 6.9) | 35.2 (12.9, 84.5) | 4.9 (3.0, 8.6) | 11.6 (6.6, 21.8) | 12.7 (6.4, 26.0) |
| 5% | 1.7 (1.1, 2.3) | 0.2 (0.1, 0.3) | 1.9 (1.2, 2.5) | 7.4 (5.6, 9.5) | 2.5 (1.6, 3.6) |
| Median | 4.6 (3.7, 5.7) | 2.6 (1.9, 3.8) | 5.3 (4.2, 6.8) | 17.1 (13.5, 24.1) | 9.1 (6.7, 13.7) |
| 95% | 12.3 (9.1, 19.8) | 35.1 (20.5, 65.0) | 15.0 (11.2, 23.8) | 39.9 (27.9, 69.6) | 33.1 (20.9, 56.7) |
| 99% | 18.6 (12.7, 34.2) | 102.8 (52.8, 218.4) | 23.2 (16.0, 41.6) | 56.7 (37.0, 109.2) | 56.7 (32.5, 125.2) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Linton, N.M.; Kobayashi, T.; Yang, Y.; Hayashi, K.; Akhmetzhanov, A.R.; Jung, S.-m.; Yuan, B.; Kinoshita, R.; Nishiura, H. Incubation Period and Other Epidemiological Characteristics of 2019 Novel Coronavirus Infections with Right Truncation: A Statistical Analysis of Publicly Available Case Data. J. Clin. Med. 2020, 9, 538. https://doi.org/10.3390/jcm9020538
Linton NM, Kobayashi T, Yang Y, Hayashi K, Akhmetzhanov AR, Jung S-m, Yuan B, Kinoshita R, Nishiura H. Incubation Period and Other Epidemiological Characteristics of 2019 Novel Coronavirus Infections with Right Truncation: A Statistical Analysis of Publicly Available Case Data. Journal of Clinical Medicine. 2020; 9(2):538. https://doi.org/10.3390/jcm9020538
Chicago/Turabian StyleLinton, Natalie M., Tetsuro Kobayashi, Yichi Yang, Katsuma Hayashi, Andrei R. Akhmetzhanov, Sung-mok Jung, Baoyin Yuan, Ryo Kinoshita, and Hiroshi Nishiura. 2020. "Incubation Period and Other Epidemiological Characteristics of 2019 Novel Coronavirus Infections with Right Truncation: A Statistical Analysis of Publicly Available Case Data" Journal of Clinical Medicine 9, no. 2: 538. https://doi.org/10.3390/jcm9020538
APA StyleLinton, N. M., Kobayashi, T., Yang, Y., Hayashi, K., Akhmetzhanov, A. R., Jung, S.-m., Yuan, B., Kinoshita, R., & Nishiura, H. (2020). Incubation Period and Other Epidemiological Characteristics of 2019 Novel Coronavirus Infections with Right Truncation: A Statistical Analysis of Publicly Available Case Data. Journal of Clinical Medicine, 9(2), 538. https://doi.org/10.3390/jcm9020538







