Increased Fecal Calprotectin Is Associated with Worse Gastrointestinal Symptoms and Quality of Life Scores in Children with Cystic Fibrosis
Abstract
1. Background
2. Methods
2.1. Design and Participants
2.2. Study Design/Procedure
2.3. Statistical Analysis
3. Results
3.1. Population Characteristics
3.2. Comparison between Scores Obtained from Children and Their Parents
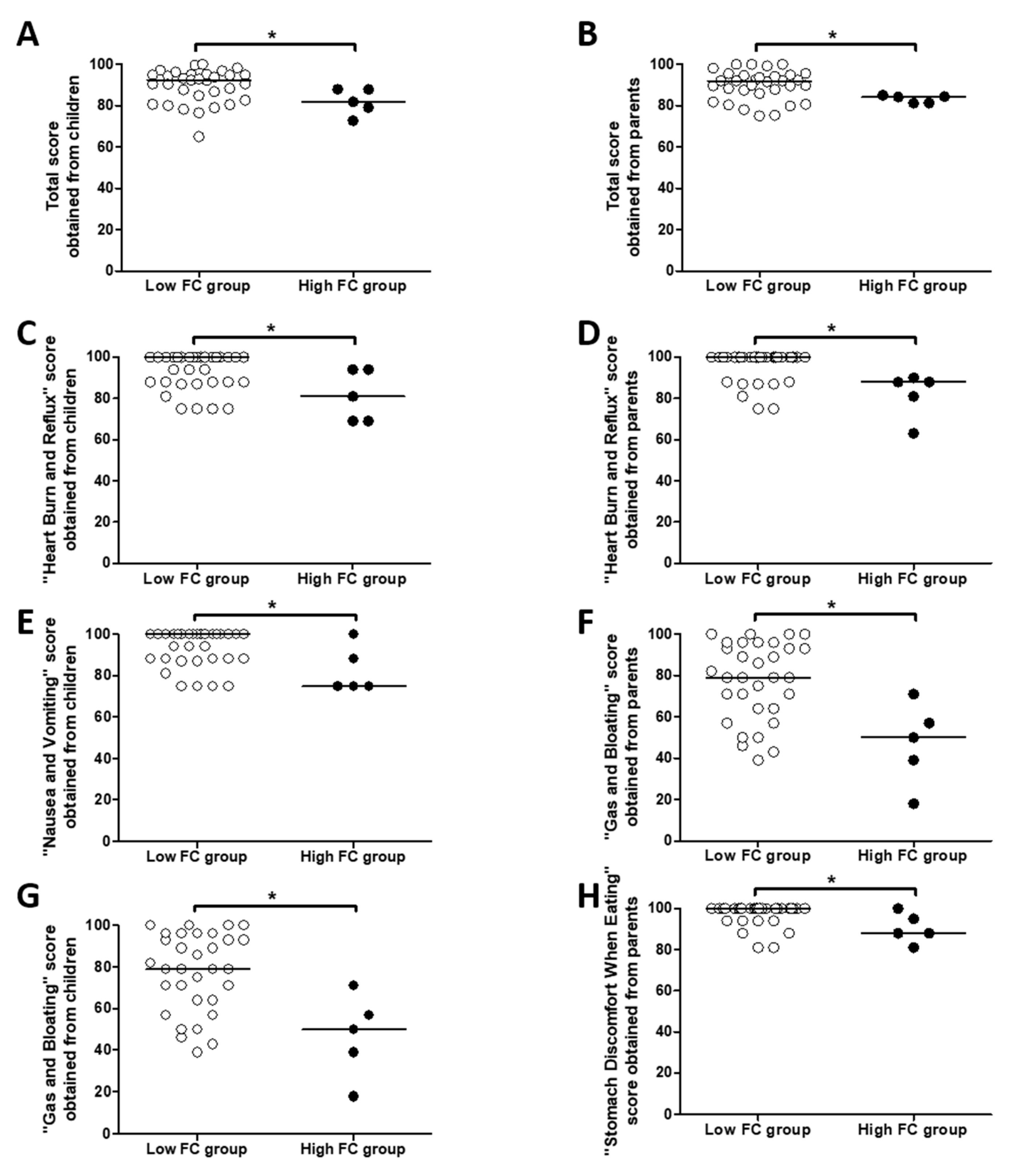
3.3. Increased Fecal Calprotectin Level Is Associated with Worse Gastrointestinal Symptomatology
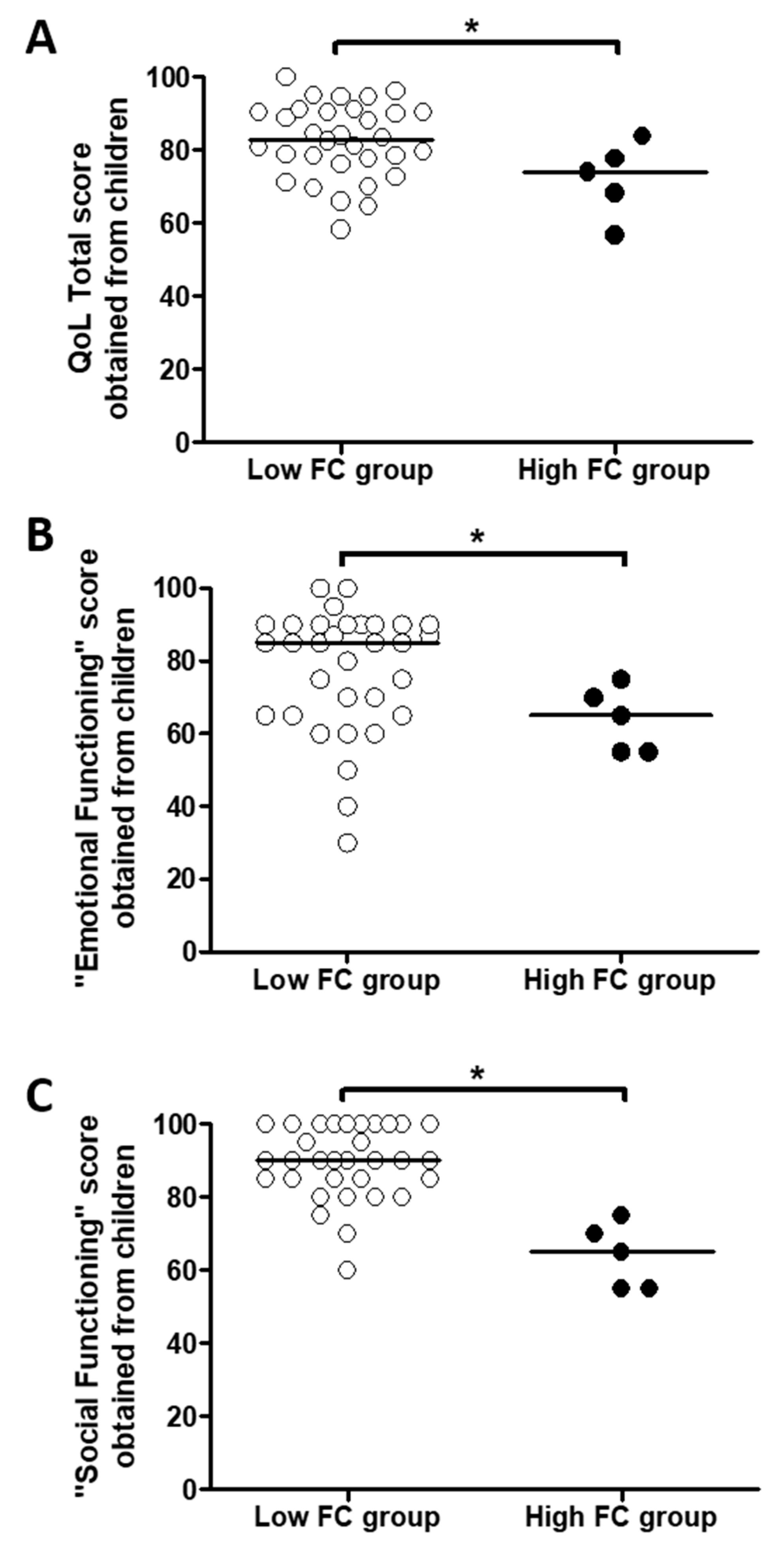
3.4. An Increased Fecal Calprotectin Level Is Associated with Worse Quality of Life Scores Assessed by Children
3.5. Associations between the Fecal Calprotectin Level and Gastrointestinal and Quality of Life Scores
3.6. Association between Fecal Calprotectin Level and Clinical Parameters
3.7. Associations of Clinical Characteristics and Lung Function of Patients with GI Symptoms and QoL
3.8. Quality of Life Scores Are Associated to Gastroinstestinal Sympotms Scores
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| CF | Cystic Fibrosis |
| FC | Fecal calprotectin |
| FEV1 | Forced expiratory volume in one second |
| FVC | Forced vital capacity |
| FEF25–75 | Forced mid-expiratory flow between 25% and 75% of forced vital capacity |
| GI | Gastrointestinal |
| IBD | Inflammatory bowel diseases |
| IQR | Interquartile range |
| QoL | Quality of life |
References
- Rowe, S.M.; Miller, S.; Sorscher, E.J. Cystic fibrosis. N. Engl. J. Med. 2005, 352, 1992–2001. [Google Scholar] [CrossRef] [PubMed]
- Ratjen, F.; Bell, S.C.; Rowe, S.M.; Goss, C.H.; Quittner, A.L.; Bush, A. Cystic fibrosis. Nat. Rev. Dis. Primers 2015, 1, 15010. [Google Scholar] [CrossRef] [PubMed]
- Munck, A. Cystic fibrosis: Evidence for gut inflammation. Int. J. Biochem. Cell Biol. 2014, 52, 180–183. [Google Scholar] [CrossRef] [PubMed]
- Bruzzese, E.; Raia, V.; Gaudiello, G.; Polito, G.; Buccigrossi, V.; Formicola, V.; Guarino, A. Intestinal inflammation is a frequent feature of cystic fibrosis and is reduced by probiotic administration. Aliment. Pharmacol. Ther. 2004, 20, 813–819. [Google Scholar] [CrossRef]
- del Campo, R.; Garriga, M.; Pérez-Aragón, A.; Guallarte, P.; Lamas, A.; Máiz, L.; Bayón, C.; Roy, G.; Cantón, R.; Zamora, J.; et al. Improvement of digestive health and reduction in proteobacterial populations in the gut microbiota of cystic fibrosis patients using a Lactobacillus reuteri probiotic preparation: A double blind prospective study. J. Cyst. Fibros. 2014, 13, 716–722. [Google Scholar] [CrossRef]
- Manor, O.; Levy, R.; Pope, C.E.; Hayden, H.S.; Brittnacher, M.J.; Carr, R.; Radey, M.C.; Hager, K.R.; Heltshe, S.L.; Ramsey, B.W.; et al. Metagenomic evidence for taxonomic dysbiosis and functional imbalance in the gastrointestinal tracts of children with cystic fibrosis. Sci. Rep. 2016, 6, 22493. [Google Scholar] [CrossRef]
- de Freitas, M.B.; Moreira, E.A.M.; Tomio, C.; Moreno, Y.M.F.; Daltoe, F.P.; Barbosa, E.; Ludwig Neto, N.; Buccigrossi, V.; Guarino, A. Altered intestinal microbiota composition, antibiotic therapy and intestinal inflammation in children and adolescents with cystic fibrosis. PLoS ONE 2018, 13, e0198457. [Google Scholar] [CrossRef]
- Enaud, R.; Hooks, K.B.; Barre, A.; Barnetche, T.; Hubert, C.; Massot, M.; Bazin, T.; Clouzeau, H.; Bui, S.; Fayon, M.; et al. Intestinal Inflammation in Children with Cystic Fibrosis Is Associated with Crohn’s-Like Microbiota Disturbances. J. Clin. Med. 2019, 8, 645. [Google Scholar] [CrossRef]
- Coffey, M.J.; Nielsen, S.; Wemheuer, B.; Kaakoush, N.O.; Garg, M.; Needham, B.; Pickford, R.; Jaffe, A.; Thomas, T.; Ooi, C.Y. Gut Microbiota in Children with Cystic Fibrosis: A Taxonomic and Functional Dysbiosis. Sci. Rep. 2019, 9, 1–14. [Google Scholar] [CrossRef]
- Raia, V.; Maiuri, L.; de Ritis, G.; de Vizia, B.; Vacca, L.; Conte, R.; Auricchio, S.; Londei, M. Evidence of Chronic Inflammation in Morphologically Normal Small Intestine of Cystic Fibrosis Patients. Pediatric Res. 2000, 47, 344–350. [Google Scholar] [CrossRef]
- Werlin, S.L.; Benuri-Silbiger, I.; Kerem, E.; Adler, S.N.; Goldin, E.; Zimmerman, J.; Malka, N.; Cohen, L.; Armoni, S.; Yatzkan-Israelit, Y.; et al. Evidence of intestinal inflammation in patients with cystic fibrosis. J. Pediatric Gastroenterol. Nutr. 2010, 51, 304–308. [Google Scholar] [CrossRef] [PubMed]
- Flass, T.; Tong, S.; Frank, D.N.; Wagner, B.D.; Robertson, C.E.; Kotter, C.V.; Sokol, R.J.; Zemanick, E.; Accurso, F.; Hoffenberg, E.J.; et al. Intestinal Lesions Are Associated with Altered Intestinal Microbiome and Are More Frequent in Children and Young Adults with Cystic Fibrosis and Cirrhosis. PLoS ONE 2015, 10, e0116967. [Google Scholar] [CrossRef] [PubMed]
- Brecelj, J.; Zidar, N.; Jeruc, J.; Orel, R. Morphological and Functional Assessment of Oesophageal Mucosa Integrity in Children with Cystic Fibrosis. J. Pediatric Gastroenterol. Nutr. 2016, 62, 757–764. [Google Scholar] [CrossRef] [PubMed]
- Bruzzese, E.; Callegari, M.L.; Raia, V.; Viscovo, S.; Scotto, R.; Ferrari, S.; Morelli, L.; Buccigrossi, V.; Lo Vecchio, A.; Ruberto, E.; et al. Disrupted Intestinal Microbiota and Intestinal Inflammation in Children with Cystic Fibrosis and Its Restoration with Lactobacillus GG: A Randomised Clinical Trial. PLoS ONE 2014, 9, e87796. [Google Scholar] [CrossRef]
- Dhaliwal, J.; Leach, S.; Katz, T.; Nahidi, L.; Pang, T.; Lee, J.M.; Strachan, R.; Day, A.S.; Jaffe, A.; Ooi, C.Y. Intestinal Inflammation and Impact on Growth in Children with Cystic Fibrosis. J. Pediatric Gastroenterol. Nutr. 2015, 60, 521–526. [Google Scholar] [CrossRef]
- Parisi, G.F.; Papale, M.; Rotolo, N.; Aloisio, D.; Tardino, L.; Scuderi, M.G.; Di Benedetto, V.; Nenna, R.; Midulla, F.; Leonardi, S. Severe disease in Cystic Fibrosis and fecal calprotectin levels. Immunobiology 2017, 222, 582–586. [Google Scholar] [CrossRef]
- Adriaanse, M.P.M.; van der Sande, L.J.T.M.; van den Neucker, A.M.; Menheere, P.P.C.A.; Dompeling, E.; Buurman, W.A.; Vreugdenhil, A.C.E. Evidence for a Cystic Fibrosis Enteropathy. PLoS ONE 2015, 10, e0138062. [Google Scholar] [CrossRef]
- Ellemunter, H.; Engelhardt, A.; Schüller, K.; Steinkamp, G. Fecal Calprotectin in Cystic Fibrosis and Its Relation to Disease Parameters: A Longitudinal Analysis for 12 Years. J. Pediatric Gastroenterol. Nutr. 2017, 65, 438–442. [Google Scholar] [CrossRef]
- Stallings, V.A.; Sainath, N.; Oberle, M.; Bertolaso, C.; Schall, J.I. Energy Balance and Mechanisms of Weight Gain with Ivacaftor Treatment of Cystic Fibrosis Gating Mutations. J. Pediatrics 2018, 201, 229–237.e4. [Google Scholar] [CrossRef]
- Ooi, C.Y.; Syed, S.A.; Rossi, L.; Garg, M.; Needham, B.; Avolio, J.; Young, K.; Surette, M.G.; Gonska, T. Impact of CFTR modulation with Ivacaftor on Gut Microbiota and Intestinal Inflammation. Sci. Rep. 2018, 8, 1–8. [Google Scholar] [CrossRef]
- Tétard, C.; Mittaine, M.; Bui, S.; Beaufils, F.; Maumus, P.; Fayon, M.; Burgel, P.-R.; Lamireau, T.; Delhaes, L.; Mas, E.; et al. Reduced Intestinal Inflammation with Lumacaftor/Ivacaftor in Adolescents with Cystic Fibrosis. J. Pediatric Gastroenterol. Nutr. 2020, 71, 778–781. [Google Scholar] [CrossRef] [PubMed]
- Rumman, N.; Sultan, M.; El-Chammas, K.; Goh, V.; Salzman, N.; Quintero, D.; Werlin, S. Calprotectin in Cystic Fibrosis. BMC Pediatrics 2014, 14, 133. [Google Scholar] [CrossRef]
- Jafari, S.-A.; Mehdizadeh-Hakkak, A.; Kianifar, H.-R.; Hebrani, P.; Ahanchian, H.; Abbasnejad, E. Effects of Probiotics on Quality of Life in Children with Cystic Fibrosis. Iran. J. Pediatr. 2013, 23, 6. [Google Scholar]
- Varni, J.W.; Bendo, C.B.; Denham, J.; Shulman, R.J.; Self, M.M.; Neigut, D.A.; Nurko, S.; Patel, A.S.; Franciosi, J.P.; Saps, M.; et al. PedsQLTM Gastrointestinal Symptoms Scales and Gastrointestinal Worry Scales in pediatric patients with functional and organic gastrointestinal diseases in comparison to healthy controls. Qual. Life Res. 2015, 24, 363–378. [Google Scholar] [CrossRef]
- Varni, J.W.; Seid, M.; Kurtin, P.S. PedsQL 4.0: Reliability and validity of the Pediatric Quality of Life Inventory version 4.0 generic core scales in healthy and patient populations. Med. Care 2001, 39, 800–812. [Google Scholar] [CrossRef]
- Koo, T.K.; Li, M.Y. A Guideline of Selecting and Reporting Intraclass Correlation Coefficients for Reliability Research. J. Chiropr. Med. 2016, 15, 155–163. [Google Scholar] [CrossRef] [PubMed]
- Gelfond, D.; Borowitz, D. Gastrointestinal Complications of Cystic Fibrosis. Clin. Gastroenterol. Hepatol. 2013, 11, 333–342. [Google Scholar] [CrossRef] [PubMed]
- Barratt, S.M.; Leeds, J.S.; Robinson, K.; Shah, P.J.; Lobo, A.J.; McAlindon, M.E.; Sanders, D.S. Reflux and irritable bowel syndrome are negative predictors of quality of life in coeliac disease and inflammatory bowel disease. Eur. J. Gastroenterol. Hepatol. 2011, 23, 159–165. [Google Scholar] [CrossRef]
- Garg, M.; Ooi, C. The Enigmatic Gut in Cystic Fibrosis: Linking Inflammation, Dysbiosis, and the Increased Risk of Malignancy. Curr. Gastroenterol. Rep. 2017, 19, 6. [Google Scholar] [CrossRef]
- Keller, J. Human pancreatic exocrine response to nutrients in health and disease. Gut 2005, 54, 1–28. [Google Scholar] [CrossRef]
- Royce, F.H.; Carl, J.C. Health-related quality of life in cystic fibrosis. Curr. Opin. Pediatric 2011, 23, 535–540. [Google Scholar] [CrossRef] [PubMed]
- Abbott, J.; Hart, A.; Havermans, T.; Matossian, A.; Goldbeck, L.; Barreto, C.; Bergsten-Brucefors, A.; Besier, T.; Catastini, P.; Lupi, F.; et al. Measuring health-related quality of life in clinical trials in cystic fibrosis. J. Cyst. Fibros. 2011, 10 (Suppl. 2), S82–S85. [Google Scholar] [CrossRef]
- Sawicki, G.S.; Rasouliyan, L.; McMullen, A.H.; Wagener, J.S.; McColley, S.A.; Pasta, D.J.; Quittner, A.L. Longitudinal assessment of health-related quality of life in an observational cohort of patients with cystic fibrosis. Pediatric Pulmonol. 2011, 46, 36–44. [Google Scholar] [CrossRef] [PubMed]
- Abbott, J. Health-related quality of life measurement in cystic fibrosis: Advances and limitations. Chronic Respir. Dis. 2009, 6, 31–41. [Google Scholar] [CrossRef]
- Goldbeck, L.; Zerrer, S.; Schmitz, T.G. Monitoring quality of life in outpatients with cystic fibrosis: Feasibility and longitudinal results. J. Cyst. Fibros. 2007, 6, 171–178. [Google Scholar] [CrossRef][Green Version]
- Gee, L.; Abbott, J.; Conway, S.P.; Etherington, C.; Webb, A.K. Quality of life in cystic fibrosis: The impact of gender, general health perceptions and disease severity. J. Cyst. Fibros. 2003, 2, 206–213. [Google Scholar] [CrossRef]
- Gee, L.; Abbott, J.; Hart, A.; Conway, S.P.; Etherington, C.; Webb, A.K. Associations between clinical variables and quality of life in adults with cystic fibrosis. J. Cyst. Fibros. 2005, 4, 59–66. [Google Scholar] [CrossRef]
- Palermo, T.M.; Harrison, D.; Koh, J.L. Effect of disease-related pain on the health-related quality of life of children and adolescents with cystic fibrosis. Clin. J. Pain 2006, 22, 532–537. [Google Scholar] [CrossRef]
- Voorend-van Bergen, S.; Brackel, H.; Caudri, D.; de Jongste, J.; Pijnenburg, M. Assessment of asthma control by children and parents. Eur. Respir. J. 2013, 41, 233. [Google Scholar] [CrossRef]
- Tabori, H.; Arnold, C.; Jaudszus, A.; Mentzel, H.-J.; Renz, D.M.; Reinsch, S.; Lorenz, M.; Michl, R.; Gerber, A.; Lehmann, T.; et al. Abdominal symptoms in cystic fibrosis and their relation to genotype, history, clinical and laboratory findings. PLoS ONE 2017, 12, e0174463. [Google Scholar] [CrossRef]
- Pang, T.; Leach, S.T.; Katz, T.; Jaffe, A.; Day, A.S.; Ooi, C.Y. Elevated fecal M2-pyruvate kinase in children with cystic fibrosis: A clue to the increased risk of intestinal malignancy in adulthood? J. Gastroenterol. Hepatol. 2015, 30, 866–871. [Google Scholar] [CrossRef] [PubMed]
- Uchmanowicz, I.; Jankowska-Polańska, B.; Wleklik, M.; Rosinczuk-Tonderys, J.; Dębska, G. Health-related quality of life of patients with cystic fibrosis assessed by the SF-36 questionnaire. Adv. Respir. Med. 2014, 82, 10–17. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.-F.; Chen, J.-M.; Zuo, J.-H.; Yu, A.; Xiao, Z.-J.; Deng, F.-H.; Nie, B.; Jiang, B. Meta-analysis: Fecal Calprotectin for Assessment of Inflammatory Bowel Disease Activity. Inflamm. Bowel. Dis. 2014, 20, 1407–1415. [Google Scholar] [CrossRef] [PubMed]
- Carlsen, K.; Jakobsen, C.; Kallemose, T.; Paerregaard, A.; Riis, L.B.; Munkholm, P.; Wewer, V. F-calprotectin and Blood Markers Correlate to Quality of Life in Pediatric Inflammatory Bowel Disease. J. Pediatric Gastroenterol. Nutr. 2017, 65, 539–545. [Google Scholar] [CrossRef] [PubMed]
- Chang, S. Disease monitoring in inflammatory bowel disease. World J. Gastroenterol. 2015, 21, 11246. [Google Scholar] [CrossRef]
- Schnapp, Z.; Hartman, C.; Livnat, G.; Shteinberg, M.; Elenberg, Y. Decreased Fecal Calprotectin Levels in Cystic Fibrosis Patients After Antibiotic Treatment for Respiratory Exacerbation. J. Pediatric Gastroenterol. Nutr. 2019, 68, 282–284. [Google Scholar] [CrossRef]
- Van Biervliet, S.; Hauser, B.; Verhulst, S.; Stepman, H.; Delanghe, J.; Warzee, J.-P.; Pot, B.; Vandewiele, T.; Wilschanski, M. Probiotics in cystic fibrosis patients: A double blind crossover placebo controlled study. Clin. Nutr. Espen 2018, 27, 59–65. [Google Scholar] [CrossRef]
| All N = 37 | Low FC Group N = 32 | High FC Group N = 5 | p | |
|---|---|---|---|---|
| Male | 22 (59.5) | 18 (56.3) | 4 (80.0) | 0.629 |
| Age (years) | 10.0 (9.2; 11.6) | 9.0 (8.8; 11.3) | 14 (7.4–18.0) | 0.146 |
| Z-Score Weight | 0.64 (0.16; 0.83) | 0.80 (0.25; 0.97) | 0.01 (−1.33; 0.95) | 0.078 |
| Z-Score Height | 1.36 (0.6; 1.7) | 1.75 (0.78; 1.94) | 0.93 (−1.84; 1.58) | 0.047 |
| Z-Score BMI | 0.09 (−0.33; 0.19) | 0.11 (−0.34; 0.22) | −0.46 (−1.03; 0.75) | 0.891 |
| CFTR mutation F508del/F508del | 21/37 (56.8) | 17/32 (53.1) | 4/5 (80.0) | 0.364 |
| Past history | ||||
| Meconial ileus | 4 (10.8) | 3 (9.4) | 1 (20.0) | 0.513 |
| DIOS | 2 (5.4) | 2 (6.3) | 0 (0.0) | 1.000 |
| CFRD | 2 (5.4) | 2 (6.3) | 0 (0.0) | 1.000 |
| Chronic colonization | ||||
| Pseudomonas aeruginosa | 9 (24.3) | 8 (25.0) | 1 (20.0) | 1.000 |
| Staphylococcus aureus | 26 (70.3) | 22 (68.8) | 4 (80.0) | 1.000 |
| Within a year prior to inclusion | ||||
| No. of patients hospitalized | 16 (43.2) | 14 (43.8) | 2 (40.0) | 1.000 |
| For Pulmonary exacerbation | 6 (16.2) | 5 (15.6) | 1 (20.0) | 1.000 |
| For recurrent IV ATB therapy | 10 (27.0) | 9 (28.1) | 1 (20.0) | 1.000 |
| No. of hospitalization per patient | 0.0 (0.6; 1.5) | 0.0 (0.5; 1.4) | 0.0 (−0.7; 3.9) | 0.362 |
| No. of IV ATB cures per patient | 0.0 (0.6; 1.5) | 0.0 (0.4; 1.4) | 1.0 (−0.2; 3.8) | 0.126 |
| Last lung function | ||||
| Time before inclusion (days) | 0.0 (1.3; 7.0) | 0.0 (1.3; 7.9) | 0.0 (−1.0; 3.4) | 0.826 |
| ppFVC | 95.0 (86.4; 95.5) | 95.0 (87.0; 96.2) | 95.0 (63.0; 111.0) | 0.657 |
| ppFEV1 | 90.0 (79.6; 91.7) | 91.0 (79.0; 92.3) | 88.0 (62.0; 108.8) | 0.824 |
| FEV1 < 80% | 14 (37.8) | 12 (37.5) | 2 (40.0) | 1.000 |
| FEV1/FVC (%) | 82.2 (76.5; 83.5) | 80.6 (75.2; 82.6) | 87.1 (76.3; 97.7) | 0.162 |
| FEV1/FVC < 80% | 14 (37.8) | 13 (40.6) | 1 (20.0) | 0.630 |
| ppFEF25–75% | 73.0 (61.8; 81.7) | 73.5 (59.5; 81.5) | 70.0 (47.2; 112.0) | 0.491 |
| ppTLC a | 94.0 (90.8; 106.8) | - | ||
| ppRV a | 99.0 (94.3; 127.8) | - | ||
| RV/TLC a | 29.0 (25.1; 32.4) | - | ||
| ppRFC a | 88.0 (86.3; 100.0) | - | ||
| ppRaw a | 66.0 (63.0; 74.5.0) | - | ||
| Treatment | ||||
| PERT U/Kg/j | 7097 (6627; 7813) | 6999 (6418; 7745) | 8496 (6786; 9420) | 0.206 |
| Proton pump inhibitor | 11 (29.7) | 9 (28.5) | 2 (40.0) | 0.623 |
| ursodeoxycolic acid | 13 (35.1) | 11 (34.4) | 2 (40.0) | 1.000 |
| Laxative treatement | 5 (13.5) | 4 (12.5) | 1 (20.0) | 0.538 |
| Inhaled antibiotic | 15 (40.5) | 13 (40.6) | 2 (40.0) | 1.000 |
| Oral ATB prophylaxis | 6 (16.2) | 5 (15.6) | 1 (20.0) | 1.000 |
| Recurrent IV ATB therapy | 15 (40.5) | 12 (32.4) | 3 (60.0) | 1.000 |
| ρ | p | |
|---|---|---|
| Gastrointestinal Symptoms Scales 3.0 PedsQLTM | ||
| By children | ||
| Total | −0.353 | 0.032 |
| Stomach Pain and Hurt | −0.165 | 0.330 |
| Stomach Discomfort When Eating | −0.274 | 0.101 |
| Food and Drink Limits | −0.135 | 0.427 |
| Trouble Swallowing | −0.361 | 0.028 |
| Heart Burn and Reflux | −0.331 | 0.046 |
| Nausea and Vomiting | −0.323 | 0.049 |
| Gas and Bloating | −0.495 | 0.002 |
| Constipation | 0.005 | 0.977 |
| Blood in Poop | −0.152 | 0.370 |
| Diarrhea | −0.130 | 0.444 |
| By parents | ||
| Total | −0.218 | 0.195 |
| Stomach Pain and Hurt | −0.057 | 0.737 |
| Stomach Discomfort When Eating | −0.358 | 0.030 |
| Food and Drink Limits | 0.037 | 0.830 |
| Trouble Swallowing | −0.115 | 0.499 |
| Heart Burn and Reflux | −0.367 | 0.026 |
| Nausea and Vomiting | −0.060 | 0.724 |
| Gas and Bloating | −0.231 | 0.169 |
| Constipation | −0.054 | 0.751 |
| Blood in Poop | 0.008 | 0.964 |
| Diarrhea | 0.047 | 0.783 |
| Quality of Life Pediatric Inventory 4.0 PedsQLTM | ||
| By children | ||
| Total | −0.206 | 0.222 |
| Physical Functioning | −0.034 | 0.841 |
| Emotional Functioning | −0.132 | 0.437 |
| Social Functioning | −0.313 | 0.049 |
| School Functioning | −0.110 | 0.516 |
| By parents | ||
| Total | −0.013 | 0.941 |
| Physical Functioning | −0.043 | 0.802 |
| Emotional Functioning | 0.091 | 0.591 |
| Social Functioning | −0.211 | 0.208 |
| School Functioning | 0.059 | 0.728 |
| QoL Total Score | Physical Functioning | Emotional Functioning | Social Functioning | School Functioning | |
|---|---|---|---|---|---|
| Obtained from children | |||||
| Gastrointestinal Total score | 0.63 a | 0.49 b | 0.44 b | 0.74 a | 0.22 |
| Stomach Pain and Hurt | 0.36 b | 0.27 | 0.22 | 0.54 a | 0.05 |
| Stomach Discomfort When Eating | 0.34 b | 0.26 | 0.21 | 0.55 a | 0.15 |
| Food and Drink Limits | 0.29 | 0.24 | 0.11 | 0.36 b | 0.23 |
| Trouble Swallowing | 0.29 | 0.25 | 0.24 | 0.42 b | 0.04 |
| Heart Burn and Reflux | 0.40 b | 0.30 | 0.28 | 0.48 a | 0.21 |
| Nausea and Vomiting | 0.37 b | 0.31 | 0.19 | 0.48 a | 0.26 |
| Gas and Bloating | 0.55 a | 0.41 b | 0.35 b | 0.55 a | 0.40 b |
| Constipation | 0.44 b | 0.47 b | 0.36 b | 0.54 a | 0.11 |
| Blood in Poop | 0.22 | 0.08 | 0.16 | 0.32 b | −0.05 |
| Diarrhea | 0.43 b | 0.38 b | 0.35 b | 0.47 b | 0.14 |
| Obtained from parents | |||||
| Gastrointestinal Total score | 0.36 b | 0.20 | 0.30 b | 0.46 b | 0.28 |
| Stomach Pain and Hurt | 0.24 | 0.10 | 0.13 | 0.25 | 0.27 |
| Stomach Discomfort When Eating | 0.12 | 0.08 | 0.14 | 0.29 | 0.06 |
| Food and Drink Limits | 0.15 | 0.13 | 0.07 | 0.35 b | 0.06 |
| Trouble Swallowing | −0.07 | −0.12 | −0.02 | 0.25 | −0.16 |
| Heart Burn and Reflux | −0.05 | 0.02 | −0.04 | 0.22 | −0.04 |
| Nausea and Vomiting | 0.22 | 0.07 | 0.07 | 0.26 | 0.25 |
| Gas and Bloating | 0.37 b | 0.12 | 0.40 b | 0.24 | 0.29 |
| Constipation | 0.37 b | 0.35 b | 0.32 b | 0.36 b | 0.25 |
| Blood in Poop | 0.08 | −0.06 | 0.08 | 0.17 | 0.16 |
| Diarrhea | 0.38 b | 0.33 b | 0.40 b | 0.33 b | 0.20 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Beaufils, F.; Mas, E.; Mittaine, M.; Addra, M.; Fayon, M.; Delhaes, L.; Clouzeau, H.; Galode, F.; Lamireau, T.; Bui, S.; et al. Increased Fecal Calprotectin Is Associated with Worse Gastrointestinal Symptoms and Quality of Life Scores in Children with Cystic Fibrosis. J. Clin. Med. 2020, 9, 4080. https://doi.org/10.3390/jcm9124080
Beaufils F, Mas E, Mittaine M, Addra M, Fayon M, Delhaes L, Clouzeau H, Galode F, Lamireau T, Bui S, et al. Increased Fecal Calprotectin Is Associated with Worse Gastrointestinal Symptoms and Quality of Life Scores in Children with Cystic Fibrosis. Journal of Clinical Medicine. 2020; 9(12):4080. https://doi.org/10.3390/jcm9124080
Chicago/Turabian StyleBeaufils, Fabien, Emmanuel Mas, Marie Mittaine, Martin Addra, Michael Fayon, Laurence Delhaes, Haude Clouzeau, François Galode, Thierry Lamireau, Stéphanie Bui, and et al. 2020. "Increased Fecal Calprotectin Is Associated with Worse Gastrointestinal Symptoms and Quality of Life Scores in Children with Cystic Fibrosis" Journal of Clinical Medicine 9, no. 12: 4080. https://doi.org/10.3390/jcm9124080
APA StyleBeaufils, F., Mas, E., Mittaine, M., Addra, M., Fayon, M., Delhaes, L., Clouzeau, H., Galode, F., Lamireau, T., Bui, S., & Enaud, R. (2020). Increased Fecal Calprotectin Is Associated with Worse Gastrointestinal Symptoms and Quality of Life Scores in Children with Cystic Fibrosis. Journal of Clinical Medicine, 9(12), 4080. https://doi.org/10.3390/jcm9124080






