Real-World Safety of CFTR Modulators in the Treatment of Cystic Fibrosis: A Systematic Review
Abstract
1. Introduction
2. Methods
2.1. Search Strategy
2.2. Selection of Studies
2.3. Data Extraction and Management
2.4. Quality Assessment
2.5. Data Analysis
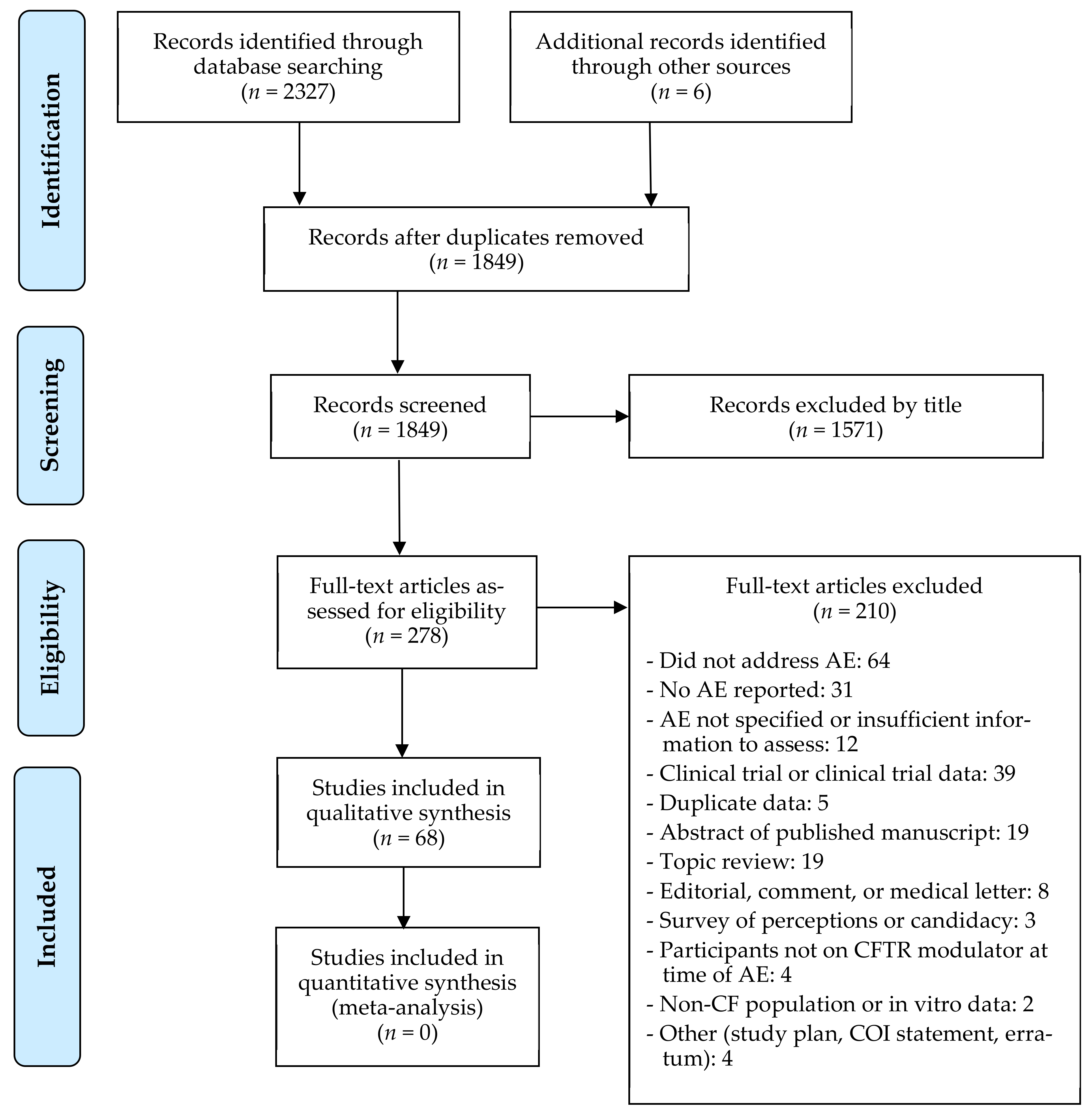
3. Results
3.1. Description of Studies
3.2. Quality Assessment
3.3. AE-Related Outcomes
3.3.1. Ivacaftor (IVA)
3.3.2. Lumacaftor/Ivacaftor (LUM/IVA)
3.3.3. Tezacaftor/Ivacaftor (TEZ/IVA)
3.3.4. Elexacaftor/Tezacaftor/Ivacaftor (ELX/TEZ/IVA)
3.3.5. Described Strategies to Address or Mitigate Reported Adverse Events
4. Discussion
4.1. Putting Real-World Adverse Events into Context
4.2. Role of the CF Pharmacist in the Safe Prescribing of CFTR Modulators
4.2.1. Selection of a CFTR Modulator
4.2.2. Patient Counseling and Education
4.2.3. Initiation and Monitoring Plan
4.3. Limitations
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Appendix A. Search Terms and Boolean Operators Used in Medline and Embase Electronic Databases
- exp Cystic Fibrosis Transmembrane Conductance Regulator/or CFTR modulator*.mp.
- ivacaftor.mp.
- lumacaftor.mp.
- tezacaftor.mp.
- elexacaftor.mp.
- kalydeco.mp.
- orkambi.mp.
- symdeko.mp.
- trikafta.mp.
- VX-770.mp.
- VX-445.mp.
- VX-809.mp.
- VX-661.mp.
- adverse event*.mp.
- side effect*.mp. or exp Treatment Outcome/
- phase IV.mp.
- phase 4.mp.
- adverse reaction*.mp.
- toxicity.mp.
- toxicities.mp.
- exp Product Surveillance, Postmarketing/or pharmacovigilance.mp. or exp Pharmacovigilance/or exp “Drug-Related Side Effects and Adverse Reactions”/or exp Adverse Drug Reaction Reporting Systems/
- post-marketing.mp.
- tolerability.mp.
- exp Drug Tolerance/or tolerance.mp.
- harm*.mp
- complication*.mp.
- drug safety.mp.
- drug hypersensitivity.mp. or exp Drug Hypersensitivity/
- 14 or 15 or 16 or 17 or 18 or 19 or 20 or 21 or 22 or 23 or 24 or 25 or 26 or 27 or 28
- 1 or 2 or 3 or 4 or 5 or 6 or 7 or 8 or 9 or 10 or 11 or 12 or 13
- 29 and 30
- exp cystic fibrosis transmembrane conductance regulator/or CFTR modulator *.mp.
- exp ivacaftor plus tezacaftor/or exp ivacaftor/or exp ivacaftor plus lumacaftor/or exp elexacaftor plus ivacaftor plus tezacaftor/or ivacaftor.mp.
- tezacaftor.mp. or exp tezacaftor/
- exp lumacaftor/or lumacaftor.mp.
- exp elexacaftor/or elexacaftor.mp.
- kalydeco.mp.
- orkambi.mp.
- symdeko.mp.
- symkevi.mp.
- trikafta.mp.
- VX-770.mp.
- VX-445.mp.
- VX-809.mp.
- VX-661.mp.
- 1 or 2 or 3 or 4 or 5 or 6 or 7 or 8 or 9 or 10 or 11 or 12 or 13 or 14
- exp adverse event/or adverse event*.mp. or exp adverse drug reaction/
- exp side effect/or side effect*.mp.
- adverse reaction*.mp. or exp drug hypersensitivity/
- toxicity/or toxicity.mp. or exp drug toxicity/
- toxicities.mp.
- post-marketing.mp. or exp drug surveillance program/or exp postmarketing surveillance/
- exp drug safety/or exp pharmacovigilance/or pharmacovigilance.mp.
- phase 4.mp.
- exp phase 4 clinical trial/or phase IV.mp.
- drug tolerance.mp. or exp drug tolerance/
- drug hypersensitivity.mp.
- complication*.mp.
- exp patient harm/or harm*.mp.
- 16 or 17 or 18 or 19 or 20 or 21 or 22 or 23 or 24 or 25 or 26 or 27 or 28
- 15 and 29
Appendix B
| Criteria | Burgel et al. 2020 [42] | Diab-Cáceres et al. 2018 [34] | Dryden et al. 2018 [57] | Finnegan et al. 2020 [43] | Gomez-Pastrana et al. 2019 [56] | Hebestreit et al. 2013 [30] | Hubert et al. 2017 [33] |
| 1. Study objective clearly stated | Y | Y | Y | Y | Y | Y | Y |
| 2. Study population clearly defined | Y | Y | N | Y | Y | Y | Y |
| 3. Participation ≥ 50% of eligible persons | Y | CD | CD | Y | Y | Y | Y |
| 4. Study subjects from same or similar population, inclusion/exclusion criteria pre-specified and uniformly applied | Y | Y | Y | Y | Y | Y | Y |
| 5. Sample size justification, power description, or variance and effect estimates provided | N/A | N/A | N/A | N/A | N/A | N/A | N/A |
| 6. Exposure of interest measured prior to outcome assessed | Y | Y | Y | Y | Y | Y | Y |
| 7. Timeframe sufficient to observe association between exposure/outcome | Y | Y | Y | Y | Y | Y | Y |
| 8. Study examined different levels of exposure as related to outcome | N/A | N/A | N/A | N/A | N/A | N/A | N/A |
| 9. Exposure measures clearly defined, valid, reliable, implemented consistently | N/A | N/A | N/A | N/A | N/A | N/A | N/A |
| 10. Exposure assessed more than once | N/A | N/A | N/A | N/A | N/A | N/A | N/A |
| 11. Outcome measures clearly defined, valid, reliable, implemented consistently | Y | Y | N | Y | CD | CD | Y |
| 12. Outcome assessors blinded to exposure status of study subjects | N/A | N/A | N/A | N/A | N/A | N/A | N/A |
| 13. Loss to follow-up ≤ 20% | Y | Y | Y | Y | Y | Y | Y |
| 14. Key potential confounders measured and adjusted statistically for impact on relationship between exposure/outcome | Y | N | N | Y | N | N | N |
| Final rating | GOOD | GOOD | FAIR | GOOD | FAIR | FAIR | GOOD |
| Criteria | Hubert et al. 2018 [58] | Jennings et al. 2017 [44] | Kopp et al. 2018 [59] | Labaste et al. 2017 [41] | Loukou et al. 2020 [60] | Murer et al. 2018 [31] | Pohl et al. 2018 [61] |
| 1. Study objective clearly stated | Y | Y | Y | Y | Y | Y | Y |
| 2. Study population clearly defined | Y | Y | Y | Y | Y | Y | Y |
| 3. Participation ≥ 50% of eligible persons | CD | Y | CD | CD | Y | Y | Y |
| 4. Study subjects from same or similar population, inclusion/exclusion criteria pre-specified and uniformly applied | Y | Y | CD | CD | Y | Y | Y |
| 5. Sample size justification, power description, or variance and effect estimates provided | N/A | N/A | N/A | N/A | N/A | N/A | N/A |
| 6. Exposure of interest measured prior to outcome assessed | Y | Y | Y | Y | Y | Y | Y |
| 7. Timeframe sufficient to observe association between exposure/outcome | Y | Y | Y | Y | Y | Y | Y |
| 8. Study examined different levels of exposure as related to outcome | N/A | N/A | N/A | N/A | N/A | N/A | N/A |
| 9. Exposure measures clearly defined, valid, reliable, implemented consistently | N/A | N/A | N/A | N/A | N/A | N/A | N/A |
| 10. Exposure assessed more than once | N/A | N/A | N/A | N/A | N/A | N/A | N/A |
| 11. Outcome measures clearly defined, valid, reliable, implemented consistently | CD | CD | N | Y | CD | Y | CD |
| 12. Outcome assessors blinded to exposure status of study subjects | N/A | N/A | N/A | N/A | N/A | N/A | N/A |
| 13. Loss to follow-up ≤ 20% | Y | Y | Y | Y | Y | Y | Y |
| 14. Key potential confounders measured and adjusted statistically for impact on relationship between exposure/outcome | N | Y | N | Y | N | N | Y |
| Final rating | FAIR | GOOD | POOR | GOOD | FAIR | GOOD | GOOD |
| Criteria | Popowicz et al. 2017 [32] | Sergeev et al. 2019 [45] | Stalling et al. 2018 [55] | Taylor-Cousar et al. 2016 [29] | Tong et al. 2019 [26] | Wark et al. 2019 [25] | |
| 1. Study objective clearly stated | Y | Y | Y | Y | Y | Y | |
| 2. Study population clearly defined | Y | Y | Y | Y | Y | Y | |
| 3. Participation ≥ 50% of eligible persons | Y | Y | N | CD | Y | Y | |
| 4. Study subjects from same or similar population, inclusion/exclusion criteria pre-specified and uniformly applied | Y | Y | Y | Y | Y | Y | |
| 5. Sample size justification, power description, or variance and effect estimates provided | N/A | N/A | N/A | N/A | N/A | N/A | |
| 6. Exposure of interest measured prior to outcome assessed | Y | Y | Y | Y | Y | Y | |
| 7. Timeframe sufficient to observe association between exposure/outcome | Y | Y | Y | Y | Y | Y | |
| 8. Study examined different levels of exposure as related to outcome | N/A | N/A | N/A | N/A | N/A | N/A | |
| 9. Exposure measures clearly defined, valid, reliable, implemented consistently | N/A | N/A | N/A | N/A | N/A | N/A | |
| 10. Exposure assessed more than once | N/A | N/A | N/A | N/A | N/A | N/A | |
| 11. Outcome measures clearly defined, valid, reliable, implemented consistently | Y | N | N | CD | CD | N | |
| 12. Outcome assessors blinded to exposure status of study subjects | N/A | N/A | N/A | N/A | N/A | N/A | |
| 13. Loss to follow-up ≤ 20% | Y | Y | Y | Y | Y | Y | |
| 14. Key potential confounders measured and adjusted statistically for impact on relationship between exposure/outcome | N | N | N | N | Y | N | |
| Final rating | GOOD | FAIR | POOR | FAIR | GOOD | FAIR |
Appendix C
| Criteria | McKinzie et al., 2017 [47] | Nash et al., 2020 [27] | Rotolo et al., 2020 [52] | Safirstein et al., 2020 [53] | Talwalkar et al., 2017 [48] |
|---|---|---|---|---|---|
| 1. Study objective clearly stated | Y | Y | N | Y | Y |
| 2. Study population clearly defined, using case definition | N | N | N | N | N |
| 3. Cases consecutive | NR | NR | NR | NR | NR |
| 4. Subjects comparable | CD | CD | CD | N | N |
| 5. Intervention clearly described | Y | Y | Y | Y | Y |
| 6. Outcome measures clearly defined, valid, reliable, implemented consistently | N | N | N | Y | N |
| 7. Adequate length of follow-up | Y | Y | Y | Y | CD |
| 8. Statistical methods well-described | N/A | N/A | N/A | N/A | N |
| 9. Results well-described | Y | N | Y | Y | Y |
| Final rating | Poor | Poor | Fair | Good | Poor |
References
- Elborn, J.S. Cystic Fibrosis. Lancet 2016, 388, 2519–2531. [Google Scholar] [CrossRef]
- Sawicki, G.S.; Sellers, D.E.; Robinson, W.M. High treatment burden in adults with cystic fibrosis: Challenges to disease self-management. J. Cyst. Fibros. 2009, 8, 91–96. [Google Scholar] [CrossRef]
- Cystic Fibrosis Foundation. Your CF Care Team. Available online: https://www.cff.org/Care/Your-CF-Care-Team/ (accessed on 12 October 2020).
- McIntosh, I.D. Health Human Resources Guidelines: Minimum Staffing Standards for CF Healthcare Teams. Cystic Fibrosis Canada. 2014. Available online: https://www.cysticfibrosis.ca/uploads/cf%20care/Health-Human-Resources-Guidelines.pdf (accessed on 12 October 2020).
- Conway, S.; Balfour-Lynn, I.M.; De Rijcke, K.; Drevinek, P.; Foweraker, J.; Havermans, T.; Heijerman, H.; Lannefors, L.; Lindblad, A.; Macek, M.; et al. European Cystic Fibrosis Society standards of care: Framework for the cystic fibrosis centre. J. Cyst. Fibros. 2014, 13, S3–S22. [Google Scholar] [CrossRef]
- Redfern, J.; Webb, A.K. Benefits of a dedicated cystic fibrosis pharmacist. J. R. Soc. Med. 2004, 97, 2–7. [Google Scholar] [PubMed]
- Habib, A.-R.R.; Kajbafzadeh, M.; Desai, S.; Yang, C.L.; Skolnik, K.; Quon, B.S. A systematic review of the clinical efficacy and safety of CFTR modulators in cystic fibrosis. Sci. Rep. 2019, 9, 7234:1–7234:9. [Google Scholar] [CrossRef]
- Vertex. Kalydeco® Product Monograph. 2019. Available online: https://pi.vrtx.com/files/Canadapm_kalydeco_en.pdf (accessed on 21 September 2020).
- Vertex. Orkambi® Product Monograph. 2019. Available online: https://pi.vrtx.com/files/Canadapm_orkambi_en.pdf (accessed on 21 September 2020).
- Vertex. Symdeko® Product Monograph. 2020. Available online: https://pi.vrtx.com/files/Canadapm_symdeko_en.pdf (accessed on 21 September 2020).
- European Medicines Agency. Symkevi. 2020. Available online: https://www.ema.europa.eu/en/medicines/human/EPAR/Symkevi (accessed on 21 September 2020).
- Vertex. Trikafta® Prescribing Information. 2020. Available online: https://pi.vrtx.com/files/uspi_elexacaftor_tezacaftor_ivacaftor.pdf (accessed on 21 September 2020).
- European Medicines Agency. Kaftrio. 2020. Available online: https://www.ema.europa.eu/en/medicines/human/EPAR/Kaftrio (accessed on 7 November 2020).
- Davies, J.C.; Wainwright, C.E.; Canny, G.J.; Chilvers, M.A.; Howenstine, M.S.; Munck, A.; Mainz, J.G.; Rodriguez, S.; Li, H.; Yen, K. Efficacy and safety of ivacaftor in patients aged 6 to 11 years with cystic fibrosis with a G551D mutation. Am. J. Respir. Crit. Care Med. 2013, 187, 1219–1225. [Google Scholar] [CrossRef] [PubMed]
- De Boeck, K.; Munck, A.; Walker, S.; Faro, A.; Hiatt, P.; Gilmartin, G.; Higgins, M. Efficacy and safety of ivacaftor in patients with cystic fibrosis and a non-G551D gating mutation. J. Cyst. Fibros. 2014, 13, 674–680. [Google Scholar] [CrossRef] [PubMed]
- Ramsey, B.W.; Davies, J.; McElvaney, N.G.; Tullis, E.; Bell, S.C.; Drevinek, P.; Griese, M.; McKone, E.F.; Wainwright, C.E.; Konstan, M.W. A CFTR potentiator in patients with cystic fibrosis and the G551D mutation. N. Eng. J. Med. 2011, 365, 1663–1672. [Google Scholar] [CrossRef] [PubMed]
- Moss, R.B.; Flume, P.A.; Elborn, J.S.; Cooke, J.; Rowe, S.M.; McColley, S.A.; Rubenstein, R.C.; Higgins, M. Efficacy and safety of ivacaftor treatment: Randomized trial in subjects with cystic fibrosis who have an R117H-CFTR mutation. Lancet Respir. Med. 2015, 3, 524–533. [Google Scholar] [CrossRef]
- Rowe, S.M.; Daines, C.; Ringshausen, F.C.; Kerem, E.; Wilson, J.; Tullis, E.; Nair, N.; Simard, C.; Han, L.; Ingenito, E.P.; et al. Tezacaftor-ivacaftor in residual-function heterozygotes with cystic fibrosis. N. Eng. J. Med. 2017, 377, 2024–2035. [Google Scholar] [CrossRef]
- Wainwright, C.E.; Elborn, J.S.; Ramsey, B.W.; Marigowda, G.; Huang, X.; Cipolli, M.; Colombo, C.; Davies, J.C.; De Boeck, K.; Flume, P.A.; et al. Lumacaftor-ivacaftor in patients with cystic fibrosis homozygous for Phe508del CFTR. N. Eng. J. Med. 2015, 373, 220–231. [Google Scholar] [CrossRef] [PubMed]
- Ratjen, F.; Hug, C.; Marigowda, G.; Tian, S.; Huang, X.; Stanojevic, S.; Milla, C.E.; Robinson, P.D.; Waltz, D.; Davies, J.C. Efficacy and safety of lumacaftor and ivacaftor in patients aged 6–11 years with cystic fibrosis homozygous for F508del-CFTR: A randomized, placebo-controlled, phase 3 trial. Lancet Respir. Med. 2017, 5, 557–567. [Google Scholar] [CrossRef]
- Taylor-Cousar, J.L.; Munck, A.; McKone, E.F.; Van der Ent, C.K.; Moeller, A.; Simard, C.; Wang, L.T.; Ingenito, E.P.; McKee, C.; Lu, Y.; et al. Tezacaftor-ivacaftor in patients with cystic fibrosis homozygous for Phe508del. N. Eng. J. Med. 2017, 377, 2013–2023. [Google Scholar] [CrossRef] [PubMed]
- Heijerman, H.G.; McKone, E.F.; Downey, D.G.; Van Braeckel, E.; Rowe, S.M.; Tullis, E.; Mall, M.A.; Welter, J.J.; Ramsey, B.W.; McKee, C.M.; et al. Efficacy and safety of the elexacaftor plus tezacaftor plus ivacaftor combination regimen in people with cystic fibrosis homozygous for the F508del mutation: A double-blind, randomised, phase 3 trial. Lancet 2019, 394, 1940–1948. [Google Scholar] [CrossRef]
- Middleton, P.G.; Mall, M.A.; Drevinek, P.; Lands, L.C.; McKone, E.F.; Polineni, D.; Ramsey, B.W.; Taylour-Cousar, J.L.; Tullis, E.; Vermeulen, F.; et al. Elexacaftor-tezacaftor-ivacaftor for cystic fibrosis with a single Phe508del allele. N. Eng. J. Med. 2019, 381, 1809–1819. [Google Scholar] [CrossRef] [PubMed]
- NIH National Heart, Lung, and Blood Institute. Study Quality Assessment Tools. Available online: https://www.nhlbi.nih.gov/health-topics/study-quality-assessment-tools (accessed on 15 July 2020).
- Wark, P.A.; Cookson, K.; Thiruchelvam, T.; Brannan, J.; Dorahy, D.J. Lumacaftor/ivacaftor improves exercise tolerance in patients with cystic fibrosis and severe airflow obstruction. BMC Pulm. Med. 2019, 19, 106:1–106:8. [Google Scholar] [CrossRef]
- Tong, K.; Barker, D.; France, M.; Burr, L.; Greville, H.; Visser, S.; Middleton, P.; Wainwright, C.; Dorahy, D.; Wark, P. Lumacaftor/ivacaftor reduces exacerbations in adults homozygous for Phe508del mutation with severe lung disease. J. Cyst. Fibros. 2020, 19, 415–420. [Google Scholar] [CrossRef]
- Nash, E.F.; Middleton, P.G.; Taylor-Cousar, J.L. Outcomes of pregnancy in women with cystic fibrosis (CF) taking CFTR modulators—An international survey. J. Cyst. Fibros. 2020, 19, 521–526. [Google Scholar] [CrossRef]
- Patterson, A.; Autry, E.; Kuhn, R.; Murth, M. Ivacaftor drug desensitization. Pediatr. Pulmonol. 2019, 54, 672–674. [Google Scholar] [CrossRef]
- Taylor-Cousar, J.; Niknian, M.; Gilmartin, G.; Pilewski, J.M. Effect of ivacaftor in patients with advanced cystic fibrosis and a G551D-CFTR mutation: Safety and efficacy in an expanded access program in the United States. J. Cyst. Fibros. 2016, 15, 116–122. [Google Scholar] [CrossRef]
- Hebestreit, H.; Sauer-Heilborn, A.; Fischer, R.; Kading, M.; Mainz, J.G. Effects of ivacaftor on severely ill patients with cystic fibrosis carrying a G551D mutation. J. Cyst. Fibros. 2013, 12, 599–603. [Google Scholar] [CrossRef] [PubMed]
- Murer, C.; Huber, L.C.; Kurowski, T.; Hirt, A.; Robinson, C.; Bɒrgi, U.; Benden, C. First experience in Switzerland in Phe508del homozygous cystic fibrosis patients with end-stage pulmonary disease enrolled in a lumacaftor-ivacaftor therapy trial—Preliminary results. Swiss Med. Wkly. 2018, 148, w14593:1–w14593:5. [Google Scholar] [CrossRef]
- Popowicz, N.; Wood, J.; Tai, A.; Morey, S.; Mulrennan, S. Immediate effects of lumacaftor/ivacaftor administration on lung function in patients with severe cystic fibrosis lung disease. J. Cyst. Fibros. 2017, 16, 392–394. [Google Scholar] [CrossRef] [PubMed]
- Hubert, D.; Chiron, R.; Camara, B.; Grenet, D.; Prévotat, A.; Bassinet, L.; Dominique, S.; Rault, G.; Macey, J.; Honoré, I.; et al. Real-life initiation of lumacaftor/ivacaftor combination in adults with cystic fibrosis homozygous for the Phe508del CFTR mutation and severe lung disease. J. Cyst. Fibros. 2017, 16, 388–391. [Google Scholar] [CrossRef]
- Diab-Cáceres, L.; Girón-Moreno, R.M.; Pastor-Sanz, M.T.; Quintana-Gallego, E.; Delgado-Pecellín, I.; Blanco-Aparicio, B.; Maiz, L.; García-Clemente, M.M.; Luna-Paredes, C.; Mondéjar-Lopez, P.; et al. Compassionate use of lumacaftor/ivacaftor in cystic fibrosis: Spanish experience. Arch. Bronconeumol. 2018, 54, 614–618. [Google Scholar] [CrossRef]
- Etherington, C.; Graham, T.; Spoletini, G.; Shaw, N.; Whitaker, P.; Clifton, I.; Peckham, D. Early experience of the Orkambi “managed Access Programme” in a regional adult UK CF centre. In Proceedings of the 40th European Cystic Fibrosis Conference, Seville, Spain, 7–10 June 2017; p. 56. [Google Scholar]
- Wareham, J.M.; Webb, K.A.; Jones, A.M.; Brennan, A.L.; Bright-Thomas, R.J.; Horsley, A.R.; Barry, P.J. Lumacaftor/ivacaftor is associated with high discontinuation rates in patients with baseline severe lung function but also benefits in those who tolerate therapy. In Proceedings of the British Thoracic Society Winter Meeting, London, UK, 6–8 December 2017; p. 247. [Google Scholar]
- Wadsworth, L.; Riley, D.; Johnson, S.; Barry, P. Physiotherapeutic objective and subjective evaluation of lumacaftor/ivacaftor initiation in patients with severe lung disease in a large adult CF centre. In Proceedings of the 33rd Annual North American Cystic Fibrosis Conference, Nashville, TN, USA, 31 October–2 November 2019; p. 468. [Google Scholar]
- Carter, S.C.; Kearns, S.; Grogan, B.; Hisert, K.B.; Cooke, G.; Singh, P.K.; Gallagher, C.G.; McKone, E.F. Effects of lumacaftor/ivacaftor in patients homozygous for F508del mutation with very advanced lung disease. In Proceedings of the 40th European Cystic Fibrosis Conference, Seville, Spain, 7–10 June 2017; p. 47. [Google Scholar]
- Robinson, R.; FitzMaurice, T.; Shaw, M.; Dawood, S.; Nazareth, D.; Walshaw, M. Symkevi: Real-world experience in an ill cohort at an adult cystic fibrosis centre. In Proceedings of the 43rd European Cystic Fibrosis Conference, Lyon, France, 3–6 June 2020; p. 219. [Google Scholar]
- Bowen, M.; Alemayehu, M.; Battle, E.; Brown, A.W. Promising results with elexacaftor/tezacaftor/ivacaftor use in cystic fibrosis patients with advanced lung disease: Beyond the clinical trial inclusion criteria. In Proceedings of the American Thoracic Society 2020 International Conference, Philadelphia, PA, USA, 15–20 May 2020; 1164. Available online: https://www.atsjournals.org/doi/book/10.1164/ajrccm-conference.2020.D12 (accessed on 1 August 2020).
- Labaste, A.; Ohlmann, C.; Mainguy, C.; Jubin, V.; Perceval, M.; Coutier, L.; Reix, P. Real-life acute lung function changes after lumacaftor/ivacaftor first administration in pediatric patients with cystic fibrosis. J. Cyst. Fibros. 2017, 16, 709–712. [Google Scholar] [CrossRef]
- Burgel, P.R.; Munck, A.; Durieu, I.; Chiron, R.; Mely, L.; Prevotat, A.; Murris-Espin, M.; Porzio, M.; Abely, M.; Reix, P.; et al. Real-life safety and effectiveness of lumacaftor-ivacaftor in patients with cystic fibrosis. Am. J. Respir. Crit. Care Med. 2020, 201, 188–197. [Google Scholar] [CrossRef]
- Finnegan, R.; O’Grady, E.; Smyth, A.; Ryan, S.; Williamson, M. Evidence of small airways disease and the immediate effects of lumacaftor/ivacaftor in children with cystic fibrosis. Ir. Med. J. 2020, 113, 70:1–70:7. [Google Scholar]
- Jennings, M.T.; Dezube, R.; Paranjape, S.; West, N.E.; Hong, G.; Braun, A.; Grant, J.; Merlo, C.A.; Lechtzin, N. An observational study of outcomes and tolerances in patients with cystic fibrosis initiated on lumacaftor/ivacaftor. Ann. Am. Thorac. Soc. 2017, 14, 1662–1666. [Google Scholar] [CrossRef] [PubMed]
- Sergeev, V.; Desai, S.; Flores, E.; Kerr, J.; Su, V.; Wilcox, P.; Quon, B.S. Safety and effectiveness of lumacaftor-ivacaftor in adults with cystic fibrosis: A single-centre Canadian experience. Can. J. Respir. Crit. Care Sleep Med. 2019, 4, 174–179. [Google Scholar] [CrossRef]
- Welsner, M.; Strasburg, S.; Taube, C.; Sivagurunathan, S. Use of ivacaftor in late diagnosed cystic fibrosis monozygotic twins heterozygous for F508del and R117H-7T: A case report. BMC Pulm. Med. 2019, 19, 76:1–76:6. [Google Scholar] [CrossRef] [PubMed]
- McKinzie, C.J.; Goralski, J.L.; Noah, T.L.; Retsch-Bogart, G.Z.; Prieur, M.B. Worsening anxiety and depression after initiation of lumacaftor/ivacaftor combination therapy in adolescent females with cystic fibrosis. J. Cyst. Fibros. 2017, 16, 525–527. [Google Scholar] [CrossRef] [PubMed]
- Talwalkar, J.S.; Koff, J.L.; Lee, H.B.; Britto, C.J.; Mulenos, A.M.; Georgiopoulos, A.M. Cystic fibrosis transmembrane regulator modulators: Implications for the management of depression and anxiety in cystic fibrosis. Psychosomatics 2017, 58, 343–354. [Google Scholar] [CrossRef] [PubMed]
- Walayat, S.; Hussain, N.; Patel, J.; Hussain, F.; Patel, P.; Dhillon, S.; Aulakh, B.; Chittivelu, S. Drug-induced dyspnea versus cystic fibrosis exacerbation: A diagnostic dilemma. Int. Med. Case Rep. J. 2017, 10, 243–246. [Google Scholar] [CrossRef]
- Roehmel, J.F.; Ogese, M.O.; Rohrbach, A.; Mall, M.A.; Naisbitt, D.J. Drug allergy to CFTR modulator therapy associated with lumacaftor-specific CD4+ T lymphocytes. J. Allergy Clin. Immunol. 2020. [Google Scholar] [CrossRef]
- Song, Y.; Palacios, A.C.; Thiagalingam, A.; Middleton, P.G. Azithromycin and tezacaftor/ivacaftor is associated with first-degree heart block in an adult with cystic fibrosis. J. Cyst. Fibros. 2020. [Google Scholar] [CrossRef] [PubMed]
- Rotolo, S.M.; Duehlmeyer, S.; Slack, S.M.; Jacobs, H.R.; Heckman, B. Testicular pain following initiation of elexacaftor/tezacaftor/ivacaftor in males with cystic fibrosis. J. Cyst. Fibros. 2020, 19, e39–e41. [Google Scholar] [CrossRef] [PubMed]
- Safirstein, J.; Grant, J.J.; Clausen, E.; Savant, D.; Dezube, R.; Hong, G. Biliary disease and cholecystectomy after initiation of elexacaftor/ivacaftor/tezacaftor in adults with cystic fibrosis. J. Cyst. Fibros. 2020. [Google Scholar] [CrossRef] [PubMed]
- Tindell, W.; Su, A.; Oros, S.M.; Rayapati, A.O.; Rakesh, G. Trikafta and psychopathology in cystic fibrosis: A case report. Psychosomatics 2020, 61, 735–738. [Google Scholar] [CrossRef] [PubMed]
- Stalling, V.A.; Sainath, N.; Oberle, M.; Bertolaso, C.; Schall, J.I. Energy balance and mechanisms of weight gain with ivacaftor treatment of cystic fibrosis gating mutations. J. Pediatr. 2018, 201, 229–237. [Google Scholar] [CrossRef] [PubMed]
- Gomez-Pastrana, D.; Nwokoro, C.; McLean, M.; Brown, S.; Christiansen, N.; Pao, C.S. Real-world effectiveness of ivacaftor in children with cystic fibrosis and the G551D mutation. An. Pediatr. (Barc.) 2019, 90, 148–156. [Google Scholar] [CrossRef] [PubMed]
- Dryden, C.; Wilkinson, J.; Young, D.; Brooker, J.R. The impact of 12 months treatment with ivacaftor on Scottish paediatric patients with cystic fibrosis with the G115D mutation: A review. Arch. Dis. Child. 2018, 103, 68–70. [Google Scholar] [CrossRef] [PubMed]
- Hubert, D.; Dehillotte, C.; Munck, A.; David, V.; Baek, J.; Mely, L.; Dominique, S.; Ramel, S.; Danner-Boucher, I.; Lefeuvre, S.; et al. Retrospective observational study of French patients with cystic fibrosis and a Gly551Asp-CFTR mutation after 1 and 2 years of treatment with ivacaftor in a real-world setting. J. Cyst. Fibros. 2018, 17, 89–95. [Google Scholar] [CrossRef]
- Kopp, B.T.; McCulloch, S.; Shrestha, C.L.; Zhang, S.; Sarzynski, L.; Woodley, F.W.; Hayes, D., Jr. Metabolomic responses to lumacaftor/ivacaftor in cystic fibrosis. Pediatr. Pulmonol. 2018, 53, 583–591. [Google Scholar] [CrossRef] [PubMed]
- Loukou, I.; Moustaki, M.; Plyta, M.; Douros, K. Longitudinal changes in lung function following initiation of lumacaftor/ivacaftor combination. J. Cyst. Fibros. 2020, 19, 534–539. [Google Scholar] [CrossRef]
- Pohl, K.; Nichols, D.P.; Taylor-Cousar, J.L.; Saavedra, M.T.; Strand, M.J.; Nick, J.A.; Bratcher, P.E. Corticosteroid use and increased CXCR2 levels on leukocytes are associated with lumacaftor/ivacaftor discontinuation in cystic fibrosis patients homozygous for the F508del CFTR mutation. PLoS ONE 2018, 13, e0209026:1–e0209026:19. [Google Scholar] [CrossRef]
- De Gregorio, F.; Tosco, A.; Casale, A.; Sepe, A.; Cimbalo, C.; Catzola, A.; Castaldo, A.; Slavadori, L.; Raia, V. Efficacy and safety of Kalydeco oral granules: The experience of our centre. In Proceedings of the 23rd Italian Congress of Cystic Fibrosis and the 13th National Congress of Cystic Fibrosis Italian Society, Naples, Italy, 22–25 November 2017; p. 29. [Google Scholar]
- Robson, E.A.; Feltbower, R.; Lee, T. Real world ivacaftor efficacy in children: Five years on…. In Proceedings of the 42nd European Cystic Fibrosis Conference, Liverpool, UK, 5–8 June 2019; p. 252. [Google Scholar]
- Al-Rashdi, Z.; Al-Busaidi, N. The effect of ivacaftor on adult cystic fibrosis patients at the Royal Hospital in Oman. In Proceedings of the 42nd European Cystic Fibrosis Conference, Liverpool, UK, 5–8 June 2019; p. 256. [Google Scholar]
- Spencer-Clegg, E.; Sapina-Vivo, R.; Cullen, D.; Lloyd, E.; Walshaw, M.J. Ivacaftor in the real world-early experience in a large adult CF centre. In Proceedings of the 27th Annual North American Cystic Fibrosis Conference, Salt Lake City, UT, USA, 17–19 October 2013; p. 228. [Google Scholar]
- Flanagan, M.; Donovan, D.; Murphy, C.; Keating, E.; Jennings, R.; Shanahan, P.; Crowley, J.; Cronin, K.; Mullane, D.; Ni Chroinin, M. Orkambi: The Cork pediatric experience Cork University Hospital. In Proceedings of the Irish Thoracic Society Annual Scientific Meeting, Limerick, Ireland, 10–11 November 2017. [Google Scholar]
- McKinney, M.L.; Noizelier, S.; Tellier, I.; Marcottee, J.; Berube, D.; Tse, S. Change in FEV1 and bronchodilator response after first dose of lumacaftor/ivacaftor. In Proceedings of the 31st Annual North American Cystic Fibrosis Conference, Indianapolis, IN, USA, 2–4 November 2017; p. 314. [Google Scholar]
- Chiron, R.; Hubert, D.; Reix, P.; Collas-Aubert, V.; Tian, S.; Kiefer, P.; Kinnman, N. Lumacaftor/ivacaftor (LUM/IVA) treatment in patients (pts) with cystic fibrosis (CF) aged ≥12 years homozygous for F508del-CFTR: Description of a French Temporary Authorization for Use (ATU) cohort. In Proceedings of the 40th European Cystic Fibrosis Conference, Seville, Spain, 7–10 June 2017; p. 49. [Google Scholar]
- Brokaar, E.; Van Leeuwen, M.; Leegwater, E.; Ploeger, M.; Van der Meer, R. Adverse drug reactions and discontinuation rate during the first year on Orkambi—The earliest results of the STORM study. In Proceedings of the 42nd European Cystic Fibrosis Conference, Liverpool, UK, 5–8 June 2019; p. 260. [Google Scholar]
- Anstead, M.; Tupayachi, G.; Murphy, D.; Autry, E.; Bulkley, V.; Kuhn, R. Lumacaftor/ivacaftor: Real world experience in a CF center. In Proceedings of the 30th Annual North American Cystic Fibrosis Conference, Orlando, FL, USA, 27–29 October 2016; p. 290. [Google Scholar]
- Taylor, T.; Brown, R.F.; Tolle, J. Lumacaftor-ivacaftor tolerance in cystic fibrosis patients homozygous for Phe508del CFTR: A single-center observational study. In Proceedings of the 30th Annual North American Cystic Fibrosis Conference, Orlando, FL, USA, 27–29 October 2016; p. 264. [Google Scholar]
- Ahmad, D.; Zanni, R.; Marmolegos, G.; Allwein, L.; Kitch, D.; Brosius, H.; Isenberg, M.; Patel, M.; Mills, N.; Stephen, M.J. Side effect profile of lumacaftor/ivacaftor. In Proceedings of the 31st Annual North American Cystic Fibrosis Conference, Indianapolis, IN, USA, 2–4 November 2017; p. 310. [Google Scholar]
- Hubert, D.; Kanaan, R.; Honoré, I.; Martin, C.; Burgel, P. Reasons for not starting or discontinuing lumacaftor/ivacaftor: A single center observational study. In Proceedings of the 31st Annual North American Cystic Fibrosis Conference, Indianapolis, IN, USA, 2–4 November 2017; p. 224. [Google Scholar]
- Walker, S.D.; Henry, B.; Sueblinvong, V.; Hunt, W.R. Experience of a large academic CF center with lumcaftor/ivacaftor. In Proceedings of the 30th Annual North American Cystic Fibrosis Conference, Orlando, FL, USA, 27–29 October 2016; p. 291. [Google Scholar]
- Wang, J.; Lester, M.K.; Benitez, D.; Rao, A.; Beringer, P. Use of in-clinic challenge dosing to assess safety and tolerability of lumcaftor/ivacaftor (L/I). In Proceedings of the 30th Annual North American Cystic Fibrosis Conference, Orlando, FL, USA, 27–29 October 2016; p. 448. [Google Scholar]
- Indihar, M.V.; Trapnell, J.P.; Burns, L.A.; Meyers, M.; Major, T.; Montag, K. Early experience initiating Orkambi therapy in adult cystic fibrosis patients. In Proceedings of the 30th Annual North American Cystic Fibrosis Conference, Orlando, FL, USA, 27–29 October 2016; p. 286. [Google Scholar]
- Albon, D.; Cagnina, E. Patient factors contributing to Orkambi discontinuation in adult cystic fibrosis, single center experience. In Proceedings of the 31st Annual North American Cystic Fibrosis Conference, Indianapolis, IN, USA, 2–4 November 2017; p. 305. [Google Scholar]
- Ahmad, D.; Patel, M.; Mills, N.; Stephen, M.J. Real world experience of lumacaftor-ivacaftor at an urban cystic fibrosis center. In Proceedings of the 30th Annual North American Cystic Fibrosis Conference, Orlando, FL, USA, 27–29 October 2016; p. 32. [Google Scholar]
- Roth, K.; Novak, K.J. Retrospective review of proton pump inhibitor use in cystic fibrosis patients initiating lumacaftor-ivacaftor (Orkambi) treatment. In Proceedings of the 31st Annual North American Cystic Fibrosis Conference, Indianapolis, IN, USA, 2–4 November 2017; p. 624. [Google Scholar]
- Wang, J.; Rao, A.; Beringer, P. Efficacy of lumacaftor/ivacaftor in patients with mild lung disease. In Proceedings of the 31st Annual North American Cystic Fibrosis Conference, Indianapolis, IN, USA, 2–4 November 2017; p. 40. [Google Scholar]
- Paluck, F.; Linnane, B. Liver tests in F508del homozygous cystic fibrosis patients on Orkambi. In Proceedings of the 9th Eruopaediatrics Congress of Royal College of Paediatrics and Child Health, Dublin, Ireland, 13–15 June 2019; p. 274. [Google Scholar]
- Melotti, P.; Tridello, G.; Pintani, E.; Meneghelli, I.; Volpi, S.; Spinelli, E.; D’Orazio, C. Effects of ivacaftor/lumacaftor in cystic fibrosis patients homozygous for F508del CFTR mutation. In Proceedings of the 24th Congress of the Italian Society of Cystic Fibrosis and the 14th National Congress of Cystic Fibrosis Italian Society, Salerno, Italy, 8–10 November 2018; p. 251. [Google Scholar]
- Bourgani, E.; Stagaki, E.; Gioka, M.; Nakou, A.; Antonogiannaki, E.-M.; Padapopoulos, D.; Diamantea, F. Two years’ experience of lumacaftor/ivacaftor treatment at an adult cystic fibrosis centre in Athens, Greece. In Proceedings of the 42nd European Cystic Fibrosis Conference, Liverpool, UK, 5–8 June 2019; p. 267. [Google Scholar]
- Bonizzoni, G.D.M.; Bellocchia, M.; Bena, C.; Clivati, E.; Biglia, C.; Demichelis, S.; Bandelli, G.; Grande, A.; Traversa, F.; Albera, C.; et al. Real life experience of lumacaftor/ivacaftor in adult Phe508del homozygous cystic fibrosis patients. In Proceedings of the European Respiratory Society International Congress, Paris, France, 15–19 September 2018; p. 1322. [Google Scholar]
- Ong, T.; Cassidy, J.; McNamara, S.; Davis, C.; Gibson, R.; Lion, K. Standardized clinical assessment of CFTR modulator use and safety monitoring: A descriptive quality improvement project. In Proceedings of the 30th Annual North American Cystic Fibrosis Conference, Orlando, FL, USA, 27–29 October 2016; p. 554. [Google Scholar]
- Pan, A.; Autry, E.; McKinzie, C.J. Real world multicenter experience of pediatric cystic fibrosis patients started on tezacaftor/ivacaftor + ivacaftor. In Proceedings of the 33rd Annual North American Cystic Fibrosis Conference, Nashville, TN, USA, 31 October–2 November 2019; p. 477. [Google Scholar]
- Robinson, N.J.; McMullan, C.; Berwick, A.; Robertson, J.; McIntosh, L.; McCabe, D.; Hardisty, G.R.; Gray, R.D. CFTR modulators in the real world: An observational study of patient response to tezacaftor/ivacaftor therapy. In Proceedings of the 43rd European Cystic Fibrosis Conference, Lyon, France, 3–6 June 2020; p. 218. [Google Scholar]
- Perez, A.; Jue, I.; Dawson, D.; Kleinhenz, M. Neurocognitive side effects thwart transition to tezacaftor/ivacaftor CFTR modulator therapy in patients with F508del homozygous cystic fibrosis. In Proceedings of the American Thoracic Society International Conference, Dallas, TX, USA, 17–22 May 2019. [Google Scholar]
- Ridge, P.C.; Campbell, C.D.; O’Mahony, M. Real-world outcomes of adult patients transitioning from Orkambi to Symkevi. In Proceedings of the 43rd European Cystic Fibrosis Conference, Lyon, France, 3–6 June 2020; p. 217. [Google Scholar]
- Richards, C.J.; Sicilian, L.; Neuringer, I. Decline and recovery of lung function with the initiation and cessation of lumacaftor-ivacaftor. In Proceedings of the American Thoracic Society 2017 International Conference, Washington, DC, USA, 19–24 May 2017. [Google Scholar]
- Howsare, M.M.; El-Kersh, K. Isolated extreme elevation of alkaline phosphatase associated with lumacaftor-ivacaftor therapy: First report. In Proceedings of the American Thoracic Society 2017 International Conference, Washington, DC, USA, 19–24 May 2017. [Google Scholar]
- Joyau, C.; Veyrac, G.; Haloun, A.; Jolliet, P. Severe rhabdomyolysis in a patient treated with lumacaftor/ivacaftor association and aciclovir. In Proceedings of the Annual Meeting of French Society of Pharmacology and Therapeutics, and INSERM Clinical Research Centers, CIC Meeting, Rouen, France, 19–21 April 2017. [Google Scholar]
- SØrensen, H.T.; Lash, T.L.; Rothman, K.J. Beyond randomized controlled trials: A critical comparison of trials with nonrandomized studies. Hepatology 2006, 44, 1075–1082. [Google Scholar] [CrossRef]
- Shteinberg, M.; Taylor-Cousar, J.L. Impact of CFTR modulator use on outcomes in people with severe cystic fibrosis lung disease. Eur. Respir. Rev. 2020, 29, 190112:1–190112:8. [Google Scholar] [CrossRef]
- Gramegna, A.; Contarini, M.; Aliberti, S.; Casciaro, R.; Blasi, F.; Castellani, C. From ivacaftor to triple combination: A systematic review of efficacy and safety of CFTR modulators in people with cystic fibrosis. Int. J. Mol. Sci. 2020, 21, 5882. [Google Scholar] [CrossRef]
- Schwarz, C.; Sivagurunathan, S.; Epaud, R.; Klingsberg, R.C.; Fischer, R.; Rowe, S.M.; Audhya, P.K.; Ahluwalia, N.; You, X.; Ferro, T.J.; et al. Tezacaftor/ivacaftor in people with cystic fibrosis who stopped lumacaftor/ivacaftor due to respiratory adverse events. J. Cyst. Fibros. 2020. [Google Scholar] [CrossRef]
- Burgel, P.-R.; Durieu, I.; Chiron, R.; Mely, L.; Prevotat, A.; Murris-Espin, M.; Porzio, M.; Abely, M.; Reix, P.; Marguet, C.; et al. Clinical response to lumacaftor-ivacaftor in patients with cystic fibrosis according to baseline lung function. J. Cyst. Fibros. 2020. [Google Scholar] [CrossRef] [PubMed]
- Elborn, J.S.; Ramsey, B.W.; Boyle, M.P.; Konstan, M.W.; Huang, X.; Marigowda, G.; Waltz, D.; Wainwright, C.E. Efficacy and safety of lumacaftor/ivacaftor combination therapy in patients with cystic fibrosis homozygous for Phe508del CFTR by pulmonary function subgroup: A pooled analysis. Lancet Respir. Med. 2016, 4, 617–626. [Google Scholar] [CrossRef]
- Marigowda, G.; Liu, F.; Waltz, D. Effect of bronchodilators in healthy individuals receiving lumacaftor/ivacaftor combination therapy. J. Cyst. Fibros. 2017, 16, 246–249. [Google Scholar] [CrossRef] [PubMed]
- Taylor-Cousar, J.L.; Jain, M.; Barto, T.L.; Haddad, T.; Atkinson, J.; Tian, S.; Tang, R.; Marigowda, G.; Waltz, D.; Pilewski, J. Lumacaftor/ivacaftor in patients with cystic fibrosis and advanced lung disease homozygous for F508del-CFTR. J. Cyst. Fibros. 2018, 17, 228–235. [Google Scholar] [CrossRef] [PubMed]
- Granados, A.; Chan, C.L.; Ode, K.L.; Moheet, A.; Moran, A.; Holl, R. Cystic fibrosis related diabetes: Pathophysiology, screening and diagnosis. J. Cyst. Fibros. 2019, 18, S3–S9. [Google Scholar] [CrossRef] [PubMed]
- Culhane, S.; George, C.; Pearo, B.; Spoede, E. Malnutrition in cystic fibrosis: A review. Nutr. Clin. Pract. 2013, 28, 676–683. [Google Scholar] [CrossRef] [PubMed]
- Keating, D.; Marigowda, G.; Burr, L.; Daines, C.; Mall, M.M.; McKone, E.F.; Ramsey, B.W.; Rowe, S.M.; Sass, L.A.; Tullis, E.; et al. VX-445-tezacaftor-ivacaftor in patients with cystic fibrosis and one or two Phe508del alleles. N. Eng. J. Med. 2018, 379, 1612–1620. [Google Scholar] [CrossRef]
- Milla, C.E.; Ratjen, F.; Marigowda, G.; Liu, F.; Waltz, D.; Rosenfeld, M. Lumacaftor/ivacaftor in patients aged 6–11 years with cystic fibrosis and homozygous for F508del-CFTR. Am. J. Respir. Crit. Care Med. 2017, 195, 912–920. [Google Scholar] [CrossRef]
- Scherer, K.; Brockow, K.; Aberer, W.; Gooi, J.H.C.; Demoly, P.; Romano, A.; Schnyder, B.; Whitaker, P.; Cernadas, J.S.R.; Bircher, A.J. Desensitization in delayed drug hypersensitivity reactions—An EAACI position paper of the Drug Allergy Interest Group. Allergy 2013, 68, 844–852. [Google Scholar] [CrossRef]
- McKone, E.F.; Borowitz, D.; Drevinek, P.; Griese, M.; Konstan, M.W.; Wainwright, C.; Ratjen, F.; Sermet-Gaudelus, I.; Plant, B.; Munck, A.; et al. Long-term safety and efficacy of ivacaftor in patients with cystic fibrosis who have the Gly551Asp-CFTR mutation: A phase 3, open-label extension study (PERSIST). Lancet Respir. Med. 2014, 2, 902–910. [Google Scholar] [CrossRef]
- Fidika, A.; Herle, M.; Goldbeck, L. Symptoms of depression impact the course of lung function in adolescents and adults with cystic fibrosis. BMC Pulm. Med. 2014, 14, 205:1–205:6. [Google Scholar] [CrossRef] [PubMed]
- Schechter, M.S.; Ostrenga, J.S.; Fink, A.K.; Barker, D.H.; Sawicki, G.S.; Quittner, A.L. Decreased survival in cystic fibrosis patients with a positive screen for depression. J. Cyst. Fibros. 2020. [Google Scholar] [CrossRef] [PubMed]
- Jordan, C.L.; Noah, T.L.; Henry, M.M. Therapeutic challenges posed by critical drug-drug interactions in cystic fibrosis. Pediatr. Pulmonol. 2016, 51, S61–S70. [Google Scholar] [CrossRef] [PubMed]
- Assis, D.N.; Debray, D. Gallbladder and bile duct disease in cystic fibrosis. J. Cyst. Fibros. 2017, 16, S62–S69. [Google Scholar] [CrossRef] [PubMed]
- Davies, J.; Sheridan, H.; Bell, N.; Cunningham, S.; Davis, S.D.; Elborn, J.S.; Milla, C.E.; Starner, T.D.; Weiner, D.J.; Lee, P.-S.; et al. Assessment of clinical response to ivacaftor with lung clearance index in cystic fibrosis patients with a G551D-CFTR mutation and preserved spirometry: A randomized controlled trial. Lancet Respir. Med. 2013, 1, 630–638. [Google Scholar] [CrossRef]
- Griese, M.; Costa, S.; Linnemann, R.W.; Mall, M.A.; McKone, E.F.; Polineni, D.; Quon, B.S.; Ringshausen, F.C.; Taylor-Cousar, J.L.; Withers, N.J.; et al. Safety and efficacy of elexacaftor/tezacaftor/ivacaftor for ≥24 weeks in people with CF and ≥1 F508del allele: Interim results of an open-label phase three clinical trial. Am. J. Respir. Crit. Care Med. 2020. [Google Scholar] [CrossRef] [PubMed]
- Konstan, M.W.; McKone, E.F.; Moss, R.B.; Marigowda, G.; Tian, S.; Waltz, D.; Huang, X.; Lubarsky, B.; Rubin, J.; Millar, S.J.; et al. Assessment of safety and efficacy of long-term treatment with combination lumacaftor and ivacaftor therapy in patients with cystic fibrosis homozygous for the F508del-CFTR mutation (PROGRESS): A phase 3, extension study. Lancet Respir. Med. 2017, 5, 107–118. [Google Scholar] [CrossRef]
- U.S. National Library of Medicine. A study to Evaluate the Safety and Efficacy of Long Term Treatment with VX-661 in Combination with Ivacaftor in Subjects with Cystic Fibrosis Who Have an F508del-CFTR Mutation. Identifier NCT02565914. October 2015. Available online: https://clinicaltrials.gov/ct2/show/results/NCT02565914?view=results (accessed on 12 October 2020).
- Assis, D.N.; Freedman, S.D. Gastrointestinal disorders in cystic fibrosis. Clin. Chest Med. 2016, 37, 109–118. [Google Scholar] [CrossRef]
- Edenborough, F.P. Women with cystic fibrosis and their potential for reproduction. Thorax 2001, 56, 649–655. [Google Scholar] [CrossRef]
- Agarwal, N.; Apperley, L.; Taylor, N.F.; Taylor, D.R.; Ghataore, L.; Rumsby, E.; Treslove, C.; Holt, R.; Thursfield, R.; Senniappan, S. Posaconazole-induced hypertension masquerading as congenital adrenal hyperplasia in a child with cystic fibrosis. Case Rep. Med. 2020, 8153012:1–8153012:5. [Google Scholar] [CrossRef]
- Lieberman, J.; Rodbard, S. Low blood pressure in young adults with cystic fibrosis: An effect of chronic salt loss in sweat? Ann. Int. Med. 1975, 82, 806–808. [Google Scholar] [CrossRef] [PubMed]
- Bisch, A.L.; Wheatley, C.M.; Baker, S.E.; Peitzman, E.R.; Van Iterson, E.H.; Laguna, T.A.; Morgan, W.J.; Snyder, E.M. Cystic fibrosis transmembrane conductance regulator genotype, not circulating catecholamines, influences cardiovascular function in patients with cystic fibrosis. Clin. Med. Insights Circ. Respir. Pulm. Med. 2019, 13, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Lilleng, H.; Abeler, K.; Johnsen, S.H.; Stensland, E.; LØseth, S.; Jorde, R.; Figenschau, Y.; Lindal, S.; Wilsgaard, T.; Bekkelund, S.I. Variation of serum creatinine kinase (CK) levels and prevalence of persistent hyperCKemia in a Norwegian normal population. The TromsØ Study. Neuromuscul. Disord. 2011, 21, 494–500. [Google Scholar] [CrossRef]
- Gruet, M.; Troosters, T.; Verges, S. Peripheral muscle abnormalities in cystic fibrosis: Etiology, clinical implications and response to therapeutic interventions. J. Cyst. Fibros. 2017, 16, 538–552. [Google Scholar] [CrossRef] [PubMed]
- Le-Rademacher, J.; Hillman, S.L.; Meyers, J.; Loprinzi, C.L.; Limburg, P.J.; Mandrekar, S.J. Statistical controversies in clinical research: Value of adverse events relatedness to study treatment: Analyses of data from randomized double-blind placebo-controlled clinical trials. Ann. Oncol. 2017, 28, 1183–1190. [Google Scholar] [CrossRef] [PubMed]
- Bitonti, M.; Patel, P.; Dickinson, R.; Knapp, P.; Blalock, S.J. The effect of counseling on willingness to use a hypothetical medication and perceptions of medication safety. Res. Soc. Adm. Pharm. 2018, 14, 295–302. [Google Scholar] [CrossRef] [PubMed]
- Agyapong, V.I.O.; Nwankwo, V.; Bangaru, R.; Kirrane, R. Sources of patients’ knowledge of the adverse effects of psychotropic medication and the perceived influence of adverse effects on compliance among service users attending community mental health services. J. Clin. Psychopharmacol. 2009, 29, 565–570. [Google Scholar] [CrossRef]
- Keating, D.; Wilson, L.; Williams, E.; Kotsimbos, T.; Wilson, J. Ivacaftor withdrawal syndrome during a randomised placebo-controlled cross-over study. In Proceedings of the 42nd European Cystic Fibrosis Conference, Liverpool, UK, 5–8 June 2019; p. 259. [Google Scholar]
- McKone, E.; Connolly, A.; Gallagher, C. Impact of ivacaftor treatment disruption on clinical outcomes: A single centre study. In Proceedings of the 42nd European Cystic Fibrosis Conference, Liverpool, UK, 5–8 June 2019; p. 208. [Google Scholar]
- Trimble, A.T.; Donaldson, S.H. Ivacaftor withdrawal syndrome in cystic fibrosis patients with the G551D mutation. J. Cyst. Fibros. 2018, 17, e13–e16. [Google Scholar] [CrossRef]
- Vekaria, S.; Popowicz, N.; White, S.W.; Mulrennan, S. To be or not to be on CFTR modulators during pregnancy: Risks to be considered. J. Cyst. Fibros. 2020, 19, e7–e8. [Google Scholar] [CrossRef]
- Mainz, J.G.; Michl, R.K.; Beiersdorf, N.; Lorenz, M.; Schneider, U.; Groten, T.; Jaudszus, A. Successful pregnancy of a patient with cystic fibrosis genotype F508del/F508del and progressed pulmonary destruction on lumacaftor/ivacaftor. Klin. Padiatr. 2019, 231, 271–273. [Google Scholar] [CrossRef]
- Trimble, A.; McKinzie, C.; Terrell, M.; Stringer, E.; Esther, C.R. Measured fetal and neonatal exposure to lumacaftor and ivacaftor during pregnancy and while breastfeeding. J. Cyst. Fibros. 2018, 17, 779–782. [Google Scholar] [CrossRef] [PubMed]
- Carpino, E.A.; Fowler, R.E.; Uluer, A.Z.; Sawicki, G.S. Acute clinical outcomes following participation in short-term CFTR modulator trials in adults with cystic fibrosis: A retrospective chart review. In Proceedings of the 32nd North American Cystic Fibrosis Conference, Denver, CO, USA, 18–20 October 2018; p. 300. [Google Scholar]
- Wark, P.; McDonald, V.M. Nebulized hypertonic saline for cystic fibrosis. Cochrane Database Syst. Rev. 2018, 9, CD001506:1–CD001506:123. [Google Scholar] [CrossRef]
- Yang, C.; Montgomery, M. Dornase alfa for cystic fibrosis. Cochrane Database Syst. Rev. 2018, 9, CD001127:1–CD001127:115. [Google Scholar] [CrossRef] [PubMed]
- Mogayzel, P.J.; Naureckas, E.T.; Robinson, K.A.; Mueller, G.; Hadjiliadis, D.; Hoag, J.B.; Lubsch, L.; Hazle, L.; Sabadosa, K.; Marshall, B. Cystic fibrosis pulmonary guidelines: Chronic medications for maintenance of lung health. Am. J. Respir. Crit. Care Med. 2013, 187, 680–689. [Google Scholar] [CrossRef] [PubMed]
- Nichols, D.; Mayer-Hamblett, N.; Gifford, A. Impact of Discontinuing Chronic Therapies in People with Cystic Fibrosis on Highly Effective CFTR Modulator Therapy (SIMPLIFY). Identifier NCT04378153. 7 May 2020. Available online: https://clinicaltrials.gov/ct2/show/NCT04378153 (accessed on 12 October 2020).
| Ref | Study Design & Location | Population a | n | Recruitment Period & Follow-up Duration | Overall Adverse Events (AE) b,c | Dose Modification, Interruption, or Discontinuation Due to AE b,c | ||||
|---|---|---|---|---|---|---|---|---|---|---|
| Ivacaftor | ||||||||||
| [29] | Prospective Cohort d United States | Baseline Age Pediatric and Adult - Mean: 33 yr - Range: 10–61 yr CFTR Genotype ≥ 1 copy G551D Baseline ppFEV1 Mean: 30% | 44 | Recruitment Period Prior to commercial availability Follow-up Duration NS | AE in n = 38 (86%): | n | % | Discontinuation: | n | % |
| - Pulmonary exacerbation - Hemoptysis - Increased sputum - Increased cough - URTI - Dyspnea - Abnormal respiration - Respiratory tract congestion - Headache - Rash | 20 7 7 6 6 3 3 3 5 4 | 45 16 16 14 14 7 7 7 11 9 | - Severe abdominal pain - Dizziness/tinnitus | 1 1 | 2 2 | |||||
| SAE in n = 14 (32%): | ||||||||||
| - Pulmonary exacerbation - Hemoptysis - Pneumothorax - Acute respiratory failure - URTI - Abdominal pain - Gastroenteritis - Abnormal LFTs - Syncope - Secondary adrenocortical insufficiency | NS NS NS NS NS NS NS NS NS NS | - - - - - - - - - - | ||||||||
| [55] | Prospective Cohort United States Canada Italy | Baseline Age Pediatric and Adult - Mean: 17 yr - Range: 5–61 yr CFTR Genotype ≥1 gating mutation Baseline ppFEV1 Mean: 86% | 23 | Recruitment Period Mar 2014 to Aug 2015 Follow-up Duration 3 mo | 49 AE in n = 21 (91%): | n | % | None reported | ||
| - Respiratory, unspecified - Gastrointestinal, unspecified - Infection, unspecified - Headache - Weakness - Dizziness - Fatigue | NS NS NS NS NS NS NS | - - - - - - - | ||||||||
| 5 SAE in n = 3 (13%): | n | % | ||||||||
| - Respiratory infection - Acute changes in metabolic and liver status | 4 1 | 17 4 | ||||||||
| [56] | Retrospective Cohort United Kingdom (1 center) | Baseline Age Pediatric - Mean: 9 yr - Range: 6–14 yr CFTR Genotype 1 copy G551D Baseline ppFEV1 Mean: 68% e | 4 | Recruitment Period Jan 2013 to Jun 2015 Follow-up Duration Mean: 24 mo | - Transaminitis (<3 x ULN) | n 1 | % 25 | None reported | ||
| [57] | Retrospective Cohort Scotland (11 centers) | Baseline Age Pediatric - Median: 9 yr CFTR Genotype ≥1 copy G551D Baseline ppFEV1 Mean: 85% | 26 | Recruitment Period NS (Jan 2013 to Mar 2013 for 85%) Follow-up Duration Mean: 17 mo | - Headache - Swollen ear - Cataracts | n 1 1 2 | % 4 4 17 f | None reported | ||
| [58] | Retrospective Cohort France (25 centers) | Baseline Age Pediatric and Adult - Median: 18 yr - Range: 6–52 yr CFTR Genotype ≥1 copy G551D Baseline ppFEV1 Mean: 72% | 57 | Recruitment Period Pre-1 Jun 2013 up to 30 Sep 2014 Follow-up Duration Up to 2 yr | 34 AE in n = 21 (37%): | n | % | Interruption in n = 7 (12%): | n | % |
| - Transaminitis - Rhinopharyngitis - Asthma - Fever - Chest pain - Abdominal pain - Nausea or vomiting - Intestinal dysmotility - Headache - Fatigue - Rash or eczema - Depression - Myalgia - Arthritis - Breast hypertrophy - Orchitis - Atrial fibrillation | 3 NS NS NS NS NS NS NS NS NS NS NS NS NS NS NS NS | 5 - - - - - - - - - - - - - - - - | - Hepatitis - Rhinopharyngitis - Abdominal pain - Vomiting - Headache - Rash - Severe depression | NS NS NS NS NS NS NS | - - - - - - - | |||||
| Discontinuation: | ||||||||||
| - Transaminitis - Liver cirrhosis diagnosis | 1 1 | 2 2 | ||||||||
| [30] | Retrospective Cohort d Germany (multicenter) | Baseline Age Adult - Mean: 34 yr CFTR Genotype ≥1 copy G551D Baseline ppFEV1 Mean: 25% | 14 | Recruitment Period Sep 2012 to Apr 2013 Follow-up Duration Mean: 235 days | - Increased bronchial and nasal secretions - Headache - Worsening RLS - Abdominal pain - Hyperbilirubinemia (mild) - Transaminitis (<3 to 4x ULN) | n 3 1 1 1 1 1 | % 21 7 7 7 7 7 | Discontinuation: - Increased bronchial and nasal secretions * * Trial of reduced dose before discontinuation | n 1 | % 7 |
| Lumacaftor/Ivacaftor | ||||||||||
| [41] | Prospective Cohort France (1 center) | Baseline Age Pediatric - Mean: 16 yr CFTR Genotype ∆F508/∆F508 Baseline ppFEV1 Mean: 87% | 32 | Recruitment Period Mar 2016 to Dec 2016 Follow-up Duration 4 h post-first dose | - Acute drop in ppFEV1 - Wheeze | n 32 3 | % 100 9 | None reported | ||
| [42] | Prospective Cohort France (47 centers) | Baseline Age Pediatric and Adult - Mean: 22 yr CFTR Genotype ∆F508/∆F508 Baseline ppFEV1 Mean: 65% | 845 | Recruitment Period 1 Jan 2016 to 31 Dec 2016 Follow-up Duration 12 mo | AE in n = 494 (59%): | n | % | Interruption: | n | % |
| - Respiratory - Digestive - Menstrual abnormality - Fatigue - Headache - CK > 5xULN - Transaminitis (> 3xULN) | 316 181 53 37 19 20 5 | 37 21 6 4 2 2 0.6 | - Respiratory - ‘Non-respiratory’ Discontinuation: Respiratory - Chest tightness/dyspnea - Bronchospasm - Increased cough/sputum - Hemoptysis - Pneumothorax Non-respiratory - Diarrhea, abdominal pain - CK >10xULN + myalgia - Fatigue - Headache - Depression - Metrorrhagia - Transaminitis (>6xULN) - Cutaneous rash - Tachycardia | 16 8 n 38 24 9 2 1 18 5 5 4 4 3 2 1 1 | 2 1 % 5 3 1 0.2 0.1 2 0.6 0.6 0.5 0.5 0.4 0.2 0.1 0.1 | |||||
| [59] | Prospective Cohort United States (1 center) | Baseline Age Pediatric and Adult - Mean: 23 yr - Range: 12–48 yr CFTR Genotype ∆F508/∆F508 Baseline ppFEV1 Mean: 70% | 26 | Recruitment Period NS Follow-up Duration 6 mo | See Discontinuation | Discontinuation: - Transaminitis - Unspecified AE | n 1 4 | % 4 15 | ||
| [31] | Prospective Cohort d Switzerland (1 center) | Baseline Age Adult - Mean NR CFTR Genotype ∆F508/∆F508 Baseline ppFEV1 Median: 30% | 20 | Recruitment Period Jan 2016 to Jan 2017 Follow-up Duration 1 mo | n | % | Reduced dose: | n | % | |
| - Dyspnea - 3 h - 24 h - 1 mo - Chest tightness - 3 h - 24 h - 1 mo - Increased sputum - 3 h - 24 h - 1 mo - Pulmonary exacerbation - 1 mo | 0 1 1 1 10 1 1 8 3 2 | - 5 5 5 50 5 5 40 15 10 | - Respiratory intolerance Discontinuation: - Chest tightness (at 24 h) | 3 1 | 15 5 | |||||
| [32] | Prospective Cohort Australia (1 center) | Baseline Age Adult - Mean: 27 yr CFTR Genotype ∆F508/∆F508 Baseline ppFEV1 Median: 36% | 12 | Recruitment Period Jan 2016 to Oct 2016 Follow-up Duration 1 mo | n | % | Discontinuation: | n | % | |
| - Acute drop in ppFEV1 - Respiratory AE overall - 4 h - 24 h - 1 mo - Dyspnea - 4 h - 24 h - 1 mo - Chest tightness - 4 h - 24 h - 1 mo - Increased sputum - 4 h - 24 h - 1 mo - Pulmonary exacerbation | 12 5 10 8 2 6 7 4 8 5 0 2 1 6 | 100 42 83 67 17 50 58 33 67 42 - 17 8 50 | - Chest tightness/dyspnea * * n = 2 discontinued after 1mo follow-up (5 wk and 9 wk) | 3 | 25 | |||||
| [33] | Prospective Cohort France (11 centers) | Baseline Age Adult - Mean: 31 yr CFTR Genotype ∆F508/∆F508 Baseline ppFEV1 Mean: 32% | 53 | Recruitment Period Jan 2016 to Jun 2016 Follow-up Duration 3 mo | AE in n = 34 (63%): | n | % | Discontinuation: | n | % |
| - Abnormal respiration - Dyspnea - Increased cough - Abdominal pain, nausea, diarrhea, or vomiting - Fatigue - Rash - Pruritus - Breast tension | 13 11 3 9 2 1 1 1 | 25 21 6 17 4 2 2 2 | - Respiratory intolerance - Vomiting - Fatigue | 13 1 1 | 25 2 2 | |||||
| [25] | Prospective Cohort d,g Australia (1 center) | Baseline Age Adult - Mean: 27 yr CFTR Genotype ∆F508/∆F508 Baseline ppFEV1 Mean: 36% | 10 | Recruitment Period NS Follow-up Duration 52 wk | AE in n = 6 (60%): - Chest tightness/dyspnea - Headache | n 6 2 | % 60 20 | None reported | ||
| [43] | Retrospective Cohort Ireland (1 center) | Baseline Age Pediatric - Mean: 14 yr CFTR Genotype ∆F508/∆F508 Baseline ppFEV1 Mean: 77% | 15 | Recruitment Period Sep 2016 to Aug 2017 Follow-up Duration NS | - Acute drop in ppFEV1 - Chest tightness - Increased sputum | n 14 2 2 | % 93 13 13 | None reported | ||
| [44] | Retrospective Cohort United States (1 center) | Baseline Age Pediatric and Adult - Mean: 25 yr - Range: 12–59 yr CFTR Genotype ∆F508/∆F508 Baseline ppFEV1 Mean: 67% | 116 | Recruitment Period NS Follow-up Duration Up to 11 mo | AE in n = 46 (40%): | n | % | Reduced dose: | n | % |
| - Chest tightness - Dyspnea - Increased cough - Diarrhea - Nausea - Decreased appetite - Rash | 23 12 10 5 3 2 2 | 20 10 9 4 3 2 2 | - AE not specified Discontinuation: - Reasons not specified h | 10 20 | 9 17 | |||||
| [60] | Retrospective Cohort Greece (1 center) | Baseline Age Pediatric and Adult - Mean: 16 yr i - Range: 12–23 yr CFTR Genotype ∆F508/∆F508 Baseline ppFEV1 Mean 92% i | 62 | Recruitment Period Mar 2016 to Aug 2017 Follow-up Duration 12 mo | - Chest tightness | n 2 | % 3 | Discontinuation: - Transaminitis - Cataract | n 1 1 | % 2 2 |
| [34] | Retrospective Cohort d Spain (multicenter) | Baseline Age Pediatric and Adult - Mean: 27 yr - Range: 10–45 yr CFTR Genotype ∆F508/∆F508 Baseline ppFEV1 Mean: 32% | 20 | Recruitment Period 2016 Follow-up Duration 6 mo | AE in n = 15 (75%): | n | % | Discontinuation: | n | % |
| - Chest tightness - Dyspnea - Headache - Weight loss - ‘Sickness’ (not defined) - Asthenia - Abdominal pain - Transaminitis | 9 8 5 5 3 3 2 2 | 45 40 25 25 15 15 10 10 | - Decreased ppFEV1 - AE not specified | 1 6 | 5 30 | |||||
| [45] | Retrospective Cohort Canada (1 center) | Baseline Age Adult - Median: 32 yr CFTR Genotype ∆F508/∆F508 Baseline ppFEV1 Median: 40% | 22 | Recruitment Period Apr 2016 to Jun 2018 Follow-up Duration Median: 10 mo | AE in n = 19 (86%): | n | % | Discontinuation: | n | % |
| - Chest tightness - Wheeze - Dyspnea - Increased sputum - Increased cough - Flu-like symptoms - Elevated blood pressure - Headache - Nausea - Elevated AST - Anxiety - Bradycardia - Pleuritic chest pain | 14 4 3 3 2 1 5 4 2 1 1 1 1 | 64 18 14 14 9 5 23 18 9 5 5 5 5 | - Respiratory symptoms - Asymptomatic hypertension - Symptomatic hypertension - Headache - Hypertensive emergency - Anxiety | 3 2 1 1 1 | 14 9 5 5 5 | |||||
| [61] | Retrospective Cohort United States (1 center) | Baseline Age Adult - Mean NR CFTR Genotype ∆F508/∆F508 Baseline ppFEV1 Mean NR | 82 | Recruitment Period Jul 2015 to Jun 2016 Follow-up Duration 12 mo | See Discontinuation | Discontinuation: Total overall: - Chest tightness * - Diarrhea ** - Abdominal pain - Nausea ** - Dysphagia - Elevated LFTs - Pericarditis - Allergic reaction ** - Suspected Stevens–Johnson syndrome * n = 3 also had significant drop in ppFEV1 ** n = 1 also discontinued due to chest tightness | n 17 11 2 1 1 1 1 1 1 1 | % 21 13 2 1 1 1 1 1 1 1 | ||
| [26] | Retrospective Cohort d,g Australia (7 centers) | Baseline Age Adult - Mean: 31 yr CFTR Genotype ∆F508/∆F508 Baseline ppFEV1 Mean: 37% | 72 | Recruitment Period Nov 2015 to Mar 2017 Follow-up Duration 12 mo | n | % | Discontinuation: | n | % | |
| - Chest tightness/dyspnea - Increased sputum - Decrease in ppFEV1 - Headache - Fatigue - Nausea - Rash | 40 4 2 2 5 1 2 | 56 6 3 3 7 1 3 | - Chest tightness/dyspnea | 22 | 31 | |||||
| [27] | Case Series (Survey) j International (31 centers) | Baseline Age Adult - Mean: 30 yr CFTR Genotype ∆F508/∆F508 Baseline ppFEV1 Mean: 59% | 26 | Recruitment Period Questionnaire sent in 2018–2019 Follow-up Duration NS | - Pulmonary exacerbation - Post-partum acute myelocytic leukemia | n 1 1 | % 4 4 | Discontinuation: - Chest tightness | n 2 | % 8 |
| Ref | Study Design | Population | n | Overall Adverse Events (AE) a,b | Dose Modification, Interruption, or Discontinuation Due to AE a,b | ||||
|---|---|---|---|---|---|---|---|---|---|
| Ivacaftor | |||||||||
| [62] | Prospective Cohort | Baseline Age Pediatric - Mean: 5 yr CFTR Genotype ≥1 gating mutation Baseline ppFEV1 Mean NR | 4 | AE in n = 2 (50%): - URTI - Nasal congestion - Headache | n NS NS NS | % - - - | None reported | ||
| [63] | Retrospective Cohort | Baseline Age Pediatric - Mean: 6 yr CFTR Genotype ≥1 gating mutation Baseline ppFEV1 Median: 87% | 10 | - Transient rash - Increased obesity | n 2 1 | % 20 10 | None reported | ||
| [64] | Prospective Cohort | Baseline Age Pediatric and Adult - Mean NR CFTR Genotype ≥1 copy S549R Baseline ppFEV1 Mean: 54% | 15 | - Liver enzyme derangement | n 2 | % 13 | None reported | ||
| [65] | Cross-sectional Survey | Baseline Age Adult - Mean: 26 yr CFTR Genotype ≥1G551D Baseline ppFEV1 Mean: 62% | 11 d | AE in n = 8 (73%) d: - Transient rash - Dizziness - Unspecified AE | n NS NS NS | % - - - | None reported | ||
| Lumacaftor/Ivacaftor | |||||||||
| [66] | Prospective Cohort | Baseline Age Pediatric - Mean: 13 yr CFTR Genotype ∆F508/∆F508 Baseline ppFEV1 Mean: 91% | 14 | - Acute drop in ppFEV1 (asymptomatic) - Chest tightness, tachypnea (requiring oxygen) | n 1 1 | % 7 7 | Reduced dose *: - Chest tightness, tachypnea * Eventual titration to full dose | n 1 | % 7 |
| [67] | Prospective Cohort | Baseline Age Pediatric - Mean: 14 yr CFTR Genotype ∆F508/∆F508 Baseline ppFEV1 Mean: 87% | 13 | - Drop in ppFEV1 requiring salbutamol | n 7 | % 54 | None reported | ||
| [68] | Prospective Cohort | Baseline Age Pediatric and Adult - Mean: 23 yr e CFTR Genotype ∆F508/∆F508 Baseline ppFEV1 Mean 61% e | 369 | n | % | Discontinuation: | n | % | |
| - Bronchospasm - Dyspnea - Abnormal respiration - Unspecified respiratory AE - Unspecified AE | 15 12 7 4 120 | 4 3 2 1 33 | - Unspecified AE | 16 | 4 | ||||
| [69] | Prospective Cohort | Baseline Age Pediatric and Adult - Mean: 25 yr CFTR Genotype ∆F508/∆F508 Baseline ppFEV1 Mean NR | 311 | 379 AE in n = 213 (68%): - Dyspnea - Cough - GI discomfort (e.g., diarrhea, nausea, abdominal pain) - Headache - Fatigue - Unspecified | n f NS NS NS NS NS NS | % 31 6 31 6 5 NR | Interruption (stop/restart): - Unspecified AE and other reasons g Discontinuation: - Unspecified AE and other reasons g | n 12 42 | % 4 14 |
| [35] | Prospective Cohort c | Baseline Age Adult - Median: 31 yr CFTR Genotype ∆F508/∆F508 Baseline ppFEV1 Median: 28% | 14 | - Chest tightness, breathless - Rash | n 7 1 | % 50 7 | Discontinuation: - Respiratory AE and/or rash | n 4 | % 29 |
| [70] | Prospective Cohort | Baseline Age Adult - Mean NR CFTR Genotype ∆F508/∆F508 Baseline ppFEV1 Mean NR | 29 | - Chest tightness * * n = 4 cases severe, requiring hospitalization for IV steroids and antibiotics | n 13 | % 45 | Reduced dose: - Chest tightness Discontinuation: - Chest tightness | n 2 5 | % 7 17 |
| [36] | Prospective Cohort c | Baseline Age Mean NR h CFTR Genotype ∆F508/∆F508 Baseline ppFEV1 Mean NR | 32 | AE in 88%: | n f | % | Interruption (stop/restart): | n | % |
| - Respiratory AE - Drop in ppFEV1 | NS NS | 87 - | - Unspecified AEA Discontinuation: - Unspecified AE | 1 8 | 3 25 | ||||
| [71] | Retrospective Cohort | Baseline Age Pediatric and Adult - Mean NR CFTR Genotype ∆F508/∆F508 Baseline ppFEV1 Mean NR | 34 | AE in n = 29 (85%): - Pulmonary exacerbation - Chest tightness - Dyspnea - Diarrhea - Abdominal pain | n 16 9 3 3 3 | % 47 26 9 9 9 | Discontinuation: - Unspecified AE | n 10 | % 29 |
| Serious AE in n = 8 (24%): | |||||||||
| - Respiratory failure i - Unspecified AE | 1 7 | 3 21 | |||||||
| [72] | Retrospective Cohort | Baseline Age Pediatric and Adult - Mean: 26 yr CFTR Genotype ∆F508/∆F508 Baseline ppFEV1 Mean: 68% | 103 | See Discontinuation | Interruption/discontinuation j: - Chest tightness and/or pain - Elevated LFTs | n 17 NS | % 17 - | ||
| [73] | Retrospective Cohort | Baseline Age Adult - Mean: 31 yr CFTR Genotype ∆F508/∆F508 Baseline ppFEV1 Mean: 50% | 71 | AE in n = 41 (58%): | n | % | Discontinuation: | n | % |
| - Chest tightness - Dyspnea - Increased cough - GI (pain, constipation, or diarrhea) - Rash - Pruritus - Irregular menses or metrorrhagia - Breast tension - Headache - Myalgia | 22 8 4 6 4 1 3 2 1 1 | 31 11 6 9 6 1 4 3 1 1 | - Dyspnea - Chest tightness - Increased cough - Fatigue | 7 6 3 1 | 10 9 4 1 | ||||
| [74] | Retrospective Cohort | Baseline Age Mean NR h CFTR Genotype ∆F508/∆F508 Baseline ppFEV1 Mean NR | 54 | See Discontinuation | Discontinuation: - Chest tightness, dyspnea, and/or drop in ppFEV1 | n 8 | % 15 | ||
| [75] | Retrospective Cohort | Baseline Age Adult - Mean: 31 yr CFTR Genotype ∆F508/∆F508 Baseline ppFEV1 Mean NR | 28 | - Increased work of breathing or chest tightness - Drop in ppFEV1 | n 12 5 | % 43 18 | Discontinuation - Respiratory intolerance vs. pulmonary exacerbation - Persistent respiratory intolerance/chest tightness - Rash and swelling of face - Increased anxiety | n 1 3 1 1 | % 4 11 4 4 |
| [76] | Retrospective Cohort | Baseline Age Adult - Mean NR CFTR Genotype ∆F508/∆F508 Baseline ppFEV1 Mean NR | 46 | n | % | Discontinuation: | n | % | |
| - Drop in ppFEV1 - Transaminitis | 21 2 | 46 4 | - Dyspnea, cough, CFPEx, and/or chest tightness - Transaminitis - Headache - Muscle ache - Fatigue - Rash | 4 1 NS NS NS NS | 9 2 - - - - | ||||
| [77] | Retrospective Cohort | Baseline Age Adult - Mean NR CFTR Genotype ∆F508/∆F508 Baseline ppFEV1 Mean NR | 28 | See Discontinuation | Discontinuation: - SOB and/or drop in ppFEV1 | n 15 | % 54 | ||
| [78] | Retrospective Cohort | Baseline Age Mean: 32 yr h CFTR Genotype ∆F508/∆F508 Baseline ppFEV1 Mean: 62% | 20 | Overall AE: | n | % | Interruption (stop/restart): | n | % |
| - Chest tightness - Elevated LFTs - Upset stomach - Increased stool output - Rash - Elevated thyroid function test - RA exacerbation | NS NS NS NS NS NS NS | - - - - - - - | - Unspecified AE - full-dose restart - half-dose restart Discontinuation: - Unspecified AE | 2 4 2 | 10 20 10 | ||||
| [79] | Retrospective Cohort | Baseline Age Mean NR h CFTR Genotype ∆F508/∆F508 Baseline ppFEV1 Mean NR | 60 | - Heartburn/reflux - Abdominal pain - Loose/oily stools | n 20 19 17 | % 33 32 28 | None reported | ||
| [80] | Retrospective Cohort | Baseline Age Mean: 29 yr h,k CFTR Genotype ∆F508/∆F508 Baseline ppFEV1 Mean: 80% k | 34 | See Discontinuation | Discontinuation: Overall total: - Respiratory AE (70%) f - Unspecified reasons g | n 11 NS NS | % 32 - - | ||
| [81] | Cohort l | Baseline Age Pediatric - Mean NR CFTR Genotype ∆F508/∆F508 Baseline ppFEV1 Mean NR | 39 | - AST >3x ULN | n 2 | % 5 | None reported | ||
| [82] | Cohort l | Baseline Age Pediatric and Adult - Range: 13–48 yr CFTR Genotype ∆F508/∆F508 Baseline ppFEV1 Mean NR | 47 | See Discontinuation | Discontinuation: - Thoracic oppression and unspecified AE | n 4 | % 9 | ||
| [83] | Cohort l | Baseline Age Adult - Mean: 28 yr CFTR Genotype ∆F508/∆F508 Baseline ppFEV1 Mean: 61% | 46 | See Discontinuation | Discontinuation: - Dyspnea, increased sputum, and unspecified AE | n 6 | % 13 | ||
| [37] | Cohort c,l | Baseline Age Adult - Mean: 31 yr CFTR Genotype ∆F508/∆F508 Baseline ppFEV1 Mean: 28% | 30 | - Drop in ppFEV1 - Dyspnea, chest tightness, or chest pain - Increased sputum * Based on 31 trials of LUM/IVA in 30 patients | n 30 * 25 * NS | % 97 81 - | Discontinuation: - Respiratory AE, unspecified - Hypertension | n 3 1 | % 10 3 |
| [84] | Cohort l | Baseline Age Adult - Mean: 31yr CFTR Genotype ∆F508/∆F508 Baseline ppFEV1 Mean: 40% | 8 | See Interruption | Interruption in n = 1 (13%): - Drop in ppFEV1 - Eczema | n 1 1 | % 13 13 | ||
| [38] | Cohort c,l | Baseline Age Mean NR h CFTR Genotype ∆F508/∆F508 Baseline ppFEV1 Mean NR | 19 | See Discontinuation | Discontinuation: - Chest tightness and dyspnea | n 4 | % 21 | ||
| [85] | Cross-sectional questionnaire | Baseline Age Mean NR h CFTR Genotype ∆F508/∆F508 Baseline ppFEV1 Mean NR | 11 | AE in n = 5 (46%): - Increased cough - Chest pain - Trouble breathing - Chest tightness - Stomach pain | n | % | Discontinuation: | n | % |
| 4 2 2 1 1 | 36 18 18 9 9 | - Increased cough | 1 | 9 | |||||
| Tezacaftor/Ivacaftor | |||||||||
| [86] | Prospective Cohort | Baseline Age Pediatric - Mean: 16 yr CFTR Genotype ∆F508 homozygous or heterozygous Baseline ppFEV1 Mean: 82% | 72 | See Discontinuation | Discontinuation: Overall total: - New-onset hemoptysis - Persistent nausea/vomiting - Elevated LFTs - Mental health changes - Alterations in blood glucose - Acholic stools | n 8 NS NS NS NS NS NS | % 11 - - - - - - | ||
| [87] | Prospective Cohort | Baseline Age Mean NR h CFTR Genotype NR Baseline ppFEV1 Mean NR | 50 | - AE not specified | n 5 | % 10 | Discontinuation: - Liver function abnormalities | n 1 | % 2 |
| [88] | Prospective Cohort | Baseline Age Adult - Mean: 34 yr CFTR Genotype ∆F508/∆F508 Baseline ppFEV1 Mean: 51% | 5 m | AE in n = 5 (11%) m: | n | % m | Discontinuation: | n | % m |
| - Sleep pattern disturbance - Out of body experience - Visual hallucination - Depersonalization - “Brain fog” - Severe migraine | 2 1 1 1 1 1 | 5 2 2 2 2 2 | - Out of body experience, visual hallucination - Depersonalization, “brain fog” - Severe migraine | 1 1 1 | 2 2 2 | ||||
| [89] | Retrospective Cohort | Baseline Age Adult - Mean NR CFTR Genotype ∆F508/∆F508 Baseline ppFEV1 Mean NR | 18 | See Discontinuation | Discontinuation: - Hair loss and fatigue | n 1 | % 6 | ||
| [39] | Cohort c,l | Baseline Age Adult - Mean NR CFTR Genotype NR Baseline ppFEV1 Mean: 34% | 22 | AE in n = 3 (14%): - Rash - Blurred vision - Viral symptoms | n 2 1 1 | % 9 5 5 | Discontinued: - Blurred vision | n 1 | % 5 |
| Elexacaftor/Tezacaftor/Ivacaftor | |||||||||
| [40] | Retrospective Cohort | Baseline Age Adult - Mean: 36 yr CFTR Genotype ≥1 copy ∆F508 Baseline ppFEV1 Mean: 31% | 11 | - Transaminitis | n 4 | % 36 | None reported | ||
| Ref | Location | n | Patient Information | Description of Adverse Events (AE) a | Dose Modification, Interruption or Discontinuation of Therapy, or Other Intervention(s) Due to AE |
|---|---|---|---|---|---|
| Ivacaftor | |||||
| [46] | Germany | 2 | Case 1: 55F Case 2: 55F | – Bronchial tightness – AST and ALT 2-3x ULN | – No interventions reported – resolved by approximately 14 days – IVA decreased to half-dose; transaminase subsequently normalized |
| Lumacaftor/Ivacaftor | |||||
| [47] | United States | 5 | Case 1: 15F | – 2 wk: new depression and SI – Within 7 mo: increased anxiety attack | – Initiation of CBT and sertraline – LUM/IVA discontinued at 11 mo (anxiety, menstrual irregularity); mood improved within 10 days of discontinuation |
| Case 2: 17F | – Within 2 mo: new worsening baseline depression, new SI | – Initiation of CBT, increased fluoxetine dose – LUM/IVA discontinued at 9 mo (GI intolerance); mood improved following discontinuation | |||
| Case 3: 14F | – Within 9 mo: new depression and SI – Suicide attempt (ibuprofen overdose) | – Initiation of CBT and fluoxetine; psychiatric hospitalization – LUM/IVA discontinued at 12 mo (depression, SI); mood improved following discontinuation | |||
| Case 4: 12F | – 2 mo: new depression and SI – Suicide attempt (cutting) | – Initiation of CBT; psychiatric hospitalization; After LUM/IVA discontinuation: initiation of sertraline – LUM/IVA discontinued at 3 mo (depression, SI); mood improved within 3 wk of discontinuation | |||
| Case 5: 17F | – 7 mo: worsening baseline depression – After LUM/IVA discontinuation: suicide attempt (escitalopram overdose) | – Initiation of escitalopram; After LUM/IVA discontinuation: trials of mirtazapine and fluoxetine; psychiatric hospitalization – LUM/IVA discontinued at 7 mo (depression) – Continued worsening depression; LUM/IVA restarted months later | |||
| [48] | United States | 4 | Case 1: 44F | – Within 1 mo: worsening baseline depression and anxiety, limiting adherence to CF therapies – Increased cough and sputum, ppFEV1 decline from 74% to 49% | – Increased citalopram dose; citalopram replaced by alternative psychotropic medications; hospitalizations for CFPEx management – LUM/IVA discontinued at 4 mo (depression, anxiety); mood and anxiety improved, but not to baseline until prior citalopram resumed |
| Case 2: 26M | – Within 3 mo: worsened depression (baseline bipolar disorder and recent bereavement) – ppFEV1 decline from 69% to 44% | – Change in psychotropic medications; CFPEx management – LUM/IVA continued unchanged – Mood and ppFEV1 back to baseline several months after interventions | |||
| Case 3: 36M | – Worsening baseline anxiety and opioid use disorder; subsequent worsening baseline depression, limiting medication adherence – ppFEV1 decline from 38% to 28% | – Change in psychotropic medications; hospitalization for psychiatric evaluation and CFPEx management; intensive outpatient mental health program – LUM/IVA discontinued (non-adherence) | |||
| Case 4: 13M | – 9 mo: worsening baseline anxiety (supported by increased GAD-7 score) with clinical impairment | – Re-initiation of CBT; increased citalopram dose – LUM/IVA continued unchanged; ongoing residual anxiety | |||
| [49] | United States | 1 | Case: 31F | – Within 6 wk: increased fatigue, “more winded”, dyspnea, “drowning” in sputum and sinus secretions – ppFEV1 decline from 58% to 34% | – Hospitalization for CFPEx management and aggressive pulmonary rehab – LUM/IVA continued unchanged; respiratory symptoms ‘near’ baseline and ppFEV1 improved to 42% (below baseline) after 2-wk hospitalization |
| [90] | United States | 1 | Case: 36M | – Immediate severe dyspnea, chest tightness – ppFEV1 drop from 31% to 23%; SpO2 drop from 95% at rest to 87% on exertion | – LUM/IVA decreased to half-dose (respiratory AE) – LUM/IVA discontinued at 6 mo (ongoing respiratory AE despite dose reduction); immediate improvement in respiratory AE, resolution of hypoxia on exertion, 1 mo ppFEV1 returned to baseline |
| [50] | Germany | 1 | Case: 20F | – 2 wk: malaise, severe progressive rash, pruritus, and facial swelling – Skin prick test negative for acute or delayed reaction to IVA, LUM/IVA, and TEZ/IVA – In vitro T-cell-mediated reaction to LUM | – Hospitalization for high-dose corticosteroids – LUM/IVA discontinued; rapid resolution of symptoms following discontinuation |
| [28] | United States | 1 | Case: 18F | – 7 days: non-confluent, red, bumpy, pruritic rash on shins and forearms; persisted 1 mo – Symptom recurrence upon rechallenging (within 3 and 7 days of first and second rechallenge, respectively) | – LUM/IVA discontinued; resolution of rash – Retrialed at full- and half-dose (retrials both 1 wk after discontinuation) – LUM/IVA discontinued indefinitely (recurrent rash); resolution of rash following discontinuation |
| [91] | United States | 1 | Case: 27M | – 4 wk: abdominal pain, diarrhea, N/V – AlkP 8477 U/L (other LFTs WNL) | – Hospitalization for supportive care, rule-out other cause – LUM/IVA discontinued; AlkP declined to WNL by 6 mo later |
| [92] | France | 1 | Case: 26M | – New myalgia following acyclovir initiation – Weeks later: rhabdomyolysis with worsening myalgia, muscle edema, dark urine, CK 17,582 U/L, AST 10x ULN, and ALT 4x ULN | – Hospitalization for supportive care – LUM/IVA discontinued (along with acyclovir); CK returned to normal, but residual lower limb myalgia |
| Tezacaftor/Ivacaftor | |||||
| [28] | United States | 1 | Case: 18F | – 3 days: non-confluent, red, bumpy, pruritic rash on shins and forearms – Symptom recurrence within 7 days upon rechallenging | – TEZ/IVA discontinued; resolution of rash – Retrialed 2 mo later, then discontinued (recurrent rash) – IVA desensitization protocol, titrating to full-dose IVA over 10 days; TEZ/IVA restarted successfully thereafter |
| [51] | Australia | 1 | Case: 21F | – Long-term azithromycin therapy and no past cardiac arrhythmia before initiation – 36 wk: PR interval of 334 ms (WNL at baseline then 4, 12, and 24 wk). QT and QTc normal – After TEZ/IVA held, PR interval WNL at 4 wk – After TEZ/IVA retrialed: 39 wk, PR interval 25–30 ms longer than at 4, 8, 12, and 24 wk | – Rule-out other causes (e.g., echocardiogram, blood work, viral testing) – TEZ/IVA held (PR prolonged on repeat ECG at 37 and 38 wk) – TEZ/IVA retrialed ~8 mo later – Azithromycin discontinued at 39 wk (PR interval lengthening on ECG); PR interval back to baseline 4 wk later and remained normal thereafter – TEZ/IVA continued |
| Elexacaftor/Tezacaftor/Ivacaftor | |||||
| [52] | United States | 7 | Case 1: 23M | – 12 days: new right testicular discomfort, intermittent sharp pain, sensitivity to touch and pressure | – OTC analgesics – ELX/TEZ/IVA unchanged; symptoms resolved in 12 days |
| Case 2: 23M | – 3 days: new right testicular soreness | – OTC analgesics – ELX/TEZ/IVA unchanged; symptoms resolved in 7 days | |||
| Case 3: 17M | – 2 days: new left testicular pain, difficulty urinating, lower abdominal pain | – No interventions reported – ELX/TEZ/IVA unchanged; symptoms resoled in 1 day | |||
| Case 4: 22M | – 7 days: new sharp, twisting, cramping testicular pain | – ELX/TEZ/IVA interrupted for 2 days, restarted at full dose; symptoms resolved in 7 days, no recurrence reported | |||
| Case 5: 30M | – 3–4 days: new right testicular pain and discomfort, increased ejaculate volume – Constipation identified on abdominal CT | – No interventions reported – ELX/TEZ/IVA unchanged; symptoms resolved in 21 days | |||
| Case 6: 37M | – 2–3 days: new testicular pain and discomfort when driving “bumpy roads” | – No interventions reported – ELX/TEZ/IVA unchanged; symptoms resolved in 2–3 days | |||
| Case 7: 39M | – 3 days: new left testicular pain – PE and ultrasound findings of bilateral epididymoorchitis and scrotal wall cellulitis | – OTC analgesics; antibiotic for epididymoorchitis – ELX/TEZ/IVA dose reduced to 1 tab daily, titrated to full dose; symptoms resolved in 1–2 days, no recurrence reported | |||
| [53] | United States | 7 | Case 1: 38F | – Within 1 mo: RUQ pain – Peak LFTs: AST 79 U/L, ALT 59 U/L, AlkP 103 U/L, total bili 12 umol/L | – Laparoscopic cholecystectomy (chronic cholecystitis with cholelithiasis; serosal fibrous adhesions) b – ELX/TEZ/IVA unchanged (except morning dose held pre-op) |
| Case 2: 33F | – Day 1: RUQ pain, nausea – Peak LFTs: AST 69 U/L, ALT 106 U/L, AlkP 138 U/L, total bili 55 umol/L | – Laparoscopic cholecystectomy (acute cholecystitis with cholelithiasis, mucosal necrosis) b; common biliary duct stent for persistent bile leak – ELX/TEZ/IVA held Day 3–7 (biliary colic), restarted post-op; held 1.5 days peri-op for stent placement (post-op complications) c – Resumed full dose post-stent placement; symptoms resolved | |||
| Case 3: 28M | – Day 3: epigastric radiating to RUQ; N/V – Peak LFTs: AST 34 U/L, ALT 37 U/L, AlkP 259 U/L, total bili 9 umol/L | – Laparoscopic cholecystectomy (chronic cholecystitis with cholelithiasis, extensive mucosal erosion, serositis, wall fibrosis) b – ELX/TEZ/IVA unchanged (except morning dose held pre-op) | |||
| Case 4: 28M | – Within 1 day: RUQ pain, nausea c – Peak LFTs: AST 42 U/L, ALT 65 U/L, AlkP 121 U/L, total bili 22 umol/L | – Laparoscopic cholecystectomy (chronic cholecystitis with cholelithiasis) b – ELX/TEZ/IVA held 2 wk pre-op, resumed 4 wk post-op, symptoms resolved – ELX/TEZ/IVA subsequently held (transaminitis), plans to retrial reduced dose upon transaminase normalization c | |||
| Case 5: 40F | – Day 1: lower abdominal and epigastric pain – Day 7: severe RUQ pain – Peak LFTs: AST 49 U/L, ALT 56 U/L, AlkP 335 U/L, total bili 10 umol/L | – No surgical intervention – ELX/TEZ/IVA unchanged | |||
| Case 6: 26F | – 2 wk: progressive abdominal pain; N/V – Peak LFTs: AST 516 U/L, ALT 283 U/L, AlkP 134 U/L, total bili 26 umol/L | – Laparoscopic cholecystectomy (chronic cholecystitis with cholelithiasis) b – ELX/TEZ/IVA unchanged (except morning dose held pre-op) | |||
| Case 7: 27F | – Day 1: central abdominal pain, anorexia, N/V – Peak LFTs of AST 103 U/L, ALT 36 U/L, AlkP 253 U/L, total bili 10 umol/L | – Hospitalization for symptom management; elective laparoscopic cholecystectomy at later date (chronic cholecystitis with cholelithiasis) b – ELX/TEZ/IVA held Day 5 (biliary colic), restarted Day 6 c – ELX/TEZ/IVA held Day 7 (symptom recurrence), restarted Day 8 c – Symptoms resolved (only held morning dose pre-op thereafter) c | |||
| [54] | United States | 1 | Case: 19F | – Within 2 wk: worsening baseline depression with new SI. At 3 wk, sleep paralysis with vivid hypnopompic hallucinations on 3 consecutive nights (history of 3 episodes of sleep paralysis previously) – After ELX/TEZ/IVA restarted: worsening depression and anxiety; subsequent improved depression and anxiety, resolved SI and sleep paralysis/hallucinations – 1–2 mo after full-dose ELX/TEZ/IVA: oscillating anxiety and depression, intermittent SI and sleep paralysis – After ELX/TEZ/IVA and IVA dose times switched: worsening depression and anxiety – After decrease to ELX/TEZ/IVA 2 tabs in AM: some improvement in depression; recurrence of sleep paralysis with hypnopompic hallucination | – ELX/TEZA/IVA discontinued at ~3 wk (worsening mood, sleep paralysis); resolution of sleep paralysis and hypnopompic hallucinations within 2 days – Sertraline initiated for ongoing depression. Subsequently, sertraline and baseline quetiapine doses increased, clonazepam initiated – ELX/TEZA/IVA restarted ~1 mo after discontinuation (respiratory status worsened); titrated to 1.5 ELX/TEZA/IVA tabs + 1.5 IVA tabs in AM – Sertraline and clonazepam doses increased, quetiapine dose decreased, bupropion initiated adjunctively (bupropion later stopped due to intolerance) – ELX/TEZA/IVA titrated to full-dose over ~1 mo; ELX/TEZ/IVA tabs moved to PM, IVA tab to AM 1–2 mo later (worsening psychiatric symptoms) – ELX/TEZA/IVA dose decreased to 2 ELX/TEZ/IVA tabs in AM ~2 mo later (worsening depression and anxiety); improvement in depressive symptoms and cognition, sertraline, quetiapine, and clonazepam doses increased – Continued 2 ELX/TEZ/IVA tabs in AM; symptoms of depression, anxiety, and sleep paralysis remained above baseline |
| CFTR Modulator | Ref | Adverse Event (AE) | Study Protocol/Strategy or Suggested Management/Monitoring for AE |
|---|---|---|---|
| Protocols/Strategies utilized in study | |||
| LUM/IVA | [43] | Respiratory AE and acute drop in ppFEV1 | Initiation protocol:
|
| LUM/IVA | [31] | Respiratory AE resulting in discontinuation | Initiation protocol:
|
| LUM/IVA | [44] | Respiratory AE resulting in discontinuation | Initiation strategy: For patients with ppFEV1 ≤ 40%, make effort to ensure use of combination inhaled beta2-agonist and corticosteroid, and lower initial dose for ≥7 days before increasing to recommended full dose. |
| TEZ/IVA and IVA a | [28] | Morbilliform drug eruption (Type IV drug allergy), recurrent upon rechallenge | Desensitization protocol:
|
| Suggested Strategy, Management, and/or Monitoring as a Result of Observations | |||
| IVA | [30] | Difficulty clearing liquefied airway secretions | For patients with severe lung disease, consider hospitalization for IV antibiotics and intense physiotherapy prior to initiation and for the first week of therapy. |
| IVA b | [57] | Cataract formation | In pediatric patients, baseline and follow-up ophthalmological examinations should be performed to assess for cataract formation. |
| LUM/IVA | [41] | Respiratory AE and acute drop in ppFEV1 | Closely monitor patients in clinic in the hours following the first dose, especially if low baseline ppFEV1 and/or known reversible airway obstruction ≥ 12%. |
| LUM/IVA | [32] | Respiratory AE and acute drop in ppFEV1 | For patients with ppFEV1 < 40%, consider lower initiation dose and monitor closely following initiation for lung function decline and respiratory AE. |
| LUM/IVA | [25] | Respiratory AE resulting in discontinuation | In patients with ppFEV1 < 40%, use caution with administration in case of poor tolerance. |
| LUM/IVA | [33] | Respiratory AE resulting in discontinuation | Pre-treat with long-acting bronchodilator therapy before initiation, as a strategy to mitigate respiratory AEs and potential subsequent discontinuation. |
| LUM/IVA | [61] | Respiratory AE resulting in discontinuation | Consider slow titration of dose during treatment initiation to mitigate AE. |
| LUM/IVA | [75] | Respiratory AE resulting in discontinuation | Consider slower titration of dose or increased use of bronchodilators to mitigate AE on initiation, especially in patients with ppFEV1 ≤ 40%. |
| LUM/IVA | [26] | Respiratory AE resulting in discontinuation | Suggested consideration for patients homozygous for F508del mutation: Preference for TEZ/IVA over LUM/IVA may be considered in patients with ppFEV1 < 40%, as TEZ/IVA does not share the same respiratory AE profile of chest tightness and dyspnea causing high rates of discontinuation. |
| LUM/IVA | [49] | Respiratory AE upon initiation, presenting as pulmonary exacerbation |
|
| LUM/IVA | [45] | Hypertension | Monitor blood pressure routinely both short- and long-term, and manage accordingly. |
| LUM/IVA | [47] | New or worsening depression and anxiety |
|
| LUM/IVA | [48] | Worsening depression and anxiety | Suggested initiation monitoring and management in patients already on citalopram, escitalopram, or sertraline:
|
| LUM/IVA | [79] | Breakthrough heartburn or GERD due to increased PPI metabolism | Suggested initiation monitoring and management for patients already on a PPI: Maintain patients on the lowest effective dose, increasing PPI dose only if patients present with breakthrough symptoms of heartburn/GERD. |
| TEZ/IVA | [88] | Neurocognitive side effects | Counsel patients regarding the possibility of neurocognitive AE (e.g., sleep pattern disturbances, visual hallucinations, out-of-body experiences) when initiating. |
| TEZ/IVA and ELX/TEZ/IVA c | [51] | First-degree heart block in combination with azithromycin | Suggested monitoring for patients already on azithromycin (or initiating azithromycin in patients already on TEZ/IVA or ELX/TEZ/IVA b): Obtain ECG at baseline and 3, 6, 9, and 12 months after combination to monitor for PR interval prolongation (onset may be delayed >8 months). |
| ELX/TEZ/IVA | [52] | Testicular pain following initiation | Until more is known about its impact on the reproductive system and fertility, may consider advising male patients to use contraception if having intercourse with females of child-bearing age, or refer to appropriate family planning resources if interested in pursuing pregnancy. |
| ELX/TEZ/IVA | [53] | Biliary colic following initiation | Prior to initiation, assess for history of postprandial RUQ pain and/or biliary colic. Following initiation, closely monitor LFTs and symptoms of biliary colic. |
| ELX/TEZ/IVA | [54] | New or worsening depression, SI, anxiety, and/or sleep paralysis with hypnopompic hallucinations | Suggested initiation monitoring: Following initiation, screen and closely monitor for new and/or worsening symptoms of depression, SI, and anxiety hallucinations. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dagenais, R.V.E.; Su, V.C.; Quon, B.S. Real-World Safety of CFTR Modulators in the Treatment of Cystic Fibrosis: A Systematic Review. J. Clin. Med. 2021, 10, 23. https://doi.org/10.3390/jcm10010023
Dagenais RVE, Su VC, Quon BS. Real-World Safety of CFTR Modulators in the Treatment of Cystic Fibrosis: A Systematic Review. Journal of Clinical Medicine. 2021; 10(1):23. https://doi.org/10.3390/jcm10010023
Chicago/Turabian StyleDagenais, Renée V. E., Victoria C. Su, and Bradley S. Quon. 2021. "Real-World Safety of CFTR Modulators in the Treatment of Cystic Fibrosis: A Systematic Review" Journal of Clinical Medicine 10, no. 1: 23. https://doi.org/10.3390/jcm10010023
APA StyleDagenais, R. V. E., Su, V. C., & Quon, B. S. (2021). Real-World Safety of CFTR Modulators in the Treatment of Cystic Fibrosis: A Systematic Review. Journal of Clinical Medicine, 10(1), 23. https://doi.org/10.3390/jcm10010023





