Addition of Lymphatic Stimulating Self-Care Practices Reduces Acute Attacks among People Affected by Moderate and Severe Lower-Limb Lymphedema in Ethiopia, a Cluster Randomized Controlled Trial
Abstract
1. Introduction
2. Materials and Methods
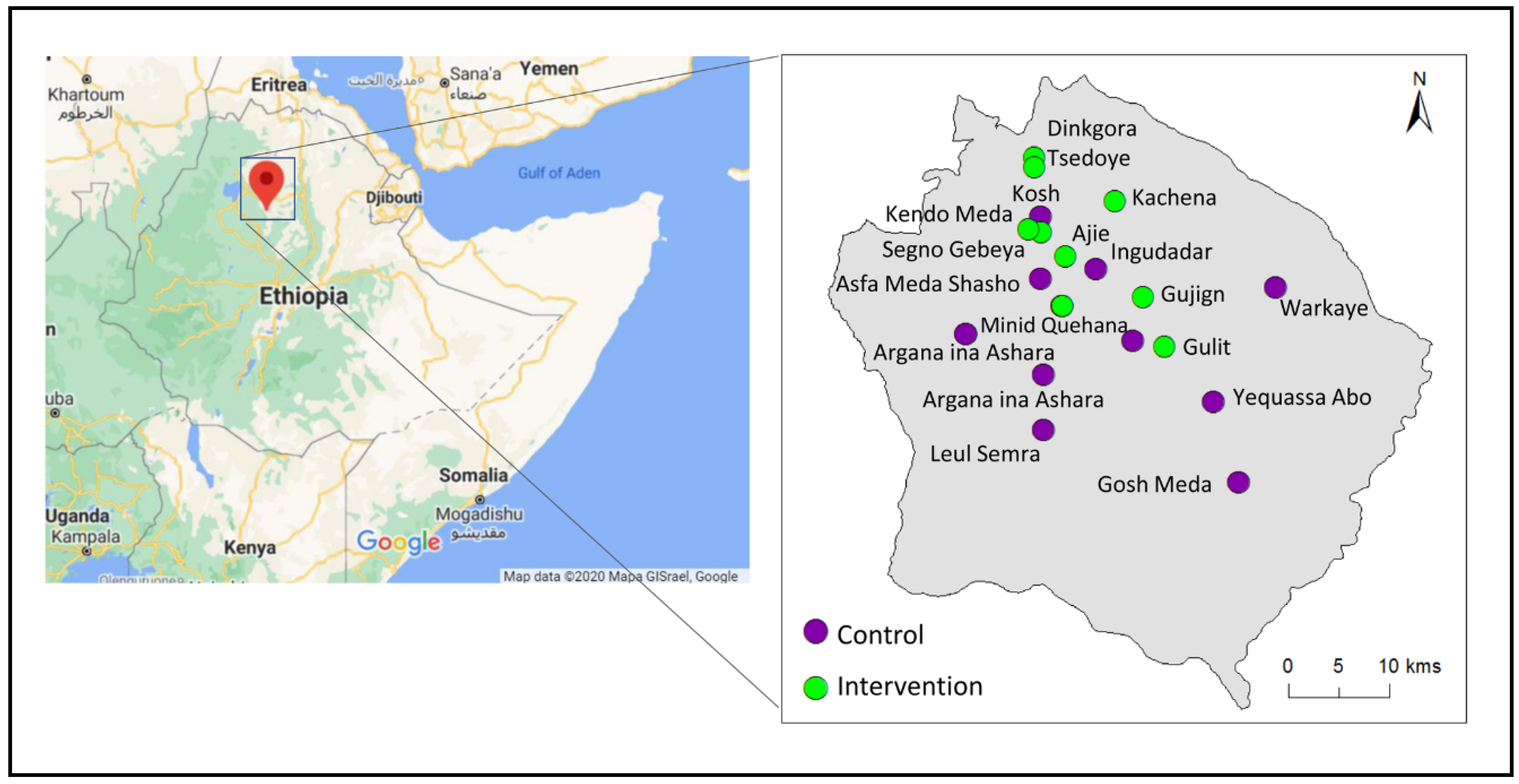
2.1. Study Design, Research Personnel and Data Collection
2.2. Outcome Measures and Data Collection
3. Intervention
4. Analysis
5. Results
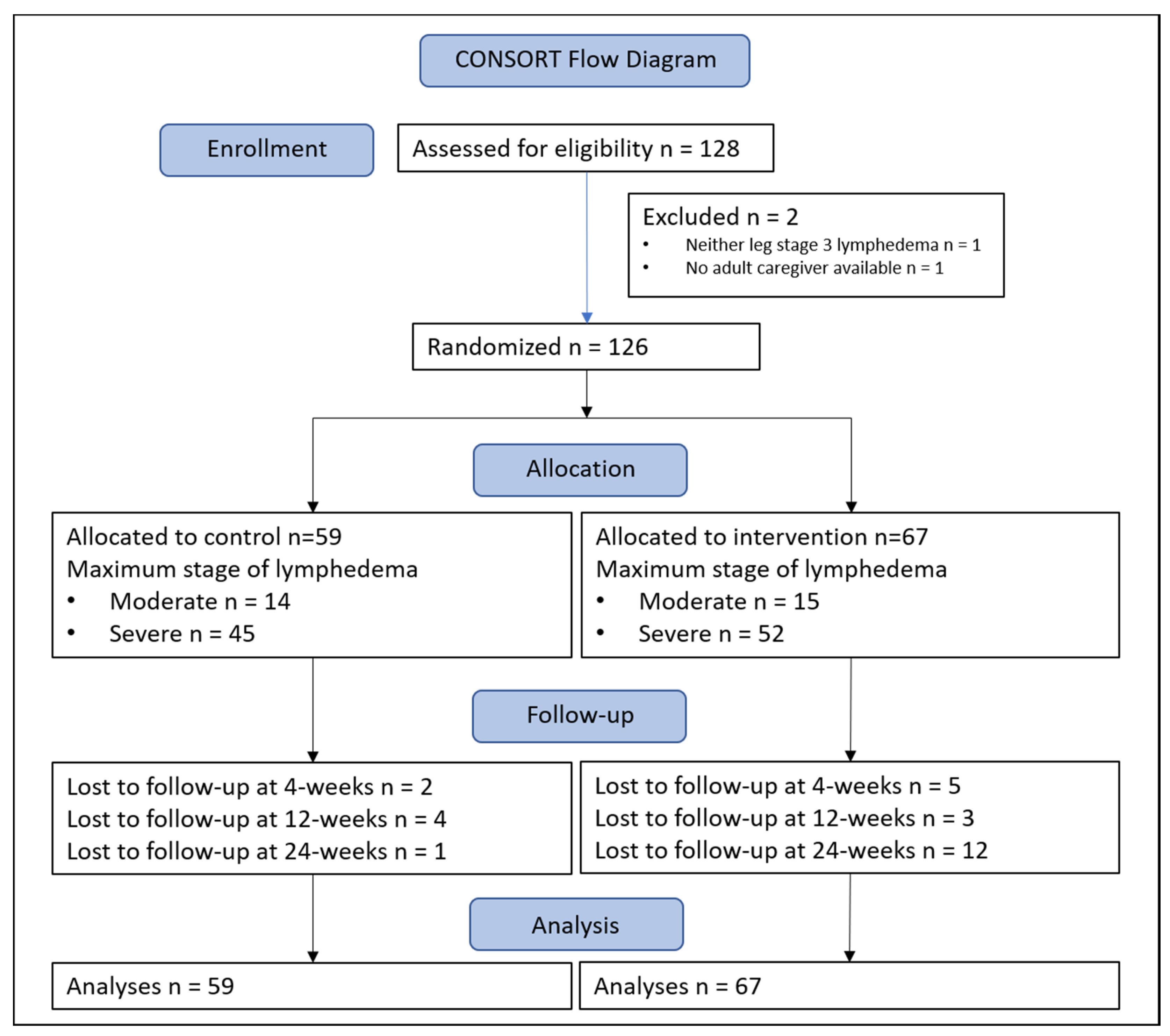
5.1. Participants
5.2. Primary Outcomes
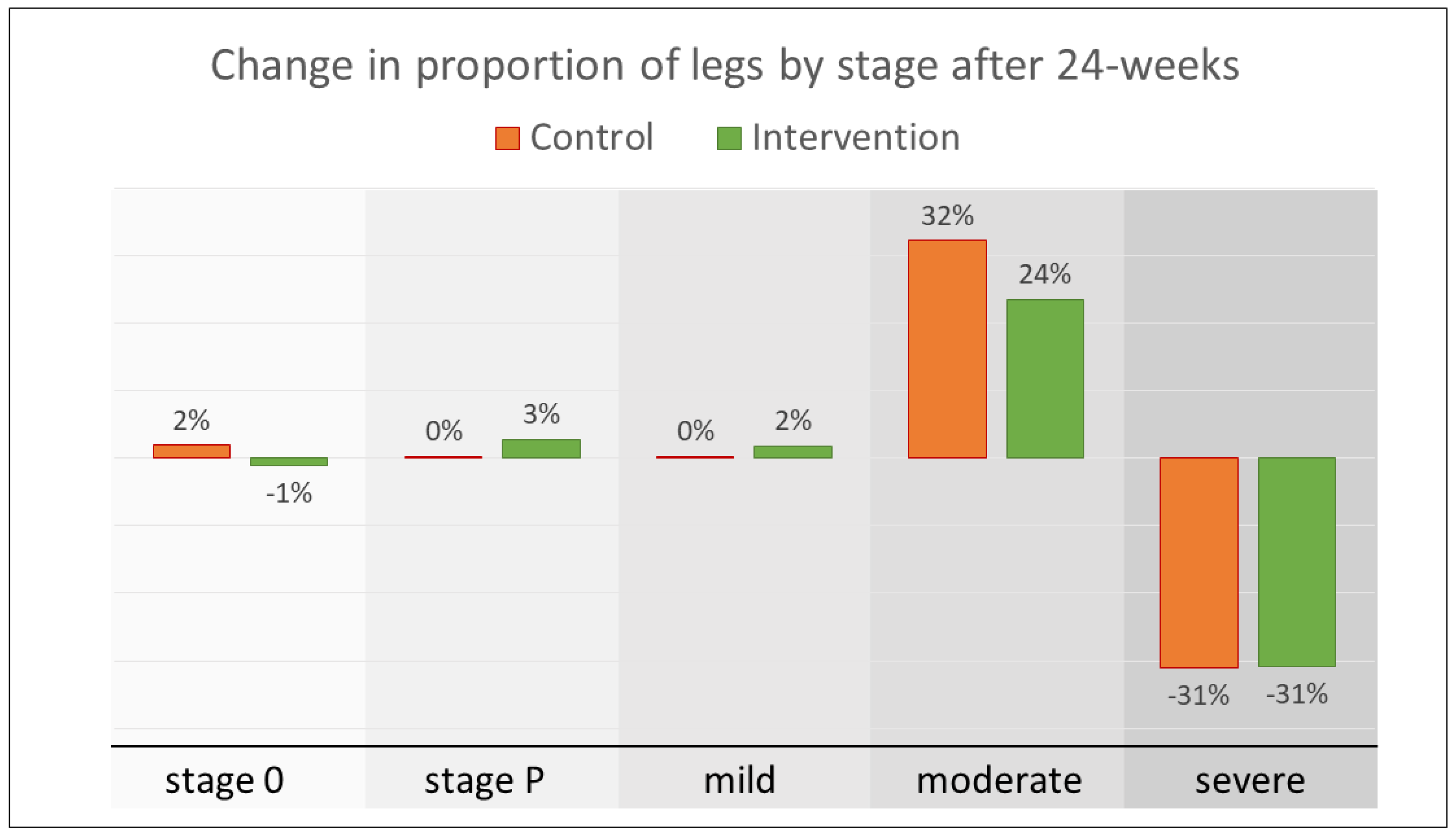
5.2.1. Change in Lymphedema Stage
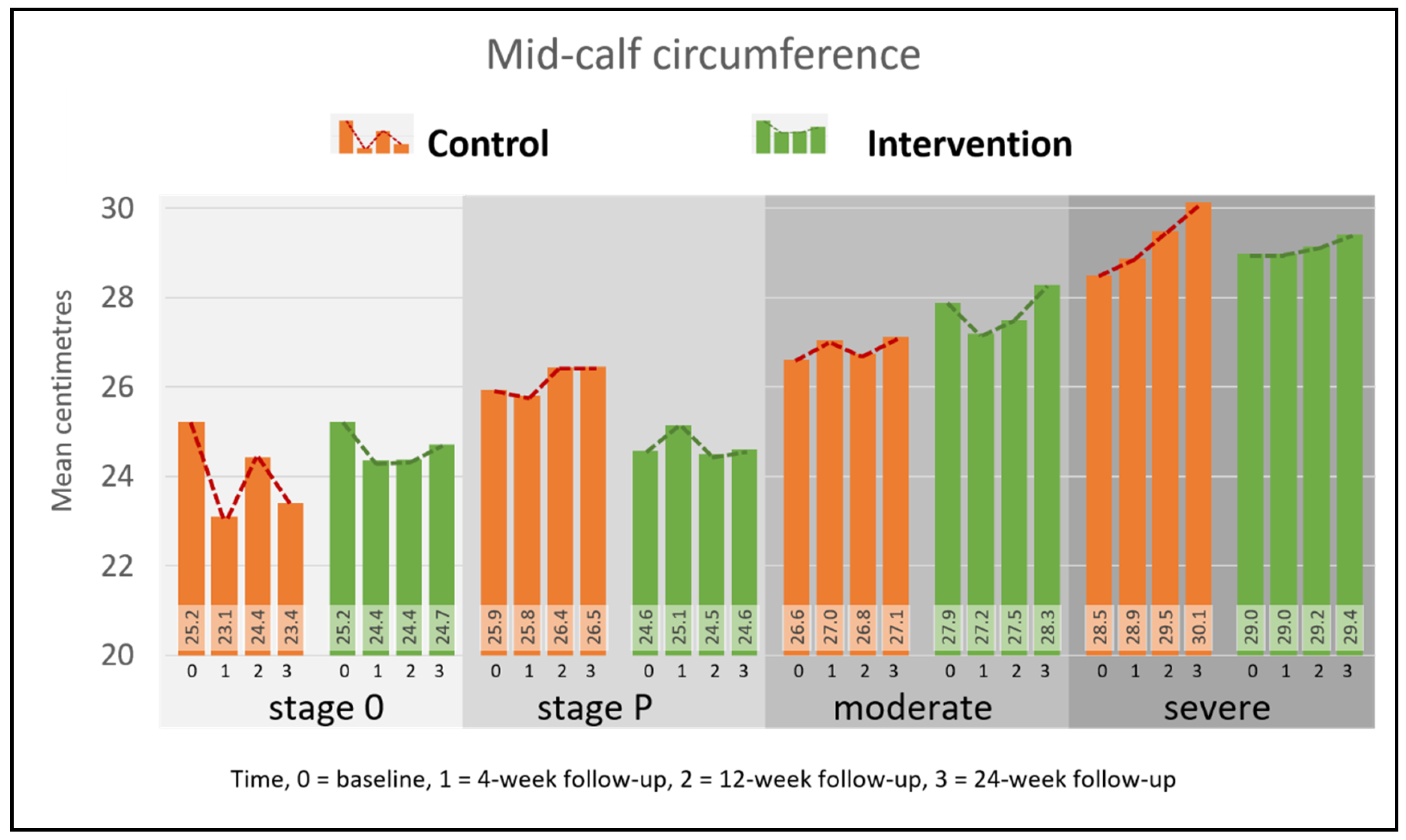
5.2.2. Mid-Calf Circumference
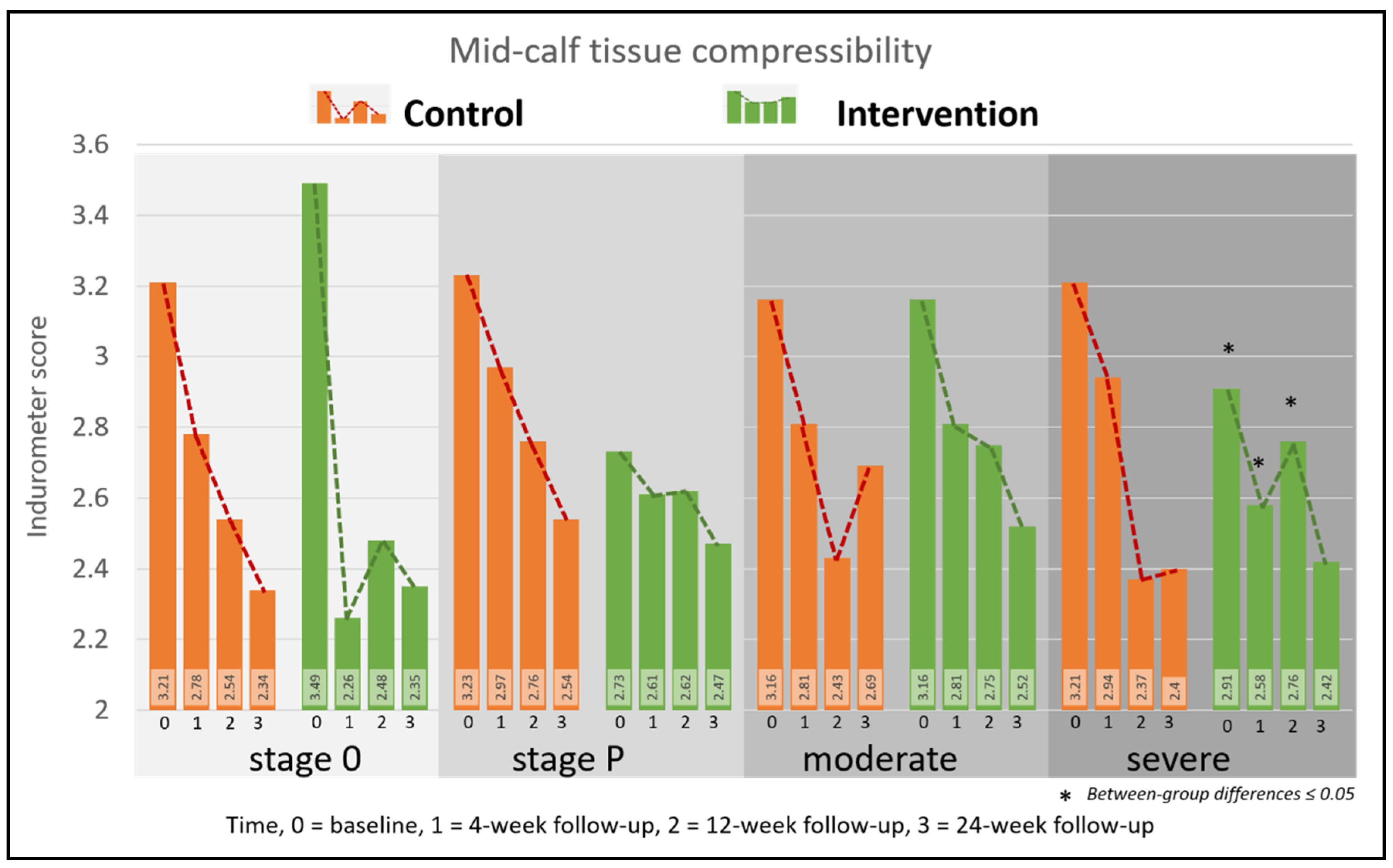
5.2.3. Midcalf Tissue Compressibility
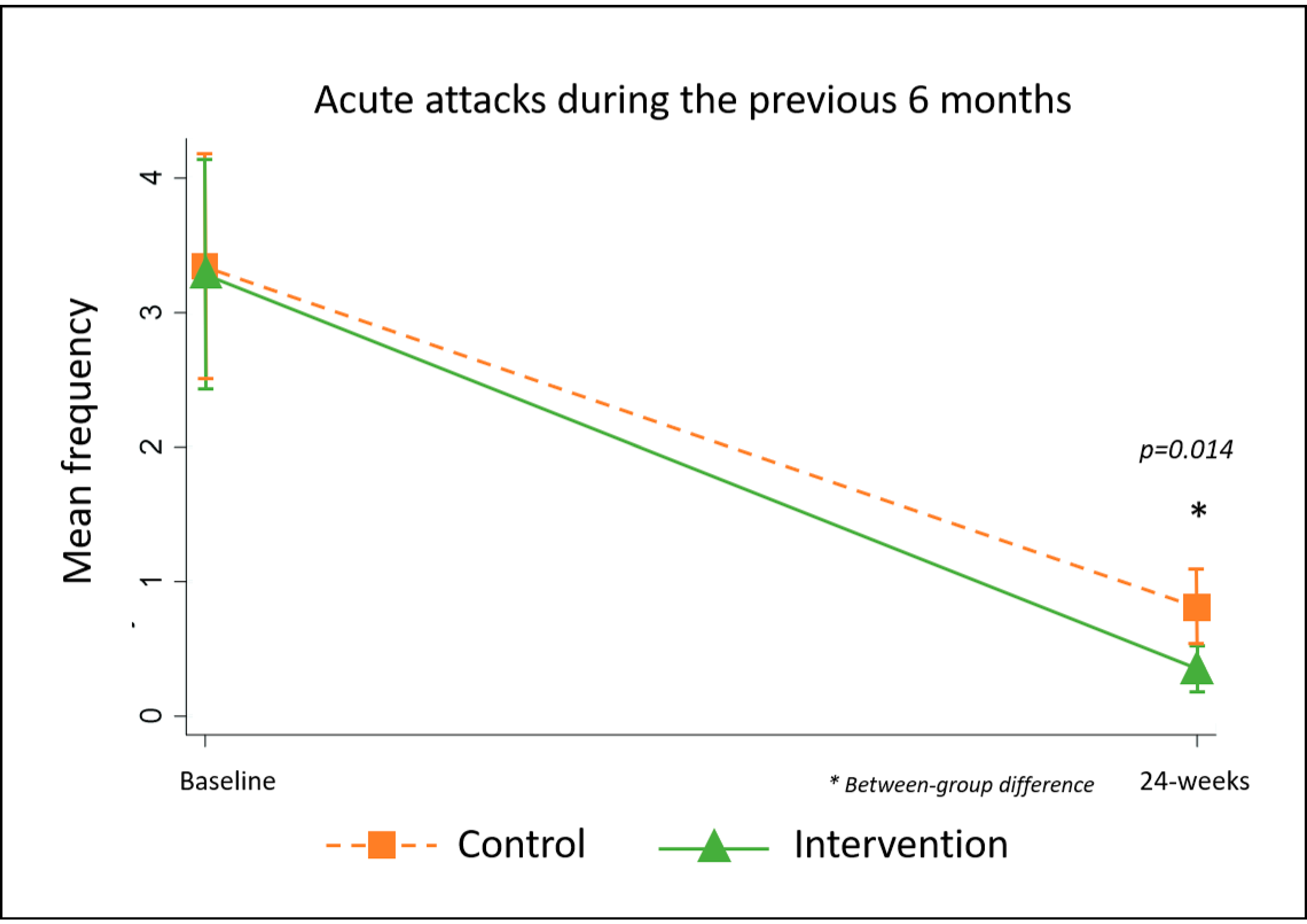
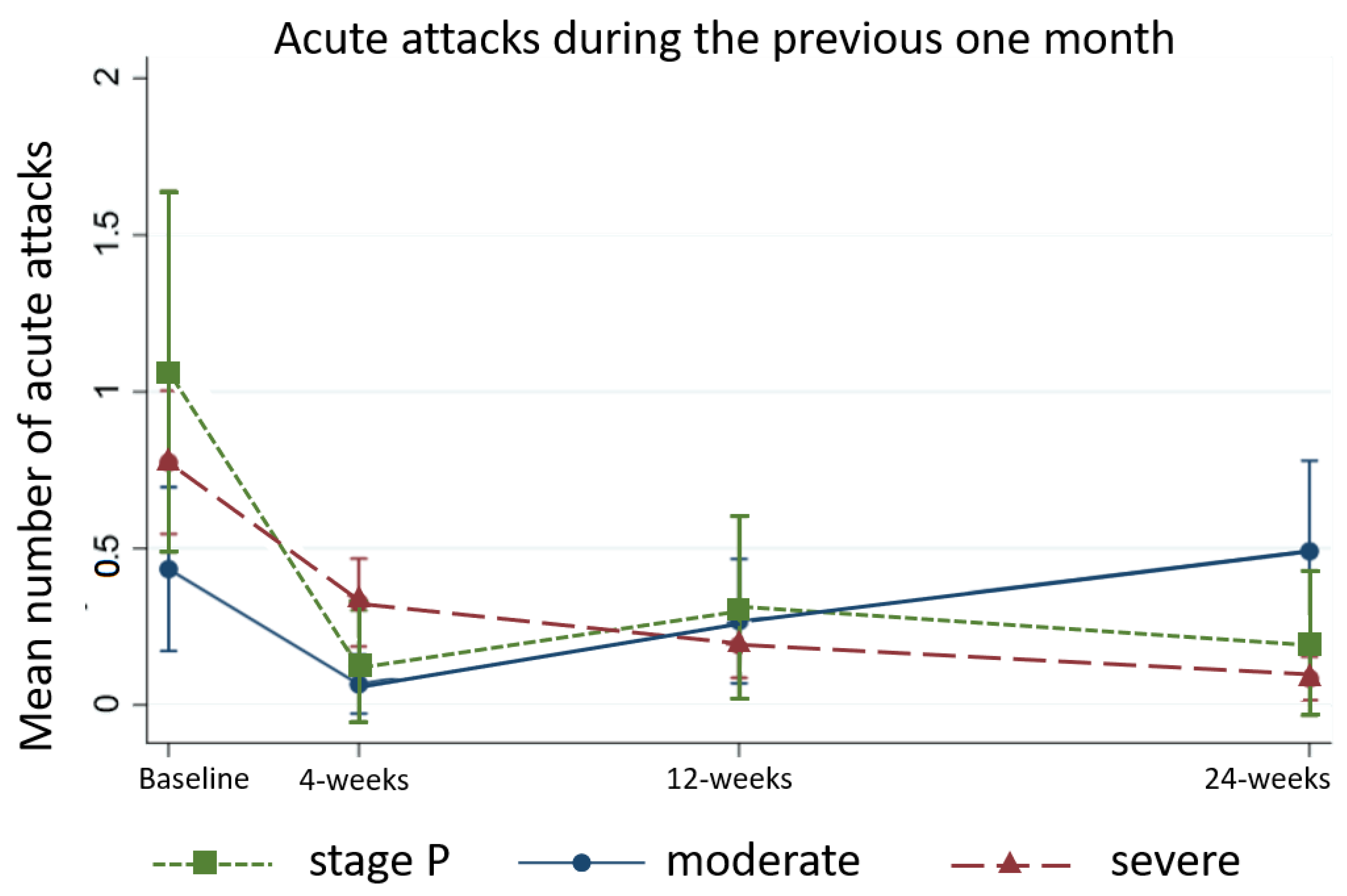
5.2.4. Frequency of Acute Attacks Previous One- and Six-Months
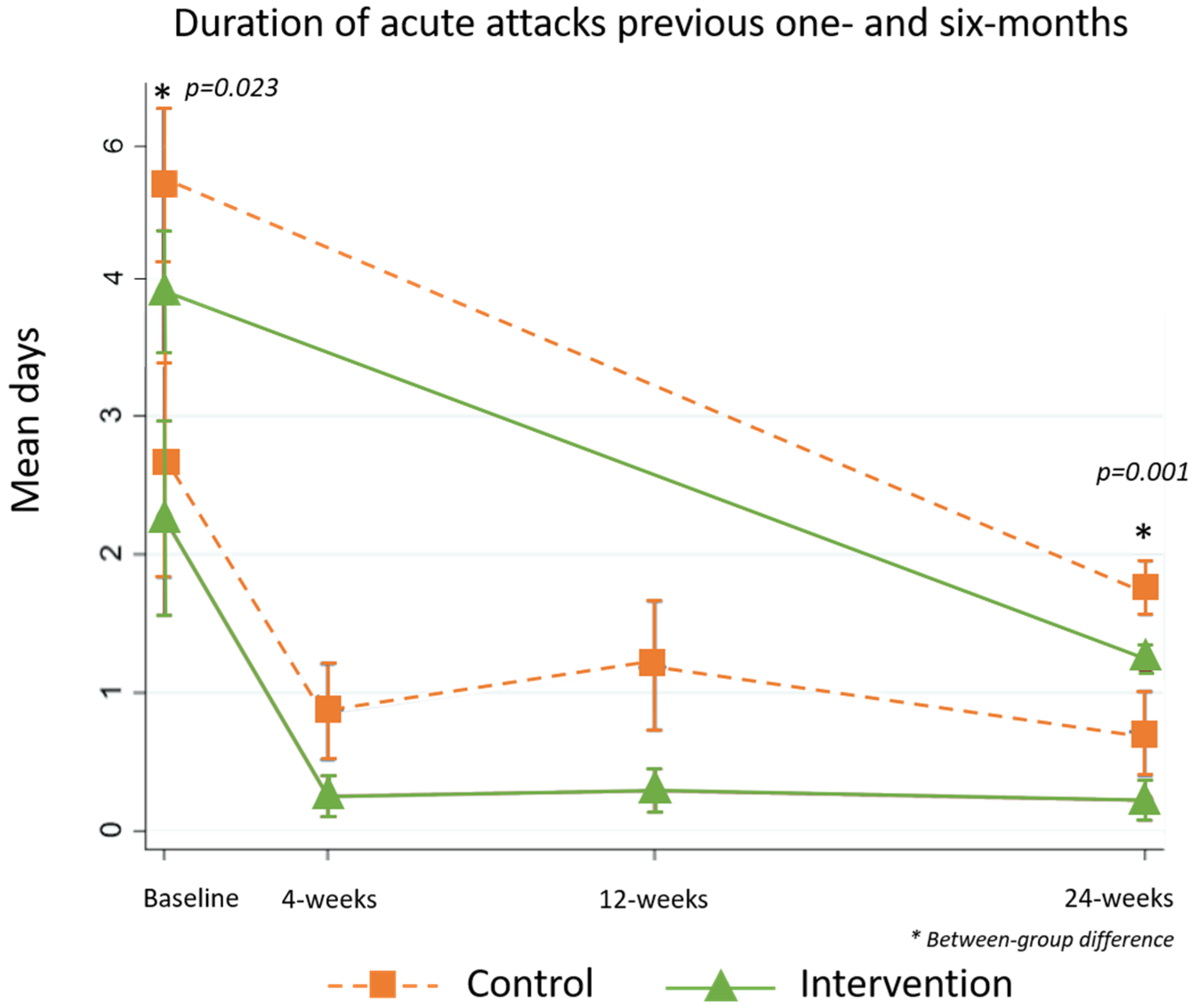
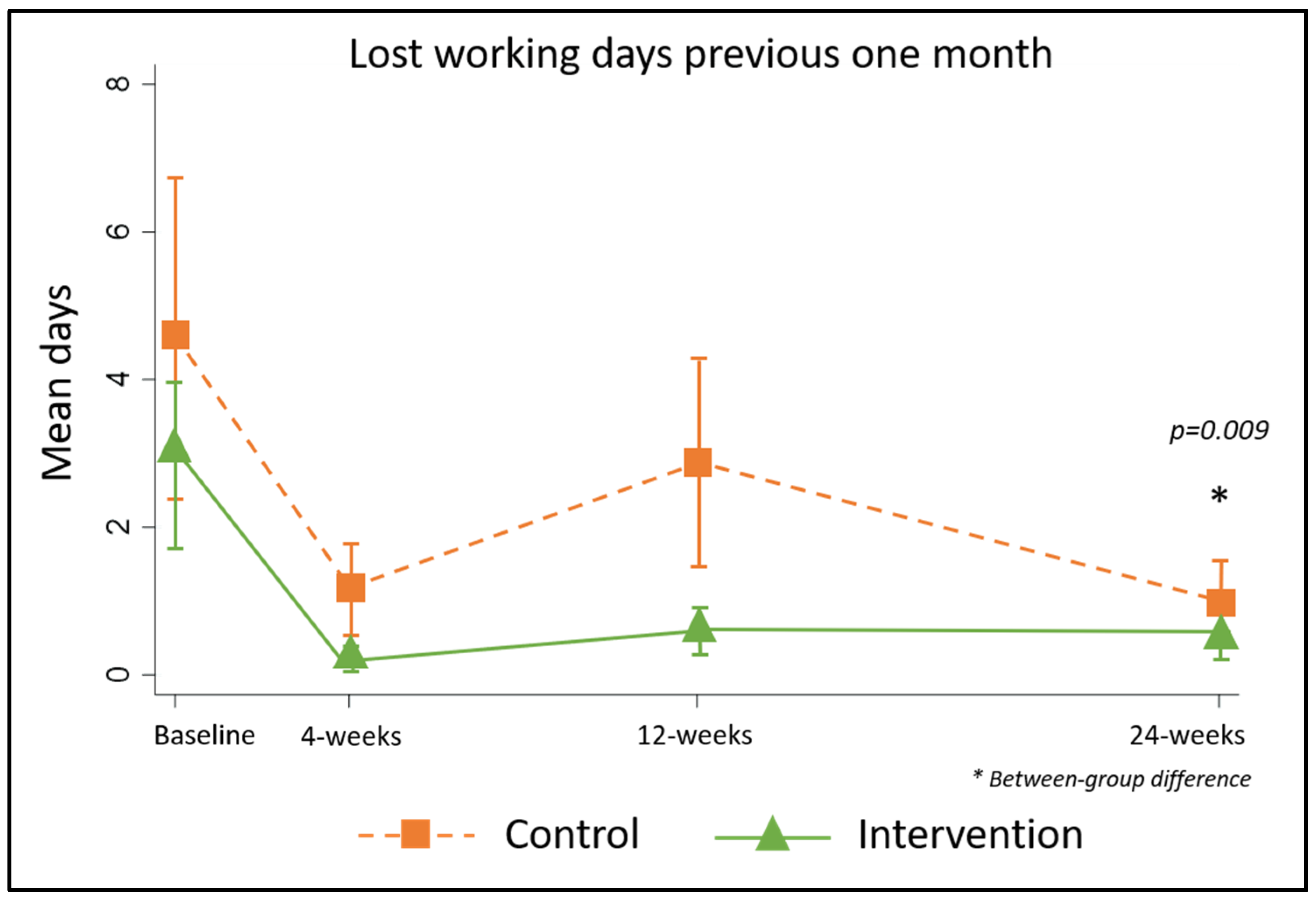
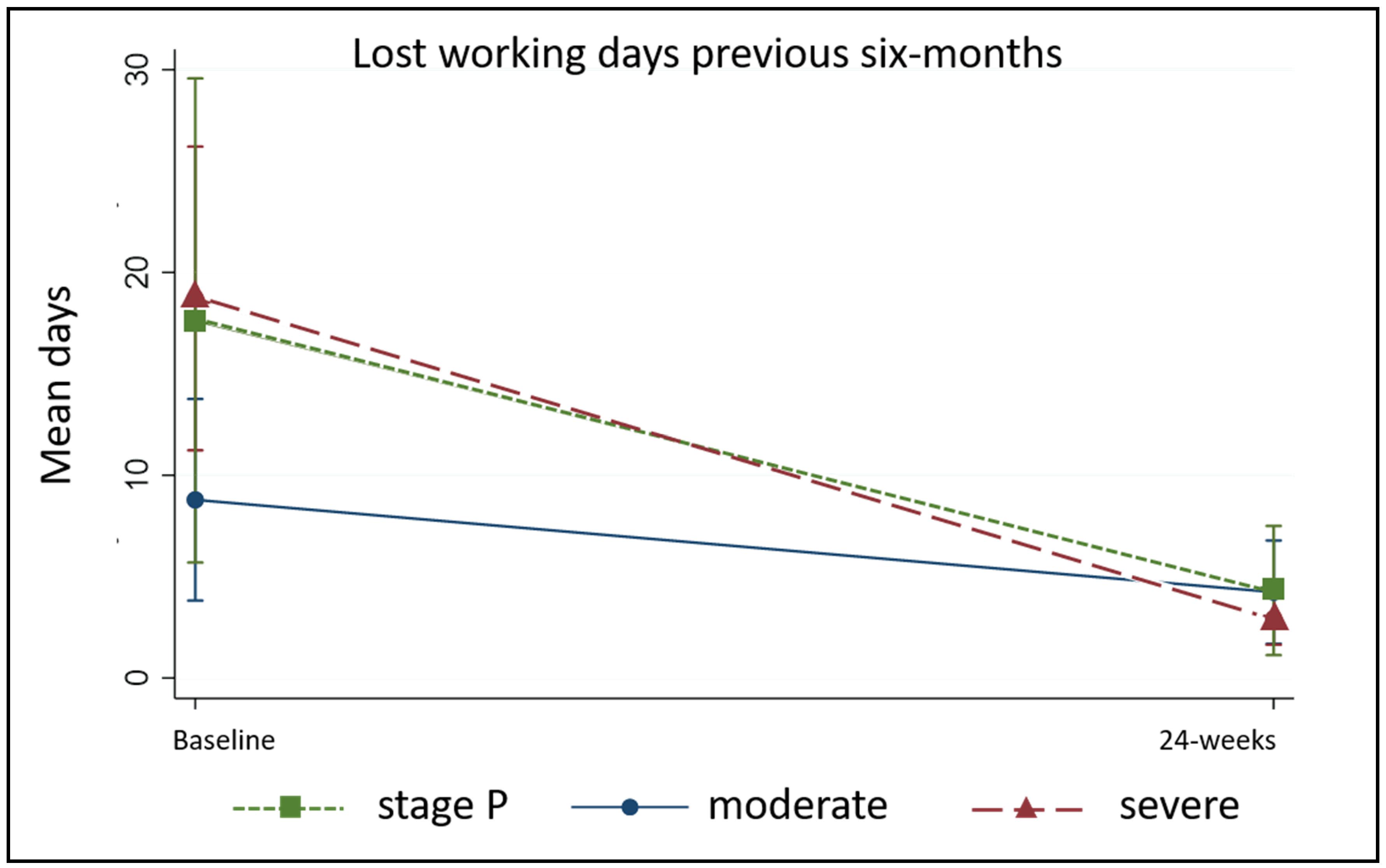
5.2.5. Duration of Acute Attacks and Days of Work Lost to Lymphedema
5.2.6. Entry Lesions and Interdigital Lesions
6. Discussion
7. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- WHO. Global Programme to Eliminate Lymphatic Filariasis; Annual report of lymphatic filariasis 2000; Rio, D.F., Ed.; Department of Communicable Disease Prevention, Control and Eradication: Geneva, Switzerland, 1999. [Google Scholar]
- WHO. Lymphatic Filariasis: Managing Morbidity and Preventing Disability: An Aide-Memoire for National Programme Managers; WHO: Geneva, Switzerland, 2013. [Google Scholar]
- Deribe, K.; Kebede, B.; Tamiru, M.; Mengistu, B.; Kebede, F.; Martindale, S.; Sime, H.; Mulugeta, A.; Kebede, B.; Sileshi, M.; et al. Integrated morbidity management for lymphatic filariasis and podoconiosis, Ethiopia. Belletin WHO 2017, 95, 652–656. [Google Scholar] [CrossRef]
- Deribe, K.; Cano, J.; Trueba, M.L.; Newport, M.J.; Davey, G. Global epidemiology of podoconiosis: A systematic review. PLoS Negl. Trop. Dis. 2018, 12, e0006324. [Google Scholar] [CrossRef]
- Sawers, L.; Stillwaggon, E. Economic Costs and Benefits of Community-Based Lymphedema-Management Programs for Lymphatic Filariasis in India. Am. J. Trop. Med. Hyg. 2020, 103, 295–302. [Google Scholar] [CrossRef]
- Hounsome, N.; Kassahun, M.M.; Ngari, M.; Berkley, J.A.; Kivaya, E.; Njuguna, P.; Fegan, G.; Tamiru, A.; Kelemework, A.; Amberbir, T.; et al. Cost-effectiveness and social outcomes of a community-based treatment for podoconiosis lymphoedema in the East Gojjam zone, Ethiopia. PLoS Negl. Trop. Dis. 2019, 13, e0007780. [Google Scholar] [CrossRef]
- Caprioli, T.; Martindale, S.; Mengiste, A.; Assefa, D.; Hailekiros, F.; Tamiru, M.; Negussu, N.; Taylor, M.; Betts, H.; Kelly-Hope, L.A. Quantifying the socio-economic impact of leg lymphoedema on patient caregivers in a lymphatic filariasis and podoconiosis co-endemic district of Ethiopia. PLoS Negl. Trop. Dis. 2020, 14, e0008058. [Google Scholar] [CrossRef]
- WHO. Weekly Epidemiological Record; WHO: Geneva, Switzerland, 2020; Volume 43, pp. 509–524. [Google Scholar]
- Davey, G.; Tekola, F.; Newport, M.J. Podoconiosis: Non-infectious geochemical elephantiasis. Trans. R. Soc. Trop. Med. Hyg. 2007, 101, 1175–1180. [Google Scholar] [CrossRef]
- ISL. The diagnosis and treatment of peripheral lymphedema: 2020 consensus document of the international society of lymphology. Lymphology 2020, 53, 3–19. [Google Scholar]
- Tora, A.; Davey, G.; Tadele, G. A qualitative study on stigma and coping strategies of patients with podoconiosis in Wolaita zone, Southern Ethiopia. Int. Health 2011, 3, 176–181. [Google Scholar] [CrossRef]
- Yakob, B.; Deribe, K.; Davey, G. High levels of misconceptions and stigma in a community highly endemic for podoconiosis in southern Ethiopia. Trans. R. Soc. Trop. Med. Hyg. 2008, 102, 439–444. [Google Scholar] [CrossRef]
- Stocks, M.E.; Freeman, M.C.; Addiss, D.G. The Effect of Hygiene-Based Lymphedema Management in Lymphatic Filariasis-Endemic Areas: A Systematic Review and Meta-analysis. PLoS Negl. Trop. Dis. 2015, 9, e0004171. [Google Scholar] [CrossRef]
- Moseley, A.L.; Piller, N.B.; Carati, C.J. The effect of gentle arm exercise and deep breathing on secondary arm lymphedema. Lymphology 2005, 38, 136–145. [Google Scholar] [PubMed]
- Douglass, J.; Immink, M.; Piller, N.; Ullah, S. Yoga for women with breast cancer-related lymphoedema: A preliminary 6-month study. J. Lymphoedema 2012, 7, 30–38. [Google Scholar]
- Fisher, M.I.; Donahoe-Fillmore, B.; Leach, L.; O’Malley, C.; Paeplow, C.; Prescott, T.; Merriman, H. Effects of yoga on arm volume among women with breast cancer related lymphedema: A pilot study. J. Bodyw. Mov. Ther. 2014, 18, 559–565. [Google Scholar] [CrossRef] [PubMed]
- Hayes, S.C.; Reul-Hirche, H.; Turner, J. Exercise and secondary lymphedema: Safety, potential benefits, and research issues. Med. Sci. Sports Exerc. 2009, 41, 483–489. [Google Scholar] [CrossRef] [PubMed]
- Kriederman, B.M.T.; Bernas, M.; Lee-Donaldson, L.; Preciado, S.; Lynch, M.S.B.; Summers, P.; Witte, C.; Witte, M. Limb volume reduction after physical treatment by compression and/or massage in a rodent model of peripheral lymphedema. Lymphology 2002, 35, 23–27. [Google Scholar] [PubMed]
- Douglass, J.; Graves, P.; Gordon, S. Self-Care for Management of Secondary Lymphedema: A Systematic Review. PLoS Negl. Trop. Dis. 2016, 10, e0004740. [Google Scholar] [CrossRef]
- Thompson, B.; Gaitatzis, K.; Janse de Jonge, X.; Blackwell, R.; Koelmeyer, L.A. Manual lymphatic drainage treatment for lymphedema: A systematic review of the literature. J. Cancer Surviv. 2020. [Google Scholar] [CrossRef]
- Douglass, J.; Mableson, H.; Martindale, S.; Jhara, S.T.; Karim, M.J.; Rahman, M.M.; Kawsar, A.A.; Khair, A.; Mahmood, A.S.; Rahman, A.F.; et al. Effect of an Enhanced Self-Care Protocol on Lymphedema Status among People Affected by Moderate to Severe Lower-Limb Lymphedema in Bangladesh, a Cluster Randomized Controlled Trial. J. Clin. Med. 2020, 9, 2444. [Google Scholar] [CrossRef]
- Vaqas, B.; Ryan, T.J. Lymphoedema: Pathophysiology and management in resource-poor settings-Relevance for lymphatic filariasis control programmes. Filaria J. 2003, 2. [Google Scholar] [CrossRef]
- Szuba, A.; Rockson, S.G. Lymphedema: Anatomy, physiology and pathogenesis. Vasc. Med. 1997, 2, 321–326. [Google Scholar] [CrossRef]
- Lawenda, B.D.; Mondry, T.E.; Johnstone, P.A.S. Lymphedema: A primer on the identification and management of a chronic condition in oncologic treatment. CA Cancer J. Clin. 2009, 59, 8–24. [Google Scholar] [CrossRef]
- Gordon, S.; Melrose, W.; Warner, J.; Buttner, P.; Ward, L. Lymphatic filariasis: A method to identify subclinical lower limb change in PNG adolescents. PLoS Negl. Trop. Dis. 2011, 5, e1242. [Google Scholar] [CrossRef]
- Soran, A.; Ozmen, T.; McGuire, K.P.; Diego, E.J.; McAuliffe, P.F.; Bonaventura, M.; Ahrendt, G.M.; Degore, L.; Johnson, R. The importance of detection of subclinical lymphedema for the prevention of breast cancer-related clinical lymphedema after axillary lymph node dissection; A prospective observational study. Lymphat. Res. Biol. 2014, 12, 289–294. [Google Scholar] [CrossRef]
- Douglass, J.; Graves, P.; Lindsay, D.; Becker, L.; Roineau, M.; Masson, J.; Aye, N.N.; Win, S.S.; Wai, T.; Win, Y.Y.; et al. Lymphatic Filariasis Increases Tissue Compressibility and Extracellular Fluid in Lower Limbs of Asymptomatic Young People in Central Myanmar. Trop. Med. Infect. Dis. 2017, 2, 50. [Google Scholar] [CrossRef]
- Douglass, J.; Mableson, H.E.; Martindale, S.; Kelly-Hope, L.A. An Enhanced Self-Care Protocol for People Affected by Moderate to Severe Lymphedema. Methods Protoc. 2019, 2, 77. [Google Scholar] [CrossRef] [PubMed]
- Niessen, L.W.; Taylor, M.J. Eliminating Lymphatic Filariasis: Is it Worth it? Clin. Infect. Dis. 2019. [Google Scholar] [CrossRef]
- Karim, M.J.; Haq, R.; Mableson, H.E.; Mahmood, A.S.M.; Rahman, M.; Chowdhury, S.M.; Rahman, A.; Hafiz, I.; Betts, H.; Mackenzie, C.; et al. Developing the first national database and map of lymphatic filariasis clinical cases in Bangladesh: Another step closer to the elimination goals. PLoS Negl. Trop. Dis. 2019, 13, e0007542. [Google Scholar] [CrossRef]
- Mengistu, B.; Deribe, K.; Kebede, F.; Martindale, S.; Hassan, M.; Sime, H.; Mackenzie, C.; Mulugeta, A.; Tamiru, M.; Sileshi, M.; et al. The National Programme to Eliminate Lymphatic Filariasis from Ethiopia. Ethiop. Med. J. 2017, 55, 45–54. [Google Scholar]
- Kebede, B.; Martindale, S.; Mengistu, B.; Kebede, B.; Mengiste, A.; Tamiru, A.; Davey, G.; Kelly-Hope, L.A.; Mackenzie, C.D. Integrated morbidity mapping of lymphatic filariasis and podoconiosis cases in 20 co-endemic districts of Ethiopia. PLoS Negl. Trop. Dis. 2018, 12, e0006491. [Google Scholar] [CrossRef]
- Dreyer, G.; Addiss, D.; Dreyer, P.; Noroes, J. Basic Lymphoedema Management, Treatment and Prevention Problems Associated with Lymphatic Filariasis; Hollis Publishing Company: Hollis, NH, USA, 2002. [Google Scholar]
- Douglass, J.; Mableson, H.; Martindale, S.; Karim, J.; Mahmood, A.S.M.S.; Hailekiros, F.; Kelly-Hope, L. Intra-Rater Reliability and Agreement of the Indurometer When Used to Assess Mid-Calf Tissue Compressibility Among People Affected by Moderate to Severe Lymphedema in Bangladesh and Ethiopia. Lymphat. Res. Biol. 2019. [Google Scholar] [CrossRef]
- Vanderstelt, S.; Pallotta, O.J.; McEwen, M.; Ullah, S.; Burrow, L.; Piller, N. Indurometer vs. Tonometer: Is the Indurometer Currently Able to Replace and Improve Upon the Tonometer? Lymphat. Res. Biol. 2015, 13, 131–136. [Google Scholar] [CrossRef] [PubMed]
- Addiss, D.G.; Louis-Charles, J.; Roberts, J.; Leconte, F.; Wendt, J.M.; Milord, M.D.; Lammie, P.J.; Dreyer, G. Feasibility and effectiveness of basic lymphedema management in Leogane, Haiti, an area endemic for bancroftian filariasis. PLoS Negl. Trop. Dis. 2010, 4, e668. [Google Scholar] [CrossRef]
- Tekola, F.; Ayele, Z.; Mariam, D.H.; Fuller, C.; Davey, G. Development and testing of a de novo clinical staging system for podoconiosis (endemic non-filarial elephantiasis). Trop. Med. Int. Health 2008, 13, 1277–1283. [Google Scholar] [CrossRef] [PubMed]
- Wilson, S.F.; Guarner, J.; Valme, A.L.; Louis-Charles, J.; Jones, T.L.; Addiss, D.G. Histopathologic improvement with lymphedema management, Leogane, Haiti. Emerg. Infect. Dis. 2004, 10, 1938–1946. [Google Scholar] [CrossRef]
- Negussie, H.; Molla, M.; Ngari, M.; Berkley, J.A.; Kivaya, E.; Njuguna, P.; Fegan, G.; Tamiru, A.; Kelemework, A.; Lang, T.; et al. Lymphoedema management to prevent acute dermatolymphangioadenitis in podoconiosis in northern Ethiopia (GoLBeT): A pragmatic randomised controlled trial. Lancet Glob. Health 2018. [Google Scholar] [CrossRef]
- Sikorski, C.; Ashine, M.; Zeleke, Z.; Davey, G. Effectiveness of a simple lymphoedema treatment regimen in podoconiosis management in southern ethiopia: One year follow-up. PLoS Negl. Trop. Dis. 2010, 4, e902. [Google Scholar] [CrossRef]
- Kerketta, A.S.; Babu, B.V.; Rath, K.; Jangid, P.K.; Nayak, A.N.; Kar, S.K. A randomized clinical trial to compare the efficacy of three treatment regimens along with footcare in the morbidity management of filarial lymphoedema. Trop. Med. Int. Health 2005, 10, 698–705. [Google Scholar] [CrossRef]
- Pani, S.P.; Vanamail, P.; Yuvaraj, J. Limb circumference measurement for recording edema volume in patients with filarial lymphedema. Lymphology 1995, 28, 57–63. [Google Scholar]
- Stillwaggon, E.; Sawers, L.; Rout, J.; Addiss, D.; Fox, L. Economic Costs and Benefits of a Community-Based Lymphedema Management Program for Lymphatic Filariasis in Odisha State, India. Am. J. Trop. Med. Hyg. 2016, 95, 877–884. [Google Scholar] [CrossRef]
- McPherson, T.; Persaud, S.; Singh, S.; Fay, M.P.; Addiss, D.; Nutman, T.B.; Hay, R. Interdigital lesions and frequency of acute dermatolymphangioadenitis in lymphoedema in a filariasis-endemic area. Br. J. Dermatol. 2006, 154, 933–941. [Google Scholar] [CrossRef]
- Douglass, J.; Kelly-Hope, L. Comparison of Staging Systems to Assess Lymphedema Caused by Cancer Therapies, Lymphatic Filariasis, and Podoconiosis. Lymphat. Res. Biol. 2018. [Google Scholar] [CrossRef] [PubMed]
- Douglass, J.; Graves, P.; Gordon, S. Moderating Factors in Tissue Tonometry and Bioimpedance Spectroscopy Measures in the Lower Extremity of Healthy Young People in Australia and Myanmar. Lymphat. Res. Biol. 2018, 16, 309–316. [Google Scholar] [CrossRef] [PubMed]

| Standard Self-Care (Control) | Enhanced Self-Care (Intervention) | p = | |
|---|---|---|---|
| Participants | n = 59 | n = 67 | |
| Age, median (IQR) | 58 (50, 65) | 57 (50, 68) | 0.77 |
| Gender = female, n (%) | 24 (41%) | 32 (48%) | 0.42 |
| Marital status = married, n (%) | 40 (68%) | 46 (67%) | 0.17 |
| Literacy = illiterate, n (%) | 44 (74%) | 49 (73%) | 0.56 |
| Maximum stage of lymphedema = | |||
| Moderate n (%) | 14 (24%) | 15 (22%) | 0.86 |
| Severe n (%) | 45 (76%) | 52 (78%) | |
| Severe cases reassigned as stage P n (%) | 7 (12%) | 11 (16%) | 0.47 |
| Interdigital lesions ≥ 1 lesion n (%) | 54 (46%) | 57 (43%) | 0.68 |
| Entry lesions ≥ 1 lesion n (%) | 7 (6%) | 15 (11%) | 0.13 |
| Acute attacks 1-month, median (IQR) | 1 (0, 1) | 1 (0, 1) | 0.48 |
| Lost workdays 1-month, median (IQR) | 3 (0, 6) | 1.5 (0, 4) | 0.22 |
| Acute attacks 6-months, median (IQR) | 3 (1, 5) | 3 (1, 4) | 0.74 |
| Lost workdays 6-months, median (IQR) | 10 (4, 20) | 7 (0, 15) | 0.17 |
| Appropriate footwear n (%) | 20 (34%) | 41 (62%) | 0.004 |
| Circumference, mean cm (SD) | 27.3 (3.46) | 27.4 (3.93) | 0.78 |
| Indurometer score, mean (SD) | 3.20 (0.60) | 2.98 (0.77) | 0.011 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Douglass, J.; Hailekiros, F.; Martindale, S.; Mableson, H.; Seife, F.; Bishaw, T.; Nigussie, M.; Meribo, K.; Tamiru, M.; Agidew, G.; et al. Addition of Lymphatic Stimulating Self-Care Practices Reduces Acute Attacks among People Affected by Moderate and Severe Lower-Limb Lymphedema in Ethiopia, a Cluster Randomized Controlled Trial. J. Clin. Med. 2020, 9, 4077. https://doi.org/10.3390/jcm9124077
Douglass J, Hailekiros F, Martindale S, Mableson H, Seife F, Bishaw T, Nigussie M, Meribo K, Tamiru M, Agidew G, et al. Addition of Lymphatic Stimulating Self-Care Practices Reduces Acute Attacks among People Affected by Moderate and Severe Lower-Limb Lymphedema in Ethiopia, a Cluster Randomized Controlled Trial. Journal of Clinical Medicine. 2020; 9(12):4077. https://doi.org/10.3390/jcm9124077
Chicago/Turabian StyleDouglass, Jan, Fikre Hailekiros, Sarah Martindale, Hayley Mableson, Fikre Seife, Tesfahun Bishaw, Mekdes Nigussie, Kadu Meribo, Mossie Tamiru, Getnet Agidew, and et al. 2020. "Addition of Lymphatic Stimulating Self-Care Practices Reduces Acute Attacks among People Affected by Moderate and Severe Lower-Limb Lymphedema in Ethiopia, a Cluster Randomized Controlled Trial" Journal of Clinical Medicine 9, no. 12: 4077. https://doi.org/10.3390/jcm9124077
APA StyleDouglass, J., Hailekiros, F., Martindale, S., Mableson, H., Seife, F., Bishaw, T., Nigussie, M., Meribo, K., Tamiru, M., Agidew, G., Kim, S., Betts, H., Taylor, M., & Kelly-Hope, L. (2020). Addition of Lymphatic Stimulating Self-Care Practices Reduces Acute Attacks among People Affected by Moderate and Severe Lower-Limb Lymphedema in Ethiopia, a Cluster Randomized Controlled Trial. Journal of Clinical Medicine, 9(12), 4077. https://doi.org/10.3390/jcm9124077





