The Incremental Role of Coronary Computed Tomography in Chronic Coronary Syndromes
Abstract
1. Introduction
2. State of the Art: Technology
2.1. Evolution of Cardiac CT Scanners
2.2. Radiation Exposure and Principle Cardiac CT-Related Risks
2.3. Future Technical Perspectives
3. Screening of Patients: The Calcium Score
4. Clinical Indications of CCTA in the Context of Chronic Coronary Syndromes
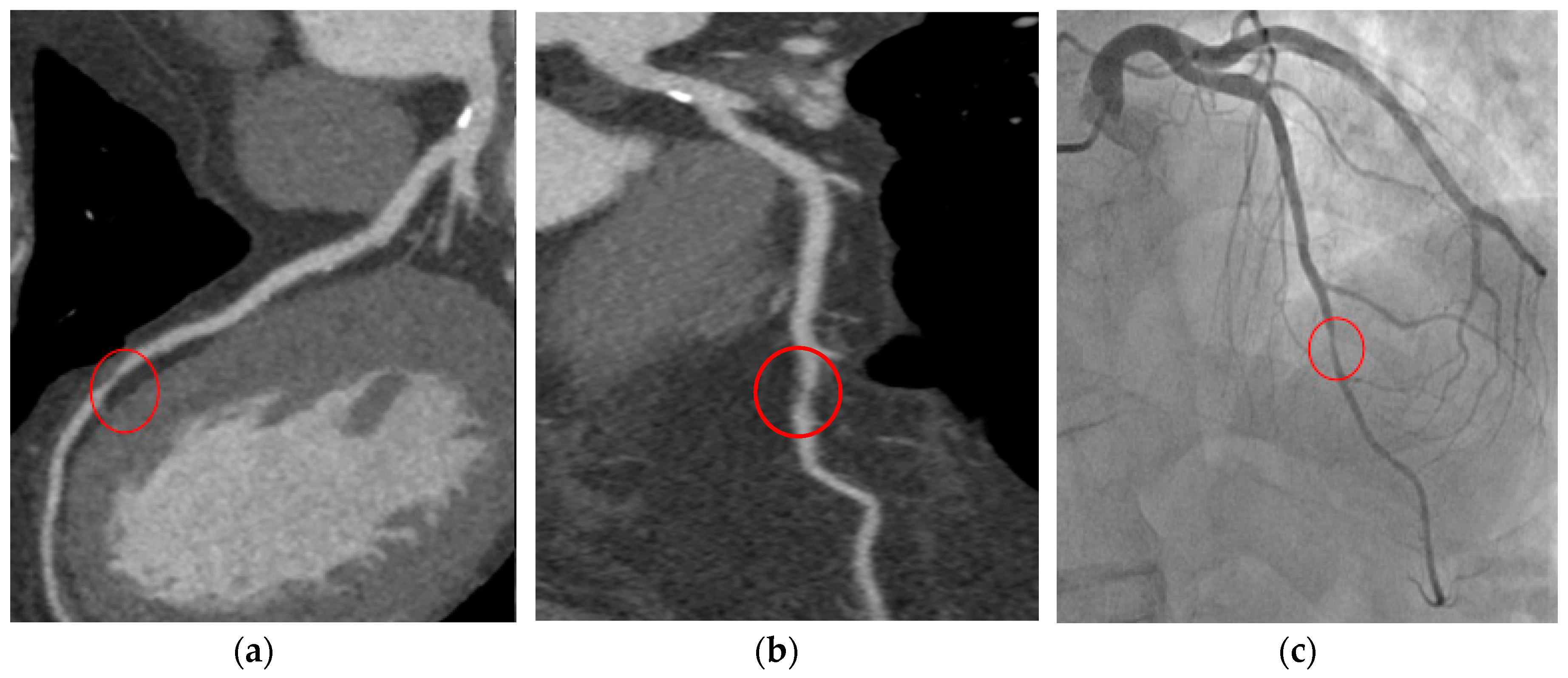
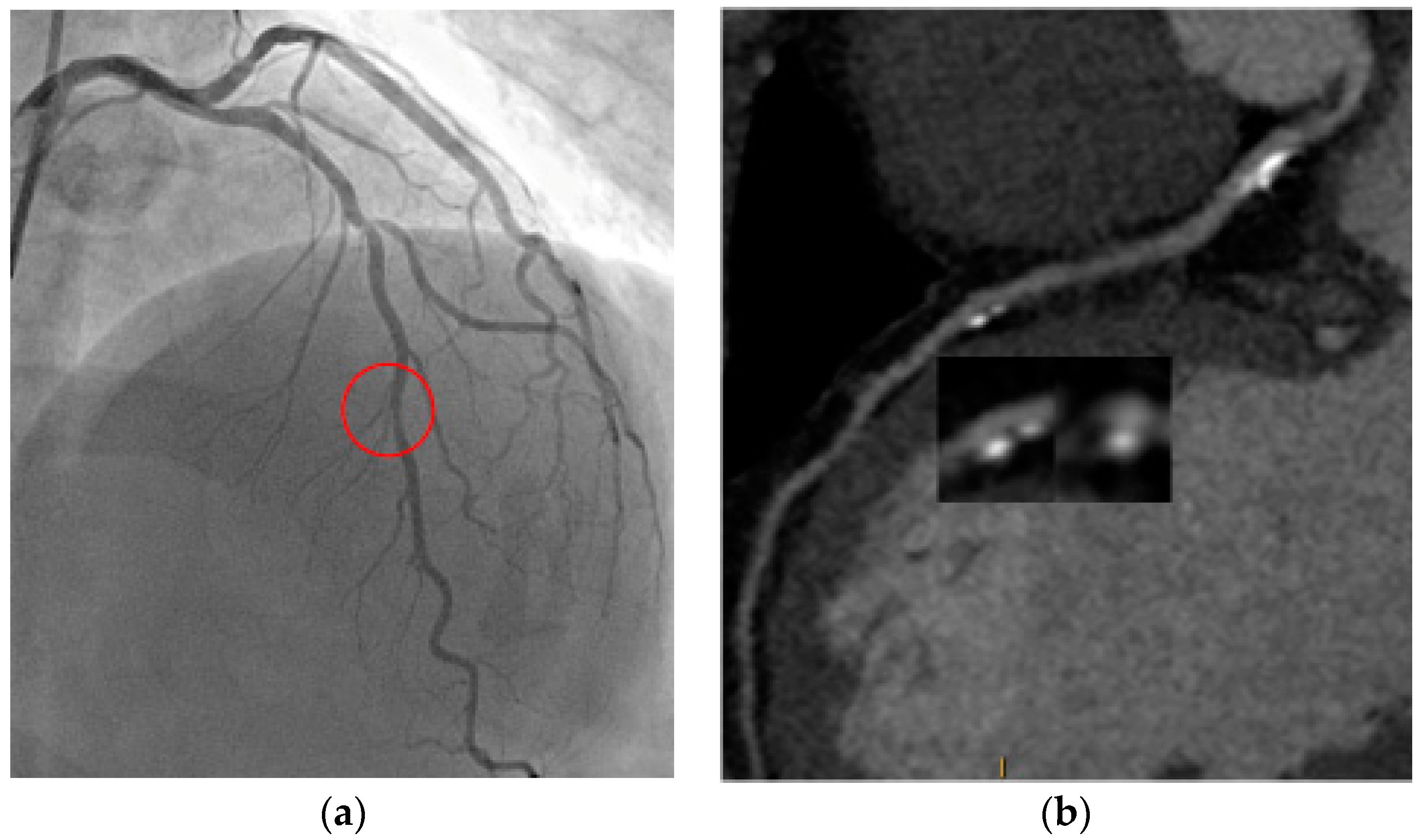
5. Prognosis and Risk Stratification: Plaque Imaging
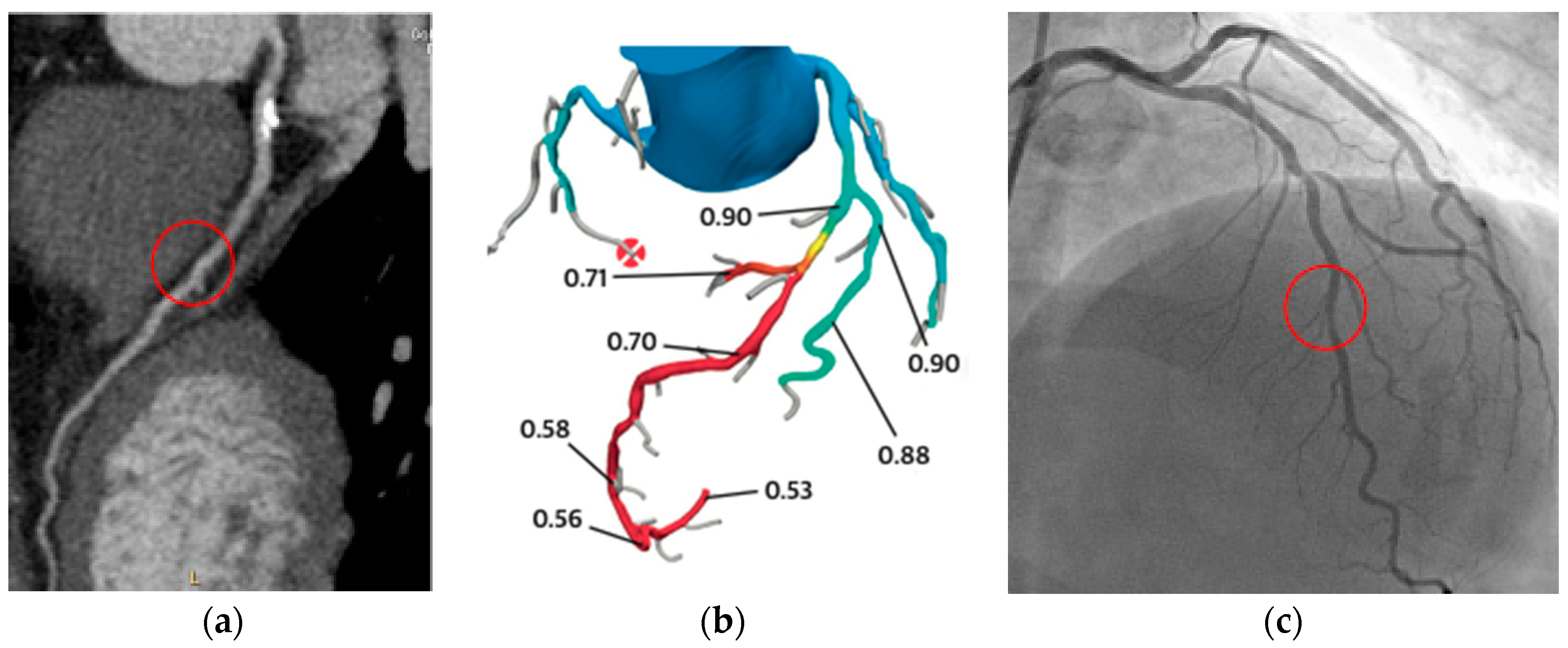
6. Advanced Techniques for Evaluation of Myocardial Ischemia: FFR-CT and Stress-CTP
6.1. FFR-CT
6.2. Stress CTP
7. Specific Roles of Cardiac CT: TAVI Planning and Follow-Up of Heart Transplantation
7.1. Cardiac CT and TAVI
7.2. Cardiac CT and Heart Transplantation
8. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Knuuti, J.; Ballo, H.; Juarez-Orozco, L.E.; Saraste, A.; Kolh, P.; Rutjes, A.W.S.; Jüni, P.; Windecker, S.; Bax, J.J.; Wijns, W. The performance of non-invasive tests to rule-in and rule-out significant coronary artery stenosis in patients with stable angina: A meta-analysis focused on post-test disease probability. Eur. Heart J. 2018, 39, 3322–3330. [Google Scholar] [CrossRef]
- Pontone, G.; Andreini, D.; Quaglia, C.; Ballerini, G.; Nobili, E.; Pepi, M. Accuracy of multidetector spiral computed tomography in detecting significant coronary stenosis in patient populations with differing pre-test probabilities of disease. Clin. Radiol. 2007, 62, 978–985. [Google Scholar] [CrossRef]
- Blanke, P.; Naoum, C.; Ahmadi, A.; Cheruvu, C.; Soon, J.; Arepalli, C.; Gransar, H.; Achenbach, S.; Berman, D.S.; Budoff, M.J.; et al. Long-Term Prognostic Utility of Coronary CT Angiography in Stable Patients With Diabetes Mellitus. JACC Cardiovasc. Imaging 2016, 9, 1280–1288. [Google Scholar] [CrossRef]
- Schulman-Marcus, J.; Hartaigh, B.; Gransar, H.; Lin, F.; Valenti, V.; Cho, I.; Berman, D.; Callister, T.; DeLago, A.; Hadamitzky, M.; et al. Sex-Specific Associations Between Coronary Artery Plaque Extent and Risk of Major Adverse Cardiovascular Events: The CONFIRM Long-Term Registry. JACC Cardiovasc. Imaging 2016, 9, 364–372. [Google Scholar] [CrossRef]
- Pontone, G.; Andreini, D.; Bartorelli, A.L.; Bertella, E.; Cortinovis, S.; Mushtaq, S.; Foti, C.; Annoni, A.; Formenti, A.; Baggiano, A.; et al. A long-term prognostic value of CT angiography and exercise ECG in patients with suspected CAD. JACC Cardiovasc. Imaging 2013, 6, 641–650. [Google Scholar] [CrossRef][Green Version]
- Andreini, D.; Pontone, G.; Bartorelli, A.L.; Agostoni, P.; Mushtaq, S.; Antonioli, L.; Cortinovis, S.; Canestrari, M.; Annoni, A.; Ballerini, G.; et al. Comparison of the diagnostic performance of 64-slice computed tomography coronary angiography in diabetic and non-diabetic patients with suspected coronary artery disease. Cardiovasc. Diabetol. 2010, 9, 80. [Google Scholar] [CrossRef]
- Lewis, M.A.; Pascoal, A.; Keevil, S.F.; Lewis, C.A. Selecting a CT scanner for cardiac imaging: The heart of the matter. Br. J. Radiol. 2016, 89, 20160376. [Google Scholar] [CrossRef]
- Lawler, L.P.; Pannu, H.K.; Fishman, E.K. MDCT evaluation of the coronary arteries, 2004: How we do it--data acquisition, postprocessing, display, and interpretation. AJR Am. J. Roentgenol. 2005, 184, 1402–1412. [Google Scholar] [CrossRef]
- Pontone, G.; Andreini, D.; Guaricci, A.I.; Baggiano, A.; Fazzari, F.; Guglielmo, M.; Muscogiuri, G.; Berzovini, C.M.; Pasquini, A.; Mushtaq, S.; et al. Incremental Diagnostic Value of Stress Computed Tomography Myocardial Perfusion With Whole-Heart Coverage CT Scanner in Intermediate- to High-Risk Symptomatic Patients Suspected of Coronary Artery Disease. JACC Cardiovasc. Imaging 2019, 12, 338–349. [Google Scholar] [CrossRef] [PubMed]
- Moss, A.J.; Williams, M.C.; Newby, D.E.; Nicol, E.D. The Updated NICE Guidelines: Cardiac CT as the First-Line Test for Coronary Artery Disease. Curr. Cardiovasc. Imaging Rep. 2017, 10, 15. [Google Scholar] [CrossRef]
- Andreini, D.; Pontone, G.; Mushtaq, S.; Mancini, M.E.; Conte, E.; Guglielmo, M.; Volpato, V.; Annoni, A.; Baggiano, A.; Formenti, A.; et al. Image quality and radiation dose of coronary CT angiography performed with whole-heart coverage CT scanner with intra-cycle motion correction algorithm in patients with atrial fibrillation. Eur. Radiol. 2018, 28, 1383–1392. [Google Scholar] [CrossRef] [PubMed]
- Chaikriangkrai, K.; Choi, S.Y.; Nabi, F.; Chang, S.M. Important advances in technology and unique applications to cardiovascular computed tomography. Methodist Debakey Cardiovasc. J. 2014, 10, 152–158. [Google Scholar] [CrossRef] [PubMed]
- Katzberg, R.W.; Lamba, R. Contrast-induced nephropathy after intravenous administration: Fact or fiction? Radiol. Clin. N. Am. 2009, 47, 789–800. [Google Scholar] [CrossRef]
- McDonald, J.S.; McDonald, R.J.; Carter, R.E.; Katzberg, R.W.; Kallmes, D.F.; Williamson, E.E. Risk of intravenous contrast material-mediated acute kidney injury: A propensity score-matched study stratified by baseline-estimated glomerular filtration rate. Radiology 2014, 271, 65–73. [Google Scholar] [CrossRef]
- Johnson, P.T.; Pannu, H.K.; Fishman, E.K. IV contrast infusion for coronary artery CT angiography: Literature review and results of a nationwide survey. AJR Am. J. Roentgenol. 2009, 192, 214–221. [Google Scholar] [CrossRef]
- Hein, P.A.; May, J.; Rogalla, P.; Butler, C.; Hamm, B.; Lembcke, A. Feasibility of contrast material volume reduction in coronary artery imaging using 320-slice volume CT. Eur. Radiol. 2010, 20, 1337–1343. [Google Scholar] [CrossRef] [PubMed]
- Kim, R.; Park, E.A.; Lee, W.; Chung, J.W. Feasibility of 320-row area detector CT coronary angiography using 40 mL of contrast material: Assessment of image quality and diagnostic accuracy. Eur. Radiol. 2016, 26, 3802–3810. [Google Scholar] [CrossRef] [PubMed]
- Cademartiri, F.; Maffei, E.; Palumbo, A.A.; Malagò, R.; La Grutta, L.; Meiijboom, W.B.; Aldrovandi, A.; Fusaro, M.; Vignali, L.; Menozzi, A.; et al. Influence of intra-coronary enhancement on diagnostic accuracy with 64-slice CT coronary angiography. Eur. Radiol. 2008, 18, 576–583. [Google Scholar] [CrossRef] [PubMed]
- Cademartiri, F.; Mollet, N.R.; Lemos, P.A.; Saia, F.; Midiri, M.; de Feyter, P.J.; Krestin, G.P. Higher intracoronary attenuation improves diagnostic accuracy in MDCT coronary angiography. AJR Am. J. Roentgenol. 2006, 187, W430–W433. [Google Scholar] [CrossRef]
- Francone, M.; Napoli, A.; Carbone, I.; Cavacece, M.; Nardis, P.G.; Lanciotti, K.; Visconti, S.; Bertoletti, L.; Di Castro, E.; Catalano, C.; et al. Noninvasive imaging of the coronary arteries using a 64-row multidetector CT scanner: Initial clinical experience and radiation dose concerns. Radiol. Med. 2007, 112, 31–46. [Google Scholar] [CrossRef]
- Hsiao, E.M.; Rybicki, F.J.; Steigner, M. CT coronary angiography: 256-slice and 320-detector row scanners. Curr. Cardiol. Rep. 2010, 12, 68–75. [Google Scholar] [CrossRef] [PubMed]
- Rybicki, F.J.; Otero, H.J.; Steigner, M.L.; Vorobiof, G.; Nallamshetty, L.; Mitsouras, D.; Ersoy, H.; Mather, R.T.; Judy, P.F.; Cai, T.; et al. Initial evaluation of coronary images from 320-detector row computed tomography. Int. J. Cardiovasc. Imaging 2008, 24, 535–546. [Google Scholar] [CrossRef] [PubMed]
- Richards, C.E.; Dorman, S.; John, P.; Davies, A.; Evans, S.; Ninan, T.; Martin, D.; Kannoly, S.; Roberts-Davies, G.; Ramsey, M.; et al. Low-radiation and high image quality coronary computed tomography angiography in “real-world” unselected patients. World J. Radiol. 2018, 10, 135–142. [Google Scholar] [CrossRef]
- Chen, M.Y.; Shanbhag, S.M.; Arai, A.E. Submillisievert median radiation dose for coronary angiography with a second-generation 320-detector row CT scanner in 107 consecutive patients. Radiology 2013, 267, 76–85. [Google Scholar] [CrossRef] [PubMed]
- Kodzwa, R. ACR Manual on Contrast Media: 2018 Updates. Radiol. Technol. 2019, 91, 97–100. [Google Scholar]
- Bellin, M.F.; Jakobsen, J.A.; Tomassin, I.; Thomsen, H.S.; Morcos, S.K.; Almén, T.; Aspelin, P.; Clauss, W.; Flaten, H.; Grenier, N.; et al. Contrast medium extravasation injury: Guidelines for prevention and management. Eur. Radiol. 2002, 12, 2807–2812. [Google Scholar] [CrossRef]
- Khan, M.; Cummings, K.W.; Gutierrez, F.R.; Bhalla, S.; Woodard, P.K.; Saeed, I.M. Contraindications and side effects of commonly used medications in coronary CT angiography. Int. J. Cardiovasc. Imaging 2011, 27, 441–449. [Google Scholar] [CrossRef]
- Pourmorteza, A.; Symons, R.; Sandfort, V.; Mallek, M.; Fuld, M.K.; Henderson, G.; Jones, E.C.; Malayeri, A.A.; Folio, L.R.; Bluemke, D.A. Abdominal Imaging with Contrast-enhanced Photon-counting CT: First Human Experience. Radiology 2016, 279, 239–245. [Google Scholar] [CrossRef]
- Symons, R.; Cork, T.E.; Lakshmanan, M.N.; Evers, R.; Davies-Venn, C.; Rice, K.A.; Thomas, M.L.; Liu, C.Y.; Kappler, S.; Ulzheimer, S.; et al. Dual-contrast agent photon-counting computed tomography of the heart: Initial experience. Int. J. Cardiovasc. Imaging 2017, 33, 1253–1261. [Google Scholar] [CrossRef]
- Mannil, M.; Hickethier, T.; von Spiczak, J.; Baer, M.; Henning, A.; Hertel, M.; Schmidt, B.; Flohr, T.; Maintz, D.; Alkadhi, H. Photon-Counting CT: High-Resolution Imaging of Coronary Stents. Invest. Radiol. 2018, 53, 143–149. [Google Scholar] [CrossRef]
- Ferencik, M.; Mayrhofer, T.; Bittner, D.O.; Emami, H.; Puchner, S.B.; Lu, M.T.; Meyersohn, N.M.; Ivanov, A.V.; Adami, E.C.; Patel, M.R.; et al. Use of High-Risk Coronary Atherosclerotic Plaque Detection for Risk Stratification of Patients With Stable Chest Pain: A Secondary Analysis of the PROMISE Randomized Clinical Trial. JAMA Cardiol. 2018, 3, 144–152. [Google Scholar] [CrossRef] [PubMed]
- Muscogiuri, G.; Chiesa, M.; Trotta, M.; Gatti, M.; Palmisano, V.; Dell’Aversana, S.; Baessato, F.; Cavaliere, A.; Cicala, G.; Loffreno, A.; et al. Performance of a deep learning algorithm for the evaluation of CAD-RADS classification with CCTA. Atherosclerosis 2020, 294, 25–32. [Google Scholar] [CrossRef] [PubMed]
- Johnson, R.C.; Leopold, J.A.; Loscalzo, J. Vascular calcification: Pathobiological mechanisms and clinical implications. Circ. Res. 2006, 99, 1044–1059. [Google Scholar] [CrossRef]
- Demer, L.L.; Watson, K.E.; Boström, K. Mechanism of calcification in atherosclerosis. Trends Cardiovasc. Med. 1994, 4, 45–49. [Google Scholar] [CrossRef]
- Agatston, A.S.; Janowitz, W.R.; Hildner, F.J.; Zusmer, N.R.; Viamonte, M.; Detrano, R. Quantification of coronary artery calcium using ultrafast computed tomography. J. Am. Coll. Cardiol. 1990, 15, 827–832. [Google Scholar] [CrossRef]
- Detrano, R.; Guerci, A.D.; Carr, J.J.; Bild, D.E.; Burke, G.; Folsom, A.R.; Liu, K.; Shea, S.; Szklo, M.; Bluemke, D.A.; et al. Coronary calcium as a predictor of coronary events in four racial or ethnic groups. N. Engl. J. Med. 2008, 358, 1336–1345. [Google Scholar] [CrossRef]
- Budoff, M.J.; Mayrhofer, T.; Ferencik, M.; Bittner, D.; Lee, K.L.; Lu, M.T.; Coles, A.; Jang, J.; Krishnam, M.; Douglas, P.S.; et al. Prognostic Value of Coronary Artery Calcium in the PROMISE Study (Prospective Multicenter Imaging Study for Evaluation of Chest Pain). Circulation 2017, 136, 1993–2005. [Google Scholar] [CrossRef]
- Villines, T.C.; Hulten, E.A.; Shaw, L.J.; Goyal, M.; Dunning, A.; Achenbach, S.; Al-Mallah, M.; Berman, D.S.; Budoff, M.J.; Cademartiri, F.; et al. Prevalence and severity of coronary artery disease and adverse events among symptomatic patients with coronary artery calcification scores of zero undergoing coronary computed tomography angiography: Results from the CONFIRM (Coronary CT Angiography Evaluation for Clinical Outcomes: An International Multicenter) registry. J. Am. Coll. Cardiol. 2011, 58, 2533–2540. [Google Scholar] [CrossRef]
- Piepoli, M.F.; Hoes, A.W.; Agewall, S.; Albus, C.; Brotons, C.; Catapano, A.L.; Cooney, M.T.; Corrà, U.; Cosyns, B.; Deaton, C.; et al. 2016 European Guidelines on cardiovascular disease prevention in clinical practice: The Sixth Joint Task Force of the European Society of Cardiology and Other Societies on Cardiovascular Disease Prevention in Clinical Practice (constituted by representatives of 10 societies and by invited experts)Developed with the special contribution of the European Association for Cardiovascular Prevention & Rehabilitation (EACPR). Eur. Heart J. 2016, 37, 2315–2381. [Google Scholar] [CrossRef]
- Winther, S.; Nissen, L.; Westra, J.; Schmidt, S.E.; Bouteldja, N.; Knudsen, L.L.; Madsen, L.H.; Frost, L.; Urbonaviciene, G.; Holm, N.R.; et al. Pre-test probability prediction in patients with a low to intermediate probability of coronary artery disease: A prospective study with a fractional flow reserve endpoint. Eur. Heart J. Cardiovasc. Imaging 2019, 20, 1208–1218. [Google Scholar] [CrossRef]
- Wasfy, M.M.; Brady, T.J.; Abbara, S.; Nasir, K.; Ghoshhajra, B.B.; Truong, Q.A.; Hoffmann, U.; Di Carli, M.F.; Blankstein, R. Comparison of the Diamond-Forrester method and Duke Clinical Score to predict obstructive coronary artery disease by computed tomographic angiography. Am. J. Cardiol. 2012, 109, 998–1004. [Google Scholar] [CrossRef] [PubMed]
- Khera, A.; Budoff, M.J.; O’Donnell, C.J.; Ayers, C.A.; Locke, J.; de Lemos, J.A.; Massaro, J.M.; McClelland, R.L.; Taylor, A.; Levine, B.D. Astronaut Cardiovascular Health and Risk Modification (Astro-CHARM) Coronary Calcium Atherosclerotic Cardiovascular Disease Risk Calculator. Circulation 2018, 138, 1819–1827. [Google Scholar] [CrossRef] [PubMed]
- Greenland, P.; Blaha, M.J.; Budoff, M.J.; Erbel, R.; Watson, K.E. Coronary Calcium Score and Cardiovascular Risk. J. Am. Coll. Cardiol. 2018, 72, 434–447. [Google Scholar] [CrossRef] [PubMed]
- Grundy, S.M.; Stone, N.J. 2018 American Heart Association/American College of Cardiology/Multisociety Guideline on the Management of Blood Cholesterol-Secondary Prevention. JAMA Cardiol. 2019, 4, 589–591. [Google Scholar] [CrossRef]
- Michos, E.D.; Blumenthal, R.S.; Mathews, L.M. My approach to using coronary artery calcium scoring in primary prevention of atherosclerotic cardiovascular disease. Trends Cardiovasc. Med. 2016, 26, 205–206. [Google Scholar] [CrossRef]
- Greenland, P.; Bonow, R.O.; Brundage, B.H.; Budoff, M.J.; Eisenberg, M.J.; Grundy, S.M.; Lauer, M.S.; Post, W.S.; Raggi, P.; Redberg, R.F.; et al. ACCF/AHA 2007 clinical expert consensus document on coronary artery calcium scoring by computed tomography in global cardiovascular risk assessment and in evaluation of patients with chest pain: A report of the American College of Cardiology Foundation Clinical Expert Consensus Task Force (ACCF/AHA Writing Committee to Update the 2000 Expert Consensus Document on Electron Beam Computed Tomography) developed in collaboration with the Society of Atherosclerosis Imaging and Prevention and the Society of Cardiovascular Computed Tomography. J. Am. Coll. Cardiol. 2007, 49, 378–402. [Google Scholar] [CrossRef]
- Blaha, M.J.; Cainzos-Achirica, M.; Greenland, P.; McEvoy, J.W.; Blankstein, R.; Budoff, M.J.; Dardari, Z.; Sibley, C.T.; Burke, G.L.; Kronmal, R.A.; et al. Role of Coronary Artery Calcium Score of Zero and Other Negative Risk Markers for Cardiovascular Disease: The Multi-Ethnic Study of Atherosclerosis (MESA). Circulation 2016, 133, 849–858. [Google Scholar] [CrossRef]
- Frink, R.J.; Achor, R.W.; Brown, A.L.; Kincaid, O.W.; Brandenburg, R.O. Significance of calcification of the coronary arteries. Am. J. Cardiol. 1970, 26, 241–247. [Google Scholar] [CrossRef]
- Wexler, L.; Brundage, B.; Crouse, J.; Detrano, R.; Fuster, V.; Maddahi, J.; Rumberger, J.; Stanford, W.; White, R.; Taubert, K. Coronary artery calcification: Pathophysiology, epidemiology, imaging methods, and clinical implications. A statement for health professionals from the American Heart Association. Writing Group. Circulation 1996, 94, 1175–1192. [Google Scholar] [CrossRef]
- Knuuti, J.; Wijns, W.; Saraste, A.; Capodanno, D.; Barbato, E.; Funck-Brentano, C.; Prescott, E.; Storey, R.F.; Deaton, C.; Cuisset, T.; et al. 2019 ESC Guidelines for the diagnosis and management of chronic coronary syndromes. Eur. Heart J. 2020, 41, 407–477. [Google Scholar] [CrossRef]
- Cheng, V.Y.; Berman, D.S.; Rozanski, A.; Dunning, A.M.; Achenbach, S.; Al-Mallah, M.; Budoff, M.J.; Cademartiri, F.; Callister, T.Q.; Chang, H.J.; et al. Performance of the traditional age, sex, and angina typicality-based approach for estimating pretest probability of angiographically significant coronary artery disease in patients undergoing coronary computed tomographic angiography: Results from the multinational coronary CT angiography evaluation for clinical outcomes: An international multicenter registry (CONFIRM). Circulation 2011, 124, 2423–2432. [Google Scholar] [CrossRef] [PubMed]
- Newby, D.; Williams, M.; Hunter, A.; Pawade, T.; Shah, A.; Flapan, A.; Forbes, J.; Hargreaes, A.; Leslie, S.; Lewis, S.; et al. CT coronary angiography in patients with suspected angina due to coronary heart disease (SCOT-HEART): An open-label, parallel-group, multicentre trial. Lancet 2015, 385, 2383–2391. [Google Scholar] [CrossRef]
- Newby, D.E.; Adamson, P.D.; Berry, C.; Boon, N.A.; Dweck, M.R.; Flather, M.; Forbes, J.; Hunter, A.; Lewis, S.; MacLean, S.; et al. Coronary CT Angiography and 5-Year Risk of Myocardial Infarction. N. Engl. J. Med. 2018, 379, 924–933. [Google Scholar] [CrossRef] [PubMed]
- Douglas, P.S.; Hoffmann, U.; Patel, M.R.; Mark, D.B.; Al-Khalidi, H.R.; Cavanaugh, B.; Cole, J.; Dolor, R.J.; Fordyce, C.B.; Huang, M.; et al. Outcomes of anatomical versus functional testing for coronary artery disease. N. Engl. J. Med. 2015, 372, 1291–1300. [Google Scholar] [CrossRef]
- Neglia, D.; Rovai, D.; Caselli, C.; Pietila, M.; Teresinska, A.; Aguadé-Bruix, S.; Pizzi, M.N.; Todiere, G.; Gimelli, A.; Schroeder, S.; et al. Detection of significant coronary artery disease by noninvasive anatomical and functional imaging. Circ. Cardiovasc. Imaging 2015, 8. [Google Scholar] [CrossRef]
- Hoffmann, U.; Ferencik, M.; Udelson, J.E.; Picard, M.H.; Truong, Q.A.; Patel, M.R.; Huang, M.; Pencina, M.; Mark, D.B.; Heitner, J.F.; et al. Prognostic Value of Noninvasive Cardiovascular Testing in Patients with Stable Chest Pain: Insights From the PROMISE Trial (Prospective Multicenter Imaging Study for Evaluation of Chest Pain). Circulation 2017, 135, 2320–2332. [Google Scholar] [CrossRef]
- Menke, J.; Kowalski, J. Diagnostic accuracy and utility of coronary CT angiography with consideration of unevaluable results: A systematic review and multivariate Bayesian random-effects meta-analysis with intention to diagnose. Eur. Radiol. 2016, 26, 451–458. [Google Scholar] [CrossRef]
- Shaw, L.J.; Berman, D.S.; Picard, M.H.; Friedrich, M.G.; Kwong, R.Y.; Stone, G.W.; Senior, R.; Min, J.K.; Hachamovitch, R.; Scherrer-Crosbie, M. Comparative definitions for moderate-severe ischemia in stress nuclear, echocardiography, and magnetic resonance imaging. JACC Cardiovasc. Imaging 2014, 7, 593–604. [Google Scholar] [CrossRef]
- Chow, B.J.; Small, G.; Yam, Y.; Chen, L.; McPherson, R.; Achenbach, S.; Al-Mallah, M.; Berman, D.S.; Budoff, M.J.; Cademartiri, F.; et al. Prognostic and therapeutic implications of statin and aspirin therapy in individuals with nonobstructive coronary artery disease: Results from the CONFIRM (COronary CT Angiography EvaluatioN for Clinical Outcomes: An InteRnational Multicenter registry) registry. Arterioscler. Thromb. Vasc. Biol. 2015, 35, 981–989. [Google Scholar] [CrossRef]
- Cho, I.; Al’Aref, S.J.; Berger, A.; Ó Hartaigh, B.; Gransar, H.; Valenti, V.; Lin, F.Y.; Achenbach, S.; Berman, D.S.; Budoff, M.J.; et al. Prognostic value of coronary computed tomographic angiography findings in asymptomatic individuals: A 6-year follow-up from the prospective multicentre international CONFIRM study. Eur. Heart J. 2018, 39, 934–941. [Google Scholar] [CrossRef]
- Kelion, A.D.; Nicol, E.D. The rationale for the primacy of coronary CT angiography in the National Institute for Health and Care Excellence (NICE) guideline (CG95) for the investigation of chest pain of recent onset. J. Cardiovasc. Comput. Tomogr. 2018, 12, 516–522. [Google Scholar] [CrossRef] [PubMed]
- Al-Mallah, M.H.; Qureshi, W.; Lin, F.Y.; Achenbach, S.; Berman, D.S.; Budoff, M.J.; Callister, T.Q.; Chang, H.J.; Cademartiri, F.; Chinnaiyan, K.; et al. Does coronary CT angiography improve risk stratification over coronary calcium scoring in symptomatic patients with suspected coronary artery disease? Results from the prospective multicenter international CONFIRM registry. Eur. Heart J. Cardiovasc. Imaging 2014, 15, 267–274. [Google Scholar] [CrossRef] [PubMed]
- Chang, H.J.; Lin, F.Y.; Lee, S.E.; Andreini, D.; Bax, J.; Cademartiri, F.; Chinnaiyan, K.; Chow, B.J.W.; Conte, E.; Cury, R.C.; et al. Coronary Atherosclerotic Precursors of Acute Coronary Syndromes. J. Am. Coll. Cardiol. 2018, 71, 2511–2522. [Google Scholar] [CrossRef] [PubMed]
- Schaar, J.A.; Muller, J.E.; Falk, E.; Virmani, R.; Fuster, V.; Serruys, P.W.; Colombo, A.; Stefanadis, C.; Ward Casscells, S.; Moreno, P.R.; et al. Terminology for high-risk and vulnerable coronary artery plaques. Report of a meeting on the vulnerable plaque, June 17 and 18, 2003, Santorini, Greece. Eur. Heart J. 2004, 25, 1077–1082. [Google Scholar] [CrossRef] [PubMed]
- Falk, E.; Nakano, M.; Bentzon, J.F.; Finn, A.V.; Virmani, R. Update on acute coronary syndromes: The pathologists’ view. Eur. Heart J. 2013, 34, 719–728. [Google Scholar] [CrossRef] [PubMed]
- Burke, A.P.; Farb, A.; Malcom, G.T.; Liang, Y.H.; Smialek, J.; Virmani, R. Coronary risk factors and plaque morphology in men with coronary disease who died suddenly. N. Engl. J. Med. 1997, 336, 1276–1282. [Google Scholar] [CrossRef]
- Narula, J.; Nakano, M.; Virmani, R.; Kolodgie, F.D.; Petersen, R.; Newcomb, R.; Malik, S.; Fuster, V.; Finn, A.V. Histopathologic characteristics of atherosclerotic coronary disease and implications of the findings for the invasive and noninvasive detection of vulnerable plaques. J. Am. Coll. Cardiol. 2013, 61, 1041–1051. [Google Scholar] [CrossRef]
- Maurovich-Horvat, P.; Hoffmann, U.; Vorpahl, M.; Nakano, M.; Virmani, R.; Alkadhi, H. The napkin-ring sign: CT signature of high-risk coronary plaques? JACC Cardiovasc. Imaging 2010, 3, 440–444. [Google Scholar] [CrossRef]
- Otsuka, K.; Fukuda, S.; Tanaka, A.; Nakanishi, K.; Taguchi, H.; Yoshikawa, J.; Shimada, K.; Yoshiyama, M. Napkin-ring sign on coronary CT angiography for the prediction of acute coronary syndrome. JACC Cardiovasc. Imaging 2013, 6, 448–457. [Google Scholar] [CrossRef]
- Creager, M.D.; Hohl, T.; Hutcheson, J.D.; Moss, A.J.; Schlotter, F.; Blaser, M.C.; Park, M.A.; Lee, L.H.; Singh, S.A.; Alcaide-Corral, C.J.; et al. F-Fluoride Signal Amplification Identifies Microcalcifications Associated With Atherosclerotic Plaque Instability in Positron Emission Tomography/Computed Tomography Images. Circ. Cardiovasc. Imaging 2019, 12, e007835. [Google Scholar] [CrossRef]
- Goeller, M.; Achenbach, S.; Cadet, S.; Kwan, A.C.; Commandeur, F.; Slomka, P.J.; Gransar, H.; Albrecht, M.H.; Tamarappoo, B.K.; Berman, D.S.; et al. Pericoronary Adipose Tissue Computed Tomography Attenuation and High-Risk Plaque Characteristics in Acute Coronary Syndrome Compared With Stable Coronary Artery Disease. JAMA Cardiol. 2018, 3, 858–863. [Google Scholar] [CrossRef] [PubMed]
- Motoyama, S.; Kondo, T.; Anno, H.; Sugiura, A.; Ito, Y.; Mori, K.; Ishii, J.; Sato, T.; Inoue, K.; Sarai, M.; et al. Atherosclerotic plaque characterization by 0.5-mm-slice multislice computed tomographic imaging. Circ. J. 2007, 71, 363–366. [Google Scholar] [CrossRef] [PubMed]
- Nakazato, R.; Otake, H.; Konishi, A.; Iwasaki, M.; Koo, B.K.; Fukuya, H.; Shinke, T.; Hirata, K.; Leipsic, J.; Berman, D.S.; et al. Atherosclerotic plaque characterization by CT angiography for identification of high-risk coronary artery lesions: A comparison to optical coherence tomography. Eur. Heart J. Cardiovasc. Imaging 2015, 16, 373–379. [Google Scholar] [CrossRef] [PubMed]
- Glagov, S.; Weisenberg, E.; Zarins, C.K.; Stankunavicius, R.; Kolettis, G.J. Compensatory enlargement of human atherosclerotic coronary arteries. N. Engl. J. Med. 1987, 316, 1371–1375. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, U.; Moselewski, F.; Nieman, K.; Jang, I.K.; Ferencik, M.; Rahman, A.M.; Cury, R.C.; Abbara, S.; Joneidi-Jafari, H.; Achenbach, S.; et al. Noninvasive assessment of plaque morphology and composition in culprit and stable lesions in acute coronary syndrome and stable lesions in stable angina by multidetector computed tomography. J. Am. Coll. Cardiol. 2006, 47, 1655–1662. [Google Scholar] [CrossRef] [PubMed]
- Ahmadi, A.; Leipsic, J.; Øvrehus, K.A.; Gaur, S.; Bagiella, E.; Ko, B.; Dey, D.; LaRocca, G.; Jensen, J.M.; Bøtker, H.E.; et al. Lesion-Specific and Vessel-Related Determinants of Fractional Flow Reserve Beyond Coronary Artery Stenosis. JACC Cardiovasc. Imaging 2018, 11, 521–530. [Google Scholar] [CrossRef]
- Pontone, G. Anatomy and physiology in ischaemic heart disease: A second honeymoon? Eur. Heart J. 2016, 37, 1228–1231. [Google Scholar] [CrossRef]
- Motoyama, S.; Ito, H.; Sarai, M.; Kondo, T.; Kawai, H.; Nagahara, Y.; Harigaya, H.; Kan, S.; Anno, H.; Takahashi, H.; et al. Plaque Characterization by Coronary Computed Tomography Angiography and the Likelihood of Acute Coronary Events in Mid-Term Follow-Up. J. Am. Coll. Cardiol. 2015, 66, 337–346. [Google Scholar] [CrossRef]
- Motoyama, S.; Kondo, T.; Sarai, M.; Sugiura, A.; Harigaya, H.; Sato, T.; Inoue, K.; Okumura, M.; Ishii, J.; Anno, H.; et al. Multislice computed tomographic characteristics of coronary lesions in acute coronary syndromes. J. Am. Coll. Cardiol. 2007, 50, 319–326. [Google Scholar] [CrossRef]
- Williams, M.C.; Moss, A.J.; Dweck, M.; Adamson, P.D.; Alam, S.; Hunter, A.; Shah, A.S.V.; Pawade, T.; Weir-McCall, J.R.; Roditi, G.; et al. Coronary Artery Plaque Characteristics Associated with Adverse Outcomes in the SCOT-HEART Study. J. Am. Coll. Cardiol. 2019, 73, 291–301. [Google Scholar] [CrossRef]
- Cury, R.C.; Abbara, S.; Achenbach, S.; Agatston, A.; Berman, D.S.; Budoff, M.J.; Dill, K.E.; Jacobs, J.E.; Maroules, C.D.; Rubin, G.D.; et al. Coronary Artery Disease-Reporting and Data System (CAD-RADS): An Expert Consensus Document of SCCT, ACR and NASCI: Endorsed by the ACC. JACC Cardiovasc. Imaging 2016, 9, 1099–1113. [Google Scholar] [CrossRef] [PubMed]
- Arbab-Zadeh, A.; Fuster, V. From Detecting the Vulnerable Plaque to Managing the Vulnerable Patient: JACC State-of-the-Art Review. J. Am. Coll. Cardiol. 2019, 74, 1582–1593. [Google Scholar] [CrossRef]
- Boogers, M.J.; Schuijf, J.D.; Kitslaar, P.H.; van Werkhoven, J.M.; de Graaf, F.R.; Boersma, E.; van Velzen, J.E.; Dijkstra, J.; Adame, I.M.; Kroft, L.J.; et al. Automated quantification of stenosis severity on 64-slice CT: A comparison with quantitative coronary angiography. JACC Cardiovasc. Imaging 2010, 3, 699–709. [Google Scholar] [CrossRef]
- Øvrehus, K.A.; Gaur, S.; Leipsic, J.; Jensen, J.M.; Dey, D.; Bøtker, H.E.; Ahmadi, A.; Achenbach, S.; Ko, B.; Nørgaard, B.L. CT-based total vessel plaque analyses improves prediction of hemodynamic significance lesions as assessed by fractional flow reserve in patients with stable angina pectoris. J. Cardiovasc. Comput. Tomogr. 2018, 12, 344–349. [Google Scholar] [CrossRef] [PubMed]
- Magnoni, M.; Andreini, D.; Gorini, M.; Moccetti, T.; Modena, M.G.; Canestrari, M.; Berti, S.; Casolo, G.; Gabrielli, D.; Marraccini, P.; et al. Coronary atherosclerosis in outlier subjects at the opposite extremes of traditional risk factors: Rationale and preliminary results of the Coronary Atherosclerosis in outlier subjects: Protective and novel Individual Risk factors Evaluation (CAPIRE) study. Am. Heart J. 2016, 173, 18–26. [Google Scholar] [CrossRef] [PubMed]
- Conte, E.; Andreini, D.; Magnoni, M.; Masson, S.; Mushtaq, S.; Berti, S.; Canestrari, M.; Casolo, G.; Gabrielli, D.; Latini, R.; et al. Association of high-risk coronary atherosclerosis at CCTA with clinical and circulating biomarkers: Insight from CAPIRE study. J. Cardiovasc. Comput. Tomogr. 2020. [Google Scholar] [CrossRef]
- Lee, S.E.; Chang, H.J.; Rizvi, A.; Hadamitzky, M.; Kim, Y.J.; Conte, E.; Andreini, D.; Pontone, G.; Volpato, V.; Budoff, M.J.; et al. Rationale and design of the Progression of AtheRosclerotic PlAque DetermIned by Computed TomoGraphic Angiography IMaging (PARADIGM) registry: A comprehensive exploration of plaque progression and its impact on clinical outcomes from a multicenter serial coronary computed tomographic angiography study. Am. Heart J. 2016, 182, 72–79. [Google Scholar] [CrossRef]
- Shin, S.; Park, H.B.; Chang, H.J.; Arsanjani, R.; Min, J.K.; Kim, Y.J.; Lee, B.K.; Choi, J.H.; Hong, G.R.; Chung, N. Impact of Intensive LDL Cholesterol Lowering on Coronary Artery Atherosclerosis Progression: A Serial CT Angiography Study. JACC Cardiovasc. Imaging 2017, 10, 437–446. [Google Scholar] [CrossRef]
- Pontone, G.; Bertella, E.; Mushtaq, S.; Loguercio, M.; Cortinovis, S.; Baggiano, A.; Conte, E.; Annoni, A.; Formenti, A.; Beltrama, V.; et al. Coronary artery disease: Diagnostic accuracy of CT coronary angiography--a comparison of high and standard spatial resolution scanning. Radiology 2014, 271, 688–694. [Google Scholar] [CrossRef]
- Celeng, C.; Leiner, T.; Maurovich-Horvat, P.; Merkely, B.; de Jong, P.; Dankbaar, J.W.; van Es, H.W.; Ghoshhajra, B.B.; Hoffmann, U.; Takx, R.A.P. Anatomical and Functional Computed Tomography for Diagnosing Hemodynamically Significant Coronary Artery Disease: A Meta-Analysis. JACC Cardiovasc. Imaging 2019, 12, 1316–1325. [Google Scholar] [CrossRef]
- Rabbat, M.G.; Berman, D.S.; Kern, M.; Raff, G.; Chinnaiyan, K.; Koweek, L.; Shaw, L.J.; Blanke, P.; Scherer, M.; Jensen, J.M.; et al. Interpreting results of coronary computed tomography angiography-derived fractional flow reserve in clinical practice. J. Cardiovasc. Comput. Tomogr. 2017, 11, 383–388. [Google Scholar] [CrossRef] [PubMed]
- Pijls, N.H.; Sels, J.W. Functional measurement of coronary stenosis. J. Am. Coll. Cardiol. 2012, 59, 1045–1057. [Google Scholar] [CrossRef] [PubMed]
- Pijls, N.H.; Fearon, W.F.; Tonino, P.A.; Siebert, U.; Ikeno, F.; Bornschein, B.; van’t Veer, M.; Klauss, V.; Manoharan, G.; Engstrøm, T.; et al. Fractional flow reserve versus angiography for guiding percutaneous coronary intervention in patients with multivessel coronary artery disease: 2-year follow-up of the FAME (Fractional Flow Reserve Versus Angiography for Multivessel Evaluation) study. J. Am. Coll. Cardiol. 2010, 56, 177–184. [Google Scholar] [CrossRef] [PubMed]
- De Bruyne, B.; Pijls, N.H.; Kalesan, B.; Barbato, E.; Tonino, P.A.; Piroth, Z.; Jagic, N.; Möbius-Winkler, S.; Mobius-Winckler, S.; Rioufol, G.; et al. Fractional flow reserve-guided PCI versus medical therapy in stable coronary disease. N. Engl. J. Med. 2012, 367, 991–1001. [Google Scholar] [CrossRef] [PubMed]
- Taylor, C.A.; Fonte, T.A.; Min, J.K. Computational fluid dynamics applied to cardiac computed tomography for noninvasive quantification of fractional flow reserve: Scientific basis. J. Am. Coll. Cardiol. 2013, 61, 2233–2241. [Google Scholar] [CrossRef] [PubMed]
- Ball, C.; Pontone, G.; Rabbat, M. Fractional Flow Reserve Derived from Coronary Computed Tomography Angiography Datasets: The Next Frontier in Noninvasive Assessment of Coronary Artery Disease. BioMed Res. Int. 2018, 2018, 2680430. [Google Scholar] [CrossRef]
- Coenen, A.; Lubbers, M.M.; Kurata, A.; Kono, A.; Dedic, A.; Chelu, R.G.; Dijkshoorn, M.L.; Gijsen, F.J.; Ouhlous, M.; van Geuns, R.J.; et al. Fractional flow reserve computed from noninvasive CT angiography data: Diagnostic performance of an on-site clinician-operated computational fluid dynamics algorithm. Radiology 2015, 274, 674–683. [Google Scholar] [CrossRef]
- Pontone, G.; Muscogiuri, G.; Andreini, D.; Guaricci, A.I.; Guglielmo, M.; Mushtaq, S.; Baggiano, A.; Conte, E.; Beltrama, V.; Annoni, A.; et al. The New Frontier of Cardiac Computed Tomography Angiography: Fractional Flow Reserve and Stress Myocardial Perfusion. Curr. Treat. Options Cardiovasc. Med. 2016, 18, 74. [Google Scholar] [CrossRef]
- Koo, B.K.; Erglis, A.; Doh, J.H.; Daniels, D.V.; Jegere, S.; Kim, H.S.; Dunning, A.; DeFrance, T.; Lansky, A.; Leipsic, J.; et al. Diagnosis of ischemia-causing coronary stenoses by noninvasive fractional flow reserve computed from coronary computed tomographic angiograms. Results from the prospective multicenter DISCOVER-FLOW (Diagnosis of Ischemia-Causing Stenoses Obtained Via Noninvasive Fractional Flow Reserve) study. J. Am. Coll. Cardiol. 2011, 58, 1989–1997. [Google Scholar] [CrossRef]
- Nørgaard, B.L.; Leipsic, J.; Gaur, S.; Seneviratne, S.; Ko, B.S.; Ito, H.; Jensen, J.M.; Mauri, L.; De Bruyne, B.; Bezerra, H.; et al. Diagnostic performance of noninvasive fractional flow reserve derived from coronary computed tomography angiography in suspected coronary artery disease: The NXT trial (Analysis of Coronary Blood Flow Using CT Angiography: Next Steps). J. Am. Coll. Cardiol. 2014, 63, 1145–1155. [Google Scholar] [CrossRef]
- Douglas, P.S.; De Bruyne, B.; Pontone, G.; Patel, M.R.; Norgaard, B.L.; Byrne, R.A.; Curzen, N.; Purcell, I.; Gutberlet, M.; Rioufol, G.; et al. 1-Year Outcomes of FFRCT-Guided Care in Patients With Suspected Coronary Disease: The PLATFORM Study. J. Am. Coll. Cardiol. 2016, 68, 435–445. [Google Scholar] [CrossRef] [PubMed]
- Hlatky, M.A.; De Bruyne, B.; Pontone, G.; Patel, M.R.; Norgaard, B.L.; Byrne, R.A.; Curzen, N.; Purcell, I.; Gutberlet, M.; Rioufol, G.; et al. Quality-of-Life and Economic Outcomes of Assessing Fractional Flow Reserve With Computed Tomography Angiography: PLATFORM. J. Am. Coll. Cardiol. 2015, 66, 2315–2323. [Google Scholar] [CrossRef] [PubMed]
- Curzen, N.; Rana, O.; Nicholas, Z.; Golledge, P.; Zaman, A.; Oldroyd, K.; Hanratty, C.; Banning, A.; Wheatcroft, S.; Hobson, A.; et al. Does routine pressure wire assessment influence management strategy at coronary angiography for diagnosis of chest pain?: The RIPCORD study. Circ. Cardiovasc. Interv. 2014, 7, 248–255. [Google Scholar] [CrossRef] [PubMed]
- Cook, C.M.; Petraco, R.; Shun-Shin, M.J.; Ahmad, Y.; Nijjer, S.; Al-Lamee, R.; Kikuta, Y.; Shiono, Y.; Mayet, J.; Francis, D.P.; et al. Accuracy of Computed Tomography-Derived Fractional Flow Reserve: A Systematic Review. JAMA Cardiol. 2017, 2, 803–810. [Google Scholar] [CrossRef]
- Fairbairn, T.A.; Nieman, K.; Akasaka, T.; Nørgaard, B.L.; Berman, D.S.; Raff, G.; Hurwitz-Koweek, L.M.; Pontone, G.; Kawasaki, T.; Jensen, J.M.; et al. Real-world clinical utility and impact on clinical decision-making of coronary computed tomography angiography-derived fractional flow reserve: Lessons from the ADVANCE Registry. Eur. Heart J. 2018, 39, 3701–3711. [Google Scholar] [CrossRef]
- Rabbat, M.; Leipsic, J.; Bax, J.; Kauh, B.; Verma, R.; Doukas, D.; Allen, S.; Pontone, G.; Wilber, D.; Mathew, V.; et al. Fractional Flow Reserve Derived from Coronary Computed Tomography Angiography Safely Defers Invasive Coronary Angiography in Patients with Stable Coronary Artery Disease. J. Clin. Med. 2020, 9, 604. [Google Scholar] [CrossRef]
- Andreini, D.; Modolo, R.; Katagiri, Y.; Mushtaq, S.; Sonck, J.; Collet, C.; De Martini, S.; Roberto, M.; Tanaka, K.; Miyazaki, Y.; et al. Impact of Fractional Flow Reserve Derived from Coronary Computed Tomography Angiography on Heart Team Treatment Decision-Making in Patients With Multivessel Coronary Artery Disease: Insights From the SYNTAX III REVOLUTION Trial. Circ. Cardiovasc. Interv. 2019, 12, e007607. [Google Scholar] [CrossRef]
- Driessen, R.S.; Danad, I.; Stuijfzand, W.J.; Raijmakers, P.G.; Schumacher, S.P.; van Diemen, P.A.; Leipsic, J.A.; Knuuti, J.; Underwood, S.R.; van de Ven, P.M.; et al. Comparison of Coronary Computed Tomography Angiography, Fractional Flow Reserve, and Perfusion Imaging for Ischemia Diagnosis. J. Am. Coll. Cardiol. 2019, 73, 161–173. [Google Scholar] [CrossRef]
- Kim, K.H.; Doh, J.H.; Koo, B.K.; Min, J.K.; Erglis, A.; Yang, H.M.; Park, K.W.; Lee, H.Y.; Kang, H.J.; Kim, Y.J.; et al. A novel noninvasive technology for treatment planning using virtual coronary stenting and computed tomography-derived computed fractional flow reserve. JACC Cardiovasc. Interv. 2014, 7, 72–78. [Google Scholar] [CrossRef]
- Nagel, E.; Greenwood, J.P.; McCann, G.P.; Bettencourt, N.; Shah, A.M.; Hussain, S.T.; Perera, D.; Plein, S.; Bucciarelli-Ducci, C.; Paul, M.; et al. Magnetic Resonance Perfusion or Fractional Flow Reserve in Coronary Disease. N. Engl. J. Med. 2019, 380, 2418–2428. [Google Scholar] [CrossRef]
- Rossi, A.; Merkus, D.; Klotz, E.; Mollet, N.; de Feyter, P.J.; Krestin, G.P. Stress myocardial perfusion: Imaging with multidetector CT. Radiology 2014, 270, 25–46. [Google Scholar] [CrossRef] [PubMed]
- Pontone, G.; Andreini, D.; Baggiano, A.; Bertella, E.; Mushtaq, S.; Conte, E.; Beltrama, V.; Guaricci, A.I.; Pepi, M. Functional relevance of coronary artery disease by cardiac magnetic resonance and cardiac computed tomography: Myocardial perfusion and fractional flow reserve. BioMed Res. Int. 2015, 2015, 297696. [Google Scholar] [CrossRef] [PubMed]
- Pontone, G.; Baggiano, A.; Andreini, D.; Guaricci, A.I.; Guglielmo, M.; Muscogiuri, G.; Fusini, L.; Soldi, M.; Del Torto, A.; Mushtaq, S.; et al. Diagnostic accuracy of simultaneous evaluation of coronary arteries and myocardial perfusion with single stress cardiac computed tomography acquisition compared to invasive coronary angiography plus invasive fractional flow reserve. Int. J. Cardiol. 2018, 273, 263–268. [Google Scholar] [CrossRef] [PubMed]
- Pontone, G.; Baggiano, A.; Andreini, D.; Guaricci, A.I.; Guglielmo, M.; Muscogiuri, G.; Fusini, L.; Fazzari, F.; Mushtaq, S.; Conte, E.; et al. Stress Computed Tomography Perfusion Versus Fractional Flow Reserve CT Derived in Suspected Coronary Artery Disease: The PERFECTION Study. JACC Cardiovasc. Imaging 2019, 12, 1487–1497. [Google Scholar] [CrossRef]
- Mehra, V.C.; Valdiviezo, C.; Arbab-Zadeh, A.; Ko, B.S.; Seneviratne, S.K.; Cerci, R.; Lima, J.A.; George, R.T. A stepwise approach to the visual interpretation of CT-based myocardial perfusion. J. Cardiovasc. Comput. Tomogr. 2011, 5, 357–369. [Google Scholar] [CrossRef]
- Yang, D.H.; Kim, Y.H.; Roh, J.H.; Kang, J.W.; Han, D.; Jung, J.; Kim, N.; Lee, J.B.; Ahn, J.M.; Lee, J.Y.; et al. Stress Myocardial Perfusion CT in Patients Suspected of Having Coronary Artery Disease: Visual and Quantitative Analysis-Validation by Using Fractional Flow Reserve. Radiology 2015, 276, 715–723. [Google Scholar] [CrossRef]
- Cury, R.C.; Magalhães, T.A.; Paladino, A.T.; Shiozaki, A.A.; Perini, M.; Senra, T.; Lemos, P.A.; Rochitte, C.E. Dipyridamole stress and rest transmural myocardial perfusion ratio evaluation by 64 detector-row computed tomography. J. Cardiovasc. Comput. Tomogr. 2011, 5, 443–448. [Google Scholar] [CrossRef]
- Pontone, G.; Andreini, D.; Guaricci, A.I.; Guglielmo, M.; Baggiano, A.; Muscogiuri, G.; Fusini, L.; Soldi, M.; Fazzari, F.; Berzovini, C.; et al. Quantitative vs. qualitative evaluation of static stress computed tomography perfusion to detect haemodynamically significant coronary artery disease. Eur. Heart J. Cardiovasc. Imaging 2018, 19, 1244–1252. [Google Scholar] [CrossRef]
- Ho, K.T.; Chua, K.C.; Klotz, E.; Panknin, C. Stress and rest dynamic myocardial perfusion imaging by evaluation of complete time-attenuation curves with dual-source CT. JACC Cardiovasc. Imaging 2010, 3, 811–820. [Google Scholar] [CrossRef]
- Bamberg, F.; Marcus, R.P.; Becker, A.; Hildebrandt, K.; Bauner, K.; Schwarz, F.; Greif, M.; von Ziegler, F.; Bischoff, B.; Becker, H.C.; et al. Dynamic myocardial CT perfusion imaging for evaluation of myocardial ischemia as determined by MR imaging. JACC Cardiovasc. Imaging 2014, 7, 267–277. [Google Scholar] [CrossRef]
- Ko, S.M.; Choi, J.W.; Song, M.G.; Shin, J.K.; Chee, H.K.; Chung, H.W.; Kim, D.H. Myocardial perfusion imaging using adenosine-induced stress dual-energy computed tomography of the heart: Comparison with cardiac magnetic resonance imaging and conventional coronary angiography. Eur. Radiol. 2011, 21, 26–35. [Google Scholar] [CrossRef]
- Rochitte, C.E.; George, R.T.; Chen, M.Y.; Arbab-Zadeh, A.; Dewey, M.; Miller, J.M.; Niinuma, H.; Yoshioka, K.; Kitagawa, K.; Nakamori, S.; et al. Computed tomography angiography and perfusion to assess coronary artery stenosis causing perfusion defects by single photon emission computed tomography: The CORE320 study. Eur. Heart J. 2014, 35, 1120–1130. [Google Scholar] [CrossRef] [PubMed]
- Andreini, D.; Mushtaq, S.; Pontone, G.; Conte, E.; Sonck, J.; Collet, C.; Guglielmo, M.; Baggiano, A.; Trabattoni, D.; Galli, S.; et al. Rationale and design of advantage (additional diagnostic value of CT perfusion over coronary CT angiography in stented patients with suspected in-stent restenosis or coronary artery disease progression) prospective study. J. Cardiovasc. Comput. Tomogr. 2018, 12, 411–417. [Google Scholar] [CrossRef] [PubMed]
- Bamberg, F.; Becker, A.; Schwarz, F.; Marcus, R.P.; Greif, M.; von Ziegler, F.; Blankstein, R.; Hoffmann, U.; Sommer, W.H.; Hoffmann, V.S.; et al. Detection of hemodynamically significant coronary artery stenosis: Incremental diagnostic value of dynamic CT-based myocardial perfusion imaging. Radiology 2011, 260, 689–698. [Google Scholar] [CrossRef] [PubMed]
- So, A.; Wisenberg, G.; Islam, A.; Amann, J.; Romano, W.; Brown, J.; Humen, D.; Jablonsky, G.; Li, J.Y.; Hsieh, J.; et al. Non-invasive assessment of functionally relevant coronary artery stenoses with quantitative CT perfusion: Preliminary clinical experiences. Eur. Radiol. 2012, 22, 39–50. [Google Scholar] [CrossRef] [PubMed]
- Lubbers, M.; Coenen, A.; Kofflard, M.; Bruning, T.; Kietselaer, B.; Galema, T.; Kock, M.; Niezen, A.; Das, M.; van Gent, M.; et al. Comprehensive Cardiac CT With Myocardial Perfusion Imaging Versus Functional Testing in Suspected Coronary Artery Disease: The Multicenter, Randomized CRESCENT-II Trial. JACC Cardiovasc. Imaging 2018, 11, 1625–1636. [Google Scholar] [CrossRef] [PubMed]
- Lu, M.; Wang, S.; Sirajuddin, A.; Arai, A.E.; Zhao, S. Dynamic stress computed tomography myocardial perfusion for detecting myocardial ischemia: A systematic review and meta-analysis. Int. J. Cardiol. 2018, 258, 325–331. [Google Scholar] [CrossRef]
- van Assen, M.; De Cecco, C.N.; Eid, M.; von Knebel Doeberitz, P.; Scarabello, M.; Lavra, F.; Bauer, M.J.; Mastrodicasa, D.; Duguay, T.M.; Zaki, B.; et al. Prognostic value of CT myocardial perfusion imaging and CT-derived fractional flow reserve for major adverse cardiac events in patients with coronary artery disease. J. Cardiovasc. Comput. Tomogr. 2019, 13, 26–33. [Google Scholar] [CrossRef]
- Osnabrugge, R.L.; Mylotte, D.; Head, S.J.; Van Mieghem, N.M.; Nkomo, V.T.; LeReun, C.M.; Bogers, A.J.; Piazza, N.; Kappetein, A.P. Aortic stenosis in the elderly: Disease prevalence and number of candidates for transcatheter aortic valve replacement: A meta-analysis and modeling study. J. Am. Coll. Cardiol. 2013, 62, 1002–1012. [Google Scholar] [CrossRef]
- Chiocchi, M.; Ricci, F.; Pasqualetto, M.; D’Errico, F.; Benelli, L.; Pugliese, L.; Cavallo, A.U.; Forcina, M.; Presicce, M.; De Stasio, V.; et al. Role of computed tomography in transcatheter aortic valve implantation and valve-in-valve implantation: Complete review of preprocedural and postprocedural imaging. J. Cardiovasc. Med. 2020, 21, 182–191. [Google Scholar] [CrossRef]
- Blanke, P.; Weir-McCall, J.R.; Achenbach, S.; Delgado, V.; Hausleiter, J.; Jilaihawi, H.; Marwan, M.; Norgaard, B.L.; Piazza, N.; Schoenhagen, P.; et al. Computed tomography imaging in the context of transcatheter aortic valve implantation (TAVI) / transcatheter aortic valve replacement (TAVR): An expert consensus document of the Society of Cardiovascular Computed Tomography. J. Cardiovasc. Comput. Tomogr. 2019, 13, 1–20. [Google Scholar] [CrossRef]
- Tops, L.F.; Wood, D.A.; Delgado, V.; Schuijf, J.D.; Mayo, J.R.; Pasupati, S.; Lamers, F.P.; van der Wall, E.E.; Schalij, M.J.; Webb, J.G.; et al. Noninvasive evaluation of the aortic root with multislice computed tomography implications for transcatheter aortic valve replacement. JACC Cardiovasc. Imaging 2008, 1, 321–330. [Google Scholar] [CrossRef]
- Yeung, A.C.; Davis, S.F.; Hauptman, P.J.; Kobashigawa, J.A.; Miller, L.W.; Valantine, H.A.; Ventura, H.O.; Wiedermann, J.; Wilensky, R. Incidence and progression of transplant coronary artery disease over 1 year: Results of a multicenter trial with use of intravascular ultrasound. Multicenter Intravascular Ultrasound Transplant Study Group. J. Heart Lung Transplant. 1995, 14, S215–S220. [Google Scholar] [PubMed]
- Stone, G.W.; Maehara, A.; Lansky, A.J.; de Bruyne, B.; Cristea, E.; Mintz, G.S.; Mehran, R.; McPherson, J.; Farhat, N.; Marso, S.P.; et al. A prospective natural-history study of coronary atherosclerosis. N. Engl. J. Med. 2011, 364, 226–235. [Google Scholar] [CrossRef] [PubMed]
- Mittal, T.K.; Panicker, M.G.; Mitchell, A.G.; Banner, N.R. Cardiac allograft vasculopathy after heart transplantation: Electrocardiographically gated cardiac CT angiography for assessment. Radiology 2013, 268, 374–381. [Google Scholar] [CrossRef] [PubMed]
- Bastarrika, G.; Calvo, M.; Ezponda, A.; García-Baizán, A.; Caballeros, M.; Rábago, G. Systolic High-Pitch Coronary CT Angiography for Evaluation of the Coronary Arteries in Heart Transplant Recipients. AJR Am. J. Roentgenol. 2020, 215, 828–833. [Google Scholar] [CrossRef] [PubMed]
- Miller, R.J.H.; Kwiecinski, J.; Shah, K.S.; Eisenberg, E.; Patel, J.; Kobashigawa, J.A.; Azarbal, B.; Tamarappoo, B.; Berman, D.S.; Slomka, P.J.; et al. Coronary computed tomography-angiography quantitative plaque analysis improves detection of early cardiac allograft vasculopathy: A pilot study. Am. J. Transplant. 2020, 20, 1375–1383. [Google Scholar] [CrossRef] [PubMed]
- Oebel, S.; Hamada, S.; Higashigaito, K.; von Spiczak, J.; Klotz, E.; Enseleit, F.; Hinzpeter, R.; Ruschitzka, F.; Manka, R.; Alkadhi, H. Comprehensive morphologic and functional imaging of heart transplant patients: First experience with dynamic perfusion CT. Eur. Radiol. 2018, 28, 4111–4121. [Google Scholar] [CrossRef]

| Year | Detector Rows | Detector z-axis Resolution (mm) | Detector z-axis Coverage (mm) | Temporal Resolution (ms) | Gantry Rotation Time (ms) |
|---|---|---|---|---|---|
| 1998 | 4 | 1–1.25 | 20 | 400 | 500–800 |
| 2001 | 16 | 0.5–0.75 | 24 | 190–250 | 380–500 |
| 2004 | 64 | 0.625 | 40 | 175 | 330–400 |
| 2007–2008 | 256–320 | 0.5–0.625 | 160 | 140–175 | 280–350 |
| 2012 | 640 | 0.5 | 160 | 137 | 275 |
| CT Modality | Effective Dose (mSv) | Additional Risks |
|---|---|---|
| CACS | 1.0–1.5 | - |
| CCTA | <1.0–13.5 | Contrast-related, Beta-blockers/Nitroglycerine |
| FFR-CT | <1.0–13.5 | Contrast-related, Beta-blockers/Nitroglycerine |
| Stress-CTP | 2.5–21.6 | Contrast-related, Beta-blockers/Nitroglycerine, Adenosine |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Baessato, F.; Guglielmo, M.; Muscogiuri, G.; Baggiano, A.; Fusini, L.; Scafuri, S.; Babbaro, M.; Mollace, R.; Collevecchio, A.; Guaricci, A.I.; et al. The Incremental Role of Coronary Computed Tomography in Chronic Coronary Syndromes. J. Clin. Med. 2020, 9, 3925. https://doi.org/10.3390/jcm9123925
Baessato F, Guglielmo M, Muscogiuri G, Baggiano A, Fusini L, Scafuri S, Babbaro M, Mollace R, Collevecchio A, Guaricci AI, et al. The Incremental Role of Coronary Computed Tomography in Chronic Coronary Syndromes. Journal of Clinical Medicine. 2020; 9(12):3925. https://doi.org/10.3390/jcm9123925
Chicago/Turabian StyleBaessato, Francesca, Marco Guglielmo, Giuseppe Muscogiuri, Andrea Baggiano, Laura Fusini, Stefano Scafuri, Mario Babbaro, Rocco Mollace, Ada Collevecchio, Andrea I. Guaricci, and et al. 2020. "The Incremental Role of Coronary Computed Tomography in Chronic Coronary Syndromes" Journal of Clinical Medicine 9, no. 12: 3925. https://doi.org/10.3390/jcm9123925
APA StyleBaessato, F., Guglielmo, M., Muscogiuri, G., Baggiano, A., Fusini, L., Scafuri, S., Babbaro, M., Mollace, R., Collevecchio, A., Guaricci, A. I., Rabbat, M., D’Andrea, A., & Pontone, G. (2020). The Incremental Role of Coronary Computed Tomography in Chronic Coronary Syndromes. Journal of Clinical Medicine, 9(12), 3925. https://doi.org/10.3390/jcm9123925











