Orthopedic Treatment for Class II Malocclusion with Functional Appliances and Its Effect on Upper Airways: A Systematic Review with Meta-Analysis
Abstract
1. Introduction
1.1. Background
1.2. Objective
2. Materials and Methods
2.1. Protocol, Registration, and Review Question
2.2. Eligibility Criteria
2.3. Information Sources and Search
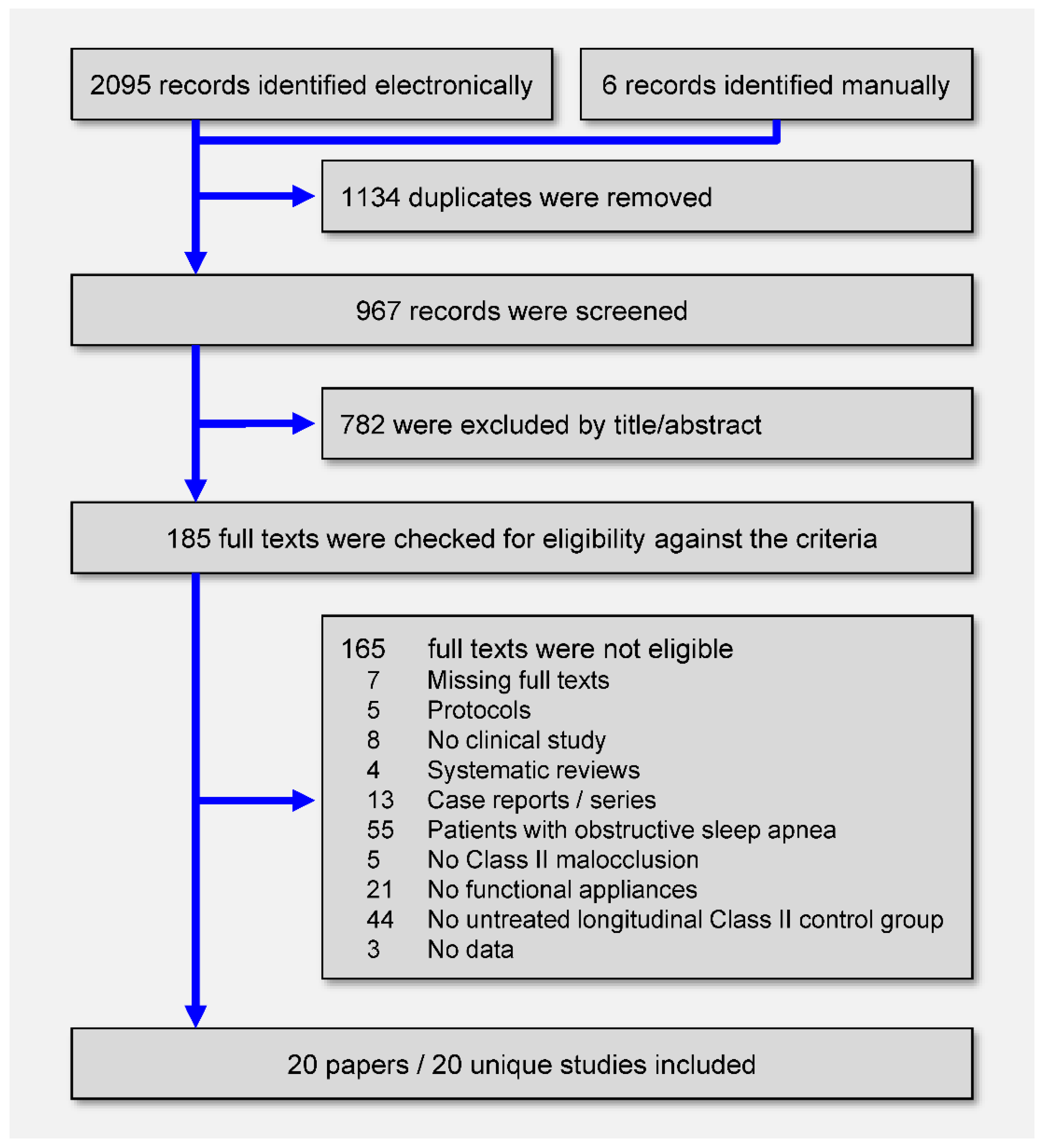
2.4. Study Selection
2.5. Data Collection Process and Items
2.6. Risk of Bias of Individual Studies
2.7. Data Synthesis and Summary Measures
2.8. Additional Analyses and Risk of Bias across Studies
3. Results
3.1. Study Selection
3.2. Study Characteristics
3.3. Risk of Bias within Studies
3.4. Results of Individual Studies and Data Synthesis
3.5. Subgroup and Meta-Regression Analyses
3.6. Reporting Biases and Sensitivity Analyses
3.7. Quality of Evidence
4. Discussion
4.1. Evidence in Context
4.2. Strengths and Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Appendix A
- When before-and-after treatment values were provided, these were converted to treatment-induced increments (post minus pre) according to Cochrane guidelines assuming a pre/post correlation of 0.75 [19].
- The produced forest plots were augmented with contours denoting the magnitude of expected effects: small (up to half a Standard Deviation [SD]), moderate (half to one SD), large (one to two SDs) and very large (more than 2 SDs). The SD for each outcome’s forest plot was based on the average pre-treatment SD of the response variable across all eligible studies for each meta-analysis (rounded up to 2 decimals).
- The standardized mean difference was chosen over the mean difference for the meta-analysis of nasopharynx volume since considerable differences were seen among the measurements of the included studies (one study reporting baseline volume in the control group being twice as large as the volume in the other two studies).
References
- Josefsson, E.; Bjerklin, K.; Lindsten, R. Malocclusion frequency in Swedish and immigrant adolescents—Influence of origin on orthodontic treatment need. Eur. J. Orthod. 2007, 29, 79–87. [Google Scholar] [CrossRef] [PubMed]
- McNamara, J.A., Jr. Components of class II malocclusion in children 8–10 years of age. Angle Orthod. 1981, 51, 177–202. [Google Scholar] [CrossRef] [PubMed]
- Koretsi, V.; Zymperdikas, V.F.; Papageorgiou, S.N.; Papadopoulos, M.A. Treatment effects of removable functional appliances in patients with Class II malocclusion: A systematic review and meta-analysis. Eur. J. Orthod. 2015, 37, 418–434. [Google Scholar] [CrossRef] [PubMed]
- Zymperdikas, V.F.; Koretsi, V.; Papageorgiou, S.N.; Papadopoulos, M.A. Treatment effects of fixed functional appliances in patients with Class II malocclusion: A systematic review and meta-analysis. Eur. J. Orthod. 2016, 38, 113–126. [Google Scholar] [CrossRef]
- Franchi, L.; Pavoni, C.; Faltin, K., Jr.; McNamara, J.A., Jr.; Cozza, P. Long-term skeletal and dental effects and treatment timing for functional appliances in Class II malocclusion. Angle Orthod. 2013, 83, 334–340. [Google Scholar] [CrossRef]
- Batista, K.B.; Thiruvenkatachari, B.; Harrison, J.E.; O’Brien, K.D. Orthodontic treatment for prominent upper front teeth (Class II malocclusion) in children and adolescents. Cochrane Database Syst. Rev. 2018, 3, CD003452. [Google Scholar] [CrossRef]
- Kyburz, K.S.; Eliades, T.; Papageorgiou, S.N. What effect does functional appliance treatment have on the temporomandibular joint? A systematic review with meta-analysis. Prog. Orthod. 2019, 20, 32. [Google Scholar] [CrossRef]
- Arens, R.; Marcus, C.L. Pathophysiology of upper airway obstruction: A developmental perspective. Sleep 2004, 27, 997–1019. [Google Scholar] [CrossRef]
- Schellenberg, J.B.; Maislin, G.; Schwab, R.J. Physical findings and the risk for obstructive sleep apnea. The importance of oropharyngeal structures. Am. J. Respir. Crit. Care Med. 2000, 162, 740–748. [Google Scholar] [CrossRef]
- Claudino, L.V.; Mattos, C.T.; Ruellas, A.C.; Sant’ Anna, E.F. Pharyngeal airway characterization in adolescents related to facial skeletal pattern: A preliminary study. Am. J. Orthod. Dentofac. Orthop. 2013, 143, 799–809. [Google Scholar] [CrossRef]
- Koretsi, V.; Eliades, T.; Papageorgiou, S.N. Oral Interventions for Obstructive Sleep Apnea. Dtsch. Arztebl. Int. 2018, 115, 200–207. [Google Scholar] [CrossRef] [PubMed]
- Lanteri, V.; Farronato, M.; Ugolini, A.; Cossellu, G.; Gaffuri, F.; Parisi, F.M.R.; Cavagnetto, D.; Abate, A.; Maspero, C. Volumetric Changes in the Upper Airways after Rapid and Slow Maxillary Expansion in Growing Patients: A Case-Control Study. Materials 2020, 13, 2239. [Google Scholar] [CrossRef] [PubMed]
- Shete, C.S.; Bhad, W.A. Three-dimensional upper airway changes with mandibular advancement device in patients with obstructive sleep apnea. Am. J. Orthod. Dentofac. Orthop. 2017, 151, 941–948. [Google Scholar] [CrossRef] [PubMed]
- Xiang, M.; Hu, B.; Liu, Y.; Sun, J.; Song, J. Changes in airway dimensions following functional appliances in growing patients with skeletal class II malocclusion: A systematic review and meta-analysis. Int. J. Pediatr. Otorhinolaryngol. 2017, 97, 170–180. [Google Scholar] [CrossRef]
- Sideri, S.; Papageorgiou, S.N.; Eliades, T. Registration in the international prospective register of systematic reviews (PROSPERO) of systematic review protocols was associated with increased review quality. J. Clin. Epidemiol. 2018, 100, 103–110. [Google Scholar] [CrossRef]
- Sterne, J.A.; Hernán, M.A.; Reeves, B.C.; Savović, J.; Berkman, N.D.; Viswanathan, M.; Henry, D.; Altman, D.G.; Ansari, M.T.; Boutron, I.; et al. ROBINS-I: A tool for assessing risk of bias in non-randomised studies of interventions. BMJ 2016, 355, i4919. [Google Scholar] [CrossRef]
- Papageorgiou, S.N. Meta-analysis for orthodontists: Part II—Is all that glitters gold? J. Orthod. 2014, 41, 327–336. [Google Scholar] [CrossRef]
- Guyatt, G.H.; Oxman, A.D.; Schünemann, H.J.; Tugwell, P.; Knottnerus, A. GRADE guidelines: A new series of articles in the Journal of Clinical Epidemiology. J. Clin. Epidemiol. 2011, 64, 380–382. [Google Scholar] [CrossRef]
- Higgins, J.P.T.; Green, S. Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0; The Cochrane Collaboration: London, UK, 2011. [Google Scholar]
- Liberati, A.; Altman, D.G.; Tetzlaff, J.; Mulrow, C.; Gøtzsche, P.C.; Ioannidis, J.P.A.; Clarke, M.; Devereaux, P.J.; Kleijnen, J.; Moher, D. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: Explanation and elaboration. J. Clin. Epidemiol. 2009, 62, e1–e34. [Google Scholar] [CrossRef]
- Papageorgiou, S.N. Meta-analysis for orthodontists: Part I—How to choose effect measure and statistical model. J. Orthod. 2014, 41, 317–326. [Google Scholar] [CrossRef]
- Langan, D.; Higgins, J.P.T.; Jackson, D.; Bowden, J.; Veroniki, A.A.; Kontopantelis, E.; Viechtbauer, W.; Simmonds, M. A comparison of heterogeneity variance estimators in simulated random-effects meta-analyses. Res. Synth. Methods 2019, 10, 83–98. [Google Scholar] [CrossRef] [PubMed]
- Higgins, J.P.T.; Thompson, S.G.; Deeks, J.J.; Altman, D.G. Measuring inconsistency in meta-analyses. BMJ 2003, 327, 557–560. [Google Scholar] [CrossRef] [PubMed]
- IntHout, J.; Ioannidis, J.P.A.; Rovers, M.M.; Goeman, J.J. Plea for routinely presenting prediction intervals in meta-analysis. BMJ Open 2016, 6, e010247. [Google Scholar] [CrossRef]
- Egger, M.; Davey Smith, G.; Schneider, M.; Minder, C. Bias in meta-analysis detected by a simple, graphical test. BMJ 1997, 315, 629–634. [Google Scholar] [CrossRef] [PubMed]
- Schünemann, H.J.; Cuello, C.; Akl, E.A.; Mustafa, R.A.; Meerpohl, J.J.; Thayer, K.; Morgan, R.L.; Gartlehner, G.; Kunz, R.; Katikireddi, S.V.; et al. GRADE guidelines: 18. How ROBINS-I and other tools to assess risk of bias in nonrandomized studies should be used to rate the certainty of a body of evidence. J. Clin. Epidemiol. 2019, 111, 105–114. [Google Scholar] [CrossRef] [PubMed]
- Bidjan, D.; Sallmann, R.; Eliades, T.; Papageorgiou, S.N. Orthopedic treatment for Class II malocclusion with functional appliances and its effect on upper airways: A systematic review with meta-analysis [dataset]. Zenodo 2020. [Google Scholar] [CrossRef]
- Ioannidis, J.P.A. Interpretation of tests of heterogeneity and bias in meta-analysis. J. Eval. Clin. Pract. 2008, 14, 951–957. [Google Scholar] [CrossRef]
- Aksu, M.; Gorucu-Coskuner, H.; Taner, T. Assessment of upper airway size after orthopedic treatment for maxillary protrusion or mandibular retrusion. Am. J. Orthod. Dentofac. Orthop. 2017, 152, 364–370. [Google Scholar] [CrossRef]
- Alhammadi, M.S.; Elfeky, H.Y.; Fayed, M.S.; Ishaq, R.A.R.; Halboub, E.; Al-Mashraqi, A.A. Three-dimensional skeletal and pharyngeal airway changes following therapy with functional appliances in growing skeletal Class II malocclusion patients: A controlled clinical trial. J. Orofac. Orthop. 2019, 80, 254–265. [Google Scholar] [CrossRef]
- Ali, B.; Shaikh, A.; Fida, M. Effect of Clark’s twin-block appliance (CTB) and non-extraction fixed mechano-therapy on the pharyngeal dimensions of growing children. Dent. Press J. Orthod. 2015, 20, 82–88. [Google Scholar] [CrossRef]
- Atik, E.; Gorucu-Coskuner, H.; Kocadereli, I. Dentoskeletal and airway effects of the X-Bow appliance versus removable functional appliances (Frankel-2 and Trainer) in prepubertal Class II division 1 malocclusion patients. Aust. Orthod. J. 2017, 33, 3–13. [Google Scholar]
- Bavbek, N.C.; Tuncer, B.B.; Turkoz, C.; Ulusoy, C.; Tuncer, C. Changes in airway dimensions and hyoid bone position following class II correction with forsus fatigue resistant device. Clin. Oral Investig. 2016, 20, 1747–1755. [Google Scholar] [CrossRef] [PubMed]
- Cortese, M.; Pigato, G.; Casiraghi, G.; Ferrari, M.; Bianco, E.; Maddalone, M. Evaluation of the Oropharyngeal Airway Space in Class II Malocclusion Treated with Mandibular Activator: A Retrospective Study. J. Contemp. Dent. Pract. 2020, 21, 666–672. [Google Scholar] [PubMed]
- Drosen, C.; Bock, N.C.; von Bremen, J.; Pancherz, H.; Ruf, S. Long-term effects of Class II Herbst treatment on the pharyngeal airway width. Eur. J. Orthod. 2018, 40, 82–89. [Google Scholar] [CrossRef] [PubMed]
- Elfeky, H.Y.; Fayed, M.M.S. Three-dimensional effects of twin block therapy on pharyngeal airway parameters in Class II malocclusion patients. J. World Fed. Orthod. 2015, 4, 114–119. [Google Scholar] [CrossRef]
- Entrenas, I.; González-Chamorro, E.; Álvarez-Abad, C.; Muriel, J.; Menéndez-Díaz, I.; Cobo, T. Evaluation of changes in the upper airway after Twin Block treatment in patients with Class II malocclusion. Clin. Exp. Dent. Res. 2019, 5, 259–268. [Google Scholar] [CrossRef] [PubMed]
- Fabiani, G.; Galván Galván, J.; Raucci, G.; Elyasi, M.; Pachêco-Pereira, C.; Flores-Mir, C.; Perillo, L. Pharyngeal airway changes in pre-pubertal children with Class II malocclusion after Frankel-2 treatment. Eur. J. Paediatr. Dent. 2017, 18, 291–295. [Google Scholar] [CrossRef] [PubMed]
- Ghodke, S.; Utreja, A.K.; Singh, S.P.; Jena, A.K. Effects of twin-block appliance on the anatomy of pharyngeal airway passage (PAP) in class II malocclusion subjects. Prog. Orthod. 2014, 15, 68. [Google Scholar] [CrossRef]
- Göymen, M.; Mourad, D.; Güleç, A. Evaluation of Airway Measurements in Class II Patients Following Functional Treatment. Turk. J. Orthod. 2019, 32, 6–10. [Google Scholar] [CrossRef]
- Jena, A.K.; Singh, S.P.; Utreja, A.K. Effectiveness of twin-block and Mandibular Protraction Appliance-IV in the improvement of pharyngeal airway passage dimensions in Class II malocclusion subjects with a retrognathic mandible. Angle Orthod. 2013, 83, 728–734. [Google Scholar] [CrossRef]
- Kilinc, D.D.; Sayar, G. Pharyngeal airway changes of patients after Class II activator treatment. Selcuk Dent. J. 2018, 5, 8–12. [Google Scholar] [CrossRef]
- Oliveira, P.M.; Cheib-Vilefort, P.L.; de Pársia Gontijo, H.; Melgaço, C.A.; Franchi, L.; McNamara, J.A., Jr.; Souki, B.Q. Three-dimensional changes of the upper airway in patients with Class II malocclusion treated with the Herbst appliance: A cone-beam computed tomography study. Am. J. Orthod. Dentofac. Orthop. 2020, 157, 205–211. [Google Scholar] [CrossRef]
- Ozbek, M.M.; Memikoglu, T.U.; Gogen, H.; Lowe, A.A.; Baspinar, E. Oropharyngeal airway dimensions and functional-orthopedic treatment in skeletal Class II cases. Angle Orthod. 1998, 68, 327–336. [Google Scholar] [CrossRef] [PubMed]
- Pavoni, C.; Cretella Lombardo, E.; Franchi, L.; Lione, R.; Cozza, P. Treatment and post-treatment effects of functional therapy on the sagittal pharyngeal dimensions in Class II subjects. Int. J. Pediatr. Otorhinolaryngol. 2017, 101, 47–50. [Google Scholar] [CrossRef] [PubMed]
- Rizk, S.; Kulbersh, V.P.; Al-Qawasmi, R. Changes in the oropharyngeal airway of Class II patients treated with the mandibular anterior repositioning appliance. Angle Orthod. 2016, 86, 955–961. [Google Scholar] [CrossRef] [PubMed]
- Rongo, R.; Martina, S.; Bucci, R.; Festa, P.; Galeotti, A.; Alessandri Bonetti, G.; Michelotti, A.; D’Antò, V. Short-term effects of the Sander bite-jumping appliance on the pharyngeal airways in subjects with skeletal Class II malocclusion: A retrospective case-control study. J. Oral Rehabil. 2020. [Google Scholar] [CrossRef] [PubMed]
- Ulusoy, C.; Canigur Bavbek, N.; Tuncer, B.B.; Tuncer, C.; Turkoz, C.; Gencturk, Z. Evaluation of airway dimensions and changes in hyoid bone position following class II functional therapy with activator. Acta Odontol. Scand. 2014, 72, 917–925. [Google Scholar] [CrossRef] [PubMed]
- Papageorgiou, S.N.; Koletsi, D.; Eliades, T. What evidence exists for myofunctional therapy with prefabricated appliances? A systematic review with meta-analyses of randomised trials. J. Orthod. 2019, 46, 297–310. [Google Scholar] [CrossRef]
- Schwab, R.J.; Gupta, K.B.; Gefter, W.B.; Metzger, L.J.; Hoffman, E.A.; Pack, A.I. Upper airway and soft tissue anatomy in normal subjects and patients with sleep-disordered breathing. Significance of the lateral pharyngeal walls. Am. J. Respir. Crit. Care Med. 1995, 152, 1673–1689. [Google Scholar] [CrossRef]
- Schwab, R.J.; Gefter, W.B.; Hoffman, E.A.; Gupta, K.B.; Pack, A.I. Dynamic upper airway imaging during awake respiration in normal subjects and patients with sleep disordered breathing. Am. Rev. Respir. Dis. 1993, 148, 1385–1400. [Google Scholar] [CrossRef]
- Poole, M.N.; Engel, G.A.; Chaconas, S.J. Nasopharyngeal cephalometrics. Oral Surg. Oral Med. Oral Pathol. 1980, 49, 266–271. [Google Scholar] [CrossRef]
- Schütz, T.C.; Dominguez, G.C.; Hallinan, M.P.; Cunha, T.C.; Tufik, S. Class II correction improves nocturnal breathing in adolescents. Angle Orthod. 2011, 81, 222–228. [Google Scholar] [CrossRef] [PubMed]
- Cozza, P.; Baccetti, T.; Franchi, L.; De Toffol, L.; McNamara, J.A., Jr. Mandibular changes produced by functional appliances in Class II malocclusion: A systematic review. Am. J. Orthod. Dentofac. Orthop. 2006, 129, 599.e1–599.e12. [Google Scholar] [CrossRef] [PubMed]
- Havakeshian, G.; Koretsi, V.; Eliades, T.; Papageorgiou, S.N. Effect of Orthopedic Treatment for Class III Malocclusion on Upper Airways: A Systematic Review and Meta-Analysis. J. Clin. Med. 2020, 9, 3015. [Google Scholar] [CrossRef]
- Papageorgiou, S.N.; Cobourne, M.T. Data sharing in orthodontic research. J. Orthod. 2018, 45, 1–3. [Google Scholar] [CrossRef]
- Papageorgiou, S.N.; Xavier, G.M.; Cobourne, M.T. Basic study design influences the results of orthodontic clinical investigations. J. Clin. Epidemiol. 2015, 68, 1512–1522. [Google Scholar] [CrossRef]
- Papageorgiou, S.N.; Koretsi, V.; Jäger, A. Bias from historical control groups used in orthodontic research: A meta-epidemiological study. Eur. J. Orthod. 2016, 39, 98–105. [Google Scholar] [CrossRef]
- Cappelleri, J.C.; Loannidis, J.P.A.; Schmid, C.H.; De Ferranti, S.D.; Aubert, M.; Chalmers, T.C.; Lau, J. Large Trials vs. Meta-analysis of Smaller Trials. JAMA 1996, 276, 1332–1338. [Google Scholar] [CrossRef]
- Papageorgiou, S.N.; Höchli, D.; Eliades, T. Outcomes of comprehensive fixed appliance orthodontic treatment: A systematic review with meta-analysis and methodological overview. Korean J. Orthod. 2017, 47, 401–413. [Google Scholar] [CrossRef]
- Konstantonis, D.; Vasileiou, D.; Papageorgiou, S.N.; Eliades, T. Soft tissue changes following extraction vs. nonextraction orthodontic fixed appliance treatment: A systematic review and meta-analysis. Eur. J. Oral Sci. 2018, 126, 167–179. [Google Scholar] [CrossRef]
- Battagel, J.M.; Johal, A.; Smith, A.M.; Kotecha, B. Postural variation in oropharyngeal dimensions in subjects with sleep disordered breathing: A cephalometric study. Eur. J. Orthod. 2002, 24, 263–276. [Google Scholar] [CrossRef] [PubMed]
- Hsu, W.E.; Wu, T.Y. Comparison of upper airway measurement by lateral cephalogram in upright position and CBCT in supine position. J. Dent. Sci. 2019, 14, 185–191. [Google Scholar] [CrossRef] [PubMed]
- Eslami, E.; Katz, E.S.; Baghdady, M.; Abramovitch, K.; Masoud, M.I. Are three-dimensional airway evaluations obtained through computed and cone-beam computed tomography scans predictable from lateral cephalograms? A systematic review of evidence. Angle Orthod. 2017, 87, 159–167. [Google Scholar] [CrossRef] [PubMed]
| Study | Design; Setting; Country * | Patients (M/F); Age † | Appliance (Active Duration) | Radio-Graph |
|---|---|---|---|---|
| Aksu 2017 [29] | rNRS; Uni; TR | Exp: 16 (4/12); 10.3 Control: 19 (9/10); 10.2 | Exp: Activator (15.6) Control—(12.0) | Lateral ceph |
| Alhammadi 2019 [30] | pNRS; Uni; EG | Exp1: 23 (0/23); 11.9 Exp2: 21 (0/21); 13.5 Control: 18 (0/18); 11.3 | Exp1: Twin Block (Tx end) Exp2: Forsus FRD (Tx end) Control—(as Exp1–2) | CBCT |
| Ali 2015 [31] | rNRS; Uni; PK | Exp: 42 (21/21); 10.4 Control ‡: 32 (16/16); 10.1 | Exp: Twin Block +FA (36.4) Control—(36.0) | Lateral ceph |
| Atik 2017 [32] | rNRS; Uni; TR | Exp1: 15 (4/11); 8.9 Exp2: 15 (6/9); 10.6 Control: 10 (6/4); 9.3 | Exp1: Fränkel-2 (14.3) Exp2: X-Bow (8.6) Control—(14.8) | Lateral ceph |
| Bavbek 2016 [33] | rNRS; Uni; TR | Exp: 18 (10/8); 13.6 Control: 19 (8/11); 12.7 | Exp: Forsus FRD (8.7) Control—(11.9) | Lateral ceph |
| Cortese 2020 [34] | rNRS; Uni; IT | Exp: 10 (7/3); 10.9 Control: 10 (5/5); 10.1 | Exp: Activator/Twin Block (21.6) Control—(40.8) | Lateral ceph |
| Drosen 2018 [35] | rNRS; Uni; SE | Exp: 13 (13/0); 12.4 Control ‡: 13 (13/0); 12.1 | Exp: Herbst (21.6) Control—(25.2) | Lateral ceph |
| Elfeky 2015 [36] | pNRS; Uni; EG | Exp: 18 (0/18); 10.0–12.0 Control: 18 (0/18); 10.0–12.0 | Exp: Twin Block (8.0) Control—(8.0) | CBCT |
| Entrenas 2019 [37] | pNRS; Uni; ES | Exp: 40 (20/20); 9.8 Control: 20 (10/10); 9.1 | Exp: Twin Block (Tx end) Control—(12.0–24.0) | Lateral ceph |
| Fabiani 2017 [38] | rNRS; Uni; IT | Exp: 28 (13/15); 8.4 Control ‡: 21 (14/7); 8.5 | Exp: Fränkel-2 (14.6) Control—(16.0) | Lateral ceph |
| Ghodke 2014 [39] | pNRS; Uni; IN | Exp: 20 (11/9); 8.0–13.0 Control: 18 (9/9); 8.0–14.0 | Exp: Twin Block (6.0) Control: ± sectionals (6.0) | Lateral ceph |
| Goymen 2019 [40] | rNRS; Uni, TR | Exp1: 15 (7/8); 12.1 Exp2: 15 (7/8); 14.5 Control ‡: 10 (NR); 13.0 | Exp1: Twin Block (Tx end) Exp2: Forsus FRD (Tx end) Control—(6.0) | Lateral ceph |
| Jena 2013 [41] | rNRS; Uni; IN | Exp1: 16 (9/7); 12.8 Exp2: 21 (11/10); 11.4 Control 16 (9/7); 10.6 | Exp1: MAPA4 (6.2) Exp2: Twin Block (9.4) Control: ± sectionals (9.9) | Lateral ceph |
| Kilinc 2018 [42] | uNRS; Uni; TR | Exp: 19 (11/8); NR Control: 19 (7/12); NR | Exp: Activator (11.5) Control—(11.3) | Lateral ceph |
| Oliveira 2020 [43] | rNRS; Uni; BR | Exp: 24 (15/9); NR Control: 18 (10/8); NR | Exp: Herbst (8.0) Control: Pre-Tx (10.4) | CBCT |
| Ozbek 1998 [44] | rNRS; Uni; TR | Exp: 26 (11/15) 11.5 Control: 15 (7/8) 11.3 | Exp: Activator±headgear (17.4) Control—(23.0) | Lateral ceph |
| Pavoni 2017 [45] | uNRS; Uni; IT | Exp: 51 (27/24); 9.9 Control: 31 (15/16); 10.1 | Exp: Activator (21.6) Control—(22.8) | Lateral ceph |
| Rizk 2016 [46] | rNRS; Uni; US | Exp: 20 (7/13); 11.7 Control: 73 (NR); NR | Exp: MARA+FA (27.4) Control—(NR) | CBCT |
| Rongo 2020 [47] | rNRS; Hosp/Uni; IT | Exp: 34 (21/13); 11.1 Control: 34 (25/9); 10.4 | Exp: Sander (14.8) Control—(13.9) | Lateral ceph |
| Ulusoy 2014 [48] | rNRS; Uni; TR | Exp: 16 (8/8); 11.4 Control: 19 (8/11); 12.1 | Exp: Activator (11.0) Control—(11.4) | Lateral ceph |
| Question | Yes/Probably Yes | No/Probably No | No Information |
|---|---|---|---|
| Was the study prospective? | 5 (25%) | 15 (75%) | – |
| Was selection of patients based on any factor that could influence the outcome (malocclusion, airways, compliance, missed appointments, breakages)? | 3 (15%) | 17 (85%) | – |
| Were FA/CTR groups clearly defined? | 19 (95%) | 1 (5%) | – |
| Were FA/CTR patients treated/observed at the same place/time? | 10 (50%) | 4 (20%) | 6 (30%) |
| Were FA/CTR patients matched for baseline age? | 11 (55%) | 5 (25%) | 4 (20%) |
| Were FA/CTR patients matched for baseline sex? | 13 (65%) | 5 (25%) | 2 (10%) |
| Were FA/CTR patients matched for baseline malocclusion? | 12 (60%) | 5 (25%) | 3 (15%) |
| Were FA/CTR patients matched for baseline airway measurements? | 7 (35%) | 13 (65%) | – |
| Was the use of other appliances the same among FA/CTR patients? | 14 (70%) | 6 (30%) | – |
| Was the observation period similar for FA/CTR patients? | 9 (45%) | 7 (35%) | 4 (20%) |
| Were FA/CTR patients measured exactly the same way? | 20 (100%) | – | – |
| Were FA/CTR patients measured blindly? | – | 20 (100%) | – |
| Was the adequate sample? (25 patients per group) | 4 (20%) | 16 (80%) | – |
| Outcome | Studies | MD (95% CI) | p | I2 (95% CI) | tau2 (95% CI) | 95% Prediction |
|---|---|---|---|---|---|---|
| Superoposterior airway space (mm) | 8 | 1.63 (1.03, 2.23) | <0.001 | 68% (28%, 92%) | 0.42 (0.08, 2.15) | −0.13, 3.39 |
| Posterior airway space (mm) | 8 | 0.52 (−0.20, 1.24) | 0.15 | 47% (0%, 87%) | 0.44 (0, 3.44) | −1.34, 2.38 |
| Middle airway space (mm) | 11 | 1.25 (0.53, 1.98) | 0.001 | 81% (58%, 93%) | 1.09 (0.36, 3.69) | −1.25, 3.76 |
| Inferior airway space (mm) | 10 | 1.32 (0.34, 2.31) | 0.009 | 90% (76%, 97%) | 1.97 (0.75, 6.47) | −2.12, 4.76 |
| McNamara’s upper pharynx (mm) | 3 | 1.35 (−0.57, 3.27) | 0.17 | 87% (45%, 99%) | 2.45 (0.31, 48.50) | −22.12, 24.82 |
| McNamara’s lower pharynx (mm) | 3 | 2.31 (0.79, 3.82) | 0.003 | 70% (0%, 99%) | 1.18 (0, 41.05) | −14.64, 19.25 |
| Upper adenoid thickness (AD2-H; mm) | 2 | 0.24 (−2.10, 2.58) | 0.84 | 93% (NE) | 2.65 (NE) | NE |
| Lower adenoid thickness (AD1-Ba; mm) | 2 | 1.16 (0.46, 1.86) | 0.001 | 0% (NE) | 0 (NE) | NE |
| Upper airway thickness (PNS-AD2; mm) | 5 | 0.38 (−0.18, 0.94) | 0.19 | 13% (0%, 89%) | 0.06 (0, 3.00) | −0.81, 1.57 |
| Nasopharynx height (PNS-BaN; mm) | 2 | 0.13 (−0.77, 1.02) | 0.78 | 51% (NE) | 0.21 (NE)) | NE |
| Upper pharyngeal airway passage (Ptm-UPW; mm) | 2 | −0.37 (−1.73, 0.99) | 0.60 | 0% (NE) | 0 (NE) | NE |
| Base of epiglottis-posterior pharyngeal wall (V-LPW; mm) | 4 | 0.70 (0.11, 1.29) | 0.02 | 14% (0%, 93%) | 0.05 (0, 4.46) | −0.93, 2.33 |
| Sagittal depth of bony nasopharynx (Ba-PNS; mm) | 2 | 1.25 (0.06, 2.43) | 0.04 | 21% (NE) | 0.18 (NE) | NE |
| Minimum axial area (mm2) | 2 | 59.91 (41.46, 78.35) | <0.001 | 0% (NE) | 0 (NE) | NE |
| Oropharynx sagittal dimension (mm) | 2 | 1.20 (−2.12, 4.52) | 0.48 | 97% (80%, 100%) | 5.58 (0.68, 721.82) | NE |
| Oropharynx area (units) | 2 * | 556.10 (−279.88, 1392.08) | 0.19 | 0% (NE) | 0 (NE) | NE |
| Nasopharynx volume (mm3) | 3 | 0.95 † (0.36, 1.54) | 0.002 | 60% (0%, 98%) | 0.16 (0, 5.02) | −5.44, 7.34 |
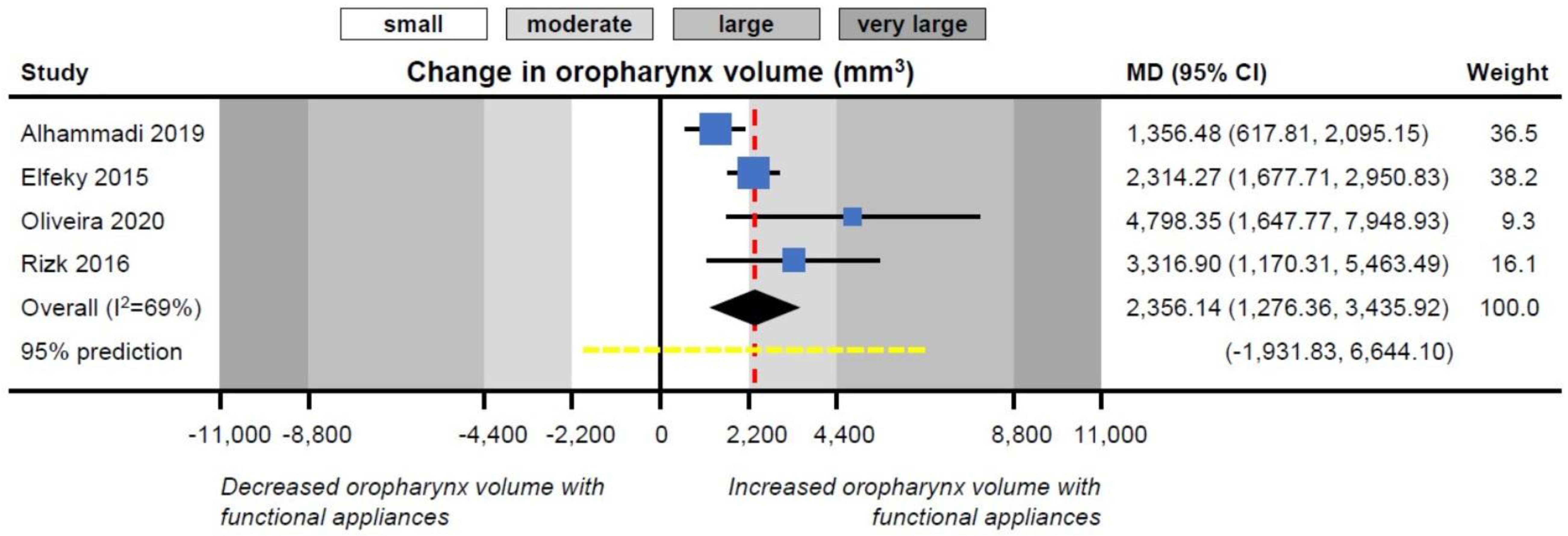
| Oropharynx volume (mm3) | 4 | 2356.14 (1276.36, 3435.92) | <0.001 | 69% (0%, 98%) | >100 (0, >100) | −1931.83, 6644.10 |
| All Appliances | Removable Appliances | Fixed Appliances | |||||
|---|---|---|---|---|---|---|---|
| Outcome | MD (95% CI) | p | MD (95% CI) | p | MD (95% CI) | p | PSG |
| Superoposterior airway space (mm) | n = 8 1.63 (1.03, 2.23) | <0.001 | n = 7 1.41 (0.65, 2.17) | <0.001 | n = 2 1.08 (0.35, 1.82) | 0.004 | 0.20 |
| Posterior airway space (mm) | n = 8 0.52 (−0.20, 1.24) | 0.15 | n = 6 0.83 (−0.18, 1.84) | 0.11 | n = 2 0.14 (−0.77, 1.06) | 0.76 | 0.35 |
| Middle airway space (mm) | n = 11 1.25 0.53, 1.98) | 0.001 | n = 9 1.37 (0.47, 2.26) | 0.003 | n = 3 1.02 (0.29, 1.75) | 0.006 | 0.26 |
| Inferior airway space (mm) | n = 10 1.32 (0.34, 2.31) | 0.009 | n = 8 1.52 (0.32, 2.72) | 0.01 | n = 2 0.79 (0.03, 1.54) | 0.04 | 0.15 |
| McNamara’s upper pharynx (mm) | n = 3 1.35 (−0.57, 3.27) | 0.17 | n = 2 2.05 (−0.04, 4.14) | 0.06 | n = 1 −0.20 (−1.81, 1.41) | 0.81 | NT |
| McNamara’s lower pharynx (mm) | n = 3 2.31 (0.79, 3.82) | 0.003 | n = 2 2.95 (2.13, 3.78) | <0.001 | n = 1 0 (−2.67, 2.67) | 1.00 | NT |
| Upper adenoid thickness (AD2-H; mm) | n = 2 0.24 (−2.10, 2.58) | 0.84 | n = 2 0.24 (−2.10, 2.58) | 0.84 | − | NT | |
| Lower adenoid thickness (AD1-Ba; mm) | n = 2 1.16 (0.46, 1.86) | 0.001 | n = 2 1.16 (0.46, 1.86) | 0.001 | − | NT | |
| Upper airway thickness (PNS-AD2; mm) | n = 5 0.38 (−0.18, 0.94) | 0.19 | n = 4 0.13 (−0.51, 0.78) | 0.69 | n = 1 0.61 (−1.90, 3.12) | 0.63 | 0.73 |
| Nasopharynx height (PNS-BaN; mm) | n = 2 0.13 (−0.77, 1.02) | 0.78 | n = 2 0.27 (−1.01, 1.56) | 0.68 | n = 1 0.02 (−0.88, 0.92) | 0.97 | NT |
| Upper pharyngeal airway passage (Ptm-UPW; mm) | n = 2 −0.37 (−1.73, 0.99) | 0.60 | n = 2 −0.04 (−1.52, 1.44) | 0.96 | n = 1 −1.12 (−3.06, 0.82) | 0.26 | NT |
| Base of epiglottis-posterior pharyngeal wall (V-LPW; mm) | n = 4 0.70 (0.11, 1.29) | 0.02 | n = 3 0.65 (−0.33, 1.62) | 0.19 | n = 2 0.51 (−0.46, 1.48) | 0.30 | NT |
| Sagittal depth of bony nasopharynx (Ba-PNS; mm) | n = 2 1.25 (0.06, 2.43) | 0.04 | n = 2 1.62 (0.57, 2.68) | 0.003 | n = 1 −0.71 (−2.91, 1.49) | 0.53 | NT |
| Minimum axial area (mm2) | n = 2 59.91(41.46, 78.35) | <0.001 | n = 2 91.60 (19.14, 197.56) | 0.01 | n = 1 −26.97 (−44.18, −9.76) | 0.002 | NT |
| Oropharynx sagittal dimension (mm) | n = 2 1.20 (−2.12, 4.52) | 0.48 | n = 1 −0.65 (−0.89, −0.42) | <0.001 | n = 2 1.30 (−1.83, 4.42) | 0.42 | NT |
| Oropharynx area (units) | n = 2 * 556.10 (−279.88, 1392.08) | 0.19 | n = 2 114.35 (98.61, 130.09) | <0.001 | n = 1 607.00 (−452.17, 1666.17) | 0.26 | NT |
| Nasopharynx volume (mm3) | n = 3 0.95 † (0.36, 1.54) | 0.002 | n = 1 1.64 † (0.88, 2.40) | <0.001 | n = 1 0.73 † (0.10, 1.36) | 0.02 | NT |
| Oropharynx volume (mm3) | n = 4 2356.14 (1276.36, 3435.92) | <0.001 | n = 2 2595.56 (2013.07, 3178.05) | <0.001 | n = 3 2303.57 (−808.11, 5415.25) | 0.15 | 0.04 |
| Any Functional Appliance (Removable/Fixed) | Only Removable Appliances | |||||
|---|---|---|---|---|---|---|
| Outcome | Patient Age (Per Year) | Male % in Sample (Per %) | Treatment Duration (Per Month) | Patient Age (Per Year) | Male % in Sample (Per %) | Treatment Duration (Per Month) |
| Upper airway thickness (PNS-AD2; mm) | b = −0.55 p = 0.30 | b = −4.03 p = 0.43 | b = 0.03 p = 0.26 | NT | b = −4.71 p = 0.46 | NT |
| Superoposterior airway space (mm) | b = 0.11 p = 0.69 | b = 1.99 p = 0.47 | b = 0.12 p = 0.09 | b = 0.41 p = 0.41 | b = 2.13 p = 0.49 | b = 0.17 p = 0.12 |
| Posterior airway space (mm) | b = −0.36 p = 0.06 | b = −6.00 p = 0.15 | b = 0.07 p = 0.45 | b = −0.99 p = 0.04 | b = −6.15 p = 0.24 | b = 0.07 p = 0.70 |
| Middle airway space (mm) | b = 0.03 p = 0.91 | b = −2.59 p = 0.41 | b = 0.09 p = 0.28 | b = 0.10 p = 0.84 | b = −3.40 p = 0.37 | b = 0.09 p = 0.48 |
| Inferior airway space (mm) | b = −0.13 p = 0.78 | b = 1.56 p = 0.71 | b = 0.29 p = 0.003 | b = 0.02 p = 0.98 | b = 1.61 p = 0.73 | b = 0.41 p < 0.001 |
| Base of epiglottis-posterior pharyngeal wall (V-LPW; mm) | b = 0.29 p = 0.46 | b = −12.09 p = 0.41 | b = −0.13 p = 0.38 | NT | b = −13.62 p = 0.55 | NT |
| Oropharynx volume (mm3) | b = −1256.28 p = 0.17 | b = 5027.43 p = 0.23 | b = 13.37 p = 0.92 | NT | NT | NT |
| Outcome | Prospective Vs Retrospective (Ref) | Tx-Ctr Difference in Duration (Per Month) | Tx-Ctr Difference in Baseline Outcome (In Absolute Cohen’s d) | Adequate Sample (≥50) vs. Inadequate (Ref) |
|---|---|---|---|---|
| Upper airway thickness (PNS-AD2; mm) | NE | b = 0.05 p = 0.82 | b = 0.38 p = 0.26 | b = 0.39 p = 0.59 |
| Superoposterior airway space (mm) | NE | b = −0.07 p = 0.32 | b = 0.82 p = 0.15 | b = 0.91 p = 0.36 |
| Posterior airway space (mm) | NE | b = 0.04 p = 0.83 | b = 1.86 p = 0.08 | b = −0.46 p = 0.65 |
| Middle airway space (mm) | b = −0.65 p = 0.62 | b = 0.01 p = 0.91 | b = 0.93 p = 0.04 | b = −1.56 p = 0.27 |
| Inferior airway space (mm) | NE | b = −0.03 p = 0.79 | b = 0.51 p = 0.64 | b = 2.05 p = 0.09 |
| Base of epiglottis-posterior pharyngeal wall (V-LPW; mm) | b = 0.95 p = 0.26 | b = 0.03 p = 0.90 | b = −0.48 p = 0.60 | b = −1.45 p = 0.29 |
| Oropharynx volume (mm3) | b = −2334.16 p = 0.27 | NE | b = −3586.53 p = 0.36 | b = −1406.38 p = 0.51 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bidjan, D.; Sallmann, R.; Eliades, T.; Papageorgiou, S.N. Orthopedic Treatment for Class II Malocclusion with Functional Appliances and Its Effect on Upper Airways: A Systematic Review with Meta-Analysis. J. Clin. Med. 2020, 9, 3806. https://doi.org/10.3390/jcm9123806
Bidjan D, Sallmann R, Eliades T, Papageorgiou SN. Orthopedic Treatment for Class II Malocclusion with Functional Appliances and Its Effect on Upper Airways: A Systematic Review with Meta-Analysis. Journal of Clinical Medicine. 2020; 9(12):3806. https://doi.org/10.3390/jcm9123806
Chicago/Turabian StyleBidjan, Darius, Rahel Sallmann, Theodore Eliades, and Spyridon N. Papageorgiou. 2020. "Orthopedic Treatment for Class II Malocclusion with Functional Appliances and Its Effect on Upper Airways: A Systematic Review with Meta-Analysis" Journal of Clinical Medicine 9, no. 12: 3806. https://doi.org/10.3390/jcm9123806
APA StyleBidjan, D., Sallmann, R., Eliades, T., & Papageorgiou, S. N. (2020). Orthopedic Treatment for Class II Malocclusion with Functional Appliances and Its Effect on Upper Airways: A Systematic Review with Meta-Analysis. Journal of Clinical Medicine, 9(12), 3806. https://doi.org/10.3390/jcm9123806







