Novel Sub-Clustering of Class III Skeletal Malocclusion Phenotypes in a Southern European Population Based on Proportional Measurements
Abstract
:1. Introduction
2. Experimental Section
2.1. Study Sample
2.2. Measurements/Assessments: Cephalometric Measurement Selection and Analysis
2.3. Statistical Analysis
Multivariate Analysis
2.4. Method Error
3. Results
3.1. Method Error
3.2. Description of the Main Components
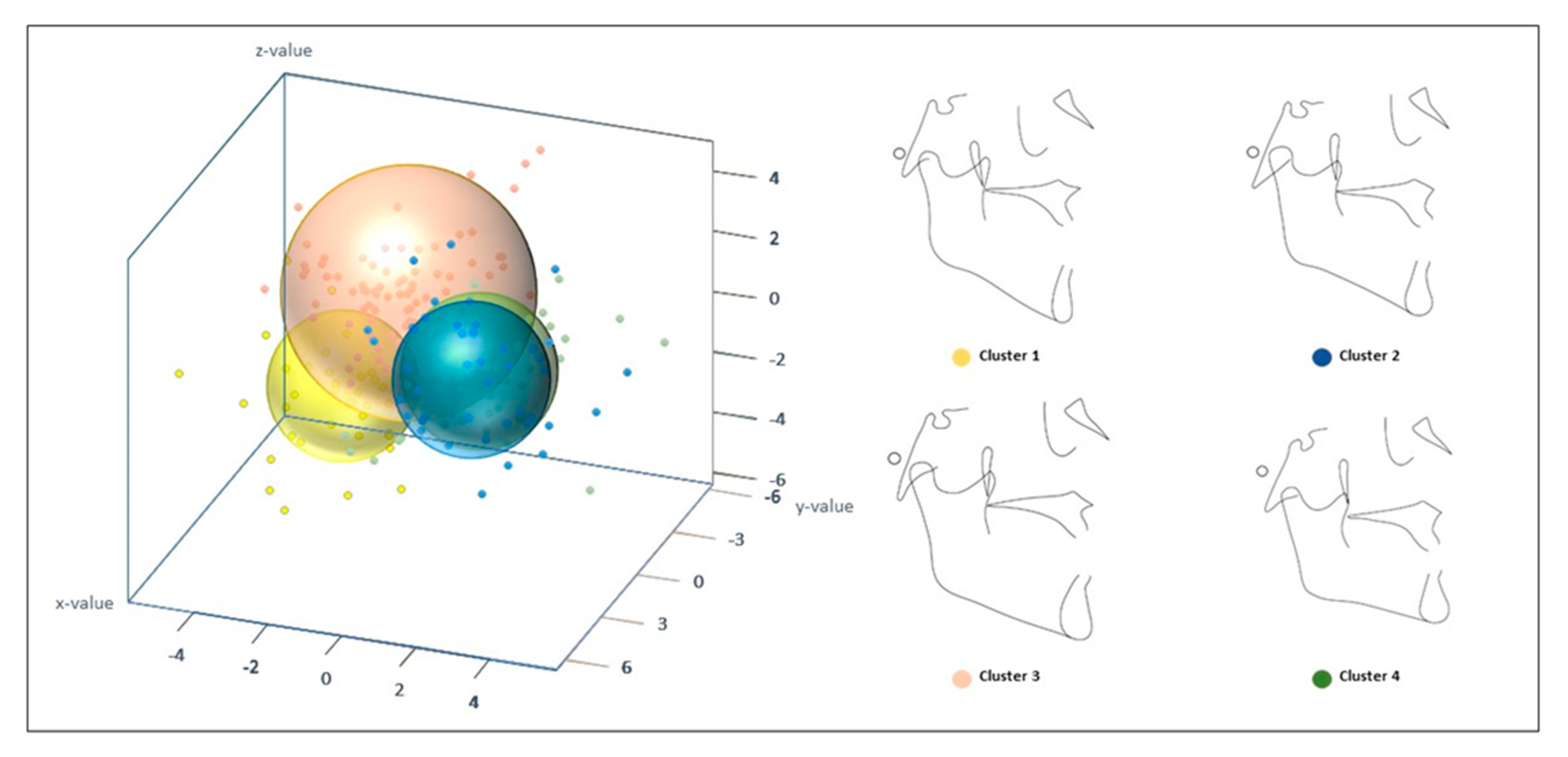
3.3. Proportional Sub-Phenotypic Patterns
3.4. Description of the Supplementary Variables in Each Proportional Sub-Phenotypic Cluster
3.5. Gender and Maturation Stage Distribution in Each Proportional Sub-Phenotypic Pattern
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Lin, Y.; Guo, R.; Hou, L.; Fu, Z.; Li, W. Stability of maxillary protraction therapy in children with Class III malocclusion: A systematic review and meta-analysis. Clin. Oral Investig. 2018, 22, 2639–2652. [Google Scholar] [CrossRef] [PubMed]
- Wendl, B.; Kamenica, A.; Droschl, H.; Jakse, N.; Weiland, F.; Wendl, T.; Wendl, M. Retrospective 25-year follow-up of treatment outcomes in Angle Class III patients: Success versus failure. J. Orofac. Orthop. 2017, 78, 129–136. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, C.M.; Ko, E.C.; Cheng, J.H.; Tseng, Y.C. Correlation between changes in the gonial area and postoperative stability in the treatment of mandibular prognathism. J. Stomatol. Oral Maxillofac. Surg. 2019, 120, 414–418. [Google Scholar] [CrossRef] [PubMed]
- Saito, F.; Kajii, T.S.; Oka, A.; Ikuno, K.; Iida, J. Genome-wide association study for mandibular prognathism using microsatellite and pooled DNA method. Am. J. Orthod. Dentofac. Orthop. 2017, 152, 382–388. [Google Scholar] [CrossRef]
- Doraczynska-Kowalik, A.; Nelke, K.H.; Pawlak, W.; Sasiadek, M.M.; Gerber, H. Genetic Factors Involved in Mandibular Prognathism. J. Craniofac. Surg. 2017, 28, 422–431. [Google Scholar] [CrossRef]
- Cruz, C.V.; Mattos, C.T.; Maia, J.C.; Granjeiro, J.M.; Reis, M.F.; Mucha, J.N.; Vilella, B.; Ruellas, A.C.; Luiz, R.R.; Costa, M.C.; et al. Genetic polymorphisms underlying the skeletal Class III phenotype. Am. J. Orthod. Dentofac. Orthop. 2017, 151, 700–707. [Google Scholar] [CrossRef]
- Liu, H.; Wu, C.; Lin, J.; Shao, J.; Chen, Q.; Luo, E. Genetic Etiology in Nonsyndromic Mandibular Prognathism. J. Craniofac. Surg. 2017, 28, 161–169. [Google Scholar] [CrossRef]
- Genno, P.G.; Nemer, G.M.; Zein Eddine, S.B.; Macari, A.T.; Ghafari, J.G. Three novel genes tied to mandibular prognathism in eastern Mediterranean families. Am. J. Orthod. Dentofac. Orthop. 2019, 156, 104–112. [Google Scholar] [CrossRef]
- Jiang, Q.; Mei, L.; Zou, Y.; Ding, Q.; Cannon, R.D.; Chen, H.; Li, H. Genetic Polymorphisms in FGFR2 Underlie Skeletal Malocclusion. J. Dent. Res. 2019, 98, 1340–1347. [Google Scholar] [CrossRef]
- Lin, M.; Xie, C.; Yang, H.; Wu, C.; Ren, A. Prevalence of malocclusion in Chinese schoolchildren from 1991 to 2018: A systematic review and meta-analysis. Int. J. Paediatr. Dent. 2020, 30, 144–155. [Google Scholar] [CrossRef]
- Piao, Y.; Kim, S.J.; Yu, H.S.; Cha, J.Y.; Baik, H.S. Five-year investigation of a large orthodontic patient population at a dental hospital in South Korea. Korean J. Orthod. 2016, 46, 137–145. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Steinmassl, O.; Steinmassl, P.A.; Schwarz, A.; Crismani, A. Orthodontic Treatment Need of Austrian Schoolchildren in the Mixed Dentition Stage. Swiss Dent. J. 2017, 127, 122–128. [Google Scholar] [PubMed]
- Perillo, L.; Masucci, C.; Ferro, F.; Apicella, D.; Baccetti, T. Prevalence of orthodontic treatment need in southern Italian schoolchildren. Eur. J. Orthod. 2010, 32, 49–53. [Google Scholar] [CrossRef] [PubMed]
- Thilander, B.; Pena, L.; Infante, C.; Parada, S.S.; de Mayorga, C. Prevalence of malocclusion and orthodontic treatment need in children and adolescents in Bogota, Colombia. An epidemiological study related to different stages of dental development. Eur. J. Orthod. 2001, 23, 153–167. [Google Scholar] [CrossRef] [PubMed]
- Narayanan, R.K.; Jeseem, M.T.; Kumar, T.A. Prevalence of Malocclusion among 10–12-year-old Schoolchildren in Kozhikode District, Kerala: An Epidemiological Study. Int. J. Clin. Pediatr. Dent. 2016, 9, 50–55. [Google Scholar] [PubMed]
- Borzabadi-Farahani, A.; Borzabadi-Farahani, A.; Eslamipour, F. Malocclusion and occlusal traits in an urban Iranian population. An epidemiological study of 11- to 14-year-old children. Eur. J. Orthod. 2009, 31, 477–484. [Google Scholar] [CrossRef] [Green Version]
- Bilgic, F.; Gelgor, I.E.; Celebi, A.A. Malocclusion prevalence and orthodontic treatment need in central Anatolian adolescents compared to European and other nations’ adolescents. Dental Press J. Orthod. 2015, 20, 75–81. [Google Scholar] [CrossRef] [PubMed]
- Borzabadi-Farahani, A.; Eslamipour, F.; Shahmoradi, M. Functional needs of subjects with dentofacial deformities: A study using the index of orthognathic functional treatment need (IOFTN). J. Plast. Reconstr. Aesthet. Surg. 2016, 69, 796–801. [Google Scholar] [CrossRef]
- Eslamian, L.; Borzabadi-Farahani, A.; Badiee, M.R.; Le, B.T. An Objective Assessment of Orthognathic Surgery Patients. J. Craniofac. Surg. 2019, 30, 2479–2482. [Google Scholar] [CrossRef]
- Olkun, H.K.; Borzabadi-Farahani, A.; Uckan, S. Orthognathic Surgery Treatment Need in a Turkish Adult Population: A Retrospective Study. Int. J. Environ. Res. Public Health 2019, 16, 1881. [Google Scholar] [CrossRef] [Green Version]
- Bailey, L.J.; Haltiwanger, L.H.; Blakey, G.H.; Proffit, W.R. Who seeks surgical-orthodontic treatment: A current review. Int. J. Adult Orthodon. Orthognath. Surg. 2001, 16, 280–292. [Google Scholar] [PubMed]
- Ireland, A.J.; Atack, N.E.; Cunningham, S.J.; House, K.; Cobourne, M.T.; Hunt, N.P.; Sherriff, M.; Sandy, J.R. National British Orthodontic Society (BOS) Orthognathic Audit 2017–2018. J. Orthod. 2019, 46, 287–296. [Google Scholar] [CrossRef] [PubMed]
- Hong, S.X.; Yi, C.K. A classification and characterization of skeletal class III malocclusion on etio-pathogenic basis. Int. J. Oral Maxillofac. Surg. 2001, 30, 264–271. [Google Scholar] [CrossRef] [PubMed]
- Abu Alhaija, E.S.; Richardson, A. Growth prediction in Class III patients using cluster and discriminant function analysis. Eur. J. Orthod. 2003, 25, 599–608. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, S.; Xu, T.M.; Lin, J.X. Analysis of treatment templates of Angle’s Class III malocclusion patients. Hua Xi Kou Qiang Yi Xue Za Zhi 2009, 27, 637–641. [Google Scholar] [PubMed]
- Moreno Uribe, L.M.; Vela, K.C.; Kummet, C.; Dawson, D.V.; Southard, T.E. Phenotypic diversity in white adults with moderate to severe Class III malocclusion. Am. J. Orthod. Dentofac. Orthop. 2013, 144, 32–42. [Google Scholar] [CrossRef] [Green Version]
- Auconi, P.; Scazzocchio, M.; Cozza, P.; McNamara, J.A., Jr.; Franchi, L. Prediction of Class III treatment outcomes through orthodontic data mining. Eur. J. Orthod. 2015, 37, 257–267. [Google Scholar] [CrossRef]
- de Frutos-Valle, L.; Martin, C.; Alarcon, J.A.; Palma-Fernandez, J.C.; Iglesias-Linares, A. Subclustering in Skeletal Class III Phenotypes of Different Ethnic Origins: A Systematic Review. J. Evid. Based Dent. Pract. 2019, 19, 34–52. [Google Scholar] [CrossRef]
- Amini, F.; Mashayekhi, Z.; Rahimi, H.; Morad, G. Craniofacial morphologic parameters in a Persian population: An anthropometric study. J. Craniofac. Surg. 2014, 25, 1874–1881. [Google Scholar] [CrossRef]
- Zacharopoulos, G.V.; Manios, A.; Kau, C.H.; Velagrakis, G.; Tzanakakis, G.N.; de Bree, E. Anthropometric Analysis of the Face. J. Craniofac. Surg. 2016, 27, 71–75. [Google Scholar] [CrossRef]
- Celebi, A.A.; Kau, C.H.; Femiano, F.; Bucci, L.; Perillo, L. A Three-Dimensional Anthropometric Evaluation of Facial Morphology. J. Craniofac. Surg. 2018, 29, 304–308. [Google Scholar] [CrossRef] [PubMed]
- Lipiec, K.; Ryniewicz, W.I.; Groch, M.; Wieczorek, A.; Loster, J.E. The Evaluation of Anthropometric Measurements of Young Polish Women’s Faces. J. Craniofac. Surg. 2019, 30, 709–712. [Google Scholar] [CrossRef] [PubMed]
- de Frutos-Valle, L.; Martin, C.; Alarcon, J.A.; Palma-Fernandez, J.C.; Ortega, R.; Iglesias-Linares, A. Sub-clustering in skeletal Class III malocclusion phenotypes via principal component analysis in a Southern European population. Sci. Rep. 2020. submitted. [Google Scholar]
- Li, C.; Cai, Y.; Chen, S.; Chen, F. Classification and characterization of class III malocclusion in Chinese individuals. Head Face Med. 2016, 12, 31. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Baccetti, T.; Franchi, L.; McNamara, J.A., Jr. An improved version of the cervical vertebral maturation (CVM) method for the assessment of mandibular growth. Angle Orthod. 2002, 72, 316–323. [Google Scholar]
- World Medical Association Declaration of Helsinki: Ethical principles for medical research involving human subjects. JAMA 2013, 310, 2191–2194. [CrossRef] [PubMed] [Green Version]
- Doka, G. The ‘Excel 3D Scatter Plot’ v2.1—The Manual. 2006–2013. Available online: https://www.doka.ch/Excel3Dscatterplot.htm (accessed on 19 January 2020).
- Shrout, P.E.; Fleiss, J.L. Intraclass correlations: Uses in assessing rater reliability. Psychol. Bull. 1979, 86, 420–428. [Google Scholar] [CrossRef]
- Galvão, M.C.d.S.; Sato, J.R.; Coelho, E.C. Dahlberg formula: A novel approach for its evaluation. Dental Press J. Orthod. 2012, 17, 115–124. [Google Scholar] [CrossRef] [Green Version]
- Auconi, P.; Scazzocchio, M.; Defraia, E.; McNamara, J.A.; Franchi, L. Forecasting craniofacial growth in individuals with class III malocclusion by computational modelling. Eur. J. Orthod. 2014, 36, 207–216. [Google Scholar] [CrossRef]
- Bui, C.; King, T.; Proffit, W.; Frazier-Bowers, S. Phenotypic characterization of Class III patients. Angle Orthod. 2006, 76, 564–569. [Google Scholar]
- Ellis, E., III; McNamara, J.A., Jr. Components of adult Class III malocclusion. J. Oral Maxillofac. Surg. 1984, 42, 295–305. [Google Scholar] [CrossRef]
- Alkadhi, R.M.; Finkelman, M.D.; Trotman, C.A.; Kanavakis, G. The role of lip thickness in upper lip response to sagittal change of incisor position. Orthod. Craniofac. Res. 2019, 22, 53–57. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ali, B.; Shaikh, A.; Fida, M. Factors affecting treatment decisions for Class I malocclusions. Am. J. Orthod. Dentofacial Orthop. 2018, 154, 234–237. [Google Scholar] [CrossRef] [PubMed]
- Zednikova Mala, P.; Krajicek, V.; Veleminska, J. How tight is the relationship between the skeletal and soft-tissue facial profile: A geometric morphometric analysis of the facial outline. Forensic Sci. Int. 2018, 292, 212–223. [Google Scholar] [CrossRef]
- da Fontoura, C.S.; Miller, S.F.; Wehby, G.L.; Amendt, B.A.; Holton, N.E.; Southard, T.E.; Allareddy, V.; Moreno Uribe, L.M. Candidate Gene Analyses of Skeletal Variation in Malocclusion. J. Dent. Res. 2015, 94, 913–920. [Google Scholar] [CrossRef] [Green Version]
- Ko, J.M.; Suh, Y.J.; Hong, J.; Paeng, J.Y.; Baek, S.H.; Kim, Y.H. Segregation analysis of mandibular prognathism in Korean orthognathic surgery patients and their families. Angle Orthod. 2013, 83, 1027–1035. [Google Scholar] [CrossRef] [Green Version]

| P-A Face Height (S-Go/N-Me) (%) | Anterior Cranial Base (SN)/Length of Mand Base (Go-Pg) (%) |
| PFH:AFH (%) | Maxillary length (ANS-PNS)/Anterior Cranial Base (SN) (%) |
| S-Ar/Ar-Go (%) | Maxillary length (ANS-PNS)/Midface Length (Co-A) (%) |
| UFH (N-ANS/(N-ANS+ANS-Me)) (%) | Maxillary length (ANS-PNS)/Length of Mand Base (Go-Pg) (%) |
| LFH/TFH (ANS-Me:N-Me) (%) | Midface Length (Co-A)/Mandibular length (Co-Gn) (%) |
| Face Ht Ratio (N-ANS/ANS-Me) (%) | Mandibular Body Length (Go-Gn)/Mandibular length (Co-Gn) (%) |
| SN/GoMe (%) | Ar - A/Ar - Gn (%) |
| ANS-PNS/Me-Go (%) | Posterior Cranial Base (S-Ar)/Posterior Face Height (SGo) (%) |
| Articular Angle/SNB (%) | Ramus Height (Ar-Go)/Posterior Face Height (SGo) (%) |
| Saddle-Sella Angle (SN-Ar)/SNA (%) | Posterior Cranial Base (S-Ar)/Upper Face Height (N-ANS) (%) |
| Occ Plane to FH/FMA (MP-FH) (%) | Ramus Height (Ar-Go)/Lower Face Height (ANS-Me) (%) |
| Occ Plane to SN/SN - GoGn (%) | Maxillary Skeletal (A-N Perp)/Mand. Skeletal (Pg-Na Perp) (%) |
| Cranio-Mx Base (SN-Palatal Plane)/SN - GoGn (%) | Convexity (A-NPg)/Pg - NB (%) |
| Principal Component | PC 1 | PC 2 | PC 3 | PC 4 | PC 5 | PC 6 | PC 7 | PC 8 |
|---|---|---|---|---|---|---|---|---|
| % of explained variance (a) | 21.7 | 15.6 | 14.1 | 11.7 | 8.0 | 5.5 | 4.5 | 4.1 |
| Cumulated % of explained variance (b) | 21.7 | 37.3 | 51.4 | 63.1 | 71.1 | 76.5 | 81.8 | 85.1 |
| Cephalometric variables (c) | SN/GoMe (%) | UFH (N-ANS/(N-ANS+ANS-Me)) (%) | P-A Face Height (S-Go/N-Me) (%) | S-Ar/Ar-Go (%) | Maxillary length (ANS-PNS)/Anterior Cranial Base (SN) (%) | SNA/Saddle-Sella Angle (SN-Ar) (%) | Midface Length (Co-A)/Mandibular length (Co-Gn)(%) | SNB/Articular Angle (%) |
| ANS-PNS/Me-Go (%) | LFH/TFH (ANS-Me:N-Me) (%) | PFH:AFH (%) | Posterior Cranial Base (S-Ar)/Posterior Face Height (SGo) (%) | Maxillary length (ANS-PNS)/Midface Length (Co-A) (%) | Occ Plane to FH/FMA (MP-FH) (%) | Ar - A/Ar - Gn (%) | Pog - NB/Convexity (A-NPo) (%) | |
| Anterior Cranial Base (SN)/Length of Mand Base (Go-Pg) (%) | Face Ht Ratio (N-ANS/ANS-Me) (%) | Ramus Height (Ar-Go)/Lower Face Height (ANS-Me) (%) | Ramus Height (Ar-Go)/Posterior Face Height (SGo) (%) | Occ Plane to SN/SN – GoGn (%) | Maxillary Skeletal (A-N Perp)/Mand. Skeletal (Pg-N Perp) (%) | |||
| Maxillary length (ANS-PNS)/Length of Mand Base (Go-Pg) (%) | Cranio-Mx Base (SN-Palatal Plane)/SN – GoGn (%) | Posterior Cranial Base (S-Ar)/Upper Face Height (N-ANS) (%) | ||||||
| Mandibular Body Length (Go-Gn)/Mandibular length (Co-Gn) (%) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Frutos-Valle, L.d.; Martín, C.; Alarcón, J.A.; Palma-Fernández, J.C.; Ortega, R.; Iglesias-Linares, A. Novel Sub-Clustering of Class III Skeletal Malocclusion Phenotypes in a Southern European Population Based on Proportional Measurements. J. Clin. Med. 2020, 9, 3048. https://doi.org/10.3390/jcm9093048
Frutos-Valle Ld, Martín C, Alarcón JA, Palma-Fernández JC, Ortega R, Iglesias-Linares A. Novel Sub-Clustering of Class III Skeletal Malocclusion Phenotypes in a Southern European Population Based on Proportional Measurements. Journal of Clinical Medicine. 2020; 9(9):3048. https://doi.org/10.3390/jcm9093048
Chicago/Turabian StyleFrutos-Valle, Leixuri de, Conchita Martín, José Antonio Alarcón, Juan Carlos Palma-Fernández, Ricardo Ortega, and Alejandro Iglesias-Linares. 2020. "Novel Sub-Clustering of Class III Skeletal Malocclusion Phenotypes in a Southern European Population Based on Proportional Measurements" Journal of Clinical Medicine 9, no. 9: 3048. https://doi.org/10.3390/jcm9093048
APA StyleFrutos-Valle, L. d., Martín, C., Alarcón, J. A., Palma-Fernández, J. C., Ortega, R., & Iglesias-Linares, A. (2020). Novel Sub-Clustering of Class III Skeletal Malocclusion Phenotypes in a Southern European Population Based on Proportional Measurements. Journal of Clinical Medicine, 9(9), 3048. https://doi.org/10.3390/jcm9093048





