Inhaled Corticosteroids and COVID-19 Risk and Mortality: A Nationwide Cohort Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population and Data Sources
2.2. Use of ICS and Other Drugs for Respiratory Diseases
2.3. Data Collection and Definitions
2.4. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Zhu, N.; Zhang, D.; Wang, W.; Li, X.; Yang, B.; Song, J.; Zhao, X.; Huang, B.; Shi, W.; Lu, R.; et al. A Novel Coronavirus from Patients with Pneumonia in China, 2019. N. Engl. J. Med. 2020, 382, 727–733. [Google Scholar] [CrossRef] [PubMed]
- Feng, Y.; Ling, Y.; Bai, T.; Xie, Y.; Huang, J.; Li, J.; Xiong, W.; Yang, D.; Chen, R.; Lu, F.; et al. COVID-19 with Different Severities: A Multicenter Study of Clinical Features. Am. J. Respir. Crit. Care Med. 2020, 201, 1380–1388. [Google Scholar] [CrossRef] [PubMed]
- Global Initiative for Chronic Obstructive Lung Disease (GOLD). Global Strategy for the Diagnosis, Management, and Prevention of Chronic Obstructive Pulmonary Disease. Available online: https://www.goldcopd.org/ (accessed on 14 October 2020).
- Global Initiative for Asthma. Global Strategy for Asthma Management and Prevention. Available online: https://www.ginasthma.org (accessed on 14 October 2020).
- Stockman, L.J.; Bellamy, R.; Garner, P. SARS: Systematic review of treatment effects. PLoS Med. 2006, 3, e343. [Google Scholar] [CrossRef] [PubMed]
- Yamaya, M.; Nishimura, H.; Deng, X.; Sugawara, M.; Watanabe, O.; Nomura, K.; Shimotai, Y.; Momma, H.; Ichinose, M.; Kawase, T. Inhibitory effects of glycopyrronium, formoterol, and budesonide on coronavirus HCoV-229E replication and cytokine production by primary cultures of human nasal and tracheal epithelial cells. Respir. Investig. 2020, 58, 155–168. [Google Scholar] [CrossRef]
- Iwabuchi, K.; Yoshie, K.; Kurakami, Y.; Takahashi, K.; Kato, Y.; Morishima, T. Therapeutic potential of ciclesonide inhalation for COVID-19 pneumonia: Report of three cases. J. Infect. Chemother. 2020, 26, 625–632. [Google Scholar] [CrossRef]
- Chhiba, K.D.; Patel, G.B.; Vu, T.H.T.; Chen, M.M.; Guo, A.; Kudlaty, E.; Mai, Q.; Yeh, C.; Muhammad, L.N.; Harris, K.E.; et al. Prevalence and characterization of asthma in hospitalized and non-hospitalized patients with COVID-19. J. Allergy Clin. Immunol. 2020, 146, 307–314. [Google Scholar] [CrossRef]
- Wang, L.; Foer, D.; Bates, D.W.; Boyce, J.A.; Zhou, L. Risk factors for hospitalization, intensive care, and mortality among patients with asthma and COVID-19. J. Allergy Clin. Immunol. 2020, 146, 808–812. [Google Scholar] [CrossRef]
- Ministry of Health and Welfare; Health Insurance Review and Assessment Service. #opendata4covid19. Available online: https://hira-covid19.net/ (accessed on 14 October 2020).
- Jung, S.Y.; Choi, J.C.; You, S.H.; Kim, W.Y. Association of renin-angiotensin-aldosterone system inhibitors with COVID-19-related outcomes in Korea: A nationwide population-based cohort study. Clin. Infect. Dis 2020, in press. [Google Scholar]
- Corman, V.M.; Landt, O.; Kaiser, M.; Molenkamp, R.; Meijer, A.; Chu, D.K.; Bleicker, T.; Brünink, S.; Schneider, J.; Schmidt, M.L.; et al. Detection of 2019 novel coronavirus (2019-nCoV) by real-time RT-PCR. Euro Surveill. 2020, 25, 2000045. [Google Scholar] [CrossRef]
- Bateman, E.D.; Hurd, S.S.; Barnes, P.J.; Bousquet, J.; Drazen, J.M.; FitzGerald, J.M.; Gibson, P.; Ohta, K.; O’Byrne, P.; Pedersen, S.E.; et al. Global strategy for asthma management and prevention: GINA executive summary. Eur. Respir. J. 2008, 31, 143–178. [Google Scholar] [CrossRef] [PubMed]
- Charlson, M.E.; Pompei, P.; Ales, K.L.; MacKenzie, C.R. A new method of classifying prognostic comorbidity in longitudinal studies: Development and validation. J. Chronic Dis. 1987, 40, 373–383. [Google Scholar] [CrossRef]
- Yang, X.; Yu, Y.; Xu, J.; Shu, H.; Xia, J.; Liu, H.; Wu, Y.; Zhang, L.; Yu, Z.; Fang, M.; et al. Clinical course and outcomes of critically ill patients with SARS-CoV-2 pneumonia in Wuhan, China: A single-centered, retrospective, observational study. Lancet Respir. Med. 2020, 8, 475–481. [Google Scholar] [CrossRef]
- Guan, W.J.; Ni, Z.Y.; Hu, Y.; Liang, W.H.; Ou, C.Q.; He, J.X.; Liu, L.; Shan, H.; Lei, C.L.; Hui, D.S.C.; et al. Clinical Characteristics of Coronavirus Disease 2019 in China. N. Engl. J. Med. 2020, 382, 1708–1720. [Google Scholar] [CrossRef]
- Halpin, D.M.G.; Faner, R.; Sibila, O.; Badia, J.R.; Agusti, A. Do chronic respiratory diseases or their treatment affect the risk of SARS-CoV-2 infection? Lancet Respir. Med. 2020, 8, 436–438. [Google Scholar] [CrossRef]
- Liu, S.; Zhi, Y.; Ying, S. COVID-19 and Asthma: Reflection during the Pandemic. Clin. Rev. Allergy Immunol. 2020, 59, 78–88. [Google Scholar] [CrossRef]
- Contoli, M.; Message, S.D.; Laza-Stanca, V.; Edwards, M.R.; Wark, P.A.; Bartlett, N.W.; Kebadze, T.; Mallia, P.; Stanciu, L.A.; Parker, H.L.; et al. Role of deficient type III interferon-lambda production in asthma exacerbations. Nat. Med. 2006, 12, 1023–1026. [Google Scholar] [CrossRef] [PubMed]
- Rosenberg, H.F.; Dyer, K.D.; Domachowske, J.B. Respiratory viruses and eosinophils: Exploring the connections. Antiviral Res. 2009, 83, 1–9. [Google Scholar] [CrossRef]
- te Velde, A.A.; Huijbens, R.J.; Heije, K.; de Vries, J.E.; Figdor, C.G. Interleukin-4 (IL-4) inhibits secretion of IL-1 beta, tumor necrosis factor alpha, and IL-6 by human monocytes. Blood 1990, 76, 1392–1397. [Google Scholar] [CrossRef]
- de Vries, J.E. The role of IL-13 and its receptor in allergy and inflammatory responses. J. Allergy Clin. Immunol. 1998, 102, 165–169. [Google Scholar] [CrossRef]
- Pilette, C.; Ouadrhiri, Y.; Van Snick, J.; Renauld, J.C.; Staquet, P.; Vaerman, J.P.; Sibille, Y. IL-9 inhibits oxidative burst and TNF-alpha release in lipopolysaccharide-stimulated human monocytes through TGF-beta. J. Immunol. 2002, 168, 4103–4111. [Google Scholar] [CrossRef]
- Peters, M.C.; Sajuthi, S.; Deford, P.; Christenson, S.; Rios, C.L.; Montgomery, M.T.; Woodruff, P.G.; Mauger, D.T.; Erzurum, S.C.; Johansson, M.W.; et al. COVID-19-related Genes in Sputum Cells in Asthma. Relationship to Demographic Features and Corticosteroids. Am. J. Respir. Crit. Care Med. 2020, 202, 83–90. [Google Scholar] [CrossRef] [PubMed]
- Yoo, K.H.; Kim, Y.S.; Sheen, S.S.; Park, J.H.; Hwang, Y.I.; Kim, S.H.; Yoon, H.I.; Lim, S.C.; Park, J.Y.; Park, S.J.; et al. Prevalence of chronic obstructive pulmonary disease in Korea: The fourth Korean National Health and Nutrition Examination Survey, 2008. Respirology 2011, 16, 659–665. [Google Scholar] [CrossRef]
- Masoli, M.; Fabian, D.; Holt, S.; Beasley, R.; Global Initiative for Asthma (GINA) Program. The global burden of asthma: Executive summary of the GINA Dissemination Committee report. Allergy 2004, 59, 469–478. [Google Scholar] [CrossRef] [PubMed]
- Williamson, E.J.; Walker, A.J.; Bhaskaran, K.; Bacon, S.; Bates, C.; Morton, C.E.; Curtis, H.J.; Mehrkar, A.; Evans, D.; Inglesby, P.; et al. Factors associated with COVID-19-related death using OpenSAFELY. Nature 2020, 584, 430–436. [Google Scholar] [CrossRef] [PubMed]
- Maes, T.; Bracke, K.; Brusselle, G.G. Reply to Lipworth et al.: Inhaled Corticosteroids and COVID-19. Am. J. Respir. Crit. Care Med. 2020, 202, 900–902. [Google Scholar] [CrossRef] [PubMed]
- Yang, I.A.; Clarke, M.S.; Sim, E.H.; Fong, K.M. Inhaled corticosteroids for stable chronic obstructive pulmonary disease. Cochrane Database Syst. Rev. 2012, CD002991. [Google Scholar] [CrossRef]
- McKeever, T.; Harrison, T.W.; Hubbard, R.; Shaw, D. Inhaled corticosteroids and the risk of pneumonia in people with asthma: A case-control study. Chest 2013, 144, 1788–1794. [Google Scholar] [CrossRef]
- Horby, P.; Lim, W.S.; Emberson, J.R.; Mafham, M.; Bell, J.L.; Linsell, L.; Staplin, N.; Brightling, C.; Ustianowski, A.; Elmahi, E.; et al. Dexamethasone in Hospitalized Patients with Covid-19—Preliminary Report. N. Engl. J. Med. 2020, in press. [Google Scholar]
- World Health Organization. Clinical management of COVID-19. Interim Guidance. Available online: https://www.who.int/publications-detail/clinical-management-of-severe-acute-respiratory-infection-when-novel-coronavirus-(ncov)-infection-is-suspected (accessed on 14 October 2020).
- Schultze, A.; Walker, A.J.; MacKenna, B.; Morton, C.E.; Bhaskaran, K.; Brown, J.P.; Rentsch, C.T.; Williamson, E.; Drysdale, H.; Croker, R.; et al. Risk of COVID-19-related death among patients with chronic obstructive pulmonary disease or asthma prescribed inhaled corticosteroids: An observational cohort study using the OpenSAFELY platform. Lancet Respir. Med. 2020, in press. [Google Scholar] [CrossRef]
- Choi, Y.J.; Park, J.Y.; Lee, H.S.; Suh, J.; Song, J.Y.; Byun, M.K.; Cho, J.H.; Kim, H.J.; Lee, J.H.; Park, J.W.; et al. Effect of Asthma and Asthma Medication on the Prognosis of Patients with COVID-19. Eur. Respir. J. 2020, in press. [Google Scholar]
| All (n = 7341) | ICS Status | p | ||
|---|---|---|---|---|
| Users (n = 114) | Nonusers (n = 7227) | |||
| Age, years | 47.1 (19.0) | 57.4 (19.3) | 46.9 (19.0) | <0.001 |
| Male sex | 2970 (40) | 48 (42) | 2922 (40) | 0.72 |
| Date of COVID-19 diagnosis | 0.13 | |||
| March 1 and earlier | 2349 (32) | 44 (39) | 2305 (32) | |
| March 2 and later | 4992 (68) | 70 (61) | 4922 (68) | |
| Comorbidities | ||||
| Diabetes | 1299 (18) | 38 (33) | 1261 (17) | <0.001 |
| Hypertension | 1782 (24) | 46 (40) | 1736 (24) | <0.001 |
| Myocardial infarction | 71 (1) | 3 (3) | 68 (1) | 0.10 |
| Congestive heart failure | 311 (4) | 21 (18) | 290 (4) | <0.001 |
| Cerebrovascular disease | 514 (7) | 18 (16) | 496 (7) | <0.001 |
| COPD | 678 (9) | 52 (46) | 626 (9) | <0.001 |
| Asthma | 123 (2) | 61 (54) | 62 (1) | <0.001 |
| Chronic liver disease | 1474 (20) | 44 (39) | 1430 (20) | <0.001 |
| Chronic kidney disease | 142 (2) | 4 (4) | 138 (2) | 0.28 |
| Malignancy | 329 (4) | 8 (7) | 321 (4) | 0.19 |
| Charlson Comorbidity Index | 1.4 (1.9) | 3.3 (2.5) | 1.3 (1.9) | <0.001 |
| Other drugs for respiratory diseases | ||||
| LABA | 121 (2) | 110 (96) | 11 (0.2) | <0.001 |
| SABA | 37 (1) | 21 (18) | 16 (0.2) | <0.001 |
| LAMA | 32 (0.4) | 18 (16) | 14 (0.2) | <0.001 |
| Methylxanthine | 110 (1) | 37 (32) | 73 (1) | <0.001 |
| LTRA | 149 (2) | 52 (46) | 97 (1) | <0.001 |
| ICS use | ||||
| Cumulative dose, median (IQR), μg | 15,000 (7500–45,000) | NA | ||
| Total days of use, median (IQR) | 90 (30–150) | NA | ||
| OCS use | 93 (1) | 9 (8) | 84 (1) | |
| Cumulative dose, median (IQR), mg | 773 (605–1645) | 980 (555–2180) | 707 (605–1578) | 0.41 |
| Total days of use, median (IQR) | 167 (121–373) | 196 (111–332) | 153 (121–376) | 0.66 |
| Healthcare usage | ||||
| Emergency room visit | 24 (0.3) | 8 (7) | 16 (0.2) | <0.001 |
| Hospitalization | 54 (1) | 15 (13) | 39 (1) | <0.001 |
| No. of Patients | No. of Events | Unadjusted OR (95% CI) | p | Adjusted OR 1 (95% CI) | p | |
|---|---|---|---|---|---|---|
| Mortality | 7341 | 227 | ||||
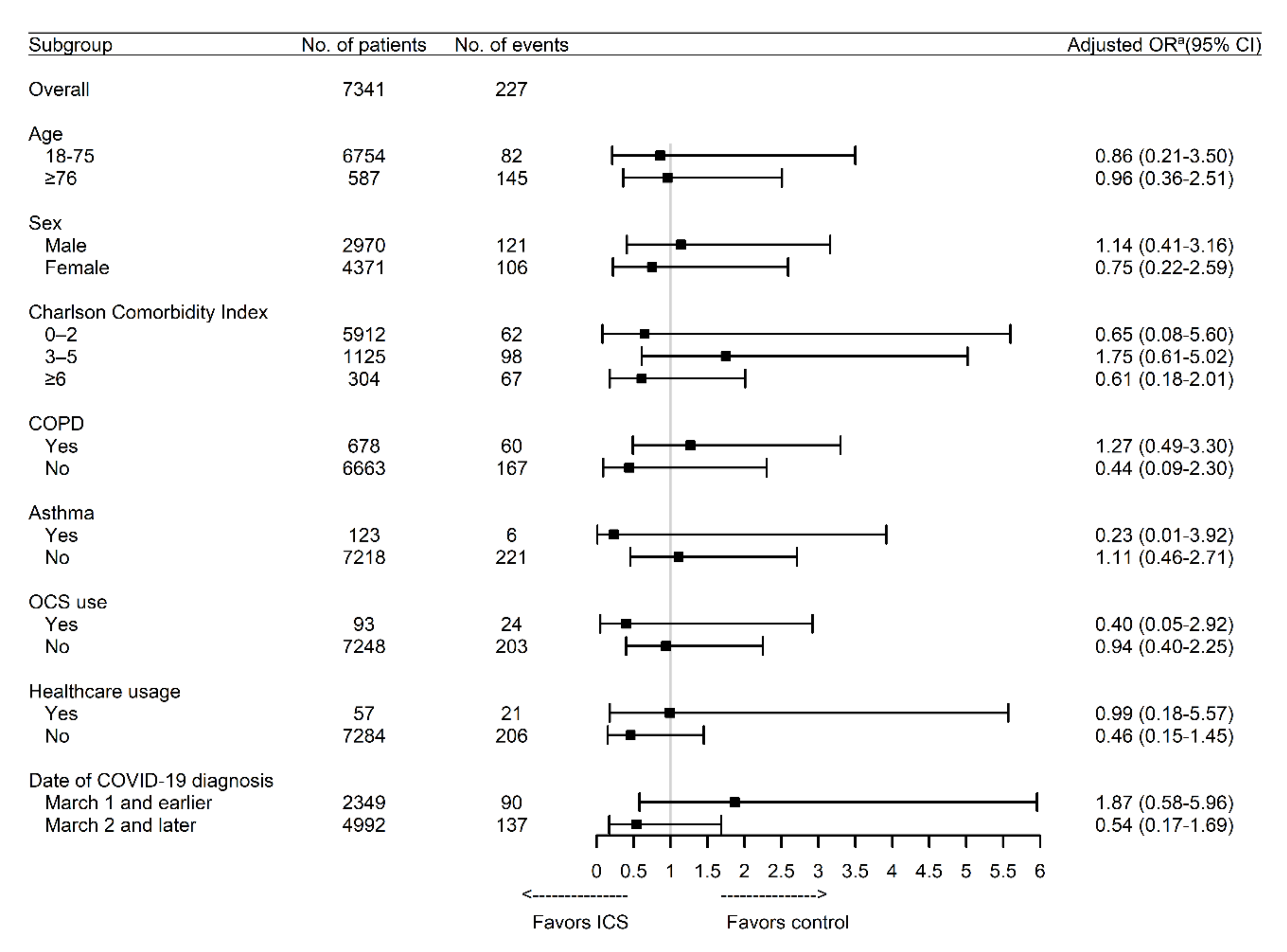
| ICS | 114 | 10 | 3.11 (1.60–6.03) | <0.001 | 0.94 (0.43–2.07) | 0.88 |
| LABA | 121 | 11 | 3.24 (1.72–6.12) | <0.001 | 0.85 (0.40–1.82) | 0.67 |
| SABA | 37 | 3 | 2.79 (0.85–9.15) | 0.09 | 0.87 (0.20–3.73) | 0.85 |
| LAMA | 32 | 4 | 4.54 (1.58–13.05) | 0.01 | 0.69 (0.20–2.32) | 0.54 |
| Methylxanthine | 110 | 20 | 7.54 (4.56–12.48) | <0.001 | 1.45 (0.78–2.69) | 0.24 |
| LTRA | 149 | 15 | 3.69 (2.13–6.39) | <0.001 | 1.36 (0.70–2.64) | 0.37 |
| OCS use | 93 | 24 | 12.07 (7.43–19.61) | <0.001 | 3.42 (1.89–6.17) | <0.001 |
| ICS cumulative dose | ||||||
| <15,000 μg | 60 | 4 | 2.31 (0.83–6.42) | 0.81 | 1.17 (0.34–3.98) | 0.71 |
| ≥15,000 μg | 54 | 6 | 4.04 (1.71–9.54) | 0.053 | 0.83 (0.31–2.24) | 0.65 |
| COPD | 678 | 60 | 3.78 (2.78–5.13) | <0.001 | 0.95 (0.65–1.39) | 0.78 |
| Asthma | 123 | 6 | 1.62 (0.71–3.73) | 0.25 | 0.92 (0.35–2.42) | 0.86 |
| Respiratory outcomes 2 | 7341 | 934 | ||||
| ICS | 114 | 34 | 2.99 (1.99–4.49) | <0.001 | 1.35 (0.80–2.26) | 0.26 |
| LABA | 121 | 36 | 2.98 (2.01–4.43) | <0.001 | 1.20 (0.72–1.99) | 0.48 |
| SABA | 37 | 11 | 2.93 (1.44–5.94) | 0.003 | 1.72 (0.65–4.55) | 0.28 |
| LAMA | 32 | 14 | 5.40 (2.68–10.90) | <0.001 | 1.14 (0.49–2.64) | 0.76 |
| Methylxanthine | 110 | 49 | 5.76 (3.93–8.44) | <0.001 | 1.81 (1.13–2.92) | 0.01 |
| LTRA | 149 | 44 | 2.97 (2.07–4.25) | <0.001 | 1.58 (1.004–2.48) | 0.048 |
| OCS use | 93 | 38 | 4.90 (3.22–7.45) | <0.001 | 1.52 (0.90–2.54) | 0.11 |
| ICS cumulative dose | ||||||
| <15,000 μg | 60 | 16 | 2.56 (1.44–4.55) | 0.34 | 2.02 (0.97–4.22) | 0.08 |
| ≥15,000 μg | 54 | 18 | 3.52 (1.99–6.22) | 0.02 | 0.96 (0.47–1.93) | 0.32 |
| COPD | 678 | 188 | 3.04 (2.53–3.66) | <0.001 | 1.31 (1.03–1.67) | 0.03 |
| Asthma | 123 | 25 | 1.77 (1.14–2.76) | 0.01 | 1.20 (0.69–2.10) | 0.52 |
| All (n = 3200) | COVID-19 (n = 640) | Non-COVID-19 (n = 2560) | p | |
|---|---|---|---|---|
| Age, years | 58.4 (19.7) | 58.4 (19.7) | 58.4 (19.7) | MV |
| Male sex | 1370 (43) | 274 (43) | 1096 (43) | MV |
| Date of COVID-19 diagnosis | ||||
| March 1 and earlier | 944 (30) | 198 (31) | NA | |
| March 2 and later | 2256 (71) | 442 (69) | NA | |
| Comorbidities | ||||
| Diabetes | 1120 (35) | 213 (33) | 907 (35) | 0.31 |
| Hypertension | 1477 (46) | 283 (44) | 1194 (47) | 0.27 |
| Myocardial infarction | 108 (3) | 13 (2) | 95 (4) | 0.04 |
| Congestive heart failure | 506 (16) | 86 (13) | 420 (16) | 0.07 |
| Cerebrovascular disease | 666 (21) | 111 (17) | 555 (22) | 0.02 |
| Chronic liver disease | 1279 (40) | 222 (35) | 1057 (41) | 0.002 |
| Chronic kidney disease | 272 (9) | 27 (4) | 245 (10) | <0.001 |
| Malignancy | 527 (16) | 66 (10) | 461 (18) | <0.001 |
| Charlson Comorbidity Index | 3.8 (3.1) | 3.3 (2.4) | 4.1 (3.1) | <0.001 |
| Other drugs for respiratory diseases | ||||
| ICS | 286 (9) | 50 (8) | 236 (9) | 0.26 |
| LABA | 368 (12) | 61 (10) | 307 (12) | 0.08 |
| SABA | 138 (4) | 15 (2) | 123 (5) | 0.01 |
| LAMA | 206 (6) | 31 (5) | 175 (7) | 0.07 |
| Methylxanthine | 444 (14) | 67 (10) | 377 (15) | 0.01 |
| LTRA | 289 (9) | 55 (9) | 234 (9) | 0.67 |
| PDE4 inhibitor | 2 (0.1) | 0 | 2 (0.1) | >0.99 |
| ICS use | ||||
| Cumulative dose, median (IQR), μg | 30,000 (12,000–69,000) | 19,500 (9000–48,000) | 33,000 (12,000–73,500) | 0.04 |
| Total days of use, median (IQR) | 120 (30–210) | 120 (30–210) | 120 (30–210) | 0.22 |
| OCS use | 176 (6) | 22 (3) | 154 (6) | |
| Cumulative dose, median (IQR), mg | 1183 (613–1835) | 689 (605–1682) | 1200 (680–1855) | 0.27 |
| Total days of use, median (IQR) | 251 (125–393) | 185 (121–398) | 255 (141–392) | 0.26 |
| Healthcare usage | ||||
| Emergency room visit | 141 (4) | 20 (3) | 121 (5) | 0.08 |
| Hospitalization | 284 (9) | 48 (8) | 236 (9) | 0.17 |
| All (n = 450) | COVID-19 (n = 90) | Non-COVID-19 (n = 360) | p | |
|---|---|---|---|---|
| Age, years | 52.8 (17.3) | 52.7 (17.5) | 52.9 (17.3) | MV |
| Male sex | 170 (38) | 34 (38) | 136 (38) | MV |
| Date of COVID-19 diagnosis | ||||
| March 1 and earlier | 120 (27) | 23 (26) | NA | |
| March 2 and later | 330 (73) | 67 (74) | NA | |
| Comorbidities | ||||
| Diabetes | 117 (26) | 25 (28) | 92 (26) | 0.67 |
| Hypertension | 158 (35) | 28 (31) | 130 (36) | 0.37 |
| Myocardial infarction | 8 (2) | 1 (1) | 7 (2) | >0.99 |
| Congestive heart failure | 43 (10) | 6 (7) | 37 (10) | 0.30 |
| Cerebrovascular disease | 58 (13) | 6 (7) | 52 (14) | 0.049 |
| Chronic liver disease | 143 (32) | 32 (36) | 111 (31) | 0.39 |
| Chronic kidney disease | 29 (6) | 3 (3) | 26 (7) | 0.18 |
| Malignancy | 49 (11) | 8 (9) | 41 (11) | 0.50 |
| Charlson Comorbidity Index | 3.0 (2.4) | 2.7 (1.9) | 3.1 (2.5) | 0.07 |
| Other drugs for respiratory diseases | ||||
| ICS | 219 (49) | 43 (48) | 176 (49) | 0.85 |
| LABA | 205 (46) | 41 (46) | 164 (46) | >0.99 |
| SABA | 87 (19) | 15 (17) | 72 (20) | 0.47 |
| LAMA | 1 (0.2) | 0 | 1 (0.3) | >0.99 |
| Methylxanthine | 101 (22) | 21 (23) | 80 (22) | 0.82 |
| LTRA | 197 (44) | 40 (44) | 157 (44) | 0.89 |
| ICS use | ||||
| Cumulative dose, median (IQR), μg | 18,000 (12,000–45,000) | 18,000 (12,000–45,000) | 18,000 (12,000–45,000) | 0.54 |
| Total days of use, median (IQR) | 60 (30–150) | 90 (30–180) | 60 (30–120) | 0.89 |
| OCS use | 51 (11) | 9 (10) | 42 (12) | |
| Cumulative dose, median (IQR), mg | 820 (570–1671) | 980 (555–1375) | 815 (600–1671) | 0.66 |
| Total days of use, median (IQR) | 162 (114–318) | 196 (111–275) | 159 (118–318) | 0.67 |
| Healthcare usage | ||||
| Emergency room visit | 13 (3) | 1 (1) | 12 (3) | 0.48 |
| Hospitalization | 25 (6) | 1 (1) | 24 (7) | 0.04 |
| No. of Patients | No. of Events | Unadjusted OR (95% CI) | p | Adjusted OR 1 (95% CI) | p | |
|---|---|---|---|---|---|---|
| COPD | 3200 | 640 | ||||
| ICS | 286 | 50 | 0.84 (0.61–1.15) | 0.27 | 1.02 (0.46–2.25) | 0.97 |
| LABA | 368 | 61 | 0.77 (0.58–1.03) | 0.08 | 0.94 (0.45–1.98) | 0.87 |
| SABA | 138 | 15 | 0.48 (0.28–0.82) | 0.01 | 0.52 (0.13–2.17) | 0.37 |
| LAMA | 206 | 31 | 0.69 (0.47–1.03) | 0.07 | 0.47 (0.11–1.94) | 0.30 |
| Methylxanthine | 444 | 67 | 0.68 (0.51–0.89) | 0.01 | 0.72 (0.33–1.58) | 0.41 |
| LTRA | 289 | 55 | 0.94 (0.69–1.27) | 0.67 | 1.20 (0.57–2.53) | 0.64 |
| OCS use | 176 | 22 | 0.56 (0.35–0.88) | 0.01 | 0.89 (0.28–2.91) | 0.85 |
| ICS cumulative dose | ||||||
| <15,000 μg | 115 | 24 | 1.04 (0.66–1.64) | 0.40 | 2.24 (0.94–5.37) | 0.02 |
| ≥15,000 μg | 171 | 26 | 0.71 (0.46–1.08) | 0.13 | 0.24 (0.03–1.73) | 0.07 |
| Asthma | 450 | 90 | ||||
| ICS | 219 | 43 | 0.96 (0.60–1.52) | 0.85 | 0.38 (0.13–1.17) | 0.09 |
| LABA | 205 | 41 | 1.00 (0.63–1.59) | >0.99 | 0.48 (0.16–1.44) | 0.19 |
| SABA | 87 | 15 | 0.80 (0.43–1.48) | 0.47 | 0.85 (0.23–3.12) | 0.80 |
| LAMA | 1 | 0 | 0.99 | 0.99 | ||
| Methylxanthine | 101 | 21 | 1.07 (0.62–1.84) | 0.82 | 0.79 (0.17–3.72) | 0.77 |
| LTRA | 197 | 40 | 1.03 (0.65–1.65) | 0.89 | 1.42 (0.50–4.04) | 0.51 |
| OCS use | 51 | 9 | 0.84 (0.39–1.80) | 0.66 | 0.87 (0.11–7.24) | 0.90 |
| ICS cumulative dose | ||||||
| <15,000 μg | 107 | 20 | 0.90 (0.50–1.61) | 0.70 | 0.16 (0.02–1.26) | 0.13 |
| ≥15,000 μg | 112 | 23 | 1.01 (0.58–1.77) | 0.82 | 0.60 (0.18–2.03) | 0.57 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Choi, J.C.; Jung, S.-Y.; Yoon, U.A.; You, S.-H.; Kim, M.-S.; Baek, M.S.; Jung, J.-W.; Kim, W.-Y. Inhaled Corticosteroids and COVID-19 Risk and Mortality: A Nationwide Cohort Study. J. Clin. Med. 2020, 9, 3406. https://doi.org/10.3390/jcm9113406
Choi JC, Jung S-Y, Yoon UA, You S-H, Kim M-S, Baek MS, Jung J-W, Kim W-Y. Inhaled Corticosteroids and COVID-19 Risk and Mortality: A Nationwide Cohort Study. Journal of Clinical Medicine. 2020; 9(11):3406. https://doi.org/10.3390/jcm9113406
Chicago/Turabian StyleChoi, Jae Chol, Sun-Young Jung, Una A. Yoon, Seung-Hun You, Myo-Song Kim, Moon Seong Baek, Jae-Woo Jung, and Won-Young Kim. 2020. "Inhaled Corticosteroids and COVID-19 Risk and Mortality: A Nationwide Cohort Study" Journal of Clinical Medicine 9, no. 11: 3406. https://doi.org/10.3390/jcm9113406
APA StyleChoi, J. C., Jung, S.-Y., Yoon, U. A., You, S.-H., Kim, M.-S., Baek, M. S., Jung, J.-W., & Kim, W.-Y. (2020). Inhaled Corticosteroids and COVID-19 Risk and Mortality: A Nationwide Cohort Study. Journal of Clinical Medicine, 9(11), 3406. https://doi.org/10.3390/jcm9113406





