Clinical Application of Virtual Reality for Upper Limb Motor Rehabilitation in Stroke: Review of Technologies and Clinical Evidence
Abstract
1. Introduction
2. Technologies Used in VR Rehabilitation
2.1. Definition of VR
2.2. Non-Immersive and Immersive VR
2.3. Technologies for Motion Tracking and Feedback for Virtual Rehabilitation
2.4. Examples of Commercialized VR Upper Limb Rehabilitation Systems
3. Clinical Evidence and Considerations for VR in Motor Rehabilitation after Stroke
3.1. Literature Search
3.2. Clinical Evidence
4. Considerations for VR Application in Stroke Rehabilitation
4.1. HMDs and Motion Sickness
4.2. Differences in Movements in VR
4.3. Transfer of Learning in VR to the Real World
4.4. Gamification
4.5. Barriers
5. Combinational Approaches with VR in Stroke Rehabilitation
6. Summary
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Collaborators, G.S. Global, regional, and national burden of stroke, 1990–2016: A systematic analysis for the Global Burden of Disease Study 2016. Lancet Neurol. 2019, 18, 439–458. [Google Scholar]
- Gorelick, P.B. The future of stroke prevention by risk factor modification. Handb. Clin. Neurol. 2009, 94, 1261–1276. [Google Scholar] [PubMed]
- Ovbiagele, B.; Goldstein, L.B.; Higashida, R.T.; Howard, V.J.; Johnston, S.C.; Khavjou, O.A.; Lackland, D.T.; Lichtman, J.H.; Mohl, S.; Sacco, R.L.; et al. Forecasting the future of stroke in the United States: A policy statement from the American Heart Association and American Stroke Association. Stroke 2013, 44, 2361–2375. [Google Scholar] [CrossRef] [PubMed]
- Lawrence, E.S.; Coshall, C.; Dundas, R.; Stewart, J.; Rudd, A.G.; Howard, R.; Wolfe, C.D. Estimates of the prevalence of acute stroke impairments and disability in a multiethnic population. Stroke 2001, 32, 1279–1284. [Google Scholar] [CrossRef]
- Faria-Fortini, I.; Michaelsen, S.M.; Cassiano, J.G.; Teixeira-Salmela, L.F. Upper extremity function in stroke subjects: Relationships between the international classification of functioning, disability, and health domains. J. Hand Ther. 2011, 24, 257–264; quiz 265. [Google Scholar] [CrossRef]
- Min, K.B.; Min, J.Y. Health-related quality of life is associated with stroke deficits in older adults. Age Ageing 2015, 44, 700–704. [Google Scholar] [CrossRef]
- Kwon, S.; Park, J.H.; Kim, W.S.; Han, K.; Lee, Y.; Paik, N.J. Health-related quality of life and related factors in stroke survivors: Data from Korea National Health and Nutrition Examination Survey (KNHANES) 2008 to 2014. PLoS ONE 2018, 13, e0195713. [Google Scholar] [CrossRef]
- Winstein, C.J.; Stein, J.; Arena, R.; Bates, B.; Cherney, L.R.; Cramer, S.C.; Deruyter, F.; Eng, J.J.; Fisher, B.; Harvey, R.L.; et al. Guidelines for Adult Stroke Rehabilitation and Recovery: A Guideline for Healthcare Professionals from the American Heart Association/American Stroke Association. Stroke 2016, 47, e98–e169. [Google Scholar] [CrossRef]
- Lohse, K.R.; Lang, C.E.; Boyd, L.A. Is more better? Using metadata to explore dose-response relationships in stroke rehabilitation. Stroke 2014, 45, 2053–2058. [Google Scholar] [CrossRef]
- Barreca, S.; Wolf, S.L.; Fasoli, S.; Bohannon, R. Treatment interventions for the paretic upper limb of stroke survivors: A critical review. Neurorehabil. Neural Repair 2003, 17, 220–226. [Google Scholar] [CrossRef]
- Levin, M.F.; Weiss, P.L.; Keshner, E.A. Emergence of virtual reality as a tool for upper limb rehabilitation: Incorporation of motor control and motor learning principles. Phys. Ther. 2015, 95, 415–425. [Google Scholar] [CrossRef] [PubMed]
- Kleim, J.A.; Jones, T.A. Principles of experience-dependent neural plasticity: Implications for rehabilitation after brain damage. J. Speech Lang. Hear. Res. 2008, 51, S225–S239. [Google Scholar] [CrossRef]
- Merians, A.S.; Jack, D.; Boian, R.; Tremaine, M.; Burdea, G.C.; Adamovich, S.V.; Recce, M.; Poizner, H. Virtual reality-augmented rehabilitation for patients following stroke. Phys. Ther. 2002, 82, 898–915. [Google Scholar] [CrossRef] [PubMed]
- Burke, J.W.; McNeill, M.; Charles, D.K.; Morrow, P.J.; Crosbie, J.H.; McDonough, S.M. Optimising engagement for stroke rehabilitation using serious games. Vis. Comput. 2009, 25, 1085. [Google Scholar] [CrossRef]
- Mihelj, M.; Novak, D.; Milavec, M.; Ziherl, J.; Olenšek, A.; Munih, M. Virtual rehabilitation environment using principles of intrinsic motivation and game design. Presence Teleoperators Virtual Environ. 2012, 21, 1–15. [Google Scholar] [CrossRef]
- Kim, W.S.; Cho, S.; Park, S.H.; Lee, J.Y.; Kwon, S.; Paik, N.J. A low cost kinect-based virtual rehabilitation system for inpatient rehabilitation of the upper limb in patients with subacute stroke: A randomized, double-blind, sham-controlled pilot trial. Medicine 2018, 97, e11173. [Google Scholar] [CrossRef]
- Holden, M.K.; Dyar, T.A.; Dayan-Cimadoro, L. Telerehabilitation using a virtual environment improves upper extremity function in patients with stroke. IEEE Trans. Neural Syst. Rehabil Eng. 2007, 15, 36–42. [Google Scholar] [CrossRef]
- Kim, W.S.; Cho, S.; Baek, D.; Bang, H.; Paik, N.J. Upper Extremity Functional Evaluation by Fugl-Meyer Assessment Scoring Using Depth-Sensing Camera in Hemiplegic Stroke Patients. PLoS ONE 2016, 11, e0158640. [Google Scholar] [CrossRef]
- Slater, M.; Wilbur, S. A Framework for Immersive Virtual Environments (FIVE): Speculations on the Role of Presence in Virtual Environments. Presence Teleoperators Virtual Environ. 1997, 6, 603–616. [Google Scholar] [CrossRef]
- Slater, M. Place illusion and plausibility can lead to realistic behaviour in immersive virtual environments. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2009, 364, 3549–3557. [Google Scholar] [CrossRef]
- Bailey, J.O.; Bailenson, J.N. When Does Virtual Embodiment Change Our Minds? Presence Teleoperators Virtual Environ. 2016, 25, 222–233. [Google Scholar] [CrossRef]
- Ventura, S.; Brivio, E.; Riva, G.; Banos, R.M. Immersive Versus Non-immersive Experience: Exploring the Feasibility of Memory Assessment through 360 degrees Technology. Front. Psychol. 2019, 10, 2509. [Google Scholar] [CrossRef] [PubMed]
- Czernuszenko, M.; Pape, D.; Sadin, D.; DeFanti, T.; Dawe, G.L.; Brown, M.D. The ImmersaDesk and infinity wall projection-based virtual reality displays. ACM Siggraph Comput. Graph. 1997, 31, 46–59. [Google Scholar] [CrossRef]
- Cruz-Neira, C.; Sandin, D.J.; DeFanti, T.A.; Kenyon, R.V.; Hart, J.C. The CAVE: Audio visual experience automatic virtual environment. Commun. ACM 1992, 35, 64–72. [Google Scholar] [CrossRef]
- Kober, S.E.; Kurzmann, J.; Neuper, C. Cortical correlate of spatial presence in 2D and 3D interactive virtual reality: An EEG study. Int. J. Psychophysiol. 2012, 83, 365–374. [Google Scholar] [CrossRef]
- Wirth, W.; Hartmann, T.; Böcking, S.; Vorderer, P.; Klimmt, C.; Schramm, H.; Saari, T.; Laarni, J.; Ravaja, N.; Gouveia, F.R.; et al. A Process Model of the Formation of Spatial Presence Experiences. Media Psychol. 2007, 9, 493–525. [Google Scholar] [CrossRef]
- Lloréns, R.; Noé, E.; Naranjo, V.; Borrego, A.; Latorre, J.; Alcañiz, M. Tracking systems for virtual rehabilitation: Objective performance vs. subjective experience. A practical scenario. Sensors 2015, 15, 6586–6606. [Google Scholar] [CrossRef]
- Juo, Y.-Y.; Abiri, A.; Pensa, J.; Sun, S.; Tao, A.; Bisley, J.; Grundfest, W.; Dutson, E. Center for Advanced Surgical and Interventional Technology Multimodal Haptic Feedback for Robotic Surgery. In Handbook of Robotic and Image-Guided Surgery; Abedin-Nasab, M.H., Ed.; Elsevier: Amsterdam, The Netherlands, 2020; pp. 285–301. [Google Scholar]
- Dimbwadyo-Terrer, I.; Trincado-Alonso, F.; de Los Reyes-Guzman, A.; Aznar, M.A.; Alcubilla, C.; Perez-Nombela, S.; Del Ama-Espinosa, A.; Polonio-Lopez, B.; Gil-Agudo, A. Upper limb rehabilitation after spinal cord injury: A treatment based on a data glove and an immersive virtual reality environment. Disabil. Rehabil. Assist. Technol. 2016, 11, 462–467. [Google Scholar] [CrossRef]
- Crosbie, J.H.; Lennon, S.; McGoldrick, M.C.; McNeill, M.D.; McDonough, S.M. Virtual reality in the rehabilitation of the arm after hemiplegic stroke: A randomized controlled pilot study. Clin. Rehabil. 2012, 26, 798–806. [Google Scholar] [CrossRef]
- Calabrò, R.S.; Accorinti, M.; Porcari, B.; Carioti, L.; Ciatto, L.; Billeri, L.; Andronaco, V.A.; Galletti, F.; Filoni, S.; Naro, A. Does hand robotic rehabilitation improve motor function by rebalancing interhemispheric connectivity after chronic stroke? Encouraging data from a randomised-clinical-trial. Clin. Neurophysiol. 2019, 130, 767–780. [Google Scholar] [CrossRef]
- Kiper, P.; Szczudlik, A.; Agostini, M.; Opara, J.; Nowobilski, R.; Ventura, L.; Tonin, P.; Turolla, A. Virtual Reality for Upper Limb Rehabilitation in Subacute and Chronic Stroke: A Randomized Controlled Trial. Arch. Phys. Med. Rehabil. 2018, 99, 834–842.e4. [Google Scholar] [CrossRef] [PubMed]
- Cho, S.; Kim, W.-S.; Paik, N.-J.; Bang, H. Upper-Limb Function Assessment Using VBBTs for Stroke Patients. IEEE Comput. Graph. Appl. 2016, 36, 70–78. [Google Scholar] [CrossRef] [PubMed]
- Askin, A.; Atar, E.; Kocyigit, H.; Tosun, A. Effects of Kinect-based virtual reality game training on upper extremity motor recovery in chronic stroke. Somatosens. Mot. Res. 2018, 35, 25–32. [Google Scholar] [CrossRef] [PubMed]
- Faria, A.L.; Cameirao, M.S.; Couras, J.F.; Aguiar, J.R.O.; Costa, G.M.; Bermudez, I.B.S. Combined Cognitive-Motor Rehabilitation in Virtual Reality Improves Motor Outcomes in Chronic Stroke—A Pilot Study. Front. Psychol. 2018, 9, 854. [Google Scholar] [CrossRef]
- Lee, M.M.; Lee, K.J.; Song, C.H. Game-Based Virtual Reality Canoe Paddling Training to Improve Postural Balance and Upper Extremity Function: A Preliminary Randomized Controlled Study of 30 Patients with Subacute Stroke. Med. Sci. Monit. Int. Med. J. Exp. Clin. Res. 2018, 24, 2590–2598. [Google Scholar] [CrossRef]
- Sucar, L.E.; Orihuela-Espina, F.; Velazquez, R.L.; Reinkensmeyer, D.J.; Leder, R.; Hernandez-Franco, J. Gesture therapy: An upper limb virtual reality-based motor rehabilitation platform. IEEE Trans. Neural Syst. Rehabil. Eng. 2014, 22, 634–643. [Google Scholar] [CrossRef]
- Ballester, B.R.; Nirme, J.; Camacho, I.; Duarte, E.; Rodríguez, S.; Cuxart, A.; Duff, A.; Verschure, P.F.M.J. Domiciliary VR-Based Therapy for Functional Recovery and Cortical Reorganization: Randomized Controlled Trial in Participants at the Chronic Stage Post Stroke. JMIR Serious Games 2017, 5, e15. [Google Scholar] [CrossRef]
- Sampson, M.; Shau, Y.W.; King, M.J. Bilateral upper limb trainer with virtual reality for post-stroke rehabilitation: Case series report. Disabil. Rehabil. Assist. Technol. 2012, 7, 55–62. [Google Scholar] [CrossRef]
- Xin, W.; Duan, F.; Yong, Y.; Tan, J.T.C.; Xiaomin, C. Design of A Multi-functional System Based on Virtual Reality for Stroke Rehabilitation. In Proceedings of the 11th World Congress on Intelligent Control and Automation, Shenyang, China, 29 June–4 July 2014; pp. 2412–2417. [Google Scholar]
- Feintuch, U.; Raz, L.; Hwang, J.; Josman, N.; Katz, N.; Kizony, R.; Rand, D.; Rizzo, A.S.; Shahar, M.; Yongseok, J.; et al. Integrating haptic-tactile feedback into a video-capture-based virtual environment for rehabilitation. Cyberpsychol. Behav. Impact Internetmultimedia Virtual Real. Behav. Soc. 2006, 9, 129–132. [Google Scholar] [CrossRef]
- Popescu, V.G.; Burdea, G.C.; Bouzit, M.; Hentz, V.R. A virtual-reality-based telerehabilitation system with force feedback. IEEE Trans. Inf. Technol. Biomed. A Publ. IEEE Eng. Med. Biol. Soc. 2000, 4, 45–51. [Google Scholar] [CrossRef]
- Prisco, G.M.; Avizzano, C.A.; Calcara, M.; Ciancio, S.; Pinna, S.; Bergamasco, M. A virtual environment with haptic feedback for the treatment of motor dexterity disabilities. In Proceedings of the 1998 IEEE International Conference on Robotics and Automation (Cat. No.98CH36146), Leuven, Belgium, 20 May 1998; Volume 4, pp. 3721–3726. [Google Scholar]
- Alamri, A.; Eid, M.; Iglesias, R.; Shirmohammadi, S.; Saddik, A.E. Haptic Virtual Rehabilitation Exercises for Poststroke Diagnosis. IEEE Trans. Instrum. Meas. 2008, 57, 1876–1884. [Google Scholar] [CrossRef]
- Kayyali, R.; Shirmohammadi, S.; Saddik, A.E.; Lemaire, E. Daily-Life Exercises for Haptic Motor Rehabilitation. In Proceedings of the 2007 IEEE International Workshop on Haptic, Audio and Visual Environments and Games, Ottawa, ON, Canada, 12–14 October 2007; pp. 118–123. [Google Scholar]
- Adamovich, S.V.; Fluet, G.G.; Mathai, A.; Qiu, Q.; Lewis, J.; Merians, A.S. Design of a complex virtual reality simulation to train finger motion for persons with hemiparesis: A proof of concept study. J. Neuroeng. Rehabil. 2009, 6, 28. [Google Scholar] [CrossRef] [PubMed]
- Molier, B.I.; Prange, G.B.; Krabben, T.; Stienen, A.H.; van der Kooij, H.; Buurke, J.H.; Jannink, M.J.; Hermens, H.J. Effect of position feedback during task-oriented upper-limb training after stroke: Five-case pilot study. J. Rehabil. Res. Dev. 2011, 48, 1109–1118. [Google Scholar] [CrossRef] [PubMed]
- Jack, D.; Boian, R.; Merians, A.S.; Tremaine, M.; Burdea, G.C.; Adamovich, S.V.; Recce, M.; Poizner, H. Virtual reality-enhanced stroke rehabilitation. IEEE Trans. Neural Syst. Rehabil. Eng. 2001, 9, 308–318. [Google Scholar] [CrossRef]
- Wille, D.; Eng, K.; Holper, L.; Chevrier, E.; Hauser, Y.; Kiper, D.; Pyk, P.; Schlegel, S.; Meyer-Heim, A. Virtual reality-based paediatric interactive therapy system (PITS) for improvement of arm and hand function in children with motor impairment—A pilot study. Dev. Neurorehabilit. 2009, 12, 44–52. [Google Scholar] [CrossRef]
- Connelly, L.; Stoykov, M.E.; Jia, Y.; Toro, M.L.; Kenyon, R.V.; Kamper, D.G. Use of a pneumatic glove for hand rehabilitation following stroke. In Proceedings of the 2009 Annual International Conference of the IEEE Engineering in Medicine and Biology Society, Minneapolis, MN, USA, 3–6 September 2009; pp. 2434–2437. [Google Scholar]
- Huang, X.; Naghdy, F.; Naghdy, G.; Du, H. Clinical effectiveness of combined virtual reality and robot assisted fine hand motion rehabilitation in subacute stroke patients. In Proceedings of the 2017 International Conference on Rehabilitation Robotics (ICORR), London, UK, 17–20 July 2017; pp. 511–515. [Google Scholar]
- Pignolo, L.; Dolce, G.; Basta, G.; Lucca, L.F.; Serra, S.; Sannita, W.G. Upper limb rehabilitation after stroke: ARAMIS a “robo-mechatronic” innovative approach and prototype. In Proceedings of the 2012 4th IEEE RAS & EMBS International Conference on Biomedical Robotics and Biomechatronics (BioRob), Rome, Italy, 24–27 June 2012; pp. 1410–1414. [Google Scholar]
- Andaluz, V.H.; Salazar, P.J.; Silva, S.M.; Escudero, V.M.; Bustamante, D.C. Rehabilitation of upper limb with force feedback. In Proceedings of the 2016 IEEE International Conference on Automatica (ICA-ACCA), Curico, Chile, 19–21 October 2016; pp. 1–6. [Google Scholar]
- Bardorfer, A.; Munih, M.; Zupan, A.; Primozic, A. Upper limb motion analysis using haptic interface. IEEE/ASME Trans. Mechatron. 2001, 6, 253–260. [Google Scholar] [CrossRef]
- Broeren, J.; Rydmark, M.; Sunnerhagen, K.S. Virtual reality and haptics as a training device for movement rehabilitation after stroke: A single-case. Arch. Phys. Med. Rehabil. 2004, 85, 1247–1250. [Google Scholar] [CrossRef]
- Adamovich, S.V.; Fluet, G.G.; Merians, A.S.; Mathai, A.; Qiu, Q. Incorporating Haptic Effects Into Three-Dimensional Virtual Environments to Train the Hemiparetic Upper Extremity. IEEE Trans. Neural Syst. Rehabil. Eng. 2009, 17, 512–520. [Google Scholar] [CrossRef]
- Merians, A.S.; Fluet, G.G.; Qiu, Q.; Saleh, S.; Lafond, I.; Davidow, A.; Adamovich, S.V. Robotically facilitated virtual rehabilitation of arm transport integrated with finger movement in persons with hemiparesis. J. Neuroeng. Rehabil. 2011, 8, 27. [Google Scholar] [CrossRef]
- Nagaraj, S.B.; Constantinescu, D. Effect of Haptic Force Feedback on Upper Limb. In Proceedings of the 2009 Second International Conference on Emerging Trends in Engineering & Technology, Nagpur, India, 16–18 December 2009; pp. 55–58. [Google Scholar]
- Chiang, V.C.; Lo, K.H.; Choi, K.S. Rehabilitation of activities of daily living in virtual environments with intuitive user interface and force feedback. Disabil. Rehabil. Assist. Technol. 2017, 12, 672–680. [Google Scholar] [CrossRef]
- Sadihov, D.; Migge, B.; Gassert, R.; Kim, Y. Prototype of a VR upper-limb rehabilitation system enhanced with motion-based tactile feedback. In Proceedings of the 2013 World Haptics Conference (WHC), Daejeon, Korea, 14–17 April 2013; pp. 449–454. [Google Scholar]
- Kapur, P.; Premakumar, S.; Jax, S.A.; Buxbaum, L.J.; Dawson, A.M.; Kuchenbecker, K.J. Vibrotactile feedback system for intuitive upper-limb rehabilitation. In Proceedings of the World Haptics 2009—Third Joint EuroHaptics Conference and Symposium on Haptic Interfaces for Virtual Environment and Teleoperator Systems, Salt Lake City, UT, USA, 18–20 March 2009; pp. 621–622. [Google Scholar]
- Ramírez-Fernández, C.; Morán, A.L.; García-Canseco, E. Haptic feedback in motor hand virtual therapy increases precision and generates less mental workload. In Proceedings of the 2015 9th International Conference on Pervasive Computing Technologies for Healthcare (PervasiveHealth), Istanbul, Turkey, 20–23 May 2015; pp. 280–286. [Google Scholar]
- Aminov, A.; Rogers, J.M.; Middleton, S.; Caeyenberghs, K.; Wilson, P.H. What do randomized controlled trials say about virtual rehabilitation in stroke? A systematic literature review and meta-analysis of upper-limb and cognitive outcomes. J. Neuroeng. Rehabil. 2018, 15, 29. [Google Scholar] [CrossRef] [PubMed]
- Lohse, K.R.; Hilderman, C.G.; Cheung, K.L.; Tatla, S.; Van der Loos, H.F. Virtual reality therapy for adults post-stroke: A systematic review and meta-analysis exploring virtual environments and commercial games in therapy. PLoS ONE 2014, 9, e93318. [Google Scholar] [CrossRef] [PubMed]
- Mekbib, D.B.; Han, J.; Zhang, L.; Fang, S.; Jiang, H.; Zhu, J.; Roe, A.W.; Xu, D. Virtual reality therapy for upper limb rehabilitation in patients with stroke: A meta-analysis of randomized clinical trials. Brain Inj. 2020, 34, 456–465. [Google Scholar] [CrossRef] [PubMed]
- Karamians, R.; Proffitt, R.; Kline, D.; Gauthier, L.V. Effectiveness of Virtual Reality- and Gaming-Based Interventions for Upper Extremity Rehabilitation Poststroke: A Meta-analysis. Arch. Phys. Med. Rehabil. 2020, 101, 885–896. [Google Scholar] [CrossRef]
- Lee, H.S.; Park, Y.J.; Park, S.W. The Effects of Virtual Reality Training on Function in Chronic Stroke Patients: A Systematic Review and Meta-Analysis. BioMed Res. Int. 2019, 2019, 7595639. [Google Scholar] [CrossRef]
- Laver, K.E.; Lange, B.; George, S.; Deutsch, J.E.; Saposnik, G.; Crotty, M. Virtual reality for stroke rehabilitation. Cochrane Database Syst. Rev. 2017, 11, Cd008349. [Google Scholar] [CrossRef]
- Shea, B.J.; Reeves, B.C.; Wells, G.; Thuku, M.; Hamel, C.; Moran, J.; Moher, D.; Tugwell, P.; Welch, V.; Kristjansson, E.; et al. AMSTAR 2: A critical appraisal tool for systematic reviews that include randomised or non-randomised studies of healthcare interventions, or both. BMJ 2017, 358, j4008. [Google Scholar] [CrossRef]
- Lang, C.E.; Edwards, D.F.; Birkenmeier, R.L.; Dromerick, A.W. Estimating minimal clinically important differences of upper-extremity measures early after stroke. Arch. Phys. Med. Rehabil. 2008, 89, 1693–1700. [Google Scholar] [CrossRef]
- Sivan, M.; O‘Connor, R.J.; Makower, S.; Levesley, M.; Bhakta, B. Systematic review of outcome measures used in the evaluation of robot-assisted upper limb exercise in stroke. J. Rehabil. Med. 2011, 43, 181–189. [Google Scholar] [CrossRef]
- Oman, C.M. Motion sickness: A synthesis and evaluation of the sensory conflict theory. Can. J. Physiol. Pharm. 1990, 68, 294–303. [Google Scholar] [CrossRef]
- Qualcomm Developer Network. Reducing Cybersickness in Virtual Reality: What is Cybersickness and how it can be Reduced or Prevented when Developing Virtual Reality Experiences. Available online: https://developer.qualcomm.com/ebook/reducing-cybersickness-virtual-reality (accessed on 20 October 2020).
- Viau, A.; Feldman, A.G.; McFadyen, B.J.; Levin, M.F. Reaching in reality and virtual reality: A comparison of movement kinematics in healthy subjects and in adults with hemiparesis. J. Neuroeng. Rehabil. 2004, 1, 11. [Google Scholar] [CrossRef]
- Hussain, N.; Alt Murphy, M.; Sunnerhagen, K.S. Upper Limb Kinematics in Stroke and Healthy Controls Using Target-to-Target Task in Virtual Reality. Front. Neurol. 2018, 9, 300. [Google Scholar] [CrossRef] [PubMed]
- Magdalon, E.C.; Michaelsen, S.M.; Quevedo, A.A.; Levin, M.F. Comparison of grasping movements made by healthy subjects in a 3-dimensional immersive virtual versus physical environment. Acta Psychol. 2011, 138, 126–134. [Google Scholar] [CrossRef] [PubMed]
- Levin, M.F.; Magdalon, E.C.; Michaelsen, S.M.; Quevedo, A.A. Quality of Grasping and the Role of Haptics in a 3-D Immersive Virtual Reality Environment in Individuals With Stroke. IEEE Trans. Neural Syst. Rehabil. Eng. 2015, 23, 1047–1055. [Google Scholar] [CrossRef] [PubMed]
- Lott, A.; Bisson, E.; Lajoie, Y.; McComas, J.; Sveistrup, H. The effect of two types of virtual reality on voluntary center of pressure displacement. Cyberpsychology Behav. 2003, 6, 477–485. [Google Scholar] [CrossRef]
- Taub, E.; Uswatte, G.; Mark, V.W.; Morris, D.M.; Barman, J.; Bowman, M.H.; Bryson, C.; Delgado, A.; Bishop-McKay, S. Method for enhancing real-world use of a more affected arm in chronic stroke: Transfer package of constraint-induced movement therapy. Stroke 2013, 44, 1383–1388. [Google Scholar] [CrossRef]
- Basteris, A.; Nijenhuis, S.M.; Stienen, A.H.; Buurke, J.H.; Prange, G.B.; Amirabdollahian, F. Training modalities in robot-mediated upper limb rehabilitation in stroke: A framework for classification based on a systematic review. J. Neuroeng. Rehabil. 2014, 11, 111. [Google Scholar] [CrossRef]
- Levac, D.E.; Huber, M.E.; Sternad, D. Learning and transfer of complex motor skills in virtual reality: A perspective review. J. Neuroeng. Rehabil. 2019, 16, 121. [Google Scholar] [CrossRef]
- Rose, F.D.; Attree, E.A.; Brooks, B.M.; Parslow, D.M.; Penn, P.R.; Ambihaipahan, N. Training in virtual environments: Transfer to real world tasks and equivalence to real task training. Ergonomics 2000, 43, 494–511. [Google Scholar] [CrossRef]
- De Mello Monteiro, C.B.; Massetti, T.; da Silva, T.D.; van der Kamp, J.; de Abreu, L.C.; Leone, C.; Savelsbergh, G.J. Transfer of motor learning from virtual to natural environments in individuals with cerebral palsy. Res. Dev. Disabil. 2014, 35, 2430–2437. [Google Scholar] [CrossRef]
- Massetti, T.; Fávero, F.M.; Menezes, L.D.C.; Alvarez, M.P.B.; Crocetta, T.B.; Guarnieri, R.; Nunes, F.L.S.; Monteiro, C.B.M.; Silva, T.D.D. Achievement of Virtual and Real Objects Using a Short-Term Motor Learning Protocol in People with Duchenne Muscular Dystrophy: A Crossover Randomized Controlled Trial. Games Health J. 2018, 7, 107–115. [Google Scholar] [CrossRef] [PubMed]
- Anglin, J.; Saldana, D.; Schmiesing, A.; Liew, S.-L. Transfer of a skilled motor learning task between virtual and conventional environments. In Proceedings of the 2017 IEEE Virtual Reality (VR), Los Angeles, CA, USA, 18–22 March 2017; pp. 401–402. [Google Scholar]
- Levac, D.E.; Jovanovic, B.B. Is children’s motor learning of a postural reaching task enhanced by practice in a virtual environment? In Proceedings of the 2017 International Conference on Virtual Rehabilitation (ICVR), Montreal, QC, Canada, 19–22 June 2017; pp. 1–7. [Google Scholar]
- Sakata, S.; Grove, P.M.; Hill, A.; Watson, M.O.; Stevenson, A.R.L. Impact of simulated three-dimensional perception on precision of depth judgements, technical performance and perceived workload in laparoscopy. Br. J. Surg. 2017, 104, 1097–1106. [Google Scholar] [CrossRef] [PubMed]
- Charles, D.; Holmes, D.; Charles, T.; McDonough, S. Virtual Reality Design for Stroke Rehabilitation. Adv. Exp. Med. Biol. 2020, 1235, 53–87. [Google Scholar] [PubMed]
- Ebrahimi, E.; Babu, S.V.; Pagano, C.C.; Jörg, S. An empirical evaluation of visuo-haptic feedback on physical reaching behaviors during 3D interaction in real and immersive virtual environments. ACM Trans. Appl. Percept. TAP 2016, 13, 1–21. [Google Scholar] [CrossRef]
- Ranganathan, R.; Wieser, J.; Mosier, K.M.; Mussa-Ivaldi, F.A.; Scheidt, R.A. Learning redundant motor tasks with and without overlapping dimensions: Facilitation and interference effects. J. Neurosci. 2014, 34, 8289–8299. [Google Scholar] [CrossRef]
- Deterding, S.; Dixon, D.; Khaled, R.; Nacke, L. From game design elements to gamefulness: Defining “gamification”. In Proceedings of the 15th International Academic MindTrek Conference: Envisioning Future Media Environments, Tampere, Finland, 28–30 September 2011; pp. 9–15. [Google Scholar]
- Yates, M.; Kelemen, A.; Sik Lanyi, C. Virtual reality gaming in the rehabilitation of the upper extremities post-stroke. Brain Inj. 2016, 30, 855–863. [Google Scholar] [CrossRef]
- Mubin, O.; Alnajjar, F.; Al Mahmud, A.; Jishtu, N.; Alsinglawi, B. Exploring serious games for stroke rehabilitation: A scoping review. Disabil. Rehabil. Assist. Technol. 2020, 1–7. [Google Scholar] [CrossRef]
- Shin, J.H.; Ryu, H.; Jang, S.H. A task-specific interactive game-based virtual reality rehabilitation system for patients with stroke: A usability test and two clinical experiments. J. Neuroeng. Rehabil. 2014, 11, 32. [Google Scholar] [CrossRef]
- Hocine, N.; Gouaïch, A.; Cerri, S.A.; Mottet, D.; Froger, J.; Laffont, I. Adaptation in serious games for upper-limb rehabilitation: An approach to improve training outcomes. User Modeling User-Adapt. Interact. 2015, 25, 65–98. [Google Scholar] [CrossRef]
- Levac, D.; Glegg, S.; Colquhoun, H.; Miller, P.; Noubary, F. Virtual Reality and Active Videogame-Based Practice, Learning Needs, and Preferences: A Cross-Canada Survey of Physical Therapists and Occupational Therapists. Games Health J. 2017, 6, 217–228. [Google Scholar] [CrossRef]
- Glegg, S.M.N.; Levac, D.E. Barriers, Facilitators and Interventions to Support Virtual Reality Implementation in Rehabilitation: A Scoping Review. PM&R 2018, 10, 1237–1251.e1. [Google Scholar]
- Johansson, B.B. Brain plasticity and stroke rehabilitation. The Willis lecture. Stroke 2000, 31, 223–230. [Google Scholar] [CrossRef] [PubMed]
- Pascual-Leone, A.; Amedi, A.; Fregni, F.; Merabet, L.B. The plastic human brain cortex. Annu. Rev. Neurosci. 2005, 28, 377–401. [Google Scholar] [CrossRef]
- Nithianantharajah, J.; Hannan, A.J. Enriched environments, experience-dependent plasticity and disorders of the nervous system. Nat. Rev. Neurosci. 2006, 7, 697–709. [Google Scholar] [CrossRef] [PubMed]
- Cervera, M.A.; Soekadar, S.R.; Ushiba, J.; Millán, J.D.R.; Liu, M.; Birbaumer, N.; Garipelli, G. Brain-computer interfaces for post-stroke motor rehabilitation: A meta-analysis. Ann. Clin. Transl. Neurol. 2018, 5, 651–663. [Google Scholar] [CrossRef] [PubMed]
- Van Dokkum, L.E.H.; Ward, T.; Laffont, I. Brain computer interfaces for neurorehabilitation—Its current status as a rehabilitation strategy post-stroke. Ann. Phys. Rehabil. Med. 2015, 58, 3–8. [Google Scholar] [CrossRef] [PubMed]
- Pichiorri, F.; Morone, G.; Petti, M.; Toppi, J.; Pisotta, I.; Molinari, M.; Paolucci, S.; Inghilleri, M.; Astolfi, L.; Cincotti, F.; et al. Brain-computer interface boosts motor imagery practice during stroke recovery. Ann. Neurol. 2015, 77, 851–865. [Google Scholar] [CrossRef]
- Vourvopoulos, A.; Pardo, O.M.; Lefebvre, S.; Neureither, M.; Saldana, D.; Jahng, E.; Liew, S.L. Effects of a Brain-Computer Interface With Virtual Reality (VR) Neurofeedback: A Pilot Study in Chronic Stroke Patients. Front. Hum. Neurosci. 2019, 13, 210. [Google Scholar] [CrossRef]
- Ward, N.S.; Cohen, L.G. Mechanisms underlying recovery of motor function after stroke. Arch. Neurol. 2004, 61, 1844–1848. [Google Scholar] [CrossRef]
- Talelli, P.; Greenwood, R.J.; Rothwell, J.C. Arm function after stroke: Neurophysiological correlates and recovery mechanisms assessed by transcranial magnetic stimulation. Clin. Neurophysiol. 2006, 117, 1641–1659. [Google Scholar] [CrossRef]
- Hummel, F.C.; Cohen, L.G. Non-invasive brain stimulation: A new strategy to improve neurorehabilitation after stroke? Lancet Neurol. 2006, 5, 708–712. [Google Scholar] [CrossRef]
- Khedr, E.M.; Ahmed, M.A.; Fathy, N.; Rothwell, J.C. Therapeutic trial of repetitive transcranial magnetic stimulation after acute ischemic stroke. Neurology 2005, 65, 466–468. [Google Scholar] [CrossRef] [PubMed]
- Gandiga, P.C.; Hummel, F.C.; Cohen, L.G. Transcranial DC stimulation (tDCS): A tool for double-blind sham-controlled clinical studies in brain stimulation. Clin. Neurophysiol. 2006, 117, 845–850. [Google Scholar] [CrossRef] [PubMed]
- Hummel, F.; Celnik, P.; Giraux, P.; Floel, A.; Wu, W.H.; Gerloff, C.; Cohen, L.G. Effects of non-invasive cortical stimulation on skilled motor function in chronic stroke. Brain 2005, 128 Pt 3, 490–499. [Google Scholar] [CrossRef]
- Mansur, C.G.; Fregni, F.; Boggio, P.S.; Riberto, M.; Gallucci-Neto, J.; Santos, C.M.; Wagner, T.; Rigonatti, S.P.; Marcolin, M.A.; Pascual-Leone, A. A sham stimulation-controlled trial of rTMS of the unaffected hemisphere in stroke patients. Neurology 2005, 64, 1802–1804. [Google Scholar] [CrossRef]
- Kim, W.-S.; Kwon, B.S.; Seo, H.G.; Park, J.; Paik, N.-J. Low-frequency repetitive transcranial magnetic stimulation over contralesional motor cortex for motor recovery in subacute ischemic stroke: A randomized sham-controlled trial. Neurorehabil. Neural Repair 2020, 34, 856–867. [Google Scholar] [CrossRef]
- Fregni, F.; Boggio, P.S.; Mansur, C.G.; Wagner, T.; Ferreira, M.J.; Lima, M.C.; Rigonatti, S.P.; Marcolin, M.A.; Freedman, S.D.; Nitsche, M.A.; et al. Transcranial direct current stimulation of the unaffected hemisphere in stroke patients. Neuroreport 2005, 16, 1551–1555. [Google Scholar] [CrossRef]
- Vines, B.W.; Cerruti, C.; Schlaug, G. Dual-hemisphere tDCS facilitates greater improvements for healthy subjects’ non-dominant hand compared to uni-hemisphere stimulation. BMC Neurosci. 2008, 9, 103. [Google Scholar] [CrossRef]
- Floel, A.; Nagorsen, U.; Werhahn, K.J.; Ravindran, S.; Birbaumer, N.; Knecht, S.; Cohen, L.G. Influence of somatosensory input on motor function in patients with chronic stroke. Ann. Neurol. 2004, 56, 206–212. [Google Scholar] [CrossRef]
- Sawaki, L.; Wu, C.W.; Kaelin-Lang, A.; Cohen, L.G. Effects of somatosensory stimulation on use-dependent plasticity in chronic stroke. Stroke 2006, 37, 246–247. [Google Scholar] [CrossRef]
- Rossi, S.; Hallett, M.; Rossini, P.M.; Pascual-Leone, A. Safety, ethical considerations, and application guidelines for the use of transcranial magnetic stimulation in clinical practice and research. Clin. Neurophysiol. 2009, 120, 2008–2039. [Google Scholar] [CrossRef] [PubMed]
- Bikson, M.; Paneri, B.; Mourdoukoutas, A.; Esmaeilpour, Z.; Badran, B.W.; Azzam, R.; Adair, D.; Datta, A.; Fang, X.H.; Wingeier, B.; et al. Limited output transcranial electrical stimulation (LOTES-2017): Engineering principles, regulatory statutes, and industry standards for wellness, over-the-counter, or prescription devices with low risk. Brain Stimul. 2018, 11, 134–157. [Google Scholar] [CrossRef] [PubMed]
- Zheng, C.J.; Liao, W.J.; Xia, W.G. Effect of combined low-frequency repetitive transcranial magnetic stimulation and virtual reality training on upper limb function in subacute stroke: A double-blind randomized controlled trail. J. Huazhong Univ. Sci. Technol. Med. Sci. 2015, 35, 248–254. [Google Scholar] [CrossRef] [PubMed]
- Johnson, N.N.; Carey, J.; Edelman, B.J.; Doud, A.; Grande, A.; Lakshminarayan, K.; He, B. Combined rTMS and virtual reality brain-computer interface training for motor recovery after stroke. J. Neural Eng. 2018, 15, 016009. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.J.; Chun, M.H. Combination transcranial direct current stimulation and virtual reality therapy for upper extremity training in patients with subacute stroke. Arch. Phys. Med. Rehabil. 2014, 95, 431–438. [Google Scholar] [CrossRef]
- Sarfo, F.S.; Ulasavets, U.; Opare-Sem, O.K.; Ovbiagele, B. Tele-Rehabilitation after Stroke: An Updated Systematic Review of the Literature. J. Stroke Cereb. Dis 2018, 27, 2306–2318. [Google Scholar] [CrossRef]
- Laver, K.E.; Adey-Wakeling, Z.; Crotty, M.; Lannin, N.A.; George, S.; Sherrington, C. Telerehabilitation services for stroke. Cochrane Database Syst. Rev. 2020, 1, Cd010255. [Google Scholar] [CrossRef]
- Piron, L.; Turolla, A.; Agostini, M.; Zucconi, C.; Cortese, F.; Zampolini, M.; Zannini, M.; Dam, M.; Ventura, L.; Battauz, M.; et al. Exercises for paretic upper limb after stroke: A combined virtual-reality and telemedicine approach. J. Rehabil. Med. 2009, 41, 1016–1102. [Google Scholar] [CrossRef]
- Choi, Y.H.; Ku, J.; Lim, H.; Kim, Y.H.; Paik, N.J. Mobile game-based virtual reality rehabilitation program for upper limb dysfunction after ischemic stroke. Restor. Neurol. Neurosci. 2016, 34, 455–463. [Google Scholar] [CrossRef]
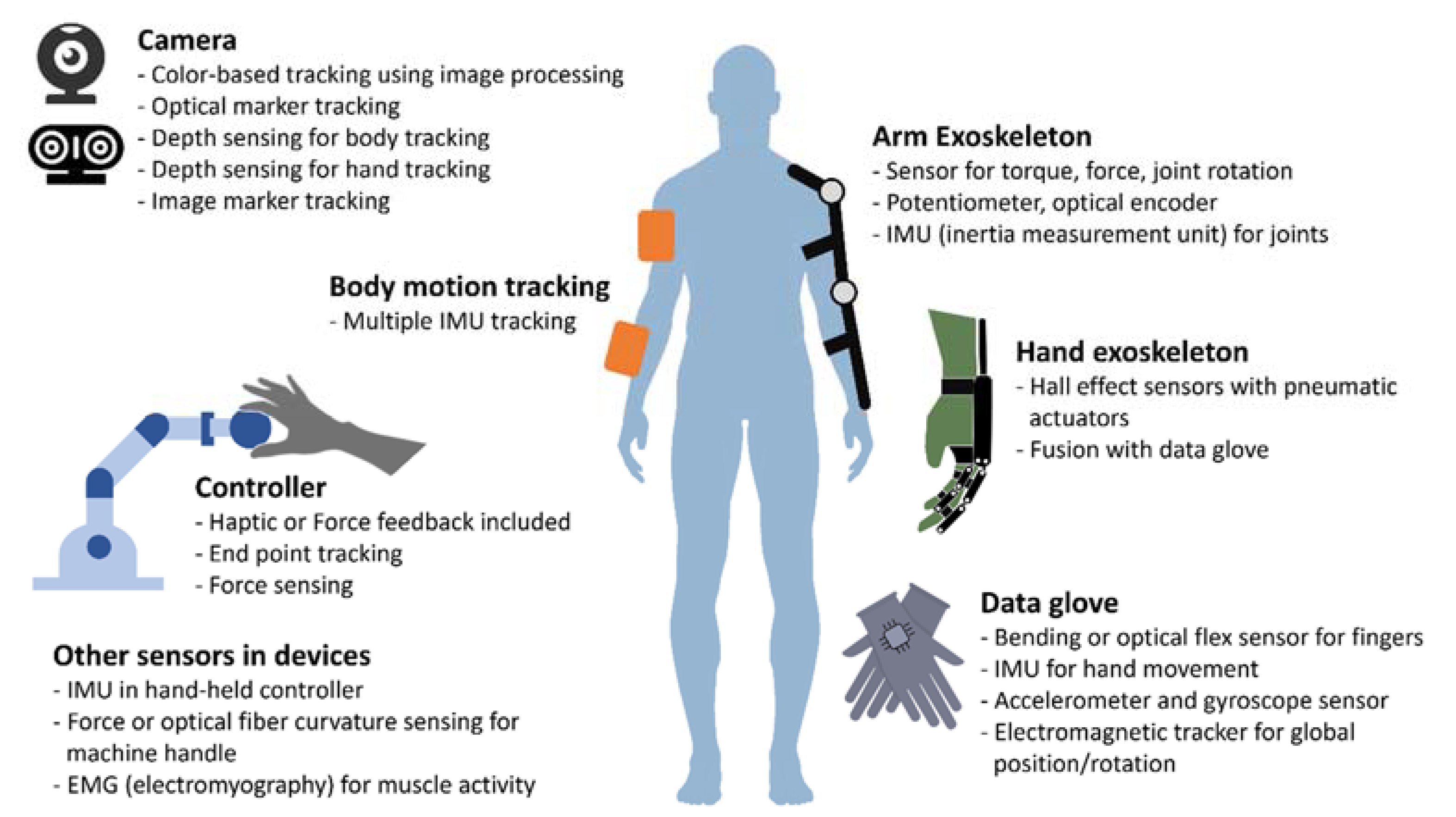
| Study | Sensor Type | Sensor Type (Detail) | Feedback Type | VR Type | Rehabilitation Part |
|---|---|---|---|---|---|
| Visuomotor Feedback | |||||
| Dimbwadyo-Terrer et al., 2016 [29] | Wearable | Data glove | V | NI | Arm, hand, finger |
| Crosbie et al., 2012 [30] | Wearable | Data glove, motion tracking sensor | V, A | I | Arm, hand, finger |
| Calabrò et al., 2019 [31] | Wearable | End-effector hand exoskeleton | V, A | NI | Finger |
| Kiper et al., 2018 [32] | Wearable | Electromagnetic sensor | V | NI | Arm, hand |
| Cho et al., 2016 [33] | Nonwearable | Motion-sensing camera (depth sensing, hand tracking) | V, A | NI | Arm, hand, finger |
| Askin et al., 2018 [34] | Nonwearable | Motion-sensing Camera (body tracking with depth sensing) | V | NI | Arm |
| Faria et al., 2018 [35] | Nonwearable | Marker-based tracking with webcam | V | NI | Arm, hand |
| Lee et al., 2018 [36] | Nonwearable | Controller for paddling movement (canoe-like apparatus) | V, A | NI | Arm, hand |
| Sucar et al., 2014 [37] | Nonwearable | Pressure sensor in custom gripper, colored object tracking with webcam | V, A | NI | Arm, hand |
| Ballester et al., 2017 [38] | Wearable, nonwearable | Motion-sensing camera (depth sensing, body tracking), data glove | V | NI | Arm, hand, finger |
| Sampson et al., 2012 [39] | Wearable, nonwearable | Colored object tracking with webcam | V | NI | Arm |
| Xin et al., 2014 [40] | Wearable, nonwearable | Motion-sensing camera (body tracking with depth sensing), EMG sensing | V | I | Arm |
| Visuohaptic Feedback | |||||
| Feintuch et al., 2006 [41] | Wearable | Colored glove tracking with webcam | T, V, A | NI | Arm |
| Popescu et al., 2000 [42] | Wearable | Non-contact position sensors | F, V, A | NI | Hand, finger |
| Prisco et al., 1998 [43] | Wearable | Glove with electromagnetic measurements, torque/force and joint rotation sensing in arm exoskeleton | F, V, A | I | Arm, hand, finger |
| Alamri et al., 2008 [44], Kayyali et al., 2007 [45] | Wearable | Data glove with hand exoskeleton | F, V | NI | Arm, hand, finger |
| Adamovich et al., 2009 [46] | Wearable | Data glove with hand exoskeleton | F, V, A | NI | Arm, hand, finger |
| Molier et al., 2011 [47] | Wearable | Potentiometer and optical encoder in arm exoskeleton | F, V, A | NI | Arm, hand |
| Jack et al., 2001 [48] Merians et al., 2002 [13] | Wearable | Non-contact position sensor, data glove | F, V, A | NI | Hand, finger |
| Wille et al., 2009 [49] | Wearable | Data glove, accelerometers, and magnetometers | T, V | NI | Arm, hand, finger |
| Connelly et al., 2009 [50] | Wearable | Data glove, magnetic tracker for head tracking | T, V, A | I | Hand, finger |
| Huang et al., 2017 [51] | Wearable | Position and force sensor in hand rehabilitation robot | V, A | I | Finger |
| Pignolo et al., 2012 [52] | Wearable | Optical encoder in arm exoskeleton | V, A | I | Arm |
| Andaluz et al., 2016 [53], Bardorfer et al., 2001 [54] | Nonwearable | 3D controller including buttons | F, V | NI | Arm, hand, finger |
| Broeren et al., 2004 [55] | Nonwearable | 3D controller | F, V | SI | Hand, finger |
| Adamovich et al., 2009 [56] | Nonwearable | Force sensor in 3 DOF admittance-controlled robot | F, V | NI | Arm, hand |
| Merians et al., 2011 [57] | Nonwearable | Data glove, optical fiber curvature sensor, force sensor in 3 DOF admittance-controlled robot | F, V | NI | Arm, hand, finger |
| Nagaraj e al., 2009 [58], Chiang et al., 2017 [59] | Nonwearable | 3D controller | F, V, A | NI | Arm, hand |
| Sadihov et al., 2013 [60] | Wearable and nonwearable | Motion sensing camera (depth sensing, body tracking), data glove (bend sensing) | T, V | NI | Arm, hand, finger |
| Kapur et al., 2009 [61] | Wearable and nonwearable | Sleeve for optical tracking (camera) | T, V | NI | Arm |
| Ramírez-Fernández et al., 2015 [62] | Wearable and nonwearable | 3D controller, motion sensing camera (depth sensing, hand tracking) | F, V, A | NI | Arm, hand |
| VR System | VR Type | Sensor Type | Body Part | Company | Country |
|---|---|---|---|---|---|
| Riablo Premium | NI | IMU sensor | Arm | CoRehab | Italy |
| SaeboVR | NI | Motion-sensing camera (depth sensing, body tracking) | Arm | Saebo | USA |
| Doctor Kinetic | NI | Motion-sensing camera (depth sensing, body tracking) | Arm | Doctor Kinetic | Netherlands |
| IREX | NI | Motion sensing with webcam | Arm | GestureTek Health | Canada |
| Virtual Rehab | NI | Motion-sensing camera (depth sensing, body tracking, and hand tracking) | Arm, hand | Evolv | Spain |
| XR Health | I | HMD, controller | Arm | XR Health | USA |
| iWall | NI | Motion-sensing camera (depth sensing, body tracking), touch screen | Arm, hand | CSE Entertainment | Finland |
| Nirvana | NI | wall or floor touch sensing | Arm, hand | BTS Bioengineering | USA |
| Myro | NI | Touch screen, touchable objects on screen | Arm, hand | Tyromotion | USA |
| DIEGO | NI | Hand suspended type | Arm | Tyromotion | USA |
| AMADEO | NI | Position and force sensor in hand rehab robot | Finger | Tyromotion | USA |
| Pablo | NI | IMU sensor | Arm, hand | Tyromotion | USA |
| EsoGLOVE | NI | Hand exoskeleton | Arm, hand, finger | Roceso Technologies | Singapore |
| Bimeo PRO | NI | IMU sensor for body, IMU sensor in objects | Arm, hand | Kinestica | Slovenia |
| HandTutor | NI | Data glove | Hand, finger | Meditouch | Israel |
| Playball | NI | IMU sensor in ball | Hand | Tonkey | Italy |
| Anika | NI | Data glove | Hand, finger | ZARYA | Russia |
| Gloreha Workstation plus | NI | Hand exoskeleton, Optical sensor | Hand, finger | Gloreha | Italy |
| Icone | NI | Machine holding and moving handle | Arm | Heaxel | Italy |
| ExoRehab X | NI | Arm exoskeleton | Arm | HoustonBionic | Turkey |
| Hand of Hope | NI | Hand exoskeleton | Hand, finger | Rehab-Robotics Company | Hong Kong |
| SaeboRejoyce | NI | 3D movable handle | Arm, hand | Saebo | USA |
| MindMotion Pro | NI | Colored object 3D tracking | Arm, hand | MindMaze | Switzerland |
| YouGrabber | NI | Data glove, infrared tracking camera | Arm, hand, finger | YouRehab | Switzerland |
| Rapel Smart Glove | NI | Data glove, IMU sensor | Arm, hand, finger | Neofect | South Korea |
| Smartboard | NI | 2D handling board | Arm | Neofect | South Korea |
| MusicGlove | NI | Finger-to-finger contact | Finger | FlintRehab | USA |
| FitMi | NI | Puck with multiple sensors for movement tracking | Arm, hand | FlintRehab | USA |
| SensoRehab | NI | Data glove | Hand, finger | SensoMed | Russia |
| Rewellio | I | HMD, controller | Arm | Rewellio Inc. | USA |
| Study | Aim | Search Strategy | Search Period | Inclusion Criteria | Included Trials, n | Participants, n |
|---|---|---|---|---|---|---|
| Lohse et al., 2014 [64] | To demonstrate the effect of virtual reality (VR) therapy among patients after stroke in both custom built virtual environments and commercial gaming systems. | MEDLINE, CINAHL, EMBASE, ERIC, PSYCInfo, DARE, PEDro, Cochrane Central Register of Controlled Trials, and Cochrane Database of Systematic Reviews | From inception to 4 April 2013 | Randomized or quasi-randomized controlled trials with adults (>18 years old) after stroke, excluding other neurological disorders. | 24 | 626 |
| Laver et al., 2017 [68] | To investigate the efficacy of VR in comparison with alternative interventions or no interventions on the function and activity of hemiparetic upper limbs. | Cochrane Stroke Group Trials Register, CENTRAL, MEDLINE, Embase, and seven additional databases | From inception to April 2017 | Randomized and quasi-randomized trials of VR rehabilitation in adults after stroke. | 72 | 2470 |
| Aminov et al., 2018 [63] | To review the evidence for VR in upper limb function and cognition after stroke. | Scopus, Cochrane Database, CINAHL, The Allied and Complementary Medicine Database, Web of Science, MEDLINE, Pre-Medline, PsycEXTRA, and PsycINFO | From inception to 28 June 2017 | Randomized controlled trials utilizing a VR to improve either motor (upper limb) function, cognitive, or activities of daily living in patients with stroke. | 31 | 971 |
| Lee et al., 2019 [67] | To evaluate the effect of VR training on lower limb, upper limb, and overall functions in patients with chronic stroke. | OVID, PubMed, and EMBASE | From January 2000 to June 2018 | Randomized controlled trials for using VR as a rehabilitation intervention in patients with chronic stroke. | 21 | 562 |
| Karamians et al., 2020 [66] | To demonstrate the efficacy of VR- and gaming-based rehabilitations for upper limb function in patients with stroke. | PubMed, CINAHL/EBSCO, SCOPUS, Ovid MEDLINE, and EMBASE | From 2005 to 2019 | Randomized controlled trials or prospective study design with outcome measures of Wolf Motor Function Test, Fugl-Meyer Assessment or Action Research Arm Test in patients who had poststroke upper extremity deficits. | 38 | 1198 |
| Mekbib et al., 2020 [65] | To evaluate the therapeutic effect of VR compared to dose-matched conventional therapy in patients with stroke. | EMBASE, MEDLINE, PubMed, and Web of Science | From 2010 to February 2019 | Randomized controlled trials that allocated patients either to a VR therapy or to a dose-matched conventional therapy. | 27 | 1094 |
| Study | Intervention | Comparison | Outcomes | Major Findings | Conclusions | Methodological Quality |
|---|---|---|---|---|---|---|
| Lohse et al., 2014 [64] | VR therapy: Custom-built VE or CG | CT | Behavioral assessment in body function, activity, or participation according to International Classification of Functioning (ICF) | (1) Body function -VE: SMD = 0.43, 95% CI = 0.22 to 0.64 -CG: SMD = 0.76, 95% CI = −0.17 to 1.70 (2) Activity -VE: SMD = 0.54, 95% CI = 0.28 to 0.81 -CG: SMD = 0.76, 95% CI = −0.25 to 1.76 (3) Participation -VE: SMD = 0.56, 95% CI = 0.02 to 1.10 | VR rehabilitation moderately improves functional outcomes compared to CT in patients with stroke. CG studies were too few and small to evaluate the benefits of CG. | High |
| Laver et al., 2017 [68] | VR rehabilitation | Alternative intervention (usually CT) or no intervention | Upper limb function and activity | (1) Upper limb function (VR versus CT) -Composite: SMD = 0.07, 95% CI = −0.05 to 0.20 -FMA: SMD = 2.85, 95% CI = 1.06 to 4.65 (2) Upper limb function (additional VR) -Composite: SMD = 0.49, 95 CI = 0.21 to 0.77 (3) Activity of daily living -VR versus CT : SMD = 0.25, 95% CI = 0.06 to 0.43 -Additional VR : SMD = 0.44, 95% CI = 0.11 to 0.76 | VR rehabilitation was not superior to CT in improving upper limb function. VR may be beneficial, when applied as an additional therapy to usual care, to improve the function of hemiparetic upper limbs and activities of daily living as additional VR therapy can increase overall therapy time. | High |
| Aminov et al., 2018 [63] | VR rehabilitation | CT | Upper limb function (e.g., FMA) and activity (e.g., BBT, BI) according to ICF | (1) Upper limb function : SMD = 0.41, 95% CI = 0.28 to 0.55 (2) Upper limb activity : SMD = 0.47, 95% CI = 0.34 to 0.60 | VR can be beneficial on outcomes of body structure/function and activity in patients with stroke. | Moderate |
| Lee et al., 2019 [67] | VR rehabilitation | CT or no intervention | Upper limb function | (1) Upper limb function : SMD = 0.43, 95% CI = 0.42 to 0.54 (2) Lower limb function : SMD = 0.42, 95% CI = 0.34 to 0.51 (3) Overall function : SMD = 0.55, 95% CI = 0.25 to 0.84 | VR training moderately improved function in patients with chronic stroke. | Low |
| Karamians et al., 2020 [66] | VR rehabilitation | CT or no intervention | Upper limb function (FMA, WMFT, ARAT) | (1) VR or gaming versus all controls : Percent possible improvement = 28.45%, 95% CI = 24.40 to 32.49% (2) VR or gaming versus CT : Percent possible improvement = 10.40%, 95% CI = 5.65 to 15.14% | VR- or gaming-based rehabilitation for upper limb function was more effective than CT in patients with stroke. | Moderate |
| Mekbib et al., 2020 [65] | VR rehabilitation | Dose-matched CT | Upper limb function (FMA, BBT, MAL) | (1) FMA : Mean difference = 3.84, 95% CI = 0.93 to 6.75 (2) BBT : Mean difference = 3.82, 95% CI = 0.26 to 7.38 (3) MAL : Mean difference = 0.80, 9% CI = 0.44 to 1.15 | VR rehabilitation was more beneficial on post-stroke upper limb function in the outcomes of FMA, BBT and MAL than dose-matched CT. | Moderate |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, W.-S.; Cho, S.; Ku, J.; Kim, Y.; Lee, K.; Hwang, H.-J.; Paik, N.-J. Clinical Application of Virtual Reality for Upper Limb Motor Rehabilitation in Stroke: Review of Technologies and Clinical Evidence. J. Clin. Med. 2020, 9, 3369. https://doi.org/10.3390/jcm9103369
Kim W-S, Cho S, Ku J, Kim Y, Lee K, Hwang H-J, Paik N-J. Clinical Application of Virtual Reality for Upper Limb Motor Rehabilitation in Stroke: Review of Technologies and Clinical Evidence. Journal of Clinical Medicine. 2020; 9(10):3369. https://doi.org/10.3390/jcm9103369
Chicago/Turabian StyleKim, Won-Seok, Sungmin Cho, Jeonghun Ku, Yuhee Kim, Kiwon Lee, Han-Jeong Hwang, and Nam-Jong Paik. 2020. "Clinical Application of Virtual Reality for Upper Limb Motor Rehabilitation in Stroke: Review of Technologies and Clinical Evidence" Journal of Clinical Medicine 9, no. 10: 3369. https://doi.org/10.3390/jcm9103369
APA StyleKim, W.-S., Cho, S., Ku, J., Kim, Y., Lee, K., Hwang, H.-J., & Paik, N.-J. (2020). Clinical Application of Virtual Reality for Upper Limb Motor Rehabilitation in Stroke: Review of Technologies and Clinical Evidence. Journal of Clinical Medicine, 9(10), 3369. https://doi.org/10.3390/jcm9103369







