Clear Cell Adenocarcinoma of the Urinary Bladder Is a Glycogen-Rich Tumor with Poorer Prognosis
Abstract
1. Introduction
2. Experimental Section
2.1. Data Source
2.2. Sample Selection and Coding
2.3. Statistical Analysis
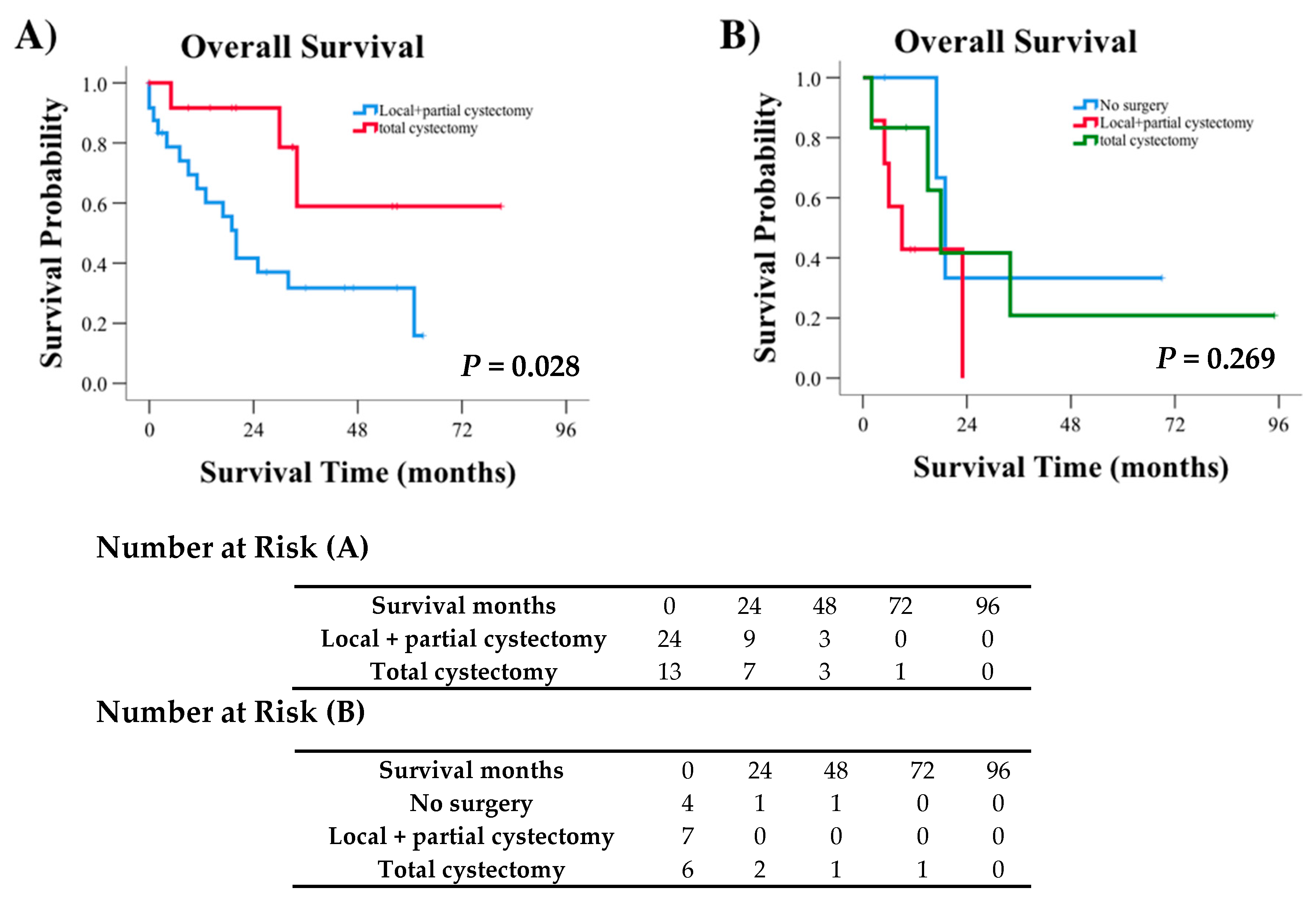
3. Results
3.1. Incidence and Mortality of CCA
3.2. Demographics and Clinical Characteristics
3.3. Survival
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References and Notes
- Young, L.E.A.; Brizzee, C.O.; Macedo, J.K.A.; Murphy, R.D.; Contreras, C.J.; DePaoli-Roach, A.A.; Roach, P.J.; Gentry, M.S.; Sun, R.C. Accurate and sensitive quantitation of glucose and glucose phosphates derived from storage carbohydrates by mass spectrometry. Carbohydr. Polym. 2019, 230, 115651. [Google Scholar] [CrossRef]
- Schulze, A.; Harris, A.L. How cancer metabolism is tuned for proliferation and vulnerable to disruption. Nature 2012, 491, 364–373. [Google Scholar] [CrossRef] [PubMed]
- Pelletier, J.; Bellot, G.; Gounon, P.; Lacas-Gervais, S.; Pouyssegur, J.; Mazure, N.M. Glycogen Synthesis is Induced in Hypoxia by the Hypoxia-Inducible Factor and Promotes Cancer Cell Survival. Front. Oncol. 2012, 2, 18. [Google Scholar] [CrossRef] [PubMed]
- Curtis, M.; Kenny, H.A.; Ashcroft, B.; Mukherjee, A.; Johnson, A.; Zhang, Y.; Helou, Y.; Batlle, R.; Liu, X.; Gutierrez, N.; et al. Fibroblasts Mobilize Tumor Cell Glycogen to Promote Proliferation and Metastasis. Cell Metab 2019, 29, 141–155. [Google Scholar] [CrossRef] [PubMed]
- Shulman, R.G.; Rothman, D.L. The Glycogen Shunt Maintains Glycolytic Homeostasis and the Warburg Effect in Cancer. Trends Cancer 2017, 3, 761–767. [Google Scholar] [CrossRef]
- Lee, W.N.; Guo, P.; Lim, S.; Bassilian, S.; Lee, S.T.; Boren, J.; Cascante, M.; Go, V.L.; Boros, L.G. Metabolic sensitivity of pancreatic tumour cell apoptosis to glycogen phosphorylase inhibitor treatment. Br. J. Cancer 2004, 91, 2094–2100. [Google Scholar] [CrossRef] [PubMed]
- Favaro, E.; Bensaad, K.; Chong, M.G.; Tennant, D.A.; Ferguson, D.J.; Snell, C.; Steers, G.; Turley, H.; Li, J.L.; Gunther, U.L.; et al. Glucose utilization via glycogen phosphorylase sustains proliferation and prevents premature senescence in cancer cells. Cell Metab. 2012, 16, 751–764. [Google Scholar] [CrossRef]
- Rousset, M.; Zweibaum, A.; Fogh, J. Presence of Glycogen and Growth-related Variations in 58 Cultured Human Tumor Cell Lines of Various Tissue Origins. Cancer Res. 1981, 41, 1165–1170. [Google Scholar]
- Rousset, M.; Chevalier, G.; Rousset, J.-P.; Dussaulx, E.; Zweibaum, A. Presence and Cell Growth-related Variations of Glycogen in Human Colorectal Adenocarcinoma Cell Lines in Culture. Cancer Res. 1979, 39, 531–534. [Google Scholar]
- Staedel, C.; Beck, J.-P. Resurgence of glycogen synthesis and storage capacity in cultured hepatoma cells. Cell Differ. 1978, 7, 61–71. [Google Scholar] [CrossRef]
- Altemus, M.A.; Yates, J.A.; Wu, Z.; Bao, L.; Merajver, S.D. Glycogen accumulation in aggressive breast cancers under hypoxia [abstract]. Mol. Cell. Biol. 2018, 78 (Suppl. 13), 1446. [Google Scholar]
- Sun, R.C.; Fan, T.W.M.; Deng, P.; Higashi, R.M.; Lane, A.N.; Le, A.-T.; Scott, T.L.; Sun, Q.; Warmoes, M.O.; Yang, Y. Noninvasive liquid diet delivery of stable isotopes into mouse models for deep metabolic network tracing. Nat. Commun. 2017, 8, 1646. [Google Scholar] [CrossRef] [PubMed]
- Cheville, J.C.; Lohse, C.M.; Zincke, H.; Weaver, A.L.; Blute, M.L. Comparisons of outcome and prognostic features among histologic subtypes of renal cell carcinoma. Am. J. Surg. Pathol. 2003, 27, 612–624. [Google Scholar] [CrossRef] [PubMed]
- Gadducci, A.; Cosio, S.; Spirito, N.; Cionini, L. Clear cell carcinoma of the endometrium: A biological and clinical enigma. Anticancer Res. 2010, 30, 1327–1334. [Google Scholar] [PubMed]
- Sugiyama, T.; Kamura, T.; Kigawa, J.; Terakawa, N.; Kikuchi, Y.; Kita, T.; Suzuki, M.; Sato, I.; Taguchi, K. Clinical characteristics of clear cell carcinoma of the ovary: A distinct histologic type with poor prognosis and resistance to platinum-based chemotherapy. Cancer 2000, 88, 2584–2589. [Google Scholar] [CrossRef]
- Zhou, Z.; Kinslow, C.J.; Hibshoosh, H.; Guo, H.; Cheng, S.K.; He, C.; Gentry, M.S.; Sun, R.C. Clinical Features, Survival and Prognostic Factors of Glycogen-Rich Clear Cell Carcinoma (GRCC) of the Breast in the U.S. Population. J. Clin. Med. 2019, 8, 246. [Google Scholar] [CrossRef]
- Dow, J.A.; Young, J.D., Jr. Mesonephric adenocarcinoma of the bladder. J. Urol. 1968, 100, 466–469. [Google Scholar] [CrossRef]
- Young, R.H.; Scully, R.E. Clear cell adenocarcinoma of the bladder and urethra. A report of three cases and review of the literature. Am. J. Surg. Pathol. 1985, 9, 816–826. [Google Scholar] [CrossRef]
- Adeniran, A.J.; Tamboli, P. Clear cell adenocarcinoma of the urinary bladder: A short review. Arch. Pathol. Lab. Med. 2009, 133, 987–991. [Google Scholar]
- Venyo, A.K. Primary Clear Cell Carcinoma of the Urinary Bladder. Int. Sch. Res. Not. 2014, 2014, 593826. [Google Scholar] [CrossRef]
- Lu, J.; Xu, Z.; Jiang, F.; Wang, Y.; Hou, Y.; Wang, C.; Chen, Q. Primary clear cell adenocarcinoma of the bladder with recurrence: A case report and literature review. World J. Surg. Oncol. 2012, 10, 33. [Google Scholar] [CrossRef] [PubMed]
- National Cancer Institute Surveillance. Epidemiology and End Result Program. Overview of the SEER Program. Available online: https://seer.cancer.gov/about/overview.html (accessed on 19 June 2016).
- National Cancer Institute. DCCPS, Surveillance Research Program. Surveillance, Epidemiology, and End Results (SEER) Program (www.seer.cancer.gov). SEER*Stat Database: Incidence—SEER 18 Regs Research Data + Hurricane Katrina Impacted Louisiana Cases, Nov 2017 Sub (2000–2015) <Katrina/Rita Population Adjustment>—Linked To County Attributes—Total U.S., 1969–2016 Counties, released April 2018, based on the November 2017 Submission.
- National Cancer Institute. DCCPS, Surveillance Research Program. Surveillance, Epidemiology, and End Results (SEER) Program (www.seer.cancer.gov) SEER*Stat Database: Incidence-Based Mortality—SEER 18 Regs (Excl Louisiana) Research Data, Nov 2017 Sub (2000–2015) <Katrina/Rita Population Adjustment>—Linked To County Attributes—Total U.S., 1969–2016 Counties, Released April 2018, based on the November 2017 Submission.
- Kinslow, C.J.; Bruce, S.S.; Rae, A.I.; Sheth, S.A.; McKhann, G.M.; Sisti, M.B.; Bruce, J.N.; Sonabend, A.M.; Wang, T.J.C. Solitary-fibrous tumor/hemangiopericytoma of the central nervous system: A population-based study. J. Neurooncol. 2018, 138, 173–182. [Google Scholar] [CrossRef] [PubMed]
- National Cancer Institute. DCCPS, Surveillance Research Program. Surveillance, Epidemiology, and End Results (SEER) Program (www.seer.cancer.gov) SEER*Stat Database: Incidence—SEER 18 Regs Research Data + Hurricane Katrina Impacted Louisiana Cases, Nov 2015 Sub (1973–2013 Varying)—Linked To County Attributes—Total U.S., 1969–2014 Counties, released April 2016, based on the November 2015 Submission.
- National Cancer Institute. DCCPS, Surveillance Research Program. Surveillance, Epidemiology, and End Results (SEER) Program (www.seer.cancer.gov) SEER*Stat Database: Incidence—SEER 18 Regs Research Data + Hurricane Katrina Impacted Louisiana Cases, Nov 2017 Sub (1973–2015 Varying)—Linked To County Attributes—Total U.S., 1969–2016 Counties, released April 2018, based on the November 2017 Submission.
- Matsuoka, Y.; Machida, T.; Oka, K.; Ishizaka, K. Clear cell adenocarcinoma of the urinary bladder inducing acute renal failure. Int. J. Urol. 2002, 9, 467–469. [Google Scholar] [CrossRef] [PubMed]
- Honda, N.; Yamada, Y.; Nanaura, H.; Fukatsu, H.; Nonomura, H.; Hatano, Y. Mesonephric adenocarcinoma of the urinary bladder: A case report. Hinyokika Kiyo 2000, 46, 27–31. [Google Scholar] [PubMed]
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines). Bladder Cancer. Version 1.2019. Available online: https://www.partnershipagainstcancer.ca/db-sage/sage20181257/# (accessed on 19 June 2018).
- Warburg, O. On the origin of cancer cells. Science 1956, 123, 309–314. [Google Scholar] [CrossRef]
- Guin, S.; Pollard, C.; Ru, Y.; Ritterson Lew, C.; Duex, J.E.; Dancik, G.; Owens, C.; Spencer, A.; Knight, S.; Holemon, H.; et al. Role in tumor growth of a glycogen debranching enzyme lost in glycogen storage disease. J. Natl. Cancer Inst. 2014, 106. [Google Scholar] [CrossRef]
- Dobruch, J.; Daneshmand, S.; Fisch, M.; Lotan, Y.; Noon, A.P.; Resnick, M.J.; Shariat, S.F.; Zlotta, A.R.; Boorjian, S.A. Gender and Bladder Cancer: A Collaborative Review of Etiology, Biology, and Outcomes. Eur. Urol. 2016, 69, 300–310. [Google Scholar] [CrossRef]
- Drew, P.A.; Murphy, W.M.; Civantos, F.; Speights, V.O. The histogenesis of clear cell adenocarcinoma of the lower urinary tract. Case series and review of the literature. Hum. Pathol. 1996, 27, 248–252. [Google Scholar] [CrossRef]
- Cohn, J.A.; Vekhter, B.; Lyttle, C.; Steinberg, G.D.; Large, M.C. Sex disparities in diagnosis of bladder cancer after initial presentation with hematuria: A nationwide claims-based investigation. Cancer 2014, 120, 555–561. [Google Scholar] [CrossRef]

| Clear Cell Adenocarcinoma (N = 91) | Non-Clear Cell Adenocarcinoma (N = 205,106) | |||||
|---|---|---|---|---|---|---|
| Count | % | Count | % | p-Value | ||
| Age | 0–60 | 27 | 29.7 | 37,649 | 18.4 | 0.005 |
| 61+ | 64 | 70.3 | 167,457 | 81.6 | ||
| Sex | Female | 54 | 59.3 | 49,241 | 24.0 | <0.001 |
| Male | 37 | 40.7 | 155,865 | 76.0 | ||
| Race | White | 77 | 84.6 | 182,492 | 89.0 | 0.001 |
| Black | 13 | 14.3 | 11,519 | 5.6 | ||
| Other | 1 | 1.1 | 8588 | 4.2 | ||
| Unknown | 0 | 0.0 | 2507 | 1.2 | ||
| Tumor primary site | Trigone of bladder | 9 | 9.9 | 12,765 | 6.2 | <0.001 |
| Dome of bladder | 4 | 4.4 | 7213 | 3.5 | ||
| Lateral wall of bladder | 6 | 6.6 | 41,041 | 20.0 | ||
| Anterior wall of bladder | 5 | 5.5 | 4334 | 2.1 | ||
| Posterior wall of bladder | 7 | 7.7 | 18,819 | 9.2 | ||
| Bladder neck | 10 | 11.0 | 6354 | 3.1 | ||
| Ureteric orifice | 4 | 4.4 | 7820 | 3.8 | ||
| Urachus | 1 | 1.1 | 310 | 0.2 | ||
| Overlapping lesion of bladder | 13 | 14.3 | 21,112 | 10.3 | ||
| Bladder, NOS | 32 | 35.2 | 85,338 | 41.6 | ||
| Grade | Grade I | 0 | 0.0 | 23,684 | 11.5 | <0.001 |
| Grade II | 5 | 5.5 | 48,123 | 23.5 | ||
| Grade III | 22 | 24.2 | 35,849 | 17.5 | ||
| Grade IV | 25 | 27.5 | 59,477 | 29.0 | ||
| Unknown | 39 | 42.9 | 37,973 | 18.5 | ||
| AJCC 6th stage | Stage 0 | 2 | 2.2 | 105,545 | 51.5 | <0.001 |
| Stage 1 | 22 | 24.2 | 46,332 | 22.6 | ||
| Stage 2 | 28 | 30.8 | 23,463 | 11.4 | ||
| Stage 3 | 9 | 9.9 | 8157 | 4.0 | ||
| Stage 4 | 17 | 18.7 | 14,012 | 6.8 | ||
| Unknown | 13 | 14.3 | 7597 | 3.7 | ||
| T stage | Tis/Ta | 2 | 2.2 | 105,545 | 51.5 | <0.001 |
| T0 | 0 | 0.0 | 91 | 0.0 | ||
| T1 | 26 | 28.6 | 49,221 | 24.0 | ||
| T2 | 35 | 38.5 | 28,776 | 14.0 | ||
| T3 | 7 | 7.7 | 8046 | 3.9 | ||
| T4 | 11 | 12.1 | 7713 | 3.8 | ||
| Unknown | 10 | 11.0 | 5714 | 2.8 | ||
| N stage | N0 | 70 | 76.9 | 189,973 | 92.6 | <0.001 |
| N1 | 2 | 2.2 | 3994 | 1.9 | ||
| N2 | 6 | 6.6 | 3806 | 1.9 | ||
| N3 | 1 | 1.1 | 166 | 0.1 | ||
| Unknown | 12 | 13.2 | 7167 | 3.5 | ||
| M stage | M0 | 75 | 82.4 | 193,071 | 94.1 | <0.001 |
| M1 | 11 | 12.1 | 7565 | 3.7 | ||
| Unknown | 5 | 5.5 | 4470 | 2.2 | ||
| Bone metastasis a | No | 42 | 97.7 | 102,083 | 97.0 | 0.697 |
| Yes | 0 | 0.0 | 1432 | 1.4 | ||
| Unknown | 1 | 2.3 | 1698 | 1.6 | ||
| Brain metastasis a | No | 41 | 95.3 | 103,393 | 98.3 | <0.001 |
| Yes | 1 | 2.3 | 122 | 0.1 | ||
| Unknown | 1 | 2.3 | 1698 | 1.6 | ||
| Liver metastasisa | No | 40 | 93.0 | 102,600 | 97.5 | 0.028 |
| Yes | 2 | 4.7 | 926 | 0.9 | ||
| Unknown | 1 | 2.3 | 1687 | 1.6 | ||
| Lung metastasis a | No | 41 | 95.3 | 102,153 | 97.1 | 0.771 |
| Yes | 1 | 2.3 | 1327 | 1.3 | ||
| Unknown | 1 | 2.3 | 1733 | 1.6 | ||
| Type of surgical procedure | No surgery | 8 | 8.8 | 15,265 | 7.4 | <0.001 |
| Local procedure/partial cystectomy | 60 | 65.9 | 170,325 | 83.0 | ||
| Complete cystectomy | 22 | 24.2 | 18,327 | 8.9 | ||
| Surgery NOS | 0 | 0.0 | 504 | 0.2 | ||
| Unknown if surgery performed | 1 | 1.1 | 685 | 0.3 | ||
| Type of radiation b | None | 67 | 85.9 | 160,440 | 94.6 | <0.001 |
| Beam radiation | 9 | 11.5 | 7485 | 4.4 | ||
| Other radiation | 1 | 1.3 | 219 | 0.1 | ||
| Unknown if radiation received | 1 | 1.3 | 1389 | 0.8 | ||
| Median Survival | 95% Confidence Interval | ||||
|---|---|---|---|---|---|
| Lower Bound | Upper Bound | p-Value | |||
| Non-muscle invasive (Stage 0–1) | Non-CCA | 119 | 0.654 | ||
| CCA | |||||
| Muscle invasive (Stage 2–3) | Non-CCA | 25 | 24.263 | 25.737 | 0.653 |
| CCA | 32 | 16.308 | 47.692 | ||
| Metastatic (Stage 4) | Non-CCA | 9 | 8.736 | 9.264 | 0.091 |
| CCA | 18 | 13.315 | 22.685 | ||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhou, Z.; Kinslow, C.J.; Wang, P.; Huang, B.; Cheng, S.K.; Deutsch, I.; Gentry, M.S.; Sun, R.C. Clear Cell Adenocarcinoma of the Urinary Bladder Is a Glycogen-Rich Tumor with Poorer Prognosis. J. Clin. Med. 2020, 9, 138. https://doi.org/10.3390/jcm9010138
Zhou Z, Kinslow CJ, Wang P, Huang B, Cheng SK, Deutsch I, Gentry MS, Sun RC. Clear Cell Adenocarcinoma of the Urinary Bladder Is a Glycogen-Rich Tumor with Poorer Prognosis. Journal of Clinical Medicine. 2020; 9(1):138. https://doi.org/10.3390/jcm9010138
Chicago/Turabian StyleZhou, Zhengqiu, Connor J. Kinslow, Peng Wang, Bin Huang, Simon K. Cheng, Israel Deutsch, Matthew S. Gentry, and Ramon C. Sun. 2020. "Clear Cell Adenocarcinoma of the Urinary Bladder Is a Glycogen-Rich Tumor with Poorer Prognosis" Journal of Clinical Medicine 9, no. 1: 138. https://doi.org/10.3390/jcm9010138
APA StyleZhou, Z., Kinslow, C. J., Wang, P., Huang, B., Cheng, S. K., Deutsch, I., Gentry, M. S., & Sun, R. C. (2020). Clear Cell Adenocarcinoma of the Urinary Bladder Is a Glycogen-Rich Tumor with Poorer Prognosis. Journal of Clinical Medicine, 9(1), 138. https://doi.org/10.3390/jcm9010138





