Bone Regeneration, Reconstruction and Use of Osteogenic Cells; from Basic Knowledge, Animal Models to Clinical Trials
Abstract
1. Introduction
2. Section I: Current State of Knowledge on Bone Regeneration—Tissue and Cell Specific Mechanisms
2.1. The Sources, Differentiation Pathways and Stemness Potency of Tissue-Derived Osteogenic Cells
2.2. The Aging of the Skeleton
3. Section II: Therapies
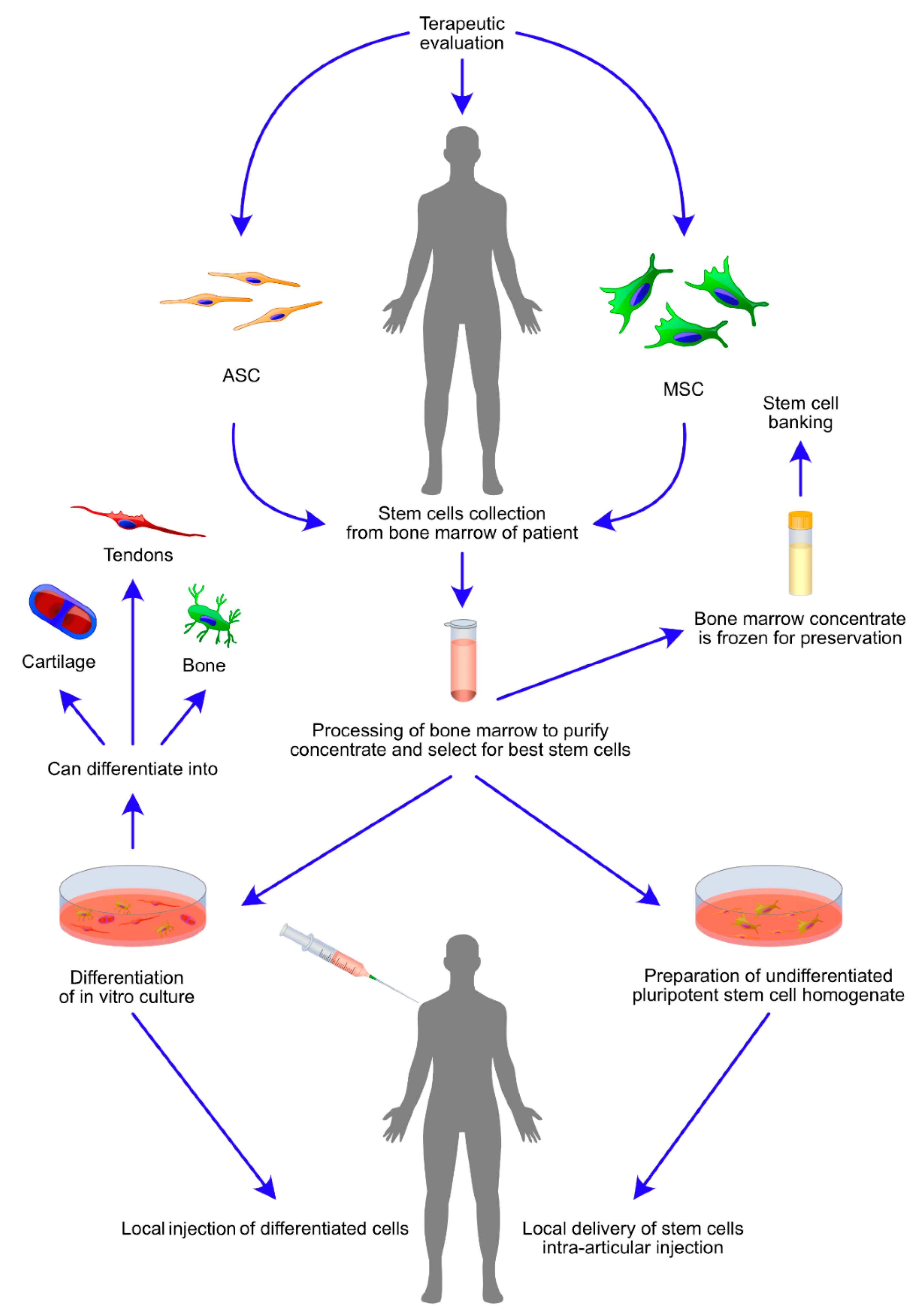
3.1. MSC- and ASC-Based Cell Therapies
3.2. Advances in Bone Reconstruction Strategies
3.2.1. Natural Grafts
3.2.2. Conventionally Fabricated Scaffolds
3.2.3. Three-Dimensionally Printing Scaffolds
3.2.4. CV-TEBG and Nanotechnology-Related Scaffolds
3.2.5. Investigating New Therapeutic Targets
Secretory Proteins
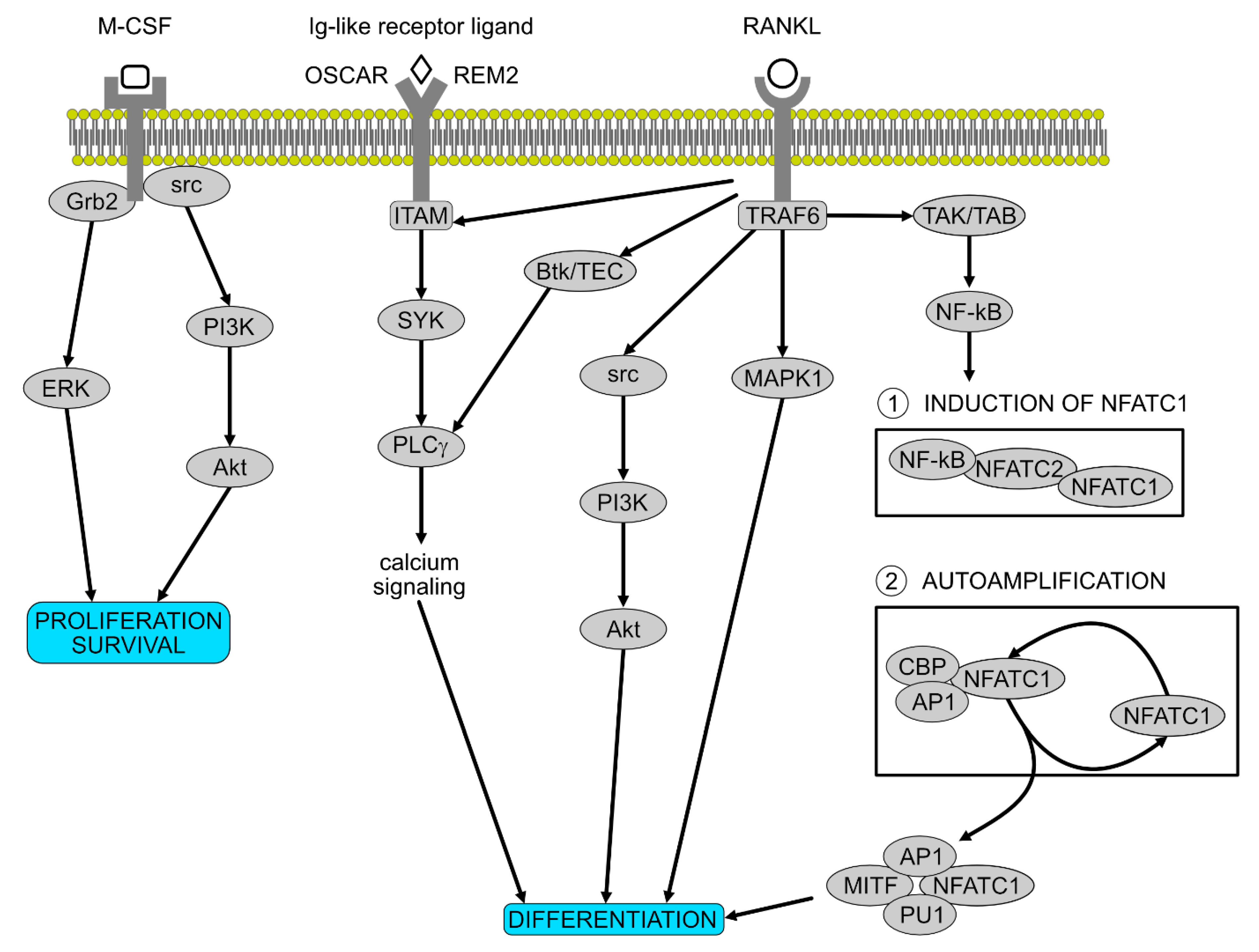
Signalling Pathways
4. Section III: Application of Osteogenic Stem Cells in Animal Model Research and Human Clinical Trials
4.1. Direct Administration of ASCs and MSCs
4.2. Bone Marrow Mononuclear Cells and Bone Marrow Concentrate
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
References
- Tang, Y.; Wu, X.; Lei, W.; Pang, L.; Wan, C.; Shi, Z.; Zhao, L.; Nagy, T.R.; Peng, X.; Hu, J.; et al. TGF-Β1-induced migration of bone mesenchymal stem cells couples bone resorption with formation. Nat. Med. 2009, 15, 757–765. [Google Scholar] [CrossRef] [PubMed]
- Paspaliaris, V.; Kolios, G. Stem cells in Osteoporosis: From Biology to New Therapeutic Approaches. Stem Cells Int. 2019, 2019, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Gómez, S.; Vlad, M.D.; López, J.; Fernández, E. Design and properties of 3D scaffolds for bone tissue engineering. Acta Biomater. 2016, 42, 341–350. [Google Scholar] [CrossRef] [PubMed]
- Sanders, R. Bone graft substitutes: Separating fact from fiction. J. Bone Joint Surg. Am. 2007, 89, 469. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z. Bone regeneration by stem cell and tissue engineering in oral and maxillofacial region. Front. Med. China 2011, 5, 401–413. [Google Scholar] [CrossRef] [PubMed]
- De Witte, T.-M.; Fratila-Apachitei, L.E.; Zadpoor, A.A.; Peppas, N.A. Bone tissue engineering via growth factor delivery: From scaffolds to complex matrices. Regen. Biomater. 2018, 5, 197–211. [Google Scholar] [CrossRef]
- Wang, L.; Zhu, L.X.; Wang, Z.; Lou, A.J.; Yang, Y.X.; Guo, Y.; Liu, S.; Zhang, C.; Zhang, Z.; Hu, H.S.; et al. Development of a centrally vascularized tissue engineering bone graft with the unique core-shell composite structure for large femoral bone defect treatment. Biomaterials 2018, 175, 44–60. [Google Scholar] [CrossRef]
- Lee, M.H.; Javed, A.; Kim, H.J.; Shin, H.I.; Gutierrez, S.; Choi, J.Y.; Rosen, V.; Stein, J.L.; van Wijnen, A.J.; Stein, G.S.; et al. Transient upregulation of CBFA1 in response to bone morphogenetic protein-2 and transforming growth factor beta1 in C2C12 myogenic cells coincides with suppression of the myogenic phenotype but is not sufficient for osteoblast differentiation. J. Cell. Biochem. 1999, 73, 114–125. [Google Scholar] [CrossRef]
- Yoshida, C.A.; Komori, H.; Maruyama, Z.; Miyazaki, T.; Kawasaki, K.; Furuichi, T.; Fukuyama, R.; Mori, M.; Yamana, K.; Nakamura, K.; et al. Sp7 inhibits osteoblast differentiation at a late stage in mice. PLoS ONE 2012, 7. [Google Scholar] [CrossRef]
- Zhang, W.; Shen, X.; Wan, C.; Zhao, Q.; Zhang, L.; Zhou, Q.; Deng, L. Effects of insulin and insulin-like growth factor 1 on osteoblast proliferation and differentiation: Differential signalling via Akt and ERK. Cell Biochem. Funct. 2012, 30, 297–302. [Google Scholar] [CrossRef]
- Celil, A.B.; Campbell, P.G. BMP-2 and insulin-like growth factor-I mediate osterix (Osx) expression in human mesenchymal stem cells via the MAPK and protein kinase D signaling pathways. J. Biol. Chem. 2005, 280, 31353–31359. [Google Scholar] [CrossRef] [PubMed]
- Leucht, P.; Lee, S.; Yim, N. Wnt signaling and bone regeneration: Can’t have one without the other. Biomaterials 2019, 196, 46–50. [Google Scholar] [CrossRef] [PubMed]
- Liang, H.; Xu, X.; Feng, X.; Ma, L.; Deng, X.; Wu, S.; Liu, X.; Yang, C. Gold nanoparticles-loaded hydroxyapatite composites guide osteogenic differentiation of human mesenchymal stem cells through Wnt/β-catenin signaling pathway. Int. J. Nanomed. 2019, 14, 6151–6163. [Google Scholar] [CrossRef] [PubMed]
- Perez, J.R.; Kouroupis, D.; Li, D.J.; Best, T.M.; Kaplan, L.; Correa, D. Tissue Engineering and Cell-Based Therapies for Fractures and Bone Defects. Front. Bioeng. Biotechnol. 2018, 6, 105. [Google Scholar] [CrossRef] [PubMed]
- Alfotawi, R.; Elsafadi, M.; Muthurangan, M.; Siyal, A.-A.; Alfayez, M.; Mahmmod, A.A. A New Procedure in Bone Engineering Using Induced Adipose Tissue. J. Investig. Surg. 2019, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Xu, T.; Yu, X.; Yang, Q.; Liu, X.; Fang, J.; Dai, X. Autologous Micro-Fragmented Adipose Tissue as Stem Cell-Based Natural Scaffold for Cartilage Defect Repair. Cell Transplant. 2019, 28, 1709–1720. [Google Scholar] [CrossRef]
- Kohno, Y.; Lin, T.; Pajarinen, J.; Romero-Lopez, M.; Maruyama, M.; Huang, J.F.; Nathan, K.; Yao, Z.; Goodman, S.B. Osteogenic ability of rat bone marrow concentrate is at least as efficacious as mesenchymal stem cells in vitro. J. Biomed. Mater. Res. Part B Appl. Biomater. 2019, 107, 2500–2506. [Google Scholar] [CrossRef]
- Stein, G.S.; Lian, J.B.; van Wijnen, A.J.; Stein, J.L.; Montecino, M.; Javed, A.; Zaidi, S.K.; Young, D.W.; Choi, J.-Y.; Pockwinse, S.M. Runx2 control of organization, assembly and activity of the regulatory machinery for skeletal gene expression. Oncogene 2004, 23, 4315–4329. [Google Scholar] [CrossRef]
- Sims, N.A.; Martin, T.J. Osteoclasts Provide Coupling Signals to Osteoblast Lineage Cells Through Multiple Mechanisms. Annu. Rev. Physiol. 2020, 82, 1–23. [Google Scholar] [CrossRef]
- Kegelman, C.D.; Coulombe, K.M.; Horan, D.J.; Qin, L.; Robling, A.G.; Ferguson, V.L.; Bellido, T.M.; Boerckel, J.D. Osteocytes Remodel Bone by TGF-β-induced YAP/TAZ Signaling. Available online: https://www.biorxiv.org/content/10.1101/611913v1 (accessed on 11 December 2019).
- Valenti, M.T.; Carbonare, L.D.; Mottes, M. Role of microRNAs in progenitor cell commitment and osteogenic differentiation in health and disease (Review). Int. J. Mol. Med. 2018, 41, 2441–2449. [Google Scholar] [CrossRef]
- Baker, N.; Boyette, L.B.; Tuan, R.S. Characterization of bone marrow-derived mesenchymal stem cells in aging. Bone 2015, 70, 37–47. [Google Scholar] [CrossRef] [PubMed]
- Long, M.W.; Robinson, J.A.; Ashcraft, E.A.; Mann, K.G. Regulation of human bone marrow-derived osteoprogenitor cells by osteogenic growth factors. J. Clin. Investig. 1995, 95, 881–887. [Google Scholar] [CrossRef] [PubMed]
- Eghbali-Fatourechi, G.Z.; Lamsam, J.; Fraser, D.; Nagel, D.; Riggs, B.L.; Khosla, S. Circulating osteoblast-lineage cells in humans. N. Engl. J. Med. 2005, 352, 1959–1966. [Google Scholar] [CrossRef] [PubMed]
- Álvarez-Viejo, M. CD271 as a marker to identify mesenchymal stem cells from diverse sources before culture. World J. Stem Cells 2015, 7, 470. [Google Scholar] [CrossRef]
- Busser, H.; Najar, M.; Raicevic, G.; Pieters, K.; Velez Pombo, R.; Philippart, P.; Meuleman, N.; Bron, D.; Lagneaux, L. Isolation and Characterization of Human Mesenchymal Stromal Cell Subpopulations: Comparison of Bone Marrow and Adipose Tissue. Stem Cells Dev. 2015, 24, 2142–2157. [Google Scholar] [CrossRef] [PubMed]
- Chava, S.; Chennakesavulu, S.; Gayatri, M.B.; Reddy, A.B.M. A novel phosphorylation by AMP-activated kinase regulates RUNX2 from ubiquitination in osteogenesis over adipogenesis article. Cell Death Dis. 2018, 9, 754. [Google Scholar] [CrossRef] [PubMed]
- Bruderer, M.; Richards, R.G.; Alini, M.; Stoddart, M.J. Role and regulation of RUNX2 in osteogenesis. Eur. Cell. Mater. 2014, 28, 269–286. [Google Scholar] [CrossRef]
- Liu, W.; Toyosawa, S.; Furuichi, T.; Kanatani, N.; Yoshida, C.; Liu, Y.; Himeno, M.; Narai, S.; Yamaguchi, A.; Komori, T. Overexpression of Cbfa1 in osteoblasts inhibits osteoblast maturation and causes osteopenia with multiple fractures. J. Cell Biol. 2001, 155, 157–166. [Google Scholar] [CrossRef]
- Cohen-Solal, K.A.; Boregowda, R.K.; Lasfar, A. RUNX2 and the PI3K/AKT axis reciprocal activation as a driving force for tumor progression. Mol. Cancer 2015, 14, 137. [Google Scholar] [CrossRef]
- Nakashima, K.; Zhou, X.; Kunkel, G.; Zhang, Z.; Deng, J.M.; Behringer, R.R.; De Crombrugghe, B. The novel zinc finger-containing transcription factor Osterix is required for osteoblast differentiation and bone formation. Cell 2002, 108, 17–29. [Google Scholar] [CrossRef]
- Nishio, Y.; Dong, Y.; Paris, M.; O’Keefe, R.J.; Schwarz, E.M.; Drissi, H. Runx2-mediated regulation of the zinc finger Osterix/Sp7 gene. Gene 2006, 372, 62–70. [Google Scholar] [CrossRef] [PubMed]
- Lv, T.; Wu, Y.; Mu, C.; Liu, G.; Yan, M.; Xu, X.; Wu, H.; Du, J.; Yu, J.; Mu, J. Insulin-like growth factor 1 promotes the proliferation and committed differentiation of human dental pulp stem cells through MAPK pathways. Arch. Oral Biol. 2016, 72, 116–123. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.; Shimizu, E.; Zhang, X.; Partridge, N.C.; Qin, L. EGFR signaling suppresses osteoblast differentiation and inhibits expression of master osteoblastic transcription factors Runx2 and Osterix. J. Cell. Biochem. 2011, 112, 1749–1760. [Google Scholar] [CrossRef] [PubMed]
- Razidlo, D.F.; Whitney, T.J.; Casper, M.E.; McGee-Lawrence, M.E.; Stensgard, B.A.; Li, X.; Secreto, F.J.; Knutson, S.K.; Hiebert, S.W.; Westendorf, J.J. Histone deacetylase 3 depletion in osteo/chondroprogenitor cells decreases bone density and increases marrow fat. PLoS ONE 2010, 5, e11492. [Google Scholar] [CrossRef]
- Lee, H.W.; Suh, J.H.; Kim, A.Y.; Lee, Y.S.; Park, S.Y.; Kim, J.B. Histone deacetylase 1-mediated histone modification regulates osteoblast differentiation. Mol. Endocrinol. 2006, 20, 2432–2443. [Google Scholar] [CrossRef]
- Jensen, E.D.; Schroeder, T.M.; Bailey, J.; Gopalakrishnan, R.; Westendorf, J.J. Histone deacetylase 7 associates with Runx2 and represses its activity during osteoblast maturation in a deacetylation-independent manner. J. Bone Miner. Res. 2008, 23, 361–372. [Google Scholar] [CrossRef]
- Lu, X.; Beck, G.R.; Gilbert, L.C.; Camalier, C.E.; Bateman, N.W.; Hood, B.L.; Conrads, T.P.; Kern, M.J.; You, S.; Chen, H.; et al. Identification of the homeobox protein Prx1 (MHox, Prrx-1) as a regulator of osterix expression and mediator of tumor necrosis factor α action in osteoblast differentiation. J. Bone Miner. Res. 2011, 26, 209–219. [Google Scholar] [CrossRef]
- Nishimura, R.; Wakabayashi, M.; Hata, K.; Matsubara, T.; Honma, S.; Wakisaka, S.; Kiyonari, H.; Shioi, G.; Yamaguchi, A.; Tsumaki, N.; et al. Osterix regulates calcification and degradation of chondrogenic matrices through matrix metalloproteinase 13 (MMP13) expression in association with transcription factor Runx2 during endochondral ossification. J. Biol. Chem. 2012, 287, 33179–33190. [Google Scholar] [CrossRef]
- Gao, M.M.; Su, Q.N.; Liang, T.Z.; Ma, J.X.; Stoddart, M.J.; Richards, R.G.; Zhou, Z.Y.; Zou, X.N. Transcriptional activation of enpp1 by osterix in osteoblasts and osteocytes. Eur. Cells Mater. 2018, 36, 1–14. [Google Scholar] [CrossRef]
- Pérez-Campo, F.M.; Santurtún, A.; García-Ibarbia, C.; Pascual, M.A.; Valero, C.; Garcés, C.; Sañudo, C.; Zarrabeitia, M.T.; Riancho, J.A. Osterix and RUNX2 are Transcriptional Regulators of Sclerostin in Human Bone. Calcif. Tissue Int. 2016, 99, 302–309. [Google Scholar] [CrossRef]
- Miyama, K.; Yamada, G.; Yamamoto, T.S.; Takagi, C.; Miyado, K.; Sakai, M.; Ueno, N.; Shibuya, H. A BMP-inducible gene, dlx5, regulates osteoblast differentiation and mesoderm induction. Dev. Biol. 1999, 208, 123–133. [Google Scholar] [CrossRef] [PubMed]
- Acampora, D.; Merlo, G.R.; Paleari, L.; Zerega, B.; Postiglione, M.P.; Mantero, S.; Bober, E.; Barbieri, O.; Simeone, A.; Levi, G. Craniofacial, vestibular and bone defects in mice lacking the Distal-less-related gene Dlx5. Development 1999, 126, 3795–3809. [Google Scholar] [PubMed]
- Hassan, M.Q.; Tare, R.S.; Suk, H.L.; Mandeville, M.; Morasso, M.I.; Javed, A.; Van Wijnen, A.J.; Stein, J.L.; Stein, G.S.; Lian, J.B. BMP2 commitment to the osteogenic lineage involves activation of Runx2 by DLX3 and a homeodomain transcriptional network. J. Biol. Chem. 2006, 281, 40515–40526. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.H.; Kim, Y.J.; Yoon, W.J.; Kim, J.I.; Kim, B.G.; Hwang, Y.S.; Wozney, J.M.; Chi, X.Z.; Bae, S.C.; Choi, K.Y.; et al. Dlx5 specifically regulates Runx2 type II expression by binding to homeodomain-response elements in the Runx2 distal promoter. J. Biol. Chem. 2005, 280, 35579–35587. [Google Scholar] [CrossRef] [PubMed]
- Heo, J.S.; Lee, S.G.; Kim, H.O. Distal-less homeobox 5 is a master regulator of the osteogenesis of human mesenchymal stem cells. Int. J. Mol. Med. 2017, 40, 1486–1494. [Google Scholar] [CrossRef]
- Lee, M.H.; Kwon, T.G.; Park, H.S.; Wozney, J.M.; Ryoo, H.M. BMP-2-induced Osterix expression is mediated by Dlx5 but is independent of Runx2. Biochem. Biophys. Res. Commun. 2003, 309, 689–694. [Google Scholar] [CrossRef]
- Zhao, G.Q.; Zhao, S.; Zhou, X.; Eberspaecher, H.; Solursh, M.; de Crombrugghe, B. rDlx, a novel distal-less-like homeoprotein is expressed in developing cartilages and discrete neuronal tissues. Dev. Biol. 1994, 164, 37–51. [Google Scholar] [CrossRef]
- Holleville, N.; Quilhac, A.; Bontoux, M.; Monsoro-Burq, A.H. BMP signals regulate Dlx5 during early avian skull development. Dev. Biol. 2003, 257, 177–189. [Google Scholar] [CrossRef]
- Lee, M.-H.; Kim, Y.-J.; Kim, H.-J.; Park, H.-D.; Kang, A.-R.; Kyung, H.-M.; Sung, J.-H.; Wozney, J.M.; Kim, H.-J.; Ryoo, H.-M. BMP-2-induced Runx2 expression is mediated by Dlx5, and TGF-beta 1 opposes the BMP-2-induced osteoblast differentiation by suppression of Dlx5 expression. J. Biol. Chem. 2003, 278, 34387–34394. [Google Scholar] [CrossRef]
- McCabe, L.R.; Banerjee, C.; Kundu, R.; Harrison, R.J.; Dobner, P.R.; Stein, J.L.; Lian, J.B.; Stein, G.S. Developmental expression and activities of specific fos and jun proteins are functionally related to osteoblast maturation: Role of Fra-2 and Jun D during differentiation. Endocrinology 1996, 137, 4398–4408. [Google Scholar] [CrossRef]
- Grigoriadis, A.E.; Schellander, K.; Wang, Z.Q.; Wagner, E.F. Osteoblasts are target cells for transformation in c-fos transgenic mice. J. Cell Biol. 1993, 122, 685–701. [Google Scholar] [CrossRef] [PubMed]
- Sabatakos, G.; Sims, N.A.; Chen, J.; Aoki, K.; Kelz, M.B.; Amling, M.; Bouali, Y.; Mukhopadhyay, K.; Ford, K.; Nestler, E.J.; et al. Overexpression of DeltaFosB transcription factor(s) increases bone formation and inhibits adipogenesis. Nat. Med. 2000, 6, 985–990. [Google Scholar] [CrossRef] [PubMed]
- Jochum, W.; David, J.P.; Elliott, C.; Wutz, A.; Plenk, H.; Matsuo, K.; Wagner, E.F. Increased bone formation and osteosclerosis in mice overexpressing the transcription factor Fra-1. Nat. Med. 2000, 6, 980–984. [Google Scholar] [CrossRef]
- Suda, T.; Takahashi, N.; Martin, T.J. Modulation of osteoclast differentiation. Endocr. Rev. 1992, 13, 66–80. [Google Scholar] [CrossRef]
- Tanaka, S.; Takahashi, N.; Udagawa, N.; Tamura, T.; Akatsu, T.; Stanley, E.R.; Kurokawa, T.; Suda, T. Macrophage colony-stimulating factor is indispensable for both proliferation and differentiation of osteoclast progenitors. J. Clin. Investig. 1993, 91, 257–263. [Google Scholar] [CrossRef]
- Suda, T.; Takahashi, N.; Udagawa, N.; Jimi, E.; Gillespie, M.T.; Martin, T.J. Modulation of osteoclast differentiation and function by the new members of the tumor necrosis factor receptor and ligand families. Endocr. Rev. 1999, 20, 345–357. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, N.; Kadono, Y.; Naito, A.; Matsumoto, K.; Yamamoto, T.; Tanaka, S.; Inoue, J.I. Segregation of TRAF6-mediated signaling pathways clarifies its role in osteoclastogenesis. EMBO J. 2001, 20, 1271–1280. [Google Scholar] [CrossRef]
- Johnson, R.S.; Spiegelman, B.M.; Papaioannou, V. Pleiotropic effects of a null mutation in the c-fos proto-oncogene. Cell 1992, 71, 577–586. [Google Scholar] [CrossRef]
- Grigoriadis, A.E.; Wang, Z.Q.; Cecchini, M.G.; Hofstetter, W.; Felix, R.; Fleisch, H.A.; Wagner, E.F. c-Fos: A key regulator of osteoclast-macrophage lineage determination and bone remodeling. Science 1994, 266, 443–448. [Google Scholar] [CrossRef]
- Yamashita, T.; Yao, Z.; Li, F.; Zhang, Q.; Badell, I.R.; Schwarz, E.M.; Takeshita, S.; Wagner, E.F.; Noda, M.; Matsuo, K.; et al. NF-κB p50 and p52 regulate receptor activator of NF-κB ligand (RANKL) and tumor necrosis factor-induced osteoclast precursor differentiation by activating c-Fos and NFATc1. J. Biol. Chem. 2007, 282, 18245–18253. [Google Scholar] [CrossRef]
- Asagiri, M.; Sato, K.; Usami, T.; Ochi, S.; Nishina, H.; Yoshida, H.; Morita, I.; Wagner, E.F.; Mak, T.W.; Serfling, E.; et al. Autoamplification of NFATc1 expression determines its essential role in bone homeostasis. J. Exp. Med. 2005, 202, 1261–1269. [Google Scholar] [CrossRef] [PubMed]
- Mócsai, A.; Humphrey, M.B.; Van Ziffle, J.A.G.; Hu, Y.; Burghardt, A.; Spusta, S.C.; Majumdar, S.; Lanier, L.L.; Lowell, C.A.; Nakamura, M.C. The immunomodulatory adapter proteins DAP12 and Fc receptor gamma-chain (FcRgamma) regulate development of functional osteoclasts through the Syk tyrosine kinase. Proc. Natl. Acad. Sci. USA 2004, 101, 6158–6163. [Google Scholar] [CrossRef] [PubMed]
- Theill, L.E.; Boyle, W.J.; Penninger, J.M. RANK-L and RANK: T cells, bone loss, and mammalian evolution. Annu. Rev. Immunol. 2002, 20, 795–823. [Google Scholar] [CrossRef] [PubMed]
- Koga, T.; Inui, M.; Inoue, K.; Kim, S.; Suematsu, A.; Kobayashi, E.; Iwata, T.; Ohnishi, H.; Matozaki, T.; Kodama, T.; et al. Costimulatory signals mediated by the ITAM motif cooperate with RANKL for bone homeostasis. Nature 2004, 428, 758–763. [Google Scholar] [CrossRef] [PubMed]
- Darnay, B.G.; Ni, J.; Moore, P.A.; Aggarwal, B.B. Activation of NF-κB by rank requires tumor necrosis factor receptor- associated factor (TRAF) 6 and NF-κB-inducing kinase. Identification of a novel TRAF6 interaction motif. J. Biol. Chem. 1999, 274, 7724–7731. [Google Scholar] [CrossRef] [PubMed]
- Gohda, J.; Akiyama, T.; Koga, T.; Takayanagi, H.; Tanaka, S.; Inoue, J. RANK-mediated amplification of TRAF6 signaling leads to NFATc1 induction during osteoclastogenesis. EMBO J. 2005, 24, 790–799. [Google Scholar] [CrossRef]
- Von Heimburg, D.; Hemmrich, K.; Haydarlioglu, S.; Staiger, H.; Pallua, N. Comparison of viable cell yield from excised versus aspirated adipose tissue. Cells Tissues Organs 2004, 178, 87–92. [Google Scholar] [CrossRef]
- Mizukami, J.; Takaesu, G.; Akatsuka, H.; Sakurai, H.; Ninomiya-Tsuji, J.; Matsumoto, K.; Sakurai, N. Receptor activator of NF-kappaB ligand (RANKL) activates TAK1 mitogen-activated protein kinase kinase kinase through a signaling complex containing RANK, TAB2, and TRAF6. Mol. Cell. Biol. 2002, 22, 992–1000. [Google Scholar] [CrossRef]
- Takatsuna, H.; Asagiri, M.; Kubota, T.; Oka, K.; Osada, T.; Sugiyama, C.; Saito, H.; Aoki, K.; Ohya, K.; Takayanagi, H.; et al. Inhibition of RANKL-induced osteoclastogenesis by (-)-DHMEQ, a novel NF-kappaB inhibitor, through downregulation of NFATc1. J. Bone Miner. Res. 2005, 20, 653–662. [Google Scholar] [CrossRef]
- Wang, Z.Q.; Ovitt, C.; Grigoriadis, A.E.; Möhle-Steinlein, U.; Rüther, U.; Wagner, E.F. Bone and haematopoietic defects in mice lacking c-fos. Nature 1992, 360, 741–745. [Google Scholar] [CrossRef]
- Gannett, P.M.; Ye, J.; Ding, M.; Powell, J.; Zhang, Y.; Darian, E.; Daft, J.; Shi, X. Activation of AP-1 through the MAP kinase pathway: A potential mechanism of the carcinogenic effect of arenediazonium ions. Chem. Res. Toxicol. 2000, 13, 1020–1027. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.; Sato, K.; Asagiri, M.; Morita, I.; Soma, K.; Takayanagi, H. Contribution of NFATc1 to the transcriptional control of immunoreceptor OSCAR but not TREM-2 during osteoclastogenesis. J. Biol. Chem. 2005, 280, 32905–32913. [Google Scholar] [CrossRef] [PubMed]
- Lu, W.; Gao, B.; Fan, J.; Cheng, P.; Hu, Y.; Jie, Q.; Luo, Z.; Yang, L. Mesenchymal progenitors derived from different locations in long bones display diverse characteristics. Stem Cells Int. 2019, 2019, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Murao, H.; Yamamoto, K.; Matsuda, S.; Akiyama, H. Periosteal cells are a major source of soft callus in bone fracture. J. Bone Miner. Metab. 2013, 31, 390–398. [Google Scholar] [CrossRef]
- Koshihara, Y.; Honda, Y. Age-related increase in collagen production in cultured human osteoblast-like periosteal cells. Mech. Ageing Dev. 1994, 74, 89–101. [Google Scholar] [CrossRef]
- De Bari, C.; Dell’Accio, F.; Vanlauwe, J.; Eyckmans, J.; Khan, I.M.; Archer, C.W.; Jones, E.A.; McGonagle, D.; Mitsiadis, T.A.; Pitzalis, C.; et al. Mesenchymal multipotency of adult human periosteal cells demonstrated by single-cell lineage analysis. Arthritis Rheum. 2006, 54, 1209–1221. [Google Scholar] [CrossRef]
- Vozzi, G.; Lucarini, G.; Dicarlo, M.; Andreoni, C.; Salvolini, E.; Ferretti, C.; Mattioli-Belmonte, M. In vitro lifespan and senescent behaviour of human periosteal derived stem cells. Bone 2016, 88, 1–12. [Google Scholar] [CrossRef]
- Shapiro, F. Bone development and its relation to fracture repair. The role of mesenchymal osteoblasts and surface osteoblasts. Eur. Cells Mater. 2008, 15, 53–76. [Google Scholar] [CrossRef]
- Stich, S.; Loch, A.; Leinhase, I.; Neumann, K.; Kaps, C.; Sittinger, M.; Ringe, J. Human periosteum-derived progenitor cells express distinct chemokine receptors and migrate upon stimulation with CCL2, CCL25, CXCL8, CXCL12, and CXCL13. Eur. J. Cell Biol. 2008, 87, 365–376. [Google Scholar] [CrossRef]
- Chappuis, V.; Gamer, L.; Cox, K.; Lowery, J.W.; Bosshardt, D.D.; Rosen, V. Periosteal BMP2 activity drives bone graft healing. Bone 2012, 51, 800–809. [Google Scholar] [CrossRef]
- Chen, G.; Deng, C.; Li, Y.P. TGF-β and BMP signaling in osteoblast differentiation and bone formation. Int. J. Biol. Sci. 2012, 8, 272–288. [Google Scholar] [CrossRef] [PubMed]
- Evans, S.F.; Chang, H.; Knothe Tate, M.L. Elucidating multiscale periosteal mechanobiology: A key to unlocking the smart properties and regenerative capacity of the periosteum? Tissue Eng. Part B. Rev. 2013, 19, 147–159. [Google Scholar] [CrossRef] [PubMed]
- Gao, B.; Deng, R.; Chai, Y.; Chen, H.; Hu, B.; Wang, X.; Zhu, S.; Cao, Y.; Ni, S.; Wan, M.; et al. Macrophage-lineage TRAP+ cells recruit periosteum-derived cells for periosteal osteogenesis and regeneration. J. Clin. Investig. 2019, 129, 2578–2594. [Google Scholar] [CrossRef] [PubMed]
- Sozen, T.; Ozisik, L.; Calik Basaran, N. An overview and management of osteoporosis. Eur. J. Rheumatol. 2017, 4, 46–56. [Google Scholar] [CrossRef] [PubMed]
- Mäkitie, R.E.; Haanpää, M.; Valta, H.; Pekkinen, M.; Laine, C.M.; Lehesjoki, A.E.; Schalin-Jäntti, C.; Mäkitie, O. Skeletal Characteristics of WNT1 Osteoporosis in Children and Young Adults. J. Bone Miner. Res. 2016, 31, 1734–1742. [Google Scholar] [CrossRef] [PubMed]
- Appelman-Dijkstra, N.M.; Papapoulos, S.E. Modulating Bone Resorption and Bone Formation in Opposite Directions in the Treatment of Postmenopausal Osteoporosis. Drugs 2015, 75, 1049–1058. [Google Scholar] [CrossRef] [PubMed]
- Boggild, M.K.; Gajic-Veljanoski, O.; McDonald-Blumer, H.; Ridout, R.; Tile, L.; Josse, R.; Cheung, A.M. Odanacatib for the treatment of osteoporosis. Expert Opin. Pharmacother. 2015, 16, 1717–1726. [Google Scholar] [CrossRef]
- Binkley, N.; Krueger, D.; de Papp, A.E. Multiple vertebral fractures following osteoporosis treatment discontinuation: A case-report after long-term Odanacatib. Osteoporos. Int. 2018, 29, 999–1002. [Google Scholar] [CrossRef]
- Stone, J.A.; McCrea, J.B.; Witter, R.; Zajic, S.; Stoch, S.A. Clinical and translational pharmacology of the cathepsin K inhibitor odanacatib studied for osteoporosis. Br. J. Clin. Pharmacol. 2019, 85, 1072–1083. [Google Scholar] [CrossRef]
- Valenti, M.T.; Carbonare, L.D.; Mottes, M. Osteogenic differentiation in healthy and pathological conditions. Int. J. Mol. Sci. 2017, 18, 41. [Google Scholar] [CrossRef]
- Roberts, S.; Colombier, P.; Sowman, A.; Mennan, C.; Rölfing, J.H.D.; Guicheux, J.; Edwards, J.R. Ageing in the musculoskeletal system: Cellular function and dysfunction throughout life. Acta Orthop. 2016, 87, 15–25. [Google Scholar] [CrossRef] [PubMed]
- Berner, A.; Reichert, J.C.; Woodruff, M.A.; Saifzadeh, S.; Morris, A.J.; Epari, D.R.; Nerlich, M.; Schuetz, M.A.; Hutmacher, D.W. Autologous vs. allogenic mesenchymal progenitor cells for the reconstruction of critical sized segmental tibial bone defects in aged sheep. Acta Biomater. 2013, 9, 7874–7884. [Google Scholar] [CrossRef] [PubMed]
- Regmi, S.; Pathak, S.; Kim, J.O.; Yong, C.S.; Jeong, J.-H. Mesenchymal stem cell therapy for the treatment of inflammatory diseases: Challenges, opportunities, and future perspectives. Eur. J. Cell Biol. 2019, 98, 151041. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Yuan, Q.; Xie, L. Mesenchymal Stem Cell-Based Immunomodulation: Properties and Clinical Application. Stem Cells Int. 2018, 2018, 3057624. [Google Scholar] [CrossRef] [PubMed]
- Ma, T.; Wang, X.; Jiao, Y.; Wang, H.; Qi, Y.; Gong, H.; Zhang, L.; Jiang, D. Interleukin 17 (IL-17)-Induced Mesenchymal Stem Cells Prolong the Survival of Allogeneic Skin Grafts. Ann. Transplant. 2018, 23, 615–621. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Shen, S.; Fu, H.; Wang, Z.; Li, X.; Sui, X.; Yuan, M.; Liu, S.; Wang, G.; Guo, Q. Immunomodulatory Functions of Mesenchymal Stem Cells in Tissue Engineering. Stem Cells Int. 2019, 2019, 1–18. [Google Scholar] [CrossRef]
- Weiss, A.R.R.; Dahlke, M.H. Immunomodulation by Mesenchymal Stem Cells (MSCs): Mechanisms of Action of Living, Apoptotic, and Dead MSCs. Front. Immunol. 2019, 10, 1191. [Google Scholar] [CrossRef]
- Lin, H.; Xu, R.; Zhang, Z.; Chen, L.; Shi, M.; Wang, F.-S. Implications of the immunoregulatory functions of mesenchymal stem cells in the treatment of human liver diseases. Cell. Mol. Immunol. 2011, 8, 19–22. [Google Scholar] [CrossRef]
- Zhao, Z.; Wang, Z.; Li, Q.; Li, W.; You, Y.; Zou, P. The Different Immunoregulatory Functions of Mesenchymal Stem Cells in Patients with Low-Risk or High-Risk Myelodysplastic Syndromes. PLoS ONE 2012, 7, e45675. [Google Scholar] [CrossRef]
- Annamalai, R.T.; Hong, X.; Schott, N.G.; Tiruchinapally, G.; Levi, B.; Stegemann, J.P. Injectable osteogenic microtissues containing mesenchymal stromal cells conformally fill and repair critical-size defects. Biomaterials 2019, 208, 32–44. [Google Scholar] [CrossRef]
- Tohma, Y.; Dohi, Y.; Ohgushi, H.; Tadokoro, M.; Akahane, M.; Tanaka, Y. Osteogenic activity of bone marrow-derived mesenchymal stem cells (BMSCs) seeded on irradiated allogenic bone. J. Tissue Eng. Regen. Med. 2012, 6, 96–102. [Google Scholar] [CrossRef] [PubMed]
- Niemeyer, P.; Kornacker, M.; Mehlhorn, A.; Seckinger, A.; Vohrer, J.; Schmal, H.; Kasten, P.; Eckstein, V.; Südkamp, N.P.; Krause, U. Comparison of Immunological Properties of Bone Marrow Stromal Cells and Adipose Tissue–Derived Stem Cells Before and After Osteogenic Differentiation In Vitro. Tissue Eng. 2007, 13, 111–121. [Google Scholar] [CrossRef] [PubMed]
- Cenni, E.; Perut, F.; Baldini, N. In vitro models for the evaluation of angiogenic potential in bone engineering. Acta Pharmacol. Sin. 2011, 32, 21–30. [Google Scholar] [CrossRef] [PubMed]
- Jing, W.; Smith, A.A.; Liu, B.; Li, J.; Hunter, D.J.; Dhamdhere, G.; Salmon, B.; Jiang, J.; Cheng, D.; Johnson, C.A.; et al. Reengineering autologous bone grafts with the stem cell activator WNT3A. Biomaterials 2015, 47, 29–40. [Google Scholar] [CrossRef] [PubMed]
- Polo-Corrales, L.; Latorre-Esteves, M.; Ramirez-Vick, J.E. Scaffold design for bone regeneration. J. Nanosci. Nanotechnol. 2014, 14, 15–56. [Google Scholar] [CrossRef] [PubMed]
- Ullah, I.; Subbarao, R.B.; Rho, G.J. Human mesenchymal stem cells—Current trends and future prospective. Biosci. Rep. 2015, 35. [Google Scholar] [CrossRef] [PubMed]
- Oryan, A. Mesenchymal Stem Cells and Immunomodulation: Implications in Bone Tissue Engineering. Int. Clin. Pathol. J. 2016, 2, 2–4. [Google Scholar] [CrossRef]
- Oryan, A.; Alidadi, S.; Moshiri, A.; Maffulli, N. Bone regenerative medicine: Classic options, novel strategies, and future directions. J. Orthop. Surg. Res. 2014, 9, 18. [Google Scholar] [CrossRef]
- Albrektsson, T.; Johansson, C. Osteoinduction, osteoconduction and osseointegration. Eur. Spine J. 2001, 10, S96–S101. [Google Scholar] [CrossRef]
- Yoo, J.U.; Johnstone, B. The role of osteochondral progenitor cells in fracture repair. In Clinical Orthopaedics and Related Research; Lippincott Williams and Wilkins: Ambler, PA, USA, 1998. [Google Scholar]
- Han, D.; Han, N.; Zhang, P.; Jiang, B. Local transplantation of osteogenic pre-differentiated autologous adipose-derived mesenchymal stem cells may accelerate non-union fracture healing with limited pro-metastatic potency. Int. J. Clin. Exp. Med. 2015, 8, 1406. [Google Scholar]
- Hernigou, P.; Beaujean, F. Bone marrow in patients with pseudarthrosis. A study of progenitor cells by in vitro cloning. Rev. Chir. orthopédique réparatrice l’appareil Mot. 1997, 83, 33–40. [Google Scholar]
- Titsinides, S.; Agrogiannis, G.; Karatzas, T. Bone grafting materials in dentoalveolar reconstruction: A comprehensive review. Jpn. Dent. Sci. Rev. 2019, 55, 26–32. [Google Scholar] [CrossRef]
- Ho-Shui-Ling, A.; Bolander, J.; Rustom, L.E.; Johnson, A.W.; Luyten, F.P.; Picart, C. Bone regeneration strategies: Engineered scaffolds, bioactive molecules and stem cells current stage and future perspectives. Biomaterials 2018, 180, 143–162. [Google Scholar] [CrossRef]
- Amini, A.R.; Laurencin, C.T.; Nukavarapu, S.P. Bone tissue engineering: Recent advances and challenges. Crit. Rev. Biomed. Eng. 2012, 40, 363–408. [Google Scholar] [CrossRef]
- El Bialy, I.; Jiskoot, W.; Reza Nejadnik, M. Formulation, Delivery and Stability of Bone Morphogenetic Proteins for Effective Bone Regeneration. Pharm. Res. 2017, 34, 1152–1170. [Google Scholar] [CrossRef]
- Yu, X.; Suárez-González, D.; Khalil, A.S.; Murphy, W.L. How does the pathophysiological context influence delivery of bone growth factors? Adv. Drug Deliv. Rev. 2015, 84, 68–84. [Google Scholar] [CrossRef]
- Visser, R.; Rico-Llanos, G.A.; Pulkkinen, H.; Becerra, J. Peptides for bone tissue engineering. J. Control. Release 2016, 244, 122–135. [Google Scholar] [CrossRef]
- Wang, W.; Yeung, K.W.K. Bone grafts and biomaterials substitutes for bone defect repair: A review. Bioact. Mater. 2017, 2, 224–247. [Google Scholar] [CrossRef]
- Loi, F.; Córdova, L.A.; Pajarinen, J.; Lin, T.H.; Yao, Z.; Goodman, S.B. Inflammation, fracture and bone repair. Bone 2016, 86, 119–130. [Google Scholar] [CrossRef]
- Seidenstuecker, M.; Lange, S.; Esslinger, S.; Latorre, S.H.; Krastev, R.; Gadow, R.; Mayr, H.O.; Bernstein, A. Inversely 3D-Printed β-TCP Scaffolds for Bone Replacement. Materials (Basel) 2019, 12, 3417. [Google Scholar] [CrossRef]
- Grellier, M.; Bordenave, L.; Amédée, J. Cell-to-cell communication between osteogenic and endothelial lineages: Implications for tissue engineering. Trends Biotechnol. 2009, 27, 562–571. [Google Scholar] [CrossRef] [PubMed]
- Kocherova, I.; Bryja, A.; Mozdziak, P.; Angelova Volponi, A.; Dyszkiewicz-Konwińska, M.; Piotrowska-Kempisty, H.; Antosik, P.; Bukowska, D.; Bruska, M.; Iżycki, D.; et al. Human Umbilical Vein Endothelial Cells (HUVECs) Co-Culture with Osteogenic Cells: From Molecular Communication to Engineering Prevascularised Bone Grafts. J. Clin. Med. 2019, 8, 1602. [Google Scholar] [CrossRef] [PubMed]
- Bose, S.; Vahabzadeh, S.; Bandyopadhyay, A. Bone tissue engineering using 3D printing. Mater. Today 2013, 16, 496–504. [Google Scholar] [CrossRef]
- Vorndran, E.; Klammert, U.; Ewald, A.; Barralet, J.E.; Gbureck, U. Simultaneous immobilization of bioactives during 3D powder printing of bioceramic drug-release matrices. Adv. Funct. Mater. 2010, 20, 1585–1591. [Google Scholar] [CrossRef]
- Orciani, M.; Fini, M.; Di Primio, R.; Mattioli-Belmonte, M. Biofabrication and bone tissue regeneration: Cell source, approaches, and challenges. Front. Bioeng. Biotechnol. 2017, 5, 17. [Google Scholar] [CrossRef]
- Popov, V.V.; Muller-Kamskii, G.; Kovalevsky, A.; Dzhenzhera, G.; Strokin, E.; Kolomiets, A.; Ramon, J. Design and 3D-printing of titanium bone implants: Brief review of approach and clinical cases. Biomed. Eng. Lett. 2018, 8, 337–344. [Google Scholar] [CrossRef]
- Midha, S.; Dalela, M.; Sybil, D.; Patra, P.; Mohanty, S. Advances in three-dimensional bioprinting of bone: Progress and challenges. J. Tissue Eng. Regen. Med. 2019, 13, 925–945. [Google Scholar] [CrossRef]
- Gao, P.; Zhang, H.; Liu, Y.; Fan, B.; Li, X.; Xiao, X.; Lan, P.; Li, M.; Geng, L.; Liu, D.; et al. Beta-tricalcium phosphate granules improve osteogenesis in vitro and establish innovative osteo-regenerators for bone tissue engineering in vivo. Sci. Rep. 2016, 6, 23367. [Google Scholar] [CrossRef]
- Seidenstuecker, M.; Kerr, L.; Bernstein, A.; Mayr, H.O.; Suedkamp, N.P.; Gadow, R.; Krieg, P.; Latorre, S.H.; Thomann, R.; Syrowatka, F.; et al. 3D powder printed bioglass and β-tricalcium phosphate bone scaffolds. Materials (Basel) 2017, 11, 13. [Google Scholar] [CrossRef]
- Kumar, V.B.; Khajuria, D.K.; Karasik, D.; Gedanken, A. Silver and gold doped hydroxyapatite nanocomposites for enhanced bone regeneration. Biomed. Mater. 2019, 14, 055002. [Google Scholar] [CrossRef]
- Taylor, U.; Garrels, W.; Barchanski, A.; Peterson, S.; Sajti, L.; Lucas-Hahn, A.; Gamrad, L.; Baulain, U.; Klein, S.; Kues, W.A.; et al. Injection of ligand-free gold and silver nanoparticles into murine embryos does not impact pre-implantation development. Beilstein J. Nanotechnol. 2014, 5, 677–688. [Google Scholar] [CrossRef] [PubMed]
- Walmsley, G.G.; McArdle, A.; Tevlin, R.; Momeni, A.; Atashroo, D.; Hu, M.S.; Feroze, A.H.; Wong, V.W.; Lorenz, P.H.; Longaker, M.T.; et al. Nanotechnology in bone tissue engineering. Nanomed. Nanotechnol. Biol. Med. 2015, 11, 1253–1263. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Li, J.J.; Zhang, J.; Wang, X.; Kawazoe, N.; Chen, G. Gold nanoparticle size and shape influence on osteogenesis of mesenchymal stem cells. Nanoscale 2016, 8, 7992–8002. [Google Scholar] [CrossRef] [PubMed]
- Dobbenga, S.; Fratila-Apachitei, L.E.; Zadpoor, A.A. Nanopattern-induced osteogenic differentiation of stem cells – A systematic review. Acta Biomater. 2016, 46, 3–14. [Google Scholar] [CrossRef] [PubMed]
- Janbaz, S.; Noordzij, N.; Widyaratih, D.S.; Hagen, C.W.; Fratila-Apachitei, L.E.; Zadpoor, A.A. Origami lattices with free-form surface ornaments. Sci. Adv. 2017, 3. [Google Scholar] [CrossRef]
- Mashinchian, O.; Turner, L.A.; Dalby, M.J.; Laurent, S.; Shokrgozar, M.A.; Bonakdar, S.; Imani, M.; Mahmoudi, M. Regulation of stem cell fate by nanomaterial substrates. Nanomedicine 2015, 10, 829–847. [Google Scholar] [CrossRef]
- Deng, L.; Wang, Y.; Peng, Y.; Wu, Y.; Ding, Y.; Jiang, Y.; Shen, Z.; Fu, Q. Osteoblast-derived microvesicles: A novel mechanism for communication between osteoblasts and osteoclasts. Bone 2015, 79, 37–42. [Google Scholar] [CrossRef]
- Ecol, M. All interstitial lung disease patients had a reduced DLCO. Mol. Ecol. 2017, 25, 1032–1057. [Google Scholar] [CrossRef]
- Fillingham, Y.; Jacobs, J. Bone grafts and their substitutes. Bone Joint J. 2016, 98-B, 6–9. [Google Scholar] [CrossRef]
- Ito, H.; Koefoed, M.; Tiyapatanaputi, P.; Gromov, K.; Goater, J.J.; Carmouche, J.; Zhang, X.; Rubery, P.T.; Rabinowitz, J.; Samulski, R.J.; et al. Remodeling of cortical bone allografts mediated by adherent rAAV-RANKL and VEGF gene therapy. Nat. Med. 2005, 11, 291–297. [Google Scholar] [CrossRef]
- Xiong, M.Y.; Liu, L.Q.; Liu, S.Q.; Liu, Z.H.; Gao, H.F. Effects of osteoprotegerin, RANK and RANKL on bone destruction and collapse in avascular necrosis femoral head. Am. J. Transl. Res. 2016, 8, 3133–3140. [Google Scholar] [PubMed]
- Jin, Q.; Cirelli, J.A.; Park, C.H.; Sugai, J.V.; Taba, M.; Kostenuik, P.J.; Giannobile, W.V. RANKL Inhibition Through Osteoprotegerin Blocks Bone Loss in Experimental Periodontitis. J. Periodontol. 2007, 78, 1300–1308. [Google Scholar] [CrossRef] [PubMed]
- Tera, T.d.M.; Prado, R.F.d.; De Marco, A.C.; Santamaria, M.P.; Jardini, M.A.N. The RANK/ RANKL/ OPG interaction in the repair of autogenous bone grafts in female rats with estrogen deficiency. Braz. Oral Res. 2014, 28, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Karner, C.M.; Long, F. Wnt signaling and cellular metabolism in osteoblasts. Cell. Mol. Life Sci. 2017, 74, 1649–1657. [Google Scholar] [CrossRef] [PubMed]
- Ng, L.F.; Kaur, P.; Bunnag, N.; Suresh, J.; Sung, I.C.H.; Tan, Q.H.; Gruber, J.; Tolwinski, N.S. WNT Signaling in Disease. Cells 2019, 8, 826. [Google Scholar] [CrossRef]
- Li, V.S.W.; Ng, S.S.; Boersema, P.J.; Low, T.Y.; Karthaus, W.R.; Gerlach, J.P.; Mohammed, S.; Heck, A.J.R.; Maurice, M.M.; Mahmoudi, T.; et al. Wnt Signaling through Inhibition of β-Catenin Degradation in an Intact Axin1 Complex. Cell 2012, 149, 1245–1256. [Google Scholar] [CrossRef]
- Gong, Y.; Allgrove, J.; van den Boogaard, M.J.; Baron, R.; Steinmann, B.; Fukai, N.; Superti-Furga, A.; Letteboer, T.; Oexle, K.; Ramesar, R.S.; et al. LDL receptor-related protein 5 (LRP5) affects bone accrual and eye development. Cell 2001, 107, 513–523. [Google Scholar] [CrossRef]
- Li, X.; Zhang, Y.; Kang, H.; Liu, W.; Liu, P.; Zhang, J.; Harris, S.E.; Wu, D. Sclerostin binds to LRP5/6 and antagonizes canonical Wnt signaling. J. Biol. Chem. 2005, 280, 19883–19887. [Google Scholar] [CrossRef]
- Semënov, M.V.; Zhang, X.; He, X. DKK1 antagonizes Wnt signaling without promotion of LRP6 internalization and degradation. J. Biol. Chem. 2008, 283, 21427–21432. [Google Scholar] [CrossRef]
- Cui, Y.; Luan, J.; Li, H.; Zhou, X.; Han, J. Exosomes derived from mineralizing osteoblasts promote ST2 cell osteogenic differentiation by alteration of microRNA expression. FEBS Lett. 2016, 590, 185–192. [Google Scholar] [CrossRef]
- Hammond, S.M. An overview of microRNAs. Adv. Drug Deliv. Rev. 2015, 87, 3–14. [Google Scholar] [CrossRef] [PubMed]
- Kang, H.; Hata, A. The role of microRNAs in cell fate determination of mesenchymal stem cells: Balancing adipogenesis and osteogenesis. BMB Rep. 2015, 48, 319–323. [Google Scholar] [CrossRef] [PubMed]
- Xiao, Y.; Li, B.; Liu, J. MiRNA-27a regulates arthritis via PPARγ in vivo and in vitro. Mol. Med. Rep. 2018, 17, 5454–5462. [Google Scholar] [CrossRef] [PubMed]
- Gu, C.; Xu, Y.; Zhang, S.; Guan, H.; Song, S.; Wang, X.; Wang, Y.; Li, Y.; Zhao, G. MIR-27a attenuates adipogenesis and promotes osteogenesis in steroid-induced rat BMSCs by targeting PPARγ 3 and GREM1. Sci. Rep. 2016, 6, 1–12. [Google Scholar] [CrossRef]
- Cheng, F.; Yang, M.; Yang, R. MiRNA-365a-3p promotes the progression of osteoporosis by inhibiting osteogenic differentiation via targeting RUNX2. Eur. Rev. Med. Pharmacol. Sci. 2019, 23, 7766–7774. [Google Scholar]
- Yang, L.; Zeng, Z.; Kang, N.; Yang, J.C.; Wei, X.; Hai, Y. Circ-VANGL1 promotes the progression of osteoporosis by absorbing miRNA-217 to regulate RUNX2 expression. Eur. Rev. Med. Pharmacol. Sci. 2019, 23, 949–957. [Google Scholar] [CrossRef]
- Weilner, S.; Schraml, E.; Wieser, M.; Messner, P.; Schneider, K.; Wassermann, K.; Micutkova, L.; Fortschegger, K.; Maier, A.B.; Westendorp, R.; et al. Secreted microvesicular miR-31 inhibits osteogenic differentiation of mesenchymal stem cells. Aging Cell 2016, 15, 744–754. [Google Scholar] [CrossRef]
- Abudusaimi, A.; Aihemaitijiang, Y.; Wang, Y.-H.; Cui, L.; Maimaitiming, S.; Abulikemu, M. Adipose-Derived Stem Cells Enhance Bone Regeneration in Vascular Necrosis of the Femoral Head in the Rabbit. J. Int. Med. Res. 2011, 39, 1852–1860. [Google Scholar] [CrossRef]
- Lange-Consiglio, A.; Corradetti, B.; Meucci, A.; Perego, R.; Bizzaro, D.; Cremonesi, F. Characteristics of equine mesenchymal stem cells derived from amnion and bone marrow: In vitro proliferative and multilineage potential assessment. Equine Vet. J. 2013, 45, 737–744. [Google Scholar] [CrossRef]
- Rawat, S.; Gupta, S.; Mohanty, S. Mesenchymal Stem Cells Modulate the Immune System in Developing Therapeutic Interventions. In Immune Response Activation and Immunomodulation; IntechOpen: London, UK, 2019. [Google Scholar]
- Taketani, T.; Oyama, C.; Mihara, A.; Tanabe, Y.; Abe, M.; Hirade, T.; Yamamoto, S.; Bo, R.; Kanai, R.; Tadenuma, T.; et al. Ex vivo expanded allogeneic mesenchymal stem cells with bone marrow transplantation improved osteogenesis in infants with severe hypophosphatasia. Cell Transplant. 2015, 24, 1931–1943. [Google Scholar] [CrossRef]
- Fernandez-Bances, I.; Perez-Basterrechea, M.; Perez-Lopez, S.; Nuñez Batalla, D.; Fernandez Rodriguez, M.A.; Alvarez-Viejo, M.; Ferrero-Gutierrez, A.; Menendez-Menendez, Y.; Garcia-Gala, J.M.; Escudero, D.; et al. Repair of long-bone pseudoarthrosis with autologous bone marrow mononuclear cells combined with allogenic bone graft. Cytotherapy 2013, 15, 571–577. [Google Scholar] [CrossRef] [PubMed]
- Martin, J.R.; Houdek, M.T.; Sierra, R.J. Use of concentrated bone marrow aspirate and platelet rich plasma during minimally invasive decompression of the femoral head in the treatment of osteonecrosis. Croat. Med. J. 2013, 54, 219–224. [Google Scholar] [CrossRef] [PubMed]
- Memeo, A.; Verdoni, F.; Ingraffia, C.; Rossi, L.; Pedretti, L.; D’Amato, R.D.; Peretto, L.; Mineo, G. V Mesenchymal stem cells as adjuvant therapy for limb lengthening in achondroplasia. J. Pediatr. Orthop. B 2019, 28, 221–227. [Google Scholar] [CrossRef] [PubMed]
- El Hadidi, Y.N.; Seif, M.; Sleem, H.; Mohsen, K.A.; El Kassaby, M. The Effect of Bone Marrow Aspirate Concentrate Application on Distracted Bone Biomechanical Properties. J. Craniofac. Surg. 2019, 30, 2650–2655. [Google Scholar] [CrossRef]
- Pilge, H.; Bittersohl, B.; Schneppendahl, J.; Hesper, T.; Zilkens, C.; Ruppert, M.; Krauspe, R.; Jäger, M. Bone Marrow Aspirate Concentrate in Combination with Intravenous Iloprost Increases Bone Healing in Patients with Avascular Necrosis of the Femoral Head: A Matched Pair Analysis. Orthop. Rev. (Pavia) 2016, 8, 6902. [Google Scholar] [CrossRef]
- Tomaru, Y.; Yoshioka, T.; Sugaya, H.; Aoto, K.; Wada, H.; Akaogi, H.; Yamazaki, M.; Mishima, H. Hip preserving surgery with concentrated autologous bone marrow aspirate transplantation for the treatment of asymptomatic osteonecrosis of the femoral head: Retrospective review of clinical and radiological outcomes at 6 years postoperatively. BMC Musculoskelet. Disord. 2017, 18, 292. [Google Scholar] [CrossRef]
- Makihara, T.; Yoshioka, T.; Sugaya, H.; Yamazaki, M.; Mishima, H. Autologous Concentrated Bone Marrow Grafting for the Treatment of Osteonecrosis of the Humeral Head: A Report of Five Shoulders in Four Cases. Case Rep. Orthop. 2017, 2017, 1–8. [Google Scholar] [CrossRef]
- De Riu, G.; Vaira, L.A.; Carta, E.; Meloni, S.M.; Sembronio, S.; Robiony, M. Bone marrow nucleated cell concentrate autograft in temporomandibular joint degenerative disorders: 1-year results of a randomized clinical trial. J. Cranio-Maxillofacial Surg. 2019, 47, 1728–1738. [Google Scholar] [CrossRef]

| Completed Research Studies and Clinical Trials on Bone Regeneration and Reconstruction | |||
| Study Title | Conditions | Interventions | Principal Investigator (Year of Study) |
| Clinical Trial of Intravenous Infusion of Fucosylated Bone Marrow Mesenchyme Cells in Patients with Osteoporosis | Osteoporosis Spinal fractures | Biological: Fucosylated MSC for Osteoporosis | Lonano et al. (2018) |
| A New Procedure in Bone Engineering Using Induced Adipose Tissue | Osteogenesis Osteonecrosis Osteoarthritis | Interconversion of mesenchymal stem cells from adipocytes to osteocytes | Alfotawi et al. (2019) |
| Repair of long-bone pseudoarthrosis with autologous bone marrow mononuclear cells combined with allogenic bone graft | Long-bone Pseudoarthrosis | Allogenic grafts supplementation with autologous BM-MNCs | Fernandez-Bances et al. (2013) |
| Use of concentrated bone marrow aspirate and platelet rich plasma during minimally invasive decompression of the femoral head in the treatment of osteonecrosis. | Osteonecrosis | Surgical procedure including decompression of femoral head and injection of mesenchymal stem cells | Martin et al. (2013) |
| Clinical Trial of Allogenic Adipose Tissue-Derived Mesenchymal Progenitor Cells Therapy for Knee Osteoarthritis | Osteoarthritis | Mesenchymal progenitor cells | Zhao X et al. (2018) |
| Bone Marrow Aspirate Concentrate in Combination with Intravenous Iloprost Increases Bone Healing in Patients With Avascular Necrosis of the Femoral Head: A Matched Pair Analysis. | Avascular necrosis of the femoral head | Intravenous injection of bone marrow concentrates in association with iloprost | Pilge et al. (2017) |
| Reduced Intensity AlloTransplant for Osteopetrosis | Osteopetrosis | Stem Cell or Umbilical Cord Blood Transplantation | Paul Orchard, MD (2008) |
| Treatment of osteonecrosis of the femoral head by core decompression and implantation of fully functional ex vivo-expanded bone marrow-derived mesenchymal stem cells: a proof-of-concept study | Osteonecrosis of the Femoral Head | Bone marrow aspirate | Mardones et al. (2019) |
| Inefficacy of autologous bone marrow concentrate in stage three osteonecrosis: a randomized controlled double-blind trial | Osteonecrosis | Bone marrow concentrate | Hauzeur et al. (2018) |
| The Effects of Intra-articular Injection of Mesenchymal Stem Cells in Knee Joint Osteoarthritis | Osteoarthritis | Mesenchymal stem cells | Mohsen Emadeddin, MD (2015) |
| Treatment of knee osteoarthritis with autologous mesenchymal stem cells: A pilot study | Osteoarthritis | Mesenchymal stem cells | Orozco et al. (2013) |
| Intra-articular Injection of Mesenchymal Stem Cells in Osteonecrosis of the Femoral Head | Osteoarthritis | Placenta Derived Mesenchymal Stem Cell | Yu tang et al. (2018) |
| Risk-Adapted Allogeneic Stem Cell Transplantation for Mixed Donor Chimerism In Patients With Non-Malignant Diseases | Bone Marrow Failure | Fludarabine, Cyclophosphamide Cyclophosphamide 40, Cyclophosphamide 30 | Garvin et al. (2011) |
| Safety Study of Gene Modified Donor T-cells Following TCR Alpha Beta Depleted Stem Cell Transplant | Osteopetrosis | Biological: BPX-501 T cells Drug: rimiducid | Locatelli et al. (2019) |
| Use of Cell Therapy to Enhance Arthroscopic Knee Cartilage Surgery | Bone fracture | Standard microfracture arthroscopic surgery, autologous stem cell administration | Burke et al. (2019) |
| T-cell Depleted Alternative Donor Transplantation | Bone Marrow Failure Osteopetrosis | CliniMACS® (T cell depletion) | Gilman et al. (2017) |
| Clinical Trial of Intravenous Infusion of Fucosylated Bone Marrow Mesenchyme Cells in Patients with Osteoporosis | Osteoporosis | Fucosylated MSC for Osteoporosis | Linares Ferrando et al. (2017) |
| Hip preserving surgery with concentrated autologous bone marrow aspirate transplantation for the treatment of asymptomatic osteonecrosis of the femoral head: retrospective review of clinical and radiological outcomes at 6 years postoperatively | Femoral head collapse | Surgical procedure including autologous bone marrow aspirate transplantation | Tomaru et al. (2017) |
| Treatment of Atrophic Non-union by Pre-osteoblast Cells | Non-union of long bone | percutaneous autologous pre-osteoblast cells implantation | Hauzeur et al. (2012) |
| Autologous Stem Cell Therapy for Fracture Non-union Healing | Non-union healing | carrier plus in vitro expanded autologous BMSCs | Richardson et al. (2014) |
| Cell Therapy by Autologous BMC for Large Bone Defect Repair | Large bone defect | Bone marrow concentrate beta-TCP Chronos® Synthes | Marzi et al. (2019) |
| Cell Therapy by Bone Marrow-derived Mononuclear Cells (BMC) for Large Bone Defect Repair: Phase-I Clinical Trial | Large bone defect | Bone marrow concentrate | Marzi et al. (2016) |
| Treatment of Atrophic Non-union Fractures by Autologous Mesenchymal Stem Cell Percutaneous Grafting | Non-union fractures of bone | Administration of Mesenchymal Stem Cells Culture medium without MSC | Hauzeur et al. (2013) |
| Autologous BM-MSC Transplantation in Combination with Platelet Lysate (PL) for Non-union Treatment | Non-union fractures of bone | Percutaneous injection | Aghdami et al. (2015) |
| Pulse Shortwave Therapy in Cervical Osteoarthritis | Osteoarthritis | ActiPatch Etoricoxib 60 mg | Mohammad et al. (2019) |
| Safety and Efficacy of Meloxicam Compared to Other Nonsteroidal Anti-inflammatory Drugs (NSAIDs) in an Observational Cohort Study of Patients with Rheumatoid Arthritis, Osteoarthritis, Lumbago, Scapulohumeral Periarthritis, Neck, Shoulder and Arm Syndrome | Osteoarthritis | Non-Steroidal Anti-Inflammatory Drugs (NSAIDs) except etodolac | Boehringer Ingelheim (2014) |
| Investigation of Mesenchymal Stem Cell Therapy for the Treatment of Osteoarthritis of the Knee | Osteoarthritis | BMAC injection | Ruane et al. (2019) |
| Ongoing Research Studies and Clinical Trials on Bone Regeneration and Reconstruction | |||
| Study Title | Conditions | Interventions | Principal Investigator (Study Start Year) |
| Stem Cell Recruitment in Osteoporosis Therapy | Low Bone Density | Teriparatide, Alendronate, calcium and vitamin D | Suzanne Jan De Beur, MD (2012) |
| Phase 2a Study on Intravenous Infusion of Autologous Osteoblastic Cells in Severe Osteoporosis | Severe Osteoporosis | PREOB® Intravenous Infusion | Anderlecht, Belgium (2019) |
| Mesenchymal Stem Cells in Osteonecrosis of the Femoral Head | Avascular Necrosis of Femur Head | XCEL-MT-OSTEO-ALPHA | Màrius Aguirre et al. (2019) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hutchings, G.; Moncrieff, L.; Dompe, C.; Janowicz, K.; Sibiak, R.; Bryja, A.; Jankowski, M.; Mozdziak, P.; Bukowska, D.; Antosik, P.; et al. Bone Regeneration, Reconstruction and Use of Osteogenic Cells; from Basic Knowledge, Animal Models to Clinical Trials. J. Clin. Med. 2020, 9, 139. https://doi.org/10.3390/jcm9010139
Hutchings G, Moncrieff L, Dompe C, Janowicz K, Sibiak R, Bryja A, Jankowski M, Mozdziak P, Bukowska D, Antosik P, et al. Bone Regeneration, Reconstruction and Use of Osteogenic Cells; from Basic Knowledge, Animal Models to Clinical Trials. Journal of Clinical Medicine. 2020; 9(1):139. https://doi.org/10.3390/jcm9010139
Chicago/Turabian StyleHutchings, Greg, Lisa Moncrieff, Claudia Dompe, Krzysztof Janowicz, Rafał Sibiak, Artur Bryja, Maurycy Jankowski, Paul Mozdziak, Dorota Bukowska, Paweł Antosik, and et al. 2020. "Bone Regeneration, Reconstruction and Use of Osteogenic Cells; from Basic Knowledge, Animal Models to Clinical Trials" Journal of Clinical Medicine 9, no. 1: 139. https://doi.org/10.3390/jcm9010139
APA StyleHutchings, G., Moncrieff, L., Dompe, C., Janowicz, K., Sibiak, R., Bryja, A., Jankowski, M., Mozdziak, P., Bukowska, D., Antosik, P., Shibli, J. A., Dyszkiewicz-Konwińska, M., Bruska, M., Kempisty, B., & Piotrowska-Kempisty, H. (2020). Bone Regeneration, Reconstruction and Use of Osteogenic Cells; from Basic Knowledge, Animal Models to Clinical Trials. Journal of Clinical Medicine, 9(1), 139. https://doi.org/10.3390/jcm9010139








