Sex-Sparing Robot-Assisted Radical Cystectomy with Intracorporeal Padua Ileal Neobladder in Female: Surgical Technique, Perioperative, Oncologic and Functional Outcomes
Abstract
1. Introduction
2. Experimental Section
2.1. Patients
2.2. Surgical Technique
2.2.1. Sex-Sparing Robot-Assisted Radical Cystectomy
2.2.2. Pelvic Lymph Node Dissection and Intracorporeal Orthotopic Neobladder
2.3. Outcomes Evaluated
2.4. Statistical Analysis
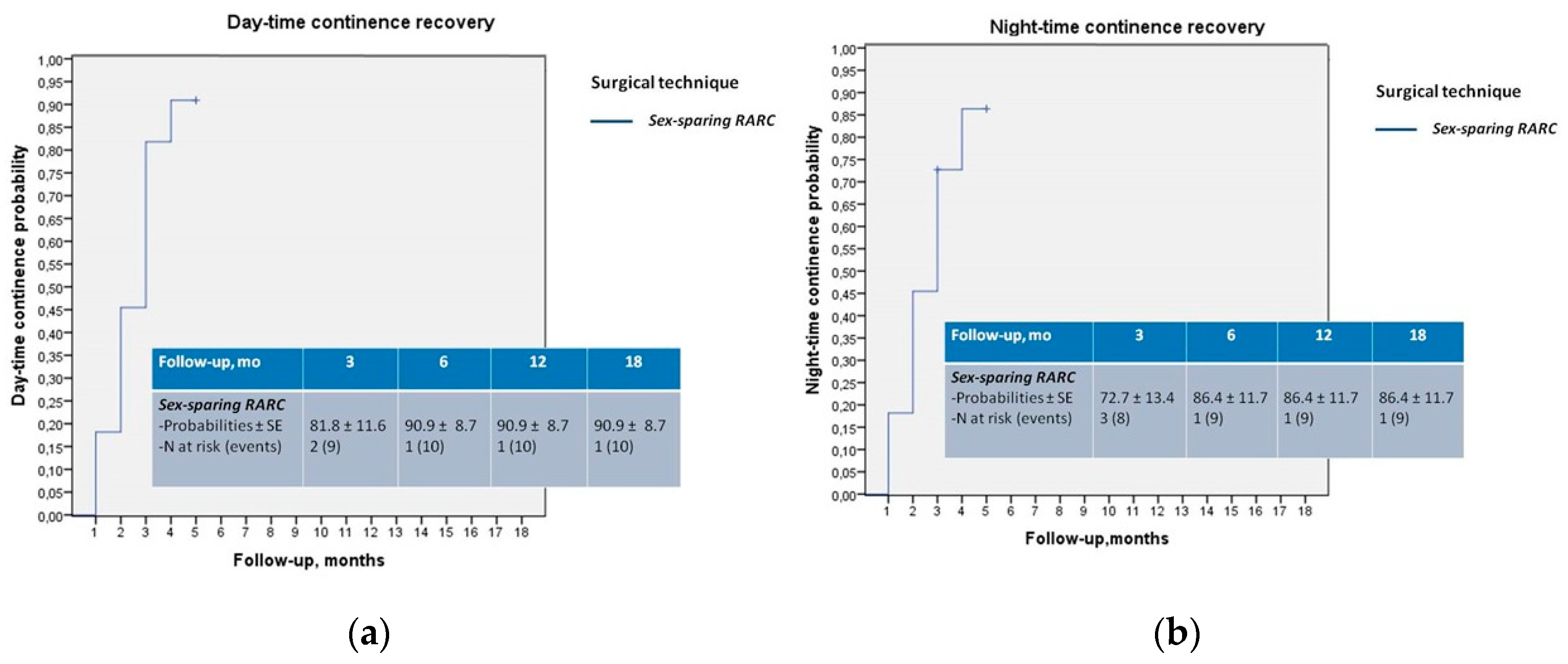
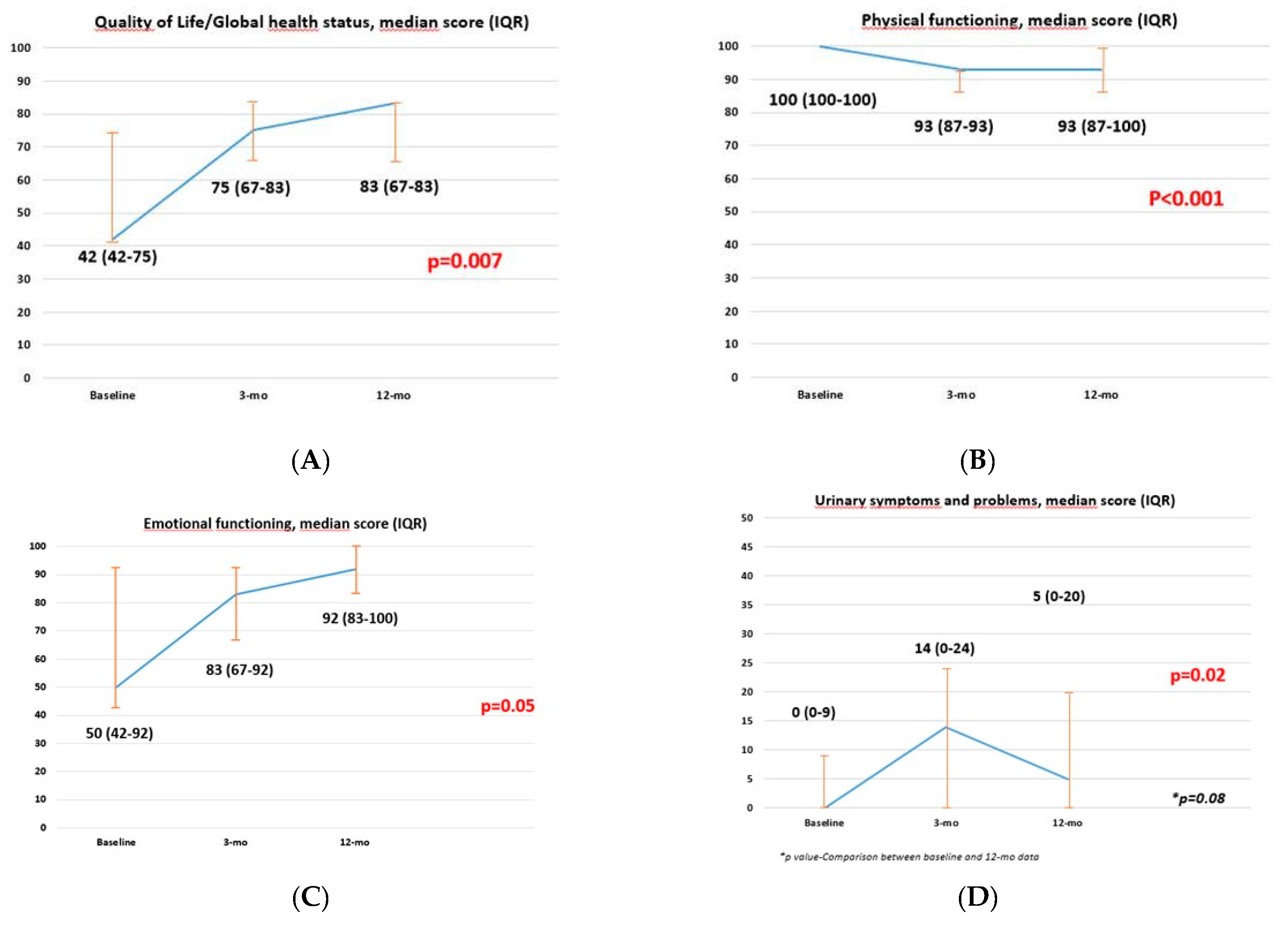
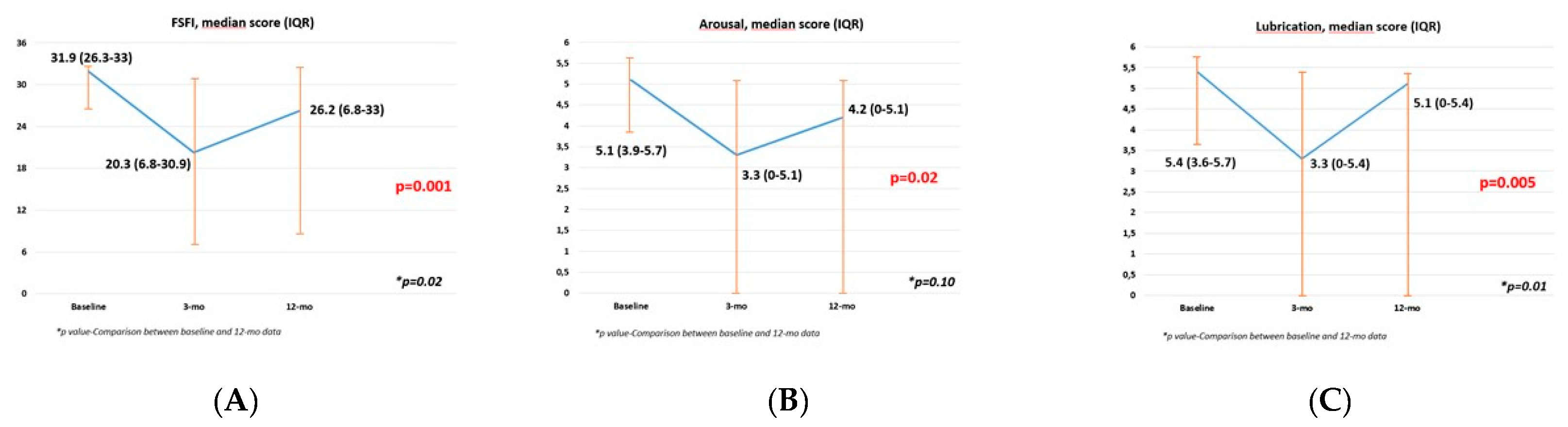
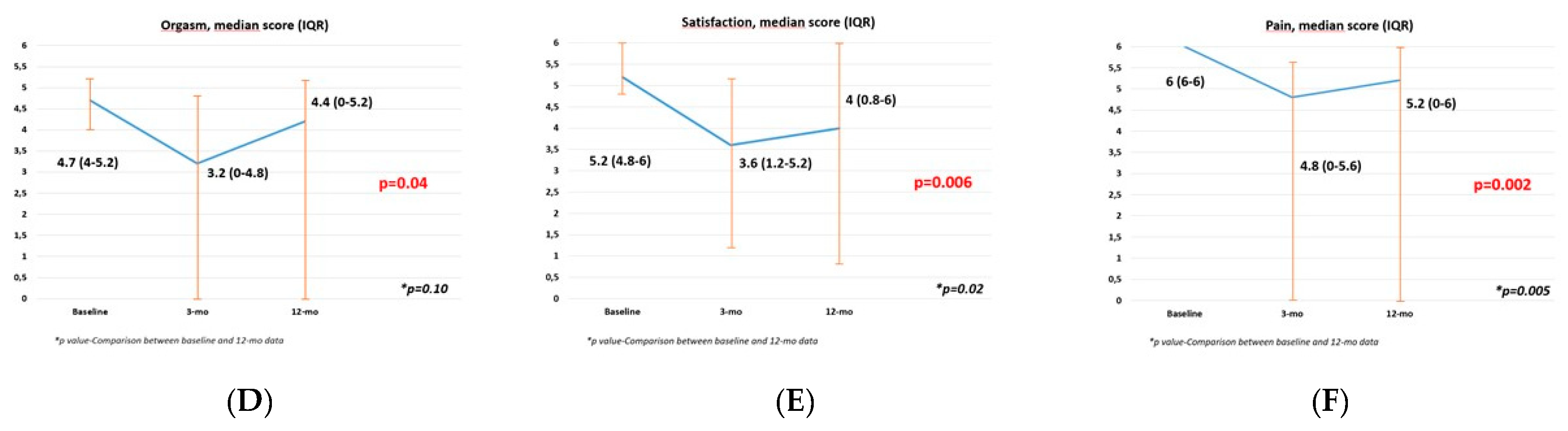
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Conflicts of Interest
References
- Witjes, J.A.; Lebret, T.; Compérat, E.; Cowan, N.C.; De Santis, M.; Bruins, H.M.; Hernández, V.; Espinos, E.L.; Dunn, J.; Rouanne, M.; et al. Updated 2016 EAU Guidelines on Muscle-invasive and Metastatic Bladder Cancer. Eur. Urol. 2017, 71, 462–475. [Google Scholar] [CrossRef]
- Burger, M.; Catto, J.; Dalbagni, G.; Grossman, H.B.; Herr, H.; Karakiewicz, P.; Kassouf, W.; Kiemeney, L.; La Vecchia, C.; Shariat, S.; et al. Epidemiology and risk factors of urothelial bladder cancer. Eur. Urol. 2013, 63, 234–241. [Google Scholar] [CrossRef] [PubMed]
- Mungan, N.; Aben, K.K.; Schoenberg, M.P.; Visser, O.; Coebergh, J.-W.W.; Witjes, J.; Kiemeney, L. Gender differences in stage-adjusted bladder cancer survival. Urology 2000, 55, 876–880. [Google Scholar] [CrossRef]
- Granberg, C.F.; Boorjian, S.A.; Crispen, P.L.; Tollefson, M.K.; Farmer, S.A.; Frank, I.; Blute, M.L. Functional and oncological outcomes after orthotopic neobladder reconstruction in women. BJU Int. 2008, 102, 1551–1555. [Google Scholar] [CrossRef] [PubMed]
- Veskimäe, E.; Neuzillet, Y.; Rouanne, M.; MacLennan, S.; Lam, T.B.L.; Yuan, Y.; Compérat, E.; Cowan, N.C.; Gakis, G.; Van Der Heijden, A.G.; et al. Systematic review of the oncological and functional outcomes of pelvic organ preserving radical cystectomy (RC) compared with standard RC in women who undergo curative surgery and orthotopic neobladder substitution for bladder cancer. BJU Int. 2017, 120, 12–24. [Google Scholar] [CrossRef]
- Zippe, C.D.; Raina, R.; Shah, A.D.; Massanyi, E.Z.; Agarwal, A.; Ulchaker, J.; Jones, S.; Klein, E. Female sexual dysfunction after radical cystectomy: A new outcome measure. Urology 2004, 63, 1153–1157. [Google Scholar] [CrossRef]
- Dhar, N.B.; Kessler, T.M.; Mills, R.D.; Burkhard, F.; Studer, U.E. Nerve-sparing radical cystectomy and orthotopic bladder replacement in female patients. Eur. Urol. 2007, 52, 1006–1014. [Google Scholar] [CrossRef]
- Simone, G.; Papalia, R.; Misuraca, L.; Tuderti, G.; Minisola, F.; Ferriero, M.; Vallati, G.E.; Guaglianone, S.; Gallucci, M. Robotic Intracorporeal Padua Ileal Bladder: Surgical Technique, Perioperative, Oncologic and Functional Outcomes. Eur. Urol. 2018, 73, 934–940. [Google Scholar] [CrossRef]
- Mitropoulos, D.; Artibani, W.; Biyani, C.S.; Jensen, J.B.; Rouprêt, M.; Truss, M. Validation of the Clavien-Dindo grading system in urology by the European Association of Urology Guidelines Ad Hoc Panel. Eur. Urol. Focus 2018, 4, 608–613. [Google Scholar] [CrossRef]
- Rosen, R.; Brown, C.; Heiman, J.; Leiblum, S.; Meston, C.; Shabsigh, R.; Ferguson, D.; D’Agostino, R., Jr. The Female Sexual Function Index (FSFI): A multidimensional self-report instrument for the assessment of female sexual function. J. Sex Marital. Ther. 2000, 26, 191–208. [Google Scholar] [CrossRef]
- Smith, A.B.; Crowell, K.; Woods, M.E.; Wallen, E.M.; Pruthi, R.S.; Nielsen, M.E.; Lee, C.T. Functional Outcomes Following Radical Cystectomy in Women with Bladder Cancer: A Systematic Review. Eur. Urol. Focus 2017, 3, 136–143. [Google Scholar] [CrossRef] [PubMed]
- Zahran, M.H.; Fahmy, O.; El-Hefnawy, A.S.; Ali-El-Dein, B. Female sexual dysfunction post radical cystectomy and urinary diversion. Climateric 2016, 19, 546–550. [Google Scholar] [CrossRef] [PubMed]
- Ripperda, C.M.; Jackson, L.A.; Phelan, J.N.; Carrick, K.S.; Corton, M.M. Anatomic relationships of the pelvic autonomic nervous system in female cadavers: Clinical applications to pelvic surgery. Am. J. Obstet. Gynecol. 2017, 216, 388.e1–388.e7. [Google Scholar] [CrossRef] [PubMed]
- Mauroy, B.; Demondion, X.; Bizet, B.; Claret, A.; Mestdagh, P.; Hurt, C. The female inferior hypogastric (= pelvic) plexus: Anatomical and radiological description of the plexus and its afferences—Applications to pelvic surgery. Surg. Radiol. Anat. 2007, 29, 55–66. [Google Scholar] [CrossRef] [PubMed]
- Mundy, A.R. An anatomical explanation for bladder dysfunction following rectal and uterine surgery. Br. J. Urol. 1982, 54, 501–504. [Google Scholar] [CrossRef] [PubMed]
- Morgan, J.L.; O’Connell, H.E.; McGuire, E.J. Is intrinsicsphincter deficiency a complication of simple hysterectomy? J. Urol. 2000, 164, 767–769. [Google Scholar] [CrossRef]
- Pederzoli, F.; Campbell, J.D.; Matsui, H.; Sopko, N.; Bivalacqua, T.J. Surgical Factors Associated with Male and Female Sexual Dysfunction After Radical Cystectomy: What Do We Know and How Can We Improve Outcomes? Sex Med. Rev. 2018, 6, 469–481. [Google Scholar] [CrossRef]
- Nandipati, C.; Bhat, A.; Zippe, C.D. Neurovascualr preservation in female orthotopic radical cystectomy significantly improves sexual function. Urology 2006, 67, 185–186. [Google Scholar] [CrossRef]
- Ali-El-Dein, B.; Mosbah, A.; Osman, Y.; El-Tabey, N.; Abdel-Latif, M.; Eraky, I.; Shaaban, A.A. Preservation of the internal genital organs during radical cystectomy in selected women with bladder cancer: A report on 15 cases with long term follow-up B. Eur. J. Surg. Oncol. 2013, 39, 358–364. [Google Scholar] [CrossRef]
- Bochner, B.H.; Dalbagni, G.; Sjoberg, D.D.; Silberstein, J.; Keren Paz, G.E.; Donat, S.M.; Coleman, J.A.; Mathew, S.; Vickers, A.; Schnorr, G.C.; et al. Comparing open radical cystectomy and robot-assisted laparoscopic radical cystectomy: A randomized clinical trial. Eur. Urol. 2015, 67, 1042–1050. [Google Scholar] [CrossRef]
- Moschini, M.; Soria, F.; Mathieu, R.; Xylinas, E.; D’Andrea, D.; Tan, W.S.; Kelly, J.D.; Simone, G.; Tuderti, G.; Meraney, A.; et al. Propensity-score-matched comparison of soft tissue surgical margins status between open and robotic-assisted radical cystectomy. Urol. Oncol. 2019, 37, 179.e1–179.e7. [Google Scholar] [CrossRef] [PubMed]
- Simone, G.; Tuderti, G.; Misuraca, L.; Anceschi, U.; Ferriero, M.; Minisola, F.; Guaglianone, S.; Gallucci, M. Perioperative and mid-term oncologic outcomes of robotic assisted radical cystectomy with totally intracorporeal neobladder: Results of a propensity score matched comparison with open cohort from a single-centre series. Eur. J. Surg. Oncol. 2018, 44, 1432–1438. [Google Scholar] [CrossRef] [PubMed]
- Asimakopoulos, A.D.; Campagna, A.; Gakis, G.; Montes, V.E.C.; Piechaud, T.; Hoepffner, J.-L.; Mugnier, C.; Gaston, R.; Corona, M.V.E. Nerve sparing robot-assisted radical cystectomy with intracorporeal bladder substitution in the male. J. Urol. 2016, 196, 1549–1557. [Google Scholar] [CrossRef] [PubMed]
- Lin, F.C.; Medendorp, A.; Van Kuiken, M.; Mills, S.A.; Tarnay, C.M. Vaginal Dehiscence and Evisceration after Robotic-Assisted Radical Cystectomy: A Case Series and Review of the Literature. Urology 2019, 134, 90–96. [Google Scholar] [CrossRef]
- Kanno, T.; Ito, K.; Sawada, A.; Saito, R.; Kobayashi, T.; Yamada, H.; Inoue, T.; Ogawa, O. Complications and reoperations after laparoscopic radical cystectomy in a Japanese multicenter cohort. Int. J. Urol. 2019, 26, 493–498. [Google Scholar] [CrossRef]
| Patients, n 11 | Sex-Sparing RARC |
|---|---|
| Age, year, mean (±SD) | 47.1 (13) |
| BMI, mean (±SD) | 23.1 (3.3) |
| ASA score, n (%) | |
| 1 | 4 (36.4) |
| 2 | 6 (54.6) |
| 3 | 1 (9) |
| 4 | - |
| Preoperative eGFR, mL/min, mean (±SD) | 84 (26.8) |
| Preoperative Hgb, g/dL, mean (±SD) | 12.6 (1.9) |
| Neoadjuvant Chemotherapy, n (%) | 4 (36.3) |
| Patients | Sex-Sparing RARC (11) |
|---|---|
| Operative time, min, median (IQR) | 255 (250–399) |
| Hgb at discharge, g/dL, median (IQR) | 10.8 (9.1–11.9) |
| Hospital stay, days, median (IQR) | 7 (7–12) |
| Complications, n (%) | 4 (36.3) |
| Clavien Low grade (1–2) | 4 (36.3) |
| Clavien High grade (≥3) | 0 (0) |
| pT stage, n (%) | |
| 0, a, is | 6 (54.6) |
| 1 | 1 (9.1) |
| 2 | - |
| 3 | 4 (36.3) |
| 4 | - |
| pN stage, n (%) | |
| 0 | 11(100) |
| 1 | - |
| 2 | - |
| Lymph node count, mean (±SD) | 26.2 (14.3) |
| Positive surgical margins, n (%) | 0 (0) |
| Follow-up, months, median (IQR) | 28 (14–51) |
| 1-Year recurrence-free survival, n (%) | 11 (100) |
| 1-Year cancer-specific survival, n (%) | 11 (100) |
| 1-Year overall survival, n (%) | 11 (100) |
| Last eGFR, mL/min, mean (±SD) | 79.2 (23.7) |
| Ureteroileal strictures, pts (%) | 1 (9) |
| Neobladder stones, n (%) | 1 (9) |
| Need for intermittent self-catheterization, n (%) | 3 (27.2) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tuderti, G.; Mastroianni, R.; Flammia, S.; Ferriero, M.; Leonardo, C.; Anceschi, U.; Brassetti, A.; Guaglianone, S.; Gallucci, M.; Simone, G. Sex-Sparing Robot-Assisted Radical Cystectomy with Intracorporeal Padua Ileal Neobladder in Female: Surgical Technique, Perioperative, Oncologic and Functional Outcomes. J. Clin. Med. 2020, 9, 577. https://doi.org/10.3390/jcm9020577
Tuderti G, Mastroianni R, Flammia S, Ferriero M, Leonardo C, Anceschi U, Brassetti A, Guaglianone S, Gallucci M, Simone G. Sex-Sparing Robot-Assisted Radical Cystectomy with Intracorporeal Padua Ileal Neobladder in Female: Surgical Technique, Perioperative, Oncologic and Functional Outcomes. Journal of Clinical Medicine. 2020; 9(2):577. https://doi.org/10.3390/jcm9020577
Chicago/Turabian StyleTuderti, Gabriele, Riccardo Mastroianni, Simone Flammia, Mariaconsiglia Ferriero, Costantino Leonardo, Umberto Anceschi, Aldo Brassetti, Salvatore Guaglianone, Michele Gallucci, and Giuseppe Simone. 2020. "Sex-Sparing Robot-Assisted Radical Cystectomy with Intracorporeal Padua Ileal Neobladder in Female: Surgical Technique, Perioperative, Oncologic and Functional Outcomes" Journal of Clinical Medicine 9, no. 2: 577. https://doi.org/10.3390/jcm9020577
APA StyleTuderti, G., Mastroianni, R., Flammia, S., Ferriero, M., Leonardo, C., Anceschi, U., Brassetti, A., Guaglianone, S., Gallucci, M., & Simone, G. (2020). Sex-Sparing Robot-Assisted Radical Cystectomy with Intracorporeal Padua Ileal Neobladder in Female: Surgical Technique, Perioperative, Oncologic and Functional Outcomes. Journal of Clinical Medicine, 9(2), 577. https://doi.org/10.3390/jcm9020577







