Overexpression of IL-10 Enhances the Efficacy of Human Umbilical-Cord-Derived Mesenchymal Stromal Cells in E. coli Pneumosepsis
Abstract
1. Introduction
2. Experimental Section
2.1. Human Mesenchymal Stromal Cells
2.2. E. coli Pneumonia Model
2.3. Ex Vivo Analyses
2.4. Assessment of Macrophage Function
2.5. Statistical Analysis
3. Results
3.1. IL-10 UC-MSC Characterization
3.2. Effects in E. coli Pneumosepsis
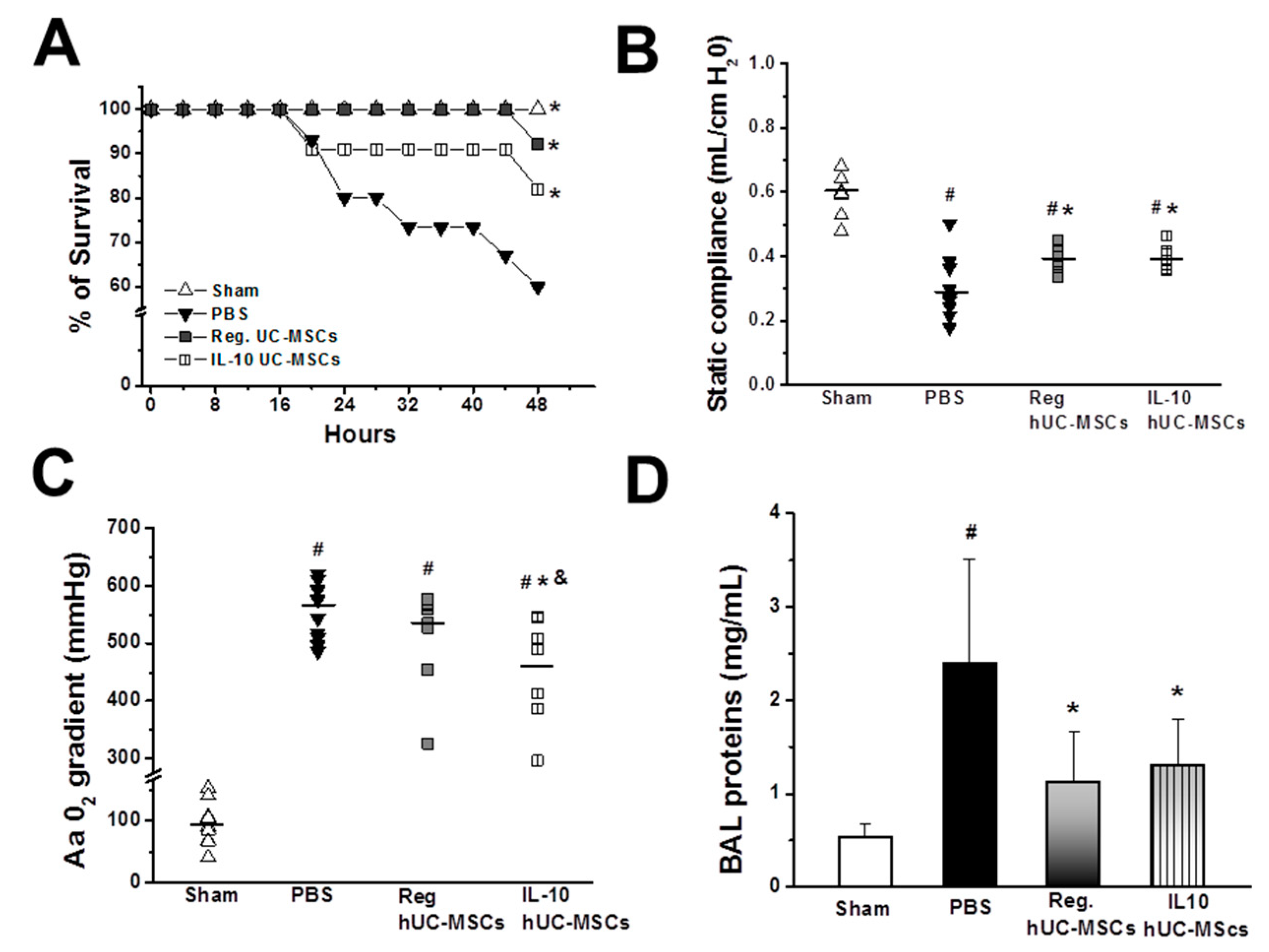
3.2.1. Animal Survival
3.2.2. Lung Injury Severity
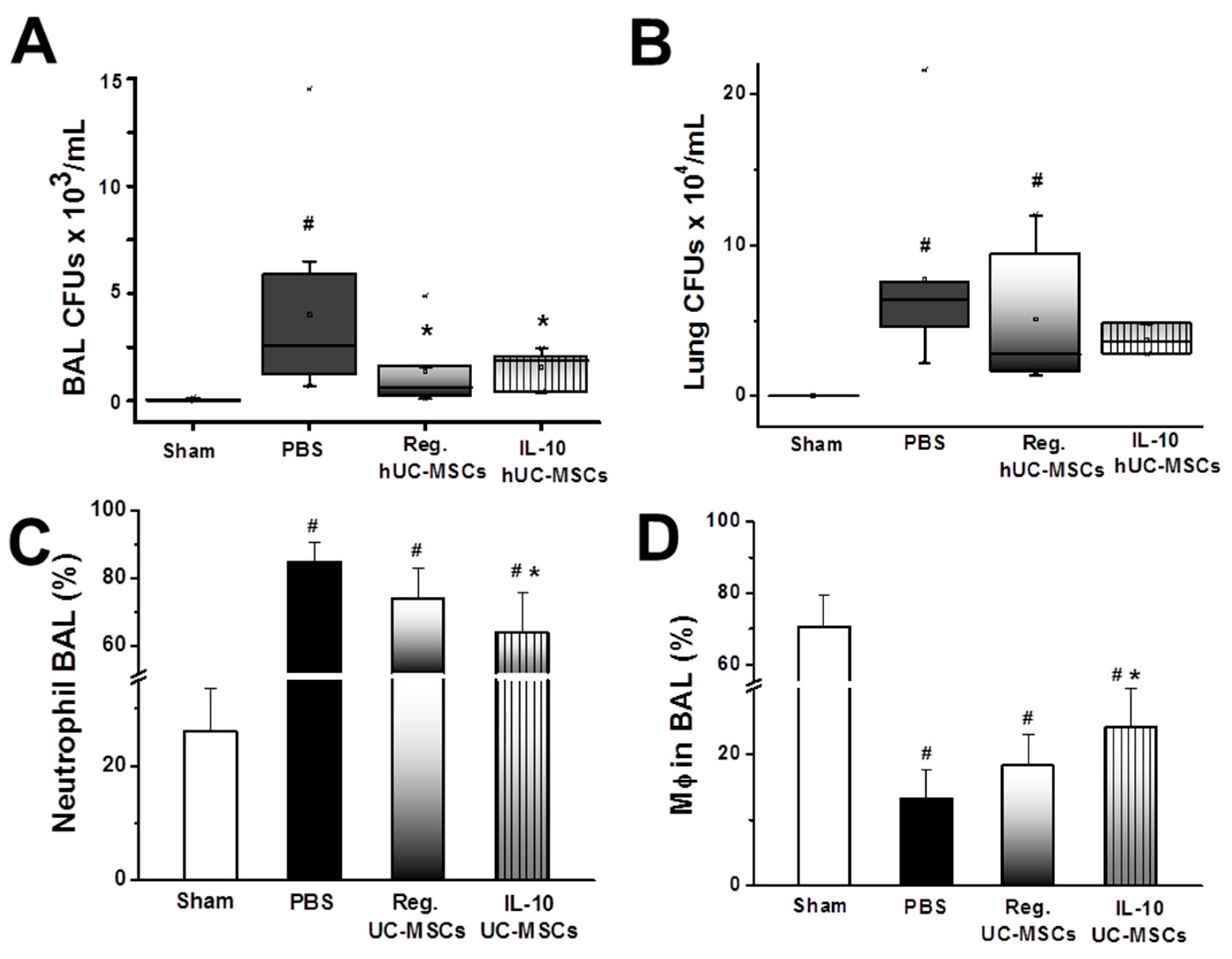
3.2.3. Bacterial Burden
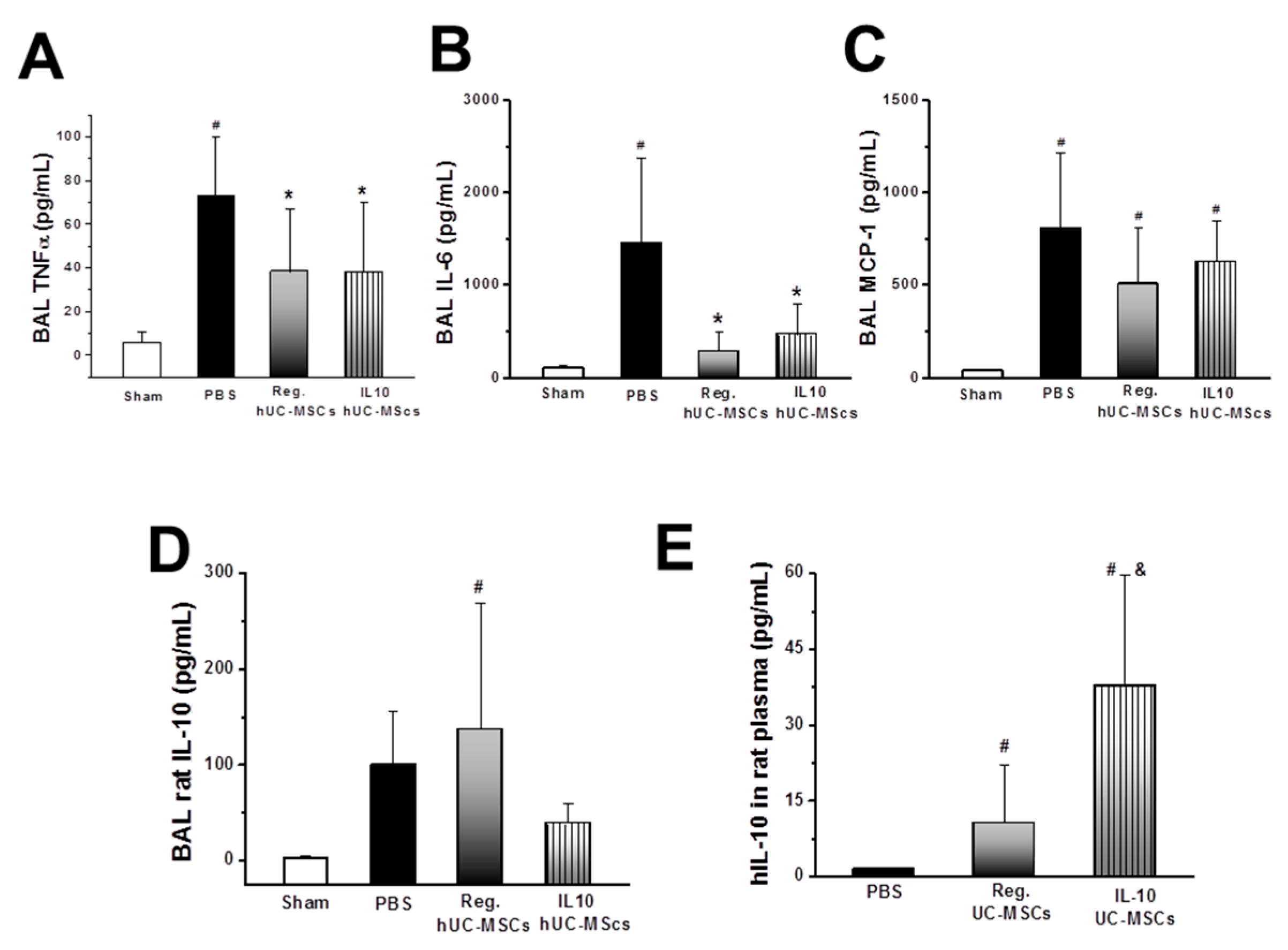
3.2.4. Inflammatory Response
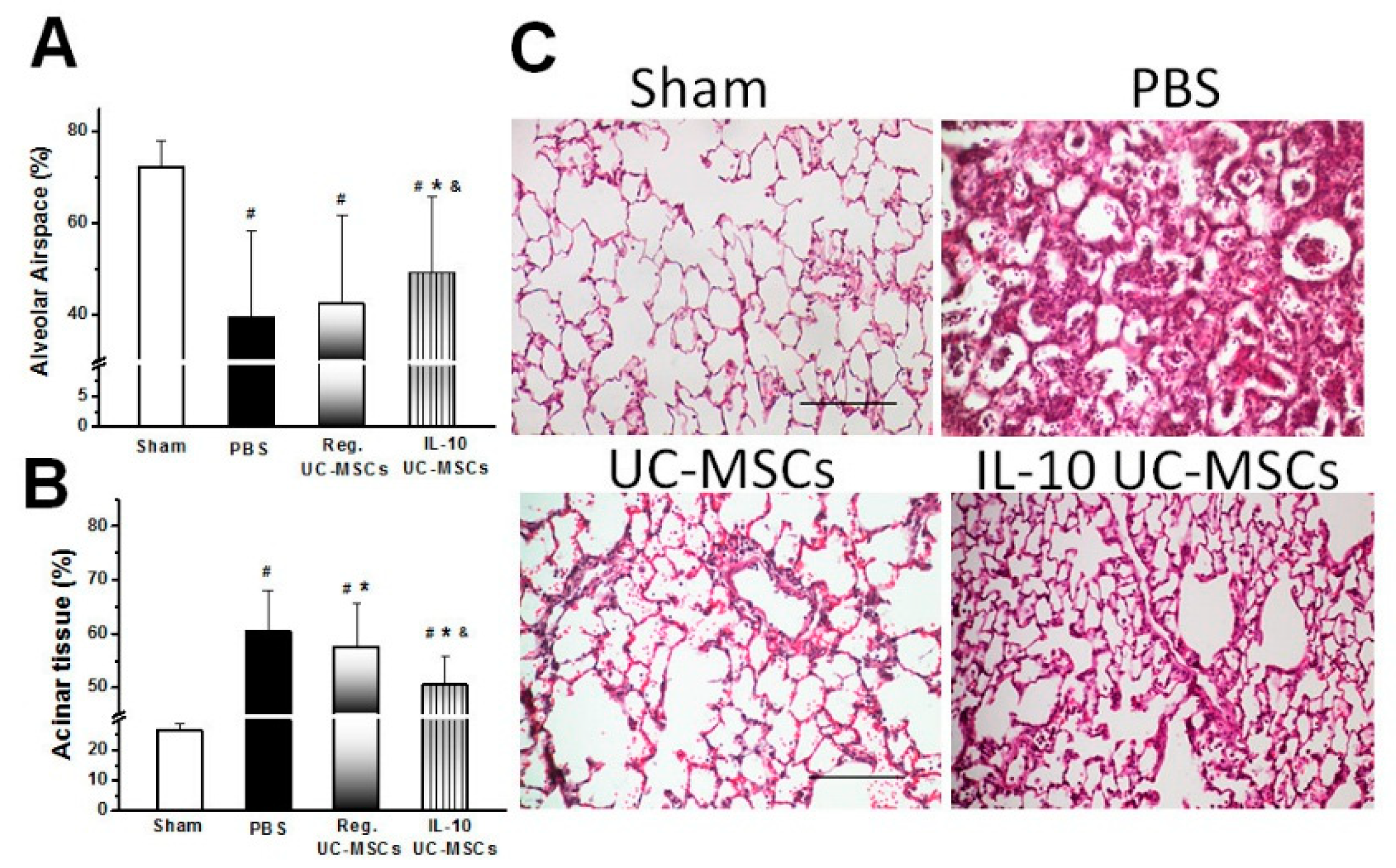
3.2.5. Structural Lung Injury
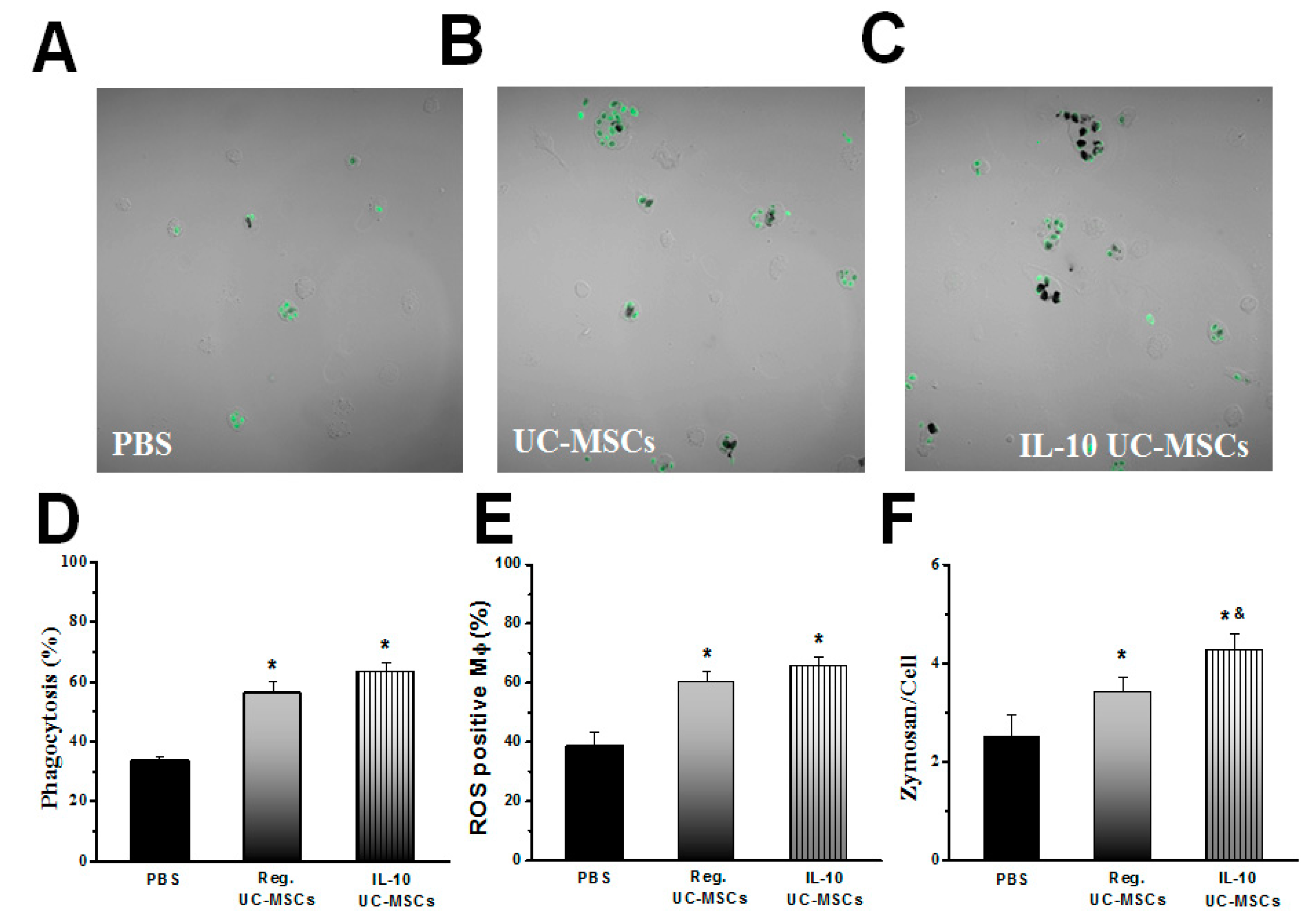
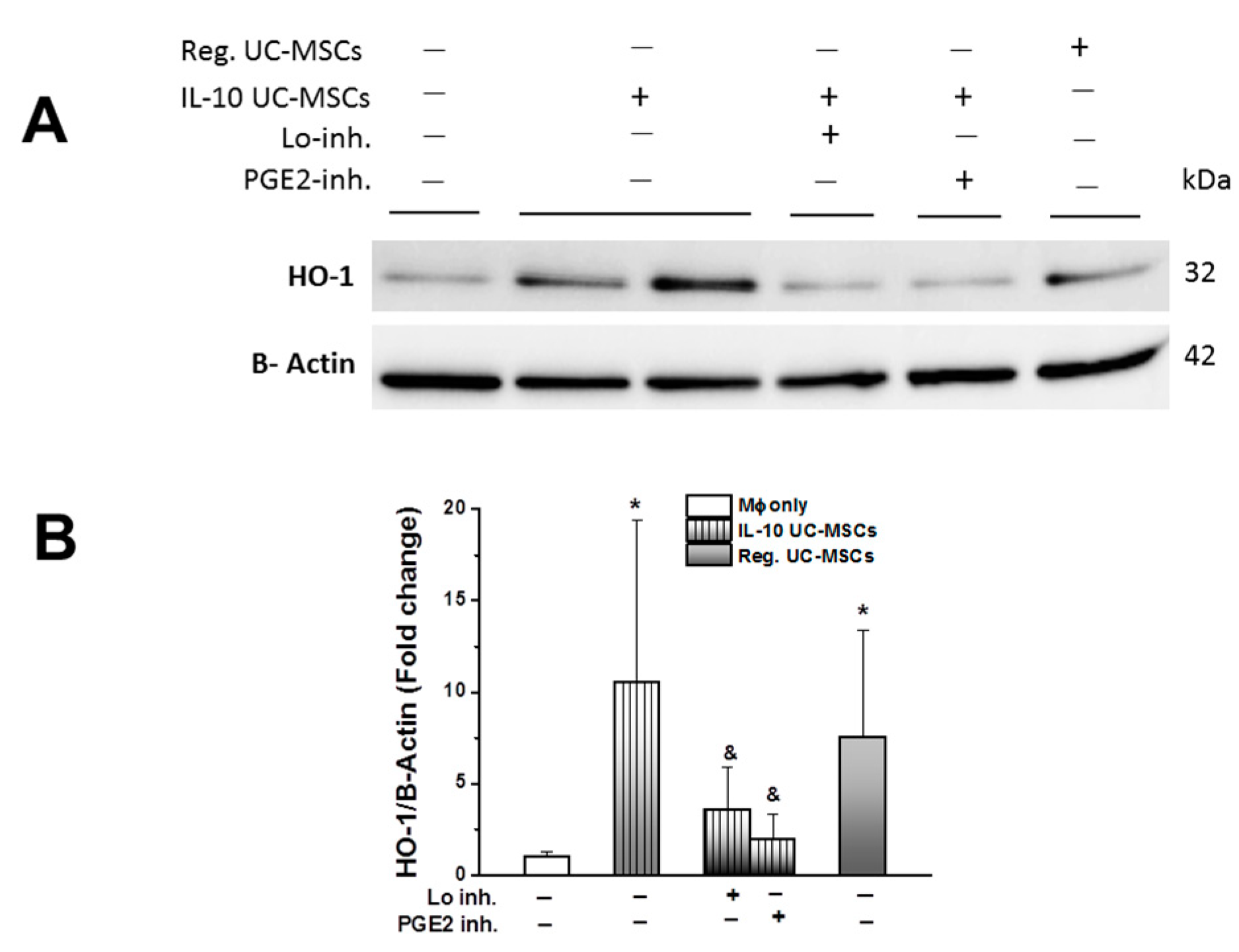
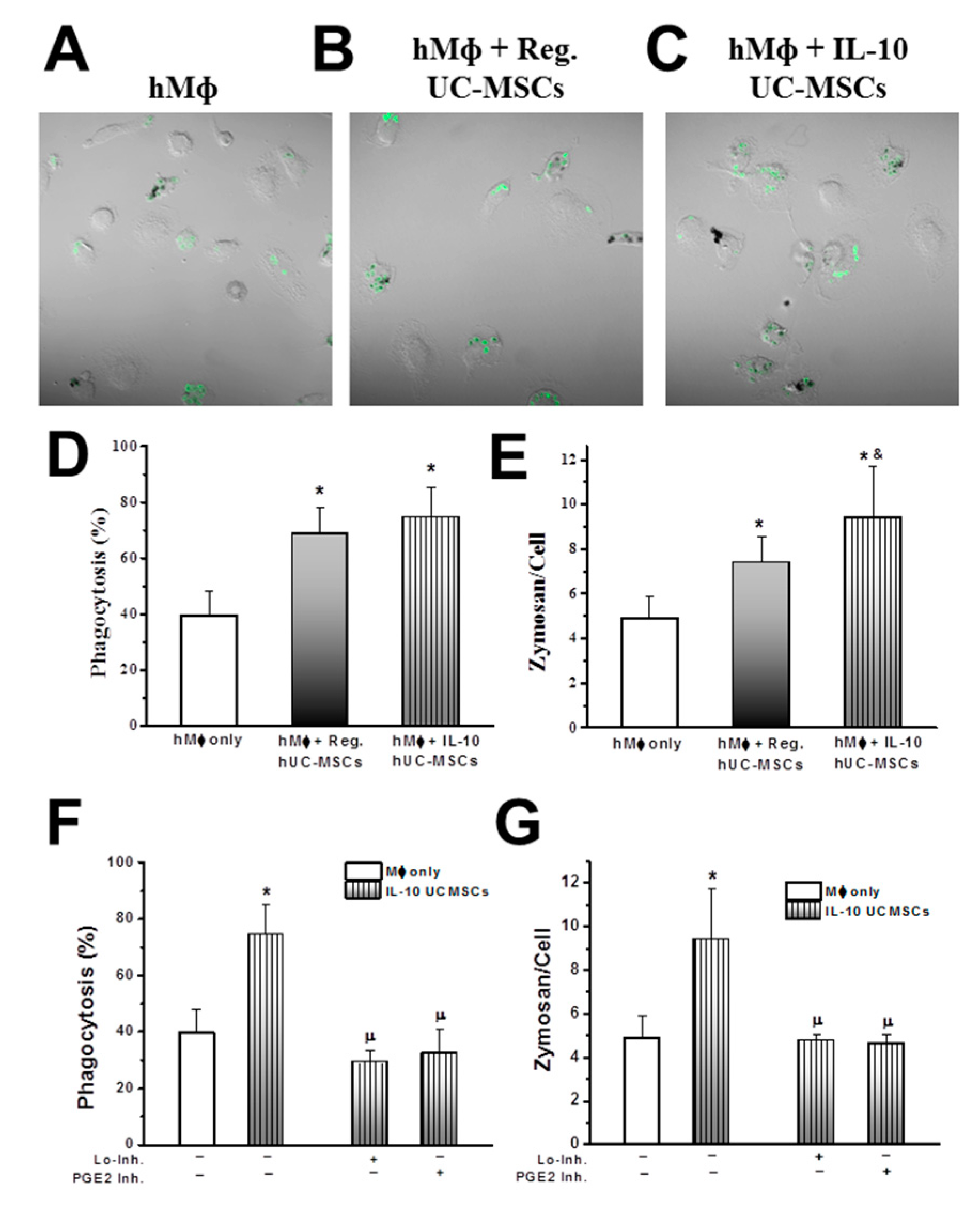
3.3. Effects on Alveolar Macrophage Function
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Keane, C.; Jerkic, M.; Laffey, J.G. Stem cell-based therapies for sepsis. Anesthesiology 2017, 127, 1017–1034. [Google Scholar] [CrossRef] [PubMed]
- Laffey, J.G.; Matthay, M.A. Fifty years of research in ARDS: Cell-based therapy for acute respiratory distress syndrome, biology and potential therapeutic value. Am. J. Respir. Crit. Care Med. 2017, 196, 266–273. [Google Scholar] [CrossRef] [PubMed]
- Curley, G.F.; Jerkic, M.; Dixon, S.; Hogan, G.; Masterson, C.; O’Toole, D.; Devaney, J.; Laffey, J.G. Cryopreserved, xeno-free human umbilical cord mesenchymal stromal cells reduce lung injury severity and bacterial burden in rodent Escherichia coli-induced acute respiratory distress syndrome. Crit. Care Med. 2017, 45, e202–e212. [Google Scholar] [CrossRef] [PubMed]
- Curley, G.; Hayes, M.; Ansari, B.; Shaw, G.; Ryan, A.; Barry, F.; O’Brien, T.; O’Toole, D.; Laffey, J. Mesenchymal stem cells enhance recovery and repair following ventilator-induced lung injury in the rat. Thorax 2012, 67, 496–501. [Google Scholar] [CrossRef] [PubMed]
- Curley, G.F.; Ansari, B.; Hayes, M.; Devaney, J.; Masterson, C.; Ryan, A.; Barry, F.; O’Brien, T.; O’Toole, D.; Laffey, J.G. Effects of intratracheal mesenchymal stromal cell therapy during recovery and resolution after ventilator-induced lung injury. Anesthesiology 2013, 118, 924–932. [Google Scholar] [CrossRef]
- Devaney, J.; Horie, S.; Masterson, C.; Elliman, E.; Barry, F.; O’Brien, T.; Curley, G.; Toole, D.; Laffey, J. Human mesenchymal stromal cells decrease the severity of acute lung injury induced by E. coli in the rat. Thorax 2015, 70, 625–635. [Google Scholar] [CrossRef]
- Hayes, M.; Curley, G.F.; Masterson, C.; Devaney, J.; O’Toole, D.; Laffey, J.G. Mesenchymal stromal cells are more effective than the MSC secretome in diminishing injury and enhancing recovery following ventilator-induced lung injury. Intensive Care Med. Exp. 2015, 3, 29. [Google Scholar] [CrossRef]
- Hayes, M.; Masterson, C.; Devaney, J.; Barry, F.; Elliman, S.; O’Brien, T.; O’Toole, D.; Curley, G.F.; Laffey, J.G. Therapeutic efficacy of human mesenchymal stromal cells in the repair of established ventilator-induced lung injury in the rat. Anesthesiology 2015, 122, 363–373. [Google Scholar] [CrossRef]
- Oggu, G.S.; Sasikumar, S.; Reddy, N.; Ella, K.K.R.; Rao, C.M.; Bokara, K.K. Gene delivery approaches for mesenchymal stem cell therapy: Strategies to increase efficiency and specificity. Stem Cell Rev. 2017, 13, 725–740. [Google Scholar] [CrossRef]
- Zhao, H.Q.; Li, W.M.; Lu, Z.Q.; Sheng, Z.Y.; Yao, Y.M. The growing spectrum of anti-inflammatory interleukins and their potential roles in the development of sepsis. J. Interferon Cytokine Res. 2015, 35, 242–251. [Google Scholar] [CrossRef]
- Li, H.D.; Zhang, Q.X.; Mao, Z.; Xu, X.J.; Li, N.Y.; Zhang, H. Exogenous interleukin-10 attenuates hyperoxia-induced acute lung injury in mice. Exp. Physiol. 2015, 100, 331–340. [Google Scholar] [CrossRef] [PubMed]
- Machuca, T.N.; Cypel, M.; Bonato, R.; Yeung, J.C.; Chun, Y.M.; Juvet, S.; Guan, Z.; Hwang, D.M.; Chen, M.; Saito, T.; et al. Safety and efficacy of ex vivo donor lung adenoviral IL-10 gene therapy in a large animal lung transplant survival model. Hum. Gene Ther. 2017, 28, 757–765. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Lv, D.; Zhang, X.; Ni, Z.A.; Sun, X.; Zhu, C. Interleukin-10-overexpressing mesenchymal stromal cells induce a series of regulatory effects in the inflammatory system and promote the survival of endotoxin-induced acute lung injury in mice model. DNA Cell Biol. 2018, 37, 53–61. [Google Scholar] [CrossRef] [PubMed]
- Manning, E.; Pham, S.; Li, S.; Vazquez-Padron, R.I.; Mathew, J.; Ruiz, P.; Salgar, S.K. Interleukin-10 delivery via mesenchymal stem cells: A novel gene therapy approach to prevent lung ischemia-reperfusion injury. Hum. Gene Ther. 2010, 21, 713–727. [Google Scholar] [CrossRef] [PubMed]
- Meng, X.; Li, J.; Yu, M.; Yang, J.; Zheng, M.; Zhang, J.; Sun, C.; Liang, H.; Liu, L. Transplantation of mesenchymal stem cells overexpressing IL10 attenuates cardiac impairments in rats with myocardial infarction. J. Cell Physiol. 2018, 233, 587–595. [Google Scholar] [CrossRef] [PubMed]
- Sarugaser, R.; Lickorish, D.; Baksh, D.; Hosseini, M.M.; Davies, J.E. Human umbilical cord perivascular (HUCPV) cells: A source of mesenchymal progenitors. Stem Cells 2005, 23, 220–229. [Google Scholar] [CrossRef] [PubMed]
- Sarugaser, R.; Ennis, J.; Stanford, W.L.; Davies, J.E. Isolation, propagation, and characterization of human umbilical cord perivascular cells (HUCPVCs). Methods Mol. Biol. 2009, 482, 269–279. [Google Scholar]
- O’Croinin, D.F.; Nichol, A.D.; Hopkins, N.; Boylan, J.; O’Brien, S.; O’Connor, C.; Laffey, J.G.; McLoughlin, P. Sustained hypercapnic acidosis during pulmonary infection increases bacterial load and worsens lung injury. Crit. Care Med. 2008, 36, 2128–2135. [Google Scholar] [CrossRef]
- Costello, J.; Higgins, B.; Contreras, M.; Chonghaile, M.N.; Hassett, P.; O’Toole, D.; Laffey, J.G. Hypercapnic acidosis attenuates shock and lung injury in early and prolonged systemic sepsis. Crit. Care Med. 2009, 37, 2412–2420. [Google Scholar] [CrossRef]
- Higgins, B.D.; Costello, J.; Contreras, M.; Hassett, P.; O’Toole, D.; Laffey, J.G. Differential effects of buffered hypercapnia versus hypercapnic acidosis on shock and lung injury induced by systemic sepsis. Anesthesiology 2009, 111, 1317–1326. [Google Scholar] [CrossRef]
- Laffey, J.G.; Honan, D.; Hopkins, N.; Hyvelin, J.M.; Boylan, J.F.; McLoughlin, P. Hypercapnic acidosis attenuates endotoxin-induced acute lung injury. Am. J. Respir. Crit. Care Med. 2004, 169, 46–56. [Google Scholar] [CrossRef] [PubMed]
- Jerkic, M.; Peter, M.; Ardelean, D.; Fine, M.; Konerding, M.A.; Letarte, M. Dextran sulfate sodium leads to chronic colitis and pathological angiogenesis in Endoglin heterozygous mice. Inflamm. Bowel Dis. 2010, 16, 1859–1870. [Google Scholar] [CrossRef] [PubMed]
- Canton, J.; Khezri, R.; Glogauer, M.; Grinstein, S. Contrasting phagosome pH regulation and maturation in human M1 and M2 macrophages. Mol. Biol. Cell 2014, 25, 3330–3341. [Google Scholar] [CrossRef] [PubMed]
- Rabani, R.; Volchuk, A.; Jerkic, M.; Ormesher, L.; Garces-Ramirez, L.; Canton, J.; Masterson, C.; Gagnon, S.; Tatham, K.C.; Marshall, J.; et al. Mesenchymal stem cells enhance NOX2-dependent reactive oxygen species production and bacterial killing in macrophages during sepsis. Eur. Respir. J. 2018, 51. [Google Scholar] [CrossRef] [PubMed]
- Masterson, C.; Devaney, J.; Horie, S.; O’Flynn, L.; Deedigan, L.; Elliman, S.; Barry, F.; O’Brien, T.; O’Toole, D.; Laffey, J.G. Syndecan-2-positive, bone marrow-derived human mesenchymal stromal cells attenuate bacterial-induced acute lung injury and enhance resolution of ventilator-induced lung injury in rats. Anesthesiology 2018, 129, 502–516. [Google Scholar] [CrossRef] [PubMed]
- Krasnodembskaya, A.; Song, Y.; Fang, X.; Gupta, N.; Serikov, V.; Lee, J.; Matthay, M. Antibacterial effect of human mesenchymal stem cells is mediated in part from secretion of the antimicrobial peptide LL-37. Stem Cells 2010, 28, 2229–2238. [Google Scholar] [CrossRef] [PubMed]
- Jackson, M.V.; Morrison, T.J.; Doherty, D.F.; McAuley, D.F.; Matthay, M.A.; Kissenpfennig, A.; O’Kane, C.M.; Krasnodembskaya, A.D. Mitochondrial transfer via tunneling nanotubes is an important mechanism by which mesenchymal stem cells enhance macrophage phagocytosis in the in vitro and in vivo models of ARDS. Stem Cells 2016, 34, 14. [Google Scholar] [CrossRef] [PubMed]
- McAuley, D.F.; Curley, G.F.; Hamid, U.I.; Laffey, J.G.; Abbott, J.; McKenna, D.H.; Fang, X.; Matthay, M.A.; Lee, J.W. Clinical grade allogeneic human mesenchymal stem cells restore alveolar fluid clearance in human lungs rejected for transplantation. Am. J. Physiol Lung Cell Mol. Physiol. 2014, 306, L809–L815. [Google Scholar] [CrossRef]
- Islam, M.N.; Das, S.R.; Emin, M.T.; Wei, M.; Sun, L.; Westphalen, K.; Rowlands, D.J.; Quadri, S.K.; Bhattacharya, S.; Bhattacharya, J. Mitochondrial transfer from bone-marrow-derived stromal cells to pulmonary alveoli protects against acute lung injury. Nat. Med. 2012, 18, 759–765. [Google Scholar] [CrossRef]
- Fang, X.; Abbott, J.; Cheng, L.; Colby, J.K.; Lee, J.W.; Levy, B.D.; Matthay, M.A. Human mesenchymal stem (stromal) cells promote the resolution of acute lung injury in part through lipoxin A4. J. Immunol. 2015, 195, 7. [Google Scholar] [CrossRef]
- Varkouhi, A.; Jerkic, M.; Ormesher, L.; Gagnon, S.; Goyal, S.; Rabani, R.; Chen, P.; Gu, F.; dos Santos, C.; Curley, G.F.; et al. Extracellular vesicles derived from Interferon-g primed human umbilical cord derived mesenchymal stromal cells demonstrate enhanced efficacy in pneumonia induced ARDS. Anesthesiology 2019, 130, 778–790. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Li, M.; Cheng, H.; Yan, Z.; Cao, J.; Pan, B.; Sang, W.; Wu, Q.; Zeng, L.; Li, Z.; et al. Overexpression of the mesenchymal stem cell Cxcr4 gene in irradiated mice increases the homing capacity of these cells. Cell Biochem. Biophys. 2013, 67, 1181–1191. [Google Scholar] [CrossRef] [PubMed]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jerkic, M.; Masterson, C.; Ormesher, L.; Gagnon, S.; Goyal, S.; Rabani, R.; Otulakowski, G.; Zhang, H.; Kavanagh, B.P.; Laffey, J.G. Overexpression of IL-10 Enhances the Efficacy of Human Umbilical-Cord-Derived Mesenchymal Stromal Cells in E. coli Pneumosepsis. J. Clin. Med. 2019, 8, 847. https://doi.org/10.3390/jcm8060847
Jerkic M, Masterson C, Ormesher L, Gagnon S, Goyal S, Rabani R, Otulakowski G, Zhang H, Kavanagh BP, Laffey JG. Overexpression of IL-10 Enhances the Efficacy of Human Umbilical-Cord-Derived Mesenchymal Stromal Cells in E. coli Pneumosepsis. Journal of Clinical Medicine. 2019; 8(6):847. https://doi.org/10.3390/jcm8060847
Chicago/Turabian StyleJerkic, Mirjana, Claire Masterson, Lindsay Ormesher, Stéphane Gagnon, Sakshi Goyal, Razieh Rabani, Gail Otulakowski, Haibo Zhang, Brian P. Kavanagh, and John G. Laffey. 2019. "Overexpression of IL-10 Enhances the Efficacy of Human Umbilical-Cord-Derived Mesenchymal Stromal Cells in E. coli Pneumosepsis" Journal of Clinical Medicine 8, no. 6: 847. https://doi.org/10.3390/jcm8060847
APA StyleJerkic, M., Masterson, C., Ormesher, L., Gagnon, S., Goyal, S., Rabani, R., Otulakowski, G., Zhang, H., Kavanagh, B. P., & Laffey, J. G. (2019). Overexpression of IL-10 Enhances the Efficacy of Human Umbilical-Cord-Derived Mesenchymal Stromal Cells in E. coli Pneumosepsis. Journal of Clinical Medicine, 8(6), 847. https://doi.org/10.3390/jcm8060847




