Dietary Profiles, Nutritional Biochemistry Status, and Attention-Deficit/Hyperactivity Disorder: Path Analysis for a Case-Control Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Subjects
2.2. Measurements
2.2.1. Demographics and Dietary Assessment
2.2.2. Food Intake Frequency Assessments
2.2.3. Food Preference Assessments
2.2.4. Nutritional Biomarker Assessment
2.3. Statistical Analysis
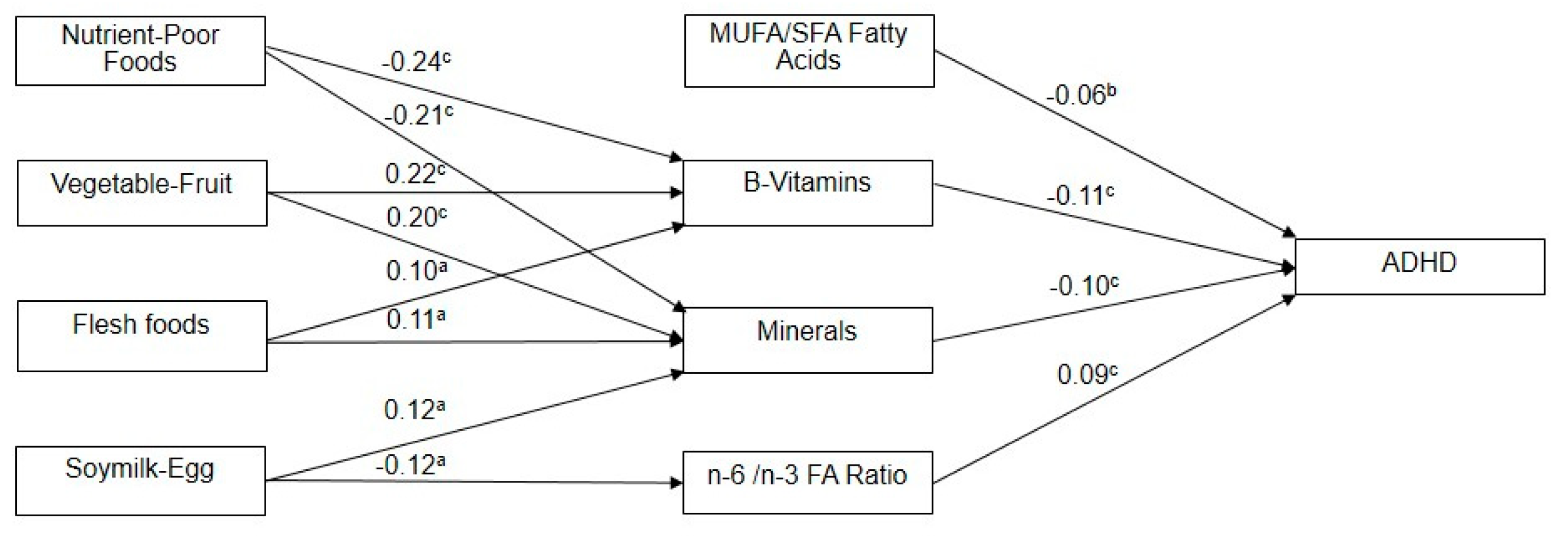
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Thapar, A.; Cooper, M. Attention deficit hyperactivity disorder. Lancet 2016, 387, 1240–1250. [Google Scholar] [CrossRef]
- Polanczyk, G.V.; Willcutt, E.G.; Salum, G.A.; Kieling, C.; Rohde, L.A. ADHD prevalence estimates across three decades: An updated systematic review and meta-regression analysis. Int. J. Epidemiol. 2014, 43, 434–442. [Google Scholar] [CrossRef] [PubMed]
- Gau, S.S.; Chong, M.Y.; Chen, T.H.; Cheng, A.T. A 3-year panel study of mental disorders among adolescents in Taiwan. Am. J. Psychiatry 2005, 162, 1344–1350. [Google Scholar] [CrossRef]
- Stevenson, J. Dietary influences on cognitive development and behaviour in children. Proc. Nutr. Soc. 2006, 65, 361–365. [Google Scholar] [CrossRef]
- Sinn, N. Nutritional and dietary influences on attention deficit hyperactivity disorder. Nutr. Rev. 2008, 66, 558–568. [Google Scholar] [CrossRef]
- Millichap, J.G.; Yee, M.M. The diet factor in attention-deficit/hyperactivity disorder. Pediatrics 2012, 129, 330–337. [Google Scholar] [CrossRef]
- Ly, V.; Bottelier, M.; Hoekstra, P.J.; Arias Vasquez, A.; Buitelaar, J.K.; Rommelse, N.N. Elimination diets’ efficacy and mechanisms in attention deficit hyperactivity disorder and autism spectrum disorder. Eur. Child Adolesc. Psychiatry 2017, 26, 1067–1079. [Google Scholar] [CrossRef]
- Thapar, A.; Cooper, M.; Jefferies, R.; Stergiakouli, E. What causes attention deficit hyperactivity disorder? Arch. Dis. Child. 2012, 97, 260–265. [Google Scholar] [PubMed]
- Fu, M.L.; Cheng, L.; Tu, S.H.; Pan, W.H. Association between unhealthful eating patterns and unfavorable overall school performance in children. J. Am. Diet. Assoc. 2007, 107, 1935–1943. [Google Scholar] [CrossRef]
- Park, S.; Cho, S.C.; Hong, Y.C.; Oh, S.Y.; Kim, J.W.; Shin, M.S.; Kim, B.N.; Yoo, H.J.; Cho, I.H.; Bhang, S.Y. Association between dietary behaviors and attention-deficit/hyperactivity disorder and learning disabilities in school-aged children. Psychiatry Res. 2012, 198, 468–476. [Google Scholar] [CrossRef] [PubMed]
- San Mauro Martin, I.; Blumenfeld Olivares, J.A.; Garicano Vilar, E.; Echeverry Lopez, M.; Garcia Bernat, M.; Quevedo Santos, Y.; Blanco Lopez, M.; Elortegui Pascual, P.; Borregon Rivilla, E.; Rincon Barrado, M. Nutritional and environmental factors in attention-deficit hyperactivity disorder (ADHD): A cross-sectional study. Nutr. Neurosci 2017, 21, 641–647. [Google Scholar] [CrossRef]
- Bowling, A.; Davison, K.; Haneuse, S.; Beardslee, W.; Miller, D.P. ADHD Medication, Dietary Patterns, Physical Activity, and BMI in Children: A Longitudinal Analysis of the ECLS-K Study. Obesity 2017, 25, 1802–1808. [Google Scholar] [CrossRef]
- Sha’ari, N.; Manaf, Z.A.; Ahmad, M.; Rahman, F.N. Nutritional status and feeding problems in pediatric attention deficit-hyperactivity disorder. Pediatr. Int. 2017, 59, 408–415. [Google Scholar] [CrossRef] [PubMed]
- Bala, K.A.; Dogan, M.; Kaba, S.; Mutluer, T.; Aslan, O.; Dogan, S.Z. Hormone disorder and vitamin deficiency in attention deficit hyperactivity disorder (ADHD) and autism spectrum disorders (ASDs). J. Pediatr. Endocrinol. Metab. 2016, 29, 1077–1082. [Google Scholar] [CrossRef]
- Focker, M.; Antel, J.; Ring, S.; Hahn, D.; Kanal, O.; Ozturk, D.; Hebebrand, J.; Libuda, L. Vitamin D and mental health in children and adolescents. Eur. Child Adolesc. Psychiatry 2017, 26, 1043–1066. [Google Scholar] [CrossRef]
- Wang, Y.; Huang, L.; Zhang, L.; Qu, Y.; Mu, D. Iron Status in Attention-Deficit/Hyperactivity Disorder: A Systematic Review and Meta-Analysis. PLoS ONE 2017, 12, e0169145. [Google Scholar] [CrossRef] [PubMed]
- Percinel, I.; Yazici, K.U.; Ustundag, B. Iron Deficiency Parameters in Children and Adolescents with Attention-Deficit/Hyperactivity Disorder. Child Psychiatry Hum. Dev. 2016, 47, 259–269. [Google Scholar] [CrossRef]
- Chen, J.R.; Hsu, S.F.; Hsu, C.D.; Hwang, L.H.; Yang, S.C. Dietary patterns and blood fatty acid composition in children with attention-deficit hyperactivity disorder in Taiwan. J. Nutr. Biochem. 2004, 15, 467–472. [Google Scholar] [CrossRef]
- Lange, K.W. Dietary factors in the etiology and therapy of attention deficit/hyperactivity disorder. Curr. Opin. Clin. Nutr. Metab. Care 2017, 20, 464–469. [Google Scholar] [CrossRef] [PubMed]
- Pelsser, L.M.; Frankena, K.; Toorman, J.; Rodrigues Pereira, R. Diet and ADHD, Reviewing the Evidence: A Systematic Review of Meta-Analyses of Double-Blind Placebo-Controlled Trials Evaluating the Efficacy of Diet Interventions on the Behavior of Children with ADHD. PLoS ONE 2017, 12, e0169277. [Google Scholar] [CrossRef]
- Wang, L.J.; Yu, Y.H.; Fu, M.L.; Yeh, W.T.; Hsu, J.L.; Yang, Y.H.; Chen, W.J.; Chiang, B.L.; Pan, W.H. Attention deficit-hyperactivity disorder is associated with allergic symptoms and low levels of hemoglobin and serotonin. Sci. Rep. 2018, 8, 10229. [Google Scholar] [CrossRef]
- Chou, W.; Wang, Y.; Chen, Y. The assessment of clinical application of activity rating scales in attention-deficit hyperactivity disorder. Chin. Psychiatry 1993, 7, 162–171. [Google Scholar]
- Lee, M.S.; Pan, W.H.; Liu, K.L.; Yu, M.S. Reproducibility and validity of a Chinese food frequency questionnaire used in Taiwan. Asia Pac. J. Clin. Nutr. 2006, 15, 161–169. [Google Scholar]
- Bates, C.J.; Pentieva, K.D.; Prentice, A. An appraisal of vitamin B6 status indices and associated confounders, in young people aged 4–18 years and in people aged 65 years and over, in two national British surveys. Public Health Nutr. 1999, 2, 529–535. [Google Scholar] [CrossRef]
- Chen, J.W.; Sperling, M.I.; Heminger, L.A. Vitamin B12. In Methods in Clinical Laboratory; Presce, A.J., Kaplan, L.A., Eds.; CV Mosby: St. Louis, MO, USA, 1987; pp. 569–573. [Google Scholar]
- Miale, J.B. Laboratory Medicine: Hematology, 6th ed.; CV Mosby: St. Louis, MO, USA, 1982. [Google Scholar]
- Konig, B.; Oed, M.; Kunz, A.; Schlett, R.; Mack, M. LIAISON Ferritin—An automated chemiluminescent immunoassay for the determination of Ferritin. Anticancer Res. 1999, 19, 2739–2741. [Google Scholar]
- Bligh, E.G.; Dyer, W.J. A rapid method of total lipid extraction and purification. Can. J. Biochem. Physiol. 1959, 37, 911–917. [Google Scholar] [CrossRef]
- Edwards, R.; Peet, M.; Shay, J.; Horrobin, D. Omega-3 polyunsaturated fatty acid levels in the diet and in red blood cell membranes of depressed patients. J. Affect. Disord. 1998, 48, 149–155. [Google Scholar] [CrossRef]
- Khaldoon, N.; Nan, H. Comparative assessment of structural equation modeling and multiple regression research methodologies: E-commerce context. Tour. Manag. 2010, 31, 314–324. [Google Scholar]
- Landaas, E.T.; Aarsland, T.I.; Ulvik, A.; Halmoy, A.; Ueland, P.M.; Haavik, J. Vitamin levels in adults with ADHD. BJPsych Open 2016, 2, 377–384. [Google Scholar] [CrossRef]
- Cruz, J.S.; Kushmerick, C.; Moreira-Lobo, D.C.; Oliveira, F.A. Thiamine deficiency in vitro accelerates A-type potassium current inactivation in cerebellar granule neurons. Neuroscience 2012, 221, 108–114. [Google Scholar] [CrossRef] [PubMed]
- Bener, A.; Kamal, M.; Bener, H.; Bhugra, D. Higher prevalence of iron deficiency as strong predictor of attention deficit hyperactivity disorder in children. Ann. Med. Health Sci. Res. 2014, 4, S291–S297. [Google Scholar] [CrossRef]
- Beard, J. Iron deficiency alters brain development and functioning. J. Nutr. 2003, 133, 1468s–1472s. [Google Scholar] [CrossRef]
- Lozoff, B.; Beard, J.; Connor, J.; Barbara, F.; Georgieff, M.; Schallert, T. Long-lasting neural and behavioral effects of iron deficiency in infancy. Nutr. Rev. 2006, 64, S34–S43, discussion S72–S91. [Google Scholar] [CrossRef]
- Othman, H.; Ammari, M.; Sakly, M.; Abdelmelek, H. Effects of prenatal exposure to WIFI signal (2.45GHz) on postnatal development and behavior in rat: Influence of maternal restraint. Behav. Brain Res. 2017, 326, 291–302. [Google Scholar] [CrossRef]
- Hong, S.H.; Park, S.J.; Lee, S.; Kim, S.; Cho, M.H. Biological effects of inorganic phosphate: Potential signal of toxicity. J. Toxicol. Sci. 2015, 40, 55–69. [Google Scholar] [CrossRef]
- Abdullah, M.M.; Jew, S.; Jones, P.J. Health benefits and evaluation of healthcare cost savings if oils rich in monounsaturated fatty acids were substituted for conventional dietary oils in the United States. Nutr. Rev. 2017, 75, 163–174. [Google Scholar] [CrossRef]
- LaChance, L.; McKenzie, K.; Taylor, V.H.; Vigod, S.N. Omega-6 to Omega-3 Fatty Acid Ratio in Patients with ADHD: A Meta-Analysis. J. Can. Acad. Child Adolesc. Psychiatry 2016, 25, 87–96. [Google Scholar]
- Parletta, N.; Niyonsenga, T.; Duff, J. Omega-3 and Omega-6 Polyunsaturated Fatty Acid Levels and Correlations with Symptoms in Children with Attention Deficit Hyperactivity Disorder, Autistic Spectrum Disorder and Typically Developing Controls. PLoS ONE 2016, 11, e0156432. [Google Scholar] [CrossRef]
- Wu, Q.; Zhou, T.; Ma, L.; Yuan, D.; Peng, Y. Protective effects of dietary supplementation with natural omega-3 polyunsaturated fatty acids on the visual acuity of school-age children with lower IQ or attention-deficit hyperactivity disorder. Nutrition 2015, 31, 935–940. [Google Scholar] [CrossRef]
- Wijendran, V.; Hayes, K.C. Dietary n-6 and n-3 fatty acid balance and cardiovascular health. Annu. Rev. Nutr. 2004, 24, 597–615. [Google Scholar] [CrossRef]
- Laasonen, M.; Hokkanen, L.; Leppamaki, S.; Tani, P.; Erkkila, A.T. Project DyAdd: Fatty acids in adult dyslexia, ADHD, and their comorbid combination. Prostaglandins Leukot. Essent. Fat. Acids 2009, 81, 89–96. [Google Scholar] [CrossRef]
| Control (N = 216) | ADHD (N = 216) | ||
|---|---|---|---|
| Demographic and Anthropometric Variables | Mean (SD) | Mean (SD) | p-Value a,b |
| Age (years) | 9.2 (1.8) | 9.2 (1.7) | 0.95 |
| Male gender, n (%) | 186 (86) | 186 (86) | 1 |
| Height (cm) | 135.2 (10.7) | 135.2 (10.7) | 0.93 |
| Weight (kg) | 33.6 (9.8) | 33.6 (9.8) | 0.95 |
| Body mass index (kg/m2) | 18.0 (3.1) | 18.1 (3.1) | 0.83 |
| Father’s education | 94 (43.5) | 123 (56.9) | 0.005 * |
| (High school or lower), n (%) | |||
| Mother’s education | 121 (56.0) | 147 (68.1) | 0.010 * |
| (High school or lower), n (%) | |||
| Expenditure balanced with revenue (yes), n (%) | 188 (87) | 162 (75) | 0.001 * |
| Nutritional biochemistry | |||
| Vitamins | |||
| Vit.B12 (pmol/L) | 462.6 (151) | 423.5 (150) | 0.007 * |
| Folate (nmol/L) | 19.6 (13.6) | 15.3 (6.3) | <0.001 * |
| Vit.B6 (nmol/L) | 58.7 (29.3) | 49.9 (29.9) | 0.002 * |
| Minerals | |||
| Ferritin (ug/L) | 44.7 (18.8) | 39.9 (17.2) | 0.006 * |
| Pi (mmol/L) | 1.5 (0.2) | 1.6 (0.2) | <0.001 * |
| Na (mmol/L) | 147.1 (5.1) | 146.5 (4.9) | 0.21 |
| K (mmol/L) | 4.3 (0.3) | 4.5 (0.3) | 0.3 |
| Ca (mmol/L) | 2.4 (0.1) | 2.4 (0.2) | 0.15 |
| Mg (mmol/L) | 0.9 (0.1) | 0.9 (0.1) | 0.09 |
| Fatty acids | |||
| SFA (%) | 40.7 (2.3) | 41.2 (2.5) | 0.023 * |
| MUFA (%) | 26.4 (3.2) | 25.4 (3.0) | 0.001 * |
| PUFA (%) | 32.9 (2.0) | 33.4 (2.1) | 0.022 * |
| n-6 FA/ n-3 FA ratio | 8.7 (1.9) | 9.4 (1.7) | <0.001 * |
| Frequency of food intakes (times per week) | |||
| Nutrient-dense foods | |||
| Vegetable | 5.8 (1.6) | 4.8 (2.6) | <0.001 * |
| Fruit | 5.0 (1.8) | 3.5 (2.6) | <0.001 * |
| Milk | 4.2 (2.3) | 3.3 (2.7) | <0.001 * |
| Yogurt | 0.4 (1.1) | 0.4 (1.0) | 0.49 |
| Meat | 6.2 (1.0) | 4.9 (2.1) | <0.001 * |
| Poultry | 3.2 (2.0) | 2.6 (2.2) | 0.003 * |
| Pork | 3.3 (2.2) | 3.2 (2.1) | 0.44 |
| Beef | 0.8 (1.4) | 0.6 (1.1) | 0.07 |
| Fish | 3.3 (2.3) | 2.4 (2.1) | <0.001 * |
| Soy milk | 1.0 (1.6) | 0.6 (1.3) | 0.01 * |
| Other soy products | 1.8 (1.8) | 1.4 (1.8) | 0.06 |
| Eggs | 4.0 (2.0) | 3.2 (2.3) | <0.001 * |
| Nutrient-poor foods | |||
| Fried foods | 0.9 (1.3) | 1.4 (1.8) | 0.001 * |
| Ice cream | 0.6 (1.1) | 0.9 (1.4) | 0.004 * |
| Sugary, high-fat foods | 2.2 (2.1) | 2.6 (2.3) | 0.07 |
| High-fat snacks | 0.9 (1.4) | 1.3 (1.8) | 0.02 * |
| Instant noodle | 0.5 (1.1) | 0.8 (1.4) | 0.01 * |
| Sweetened beverage | 1.9 (1.9) | 2.9 (2.6) | 0.001 * |
| Shaved ice desserts | 0.5 (1.0) | 0.8 (1.5) | 0.005 * |
| Candy and chocolate | 1.3 (1.5) | 1.7 (2.0) | 0.02 * |
| Blood Nutritional Biochemistry Markers | Factor 1: MUFA/SFA Fatty Acids | Factor 2: B-Vitamins | Factor 3: Minerals | Factor 4: n-6/n-3 FA Ratio |
|---|---|---|---|---|
| Vit.B12 (pmol/L) | −0.066 | 0.546 | 0.156 | −0.058 |
| Folate (nmol/L) | 0.085 | 0.807 | 0.061 | −0.043 |
| Vit.B6 (nmol/L) | 0.120 | 0.731 | −0.069 | 0.107 |
| Ferritin (ug/L) | 0.084 | 0.103 | 0.768 | 0.175 |
| Pi (mmol/L) | 0.129 | −0.037 | −0.734 | 0.161 |
| SFA (%) | −0.932 | −0.024 | −0.003 | 0.130 |
| MUFA (%) | 0.929 | 0.092 | −0.059 | 0.072 |
| n-6/n-3 FA ratio | −0.054 | −0.010 | 0.008 | 0.968 |
| Food frequency (times per week) | Factor 1: Nutrient-Poor Foods | Factor 2: Vegetable-Fruit | Factor 3: Flesh foods | Factor 4: Soymilk-Egg |
| Vegetable | −0.126 | 0.795 | 0.176 | −0.043 |
| Fruit | −0.074 | 0.819 | 0.154 | 0.094 |
| Milk | −0.112 | 0.204 | 0.275 | −0.041 |
| Meat | −0.036 | 0.211 | 0.795 | 0.042 |
| Poultry | 0.191 | −0.163 | 0.704 | 0.025 |
| Fish | 0.043 | 0.258 | 0.560 | 0.263 |
| Soy milk | 0.052 | −0.097 | −0.066 | 0.827 |
| Eggs | −0.051 | 0.130 | 0.258 | 0.610 |
| Fried foods | 0.583 | −0.285 | 0.191 | −0.172 |
| Ice cream | 0.748 | 0.018 | −0.009 | −0.038 |
| High-fat snacks | 0.646 | −0.195 | 0.085 | −0.004 |
| Instant noodle | 0.604 | −0.247 | 0.072 | 0.036 |
| Sweetened beverage | 0.505 | −0.321 | 0.024 | −0.024 |
| Shaved ice desserts | 0.744 | 0.161 | −0.053 | 0.086 |
| Candy and chocolate | 0.616 | 0.045 | −0.124 | 0.049 |
| MUFA/SFA | B-Vitamins | Minerals | n-6/n-3 FA Ratio | |||||
|---|---|---|---|---|---|---|---|---|
| r | p-Value | r | p-Value | r | p-Value | r | p-Value | |
| Nutrient-Poor Foods | 0.043 | 0.373 | −0.235 | <0.001 * | −0.205 | <0.001 * | 0.009 | 0.845 |
| Vegetable-Fruit | 0.039 | 0.414 | 0.217 | <0.001 * | 0.195 | <0.001 * | −0.057 | 0.236 |
| Flesh Foods | 0.007 | 0.883 | 0.099 | 0.039 * | 0.110 | 0.022 * | 0.012 | 0.801 |
| Soymilk-Egg | 0.028 | 0.561 | −0.004 | 0.938 | 0.116 | 0.016 * | −0.119 | 0.013 * |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, L.-J.; Yu, Y.-H.; Fu, M.-L.; Yeh, W.-T.; Hsu, J.-L.; Yang, Y.-H.; Yang, H.-T.; Huang, S.-Y.; Wei, I.-L.; Chen, W.J.; et al. Dietary Profiles, Nutritional Biochemistry Status, and Attention-Deficit/Hyperactivity Disorder: Path Analysis for a Case-Control Study. J. Clin. Med. 2019, 8, 709. https://doi.org/10.3390/jcm8050709
Wang L-J, Yu Y-H, Fu M-L, Yeh W-T, Hsu J-L, Yang Y-H, Yang H-T, Huang S-Y, Wei I-L, Chen WJ, et al. Dietary Profiles, Nutritional Biochemistry Status, and Attention-Deficit/Hyperactivity Disorder: Path Analysis for a Case-Control Study. Journal of Clinical Medicine. 2019; 8(5):709. https://doi.org/10.3390/jcm8050709
Chicago/Turabian StyleWang, Liang-Jen, Ya-Hui Yu, Ming-Ling Fu, Wen-Ting Yeh, Jung-Lung Hsu, Yao-Hsu Yang, Hui-Ting Yang, Shih-Yi Huang, Ien-Lan Wei, Wei J. Chen, and et al. 2019. "Dietary Profiles, Nutritional Biochemistry Status, and Attention-Deficit/Hyperactivity Disorder: Path Analysis for a Case-Control Study" Journal of Clinical Medicine 8, no. 5: 709. https://doi.org/10.3390/jcm8050709
APA StyleWang, L.-J., Yu, Y.-H., Fu, M.-L., Yeh, W.-T., Hsu, J.-L., Yang, Y.-H., Yang, H.-T., Huang, S.-Y., Wei, I.-L., Chen, W. J., Chiang, B.-L., & Pan, W.-H. (2019). Dietary Profiles, Nutritional Biochemistry Status, and Attention-Deficit/Hyperactivity Disorder: Path Analysis for a Case-Control Study. Journal of Clinical Medicine, 8(5), 709. https://doi.org/10.3390/jcm8050709







