Elevated CTRP1 Plasma Concentration Is Associated with Sepsis and Pre-Existing Type 2 Diabetes Mellitus in Critically Ill Patients
Abstract
:1. Introduction
2. Experimental Section
3. Results
3.1. CTRP1 Plasma Levels Are Significantly Elevated in Critically Ill Patients as Compared with Healthy Controls
3.2. Elevated CTRP1 Plasma Levels in Critically Ill Patients Are Associated with the Presence of Sepsis
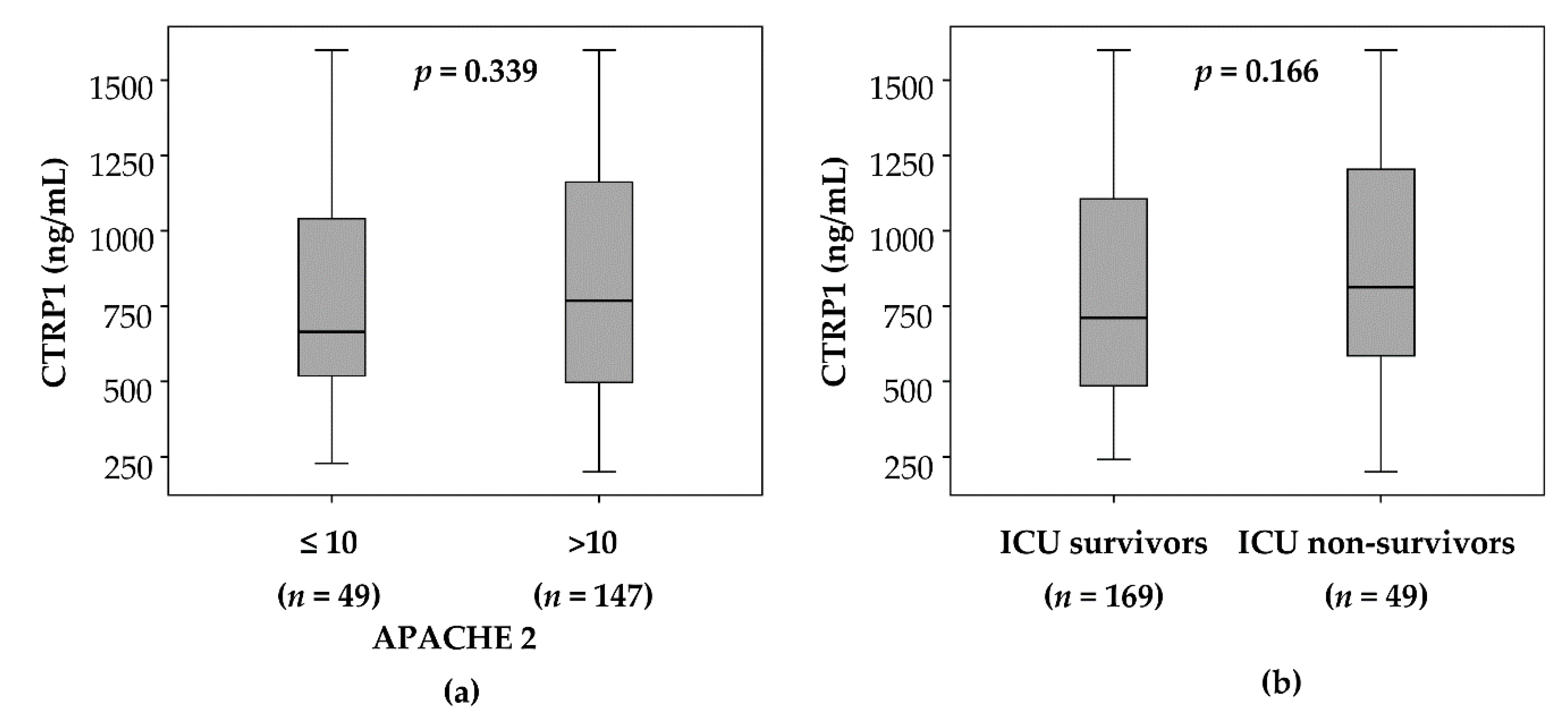
3.3. CTRP1 Plasma Levels in Critically Ill Patients Are Not Associated with Disease Severity or Mortality
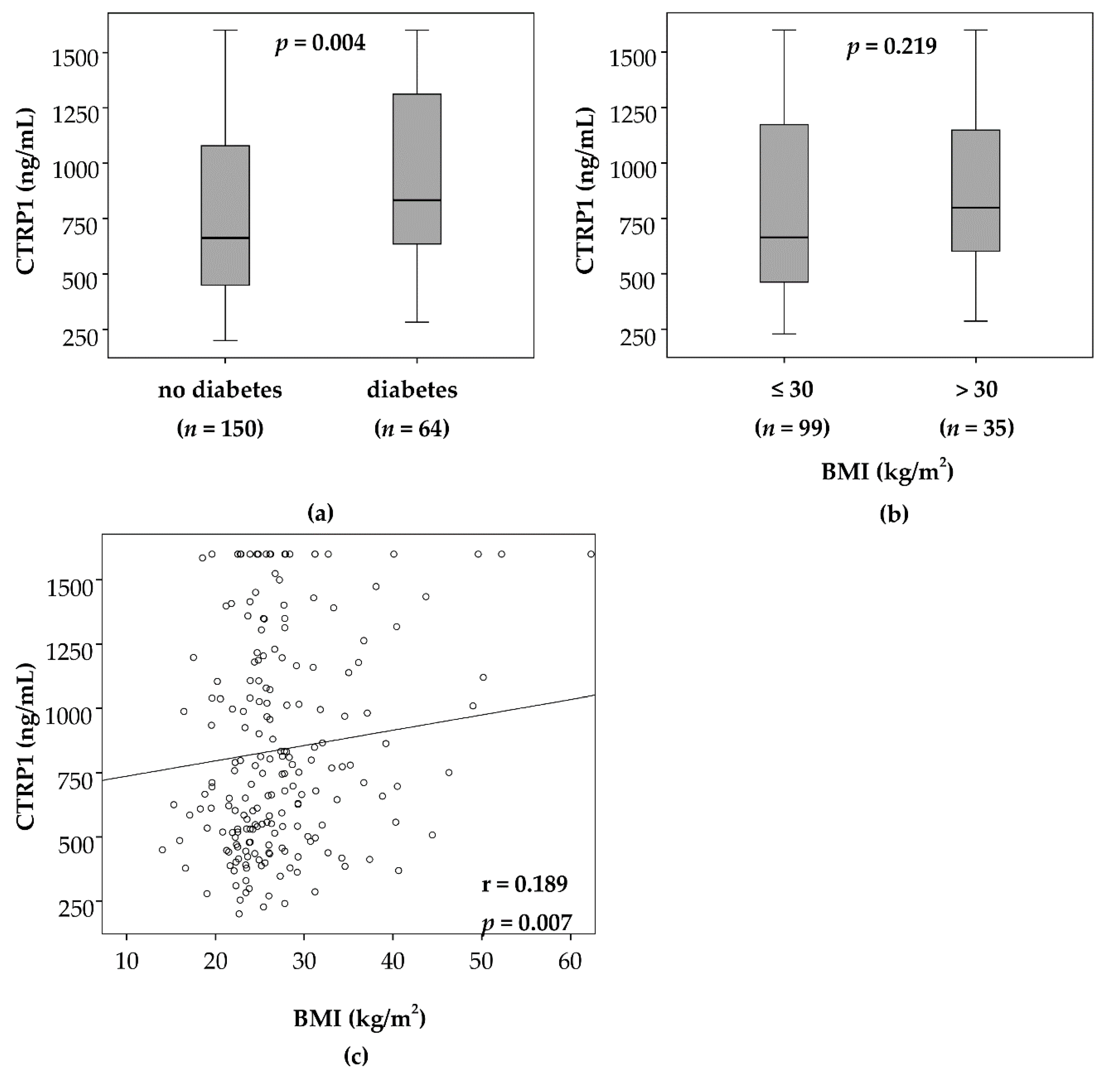
3.4. Elevated CTRP1 Plasma Levels in Critically Ill Patients Are Closely Associated with Diabetic Comorbidity but Not Pre-Existing Obesity
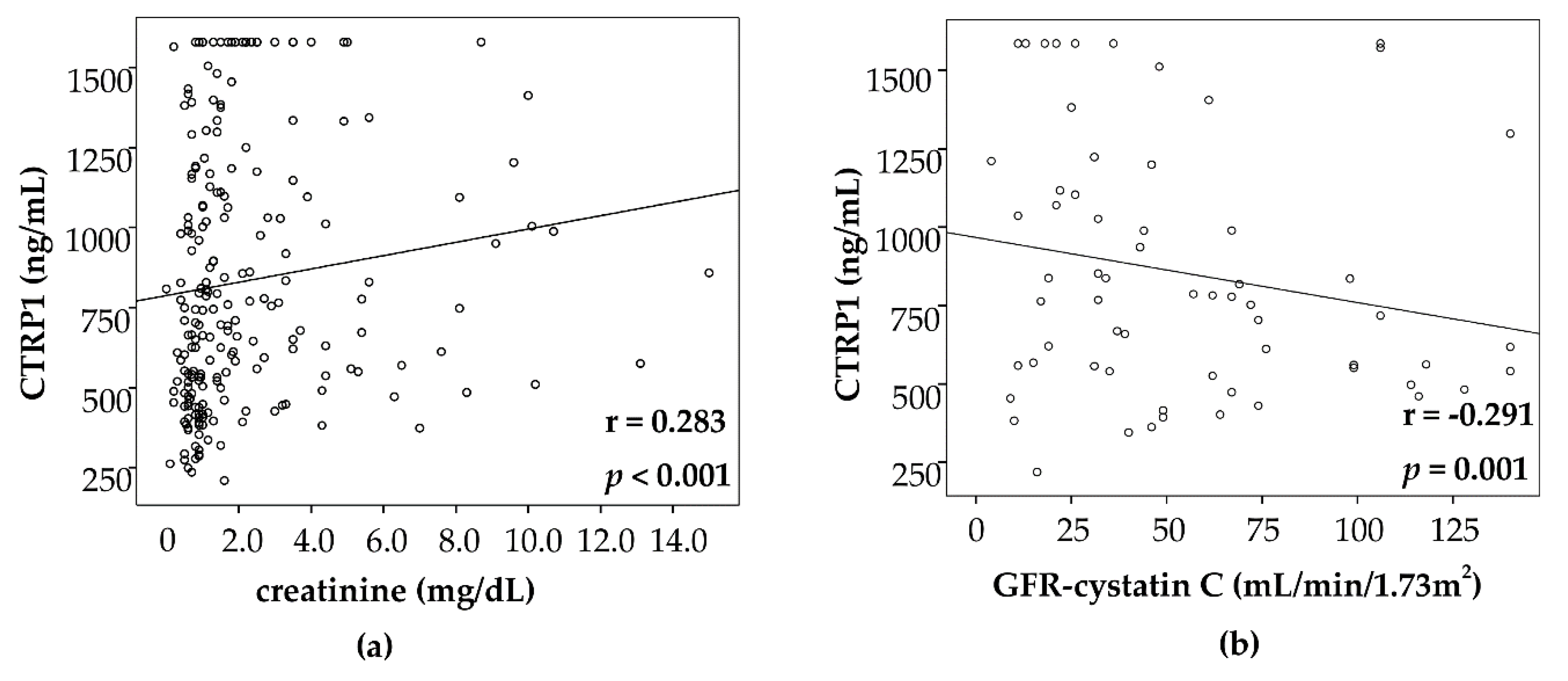
3.5. CTRP1 Levels in Critically Ill Patients Are Correlated with Biomarkers of Inflammation, Cholestasis and Renal Failure
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Bai, B.; Ban, B.; Liu, Z.; Zhang, M.M.; Tan, B.K.; Chen, J. Circulating C1q complement/TNF-related protein (CTRP) 1, CTRP9, CTRP12 and CTRP13 concentrations in Type 2 diabetes mellitus: In vivo regulation by glucose. PLoS ONE 2017, 12, e0172271. [Google Scholar] [CrossRef] [PubMed]
- Peterson, J.M.; Wei, Z.; Wong, G.W. C1q/TNF-related protein-3 (CTRP3); a novel adipokine that regulates hepatic glucose output. J. Biol. Chem. 2010, 285, 39691–39701. [Google Scholar] [CrossRef]
- Schäffler, A.; Buechler, C. CTRP family: Linking immunity to metabolism. Trends Endocrinol. Metab. 2012, 23, 194–204. [Google Scholar] [CrossRef]
- Seldin, M.M.; Tan, S.Y.; Wong, G.W. Metabolic function of the CTRP family of hormones. Rev. Endocr. Metab. Disord. 2014, 15, 111–123. [Google Scholar] [CrossRef]
- Chen, L.; Wu, F.; Yuan, S.; Feng, B. Identification and characteristic of three members of the C1q/TNF-related proteins (CTRPs) superfamily in Eudontomyzon morii. Fish Shellfish Immunol. 2016, 59, 233–240. [Google Scholar] [CrossRef]
- Shabani, P.; Emamgholipour, S.; Doosti, M. CTRP1 in Liver Disease. Adv. Clin. Chem. 2017, 79, 1–23. [Google Scholar] [PubMed]
- Kon, M.; Ebi, Y.; Nakagaki, K. Effects of a single bout of high-intensity interval exercise on C1q/TNF-related proteins. Appl. Physiol. Nutr. Metab. 2019, 44, 47–51. [Google Scholar] [CrossRef]
- Yang, Y.; Liu, S.; Zhang, R.Y.; Luo, H.; Chen, L.; He, W.F.; Lei, R.; Liu, M.R.; Hu, H.X.; Chen, M. Association Between C1q/TNF-Related Protein-1 Levels in Human Plasma and Epicardial Adipose Tissues and Congestive Heart Failure. Cell. Physiol. Biochem. 2017, 42, 2130–2143. [Google Scholar] [CrossRef] [PubMed]
- Han, S.; Yang, Y. A Novel Blood Pressure Modulator C1q/TNF-α-related Protein 1 (CTRP1). BMB Rep. 2018, 51, 611. [Google Scholar] [CrossRef]
- Chen, H.; Gao, L.; Huang, Z.; Liu, Y.; Guo, S.; Xing, J.; Meng, Z.; Liang, C.; Li, Y.; Yao, R.; et al. C1qTNF-related protein 1 attenuates doxorubicin-induced cardiac injury via activation of AKT. Life Sci. 2018, 207, 492–498. [Google Scholar] [CrossRef] [PubMed]
- Peterson, J.M.; Aja, S.; Wei, Z.; Wong, G.W. CTRP1 protein enhances fatty acid oxidation via AMP-activated protein kinase (AMPK) activation and acetyl-CoA carboxylase (ACC) inhibition. J. Biol. Chem. 2012, 287, 1576–1587. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez, S.; Lei, X.; Petersen, P.S.; Tan, S.Y.; Little, H.C.; Wong, G.W. Loss of CTRP1 disrupts glucose and lipid homeostasis. Am. J. Physiol. Endocrinol. Metab. 2016, 311, E678–E697. [Google Scholar] [CrossRef]
- Koch, A.; Weiskirchen, R.; Zimmermann, H.W.; Sanson, E.; Trautwein, C.; Tacke, F. Relevance of serum leptin and leptin-receptor concentrations in critically ill patients. Mediat. Inflamm. 2010, 2010, 473540. [Google Scholar] [CrossRef]
- Chalupova, L.; Zakovska, A.; Adamcova, K. Development of a novel enzyme-linked immunosorbent assay (ELISA) for measurement of serum CTRP1: A pilot study: Measurement of serum CTRP1 in healthy donors and patients with metabolic syndrome. Clin. Biochem. 2013, 46, 73–78. [Google Scholar] [CrossRef] [PubMed]
- Koch, A.; Weiskirchen, R.; Krusch, A.; Bruensing, J.; Buendgens, L.; Herbers, U.; Yagmur, E.; Koek, G.H.; Trautwein, C.; Tacke, F. Visfatin Serum Levels Predict Mortality in Critically Ill Patients. Dis. Markers 2018, 2018, 7315356. [Google Scholar] [CrossRef] [PubMed]
- Koch, A.; Gressner, O.A.; Sanson, E.; Tacke, F.; Trautwein, C. Serum resistin levels in critically ill patients are associated with inflammation.; organ dysfunction and metabolism and may predict survival of non-septic patients. Crit. Care 2009, 13, R95. [Google Scholar] [CrossRef]
- Luedde, M.; Benz, F.; Niedeggen, J.; Vucur, M.; Hippe, H.J.; Spehlmann, M.E.; Schueller, F.; Loosen, S.; Frey, N.; Trautwein, C.; et al. Elevated Omentin Serum Levels Predict Long-Term Survival in Critically Ill Patients. Dis. Markers 2016, 2016, 3149243. [Google Scholar] [CrossRef]
- Koch, A.; Sanson, E.; Voigt, S.; Helm, A.; Trautwein, C.; Tacke, F. Serum adiponectin upon admission to the intensive care unit may predict mortality in critically ill patients. J. Crit. Care 2011, 26, 166–174. [Google Scholar] [CrossRef]
- Kim, D.; Park, S.Y. C1q and TNF related protein 1 regulates expression of inflammatory genes in vascular smooth muscle cells. Genes Genom. 2018. [Google Scholar] [CrossRef]
- Koch, A.; Weiskirchen, R.; Kunze, J.; Dückers, H.; Bruensing, J.; Buendgens, L.; Matthes, M.; Luedde, T.; Trautwein, C.; Tacke, F. Elevated asymmetric dimethylarginine levels predict short- and long-term mortality risk in critically ill patients. J. Crit. Care 2013, 28, 947–953. [Google Scholar] [CrossRef]
- Buendgens, L.; Yagmur, E.; Bruensing, J.; Herbers, U.; Baeck, C.; Trautwein, C.; Koch, A.; Tacke, F. Growth Differentiation Factor-15 Is a Predictor of Mortality in Critically Ill Patients with Sepsis. Dis. Markers 2017, 2017, 5271203. [Google Scholar] [CrossRef]
- Rhodes, A.; Evans, L.E.; Alhazzani, W.; Levy, M.M.; Antonelli, M.; Ferrer, R.; Kumar, A.; Sevransky, J.E.; Sprung, C.L.; Nunnally, M.E.; et al. Surviving Sepsis Campaign: International Guidelines for Management of Sepsis and Septic Shock: 2016. Intensive Care Med. 2017, 43, 304–377. [Google Scholar]
- Koch, A.; Voigt, S.; Kruschinski, C.; Sanson, E.; Dückers, H.; Horn, A.; Yagmur, E.; Zimmermann, H.; Trautwein, C.; Tacke, F. Circulating soluble urokinase plasminogen activator receptor is stably elevated during the first week of treatment in the intensive care unit and predicts mortality in critically ill patients. Crit. Care 2011, 15, R63. [Google Scholar] [CrossRef]
- Feng, S.; Su, Y.; Luo, L.; Jing, F.; Yi, Q. Serum levels of C1q/tumor necrosis factor-related protein-1 in children with Kawasaki disease. Pediatr. Res. 2018, 83, 999–1003. [Google Scholar] [CrossRef] [PubMed]
- Han, S.; Jeong, A.L.; Lee, S.; Park, J.S.; Buyanravjikh, S.; Kang, W.; Choi, S.; Park, C.; Han, J.; Son, W.C.; et al. C1q/TNF-α-Related Protein 1 (CTRP1) Maintains Blood Pressure Under Dehydration Conditions. Circ. Res. 2018, 123, e5–e19. [Google Scholar] [CrossRef]
- Han, S.; Kim, J.D.; Lee, S.; Jeong, A.L.; Park, J.S.; Yong, H.J.; Boldbaatar, A.; Ka, H.I.; Rhee, E.J.; Lee, W.Y.; et al. Circulating CTRP1 Levels in Type 2 Diabetes and Their Association with FGF21. Int. J. Endocrinol. 2016, 2016, 5479627. [Google Scholar] [CrossRef]
- Backes, Y.; van der Sluijs, K.F.; Mackie, D.P.; Tacke, F.; Koch, A.; Tenhunen, J.J.; Schultz, M.J. Usefulness of suPAR as a biological marker in patients with systemic inflammation or infection: A systematic review. Intensive Care Med. 2012, 38, 1418–1428. [Google Scholar] [CrossRef]
- Pan, X.; Lu, T.; Wu, F.; Jin, L.; Zhang, Y.; Shi, L.; Li, X.; Lin, Z. Circulating complement-C1q TNF-related protein 1 levels are increased in patients with type 2 diabetes and are associated with insulin sensitivity in Chinese subjects. PLoS ONE 2014, 9, e94478. [Google Scholar] [CrossRef] [PubMed]
- Han, S.; Park, J.S.; Lee, S.; Jeong, A.L.; Oh, K.S.; Ka, H.I.; Choi, H.J.; Son, W.C.; Lee, W.Y.; Oh, S.J.; et al. CTRP1 protects against diet-induced hyperglycemia by enhancing glycolysis and fatty acid oxidation. J. Nutr. Biochem. 2016, 27, 43–52. [Google Scholar] [CrossRef] [PubMed]
- Jeon, J.H.; Kim, K.Y.; Kim, J.H.; Baek, A.; Cho, H.; Lee, Y.H.; Kim, J.W.; Kim, D.; Han, S.H.; Lim, J.S.; et al. A novel adipokine CTRP1 stimulates aldosterone production. FASEB J. 2008, 22, 1502–1511. [Google Scholar] [CrossRef] [PubMed]
- Lu, L.; Zhang, R.Y.; Wang, X.Q.; Liu, Z.H.; Shen, Y.; Ding, F.H.; Meng, H.; Wang, L.J.; Yan, X.X.; Yang, K.; et al. C1q/TNF-related protein-1: An adipokine marking and promoting atherosclerosis. Eur. Heart J. 2016, 37, 1762–1771. [Google Scholar] [CrossRef] [PubMed]
- Yuasa, D.; Ohashi, K.; Shibata, R.; Mizutani, N.; Kataoka, Y.; Kambara, T.; Uemura, Y.; Matsuo, K.; Kanemura, N.; Hayakawa, S.; et al. C1q/TNF-related protein-1 functions to protect against acute ischemic injury in the heart. FASEB J. 2016, 30, 1065–1075. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.H.; Li, C.; Chen, J.W.; Shen, Y.; Gao, J.; Shen, W.F.; Zhang, R.Y.; Wang, X.Q.; Lu, L. C1q/TNF-related protein 1 promotes endothelial barrier dysfunction under disturbed flow. Biochem. Biophys. Res. Commun. 2017, 490, 580–586. [Google Scholar] [CrossRef] [PubMed]

| Parameter | All Patients | Non-Sepsis | Sepsis | * p |
|---|---|---|---|---|
| Number n | 218 | 73 | 145 | |
| Sex (male/female) n | 133/85 | 48/25 | 85/60 | n.s. |
| Age (years) | 64 (18–90) | 61 (18–85) | 65 (20–90) | n.s. |
| APACHE-II score | 18 (2–43) | 13.5 (2–33) | 19 (4–43) | <0.001 |
| ICU days | 7 (1–137) | 6 (1–45) | 9 (1–137) | 0.004 |
| Death during ICU n (%) | 49 (22.5%) | 9 (12.3%) | 40 (27.6%) | 0.010 |
| Death during follow-up (total) n (%) | 89 (40.8%) | 22 (30.1%) | 67 (46.2%) | 0.026 |
| Mechanical ventilation n (%) | 143 (65.6%) | 46 (63%) | 97 (66.9%) | n.s. |
| Pre-existing diabetes n (%) | 64 (29.4%) | 22 (30.1%) | 42 (29.0%) | n.s. |
| BMI (m2/kg) | 25.8 (14–86) | 25.7 (15.9–40.5) | 28.9 (14–86.5) | n.s. |
| WBC (×103/µL) | 13.1 (0.1–208) | 12.5 (1.8–29.6) | 14.1 (0.1–208) | 0.024 |
| CRP (mg/dL) | 100.5 (5–230) | 17 (5–230) | 164 (5–230) | <0.001 |
| IL-6 (pg/mL) | 150.0 (2–28000) | 66.5 (1.5–5000) | 250 (0.1–28000) | <0.001 |
| Procalcitonin (ng/mL) | 0.7 (0.03–207.5) | 0.2 (0.03–100) | 2.2 (0.1–207.5) | <0.001 |
| Creatinine (mg/dL) | 1.3 (0.1–15) | 1.0 (0.2–15) | 1.5 (0.1–10.7) | 0.017 |
| GFR-Cystatin C (mL/min) | 34 (0–379) | 59 (5–379) | 21.5 (0–218) | <0.001 |
| INR | 1.16 (0.92–13) | 1.17 (0.95–6.73) | 1.16 (0.92–13) | n.s. |
| CTRP1 day 1 (ng/mL) | 747.1 (200.5–1600) | 574.2 (227.2–1600) | 779.6 (200.5–1600) | 0.006 |
| Sepsis | Non-Sepsis | |
|---|---|---|
| n = 145 | n = 73 | |
| Aetiology of sepsis critical illness | ||
| Site of infection n (%) | ||
| Pulmonary | 72 (50%) | |
| Abdominal | 28 (19%) | |
| Urogenital | 11 (8%) | |
| Other | 34 (23%) | |
| Aetiology of non-sepsis critical illness n (%) | ||
| Cardio-pulmonary disorder | 29 (40%) | |
| Acute pancreatitis | 10 (14%) | |
| Acute liver failure | 4 (5.5%) | |
| Decompensated liver cirrhosis | 9 (12%) | |
| Severe gastrointestinal hemorrhage | 4 (5.5%) | |
| Non-sepsis other | 17 (23%) |
| ICU Patients | ||
|---|---|---|
| Parameters | r | p |
| Obesity/diabetes | ||
| BMI | 0.189 | 0.007 |
| HbA1c | 0.301 | 0.011 |
| Inflammatory response | ||
| CRP | 0.238 | <0.001 |
| IL-6 | 0.317 | <0.001 |
| PCT | 0.414 | <0.001 |
| suPAR | 0.279 | 0.001 |
| Renal function | ||
| Urea | 0.324 | <0.001 |
| Creatinine | 0.283 | <0.001 |
| Cystatin C | 0.287 | 0.001 |
| GFR Cystatin C | −0.291 | 0.001 |
| Liver injury/cholestasis | ||
| Bilirubin | 0.422 | <0.001 |
| GLDH | 0.154 | 0.033 |
| γ-GT | 0.243 | <0.001 |
| AP | 0.211 | 0.003 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yagmur, E.; Buergerhausen, D.; Koek, G.H.; Weiskirchen, R.; Trautwein, C.; Koch, A.; Tacke, F. Elevated CTRP1 Plasma Concentration Is Associated with Sepsis and Pre-Existing Type 2 Diabetes Mellitus in Critically Ill Patients. J. Clin. Med. 2019, 8, 661. https://doi.org/10.3390/jcm8050661
Yagmur E, Buergerhausen D, Koek GH, Weiskirchen R, Trautwein C, Koch A, Tacke F. Elevated CTRP1 Plasma Concentration Is Associated with Sepsis and Pre-Existing Type 2 Diabetes Mellitus in Critically Ill Patients. Journal of Clinical Medicine. 2019; 8(5):661. https://doi.org/10.3390/jcm8050661
Chicago/Turabian StyleYagmur, Eray, David Buergerhausen, Ger H. Koek, Ralf Weiskirchen, Christian Trautwein, Alexander Koch, and Frank Tacke. 2019. "Elevated CTRP1 Plasma Concentration Is Associated with Sepsis and Pre-Existing Type 2 Diabetes Mellitus in Critically Ill Patients" Journal of Clinical Medicine 8, no. 5: 661. https://doi.org/10.3390/jcm8050661
APA StyleYagmur, E., Buergerhausen, D., Koek, G. H., Weiskirchen, R., Trautwein, C., Koch, A., & Tacke, F. (2019). Elevated CTRP1 Plasma Concentration Is Associated with Sepsis and Pre-Existing Type 2 Diabetes Mellitus in Critically Ill Patients. Journal of Clinical Medicine, 8(5), 661. https://doi.org/10.3390/jcm8050661






