Short and Long-Term Effectiveness of Ustekinumab in Patients with Crohn’s Disease: Real-World Data from a German IBD Cohort
Abstract
1. Introduction
2. Methods
2.1. Study Design
2.2. Study Population
2.3. Study Variables
2.4. Definitions and Outcomes
2.5. Ethics Statement
2.6. Statistical Analyses
3. Results
3.1. Baseline Characteristics
3.2. Disease Efficacy Follow-up
3.3. Combined Response and Remission Analysis
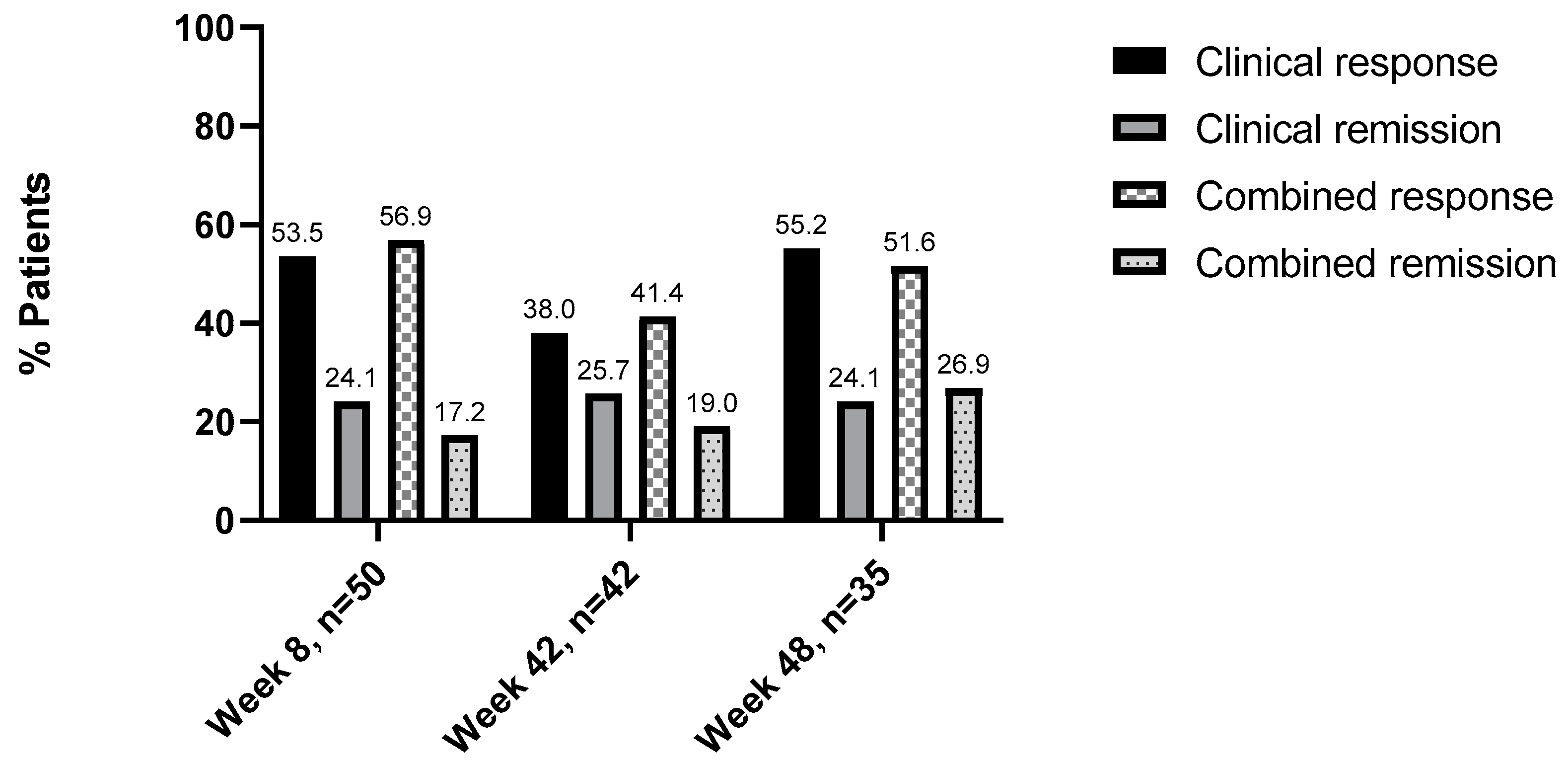
3.4. Comparing the Evaluation of Clinical and Combined Disease Activity
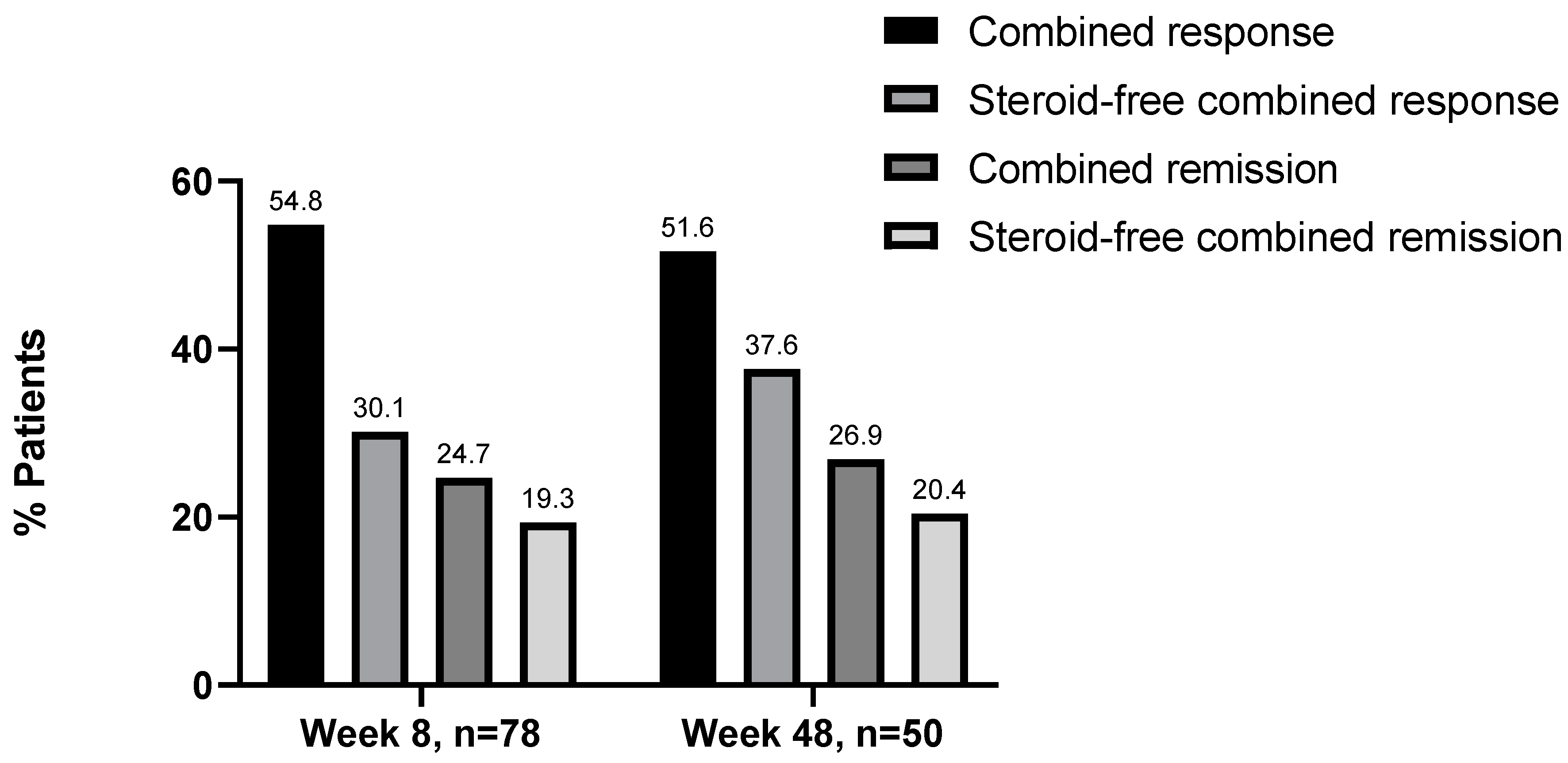
3.5. Steroid-Free Remission and Response at Week 8 and 48
3.6. Adverse Events, Non-Responders, and Re-Inductions
3.7. Predictors for Clinical Remission at Week 48 Follow-Up
4. Discussion
5. Conclusions
Author Contributions
Conflicts of Interest
References
- Hanauer, S.B.; Feagan, B.G.; Lichtenstein, G.R.; Mayer, L.F.; Schreiber, S.; Colombel, J.F.; Rachmilewitz, D.; Wolf, D.C.; Olson, A.; Bao, W.; et al. Maintenance infliximab for Crohn’s disease: The ACCENT I randomized trial. Lancet 2002, 359, 1541–1549. [Google Scholar] [CrossRef]
- Sandborn, W.J.; Feagan, B.G.; Rutgeerts, P.; Hanauer, S.; Colombel, J.F.; Sands, B.E.; Lukas, M.; Fedorak, R.N.; Lee, S.; Bressler, B.; et al. Vedolizumab as induction and maintenance therapy for Crohn’s disease. N. Engl. J. Med. 2013, 369, 711–721. [Google Scholar] [CrossRef] [PubMed]
- Papamichael, K.; Gils, A.; Rutgeerts, P.; Levesque, B.G.; Vermeire, S.; Sandborn, W.J.; Vande Casteele, N. Role for therapeutic drug monitoring during induction therapy with TNF antagonists in IBD: Evolution in the definition and management of primary nonresponse. Inflamm. Bowel Dis. 2015, 21, 182–197. [Google Scholar] [CrossRef] [PubMed]
- Reinisch, W. How to Manage Loss of Response to Anti-TNF in Crohns Disease? Curr. Drug Targets 2010, 11, 152–155. [Google Scholar] [CrossRef] [PubMed]
- Coskun, M.; Vermeire, S.; Nielsen, O.H. Novel Targeted Therapies for Inflammatory Bowel Disease. Trends Pharmacol. Sci. 2017, 38, 127–142. [Google Scholar] [CrossRef] [PubMed]
- Teng, M.W.; Bowman, E.P.; McElwee, J.J.; Smyth, M.J.; Casanova, J.L.; Cooper, A.M.; Cua, J.D. IL-12 and IL-23 cytokines: From discovery to targeted therapies for immune-mediated inflammatory diseases. Nat. Med. 2015, 21, 719–729. [Google Scholar] [CrossRef] [PubMed]
- Iborra, M.; Beltrán, B.; Fernández-Clotet, A.; Gutiérrez, A.; Antolín, B.; Huguet, J.M.; De Francisco, R.; Merino, O.; Carpio, D.; García-López, S.; et al. Real-world short-term effectiveness of ustekinumab in 305 patients with Crohn’s disease: Results from the ENEIDA registry. Aliment. Pharmacol. Ther. 2019, 50, 278–288. [Google Scholar] [CrossRef] [PubMed]
- Kopylov, U.; Afif, W.; Cohen, A.; Bitton, A.; Wild, G.; Bessissow, T.; Wyse, J.; Al-Taweel, T.; Szilagyi, A.; Seidman, E.; et al. Subcutaneous ustekinumab for the treatment of anti-TNF resistant Crohn’s disease-The McGill experience. J. Crohn’s Colitis 2014, 8, 1516–1522. [Google Scholar] [CrossRef] [PubMed]
- Harris, K.A.; Horst, S.; Gadani, A.; Nohl, A.; Annis, K.; Duley, C.; Beaulieu, D.; Ghazi, L.S.D. Patients with Refractory Crohn’s Disease Successfully Treated with Ustekinumab. Inflamm. Bowel Dis 2016, 22, 397–401. [Google Scholar] [CrossRef] [PubMed]
- Liefferinckx, C.; Verstockt, B.; Gils, A.; Noman, M.; Van Kemseke, C.; Macken, E.; De Vos, M.; Van Moerkercke, W.; Rahier, J.F.; Bossuyt, P.; et al. Long-term Clinical Effectiveness of Ustekinumab in Patients with Crohn’s Disease Who Failed Biologic Therapies: A National Cohort Study. J. Crohn’s Colitis 2019. [Google Scholar] [CrossRef] [PubMed]
- Wils, P.; Bouhnik, Y.; Michetti, P.; Flourie, B.; Brixi, H.; Bourrier, A.; Allez, M.; Duclos, B.; Serrero, M.; Buisson, A.; et al. Long-term efficacy and safety of ustekinumab in 122 refractory Crohn’s disease patients: A multicentre experience. Aliment. Pharmacol. Ther. 2018, 47, 588–595. [Google Scholar] [CrossRef] [PubMed]
- Biemans, V.B.C.; van der Meulen-de Jong, A.E.; van der Woude, C.J.; Löwenberg, M.; Dijkstra, G.; Oldenburg, B.; de Boer, N.K.; van der Marel, S.; Bodelier, A.G.; Jansen, J.M.; et al. Ustekinumab for Crohn’s disease: Results of the ICC Registry, a nationwide prospective observational cohort study. J. Crohn’s Colitis 2019. [Google Scholar] [CrossRef]
- Louis, E.; Collard, A.; Oger, A.F.; Degroote, E.; El Yafi, F.A.N.; Belaiche, J. Behavior of Crohn’s disease according to the Vienna classification: Changing pattern over the course of the disease. Gut 2001, 49, 777–782. [Google Scholar] [CrossRef] [PubMed]
- Harvey, R.F.; Bradshaw, J.M. A simple index of Crohn’s-disease activity. Lancet 1980, 1, 514. [Google Scholar] [CrossRef]
- Vermeire, S.; Schreiber, S.; Sandborn, W.J.; Dubois, C.; Rutgeerts, P. Correlation between the Crohn’s disease activity and Harvey-Bradshaw indices in assessing Crohn’s disease severity. Hepatol 2010, 8, 357–363. [Google Scholar]
- Simon, E.G.; Ghosh, S.; Iacucci, M.; Moran, G.W. Ustekinumab for the treatment of Crohn’s disease: Can it find its niche? Ther. Adv. Gastroenterol. 2016, 9, 26–36. [Google Scholar] [CrossRef] [PubMed]
- Feagan, B.G.; Sandborn, W.J.; Gasink, C.; Jacobstein, D.; Lang, Y.; Friedman, J.R.; Blank, M.A.; Johanns, J.; Gao, L.L.; Miao, Y.; et al. Ustekinumab as induction and maintenance therapy for Crohn’s disease. N. Engl. J. Med. 2016, 375, 1946–1960. [Google Scholar] [CrossRef] [PubMed]
- Feagan, B.G.; Gasink, C.; Jacobstein, D.; Zou, B.; Johanns, J.; Adedokun, O.J.; Sands, B.E.; Rutgeerts, P.; de Villiers, W.J.S.; Colombel, J.F. IM-UNITI: 3 Year Efficacy, Safety, and Immunogenicity of Ustekinumab Treatment of Crohn’s Disease. J. Crohn’s Colitis 2019, 3, jjz110. [Google Scholar] [CrossRef]
- Papp, K.; Gottlieb, A.B.; Naldi, L.; Pariser, D.; Ho, V.; Goyal, K.; Fakharzadeh, S.; Chevrier, M.; Calabro, S.; Langholff, W.K.G. Safety Surveillance for Ustekinumab and Other Psoriasis Treatments from the Psoriasis Longitudinal Assessment and Registry (PSOLAR). J. Drugs Dermatol. 2015, 14, 706–714. [Google Scholar] [PubMed]

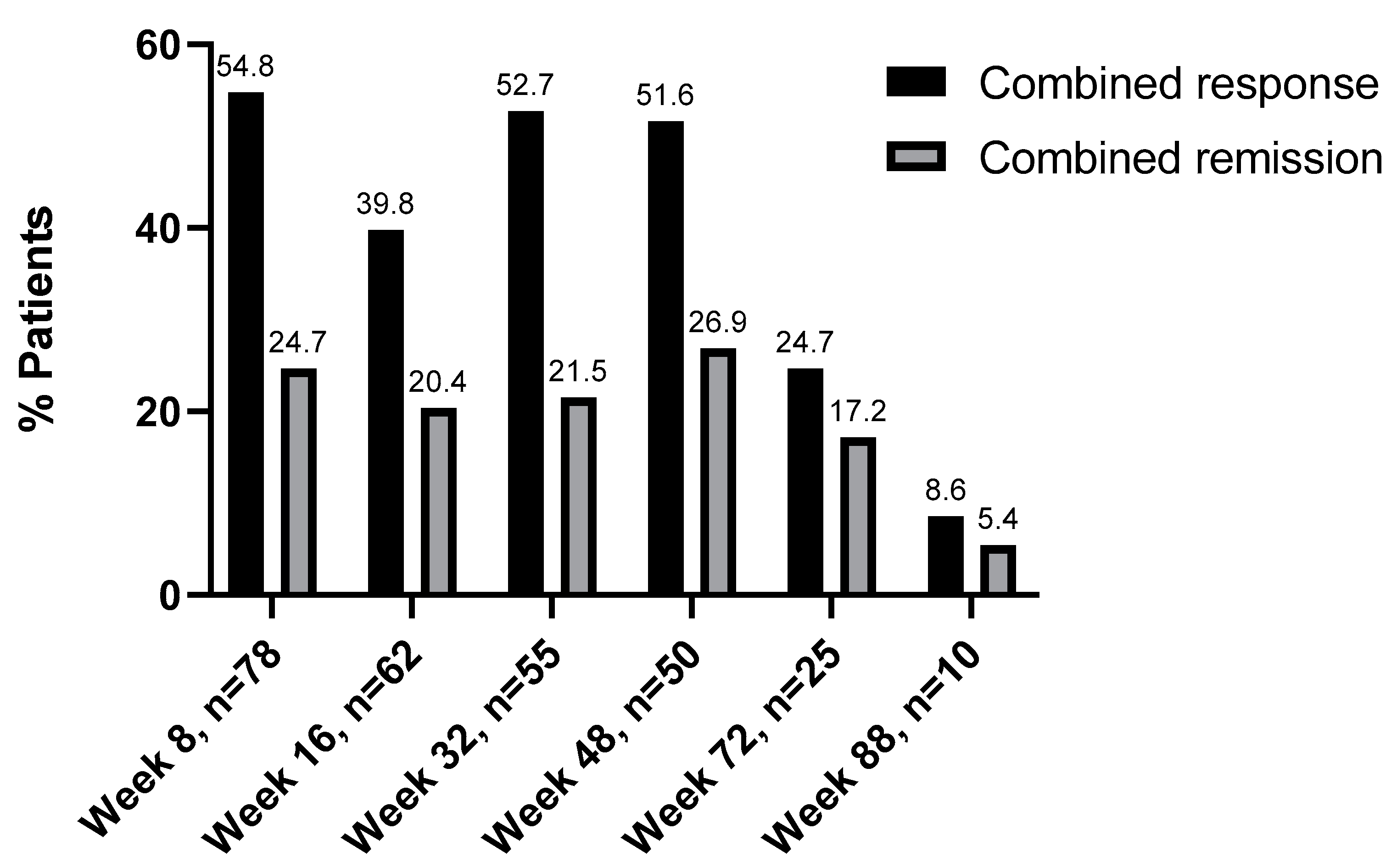

| n = 106 | |
| Female gender (%) | 62; (58.5) |
| Age (years), median (range) | 39.5; (19–73) |
| Disease duration (years), median (range) | 11; (2–39) |
| Median follow-up (weeks), median (95% CI) | 49.1 (95% CI 42.03–56.25) |
| Vienna Classification | |
| Age at diagnosis, n | |
| A1 (<40 year), n (%) | 75 (79.8) |
| A2 (≥40 year), n (%) | 19 (20.2) |
| Disease location | |
| L1 (ileal), n (%) | 21 (22.8) |
| L2 (colonic), n (%) | 16 (17.4) |
| L3 (ileocolonic), n (%) | 35 (38) |
| L4 (upper GI), n (%) | 20 (21.8) |
| Phenotype | |
| B1 (inflammatory), n (%) | 33 (34.4) |
| B2 (stenosing), n (%) | 20 (20.8) |
| B3 (penetrating), n (%) | 43 (44.8) |
| Previous anti-TNFα therapy, n (%) | 102 (96.2) |
| ≥1 TNFα, n (%) | 55 (51.9) |
| ≥2 TNFα, n (%) | 46 (43.4) |
| None, n (%) | 5 (4.7) |
| Previous anti-Integrin therapy, n (%) | 36 (34.4) |
| Exposure to both *, n (%) | 36 (34.4) |
| Previous immunosuppressants, n (%) | 95 (89.6) |
| Steroids at baseline, n (%) | 38 (35.8) |
| Baseline disease activity, n (%) | |
| Remission | 13 (12.3) |
| Mild | 11 (10.4) |
| Moderate | 45 (42.5) |
| Severe | 37 (34.9) |
| HBI score, median (range) | 8; (0–26) |
| CRP, median (range) | 0.95; (0.1–12.2) |
| fCal µg/g, median (range) | 539; (5–2100) |
| Univariate Analysis | ||
|---|---|---|
| p-Value | OR (95% CI) | |
| Age | 0.854 | 1.04 (0.96–1.05) |
| Male Gender | 0.031 | 0.26 (0.08–0.88) |
| Remission week 8 | 0.025 | 4.75 (1.21–18.58) |
| Response week 16 | 0.003 | 10.52 (2.27–48.75) |
| Steroids at baseline | 0.66 | 0.78 (0.25–2.40) |
| Penetrating behavior (B3) | 0.03 | 0.25 (0.07–0.89) |
| Anti-Integrin therapy | 0.61 | 0.73 (0.22–2.43) |
| HBI > 4 | 0.13 | 0.25 (0.43–1.45) |
| fCal > 250 µg/g | 0.48 | 1.90 (0.31–11.61) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kubesch, A.; Rueter, L.; Farrag, K.; Krause, T.; Stienecker, K.; Hausmann, J.; Filmann, N.; Dignass, A.; Stein, J.; Blumenstein, I. Short and Long-Term Effectiveness of Ustekinumab in Patients with Crohn’s Disease: Real-World Data from a German IBD Cohort. J. Clin. Med. 2019, 8, 2140. https://doi.org/10.3390/jcm8122140
Kubesch A, Rueter L, Farrag K, Krause T, Stienecker K, Hausmann J, Filmann N, Dignass A, Stein J, Blumenstein I. Short and Long-Term Effectiveness of Ustekinumab in Patients with Crohn’s Disease: Real-World Data from a German IBD Cohort. Journal of Clinical Medicine. 2019; 8(12):2140. https://doi.org/10.3390/jcm8122140
Chicago/Turabian StyleKubesch, Alica, Laurenz Rueter, Karima Farrag, Thomas Krause, Klaus Stienecker, Johannes Hausmann, Natalie Filmann, Axel Dignass, Jürgen Stein, and Irina Blumenstein. 2019. "Short and Long-Term Effectiveness of Ustekinumab in Patients with Crohn’s Disease: Real-World Data from a German IBD Cohort" Journal of Clinical Medicine 8, no. 12: 2140. https://doi.org/10.3390/jcm8122140
APA StyleKubesch, A., Rueter, L., Farrag, K., Krause, T., Stienecker, K., Hausmann, J., Filmann, N., Dignass, A., Stein, J., & Blumenstein, I. (2019). Short and Long-Term Effectiveness of Ustekinumab in Patients with Crohn’s Disease: Real-World Data from a German IBD Cohort. Journal of Clinical Medicine, 8(12), 2140. https://doi.org/10.3390/jcm8122140





