Approximate Mortality Risks between Hyperuricemia and Diabetes in the United States
Abstract
:1. Introduction
2. Materials and Methods
2.1. Data Source and Study Population
2.2. Definition of Diabetes
2.3. Mortality Outcomes
2.4. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kuo, C.F.; See, L.C.; Luo, S.F.; Ko, Y.S.; Lin, Y.S.; Hwang, J.S.; Lin, C.M.; Chen, H.W.; Yu, K.H. Gout: An independent risk factor for all-cause and cardiovascular mortality. Rheumatology 2010, 49, 141–146. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Choi, H.K.; Curhan, G. Independent impact of gout on mortality and risk for coronary heart disease. Circulation 2007, 116, 894–900. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Pandya, B.J.; Choi, H.K. Prevalence of gout and hyperuricemia in the US general population: The National Health and Nutrition Examination Survey 2007–2008. Arthritis Rheum 2011, 63, 3136–3141. [Google Scholar] [CrossRef] [PubMed]
- Ruggiero, C.; Cherubini, A.; Ble, A.; Bos, A.J.; Maggio, M.; Dixit, V.D.; Lauretani, F.; Bandinelli, S.; Senin, U.; Ferrucci, L. Uric acid and inflammatory markers. Eur. Heart J. 2006, 27, 1174–1181. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ruggiero, C.; Cherubini, A.; Miller, E.; Maggio, M.; Najjar, S.S.; Lauretani, F.; Bandinelli, S.; Senin, U.; Ferrucci, L. Usefulness of uric acid to predict changes in C-reactive protein and interleukin-6 in 3-year period in Italians aged 21 to 98 years. Am. J. Cardiol. 2007, 100, 115–121. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kienhorst, L.B.; van Lochem, E.; Kievit, W.; Dalbeth, N.; Merriman, M.E.; Phipps-Green, A.; Loof, A.; van Heerde, W.; Vermeulen, S.; Stamp, L.K.; et al. Gout Is a Chronic Inflammatory Disease in Which High Levels of Interleukin-8 (CXCL8), Myeloid-Related Protein 8/Myeloid-Related Protein 14 Complex, and an Altered Proteome Are Associated with Diabetes Mellitus and Cardiovascular Disease. Arthritis Rheumatol. 2015, 67, 3303–3313. [Google Scholar] [CrossRef] [Green Version]
- Snow, M.H.; Mikuls, T.R. Rheumatoid arthritis and cardiovascular disease: The role of systemic inflammation and evolving strategies of prevention. Curr. Opin. Rheumatol. 2005, 17, 234–241. [Google Scholar] [CrossRef]
- Full, L.E.; Ruisanchez, C.; Monaco, C. The inextricable link between atherosclerosis and prototypical inflammatory diseases rheumatoid arthritis and systemic lupus erythematosus. Arthritis Res. Ther. 2009, 11, 217. [Google Scholar] [CrossRef] [Green Version]
- Richette, P.; Latourte, A.; Bardin, T. Cardiac and renal protective effects of urate-lowering therapy. Rheumatology 2018, 57, i47–i50. [Google Scholar] [CrossRef] [Green Version]
- Ramirez, M.E.G.; Bargman, J.M. Treatment of asymptomatic hyperuricemia in chronic kidney disease: A new target in an old enemy–A review. J. Adv. Res. 2017, 8, 551–554. [Google Scholar] [CrossRef]
- Kimura, K.; Hosoya, T.; Uchida, S.; Inaba, M.; Makino, H.; Maruyama, S.; Ito, S.; Yamamoto, T.; Tomino, Y.; Ohno, I.; et al. Febuxostat Therapy for Patients with Stage 3 CKD and Asymptomatic Hyperuricemia: A Randomized Trial. Am. J. Kidney Dis. 2018, 72, 798–810. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tsujimoto, T.; Kajio, H.; Sugiyama, T. Favourable changes in mortality in people with diabetes: US NHANES 1999–2010. Diabetes Obes. Metab. 2018, 20, 85–93. [Google Scholar] [CrossRef] [PubMed]
- Kuo, C.F.; Grainge, M.J.; Mallen, C.; Zhang, W.; Doherty, M. Rising burden of gout in the UK but continuing suboptimal management: A nationwide population study. Ann. Rheum. Dis. 2015, 74, 661–667. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Graf, S.W.; Whittle, S.L.; Wechalekar, M.D.; Moi, J.H.; Barrett, C.; Hill, C.L.; Littlejohn, G.; Lynch, N.; Major, G.; Taylor, A.L.; et al. Australian and New Zealand recommendations for the diagnosis and management of gout: Integrating systematic literature review and expert opinion in the 3e Initiative. Int. J. Rheum. Dis. 2015, 18, 341–351. [Google Scholar] [CrossRef] [PubMed]
- Levey, A.S.; Stevens, L.A.; Schmid, C.H.; Zhang, Y.L.; Castro, A.F., 3rd; Feldman, H.I.; Kusek, J.W.; Eggers, P.; Van Lente, F.; Greene, T.; et al. A new equation to estimate glomerular filtration rate. Ann. Intern. Med. 2009, 150, 604–612. [Google Scholar] [CrossRef]
- NCHS 2011 Linked Mortality Files Matching Methodology; National Center for Health Statistics, Office of Analysis and Epidemiology: Hyattsville, MD, USA, 2013. Available online: http://www.cdc.gov/nchs/data_access/data_linkage/mortality/linkage_methods_analytical_support/2011_linked_mortality_file_matching_methodology.pdf (accessed on 29 October 2019).
- National Health and Nutrition Examination Survey: Analytic Guidelines, 2011–2014 and 2015–2016. Available online: https://wwwn.cdc.gov/nchs/data/nhanes/2011–2012/analyticguidelines/analytic_guidelines_11_16.pdf (accessed on 29 October 2019).
- Zuo, T.; Liu, X.; Jiang, L.; Mao, S.; Yin, X.; Guo, L. Hyperuricemia and coronary heart disease mortality: A meta-analysis of prospective cohort studies. BMC Cardiovasc. Disord. 2016, 16, 207. [Google Scholar] [CrossRef] [Green Version]
- Lehto, S.; Niskanen, L.; Ronnemaa, T.; Laakso, M. Serum uric acid is a strong predictor of stroke in patients with non-insulin-dependent diabetes mellitus. Stroke 1998, 29, 635–639. [Google Scholar] [CrossRef] [Green Version]
- Cho, S.K.; Chang, Y.; Kim, I.; Ryu, S. U-Shaped Association between Serum Uric Acid Level and Risk of Mortality: A Cohort Study. Arthritis Rheumatol. 2018, 70, 1122–1132. [Google Scholar] [CrossRef] [Green Version]
- Zhang, W.; Iso, H.; Murakami, Y.; Miura, K.; Nagai, M.; Sugiyama, D.; Ueshima, H.; Okamura, T.; Epoch-Japan, G. Serum Uric Acid and Mortality Form Cardiovascular Disease: EPOCH-JAPAN Study. J. Atheroscler. Thromb. 2016, 23, 692–703. [Google Scholar] [CrossRef] [Green Version]
- Beberashvili, I.; Sinuani, I.; Azar, A.; Shapiro, G.; Feldman, L.; Stav, K.; Sandbank, J.; Averbukh, Z. Serum uric acid as a clinically useful nutritional marker and predictor of outcome in maintenance hemodialysis patients. Nutrition 2015, 31, 138–147. [Google Scholar] [CrossRef]
- Cutler, R.G. Urate and ascorbate: Their possible roles as antioxidants in determining longevity of mammalian species. Arch. Gerontol. Geriatr. 1984, 3, 321–348. [Google Scholar] [CrossRef]
- Wakasugi, M.; Kazama, J.J.; Narita, I.; Konta, T.; Fujimoto, S.; Iseki, K.; Moriyama, T.; Yamagata, K.; Tsuruya, K.; Asahi, K.; et al. Association between hypouricemia and reduced kidney function: A cross-sectional population-based study in Japan. Am. J. Nephrol. 2015, 41, 138–146. [Google Scholar] [CrossRef] [PubMed]
- Iliesiu, A.; Campeanu, A.; Dusceac, D. Serum uric acid and cardiovascular disease. Maedica 2010, 5, 186–192. [Google Scholar] [PubMed]
- Eleftheriadis, T.; Golphinopoulos, S.; Pissas, G.; Stefanidis, I. Asymptomatic hyperuricemia and chronic kidney disease: Narrative review of a treatment controversial. J. Adv. Res. 2017, 8, 555–560. [Google Scholar] [CrossRef]
- Peng, T.C.; Wang, C.C.; Kao, T.W.; Chan, J.Y.; Yang, Y.H.; Chang, Y.W.; Chen, W.L. Relationship between hyperuricemia and lipid profiles in US adults. Biomed. Res. Int. 2015, 2015, 127596. [Google Scholar] [CrossRef]
- Krishnan, E.; Akhras, K.S.; Sharma, H.; Marynchenko, M.; Wu, E.Q.; Tawk, R.; Liu, J.; Shi, L. Relative and attributable diabetes risk associated with hyperuricemia in US veterans with gout. QJM 2013, 106, 721–729. [Google Scholar] [CrossRef] [Green Version]
- Khanna, D.; Fitzgerald, J.D.; Khanna, P.P.; Bae, S.; Singh, M.K.; Neogi, T.; Pillinger, M.H.; Merill, J.; Lee, S.; Prakash, S.; et al. 2012 American College of Rheumatology guidelines for management of gout. Part 1: Systematic nonpharmacologic and pharmacologic therapeutic approaches to hyperuricemia. Arthritis. Care Res. 2012, 64, 1431–1446. [Google Scholar] [CrossRef] [Green Version]
- Richette, P.; Doherty, M.; Pascual, E.; Barskova, V.; Becce, F.; Castaneda-Sanabria, J.; Coyfish, M.; Guillo, S.; Jansen, T.L.; Janssens, H.; et al. 2016 updated EULAR evidence-based recommendations for the management of gout. Ann. Rheum. Dis. 2017, 76, 29–42. [Google Scholar] [CrossRef]
- Hoieggen, A.; Alderman, M.H.; Kjeldsen, S.E.; Julius, S.; Devereux, R.B.; De Faire, U.; Fyhrquist, F.; Ibsen, H.; Kristianson, K.; Lederballe-Pedersen, O.; et al. The impact of serum uric acid on cardiovascular outcomes in the LIFE study. Kidney Int. 2004, 65, 1041–1049. [Google Scholar] [CrossRef] [Green Version]
- Braga, T.T.; Forni, M.F.; Correa-Costa, M.; Ramos, R.N.; Barbuto, J.A.; Branco, P.; Castoldi, A.; Hiyane, M.I.; Davanso, M.R.; Latz, E.; et al. Soluble Uric Acid Activates the NLRP3 Inflammasome. Sci. Rep. 2017, 7, 39884. [Google Scholar] [CrossRef]
- Yu, M.A.; Sanchez-Lozada, L.G.; Johnson, R.J.; Kang, D.H. Oxidative stress with an activation of the renin-angiotensin system in human vascular endothelial cells as a novel mechanism of uric acid-induced endothelial dysfunction. J. Hypertens. 2010, 28, 1234–1242. [Google Scholar] [CrossRef] [PubMed]
- Wilcox, C.S.; Shen, W.; Boulton, D.W.; Leslie, B.R.; Griffen, S.C. Interaction between the Sodium-Glucose-Linked Transporter 2 Inhibitor Dapagliflozin and the Loop Diuretic Bumetanide in Normal Human Subjects. J. Am. Heart. Assoc. 2018, 7, e007046. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kuwabara, M.; Niwa, K.; Hisatome, I.; Nakagawa, T.; Roncal-Jimenez, C.A.; Andres-Hernando, A.; Bjornstad, P.; Jensen, T.; Sato, Y.; Milagres, T.; et al. Asymptomatic Hyperuricemia Without Comorbidities Predicts Cardiometabolic Diseases: Five-Year Japanese Cohort Study. Hypertension 2017, 69, 1036–1044. [Google Scholar] [CrossRef] [PubMed]
- Tsai, C.W.; Lin, S.Y.; Kuo, C.C.; Huang, C.C. Serum Uric Acid and Progression of Kidney Disease: A Longitudinal Analysis and Mini-Review. PLoS ONE 2017, 12, e0170393. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chronic Kidney Disease Prognosis, Consortium; Matsushita, K.; van der Velde, M.; Astor, B.C.; Woodward, M.; Levey, A.S.; de Jong, P.E.; Coresh, J.; Gansevoort, R.T.; Levey, A.S. Association of estimated glomerular filtration rate and albuminuria with all-cause and cardiovascular mortality in general population cohorts: A collaborative meta-analysis. Lancet 2010, 375, 2073–2081. [Google Scholar] [CrossRef] [Green Version]
- Bibbins-Domingo, K.; Grossman, D.C.; Curry, S.J.; Davidson, K.W.; Epling, J.W., Jr.; Garcia, F.A.; Gillman, M.W.; Kemper, A.R.; Krist, A.H. Statin Use for the Primary Prevention of Cardiovascular Disease in Adults: US Preventive Services Task Force Recommendation Statement. JAMA 2016, 316, 1997–2007. [Google Scholar]
- Collins, R.; Reith, C.; Emberson, J.; Armitage, J.; Baigent, C.; Blackwell, L.; Blumenthal, R.; Danesh, J.; Smith, G.D.; DeMets, D.; et al. Interpretation of the evidence for the efficacy and safety of statin therapy. Lancet 2016, 388, 2532–2561. [Google Scholar] [CrossRef] [Green Version]
- Opie, L.H.; Dalby, A.J. Cardiovascular prevention: Lifestyle and statins–competitors or companions? S. Afr. Med. 2014, 104, 168–173. [Google Scholar] [CrossRef] [Green Version]
- Fang, J.; Alderman, M.H. Serum uric acid and cardiovascular mortality the NHANES I epidemiologic follow-up study, 1971–1992. National Health and Nutrition Examination Survey. JAMA 2000, 283, 2404–2410. [Google Scholar] [CrossRef] [Green Version]
- Krishnan, E.; Svendsen, K.; Neaton, J.D.; Grandits, G.; Kuller, L.H.; Group, M.R. Long-term cardiovascular mortality among middle-aged men with gout. Arch. Intern. Med. 2008, 168, 1104–1110. [Google Scholar] [CrossRef]
- White, W.B.; Saag, K.G.; Becker, M.A.; Borer, J.S.; Gorelick, P.B.; Whelton, A.; Hunt, B.; Castillo, M.; Gunawardhana, L.; Investigators, C. Cardiovascular Safety of Febuxostat or Allopurinol in Patients with Gout. N. Engl. J. Med. 2018, 378, 1200–1210. [Google Scholar] [CrossRef] [PubMed]
| Variable | Overall | Non-DM | DM | p-Value a |
|---|---|---|---|---|
| Number | 29,226 | 26,720 | 2506 | |
| Age, years | 44.3 ± 0.2 | 43.6 ± 0.2 | 56.9 ± 0.4 | <0.001 |
| Male | 13,736 (47.6) | 12,560 (47.5) | 1176 (46.7) | 0.52 |
| Non-Hispanic black | 5876 (10.9) | 5208 (10.4) | 668 (16.8) | <0.001 |
| Current smoker | 6012 (23.0) | 5565 (23.2) | 447 (19.1) | <0.001 |
| Cancer | 2099 (7.4) | 1805 (7.1) | 294 (12.7) | <0.001 |
| Hypertension | 8075 (25.6) | 6471 (23.2) | 1604 (61.2) | <0.001 |
| BMI, kg/m2 | 28.2 ± 0.1 | 27.9 ± 0.1 | 32.5 ± 0.2 | <0.001 |
| SBP | 122 ± 0.2 | 121 ± 0.2 | 131 ± 0.7 | <0.001 |
| HDL-C, mg/dL | 52.9 ± 0.2 | 53.2 ± 0.2 | 48.4 ± 0.4 | <0.001 |
| Fasting glucose, mg/dL | 95.5 ± 0.2 | 91.6 ± 0.2 | 152.9 ± 1.9 | <0.001 |
| Total cholesterol, mg/dL | 199.4 ± 0.4 | 199.7 ± 0.5 | 196.7 ± 1.4 | <0.001 |
| Triglycerides, mg/dL | 147.4 ± 1.3 | 143.5 ± 1.2 | 205.2 ± 7.1 | <0.001 |
| Creatinine, mg/dL | 0.84 ± 0.002 | 0.84 ± 0.002 | 0.85 ± 0.01 | <0.001 |
| DM duration, years | 10.3 ± 0.3 | 10.3±0.3 |
| Variable | Non-DM | ||||
|---|---|---|---|---|---|
| <5 (mg/dL) | 5–7 (mg/dL) | 7–9 (mg/dL) | ≥9 (mg/dL) | p-Value a | |
| Number | 11,545 | 11,895 | 3030 | 250 | |
| Age, years | 42.4 ± 0.2 | 44.3 ± 0.3 | 44.6 ± 0.4 | 45.3 ± 1.4 | <0.001 |
| Male | 2306 (19.1) | 7556 (63.8) | 2498 (84.6) | 200 (81.8) | |
| Non-Hispanic black | 2106 (10.5) | 2347 (10.2) | 680 (10.4) | 75 (17.5) | |
| Current smoker | 2141 (22.1) | 2746 (24.6) | 630 (22.3) | 48 (18.2) | |
| Cancer | 700 (6.9) | 869 (7.5) | 220 (6.6) | 16 (6.3) | |
| Hypertension | 1961 (16.4) | 3232 (26) | 1140 (35.1) | 138 (49.2) | <0.001 |
| BMI, kg/m2 | 26.0 ± 0.1 | 28.8 ± 0.1 | 30.9 ± 0.2 | 31.6 ± 0.5 | <0.001 |
| SBP | 118 ± 0.3 | 123 ± 0.2 | 126 ± 0.4 | 126 ± 1.4 | <0.001 |
| HDL-C, mg/dL | 58.4 ± 0.3 | 50.4 ± 0.2 | 46.0 ± 0.3 | 45.9 ± 1.3 | <0.001 |
| Fasting Glucose, mg/dL | 89.4 ± 0.2 | 92.6 ± 0.2 | 95.3 ± 0.4 | 96.0 ± 1.3 | <0.001 |
| Total cholesterol, mg/dL | 195.9 ± 0.5 | 201.0 ± 0.7 | 206.6 ± 1.1 | 218.3 ± 3.5 | <0.001 |
| Triglycerides, mg/dL | 118.5 ± 1.3 | 152 ± 1.6 | 193.2 ± 4.4 | 241.6 ± 22.4 | <0.001 |
| Creatinine, mg/dL | 0.74 ± 0.002 | 0.89 ± 0.003 | 0.97 ± 0.005 | 1.01 ± 0.017 | <0.001 |
| DM | |||||
| Number | 973 | 1141 | 345 | 47 | |
| Age, years | 54.9 ± 0.6 | 57.9 ± 0.5 | 59.7 ± 0.9 | 54.5 ± 2.3 | <0.001 |
| Male | 370 (37.5) | 585 (50.5) | 194 (57.5) | 27 (63.1) | |
| Non-Hispanic black | 204 (14.6) | 299 (16.1) | 142 (23.3) | 23 (27.5) | |
| Current smoker | 196 (21.4) | 198 (19.8) | 49 (12.4) | 4 (7.5) | |
| Cancer | 98 (12.4) | 137 (12.6) | 51 (14.2) | 8 (12.0) | |
| Hypertension | 510 (47.3) | 781 (67.3) | 268 (75.1) | 45 (91.9) | <0.001 |
| BMI, kg/m2 | 30.5 ± 0.3 | 33.3 ± 0.4 | 34.6 ± 0.5 | 36.3 ± 1.3 | <0.001 |
| SBP | 130 ± 1.1 | 132 ± 0.9 | 132 ± 1.4 | 130 ± 3.9 | <0.001 |
| HDL-C, mg/dL | 51.1 ± 0.7 | 47.7 ± 0.6 | 44.0 ± 0.7 | 40.7 ± 1.6 | <0.001 |
| Fasting Glucose, mg/dL | 168.4 ± 3.8 | 144.6 ± 2.3 | 136.6 ± 3.8 | 158.0 ± 13.8 | <0.001 |
| Total cholesterol, mg/dL | 197.6 ± 2.8 | 196.2 ± 1.9 | 196.3 ± 3.4 | 193.7 ± 9.6 | <0.001 |
| Triglycerides, mg/dL | 190.5 ± 10.3 | 208.5 ± 12 | 226.8 ± 14.3 | 260 ± 47.2 | <0.001 |
| Creatinine, mg/dL | 0.77 ± 0.006 | 0.88 ± 0.009 | 0.98 ± 0.017 | 1.05 ± 0.048 | <0.001 |
| DM duration, years | 10.8 ± 0.5 | 9.9 ± 0.4 | 10.5±0.9 | 7.6±1.3 | <0.001 |
| Outcomes | Non-DM | |||
|---|---|---|---|---|
| <5 (mg/dL) N = 11,545 | 5–7 (mg/dL) N = 11,895 | 7–9 (mg/dL) N = 3030 | ≥9 (mg/dL) N = 250 | |
| All-cause mortality (N = 1664) | ||||
| un-weighted | 1.02 (0.8–1.18) | 1.0 (reference) | 1.26 (1.06–1.49) * | 2.44 (1.63–3.66) * |
| weighted a | 1.03 (0.87–1.22) | 1.0 (reference) | 1.43 (1.15–1.79) * | 2.41 (1.42–4.08) * |
| CVD mortality (N = 378) | ||||
| un-weighted | 1.06 (0.78–1.44) | 1.0 (reference) | 1.37 (0.96–1.95) | 2.97 (1.37–6.45) * |
| weighted a | 1.22 (0.82-1.82) | 1.0 (reference) | 1.76 (1.22–2.56) * | 5.06 (1.69–15.15) * |
| Cancer death (N = 436) | ||||
| un-weighted | 1.06 (0.82-1.36) | 1.0 (reference) | 1.09 (0.79–1.49) | 2.39 (1.17–4.90) * |
| weighted a | 1.04 (0.75-1.45) | 1.0 (reference) | 1.25 (0.90–1.73) | 1.43 (0.64–3.21) |
| CVD or Cancer death (N = 814) | ||||
| un-weighted | 1.06 (0.87-1.29) | 1.0 (reference) | 1.19 (0.94–1.51) | 2.63 (1.56–4.45) * |
| weighted a | 1.10 (0.84-1.43) | 1.0 (reference) | 1.42 (1.08–1.87) * | 2.60 (1.21–5.58) * |
| DM | ||||
| N = 973 | N = 1141 | N = 345 | N = 47 | |
| All-cause mortality (N = 405) | ||||
| un-weighted | 1.33 (1.01–1.75) * | 1.0 (reference) | 1.69 (1.22–2.34) * | 1.41 (0.65–3.08) |
| weighted a | 1.66 (1.14–2.41) * | 1.0 (reference) | 2.17 (1.49–3.17) * | 2.10 (0.87–5.06) |
| CVD mortality (N = 117) | ||||
| un-weighted | 0.71 (0.41–1.24) | 1.0 (reference) | 1.75 (0.95–3.23) | 1.91 (0.45–8.11) |
| weighted a | 0.91 (0.38–2.18) | 1.0 (reference) | 2.53 (1.18–5.41) * | 0.89 (0.18–4.50) |
| Cancer death (N = 84) | ||||
| un-weighted | 1.82 (1.01–3.26) * | 1.0 (reference) | 1.81 (0.85–3.85) | 2.60 (0.59–11.40) |
| weighted a | 2.13 (0.84–5.42) | 1.0 (reference) | 2.13 (0.88–5.12) | 4.51 (0.89-22.78) |
| CVD or Cancer death (N = 201) | ||||
| un-weighted | 1.10 (0.74–1.63) | 1.0 (reference) | 1.75 (1.09–2.81) | 2.15 (0.77–6.04) |
| weighted a | 1.36 (0.77–2.43) | 1.0 (reference) | 2.30 (1.34–3.96) * | 2.60 (0.69–9.80) |
| Risks | Non-DM | DM | ||||||
| <5 (mg/dL) N = 11,545 | 5–7 (mg/dL) N = 11,895 | 7–9 (mg/dL) N = 3030 | ≥9 (mg/dL) N = 250 | <5 (mg/dL) N = 973 | 5–7 (mg/dL) N = 1141 | 7–9 (mg/dL) N = 345 | ≥9 (mg/dL) N = 47 | |
| All-cause mortality (N = 2069) | ||||||||
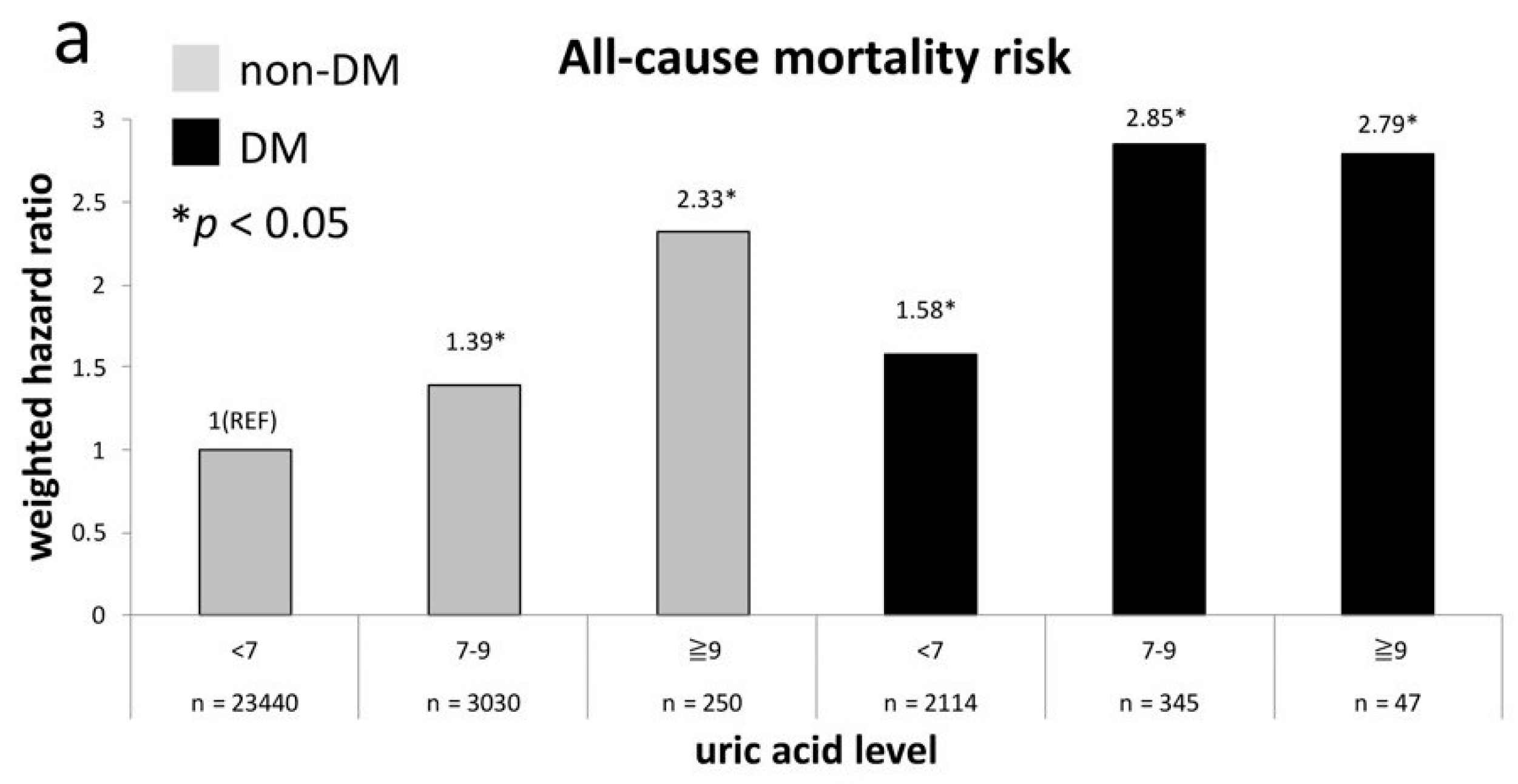
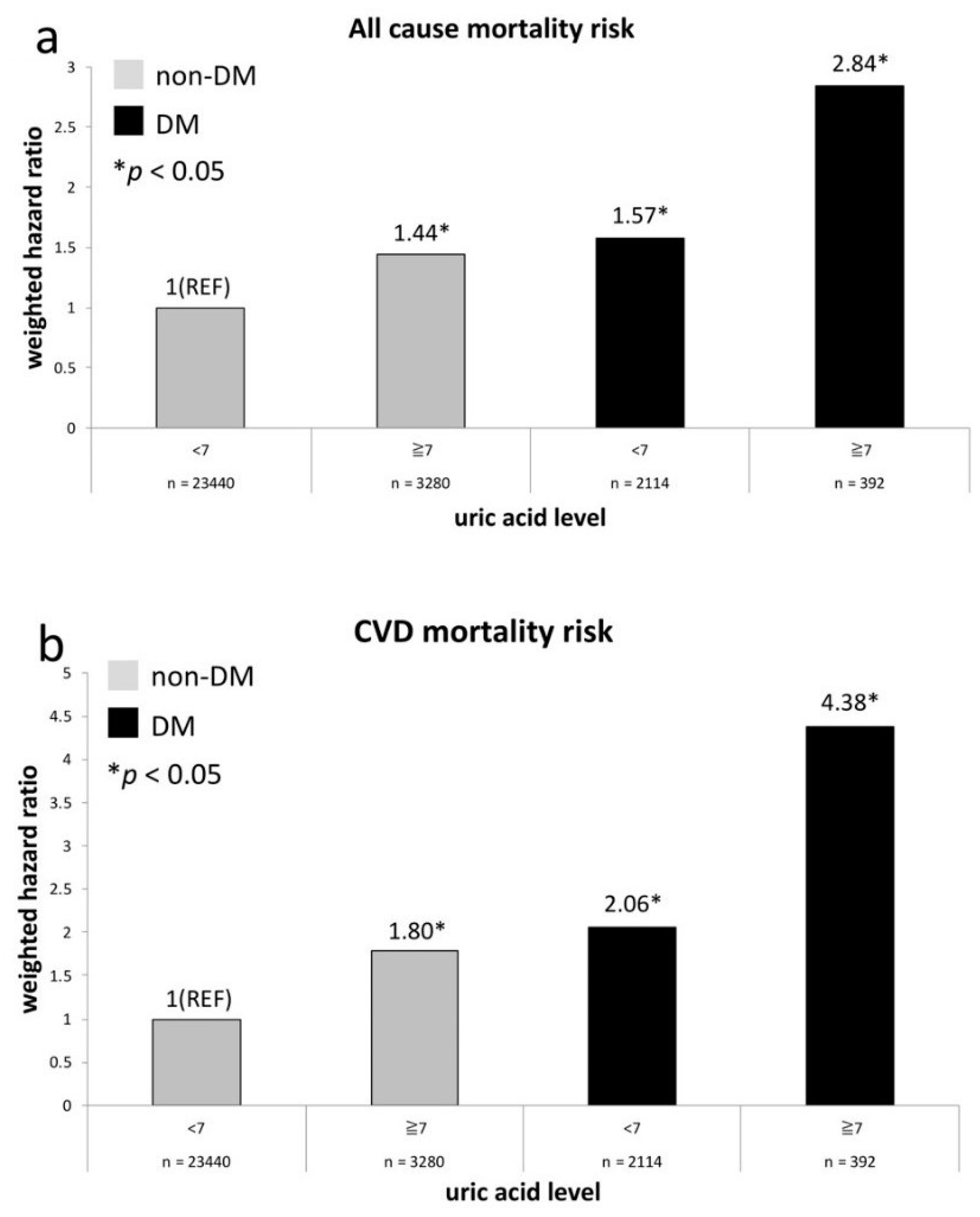
| un-weighted | 1.04 (0.9–1.19) | 1.0 (reference) | 1.24 (1.05–1.47) * | 2.37 (1.58–3.55) * | 1.77 (1.44–2.18) * | 1.43 (1.17–1.76) * | 2.55 (1.94–3.34) * | 2.25 (1.06–4.77) * |
| weighted a | 1.06 (0.9–1.26) | 1.0 (reference) | 1.40 (1.12–1.75) * | 2.35 (1.39–3.96) * | 2.01 (1.55–2.62) * | 1.30 (0.95–1.76) | 2.87 (2.14–3.85) * | 2.79 (1.28–6.08) * |
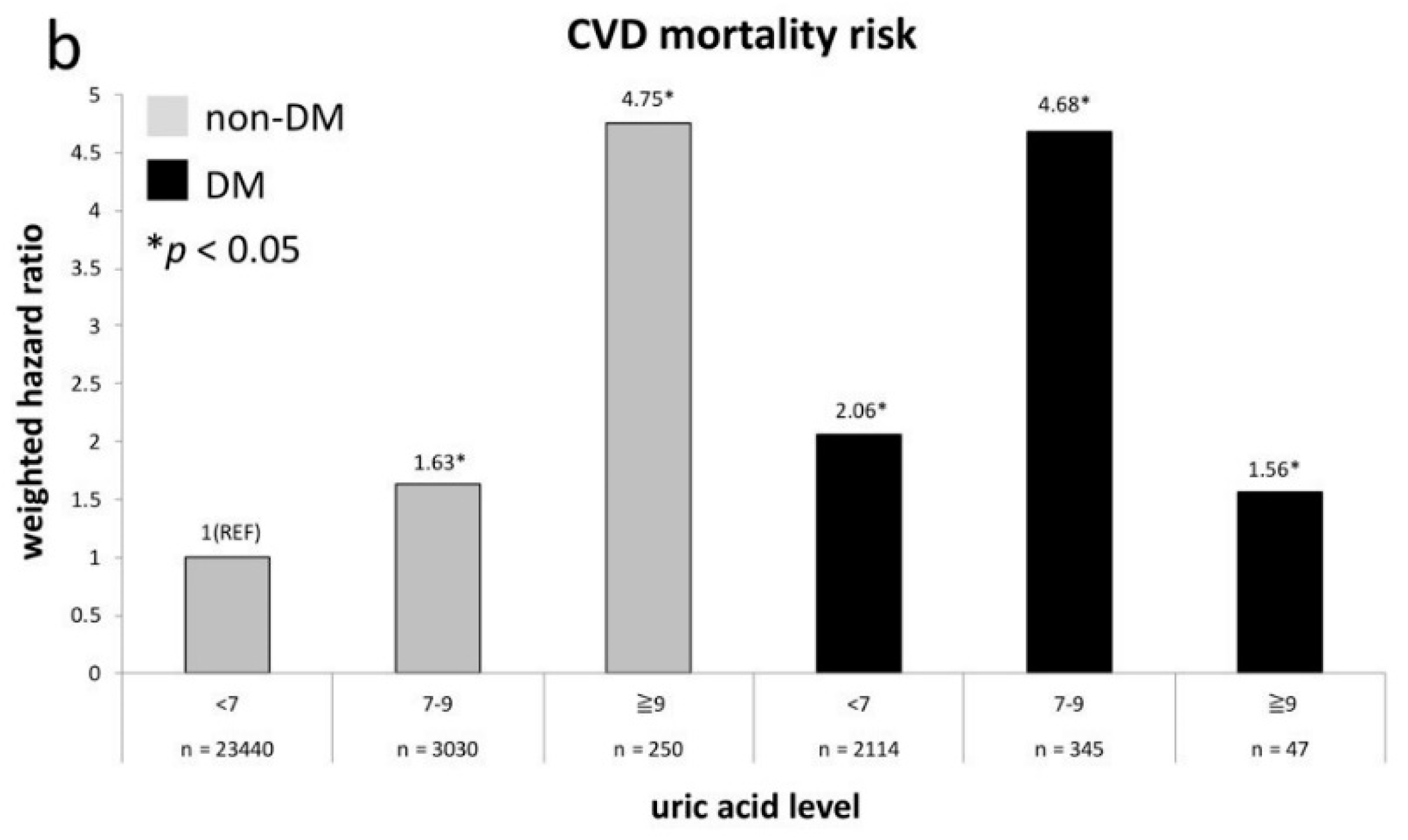
| CVD mortality (N = 495) | ||||||||
| un-weighted | 1.03 (0.77–1.39) | 1.0 (reference) | 1.39 (0.98–1.98) | 3.06 (1.42–6.62) * | 1.71 (1.08–2.69) * | 2.12 (1.46–3.09) * | 3.25 (1.89-5.61) * | 3.38 (0.82–13.87) |
| weighted a | 1.23 (0.86–1.76) | 1.0 (reference) | 1.74 (1.18–2.57) * | 5.07 (1.71–5.04) * | 2.21 (1.14–4.28) * | 2.25 (1.25–4.06) * | 4.99 (2.48–10.03) * | 1.66 (0.42–6.60) |
| Cancer death (N = 520) | ||||||||
| un-weighted | 1.04 (0.81–1.34) | 1.0 (reference) | 1.10 (0.80–1.50) | 2.42 (1.18–4.94) * | 1.58 (1.07–2.33) * | 0.82 (0.52–1.30) | 1.28 (0.69–2.39) | 1.84 (0.45–7.47) |
| weighted a | 1.07 (0.77–1.49) | 1.0 (reference) | 1.23 (0.88-1.71) | 1.43 (0.64–3.19) | 1.45 (0.77–2.72) | 0.78 (0.45–1.35) | 1.54 (0.75–3.17) | 3.30 (0.76–14.37) |
| CVD or Cancer death (N = 1015) | ||||||||
| un-weighted | 1.04 (0.86–1.26) | 1.0 (reference) | 1.22 (0.96–1.54) | 2.67 (1.58–4.51) * | 1.62 (1.21–2.18) * | 1.35 (1.02–1.80) * | 2.01 (1.34–3.02) * | 2.40 (0.89–6.50) |
| weighted a | 1.13 (0.86–1.47) | 1.0 (reference) | 1.40 (1.06–1.84) * | 2.58 (1.21–5.54) * | 1.69 (1.14–2.51) * | 1.28 (0.89–1.84) | 2.67 (1.68–4.26) * | 2.81 (0.84–9.39) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, P.-H.; Chen, Y.-W.; Liu, W.-J.; Hsu, S.-W.; Chen, C.-H.; Lee, C.-L. Approximate Mortality Risks between Hyperuricemia and Diabetes in the United States. J. Clin. Med. 2019, 8, 2127. https://doi.org/10.3390/jcm8122127
Chen P-H, Chen Y-W, Liu W-J, Hsu S-W, Chen C-H, Lee C-L. Approximate Mortality Risks between Hyperuricemia and Diabetes in the United States. Journal of Clinical Medicine. 2019; 8(12):2127. https://doi.org/10.3390/jcm8122127
Chicago/Turabian StyleChen, Po-Hsun, Yu-Wei Chen, Wei-Ju Liu, Ssu-Wei Hsu, Ching-Hsien Chen, and Chia-Lin Lee. 2019. "Approximate Mortality Risks between Hyperuricemia and Diabetes in the United States" Journal of Clinical Medicine 8, no. 12: 2127. https://doi.org/10.3390/jcm8122127
APA StyleChen, P.-H., Chen, Y.-W., Liu, W.-J., Hsu, S.-W., Chen, C.-H., & Lee, C.-L. (2019). Approximate Mortality Risks between Hyperuricemia and Diabetes in the United States. Journal of Clinical Medicine, 8(12), 2127. https://doi.org/10.3390/jcm8122127




