Periprosthetic Osteolysis: Mechanisms, Prevention and Treatment
Abstract
1. Introduction
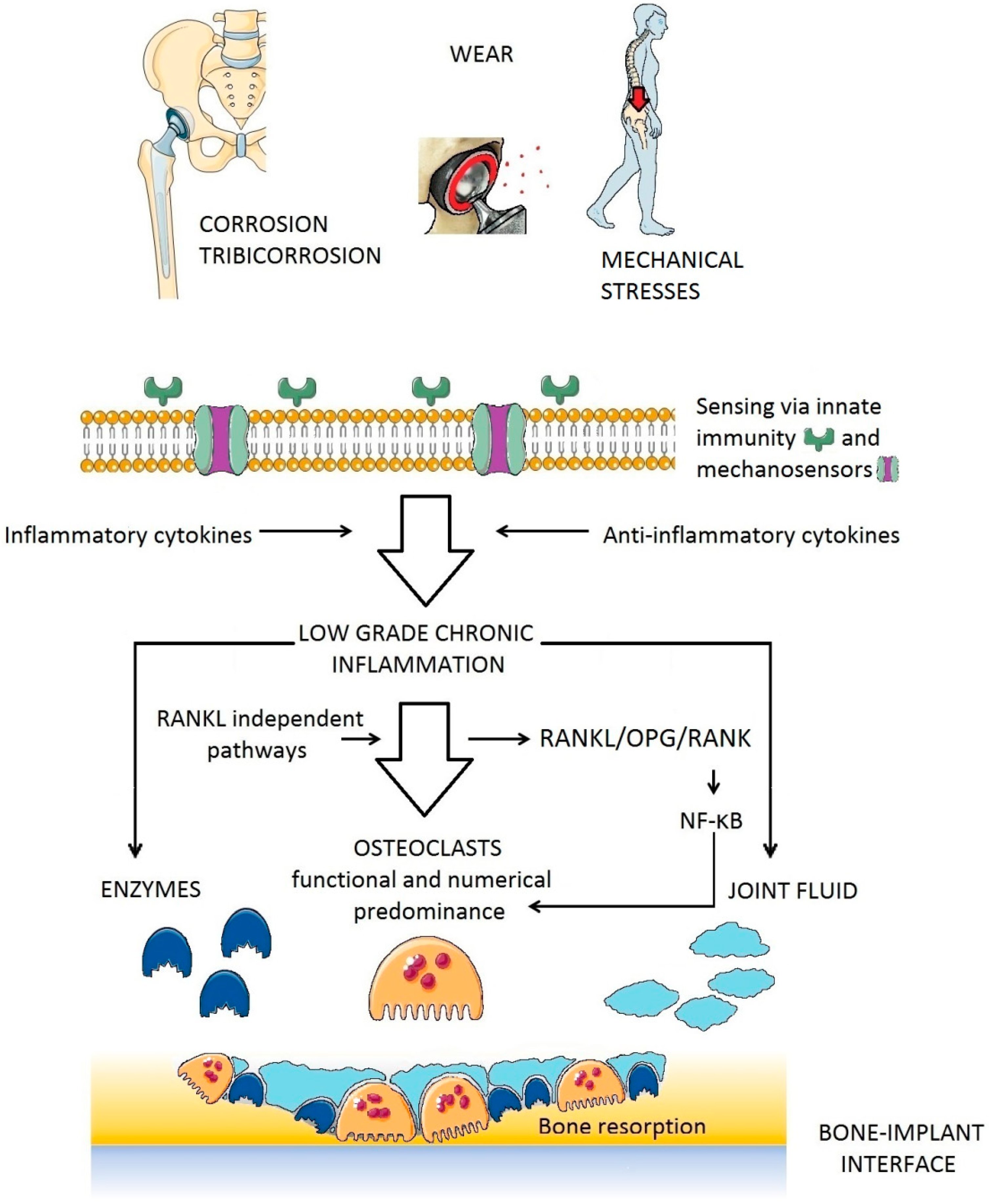
2. Mechanisms Underlying Periprosthetic Osteolysis
2.1. The Case of Inflammatory Osteolysis
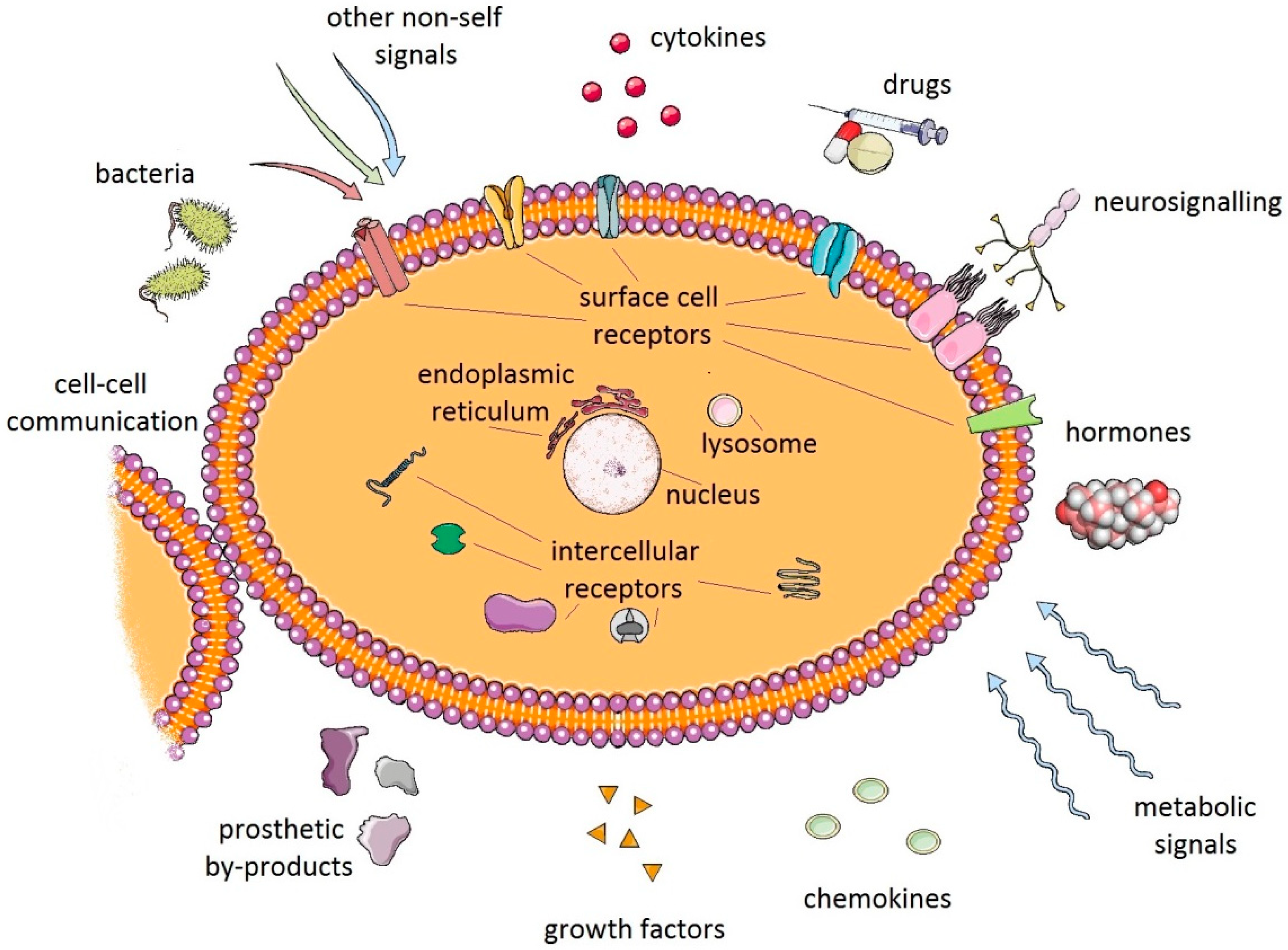
2.1.1. Stimuli of Periprosthetic Inflammation
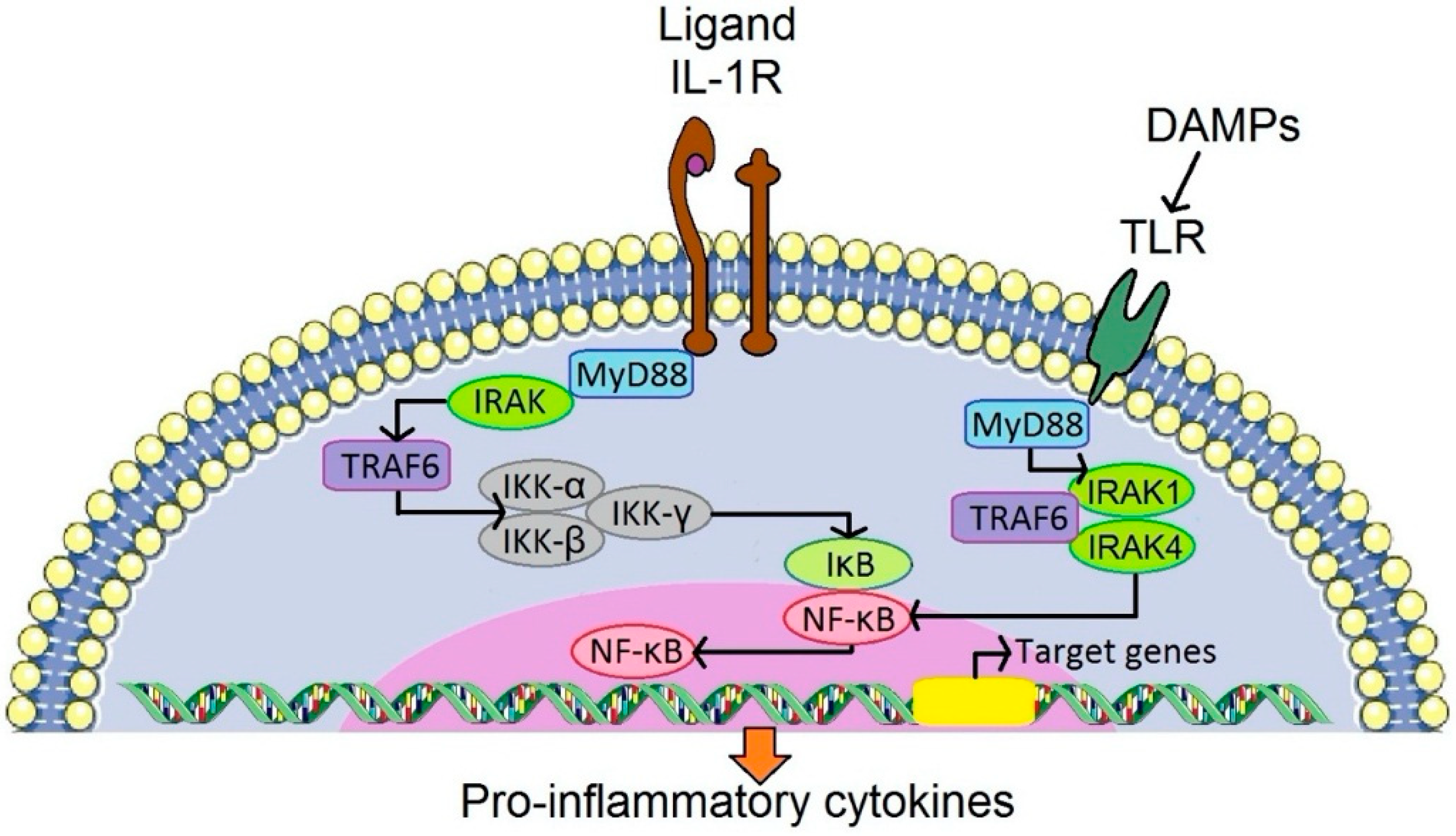
2.1.2. From Stimulation of Receptors of Innate Immunity to Fueling of Inflammation
2.1.3. Regulation of Debris-Induced Inflammation
2.2. Inflammation-Associated Bone Resorption
2.3. The Implant–Bone Interface: A Series of Bone Multicellular Units
2.4. Mechanical Osteolysis and Synergies with Inflammatory Agents
3. Treatments for Periprosthetic Osteolysis
3.1. Operative Treatment
3.2. Non-Operative Treatment
3.2.1. Bisphosphonates
3.2.2. Other Treatments Aimed at Osteoclasts
3.2.3. Treatments Aimed at Specific Pro-Inflammatory Cytokines
3.2.4. Treatments Targeted to Events More Upstream from the Inflammatory Cytokines
3.2.5. Anabolic Treatments
3.2.6. Cell Therapy
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Hart, A.; Janz, V.; Trousdale, R.T.; Sierra, R.J.; Berry, D.J.; Abdel, M.P. Long-Term Survivorship of Total Hip Arthroplasty with Highly Cross-Linked Polyethylene for Osteonecrosis. J. Bone Jt. Surg. Am. 2019, 101, 1563–1568. [Google Scholar] [CrossRef] [PubMed]
- Evans, J.T.; Evans, J.P.; Walker, R.W.; Blom, A.W.; Whitehouse, M.R.; Sayers, A. How long does a hip replacement last? A systematic review and meta-analysis of case series and national registry reports with more than 15 years of follow-up. Lancet 2019, 393, 647–654. [Google Scholar] [CrossRef]
- Pajarinen, J.; Gallo, J.; Takagi, M.; Goodman, S.B.; Mjoberg, B. Particle disease really does exist. Acta Orthop. 2018, 89, 133–136. [Google Scholar] [CrossRef] [PubMed]
- Evans, J.T.; Walker, R.W.; Evans, J.P.; Blom, A.W.; Sayers, A.; Whitehouse, M.R. How long does a knee replacement last? A systematic review and meta-analysis of case series and national registry reports with more than 15 years of follow-up. Lancet 2019, 393, 655–663. [Google Scholar] [CrossRef]
- Delanois, R.E.; Mistry, J.B.; Gwam, C.U.; Mohamed, N.S.; Choksi, U.S.; Mont, M.A. Current Epidemiology of Revision Total Knee Arthroplasty in the United States. J. Arthroplast. 2017, 32, 2663–2668. [Google Scholar] [CrossRef]
- Gwam, C.U.; Mistry, J.B.; Mohamed, N.S.; Thomas, M.; Bigart, K.C.; Mont, M.A.; Delanois, R.E. Current Epidemiology of Revision Total Hip Arthroplasty in the United States: National Inpatient Sample 2009 to 2013. J. Arthroplast. 2017, 32, 2088–2092. [Google Scholar] [CrossRef]
- Gallo, J.; Goodman, S.B.; Konttinen, Y.T.; Raska, M. Particle disease: Biologic mechanisms of periprosthetic osteolysis in total hip arthroplasty. Innate Immun. 2013, 19, 213–224. [Google Scholar] [CrossRef]
- Gallo, J.; Goodman, S.B.; Konttinen, Y.T.; Wimmer, M.A.; Holinka, M. Osteolysis around total knee arthroplasty: A review of pathogenetic mechanisms. Acta Biomater. 2013, 9, 8046–8058. [Google Scholar] [CrossRef]
- Anderson, J.M.; Rodriguez, A.; Chang, D.T. Foreign body reaction to biomaterials. Semin. Immunol. 2008, 20, 86–100. [Google Scholar] [CrossRef]
- Greenfield, E.M.; Bi, Y.; Ragab, A.A.; Goldberg, V.M.; Nalepka, J.L.; Seabold, J.M. Does endotoxin contribute to aseptic loosening of orthopedic implants? J. Biomed. Mater. Res. Part B Appl. Biomater. 2005, 72, 179–185. [Google Scholar] [CrossRef]
- Fisher, J.; Bell, J.; Barbour, P.S.; Tipper, J.L.; Matthews, J.B.; Besong, A.A.; Stone, M.H.; Ingham, E. A novel method for the prediction of functional biological activity of polyethylene wear debris. Proc. Inst. Mech. Eng. Part H J. Eng. Med. 2001, 215, 127–132. [Google Scholar] [CrossRef]
- Ihn, H.J.; Kim, K.; Cho, H.S.; Park, E.K. Pentamidine Inhibits Titanium Particle-Induced Osteolysis In Vivo and Receptor Activator of Nuclear Factor-kappaB Ligand-Mediated Osteoclast Differentiation In Vitro. Tissue Eng. Regen. Med. 2019, 16, 265–273. [Google Scholar] [CrossRef] [PubMed]
- Koehler, M.I.; Hartmann, E.S.; Schluessel, S.; Beck, F.; Redeker, J.I.; Schmitt, B.; Unger, M.; van Griensven, M.; Summer, B.; Fottner, A.; et al. Impact of Periprosthetic Fibroblast-Like Cells on Osteoclastogenesis in Co-Culture with Peripheral Blood Mononuclear Cells Varies Depending on Culture System. Int. J. Mol. Sci. 2019, 20, 2583. [Google Scholar] [CrossRef]
- Lei, P.; Dai, Z.; Zhang, Y.S.; Liu, H.; Niu, W.; Li, K.; Wang, L.; Hu, Y.; Xie, J. Macrophage inhibits the osteogenesis of fibroblasts in ultrahigh molecular weight polyethylene (UHMWPE) wear particle-induced osteolysis. J. Orthop. Surg. Res. 2019, 14, 80. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Y.; Jia, T.; Gong, W.; Wooley, P.H.; Yang, S.Y. Titanium particle-challenged osteoblasts promote osteoclastogenesis and osteolysis in a murine model of periprosthestic osteolysis. Acta Biomater. 2013, 9, 7564–7572. [Google Scholar] [CrossRef] [PubMed]
- Taki, N.; Tatro, J.M.; Nalepka, J.L.; Togawa, D.; Goldberg, V.M.; Rimnac, C.M.; Greenfield, E.M. Polyethylene and titanium particles induce osteolysis by similar, lymphocyte-independent, mechanisms. J. Orthop. Res. 2005, 23, 376–383. [Google Scholar] [CrossRef] [PubMed]
- Bechtel, C.P.; Gebhart, J.J.; Tatro, J.M.; Kiss-Toth, E.; Wilkinson, J.M.; Greenfield, E.M. Particle-Induced Osteolysis Is Mediated by TIRAP/Mal in Vitro and in Vivo: Dependence on Adherent Pathogen-Associated Molecular Patterns. J. Bone Jt. Surg. Am. 2016, 98, 285–294. [Google Scholar] [CrossRef]
- El-Warrak, A.O.; Olmstead, M.; Schneider, R.; Meinel, L.; Bettschart-Wolfisberger, R.; Akens, M.K.; Auer, J.; von Rechenberg, B. An experimental animal model of aseptic loosening of hip prostheses in sheep to study early biochemical changes at the interface membrane. BMC Musculoskelet. Disord. 2004, 5, 7. [Google Scholar] [CrossRef]
- Huang, C.H.; Lu, Y.C.; Chang, T.K.; Hsiao, I.L.; Su, Y.C.; Yeh, S.T.; Fang, H.W.; Huang, C.H. In vivo biological response to highly cross-linked and vitamin e-doped polyethylene-a particle-Induced osteolysis animal study. J. Biomed. Mater. Res. Part B Appl. Biomater. 2015, 104, 561–567. [Google Scholar] [CrossRef]
- Grosse, S.; Haugland, H.K.; Lilleng, P.; Ellison, P.; Hallan, G.; Hol, P.J. Wear particles and ions from cemented and uncemented titanium-based hip prostheses—A histological and chemical analysis of retrieval material. J. Biomed. Mater. Res. Part B Appl. Biomater. 2015, 103, 709–717. [Google Scholar] [CrossRef]
- Vaculova, J.; Gallo, J.; Hurnik, P.; Motyka, O.; Goodman, S.B.; Dvorackova, J. Low intrapatient variability of histomorphological findings in periprosthetic tissues from revised metal/ceramic on polyethylene joint arthroplasties. J. Biomed. Mater. Res. B Appl. Biomater. 2018, 106, 2008–2018. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Hall, D.J.; McCarthy, S.M.; Jacobs, J.J.; Urban, R.M.; Pourzal, R. Fourier transform infrared spectroscopic imaging of wear and corrosion products within joint capsule tissue from total hip replacements patients. J. Biomed. Mater. Res. B Appl. Biomater. 2019. [Google Scholar] [CrossRef]
- Goodman, S.B.; Huie, P.; Song, Y.; Lee, K.; Doshi, A.; Rushdieh, B.; Woolson, S.; Maloney, W.; Schurman, D.; Sibley, R. Loosening and osteolysis of cemented joint arthroplasties. A biologic spectrum. Clin. Orthop. Relat. Res. 1997, 337, 149–163. [Google Scholar] [CrossRef] [PubMed]
- Gallo, J.; Vaculova, J.; Goodman, S.B.; Konttinen, Y.T.; Thyssen, J.P. Contributions of human tissue analysis to understanding the mechanisms of loosening and osteolysis in total hip replacement. Acta Biomater. 2014, 10, 2354–2366. [Google Scholar] [CrossRef]
- Maitra, R.; Clement, C.C.; Scharf, B.; Crisi, G.M.; Chitta, S.; Paget, D.; Purdue, P.E.; Cobelli, N.; Santambrogio, L. Endosomal damage and TLR2 mediated inflammasome activation by alkane particles in the generation of aseptic osteolysis. Mol. Immunol. 2009, 47, 175–184. [Google Scholar] [CrossRef]
- O’Shea, J.J.; Gadina, M.; Siegel, R.M. Cytokines and cytokine receptors. In Clinical Immunology Principles and Practice, 5th ed.; Rich, R.R., Ed.; Elsevier: Amsterdam, The Netherlands, 2019; Volume 1, pp. 127–155. [Google Scholar]
- McKellop, H.A. The lexicon of polyethylene wear in artificial joints. Biomaterials 2007, 28, 5049–5057. [Google Scholar] [CrossRef] [PubMed]
- Singh, G.; Reichard, T.; Hameister, R.; Awiszus, F.; Schenk, K.; Feuerstein, B.; Roessner, A.; Lohmann, C. Ballooning osteolysis in 71 failed total ankle arthroplasties. Acta Orthop. 2016, 87, 401–405. [Google Scholar] [CrossRef]
- Baxter, R.M.; MacDonald, D.W.; Kurtz, S.M.; Steinbeck, M.J. Characteristics of highly cross-linked polyethylene wear debris in vivo. J. Biomed. Mater. Res. Part B Appl. Biomater. 2013, 101, 467–475. [Google Scholar] [CrossRef]
- Green, T.R.; Fisher, J.; Stone, M.; Wroblewski, B.M.; Ingham, E. Polyethylene particles of a ‘critical size’ are necessary for the induction of cytokines by macrophages in vitro. Biomaterials 1998, 19, 2297–2302. [Google Scholar] [CrossRef]
- Illgen, R.L., II; Bauer, L.M.; Hotujec, B.T.; Kolpin, S.E.; Bakhtiar, A.; Forsythe, T.M. Highly crosslinked vs. conventional polyethylene particles: Relative in vivo inflammatory response. J. Arthroplast. 2009, 24, 117–124. [Google Scholar] [CrossRef]
- Kobayashi, A.; Bonfield, W.; Kadoya, Y.; Yamac, T.; Freeman, M.A.; Scott, G.; Revell, P.A. The size and shape of particulate polyethylene wear debris in total joint replacements. Proc. Inst. Mech. Eng. Part H J. Eng. Med. 1997, 211, 11–15. [Google Scholar] [CrossRef] [PubMed]
- Bertrand, J.; Delfosse, D.; Mai, V.; Awiszus, F.; Harnisch, K.; Lohmann, C.H. Ceramic prosthesis surfaces induce an inflammatory cell response and fibrotic tissue changes. Bone Jt. J. 2018, 100B, 882–890. [Google Scholar] [CrossRef] [PubMed]
- Cooper, H.J.; Della Valle, C.J.; Berger, R.A.; Tetreault, M.; Paprosky, W.G.; Sporer, S.M.; Jacobs, J.J. Corrosion at the head-neck taper as a cause for adverse local tissue reactions after total hip arthroplasty. J. Bone Jt. Surg. Am. Vol. 2012, 94, 1655–1661. [Google Scholar] [CrossRef] [PubMed]
- Xia, Z.; Ricciardi, B.F.; Liu, Z.; von Ruhland, C.; Ward, M.; Lord, A.; Hughes, L.; Goldring, S.R.; Purdue, E.; Murray, D.; et al. Nano-analyses of wear particles from metal-on-metal and non-metal-on-metal dual modular neck hip arthroplasty. Nanomedicine 2017, 13, 1205–1217. [Google Scholar] [CrossRef] [PubMed]
- Ricciardi, B.F.; Nocon, A.A.; Jerabek, S.A.; Wilner, G.; Kaplowitz, E.; Goldring, S.R.; Purdue, P.E.; Perino, G. Histopathological characterization of corrosion product associated adverse local tissue reaction in hip implants: A study of 285 cases. BMC Clin. Pathol. 2016, 16, 3. [Google Scholar] [CrossRef]
- Cerquiglini, A.; Henckel, J.; Hothi, H.S.; Di Laura, A.; Skinner, J.A.; Hart, A.J. Inflammatory cell-induced corrosion in total knee arthroplasty: A retrieval study. J. Biomed. Mater. Res. B Appl. Biomater. 2018, 106, 460–467. [Google Scholar] [CrossRef]
- Teo, W.Z.; Schalock, P.C. Metal Hypersensitivity Reactions to Orthopedic Implants. Dermatol. Ther. (Heidelb) 2017, 7, 53–64. [Google Scholar] [CrossRef]
- Ingram, J.H.; Kowalski, R.; Fisher, J.; Ingham, E. The osteolytic response of macrophages to challenge with particles of Simplex P, Endurance, Palacos R, and Vertebroplastic bone cement particles in vitro. J. Biomed. Mater. Res. B Appl. Biomater. 2005, 75, 210–220. [Google Scholar] [CrossRef]
- Vega, F.; Bazire, R.; Belver, M.T.; Mugica, M.V.; Urquia, A.; Blanco, C. Aseptic loosening of a total knee prosthesis caused by delayed hypersensitivity to bone cement. Ann. Allergy Asthma Immunol. 2016, 117, 89–91. [Google Scholar] [CrossRef]
- Sabokbar, A.; Pandey, R.; Athanasou, N.A. The effect of particle size and electrical charge on macrophage-osteoclast differentiation and bone resorption. J. Mater. Sci. Mater. Med. 2003, 14, 731–738. [Google Scholar] [CrossRef]
- Green, T.R.; Fisher, J.; Matthews, J.B.; Stone, M.H.; Ingham, E. Effect of size and dose on bone resorption activity of macrophages by in vitro clinically relevant ultra high molecular weight polyethylene particles. J. Biomed. Mater. Res. 2000, 53, 490–497. [Google Scholar] [CrossRef]
- Nalepka, J.L.; Lee, M.J.; Kraay, M.J.; Marcus, R.E.; Goldberg, V.M.; Chen, X.; Greenfield, E.M. Lipopolysaccharide found in aseptic loosening of patients with inflammatory arthritis. Clin. Orthop. Relat. Res. 2006, 451, 229–235. [Google Scholar] [CrossRef] [PubMed]
- Hoenders, C.S.; Harmsen, M.C.; van Luyn, M.J. The local inflammatory environment and microorganisms in “aseptic” loosening of hip prostheses. J. Biomed. Mater. Res. Part B Appl. Biomater. 2008, 86, 291–301. [Google Scholar] [CrossRef] [PubMed]
- Manzano, G.W.; Fort, B.P.; Dubyak, G.R.; Greenfield, E.M. Wear Particle-induced Priming of the NLRP3 Inflammasome Depends on Adherent Pathogen-associated Molecular Patterns and Their Cognate Toll-like Receptors: An In Vitro Study. Clin. Orthop. Relat. Res. 2018, 476, 2442–2453. [Google Scholar] [CrossRef] [PubMed]
- Josse, J.; Valour, F.; Maali, Y.; Diot, A.; Batailler, C.; Ferry, T.; Laurent, F. Interaction Between Staphylococcal Biofilm and Bone: How Does the Presence of Biofilm Promote Prosthesis Loosening? Front. Microbiol. 2019, 10, 1602. [Google Scholar] [CrossRef] [PubMed]
- Ren, W.; Blasier, R.; Peng, X.; Shi, T.; Wooley, P.H.; Markel, D. Effect of oral erythromycin therapy in patients with aseptic loosening of joint prostheses. Bone 2009, 44, 671–677. [Google Scholar] [CrossRef]
- Zhu, L.; Kang, H.; Guo, C.A.; Fan, W.S.; Wang, Y.M.; Deng, L.F.; Yan, Z.Q. Rifampin suppresses osteoclastogenesis and titanium particle-induced osteolysis via modulating RANKL signaling pathways. Biochem. Biophys. Res. Commun. 2017, 484, 64–70. [Google Scholar] [CrossRef]
- Croes, M.; Kruyt, M.C.; Boot, W.; Pouran, B.; Braham, M.V.; Pakpahan, S.A.; Weinans, H.; Vogely, H.C.; Fluit, A.C.; Dhert, W.J.; et al. The role of bacterial stimuli in inflammation-driven bone formation. Eur. Cells Mater. 2019, 37, 402–419. [Google Scholar] [CrossRef]
- Pearl, J.I.; Ma, T.; Irani, A.R.; Huang, Z.; Robinson, W.H.; Smith, R.L.; Goodman, S.B. Role of the Toll-like receptor pathway in the recognition of orthopedic implant wear-debris particles. Biomaterials 2011, 32, 5535–5542. [Google Scholar] [CrossRef]
- Wang, H.; Li, Z.; Wang, C.; Xiao, F.; Gao, Y.; Zhang, X.; Wang, P.; Peng, J.; Cai, G.; Zuo, B.; et al. The inhibition of RANKL expression in fibroblasts attenuate CoCr particles induced aseptic prosthesis loosening via the MyD88-independent TLR signaling pathway. Biochem. Biophys. Res. Commun. 2018, 503, 1115–1122. [Google Scholar] [CrossRef]
- Valladares, R.D.; Nich, C.; Zwingenberger, S.; Li, C.; Swank, K.R.; Gibon, E.; Rao, A.J.; Yao, Z.; Goodman, S.B. Toll-like receptors-2 and 4 are overexpressed in an experimental model of particle-induced osteolysis. J. Biomed. Mater. Res. Part A 2014, 102, 3004–3011. [Google Scholar] [CrossRef]
- Wang, A.; Medzhitov, R. Counting Calories: The Cost of Inflammation. Cell 2019, 177, 223–224. [Google Scholar] [CrossRef]
- Seong, S.Y.; Matzinger, P. Hydrophobicity: An ancient damage-associated molecular pattern that initiates innate immune responses. Nat. Rev. Immunol. 2004, 4, 469–478. [Google Scholar] [CrossRef] [PubMed]
- Gallo, J.; Raska, M.; Konttinen, Y.T.; Nich, C.; Goodman, S.B. Innate immunity sensors participating in pathophysiology of joint diseases: A brief overview. J. Long Term Eff. Med. Implant. 2014, 24, 297–317. [Google Scholar] [CrossRef]
- Zhao, B. Does TNF Promote or Restrain Osteoclastogenesis and Inflammatory Bone Resorption? Crit. Rev. Immunol. 2018, 38, 253–261. [Google Scholar] [CrossRef]
- Adamopoulos, I.E. Inflammation in bone physiology and pathology. Curr. Opin. Rheumatol. 2018, 30, 59–64. [Google Scholar] [CrossRef]
- Alippe, Y.; Mbalaviele, G. Omnipresence of inflammasome activities in inflammatory bone diseases. Semin. Immunopathol. 2019, 41, 607–618. [Google Scholar] [CrossRef]
- Dyskova, T.; Gallo, J.; Kriegova, E. The Role of the Chemokine System in Tissue Response to Prosthetic By-products Leading to Periprosthetic Osteolysis and Aseptic Loosening. Front. Immunol. 2017, 8, 1026. [Google Scholar] [CrossRef] [PubMed]
- Maruotti, N.; Corrado, A.; Rotondo, C.; Cantatore, F.P. Janus kinase inhibitors role in bone remodeling. J. Cell. Physiol. 2019. [Google Scholar] [CrossRef]
- Murphy, P.M. Chemokines and chemokine receptors. In Clinical Immunology Principles and Practice, 5th ed.; Rich, R.R., Ed.; Elsevier: Amsterdam, The Netherlands, 2019; Volume 1, pp. 157–170. [Google Scholar]
- Gong, T.; Liu, L.; Jiang, W.; Zhou, R. DAMP-sensing receptors in sterile inflammation and inflammatory diseases. Nat. Rev. Immunol. 2019. [Google Scholar] [CrossRef]
- Tonnus, W.; Linkermann, A. Regulated necrosis and its immunogenicity. In Clinical Immunology Principles and Practice, 5th ed.; Rich, R.R., Ed.; Elsevier: Amsterdam, The Netherlands, 2019; Volume 1, pp. 197–205. [Google Scholar]
- Steinbeck, M.J.; Jablonowski, L.J.; Parvizi, J.; Freeman, T.A. The role of oxidative stress in aseptic loosening of total hip arthroplasties. J. Arthroplast. 2014, 29, 843–849. [Google Scholar] [CrossRef]
- Hukkanen, M.; Corbett, S.A.; Batten, J.; Konttinen, Y.T.; McCarthy, I.D.; Maclouf, J.; Santavirta, S.; Hughes, S.P.; Polak, J.M. Aseptic loosening of total hip replacement. Macrophage expression of inducible nitric oxide synthase and cyclo-oxygenase-2, together with peroxynitrite formation, as a possible mechanism for early prosthesis failure. J. Bone Jt. Surg. Br. Vol. 1997, 79, 467–474. [Google Scholar] [CrossRef]
- Hukkanen, M.; Corbett, S.A.; Platts, L.A.; Konttinen, Y.T.; Santavirta, S.; Hughes, S.P.; Polak, J.M. Nitric oxide in the local host reaction to total hip replacement. Clin. Orthop. Relat. Res. 1998, 352, 53–65. [Google Scholar] [CrossRef]
- Agidigbi, T.S.; Kim, C. Reactive Oxygen Species in Osteoclast Differentiation and Possible Pharmaceutical Targets of ROS-Mediated Osteoclast Diseases. Int. J. Mol. Sci. 2019, 20, 3576. [Google Scholar] [CrossRef]
- Luan, H.H.; Wang, A.; Hilliard, B.K.; Carvalho, F.; Rosen, C.E.; Ahasic, A.M.; Herzog, E.L.; Kang, I.; Pisani, M.A.; Yu, S.; et al. GDF15 Is an Inflammation-Induced Central Mediator of Tissue Tolerance. Cell 2019, 178, 1231–1244. [Google Scholar] [CrossRef] [PubMed]
- Ingham, E.; Fisher, J. The role of macrophages in osteolysis of total joint replacement. Biomaterials 2005, 26, 1271–1286. [Google Scholar] [CrossRef] [PubMed]
- Nich, C.; Takakubo, Y.; Pajarinen, J.; Gallo, J.; Konttinen, Y.T.; Takagi, M.; Goodman, S.B. The Role of Macrophages in the Biological Reaction to Wear Debris from Artificial Joints. J. Long Term Eff. Med. Implant. 2016, 26, 303–309. [Google Scholar] [CrossRef]
- Konttinen, Y.T.; Pajarinen, J.; Takakubo, Y.; Gallo, J.; Nich, C.; Takagi, M.; Goodman, S.B. Macrophage polarization and activation in response to implant debris: Influence by “particle disease” and “ion disease”. J. Long Term Eff. Med. Implant. 2014, 24, 267–281. [Google Scholar] [CrossRef]
- Jamsen, E.; Kouri, V.P.; Olkkonen, J.; Cor, A.; Goodman, S.B.; Konttinen, Y.T.; Pajarinen, J. Characterization of macrophage polarizing cytokines in the aseptic loosening of total hip replacements. J. Orthop. Res. Off. Publ. Orthop. Res. Soc. 2014, 32, 1241–1246. [Google Scholar] [CrossRef]
- Jamsen, E.; Kouri, V.P.; Ainola, M.; Goodman, S.B.; Nordstrom, D.C.; Eklund, K.K.; Pajarinen, J. Correlations between macrophage polarizing cytokines, inflammatory mediators, osteoclast activity, and toll-like receptors in tissues around aseptically loosened hip implants. J. Biomed. Mater. Res. Part A 2017, 105, 454–463. [Google Scholar] [CrossRef]
- Hopf, F.; Thomas, P.; Sesselmann, S.; Thomsen, M.N.; Hopf, M.; Hopf, J.; Krukemeyer, M.G.; Resch, H.; Krenn, V. CD3+ lymphocytosis in the peri-implant membrane of 222 loosened joint endoprostheses depends on the tribological pairing. Acta Orthop. 2017, 88, 642–648. [Google Scholar] [CrossRef] [PubMed]
- Dapunt, U.; Giese, T.; Prior, B.; Gaida, M.M.; Hansch, G.M. Infectious versus non-infectious loosening of implants: Activation of T lymphocytes differentiates between the two entities. Int. Orthop. 2014, 38, 1291–1296. [Google Scholar] [CrossRef] [PubMed]
- Christiansen, R.J.; Munch, H.J.; Bonefeld, C.M.; Thyssen, J.P.; Sloth, J.J.; Geisler, C.; Soballe, K.; Jellesen, M.S.; Jakobsen, S.S. Cytokine Profile in Patients with Aseptic Loosening of Total Hip Replacements and Its Relation to Metal Release and Metal Allergy. J. Clin. Med. 2019, 8, 1259. [Google Scholar] [CrossRef] [PubMed]
- Soehnlein, O.; Steffens, S.; Hidalgo, A.; Weber, C. Neutrophils as protagonists and targets in chronic inflammation. Nat. Rev. Immunol. 2017, 17, 248–261. [Google Scholar] [CrossRef]
- Ng, L.G.; Ostuni, R.; Hidalgo, A. Heterogeneity of neutrophils. Nat. Rev. Immunol. 2019, 19, 255–265. [Google Scholar] [CrossRef]
- Qiu, J.; Beckman, M.J.; Qian, J.; Jiranek, W. Simultaneous labeling of mast cell proteases and protease mRNAs at the bone-implant interface of aseptically loosened hip implants. J. Orthop. Res. Off. Publ. Orthop. Res. Soc. 2005, 23, 942–948. [Google Scholar] [CrossRef]
- Solovieva, S.A.; Ceponis, A.; Konttinen, Y.T.; Takagi, M.; Suda, A.; Eklund, K.K.; Sorsa, T.; Santavirta, S. Mast cells in loosening of totally replaced hips. Clin. Orthop. Relat. Res. 1996, 322, 158–165. [Google Scholar] [CrossRef]
- Kou, P.M.; Babensee, J.E. Macrophage and dendritic cell phenotypic diversity in the context of biomaterials. J. Biomed. Mater. Res. Part A 2011, 96, 239–260. [Google Scholar] [CrossRef]
- Vaculova, J.; Hurnik, P.; Gallo, J.; Ziak, D.; Motyka, O. Immunohistochemical Detection of Mast and Dendritic Cells in Periprosthetic Tissues of Aseptically Loosened Total Prostheses. Acta Chir. Orthop. Traumatol. Cechoslov. 2018, 85, 351–358. [Google Scholar]
- Ormsby, R.T.; Solomon, L.B.; Yang, D.; Crotti, T.N.; Haynes, D.R.; Findlay, D.M.; Atkins, G.J. Osteocytes respond to particles of clinically-relevant conventional and cross-linked polyethylene and metal alloys by up-regulation of resorptive and inflammatory pathways. Acta Biomater. 2019, 87, 296–306. [Google Scholar] [CrossRef]
- Ormsby, R.T.; Cantley, M.; Kogawa, M.; Solomon, L.B.; Haynes, D.R.; Findlay, D.M.; Atkins, G.J. Evidence that osteocyte perilacunar remodelling contributes to polyethylene wear particle induced osteolysis. Acta Biomater. 2016, 33, 242–251. [Google Scholar] [CrossRef] [PubMed]
- Chovatiya, R.; Medzhitov, R. Stress, inflammation, and defense of homeostasis. Mol. Cell 2014, 54, 281–288. [Google Scholar] [CrossRef] [PubMed]
- El Kholy, K.; Freire, M.; Chen, T.; Van Dyke, T.E. Resolvin E1 Promotes Bone Preservation Under Inflammatory Conditions. Front. Immunol. 2018, 9, 1300. [Google Scholar] [CrossRef] [PubMed]
- Matzinger, P.; Kamala, T. Tissue-based class control: The other side of tolerance. Nat. Rev. Immunol. 2011, 11, 221–230. [Google Scholar] [CrossRef]
- Van den Bossche, J.; O’Neill, L.A.; Menon, D. Macrophage Immunometabolism: Where Are We (Going)? Trends Immunol. 2017, 38, 395–406. [Google Scholar] [CrossRef]
- Inoue, K.; Nakano, S.; Zhao, B. Osteoclastic microRNAs and their translational potential in skeletal diseases. Semin. Immunopathol. 2019, 41, 573–582. [Google Scholar] [CrossRef]
- Lee, M.J.; Lim, E.; Mun, S.; Bae, S.; Murata, K.; Ivashkiv, L.B.; Park-Min, K.H. Intravenous Immunoglobulin (IVIG) Attenuates TNF-Induced Pathologic Bone Resorption and Suppresses Osteoclastogenesis by Inducing A20 Expression. J. Cell. Physiol. 2016, 231, 449–458. [Google Scholar] [CrossRef]
- Mediero, A.; Wilder, T.; Shah, L.; Cronstein, B.N. Adenosine A2A receptor (A2AR) stimulation modulates expression of semaphorins 4D and 3A, regulators of bone homeostasis. FASEB J. 2018, 32, 3487–3501. [Google Scholar] [CrossRef]
- Saad, S.; Dharmapatni, A.; Crotti, T.N.; Cantley, M.D.; Algate, K.; Findlay, D.M.; Atkins, G.J.; Haynes, D.R. Semaphorin-3a, neuropilin-1 and plexin-A1 in prosthetic-particle induced bone loss. Acta Biomater. 2016, 30, 311–318. [Google Scholar] [CrossRef]
- Kadoya, Y.; Revell, P.A.; Kobayashi, A.; al-Saffar, N.; Scott, G.; Freeman, M.A. Wear particulate species and bone loss in failed total joint arthroplasties. Clin. Orthop. Relat. Res. 1997, 340, 118–129. [Google Scholar] [CrossRef]
- Uehara, S.; Udagawa, N.; Kobayashi, Y. Non-canonical Wnt signals regulate cytoskeletal remodeling in osteoclasts. Cell. Mol. Life Sci. 2018, 75, 3683–3692. [Google Scholar] [CrossRef] [PubMed]
- O’Brien, W.; Fissel, B.M.; Maeda, Y.; Yan, J.; Ge, X.; Gravallese, E.M.; Aliprantis, A.O.; Charles, J.F. RANK-Independent Osteoclast Formation and Bone Erosion in Inflammatory Arthritis. Arthritis Rheumatol. 2016, 68, 2889–2900. [Google Scholar] [CrossRef] [PubMed]
- Xing, L.; Xiu, Y.; Boyce, B.F. Osteoclast fusion and regulation by RANKL-dependent and independent factors. World J. Orthop. 2012, 3, 212–222. [Google Scholar] [CrossRef] [PubMed]
- Sabokbar, A.; Mahoney, D.J.; Hemingway, F.; Athanasou, N.A. Non-Canonical (RANKL-Independent) Pathways of Osteoclast Differentiation and Their Role in Musculoskeletal Diseases. Clin. Rev. Allergy Immunol. 2016, 51, 16–26. [Google Scholar] [CrossRef]
- Mandelin, J.; Li, T.F.; Liljestrom, M.; Kroon, M.E.; Hanemaaijer, R.; Santavirta, S.; Konttinen, Y.T. Imbalance of RANKL/RANK/OPG system in interface tissue in loosening of total hip replacement. J. Bone Jt. Surg. Br. Vol. 2003, 85, 1196–1201. [Google Scholar] [CrossRef]
- Masui, T.; Sakano, S.; Hasegawa, Y.; Warashina, H.; Ishiguro, N. Expression of inflammatory cytokines, RANKL and OPG induced by titanium, cobalt-chromium and polyethylene particles. Biomaterials 2005, 26, 1695–1702. [Google Scholar] [CrossRef] [PubMed]
- Veigl, D.; Niederlova, J.; Krystufkova, O. Periprosthetic osteolysis and its association with RANKL expression. Physiol. Res. Acad. Sci. Bohemoslov. 2007, 56, 455–462. [Google Scholar]
- Wang, C.T.; Lin, Y.T.; Chiang, B.L.; Lee, S.S.; Hou, S.M. Over-expression of receptor activator of nuclear factor-kappaB ligand (RANKL), inflammatory cytokines, and chemokines in periprosthetic osteolysis of loosened total hip arthroplasty. Biomaterials 2010, 31, 77–82. [Google Scholar] [CrossRef]
- Park, J.H.; Lee, N.K.; Lee, S.Y. Current Understanding of RANK Signaling in Osteoclast Differentiation and Maturation. Mol. Cells 2017, 40, 706–713. [Google Scholar] [CrossRef]
- Altaf, H.; Revell, P.A. Evidence for active antigen presentation by monocyte/macrophages in response to stimulation with particles: The expression of NFkappaB transcription factors and costimulatory molecules. Inflammopharmacology 2013, 21, 279–290. [Google Scholar] [CrossRef]
- Chen, Y.; Aiken, A.; Saw, S.; Weiss, A.; Fang, H.; Khokha, R. TIMP Loss Activates Metalloproteinase-TNFalpha-DKK1 Axis To Compromise Wnt Signaling and Bone Mass. J. Bone Min. Res. 2019, 34, 182–194. [Google Scholar] [CrossRef] [PubMed]
- Syggelos, S.A.; Aletras, A.J.; Smirlaki, I.; Skandalis, S.S. Extracellular matrix degradation and tissue remodeling in periprosthetic loosening and osteolysis: Focus on matrix metalloproteinases, their endogenous tissue inhibitors, and the proteasome. BioMed Res. Int. 2013, 2013, 230805. [Google Scholar] [CrossRef]
- Jonitz-Heincke, A.; Lochner, K.; Schulze, C.; Pohle, D.; Pustlauk, W.; Hansmann, D.; Bader, R. Contribution of human osteoblasts and macrophages to bone matrix degradation and proinflammatory cytokine release after exposure to abrasive endoprosthetic wear particles. Mol. Med. Rep. 2016, 14, 1491–1500. [Google Scholar] [CrossRef] [PubMed]
- Takei, I.; Takagi, M.; Santavirta, S.; Ida, H.; Ishii, M.; Ogino, T.; Ainola, M.; Konttinen, Y.T. Messenger ribonucleic acid expression of 16 matrix metalloproteinases in bone-implant interface tissues of loose artificial hip joints. J. Biomed. Mater. Res. 2000, 52, 613–620. [Google Scholar] [CrossRef]
- Niarakis, A.; Giannopoulou, E.; Ravazoula, P.; Panagiotopoulos, E.; Zarkadis, I.K.; Aletras, A.J. Detection of a latent soluble form of membrane type 1 matrix metalloprotease bound with tissue inhibitor of matrix metalloproteinases-2 in periprosthetic tissues and fluids from loose arthroplasty endoprostheses. FEBS J. 2013, 280, 6541–6555. [Google Scholar] [CrossRef] [PubMed]
- Niarakis, A.; Giannopoulou, E.; Syggelos, S.A.; Panagiotopoulos, E. Effects of proteasome inhibitors on cytokines, metalloproteinases and their inhibitors and collagen type-I expression in periprosthetic tissues and fibroblasts from loose arthroplasty endoprostheses. Connect. Tissue Res. 2019, 60, 555–570. [Google Scholar] [CrossRef]
- Drake, M.T.; Clarke, B.L.; Oursler, M.J.; Khosla, S. Cathepsin K Inhibitors for Osteoporosis: Biology, Potential Clinical Utility, and Lessons Learned. Endocr. Rev. 2017, 38, 325–350. [Google Scholar] [CrossRef]
- Arias, C.F.; Herrero, M.A.; Echeverri, L.F.; Oleaga, G.E.; Lopez, J.M. Bone remodeling: A tissue-level process emerging from cell-level molecular algorithms. PLoS ONE 2018, 13, e0204171. [Google Scholar] [CrossRef]
- Rommelt, C.; Munsch, T.; Drynda, A.; Lessmann, V.; Lohmann, C.H.; Bertrand, J. Periprosthetic hypoxia as consequence of TRPM7 mediated cobalt influx in osteoblasts. J. Biomed. Mater. Res. B Appl. Biomater. 2019, 107, 1806–1813. [Google Scholar] [CrossRef]
- Queally, J.M.; Devitt, B.M.; Butler, J.S.; Malizia, A.P.; Murray, D.; Doran, P.P.; O’Byrne, J.M. Cobalt ions induce chemokine secretion in primary human osteoblasts. J. Orthop. Res. 2009, 27, 855–864. [Google Scholar] [CrossRef]
- Perni, S.; Yang, L.; Preedy, E.C.; Prokopovich, P. Cobalt and Titanium nanoparticles influence on human osteoblast mitochondrial activity and biophysical properties of their cytoskeleton. J. Colloid Interface Sci. 2018, 531, 410–420. [Google Scholar] [CrossRef] [PubMed]
- Fahlgren, A.; Bratengeier, C.; Semeins, C.M.; Klein-Nulend, J.; Bakker, A.D. Supraphysiological loading induces osteocyte-mediated osteoclastogenesis in a novel in vitro model for bone implant loosening. J. Orthop. Res. 2018, 36, 1425–1434. [Google Scholar] [CrossRef] [PubMed]
- Gibon, E.; Yao, Z.; Rao, A.J.; Zwingenberger, S.; Batke, B.; Valladares, R.; Smith, R.L.; Biswal, S.; Gambhir, S.S.; Goodman, S.B. Effect of a CCR1 receptor antagonist on systemic trafficking of MSCs and polyethylene particle-associated bone loss. Biomaterials 2012, 33, 3632–3638. [Google Scholar] [CrossRef]
- Haleem-Smith, H.; Argintar, E.; Bush, C.; Hampton, D.; Postma, W.F.; Chen, F.H.; Rimington, T.; Lamb, J.; Tuan, R.S. Biological responses of human mesenchymal stem cells to titanium wear debris particles. J. Orthop. Res. 2012, 30, 853–863. [Google Scholar] [CrossRef] [PubMed]
- Preedy, E.C.; Perni, S.; Prokopovich, P. Cobalt and titanium nanoparticles influence on mesenchymal stem cell elasticity and turgidity. Colloids Surf. B Biointerfaces 2017, 157, 146–156. [Google Scholar] [CrossRef]
- Klein-Nulend, J.; Bacabac, R.G.; Bakker, A.D. Mechanical loading and how it affects bone cells: The role of the osteocyte cytoskeleton in maintaining our skeleton. Eur. Cell Mater. 2012, 24, 278–291. [Google Scholar] [CrossRef]
- Knutsen, A.R.; Lau, N.; Longjohn, D.B.; Ebramzadeh, E.; Sangiorgio, S.N. Periprosthetic femoral bone loss in total hip arthroplasty: Systematic analysis of the effect of stem design. Hip Int. 2017, 27, 26–34. [Google Scholar] [CrossRef]
- Cristofolini, L. Critical Examination of Stress Shielding Evaluation of Hip Prostheses. Crit. Rev. Biomed. Eng. 2017, 45, 549–623. [Google Scholar] [CrossRef]
- Norris, B.L.; Lang, G.; Russell, T.A.T.; Rothberg, D.L.; Ricci, W.M.; Borrelli, J., Jr. Absolute Versus Relative Fracture Fixation: Impact on Fracture Healing. J. Orthop. Trauma 2018, 32 (Suppl. 1), S12–S16. [Google Scholar] [CrossRef]
- Castillo, A.B.; Leucht, P. Bone Homeostasis and Repair: Forced Into Shape. Curr. Rheumatol. Rep. 2015, 17, 58. [Google Scholar] [CrossRef]
- Rodriguez-Merchan, E.C.; Forriol, F. Nonunion: General principles and experimental data. Clin. Orthop. Relat. Res. 2004, 419, 4–12. [Google Scholar] [CrossRef]
- MacQuarrie, R.A.; Fang Chen, Y.; Coles, C.; Anderson, G.I. Wear-particle-induced osteoclast osteolysis: The role of particulates and mechanical strain. J. Biomed. Mater. Res. B Appl. Biomater. 2004, 69, 104–112. [Google Scholar] [CrossRef]
- Nam, D.; Bostrom, M.P.; Fahlgren, A. Emerging ideas: Instability-induced periprosthetic osteolysis is not dependent on the fibrous tissue interface. Clin. Orthop. Relat. Res. 2013, 471, 1758–1762. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Fahlgren, A.; Bostrom, M.P.; Yang, X.; Johansson, L.; Edlund, U.; Agholme, F.; Aspenberg, P. Fluid pressure and flow as a cause of bone resorption. Acta Orthop. 2010, 81, 508–516. [Google Scholar] [CrossRef]
- Van der Vis, H.M.; Aspenberg, P.; Marti, R.K.; Tigchelaar, W.; Van Noorden, C.J. Fluid pressure causes bone resorption in a rabbit model of prosthetic loosening. Clin. Orthop. Relat. Res. 1998, 350, 201–208. [Google Scholar] [CrossRef]
- van der Vis, H.; Aspenberg, P.; de Kleine, R.; Tigchelaar, W.; van Noorden, C.J. Short periods of oscillating fluid pressure directed at a titanium-bone interface in rabbits lead to bone lysis. Acta Orthop. Scand. 1998, 69, 5–10. [Google Scholar] [CrossRef]
- Solis, A.G.; Bielecki, P.; Steach, H.R.; Sharma, L.; Harman, C.C.D.; Yun, S.; de Zoete, M.R.; Warnock, J.N.; To, S.D.F.; York, A.G.; et al. Mechanosensation of cyclical force by PIEZO1 is essential for innate immunity. Nature 2019, 573, 69–74. [Google Scholar] [CrossRef]
- Bratengeier, C.; Bakker, A.D.; Fahlgren, A. Mechanical loading releases osteoclastogenesis-modulating factors through stimulation of the P2X7 receptor in hematopoietic progenitor cells. J. Cell. Physiol. 2019, 234, 13057–13067. [Google Scholar] [CrossRef]
- Estell, E.G.; Murphy, L.A.; Silverstein, A.M.; Tan, A.R.; Shah, R.P.; Ateshian, G.A.; Hung, C.T. Fibroblast-like synoviocyte mechanosensitivity to fluid shear is modulated by interleukin-1alpha. J. Biomech. 2017, 60, 91–99. [Google Scholar] [CrossRef]
- Amirhosseini, M.; Andersson, G.; Aspenberg, P.; Fahlgren, A. Mechanical instability and titanium particles induce similar transcriptomic changes in a rat model for periprosthetic osteolysis and aseptic loosening. Bone Rep. 2017, 7, 17–25. [Google Scholar] [CrossRef]
- Mjoberg, B. Theories of wear and loosening in hip prostheses. Wear-induced loosening vs loosening-induced wear—A review. Acta Orthop. Scand. 1994, 65, 361–371. [Google Scholar] [CrossRef]
- Kurcz, B.; Lyons, J.; Sayeed, Z.; Anoushiravani, A.A.; Iorio, R. Osteolysis as it Pertains to Total Hip Arthroplasty. Orthop. Clin. N. Am. 2018, 49, 419–435. [Google Scholar] [CrossRef]
- Sheth, N.P.; Rozell, J.C.; Paprosky, W.G. Evaluation and Treatment of Patients With Acetabular Osteolysis After Total Hip Arthroplasty. J. Am. Acad. Orthop. Surg. 2019, 27, e258–e267. [Google Scholar] [CrossRef]
- Gittings, D.J.; Dattilo, J.R.; Hardaker, W.; Sheth, N.P. Evaluation and Treatment of Femoral Osteolysis Following Total Hip Arthroplasty. JBJS Rev. 2017, 5, e9. [Google Scholar] [CrossRef]
- Schmalzried, T.P.; Kwong, L.M.; Jasty, M.; Sedlacek, R.C.; Haire, T.C.; O’Connor, D.O.; Bragdon, C.R.; Kabo, J.M.; Malcolm, A.J.; Harris, W.H. The mechanism of loosening of cemented acetabular components in total hip arthroplasty. Analysis of specimens retrieved at autopsy. Clin. Orthop. Relat. Res. 1992, 274, 60–78. [Google Scholar]
- Sheth, N.P.; Nelson, C.L.; Paprosky, W.G. Femoral bone loss in revision total hip arthroplasty: Evaluation and management. J. Am. Acad. Orthop. Surg. 2013, 21, 601–612. [Google Scholar] [CrossRef] [PubMed]
- Liow, M.H.L.; Kwon, Y.M. Metal-on-metal total hip arthroplasty: Risk factors for pseudotumours and clinical systematic evaluation. Int. Orthop. 2017, 41, 885–892. [Google Scholar] [CrossRef]
- Dalling, J.G.; Math, K.; Scuderi, G.R. Evaluating the progression of osteolysis after total knee arthroplasty. J. Am. Acad. Orthop. Surg. 2015, 23, 173–180. [Google Scholar] [CrossRef]
- Skoldenberg, O.; Rysinska, A.; Eisler, T.; Salemyr, M.; Boden, H.; Muren, O. Denosumab for treating periprosthetic osteolysis; study protocol for a randomized, double-blind, placebo-controlled trial. BMC Musculoskelet. Disord. 2016, 17, 174. [Google Scholar] [CrossRef]
- Goodman, S.B.; Trindade, M.; Ma, T.; Genovese, M.; Smith, R.L. Pharmacologic modulation of periprosthetic osteolysis. Clin. Orthop. Relat. Res. 2005, 430, 39–45. [Google Scholar] [CrossRef]
- Goodman, S.B.; Gibon, E.; Pajarinen, J.; Lin, T.H.; Keeney, M.; Ren, P.G.; Nich, C.; Yao, Z.; Egashira, K.; Yang, F.; et al. Novel biological strategies for treatment of wear particle-induced periprosthetic osteolysis of orthopaedic implants for joint replacement. J. R. Soc. Interface 2014, 11, 20130962. [Google Scholar] [CrossRef] [PubMed]
- Smith, R.L.; Schwarz, E.M. Are biologic treatments a potential approach to wear- and corrosion-related problems? Clin. Orthop. Relat. Res. 2014, 472, 3740–3746. [Google Scholar] [CrossRef]
- Holt, G.; Reilly, J.; Meek, R.M. Effect of alendronate on pseudomembrane cytokine expression in patients with aseptic osteolysis. J. Arthroplast. 2010, 25, 958–963. [Google Scholar] [CrossRef]
- Wilkinson, J.M.; Little, D.G. Bisphosphonates in orthopedic applications. Bone 2011, 49, 95–102. [Google Scholar] [CrossRef]
- Cordova, L.A.; Trichet, V.; Escriou, V.; Rosset, P.; Amiaud, J.; Battaglia, S.; Charrier, C.; Berreur, M.; Brion, R.; Gouin, F.; et al. Inhibition of osteolysis and increase of bone formation after local administration of siRNA-targeting RANK in a polyethylene particle-induced osteolysis model. Acta Biomater. 2015, 13, 150–158. [Google Scholar] [CrossRef]
- Cummings, S.R.; San Martin, J.; McClung, M.R.; Siris, E.S.; Eastell, R.; Reid, I.R.; Delmas, P.; Zoog, H.B.; Austin, M.; Wang, A.; et al. Denosumab for prevention of fractures in postmenopausal women with osteoporosis. N. Engl. J. Med. 2009, 361, 756–765. [Google Scholar] [CrossRef]
- Purdue, P.E.; Koulouvaris, P.; Potter, H.G.; Nestor, B.J.; Sculco, T.P. The cellular and molecular biology of periprosthetic osteolysis. Clin. Orthop. Relat. Res. 2007, 454, 251–261. [Google Scholar] [CrossRef]
- Goodman, S.B.; Gibon, E.; Yao, Z. The basic science of periprosthetic osteolysis. Instr. Course Lect. 2013, 62, 201–206. [Google Scholar]
- Hallab, N.J.; Jacobs, J.J. Chemokines Associated with Pathologic Responses to Orthopedic Implant Debris. Front/ Endocrinol/ (Lausanne) 2017, 8, 5. [Google Scholar] [CrossRef]
- Howie, D.W.; Neale, S.D.; Haynes, D.R.; Holubowycz, O.T.; McGee, M.A.; Solomon, L.B.; Callary, S.A.; Atkins, G.J.; Findlay, D.M. Periprosthetic osteolysis after total hip replacement: Molecular pathology and clinical management. Inflammopharmacology 2013, 21, 389–396. [Google Scholar] [CrossRef]
- Janeway, C.A., Jr.; Medzhitov, R. Innate immune recognition. Annu. Rev. Immunol. 2002, 20, 197–216. [Google Scholar] [CrossRef]
- Abu-Amer, Y.; Darwech, I.; Clohisy, J.C. Aseptic loosening of total joint replacements: Mechanisms underlying osteolysis and potential therapies. Arthritis Res. Ther. 2007, 9 (Suppl. 1), S6. [Google Scholar] [CrossRef] [PubMed]
- Lin, T.-H.; Pajarinen, J.; Sato, T.; Loi, F.; Fan, C.; Córdova, L.A.; Nabeshima, A.; Gibon, E.; Zhang, R.; Yao, Z.; et al. NF-κB Decoy Oligodeoxynucleotide Mitigates Wear Particle-Associated Bone Loss in the Murine Continuous Infusion Model. Acta Biomater. 2016, 41, 273–281. [Google Scholar] [CrossRef] [PubMed]
- Lin, T.-H.; Yao, Z.; Sato, T.; Keeney, M.; Li, C.; Pajarinen, J.; Yang, F.; Egashira, K.; Goodman, S.B. Suppression of wear-particle-induced pro-inflammatory cytokine and chemokine production in macrophages via NF-κB decoy oligodeoxynucleotide: A preliminary report. Acta Biomater. 2014, 10, 3747–3755. [Google Scholar] [CrossRef] [PubMed]
- Lin, T.; Pajarinen, J.; Nabeshima, A.; Cordova, L.A.; Loi, F.; Gibon, E.; Lu, L.; Nathan, K.; Jamsen, E.; Yao, Z.; et al. Orthopaedic wear particle-induced bone loss and exogenous macrophage infiltration is mitigated by local infusion of NF-kappaB decoy oligodeoxynucleotide. J. Biomed. Mater. Res. A 2017, 105, 3169–3175. [Google Scholar] [CrossRef]
- Sato, T.; Pajarinen, J.; Lin, T.-H.; Tamaki, Y.; Loi, F.; Egashira, K.; Yao, Z.; Goodman, S.B. NF-κB decoy oligodeoxynucleotide inhibits wear particle-induced inflammation in a murine calvarial model. J. Biomed. Mater. Res. Part A 2015, 103, 3872–3878. [Google Scholar] [CrossRef][Green Version]
- Clohisy, J.C.; Hirayama, T.; Frazier, E.; Han, S.K.; Abu-Amer, Y. NF-kB signaling blockade abolishes implant particle-induced osteoclastogenesis. J. Orthop. Res. 2004, 22, 13–20. [Google Scholar] [CrossRef]
- Clohisy, J.C.; Yamanaka, Y.; Faccio, R.; Abu-Amer, Y. Inhibition of IKK activation, through sequestering NEMO, blocks PMMA-induced osteoclastogenesis and calvarial inflammatory osteolysis. J. Orthop. Res. 2006, 24, 1358–1365. [Google Scholar] [CrossRef]
- Yamanaka, Y.; Abu-Amer, Y.; Faccio, R.; Clohisy, J.C. Map kinase c-JUN N-terminal kinase mediates PMMA induction of osteoclasts. J. Orthop. Res. 2006, 24, 1349–1357. [Google Scholar] [CrossRef]
- Rao, A.J.; Nich, C.; Dhulipala, L.S.; Gibon, E.; Valladares, R.; Zwingenberger, S.; Smith, R.L.; Goodman, S.B. Local effect of IL-4 delivery on polyethylene particle induced osteolysis in the murine calvarium. J. Biomed. Mater. Res. Part A 2013, 101, 1926–1934. [Google Scholar] [CrossRef]
- Sato, T.; Pajarinen, J.; Behn, A.; Jiang, X.; Lin, T.-H.; Loi, F.; Yao, Z.; Egashira, K.; Yang, F.; Goodman, S.B. The effect of local IL-4 delivery or CCL2 blockade on implant fixation and bone structural properties in a mouse model of wear particle induced osteolysis. J. Biomed. Mater. Res. Part A 2016, 104, 2255–2262. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Wu, N.N.; Mou, Y.Q.; Chen, L.; Deng, Z.L. Inhibitory effects of recombinant IL-4 and recombinant IL-13 on UHMWPE-induced bone destruction in the murine air pouch model. J. Surg. Res. 2013, 180, e73–e81. [Google Scholar] [CrossRef] [PubMed]
- Nabeshima, A.; Pajarinen, J.; Lin, T.H.; Jiang, X.; Gibon, E.; Cordova, L.A.; Loi, F.; Lu, L.; Jamsen, E.; Egashira, K.; et al. Mutant CCL2 protein coating mitigates wear particle-induced bone loss in a murine continuous polyethylene infusion model. Biomaterials 2017, 117, 1–9. [Google Scholar] [CrossRef]
- Gibon, E.; Ma, T.; Ren, P.-G.; Fritton, K.; Biswal, S.; Yao, Z.; Smith, L.; Goodman, S.B. Selective inhibition of the MCP-1-CCR2 ligand-receptor axis decreases systemic trafficking of macrophages in the presence of UHMWPE particles. J. Orthop. Res. 2012, 30, 547–553. [Google Scholar] [CrossRef] [PubMed]
- Jiang, X.; Sato, T.; Yao, Z.; Keeney, M.; Pajarinen, J.; Lin, T.-H.; Loi, F.; Egashira, K.; Goodman, S.; Yang, F. Local delivery of mutant CCL2 protein-reduced orthopaedic implant wear particle-induced osteolysis and inflammation in vivo. J. Orthop. Res. 2016, 34, 58–64. [Google Scholar] [CrossRef]
- Ren, W.; Markel, D.C.; Schwendener, R.; Ding, Y.; Wu, B.; Wooley, P.H. Macrophage depletion diminishes implant-wear-induced inflammatory osteolysis in a mouse model. J. Biomed. Mater. Res. A 2008, 85, 1043–1051. [Google Scholar] [CrossRef]
- Ulrich-Vinther, M.; Carmody, E.E.; Goater, J.J.; Søballe, K.; O’Keefe, R.J.; Schwarz, E.M. Recombinant adeno-associated virus-mediated osteoprotegerin gene therapy inhibits wear debris-induced osteolysis. J. Bone Jt. Surg. Am. 2002, 84, 1405–1412. [Google Scholar] [CrossRef]
- Wang, J.; Tao, Y.; Ping, Z.; Zhang, W.; Hu, X.; Wang, Y.; Wang, L.; Shi, J.; Wu, X.; Yang, H.; et al. Icariin attenuates titanium-particle inhibition of bone formation by activating the Wnt/beta-catenin signaling pathway in vivo and in vitro. Sci. Rep. 2016, 6, 23827. [Google Scholar] [CrossRef]
- Ping, Z.; Hu, X.; Wang, L.; Shi, J.; Tao, Y.; Wu, X.; Hou, Z.; Guo, X.; Zhang, W.; Yang, H.; et al. Melatonin attenuates titanium particle-induced osteolysis via activation of Wnt/beta-catenin signaling pathway. Acta Biomater. 2017, 51, 513–525. [Google Scholar] [CrossRef]
- Ping, Z.; Wang, Z.; Shi, J.; Wang, L.; Guo, X.; Zhou, W.; Hu, X.; Wu, X.; Liu, Y.; Zhang, W.; et al. Inhibitory effects of melatonin on titanium particle-induced inflammatory bone resorption and osteoclastogenesis via suppression of NF-kappaB signaling. Acta Biomater. 2017, 62, 362–371. [Google Scholar] [CrossRef]
- Lu, Y.C.; Chang, T.K.; Yeh, S.T.; Fang, H.W.; Lin, C.Y.; Hsu, L.I.; Huang, C.H.; Huang, C.H. The potential role of strontium ranelate in treating particle-induced osteolysis. Acta Biomater. 2015, 20, 147–154. [Google Scholar] [CrossRef] [PubMed]
- Pajarinen, J.; Lin, T.; Gibon, E.; Kohno, Y.; Maruyama, M.; Nathan, K.; Lu, L.; Yao, Z.; Goodman, S.B. Mesenchymal stem cell-macrophage crosstalk and bone healing. Biomaterials 2019, 196, 80–89. [Google Scholar] [CrossRef] [PubMed]
- Lin, T.; Pajarinen, J.; Nabeshima, A.; Lu, L.; Nathan, K.; Yao, Z.; Goodman, S.B. Establishment of NF-kappaB sensing and interleukin-4 secreting mesenchymal stromal cells as an “on-demand” drug delivery system to modulate inflammation. Cytotherapy 2017, 19, 1025–1034. [Google Scholar] [CrossRef] [PubMed]
- Lin, T.; Kohno, Y.; Huang, J.F.; Romero-Lopez, M.; Maruyama, M.; Ueno, M.; Pajarinen, J.; Nathan, K.; Yao, Z.; Yang, F.; et al. Preconditioned or IL4-Secreting Mesenchymal Stem Cells Enhanced Osteogenesis at Different Stages. Tissue Eng. Part A 2019, 25, 1096–1103. [Google Scholar] [CrossRef] [PubMed]
- Lin, T.; Pajarinen, J.; Nabeshima, A.; Lu, L.; Nathan, K.; Jamsen, E.; Yao, Z.; Goodman, S.B. Preconditioning of murine mesenchymal stem cells synergistically enhanced immunomodulation and osteogenesis. Stem Cell Res. 2017, 8, 277. [Google Scholar] [CrossRef] [PubMed]
- Lin, T.; Pajarinen, J.; Kohno, Y.; Huang, J.F.; Maruyama, M.; Romero-Lopez, M.; Nathan, K.; Yao, Z.; Goodman, S.B. Trained murine mesenchymal stem cells have anti-inflammatory effect on macrophages, but defective regulation on T-cell proliferation. FASEB J. 2019, 33, 4203–4211. [Google Scholar] [CrossRef] [PubMed]
- Lin, T.; Kohno, Y.; Huang, J.-F.; Romero-Lopez, M.; Pajarinen, J.; Maruyama, M.; Nathan, K.; Yao, Z.; Goodman, S.B. NFκB sensing IL-4 secreting mesenchymal stem cells mitigate the proinflammatory response of macrophages exposed to polyethylene wear particles. J. Biomed. Mater. Res. Part A 2018, 106, 2744–2752. [Google Scholar] [CrossRef]
| Parameter | Potential Role for | Estimated Evidence |
|---|---|---|
| Particle size (Particles between 0.1 and 1 µm seem to be the most effective ones) | Induction/perpetuation of inflammation | Strong |
| Origin of particles (Polymers such as polyethylene and PMMA, and metal particles are the most inflammatory) | Intensity of inflammation; type of inflammation; induction of hypersensitivity | Strong |
| Particle shape (Irregular shaped particles with a rough surface topography are more inflammatory) | Intensity of inflammation; type of inflammation | Weak |
| Particle amount (A threshold for PPOL has been determined) | Induction/perpetuation/intensity | Strong |
| Particle surface charge (Need to be verified) | Intensity of inflammation; type of inflammation | Weak |
| Clearing capacity of periprosthetic tissues | Intensity of inflammation; type of inflammation | Moderate |
| Anti-inflammatory mechanisms | Regulate intensity of inflammation in periprosthetic tissues | Strong |
| Individual susceptibility (genetic make-up e.g., SNPs may predispose to a more aggressive inflammatory reaction) | Intensity of inflammation; type of inflammation; tissue homeostasis; osteoclast survival/activity | Moderate |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Goodman, S.B.; Gallo, J. Periprosthetic Osteolysis: Mechanisms, Prevention and Treatment. J. Clin. Med. 2019, 8, 2091. https://doi.org/10.3390/jcm8122091
Goodman SB, Gallo J. Periprosthetic Osteolysis: Mechanisms, Prevention and Treatment. Journal of Clinical Medicine. 2019; 8(12):2091. https://doi.org/10.3390/jcm8122091
Chicago/Turabian StyleGoodman, Stuart B., and Jiri Gallo. 2019. "Periprosthetic Osteolysis: Mechanisms, Prevention and Treatment" Journal of Clinical Medicine 8, no. 12: 2091. https://doi.org/10.3390/jcm8122091
APA StyleGoodman, S. B., & Gallo, J. (2019). Periprosthetic Osteolysis: Mechanisms, Prevention and Treatment. Journal of Clinical Medicine, 8(12), 2091. https://doi.org/10.3390/jcm8122091






