The Role of Animal Models in Developing Pharmacotherapy for Obstructive Sleep Apnea
Abstract
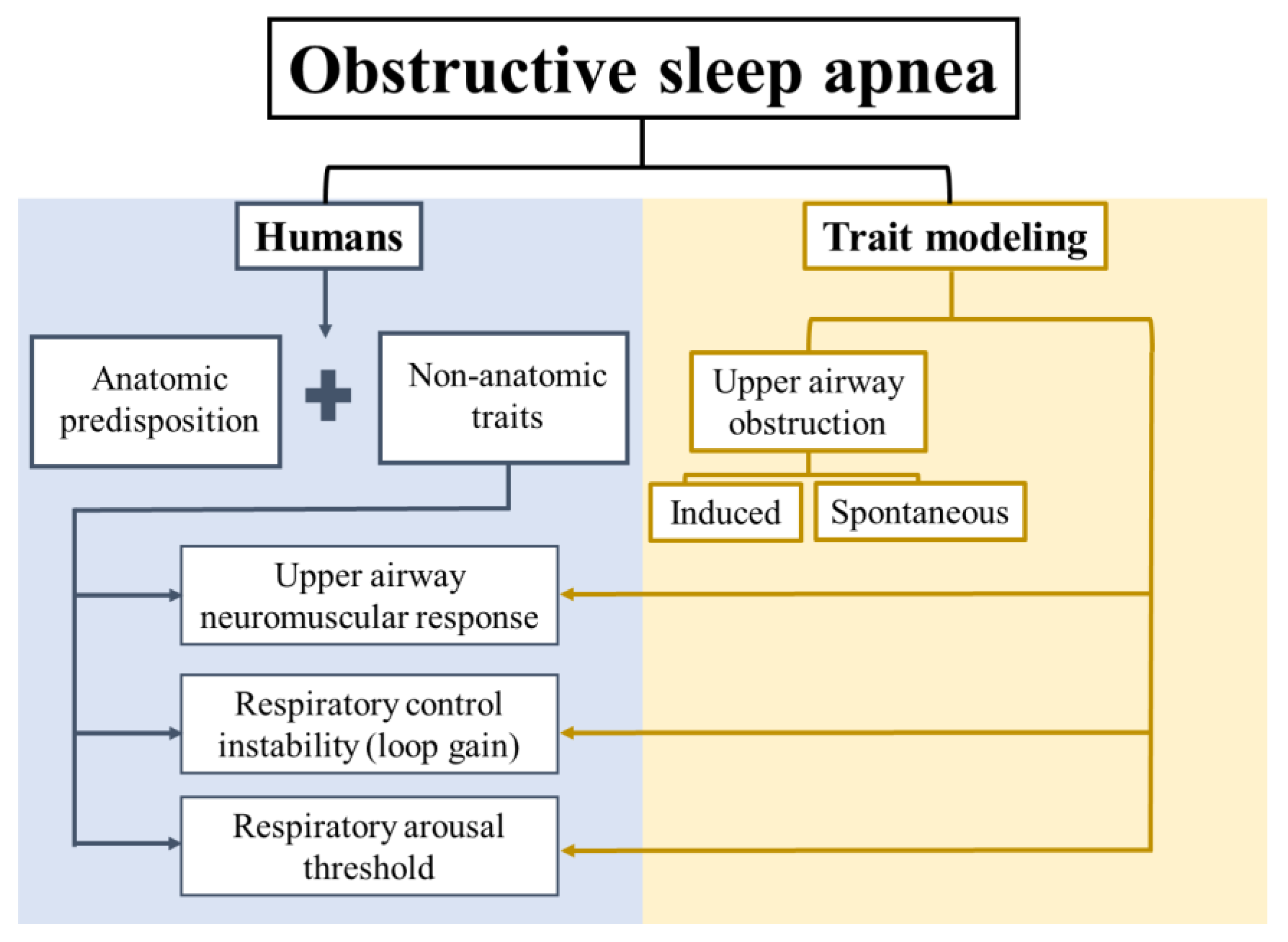
1. Introduction
2. Upper Airway Anatomy or Collapsibility
2.1. Definition and Human Evidence
2.2. Animal Models Focused on Upper Airway Anatomic Predisposition to OSA
3. Upper Airway Neuromuscular Response
3.1. Definition and Human Evidence
3.2. Animal Models of Neuromuscular Response: EMG Outcomes
3.3. Animal Models of Neuromuscular Response: Airflow Outcomes
4. Respiratory Control Instability (Loop Gain)
4.1. Definition and Human Evidence
4.2. Animal Models of Ventilatory Instability in OSA
5. Respiratory Arousal Threshold
5.1. Definition and Human Evidence
5.2. Animal Models Focusing on Arousal Threshold
6. Conclusions and Future Perspectives
Author Contributions
Funding
Conflicts of Interest
References
- Dempsey, J.A.; Veasey, S.C.; Morgan, B.J.; O’Donnell, C.P. Pathophysiology of Sleep Apnea. Physiol. Rev. 2010, 90, 47–112. [Google Scholar] [CrossRef] [PubMed]
- Tufik, S.; Santos-Silva, R.; Taddei, J.A.; Bittencourt, L.R.A. Obstructive Sleep Apnea Syndrome in the Sao Paulo Epidemiologic Sleep Study. Sleep Med. 2010, 11, 441–446. [Google Scholar] [CrossRef] [PubMed]
- Heinzer, R.; Vat, S.; Marques-Vidal, P.; Marti-Soler, H.; Andries, D.; Tobback, N.; Mooser, V.; Preisig, M.; Malhotra, A.; Waeber, G.; et al. Prevalence of sleep-disordered breathing in the general population: The HypnoLaus study. Lancet Respir. Med. 2015, 3, 310–318. [Google Scholar] [CrossRef]
- Sullivan, C.E.; Issa, F.G.; Berthon-Jones, M.; Eves, L. Reversal of obstructive sleep apnoea by continuous positive airway pressure applied through the nares. Lancet 1981, 1, 862–865. [Google Scholar] [CrossRef]
- Weaver, T.E.; Grunstein, R.R. Adherence to continuous positive airway pressure therapy: The challenge to effective treatment. Proc. Am. Thorac. Soc. 2008, 5, 173–178. [Google Scholar] [CrossRef] [PubMed]
- Kribbs, N.B.; Pack, A.I.; Kline, L.R.; Smith, P.L.; Schwartz, A.R.; Schubert, N.M.; Redline, S.; Henry, J.N.; Getsy, J.E.; Dinges, D.F. Objective measurement of patterns of nasal CPAP use by patients with obstructive sleep apnea. Am. Rev. Respir. Dis. 1993, 147, 887–895. [Google Scholar] [CrossRef]
- Owens, R.L.; Eckert, D.J.; Yeh, S.Y.; Malhotra, A. Upper airway function in the pathogenesis of obstructive sleep apnea: A review of the current literature. Curr. Opin. Pulm. Med. 2008, 14, 519–524. [Google Scholar] [CrossRef]
- Wellman, A.; Eckert, D.J.; Jordan, A.S.; Edwards, B.A.; Passaglia, C.L.; Jackson, A.C.; Gautam, S.; Owens, R.L.; Malhotra, A.; White, D.P. A method for measuring and modeling the physiological traits causing obstructive sleep apnea. J. Appl. Physiol. 2011, 110, 1627–1637. [Google Scholar] [CrossRef]
- Eckert, D.J.; White, D.P.; Jordan, A.S.; Malhotra, A.; Wellman, A. Defining phenotypic causes of obstructive sleep apnea. Identification of novel therapeutic targets. Am. J. Respir. Crit. Care Med. 2013, 188, 996–1004. [Google Scholar] [CrossRef]
- Wellman, A.; Edwards, B.A.; Sands, S.A.; Owens, R.L.; Nemati, S.; Butler, J.; Passaglia, C.L.; Jackson, A.C.; Malhotra, A.; White, D.P. A simplified method for determining phenotypic traits in patients with obstructive sleep apnea. J. Appl. Physiol. 2013, 114, 911–922. [Google Scholar] [CrossRef]
- Eckert, D.J. Phenotypic approaches to obstructive sleep apnoea–New pathways for targeted therapy. Sleep Med. Rev. 2018, 37, 45–59. [Google Scholar] [CrossRef] [PubMed]
- Neelapu, B.C.; Kharbanda, O.P.; Sardana, H.K.; Balachandran, R.; Sardana, V.; Kapoor, P.; Gupta, A.; Vasamsetti, S. Craniofacial and upper airway morphology in adult obstructive sleep apnea patients: A systematic review and meta-analysis of cephalometric studies. Sleep Med. Rev. 2017, 31, 79–90. [Google Scholar] [CrossRef] [PubMed]
- Schwab, R.J.; Pasirstein, M.; Pierson, R.; Mackley, A.; Hachadoorian, R.; Arens, R.; Maislin, G.; Pack, A.I. Identification of upper airway anatomic risk factors for obstructive sleep apnea with volumetric magnetic resonance imaging. Am. J. Respir. Crit. Care Med. 2003, 168, 522–530. [Google Scholar] [CrossRef] [PubMed]
- Lowe, A.A.; Santamaria, J.D.; Fleetham, J.A.; Price, C. Facial morphology and obstructive sleep apnea. Am. J. Orthod. Dentofac. Orthop. 1986, 90, 484–491. [Google Scholar] [CrossRef]
- Miles, P.G.; Vig, P.S.; Weyant, R.J.; Forrest, T.D.; Rockette, H.E. Craniofacial structure and obstructive sleep apnea syndrome–a qualitative analysis and meta-analysis of the literature. Am. J. Orthod. Dentofac. Orthop. 1996, 109, 163–172. [Google Scholar] [CrossRef]
- Schwartz, A.R.; O’Donnell, C.P.; Baron, J.; Schubert, N.; Alam, D.; Samadi, S.D.; Smith, P.L. The hypotonic upper airway in obstructive sleep apnea: Role of structures and neuromuscular activity. Am. J. Respir. Crit. Care Med. 1998, 157, 1051–1057. [Google Scholar] [CrossRef] [PubMed]
- Carberry, J.C.; Jordan, A.S.; White, D.P.; Wellman, A.; Eckert, D.J. Upper Airway Collapsibility (Pcrit) and Pharyngeal Dilator Muscle Activity are Sleep Stage Dependent. Sleep 2016, 39, 511–521. [Google Scholar] [CrossRef]
- Gold, A.R.; Schwartz, A.R. The pharyngeal critical pressure. The whys and hows of using nasal continuous positive airway pressure diagnostically. Chest 1996, 110, 1077–1088. [Google Scholar] [CrossRef]
- Kirkness, J.P.; Schwartz, A.R.; Schneider, H.; Punjabi, N.M.; Maly, J.J.; Laffan, A.M.; McGinley, B.M.; Magnuson, T.; Schweitzer, M.; Smith, P.L.; et al. Contribution of male sex, age, and obesity to mechanical instability of the upper airway during sleep. J. Appl. Physiol. 2008, 104, 1618–1624. [Google Scholar] [CrossRef]
- Schwartz, A.R.; Patil, S.P.; Laffan, A.M.; Polotsky, V.; Schneider, H.; Smith, P.L. Obesity and obstructive sleep apnea: Pathogenic mechanisms and therapeutic approaches. Proc. Am. Thorac. Soc. 2008, 5, 185–192. [Google Scholar] [CrossRef]
- Joosten, S.A.; Edwards, B.A.; Wellman, A.; Turton, A.; Skuza, E.M.; Berger, P.J.; Hamilton, G.S. The Effect of Body Position on Physiological Factors that Contribute to Obstructive Sleep Apnea. Sleep 2015, 38, 1469–1478. [Google Scholar] [CrossRef] [PubMed]
- Patil, S.P.; Schneider, H.; Marx, J.J.; Gladmon, E.; Schwartz, A.R.; Smith, P.L. Neuromechanical control of upper airway patency during sleep. J. Appl. Physiol. 2007, 102, 547–556. [Google Scholar] [CrossRef] [PubMed]
- Horner, R.L.; Mohiaddin, R.H.; Lowell, D.G.; Shea, S.A.; Burman, E.D.; Longmore, D.B.; Guz, A. Sites and sizes of fat deposits around the pharynx in obese patients with obstructive sleep apnoea and weight matched controls. Eur. Respir. J. 1989, 2, 613–622. [Google Scholar] [PubMed]
- Arens, R.; Sin, S.; Nandalike, K.; Rieder, J.; Khan, U.I.; Freeman, K.; Wylie-Rosett, J.; Lipton, M.L.; Wootton, D.M.; McDonough, J.M.; et al. Upper airway structure and body fat composition in obese children with obstructive sleep apnea syndrome. Am. J. Respir. Crit. Care Med. 2011, 183, 782–787. [Google Scholar] [CrossRef]
- Pahkala, R.; Seppä, J.; Ikonen, A.; Smirnov, G.; Tuomilehto, H. The impact of pharyngeal fat tissue on the pathogenesis of obstructive sleep apnea. Sleep Breath. 2014, 18, 275–282. [Google Scholar] [CrossRef]
- Kim, A.M.; Keenan, B.T.; Jackson, N.; Chan, E.L.; Staley, B.; Poptani, H.; Torigian, D.A.; Pack, A.I.; Schwab, R.J. Tongue fat and its relationship to obstructive sleep apnea. Sleep 2014, 37, 1639–1648. [Google Scholar] [CrossRef]
- Van de Graaff, W.B. Thoracic influence on upper airway patency. J. Appl. Physiol. 1988, 65, 2124–2131. [Google Scholar] [CrossRef]
- Heinzer, R.C.; Stanchina, M.L.; Malhotra, A.; Jordan, A.S.; Patel, S.R.; Lo, Y.-L.; Wellman, A.; Schory, K.; Dover, L.; White, D.P. Effect of increased lung volume on sleep disordered breathing in patients with sleep apnoea. Thorax 2006, 61, 435–439. [Google Scholar] [CrossRef]
- Kairaitis, K.; Byth, K.; Parikh, R.; Stavrinou, R.; Wheatley, J.R.; Amis, T.C. Tracheal traction effects on upper airway patency in rabbits: The role of tissue pressure. Sleep 2007, 30, 179–186. [Google Scholar] [CrossRef]
- Schwartz, A.R.; Gold, A.R.; Schubert, N.; Stryzak, A.; Wise, R.A.; Permutt, S.; Smith, P.L. Effect of weight loss on upper airway collapsibility in obstructive sleep apnea. Am. Rev. Respir. Dis. 1991, 144, 494–498. [Google Scholar] [CrossRef]
- White, D.P. Pharmacologic Approaches to the Treatment of Obstructive Sleep Apnea. Sleep Med. Clin. 2016, 11, 203–212. [Google Scholar] [CrossRef] [PubMed]
- Sutherland, K.; Lee, R.W.W.; Phillips, C.L.; Dungan, G.; Yee, B.J.; Magnussen, J.S.; Grunstein, R.R.; Cistulli, P.A. Effect of weight loss on upper airway size and facial fat in men with obstructive sleep apnoea. Thorax 2011, 66, 797–803. [Google Scholar] [CrossRef] [PubMed]
- Bucca, C.B.; Brussino, L.; Battisti, A.; Mutani, R.; Rolla, G.; Mangiardi, L.; Cicolin, A. Diuretics in obstructive sleep apnea with diastolic heart failure. Chest 2007, 132, 440–446. [Google Scholar] [CrossRef] [PubMed]
- Chopra, S.; Polotsky, V.Y.; Jun, J.C. Sleep Apnea Research in Animals. Past, Present, and Future. Am. J. Respir. Cell Mol. Biol. 2016, 54, 299–305. [Google Scholar] [CrossRef]
- Hendricks, J.C.; Kline, L.R.; Kovalski, R.J.; O’Brien, J.A.; Morrison, A.R.; Pack, A.I. The English bulldog: A natural model of sleep-disordered breathing. J. Appl. Physiol. 1987, 63, 1344–1350. [Google Scholar] [CrossRef]
- Lonergan, R.P.; Ware, J.C.; Atkinson, R.L.; Winter, W.C.; Suratt, P.M. Sleep apnea in obese miniature pigs. J. Appl. Physiol. 1998, 84, 531–536. [Google Scholar] [CrossRef]
- Farré, R.; Nácher, M.; Serrano-Mollar, A.; Gáldiz, J.B.; Alvarez, F.J.; Navajas, D.; Montserrat, J.M. Rat model of chronic recurrent airway obstructions to study the sleep apnea syndrome. Sleep 2007, 30, 930–933. [Google Scholar] [CrossRef]
- Neuzeret, P.-C.; Gormand, F.; Reix, P.; Parrot, S.; Sastre, J.-P.; Buda, C.; Guidon, G.; Sakai, K.; Lin, J.-S. A new animal model of obstructive sleep apnea responding to continuous positive airway pressure. Sleep 2011, 34, 541–548. [Google Scholar] [CrossRef]
- Crossland, R.F.; Durgan, D.J.; Lloyd, E.E.; Phillips, S.C.; Reddy, A.K.; Marrelli, S.P.; Bryan, R.M. A new rodent model for obstructive sleep apnea: Effects on ATP-mediated dilations in cerebral arteries. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2013, 305, R334–R342. [Google Scholar] [CrossRef]
- Lee, M.-C.; Lee, C.H.; Hong, S.-L.; Kim, S.-W.; Lee, W.-H.; Lim, J.Y.; Joe, S.; Yoon, I.-Y.; Kim, J.-W. Establishment of a rabbit model of obstructive sleep apnea by paralyzing the genioglossus. JAMA Otolaryngol. Head Neck Surg. 2013, 139, 834–840. [Google Scholar] [CrossRef][Green Version]
- Lu, H.; Dong, F.; Liu, C.; Wang, J.; Liu, Y.; Xiao, W. An animal model of obstructive sleep apnoea-hypopnea syndrome corrected by mandibular advancement device. Eur. J. Orthod. 2015, 37, 284–289. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.-C.; Rhee, C.-S.; Joe, S.; Yoon, I.-Y.; Kim, J.-W. A Single Primary Site Obstruction May Lead to Sleep-Disordered Breathing in Multiple Sites: An Animal Model. Ann. Otol. Rhinol. Laryngol. 2016, 125, 277–283. [Google Scholar] [CrossRef] [PubMed]
- Brennick, M.J.; Pickup, S.; Cater, J.R.; Kuna, S.T. Phasic respiratory pharyngeal mechanics by magnetic resonance imaging in lean and obese zucker rats. Am. J. Respir. Crit. Care Med. 2006, 173, 1031–1037. [Google Scholar] [CrossRef] [PubMed]
- Brennick, M.J.; Delikatny, J.; Pack, A.I.; Pickup, S.; Shinde, S.; Zhu, J.-X.; Roscoe, I.; Kim, D.Y.; Buxbaum, L.U.; Cater, J.R.; et al. Tongue fat infiltration in obese versus lean Zucker rats. Sleep 2014, 37, 1095–1102. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Brennick, M.J.; Pack, A.I.; Ko, K.; Kim, E.; Pickup, S.; Maislin, G.; Schwab, R.J. Altered upper airway and soft tissue structures in the New Zealand Obese mouse. Am. J. Respir. Crit. Care Med. 2009, 179, 158–169. [Google Scholar] [CrossRef]
- Baum, D.M.; Morales Rodriguez, B.; Attali, V.; Cauhapé, M.; Arnulf, I.; Cardot, P.; Bodineau, L.; Fiamma, M.-N. New Zealand Obese Mice as a Translational Model of Obesity-related Obstructive Sleep Apnea Syndrome. Am. J. Respir. Crit. Care Med. 2018, 198, 1336–1339. [Google Scholar] [CrossRef]
- Nishimura, Y.; Arias, R.S.; Pho, H.; Pham, L.V.; Curado, T.F.; Polotsky, V.Y.; Schwartz, A.R. A Novel Non-invasive Approach for Measuring Upper Airway Collapsibility in Mice. Front. Neurol. 2018, 9, 985. [Google Scholar] [CrossRef]
- Polotsky, M.; Elsayed-Ahmed, A.S.; Pichard, L.; Richardson, R.A.; Smith, P.L.; Schneider, H.; Kirkness, J.P.; Polotsky, V.; Schwartz, A.R. Effect of age and weight on upper airway function in a mouse model. J. Appl. Physiol. 2011, 111, 696–703. [Google Scholar] [CrossRef]
- Hernandez, A.B.; Kirkness, J.P.; Smith, P.L.; Schneider, H.; Polotsky, M.; Richardson, R.A.; Hernandez, W.C.; Schwartz, A.R. Novel whole body plethysmography system for the continuous characterization of sleep and breathing in a mouse. J. Appl. Physiol. 2012, 112, 671–680. [Google Scholar] [CrossRef]
- Pho, H.; Hernandez, A.B.; Arias, R.S.; Leitner, E.B.; Van Kooten, S.; Kirkness, J.P.; Schneider, H.; Smith, P.L.; Polotsky, V.Y.; Schwartz, A.R. The effect of leptin replacement on sleep-disordered breathing in the leptin-deficient ob/ob mouse. J. Appl. Physiol. 2016, 120, 78–86. [Google Scholar] [CrossRef]
- Fleury Curado, T.; Pho, H.; Berger, S.; Caballero-Eraso, C.; Shin, M.-K.; Sennes, L.U.; Pham, L.; Schwartz, A.R.; Polotsky, V.Y. Sleep-disordered breathing in C57BL/6J mice with diet-induced obesity. Sleep 2018, 41, zsy089. [Google Scholar] [CrossRef] [PubMed]
- O’donnell, C.P.; Schaub, C.D.; Haines, A.S.; Berkowitz, D.E.; Tankersley, C.G.; Schwartz, A.R.; Smith, P.L. Leptin prevents respiratory depression in obesity. Am. J. Respir. Crit. Care Med. 1999, 159, 1477–1484. [Google Scholar] [CrossRef] [PubMed]
- Davis, E.M.; O’Donnell, C.P. Rodent models of sleep apnea. Respir. Physiol. Neurobiol. 2013, 188, 355–361. [Google Scholar] [CrossRef] [PubMed]
- Zaidi, F.N.; Meadows, P.; Jacobowitz, O.; Davidson, T.M. Tongue anatomy and physiology, the scientific basis for a novel targeted neurostimulation system designed for the treatment of obstructive sleep apnea. Neuromodulation 2013, 16, 376–386. [Google Scholar] [CrossRef] [PubMed]
- Remmers, J.E.; deGroot, W.J.; Sauerland, E.K.; Anch, A.M. Pathogenesis of upper airway occlusion during sleep. J. Appl. Physiol. Respir. 1978, 44, 931–938. [Google Scholar] [CrossRef]
- Fuller, D.; Mateika, J.H.; Fregosi, R.F. Co-activation of tongue protrudor and retractor muscles during chemoreceptor stimulation in the rat. J. Physiol. (Lond.) 1998, 507, 265–276. [Google Scholar] [CrossRef]
- Dotan, Y.; Pillar, G.; Tov, N.; Oliven, R.; Steinfeld, U.; Gaitini, L.; Odeh, M.; Schwartz, A.R.; Oliven, A. Dissociation of electromyogram and mechanical response in sleep apnoea during propofol anaesthesia. Eur. Respir. J. 2013, 41, 74–84. [Google Scholar] [CrossRef]
- Fuller, D.D.; Williams, J.S.; Janssen, P.L.; Fregosi, R.F. Effect of co-activation of tongue protrudor and retractor muscles on tongue movements and pharyngeal airflow mechanics in the rat. J. Physiol. (Lond.) 1999, 51, 601–613. [Google Scholar] [CrossRef]
- Schwartz, A.R.; Bennett, M.L.; Smith, P.L.; De Backer, W.; Hedner, J.; Boudewyns, A.; Van de Heyning, P.; Ejnell, H.; Hochban, W.; Knaack, L.; et al. Therapeutic electrical stimulation of the hypoglossal nerve in obstructive sleep apnea. Arch. Otolaryngol. Head Neck Surg. 2001, 127, 1216–1223. [Google Scholar] [CrossRef]
- Kubin, L.; Davies, R.O.; Pack, A.I. Control of Upper Airway Motoneurons during REM Sleep. News Physiol. Sci. 1998, 13, 91–97. [Google Scholar] [CrossRef]
- Veasey, S.C. Serotonin agonists and antagonists in obstructive sleep apnea: Therapeutic potential. Am. J. Respir. Med. 2003, 2, 21–29. [Google Scholar] [CrossRef] [PubMed]
- Sood, S.; Liu, X.; Liu, H.; Horner, R.L. Genioglossus muscle activity and serotonergic modulation of hypoglossal motor output in obese Zucker rats. J. Appl. Physiol. 2007, 102, 2240–2250. [Google Scholar] [CrossRef] [PubMed]
- Chan, E.; Steenland, H.W.; Liu, H.; Horner, R.L. Endogenous excitatory drive modulating respiratory muscle activity across sleep-wake states. Am. J. Respir. Crit. Care Med. 2006, 174, 1264–1273. [Google Scholar] [CrossRef] [PubMed]
- Grace, K.P.; Hughes, S.W.; Horner, R.L. Identification of the mechanism mediating genioglossus muscle suppression in REM sleep. Am. J. Respir. Crit. Care Med. 2013, 187, 311–319. [Google Scholar] [CrossRef] [PubMed]
- Grace, K.P.; Hughes, S.W.; Shahabi, S.; Horner, R.L. K+ channel modulation causes genioglossus inhibition in REM sleep and is a strategy for reactivation. Respir. Physiol. Neurobiol. 2013, 188, 277–288. [Google Scholar] [CrossRef] [PubMed]
- Grace, K.P.; Hughes, S.W.; Horner, R.L. Identification of a Pharmacological Target for Genioglossus Reactivation throughout Sleep. Sleep 2014, 37, 41–50. [Google Scholar] [CrossRef]
- Song, G.; Poon, C.-S. α2-Adrenergic blockade rescues hypoglossal motor defense against obstructive sleep apnea. JCI Insight 2017, 2, e91456. [Google Scholar] [CrossRef]
- Fleury Curado, T.; Fishbein, K.; Pho, H.; Brennick, M.; Dergacheva, O.; Sennes, L.U.; Pham, L.V.; Ladenheim, E.E.; Spencer, R.; Mendelowitz, D.; et al. Chemogenetic stimulation of the hypoglossal neurons improves upper airway patency. Sci. Rep. 2017, 7, 44392. [Google Scholar] [CrossRef]
- Polotsky, M.; Elsayed-Ahmed, A.S.; Pichard, L.; Harris, C.C.; Smith, P.L.; Schneider, H.; Kirkness, J.P.; Polotsky, V.; Schwartz, A.R. Effects of leptin and obesity on the upper airway function. J. Appl. Physiol. 2012, 112, 1637–1643. [Google Scholar] [CrossRef]
- Yao, Q.; Pho, H.; Kirkness, J.; Ladenheim, E.E.; Bi, S.; Moran, T.H.; Fuller, D.D.; Schwartz, A.R.; Polotsky, V.Y. Localizing Effects of Leptin on Upper Airway and Respiratory Control during Sleep. Sleep 2016, 39, 1097–1106. [Google Scholar] [CrossRef]
- Berger, S.; Pho, H.; Fleury-Curado, T.; Bevans-Fonti, S.; Younas, H.; Shin, M.-K.; Jun, J.C.; Anokye-Danso, F.; Ahima, R.S.; Enquist, L.W.; et al. Intranasal Leptin Relieves Sleep-disordered Breathing in Mice with Diet-induced Obesity. Am. J. Respir. Crit. Care Med. 2019, 199, 773–783. [Google Scholar] [CrossRef] [PubMed]
- Peng, Y.-J.; Zhang, X.; Gridina, A.; Chupikova, I.; McCormick, D.L.; Thomas, R.J.; Scammell, T.E.; Kim, G.; Vasavda, C.; Nanduri, J.; et al. Complementary roles of gasotransmitters CO and H2S in sleep apnea. Proc. Natl. Acad. Sci. USA 2017, 114, 1413–1418. [Google Scholar] [CrossRef] [PubMed]
- Caballero-Eraso, C.; Shin, M.-K.; Pho, H.; Kim, L.J.; Pichard, L.E.; Wu, Z.-J.; Gu, C.; Berger, S.; Pham, L.; Yeung, H.-Y.B.; et al. Leptin acts in the carotid bodies to increase minute ventilation during wakefulness and sleep and augment the hypoxic ventilatory response. J. Physiol. (Lond.) 2019, 597, 151–172. [Google Scholar] [CrossRef] [PubMed]
- Yuan, F.; Wang, H.; Feng, J.; Wei, Z.; Yu, H.; Zhang, X.; Zhang, Y.; Wang, S. Leptin Signaling in the Carotid Body Regulates a Hypoxic Ventilatory Response Through Altering TASK Channel Expression. Front. Physiol. 2018, 9, 249. [Google Scholar] [CrossRef]
- Park, E.; Younes, M.; Liu, H.; Liu, X.; Horner, R.L. Systemic vs. central administration of common hypnotics reveals opposing effects on genioglossus muscle activity in rats. Sleep 2008, 31, 355–365. [Google Scholar] [CrossRef]
- Kaur, S.; Wang, J.L.; Ferrari, L.; Thankachan, S.; Kroeger, D.; Venner, A.; Lazarus, M.; Wellman, A.; Arrigoni, E.; Fuller, P.M.; et al. A Genetically Defined Circuit for Arousal from Sleep during Hypercapnia. Neuron 2017, 96, 1153–1167.e5. [Google Scholar] [CrossRef]
- Taranto-Montemurro, L.; Messineo, L.; Sands, S.A.; Azarbarzin, A.; Marques, M.; Edwards, B.A.; Eckert, D.J.; White, D.P.; Wellman, A. The Combination of Atomoxetine and Oxybutynin Greatly Reduces Obstructive Sleep Apnea Severity. A Randomized, Placebo-controlled, Double-Blind Crossover Trial. Am. J. Respir. Crit. Care Med. 2019, 199, 1267–1276. [Google Scholar] [CrossRef]
- Horton, G.A.; Fraigne, J.J.; Torontali, Z.A.; Snow, M.B.; Lapierre, J.L.; Liu, H.; Montandon, G.; Peever, J.H.; Horner, R.L. Activation of the Hypoglossal to Tongue Musculature Motor Pathway by Remote Control. Sci. Rep. 2017, 7, 45860. [Google Scholar] [CrossRef]
- Roth, B.L. DREADDs for Neuroscientists. Neuron 2016, 89, 683–694. [Google Scholar] [CrossRef]
- Miki, H.; Hida, W.; Shindoh, C.; Kikuchi, Y.; Chonan, T.; Taguchi, O.; Inoue, H.; Takishima, T. Effects of electrical stimulation of the genioglossus on upper airway resistance in anesthetized dogs. Am. Rev. Respir. Dis. 1989, 140, 1279–1284. [Google Scholar] [CrossRef]
- Schwartz, A.R.; Thut, D.C.; Russ, B.; Seelagy, M.; Yuan, X.; Brower, R.G.; Permutt, S.; Wise, R.A.; Smith, P.L. Effect of electrical stimulation of the hypoglossal nerve on airflow mechanics in the isolated upper airway. Am. Rev. Respir. Dis. 1993, 147, 1144–1150. [Google Scholar] [CrossRef] [PubMed]
- Oliven, A.; Odeh, M.; Schnall, R.P. Improved upper airway patency elicited by electrical stimulation of the hypoglossus nerves. Respiration 1996, 63, 213–216. [Google Scholar] [CrossRef] [PubMed]
- Eisele, D.W.; Schwartz, A.R.; Hari, A.; Thut, D.C.; Smith, P.L. The effects of selective nerve stimulation on upper airway airflow mechanics. Arch. Otolaryngol. Head Neck Surg. 1995, 121, 1361–1364. [Google Scholar] [CrossRef] [PubMed]
- Bishara, H.; Odeh, M.; Schnall, R.P.; Gavriely, N.; Oliven, A. Electrically-activated dilator muscles reduce pharyngeal resistance in anaesthetized dogs with upper airway obstruction. Eur. Respir. J. 1995, 8, 1537–1542. [Google Scholar] [PubMed]
- Goding, G.S.; Eisele, D.W.; Testerman, R.; Smith, P.L.; Roertgen, K.; Schwartz, A.R. Relief of upper airway obstruction with hypoglossal nerve stimulation in the canine. Laryngoscope 1998, 108, 162–169. [Google Scholar] [CrossRef] [PubMed]
- Yoo, P.B.; Durand, D.M. Effects of selective hypoglossal nerve stimulation on canine upper airway mechanics. J. Appl. Physiol. 2005, 99, 937–943. [Google Scholar] [CrossRef] [PubMed]
- Fleury Curado, T.A.; Pho, H.; Dergacheva, O.; Berger, S.; Lee, R.; Freire, C.; Asherov, A.; Sennes, L.U.; Mendelowitz, D.; Schwartz, A.R.; et al. Silencing of Hypoglossal Motoneurons Leads to Sleep Disordered Breathing in Lean Mice. Front. Neurol. 2018, 9, 962. [Google Scholar] [CrossRef]
- Marcus, C.L.; McColley, S.A.; Carroll, J.L.; Loughlin, G.M.; Smith, P.L.; Schwartz, A.R. Upper airway collapsibility in children with obstructive sleep apnea syndrome. J. Appl. Physiol. 1994, 77, 918–924. [Google Scholar] [CrossRef]
- Marcus, C.L. Sleep-disordered breathing in children. Am. J. Respir. Crit. Care Med. 2001, 164, 16–30. [Google Scholar] [CrossRef]
- Morris, D.L.; Rui, L. Recent advances in understanding leptin signaling and leptin resistance. Am. J. Physiol. Endocrinol. Metab. 2009, 297, E1247–E1259. [Google Scholar] [CrossRef]
- Friedman, J.M.; Halaas, J.L. Leptin and the regulation of body weight in mammals. Nature 1998, 395, 763–770. [Google Scholar] [CrossRef] [PubMed]
- Breslow, M.J.; Min-Lee, K.; Brown, D.R.; Chacko, V.P.; Palmer, D.; Berkowitz, D.E. Effect of leptin deficiency on metabolic rate in ob/ob mice. Am. J. Physiol. 1999, 276, E443–E449. [Google Scholar] [PubMed]
- Ip, M.S.; Lam, K.S.; Ho, C.; Tsang, K.W.; Lam, W. Serum leptin and vascular risk factors in obstructive sleep apnea. Chest 2000, 118, 580–586. [Google Scholar] [CrossRef] [PubMed]
- Phipps, P.R.; Starritt, E.; Caterson, I.; Grunstein, R.R. Association of serum leptin with hypoventilation in human obesity. Thorax 2002, 57, 75–76. [Google Scholar] [CrossRef] [PubMed]
- Maffei, M.; Halaas, J.; Ravussin, E.; Pratley, R.E.; Lee, G.H.; Zhang, Y.; Fei, H.; Kim, S.; Lallone, R.; Ranganathan, S. Leptin levels in human and rodent: Measurement of plasma leptin and ob RNA in obese and weight-reduced subjects. Nat. Med. 1995, 1, 1155–1161. [Google Scholar] [CrossRef] [PubMed]
- Considine, R.V.; Sinha, M.K.; Heiman, M.L.; Kriauciunas, A.; Stephens, T.W.; Nyce, M.R.; Ohannesian, J.P.; Marco, C.C.; McKee, L.J.; Bauer, T.L. Serum immunoreactive-leptin concentrations in normal-weight and obese humans. N. Engl. J. Med. 1996, 334, 292–295. [Google Scholar] [CrossRef]
- Scarpace, P.J.; Zhang, Y. Leptin resistance: A prediposing factor for diet-induced obesity. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2009, 296, R493–R500. [Google Scholar] [CrossRef]
- Wauman, J.; Tavernier, J. Leptin receptor signaling: Pathways to leptin resistance. Front. Biosci. (Landmark Ed.) 2011, 16, 2771–2793. [Google Scholar] [CrossRef]
- Deacon-Diaz, N.; Malhotra, A. Inherent vs. Induced Loop Gain Abnormalities in Obstructive Sleep Apnea. Front. Neurol. 2018, 9, 896. [Google Scholar] [CrossRef]
- Khoo, M.C. Determinants of ventilatory instability and variability. Respir. Physiol. 2000, 122, 167–182. [Google Scholar] [CrossRef]
- Younes, M.; Ostrowski, M.; Thompson, W.; Leslie, C.; Shewchuk, W. Chemical control stability in patients with obstructive sleep apnea. Am. J. Respir. Crit. Care Med. 2001, 163, 1181–1190. [Google Scholar] [CrossRef] [PubMed]
- Eckert, D.J.; Malhotra, A.; Jordan, A.S. Mechanisms of apnea. Prog. Cardiovasc. Dis. 2009, 51, 313–323. [Google Scholar] [CrossRef] [PubMed]
- Younes, M. Role of respiratory control mechanisms in the pathogenesis of obstructive sleep disorders. J. Appl. Physiol. 2008, 105, 1389–1405. [Google Scholar] [CrossRef] [PubMed]
- Deacon-Diaz, N.L.; Sands, S.A.; McEvoy, R.D.; Catcheside, P.G. Daytime loop gain is elevated in obstructive sleep apnea but not reduced by CPAP treatment. J. Appl. Physiol. 2018, 125, 1490–1497. [Google Scholar] [CrossRef] [PubMed]
- Narkiewicz, K.; van de Borne, P.J.; Pesek, C.A.; Dyken, M.E.; Montano, N.; Somers, V.K. Selective potentiation of peripheral chemoreflex sensitivity in obstructive sleep apnea. Circulation 1999, 99, 1183–1189. [Google Scholar] [CrossRef]
- Peng, Y.; Kline, D.D.; Dick, T.E.; Prabhakar, N.R. Chronic intermittent hypoxia enhances carotid body chemoreceptor response to low oxygen. Adv. Exp. Med. Biol. 2001, 499, 33–38. [Google Scholar]
- Deacon, N.L.; Catcheside, P.G. The role of high loop gain induced by intermittent hypoxia in the pathophysiology of obstructive sleep apnoea. Sleep Med. Rev. 2015, 22, 3–14. [Google Scholar] [CrossRef]
- Bach, K.B.; Mitchell, G.S. Hypoxia-induced long-term facilitation of respiratory activity is serotonin dependent. Respir. Physiol. 1996, 104, 251–260. [Google Scholar] [CrossRef]
- Mateika, J.H.; Mendello, C.; Obeid, D.; Badr, M.S. Peripheral chemoreflex responsiveness is increased at elevated levels of carbon dioxide after episodic hypoxia in awake humans. J. Appl. Physiol. 2004, 96, 1197–1205. [Google Scholar] [CrossRef]
- Pialoux, V.; Hanly, P.J.; Foster, G.E.; Brugniaux, J.V.; Beaudin, A.E.; Hartmann, S.E.; Pun, M.; Duggan, C.T.; Poulin, M.J. Effects of exposure to intermittent hypoxia on oxidative stress and acute hypoxic ventilatory response in humans. Am. J. Respir. Crit. Care Med. 2009, 180, 1002–1009. [Google Scholar] [CrossRef]
- Chowdhuri, S.; Shanidze, I.; Pierchala, L.; Belen, D.; Mateika, J.H.; Badr, M.S. Effect of episodic hypoxia on the susceptibility to hypocapnic central apnea during NREM sleep. J. Appl. Physiol. 2010, 108, 369–377. [Google Scholar] [CrossRef] [PubMed]
- Peng, Y.-J.; Overholt, J.L.; Kline, D.; Kumar, G.K.; Prabhakar, N.R. Induction of sensory long-term facilitation in the carotid body by intermittent hypoxia: Implications for recurrent apneas. Proc. Natl. Acad. Sci. USA 2003, 100, 10073–10078. [Google Scholar] [CrossRef] [PubMed]
- Kline, D.D.; Peng, Y.-J.; Manalo, D.J.; Semenza, G.L.; Prabhakar, N.R. Defective carotid body function and impaired ventilatory responses to chronic hypoxia in mice partially deficient for hypoxia-inducible factor 1 alpha. Proc. Natl. Acad. Sci. USA 2002, 99, 821–826. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, M.W.; Peskind, E.; Raskind, M.; Boyko, E.J.; Porte, D. Cerebrospinal fluid leptin levels: Relationship to plasma levels and to adiposity in humans. Nat. Med. 1996, 2, 589–593. [Google Scholar] [CrossRef]
- Berry, R.B.; Kouchi, K.; Bower, J.; Prosise, G.; Light, R.W. Triazolam in patients with obstructive sleep apnea. Am. J. Respir. Crit. Care Med. 1995, 151, 450–454. [Google Scholar] [CrossRef]
- Parrino, L.; Terzano, M.G. Polysomnographic effects of hypnotic drugs. A review. Psychopharmacology (Berl.) 1996, 126, 1–16. [Google Scholar] [CrossRef]
- Rosenberg, R.; Roach, J.M.; Scharf, M.; Amato, D.A. A pilot study evaluating acute use of eszopiclone in patients with mild to moderate obstructive sleep apnea syndrome. Sleep Med. 2007, 8, 464–470. [Google Scholar] [CrossRef]
- Heinzer, R.C.; White, D.P.; Jordan, A.S.; Lo, Y.L.; Dover, L.; Stevenson, K.; Malhotra, A. Trazodone increases arousal threshold in obstructive sleep apnoea. Eur. Respir. J. 2008, 31, 1308–1312. [Google Scholar] [CrossRef]
- Eckert, D.J.; Owens, R.L.; Kehlmann, G.B.; Wellman, A.; Rahangdale, S.; Yim-Yeh, S.; White, D.P.; Malhotra, A. Eszopiclone increases the respiratory arousal threshold and lowers the apnoea/hypopnoea index in obstructive sleep apnoea patients with a low arousal threshold. Clin. Sci. 2011, 120, 505–514. [Google Scholar] [CrossRef]
- Smales, E.T.; Edwards, B.A.; Deyoung, P.N.; McSharry, D.G.; Wellman, A.; Velasquez, A.; Owens, R.; Orr, J.E.; Malhotra, A. Trazodone Effects on Obstructive Sleep Apnea and Non-REM Arousal Threshold. Ann. Am. Thorac. Soc. 2015, 12, 758–764. [Google Scholar] [CrossRef]
- Carter, S.G.; Berger, M.S.; Carberry, J.C.; Bilston, L.E.; Butler, J.E.; Tong, B.K.Y.; Martins, R.T.; Fisher, L.P.; McKenzie, D.K.; Grunstein, R.R.; et al. Zopiclone Increases the Arousal Threshold without Impairing Genioglossus Activity in Obstructive Sleep Apnea. Sleep 2016, 39, 757–766. [Google Scholar] [CrossRef] [PubMed]
- Sands, S.A.; Terrill, P.I.; Edwards, B.A.; Taranto Montemurro, L.; Azarbarzin, A.; Marques, M.; de Melo, C.M.; Loring, S.H.; Butler, J.P.; White, D.P.; et al. Quantifying the Arousal Threshold Using Polysomnography in Obstructive Sleep Apnea. Sleep 2018, 41, zsx183. [Google Scholar] [CrossRef] [PubMed]
- Younes, M.; Park, E.; Horner, R.L. Pentobarbital sedation increases genioglossus respiratory activity in sleeping rats. Sleep 2007, 30, 478–488. [Google Scholar] [CrossRef] [PubMed]
- Kaur, S.; Pedersen, N.P.; Yokota, S.; Hur, E.E.; Fuller, P.M.; Lazarus, M.; Chamberlin, N.L.; Saper, C.B. Glutamatergic signaling from the parabrachial nucleus plays a critical role in hypercapnic arousal. J. Neurosci. 2013, 33, 7627–7640. [Google Scholar] [CrossRef]
- Yokota, S.; Kaur, S.; VanderHorst, V.G.; Saper, C.B.; Chamberlin, N.L. Respiratory-related outputs of glutamatergic, hypercapnia-responsive parabrachial neurons in mice. J. Comp. Neurol. 2015, 523, 907–920. [Google Scholar] [CrossRef]
- Kaur, S.; Saper, C.B. Neural Circuitry Underlying Waking Up to Hypercapnia. Front. Neurosci. 2019, 13, 401. [Google Scholar] [CrossRef]
| Phenotypic Features | Study | Animal Model | Pharmacotherapy Findings |
|---|---|---|---|
| Neuromuscular responses of upper airway | Chan et al. [63] | Wistar rats |
|
| Grace et al. [64,65,66] | Wistar rats |
| |
| Song and Poon [67] | Sprague-Dawley rats |
| |
| Fleury Curado et al. [68] | Lean C57BL/6J mice |
| |
| Polotsky et al. [69] | Leptin deficient ob/ob mice |
| |
| Pho et al. [50] | Leptin deficient ob/ob mice |
| |
| Yao et al. [70] | Leptin deficient ob/ob mice |
| |
| Berger et al. [71] | Diet-induced obesity (DIO) mice |
| |
| Respiratory instability (high loop gain) | Peng et al. [72] | HO-2-null mice and SHR rats |
|
| Caballero-Eraso et al. [73] | Lean C57BL/6J and LepRb deficient db/db mice |
| |
| Yuan et al. [74] | Zucker rats |
| |
| Respiratory arousal threshold | Park et al. [75] | Wistar rats |
|
| Kaur et al. [76] | CGRP-CreER mice |
|
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, L.J.; Freire, C.; Fleury Curado, T.; Jun, J.C.; Polotsky, V.Y. The Role of Animal Models in Developing Pharmacotherapy for Obstructive Sleep Apnea. J. Clin. Med. 2019, 8, 2049. https://doi.org/10.3390/jcm8122049
Kim LJ, Freire C, Fleury Curado T, Jun JC, Polotsky VY. The Role of Animal Models in Developing Pharmacotherapy for Obstructive Sleep Apnea. Journal of Clinical Medicine. 2019; 8(12):2049. https://doi.org/10.3390/jcm8122049
Chicago/Turabian StyleKim, Lenise Jihe, Carla Freire, Thomaz Fleury Curado, Jonathan C. Jun, and Vsevolod Y. Polotsky. 2019. "The Role of Animal Models in Developing Pharmacotherapy for Obstructive Sleep Apnea" Journal of Clinical Medicine 8, no. 12: 2049. https://doi.org/10.3390/jcm8122049
APA StyleKim, L. J., Freire, C., Fleury Curado, T., Jun, J. C., & Polotsky, V. Y. (2019). The Role of Animal Models in Developing Pharmacotherapy for Obstructive Sleep Apnea. Journal of Clinical Medicine, 8(12), 2049. https://doi.org/10.3390/jcm8122049





