Interstitial Lung Disease in Rheumatoid Arthritis Remains a Challenge for Clinicians
Abstract
1. Introduction
2. Epidemiology and Risk Factors
3. Pathogenesis
4. Symptoms and Clinical Features
4.1. Symptoms
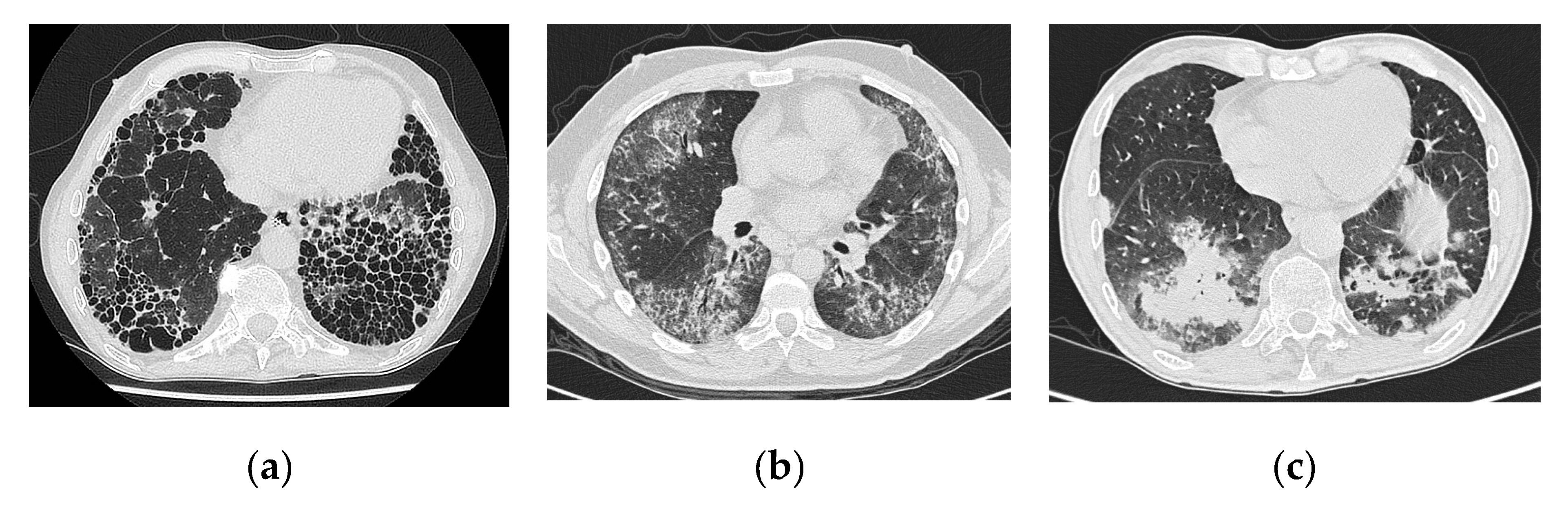
4.2. Imaging
4.3. Pulmonary Function
4.4. Bronchoscopy with Bronchoalveolar Lavage
4.5. Histopathology
5. Screening for RA-ILD
6. Differential Diagnosis
7. Treatment
8. Prognosis
9. Future Directions for RA-ILD
10. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Young, A.; Koduri, G.; Batley, M.; Kulinskaya, E.; Gough, A.; Norton, S.; Dixey, J. Mortality in rheumatoid arthritis. Increased in the early course of disease, in ischaemic heart disease and in pulmonary fibrosis. Rheumatology 2007, 46, 350–357. [Google Scholar] [CrossRef] [PubMed]
- Sparks, J.A.; Chang, S.C.; Liao, K.P.; Lu, B.; Fine, A.R.; Solomon, D.H.; Costenbader, K.H.; Karlson, E.W. Rheumatoid Arthritis and Mortality among Women during 36 Years of Prospective Follow-Up: Results from the Nurses’ Health Study. Arthritis Care Res. 2016, 68, 753–762. [Google Scholar] [CrossRef] [PubMed]
- Hyldgaard, C.; Hilberg, O.; Pedersen, A.B.; Ulrichsen, S.P.; Løkke, A.; Bendstrup, E.; Ellingsen, T. A population-based cohort study of rheumatoid arthritis-associated interstitial lung disease: Comorbidity and mortality. Ann. Rheum. Dis. 2017, 76, 1700–1706. [Google Scholar] [CrossRef] [PubMed]
- Hyldgaard, C.; Bendstrup, E.; Pedersen, A.B.; Ulrichsen, S.P.; Løkke, A.; Hilberg, O.; Ellingsen, T. Increased mortality among patients with rheumatoid arthritis and COPD: A population-based study. Respir. Med. 2018, 140, 101–107. [Google Scholar] [CrossRef]
- Restrepo, J.F.; del Rincón, I.; Battafarano, D.F.; Haas, R.W.; Doria, M.; Escalante, A. Clinical and laboratory factors associated with interstitial lung disease in rheumatoid arthritis. Clin. Rheumatol. 2015, 34, 1529–1536. [Google Scholar] [CrossRef]
- Olson, A.L.; Swigris, J.J.; Sprunger, D.B.; Fischer, A.; Fernandez-Perez, E.R.; Solomon, J.; Murphy, J.; Cohen, M.; Raghu, G.; Brown, K.K. Rheumatoid Arthritis–Interstitial Lung Disease–associated Mortality. Am. J. Respir. Crit. Care Med. 2010, 183, 372–378. [Google Scholar] [CrossRef]
- Raimundo, K.; Solomon, J.J.; Olson, A.L.; Kong, A.M.; Cole, A.L.; Fischer, A.; Swigris, J.J. Rheumatoid Arthritis-Interstitial Lung Disease in the United States: Prevalence, Incidence, and Healthcare Costs and Mortality. J. Rheumatol. 2019, 46, 360–369. [Google Scholar] [CrossRef]
- Juge, P.-A.; Lee, J.S.; Ebstein, E.; Furukawa, H.; Dobrinskikh, E.; Gazal, S.; Kannengiesser, C.; Ottaviani, S.; Oka, S.; Tohma, S.; et al. MUC5B Promoter Variant and Rheumatoid Arthritis with Interstitial Lung Disease. N. Engl. J. Med. 2018, 379, 2209–2219. [Google Scholar] [CrossRef]
- Turesson, C.; O’Fallon, W.M.; Crowson, C.S.; Gabriel, S.E.; Matteson, E.L. Extra-articular disease manifestations in rheumatoid arthritis: Incidence trends and risk factors over 46 years. Ann. Rheum. Dis. 2003, 62, 722–727. [Google Scholar] [CrossRef]
- Turesson, C.; Jacobsson, L.T.H. Epidemiology of extra-articular manifestations in rheumatoid arthritis. Scand. J. Rheumatol. 2004, 33, 65–72. [Google Scholar] [CrossRef]
- Bongartz, T.; Nannini, C.; Medina-Velasquez, Y.F.; Achenbach, S.J.; Crowson, C.S.; Ryu, J.H.; Vassallo, R.; Gabriel, S.E.; Matteson, E.L. Incidence and mortality of interstitial lung disease in rheumatoid arthritis: A population-based study. Arthritis Rheum. 2010, 62, 1583–1591. [Google Scholar] [CrossRef] [PubMed]
- Doyle, T.J.; Patel, A.S.; Hatabu, H.; Nishino, M.; Wu, G.; Osorio, J.C.; Golzarri, M.F.; Traslosheros, A.; Chu, S.G.; Frits, M.L.; et al. Detection of rheumatoid arthritis-interstitial lung disease is enhanced by serum biomarkers. Am. J. Respir. Crit. Care Med. 2015, 191, 1403–1412. [Google Scholar] [CrossRef] [PubMed]
- Svendsen, A.J.; Junker, P.; Houen, G.; Kyvik, K.O.; Nielsen, C.; Skytthe, A.; Holst, R. Incidence of Chronic Persistent Rheumatoid Arthritis and the Impact of Smoking: A Historical Twin Cohort Study. Arthritis Care Res. 2017, 69, 616–624. [Google Scholar] [CrossRef] [PubMed]
- Kelly, C.A.; Saravanan, V.; Nisar, M.; Arthanari, S.; Woodhead, F.A.; Price-Forbes, A.N.; Dawson, J.; Sathi, N.; Ahmad, Y.; Koduri, G.; et al. Rheumatoid arthritis-related interstitial lung disease: Associations, prognostic factors and physiological and radiological characteristics-a large multicentre UK study. Rheumatology 2014, 53, 1676–1682. [Google Scholar] [CrossRef]
- Gregersen, P.K.; Silver, J.; Winchester, R.J. The shared epitope hypothesis. An approach to understanding the molecular genetics of susceptibility to rheumatoid arthritis. Arthritis Rheum. 1987, 30, 1205–1213. [Google Scholar] [CrossRef]
- Charles, P.J.; Sweatman, M.C.; Markwick, J.R.; Maini, R.N. HLA-B40: A marker for susceptibility to lung disease in rheumatoid arthritis. Dis. Markers 1991, 9, 97–101. [Google Scholar]
- Scott, T.E.; Wise, R.A.; Hochberg, M.C.; Wigley, F.M. HLA-DR4 and pulmonary dysfunction in rheumatoid arthritis. Am. J. Med. 1987, 82, 765–771. [Google Scholar] [CrossRef]
- Sugiyama, Y.; Ohno, S.; Kano, S.; Maeda, H.; Kitamura, S. Diffuse Panbronchiolitis and Rheumatoid Arthritis: A Possible Correlation with HLA-B54. Intern. Med. 1994, 33, 612–614. [Google Scholar] [CrossRef]
- Demoruelle, M.K.; Deane, K.D.; Holers, V.M. When and where does inflammation begin in rheumatoid arthritis? Curr. Opin. Rheumatol. 2014, 26, 64–71. [Google Scholar] [CrossRef]
- Scally, S.W.; Petersen, J.; Law, S.C.; Dudek, N.L.; Nel, H.J.; Loh, K.L.; Wijeyewickrema, L.C.; Eckle, S.B.G.; van Heemst, J.; Pike, R.N.; et al. A molecular basis for the association of the HLA-DRB1 locus, citrullination, and rheumatoid arthritis. J. Exp. Med. 2013, 210, 2569–2582. [Google Scholar] [CrossRef]
- Makrygiannakis, D.; Hermansson, M.; Ulfgren, A.-K.; Nicholas, A.P.; Zendman, A.J.W.; Eklund, A.; Grunewald, J.; Skold, C.M.; Klareskog, L.; Catrina, A.I. Smoking increases peptidylarginine deiminase 2 enzyme expression in human lungs and increases citrullination in BAL cells. Ann. Rheum. Dis. 2008, 67, 1488–1492. [Google Scholar] [CrossRef] [PubMed]
- Meroni, R.; Piscitelli, D.; Bonetti, F.; Zambaldi, M.; Guccione, A.A.; Pillastrini, P. Rasch analysis of the Italian version of fear avoidance beliefs questionnaire (FABQ-I). Disabil. Rehabil. 2015, 37, 151–157. [Google Scholar] [CrossRef] [PubMed]
- Gochuico, B.R.; Avila, N.A.; Chow, C.K.; Novero, L.J.; Wu, H.-P.P.; Ren, P.; MacDonald, S.D.; Travis, W.D.; Stylianou, M.P.; Rosas, I.O. Progressive preclinical interstitial lung disease in rheumatoid arthritis. Arch. Intern. Med. 2008, 168, 159–166. [Google Scholar] [CrossRef] [PubMed]
- Paulin, F.; Babini, A.; Mamani, M.; Mercado, J.; Caro, F. Practical Approach to the Evaluation and Management of Rheumatoid Arthritis-Interstitial Lung Disease Based on its Proven and Hypothetical Mechanisms. Rev. Investig. Clin. 2017, 69, 235–242. [Google Scholar] [CrossRef]
- Paulin, F.; Doyle, T.J.; Fletcher, E.A.; Ascherman, D.P.; Rosas, I.O. Rheumatoid Arthritis-Associated Interstitial Lung Disease and Idiopathic Pulmonary Fibrosis: Shared Mechanistic and Phenotypic Traits Suggest Overlapping Disease Mechanisms. Rev. Investig. Clin. 2015, 67, 280–286. [Google Scholar]
- Juge, P.-A.; Borie, R.; Kannengiesser, C.; Gazal, S.; Revy, P.; Wemeau-Stervinou, L.; Debray, M.-P.; Ottaviani, S.; Marchand-Adam, S.; Nathan, N.; et al. Shared genetic predisposition in rheumatoid arthritis-interstitial lung disease and familial pulmonary fibrosis. Eur. Respir. J. 2017, 49, 1602314. [Google Scholar] [CrossRef]
- Ley, B.; Newton, C.A.; Arnould, I.; Elicker, B.M.; Henry, T.S.; Vittinghoff, E.; Golden, J.A.; Jones, K.D.; Batra, K.; Torrealba, J.; et al. The MUC5B promoter polymorphism and telomere length in patients with chronic hypersensitivity pneumonitis: An observational cohort-control study. Lancet Respir. Med. 2017, 5, 639–647. [Google Scholar] [CrossRef]
- Behr, J.; Kreuter, M.; Hoeper, M.M.; Wirtz, H.; Klotsche, J.; Koschel, D.; Andreas, S.; Claussen, M.; Grohé, C.; Wilkens, H.; et al. Management of patients with idiopathic pulmonary fibrosis in clinical practice: The INSIGHTS-IPF registry. Eur. Respir. J. 2015, 46, 186–196. [Google Scholar] [CrossRef]
- Dawson, J.K.; Fewins, H.E.; Desmond, J.; Lynch, M.P.; Graham, D.R. Fibrosing alveolitis in patients with rheumatoid arthritis as assessed by high resolution computed tomography, chest radiography, and pulmonary function tests. Thorax 2001, 56, 622–627. [Google Scholar] [CrossRef]
- Lee, H.-K.K.; Kim, D.S.; Yoo, B.; Seo, J.B.; Rho, J.-Y.Y.; Colby, T.V.; Kitaichi, M. Histopathologic pattern and clinical features of rheumatoid arthritis-associated interstitial lung disease. Chest 2005, 127, 2019–2027. [Google Scholar] [CrossRef]
- Skare, T.L.; Nakano, I.; Escuissiato, D.L.; Batistetti, R.; de Oliveira Rodrigues, T.; Silva, M.B. Pulmonary changes on high-resolution computed tomography of patients with rheumatoid arthritis and their association with clinical, demographic, serological and therapeutic variables. Rev. Bras. Reumatol. 2011, 51, 325–337. [Google Scholar] [PubMed]
- Nakamura, Y.; Suda, T.; Kaida, Y.; Kono, M.; Hozumi, H.; Hashimoto, D.; Enomoto, N.; Fujisawa, T.; Inui, N.; Imokawa, S.; et al. Rheumatoid lung disease: Prognostic analysis of 54 biopsy-proven cases. Respir. Med. 2012, 106, 1164–1169. [Google Scholar] [CrossRef] [PubMed]
- Bilgici, A.; Ulusoy, H.; Kuru, O.; Celenk, C.; Ünsal, M.; Danacı, M. Pulmonary involvement in rheumatoid arthritis. Rheumatol. Int. 2005, 25, 429–435. [Google Scholar] [CrossRef] [PubMed]
- Zrour, S.H.; Touzi, M.; Bejia, I.; Golli, M.; Rouatbi, N.; Sakly, N.; Younes, M.; Tabka, Z.; Bergaoui, N. Correlations between high-resolution computed tomography of the chest and clinical function in patients with rheumatoid arthritis. Jt. Bone Spine 2005, 72, 41–47. [Google Scholar] [CrossRef] [PubMed]
- Leonel, D.; Lucia, C.A.M.; Martha-Alicia, H.; Blanca, M. Pulmonary function test: Its correlation with pulmonary high-resolution computed tomography in patients with rheumatoid arthritis. Rheumatol. Int. 2012, 32, 2111–2116. [Google Scholar] [CrossRef]
- Youssef, A.A.; Machaly, S.A.; El-Dosoky, M.E.; El-Maghraby, N.M. Respiratory symptoms in rheumatoid arthritis: Relation to pulmonary abnormalities detected by high-resolution CT and pulmonary functional testing. Rheumatol. Int. 2012, 32, 1985–1995. [Google Scholar] [CrossRef]
- Kanat, F.; Levendoglu, F.; Teke, T. Radiological and functional assessment of pulmonary involvement in the rheumatoid arthritis patients. Rheumatol. Int. 2007, 27, 459–466. [Google Scholar] [CrossRef]
- Zhang, Y.; Li, H.; Wu, N.; Dong, X.; Zheng, Y. Retrospective study of the clinical characteristics and risk factors of rheumatoid arthritis-associated interstitial lung disease. Clin. Rheumatol. 2017, 36, 817–823. [Google Scholar] [CrossRef]
- Tanaka, N.; Kim, J.S.; Newell, J.D.; Brown, K.K.; Cool, C.D.; Meehan, R.; Emoto, T.; Matsumoto, T.; Lynch, D.A. Rheumatoid Arthritis–related Lung Diseases: CT Findings. Radiology 2004, 232, 81–91. [Google Scholar] [CrossRef]
- Zamora-Legoff, J.A.; Krause, M.L.; Crowson, C.S.; Ryu, J.H.; Matteson, E.L. Patterns of interstitial lung disease and mortality in rheumatoid arthritis. Rheumatology 2017, 56, 344–350. [Google Scholar] [CrossRef]
- Mori, S.; Cho, I.; Koga, Y.; Sugimoto, M. Comparison of pulmonary abnormalities on high-resolution computed tomography in patients with early versus longstanding rheumatoid arthritis. J. Rheumatol. 2008, 35, 1513–1521. [Google Scholar] [PubMed]
- Kim, E.J.; Elicker, B.M.; Maldonado, F.; Webb, W.R.; Ryu, J.H.; Van Uden, J.H.; Lee, J.S.; King, T.E.; Collard, H.R. Usual interstitial pneumonia in rheumatoid arthritis-associated interstitial lung disease. Eur. Respir. J. 2010, 35, 1322–1328. [Google Scholar] [CrossRef] [PubMed]
- Yunt, Z.X.; Chung, J.H.; Hobbs, S.; Fernandez-Perez, E.R.; Olson, A.L.; Huie, T.J.; Keith, R.C.; Janssen, W.J.; Goldstein, B.L.; Lynch, D.A.; et al. High resolution computed tomography pattern of usual interstitial pneumonia in rheumatoid arthritis-associated interstitial lung disease: Relationship to survival. Respir. Med. 2017, 126, 100–104. [Google Scholar] [CrossRef] [PubMed]
- Nurmi, H.M.; Kettunen, H.-P.; Suoranta, S.-K.; Purokivi, M.K.; Kärkkäinen, M.S.; Selander, T.A.; Kaarteenaho, R.L. Several high-resolution computed tomography findings associate with survival and clinical features in rheumatoid arthritis-associated interstitial lung disease. Respir. Med. 2018, 134, 24–30. [Google Scholar] [CrossRef]
- Tsuchiya, Y.; Takayanagi, N.; Sugiura, H.; Miyahara, Y.; Tokunaga, D.; Kawabata, Y.; Sugita, Y. Lung diseases directly associated with rheumatoid arthritis and their relationship to outcome. Eur. Respir. J. 2011, 37, 1411–1417. [Google Scholar] [CrossRef]
- Assayag, D.; Elicker, B.M.; Urbania, T.H.; Colby, T.V.; Kang, B.H.; Ryu, J.H.; King, T.E.; Collard, H.R.; Kim, D.S.; Lee, J.S. Rheumatoid Arthritis–associated Interstitial Lung Disease: Radiologic Identification of Usual Interstitial Pneumonia Pattern. Radiology 2014, 270, 583–588. [Google Scholar] [CrossRef]
- Morisset, J.; Vittinghoff, E.; Lee, B.Y.; Tonelli, R.; Hu, X.; Elicker, B.M.; Ryu, J.H.; Jones, K.D.; Cerri, S.; Manfredi, A.; et al. The performance of the GAP model in patients with rheumatoid arthritis associated interstitial lung disease. Respir. Med. J. 2017, 127, 51–56. [Google Scholar] [CrossRef]
- Kelly, C.; Palmer, E.; Gordon, J.; Woodhead, F.; Nisar, M.; Arthanari, S.; Forbes-Price, A.; Middleton, D.; Dempsey, O.; Dawson, J.; et al. OP0037 Pulsed Cyclophosphamide in the Treatment of Rheumatoid Arthritis-Related Interstitial Lung Disease (RA-ILD). Ann. Rheum. Dis. 2014, 73, 74. [Google Scholar] [CrossRef]
- Zhang, Y.; Lu, N.; Peloquin, C.; Dubreuil, M.; Neogi, T.; Aviña-Zubieta, J.A.; Rai, S.K.; Choi, H.K. Improved survival in rheumatoid arthritis: A general population-based cohort study. Ann. Rheum. Dis. 2017, 76, 408–413. [Google Scholar] [CrossRef]
- Dawson, J.K.; Fewins, H.E.; Desmond, J.; Lynch, M.P.; Graham, D.R. Predictors of progression of HRCT diagnosed fibrosing alveolitis in patients with rheumatoid arthritis. Ann. Rheum. Dis. 2002, 61, 517–521. [Google Scholar] [CrossRef]
- Chen, J.; Shi, Y.; Wang, X.; Huang, H.; Ascherman, D. Asymptomatic Preclinical Rheumatoid Arthritis-Associated Interstitial Lung Disease. Clin. Dev. Immunol. 2013, 2013, 10–15. [Google Scholar] [CrossRef] [PubMed]
- Cogliati, C.; Antivalle, M.; Torzillo, D.; Birocchi, S.; Norsa, A.; Bianco, R.; Costantino, G.; Ditto, M.C.; Battellino, M.; Sarzi Puttini, P.C.; et al. Standard and pocket-size lung ultrasound devices can detect interstitial lung disease in rheumatoid arthritis patients. Rheumatology 2014, 53, 1497–1503. [Google Scholar] [CrossRef] [PubMed]
- Moazedi-Fuerst, F.C.; Kielhauser, S.M.; Scheidl, S.; Tripolt, N.J.; Lutfi, A.; Yazdani-Biuki, B.; Dejaco, C.; Graninger, W.B. Ultrasound screening for interstitial lung disease in rheumatoid arthritis. Clin. Exp. Rheumatol. 2014, 32, 199–203. [Google Scholar] [PubMed]
- Wang, Y.; Gargani, L.; Barskova, T.; Furst, D.E.; Cerinic, M.M. Usefulness of lung ultrasound B-lines in connective tissue disease-associated interstitial lung disease: A literature review. Arthritis Res. Ther. 2017, 19, 206. [Google Scholar] [CrossRef] [PubMed]
- Avnon, L.S.; Manzur, F.; Bolotin, A.; Heimer, D.; Flusser, D.; Buskila, D.; Sukenik, S.; Abu-Shakra, M. Pulmonary functions testing in patients with rheumatoid arthritis. Isr. Med. Assoc. J. 2009, 11, 83–87. [Google Scholar]
- Pappas, D.A.; Giles, J.T.; Connors, G.; Lechtzin, N.; Bathon, J.M.; Danoff, S.K. Respiratory symptoms and disease characteristics as predictors of pulmonary function abnormalities in patients with rheumatoid arthritis: An observational cohort study. Arthritis Res. Ther. 2010, 12, R104. [Google Scholar] [CrossRef]
- Robles-Perez, A.; Luburich, P.; Rodriguez-Sanchon, B.; Dorca, J.; Nolla, J.M.; Molina-Molina, M.; Narvaez-Garcia, J. Preclinical lung disease in early rheumatoid arthritis. Chronic Respir. Dis. 2016, 13, 75–81. [Google Scholar] [CrossRef]
- Kolarz, G.; Scherak, O.; Popp, W.; Ritschka, L.; Thumb, N.; Wottawa, A.; Zwick, H. Bronchoalveolar lavage in rheumatoid arthritis. Br. J. Rheumatol. 1993, 32, 556–561. [Google Scholar] [CrossRef]
- Popp, W.; Ritschka, L.; Scherak, O.; Braun, O.; Kolarz, G.; Rauscher, H.; Zwick, H. Bronchoalveolar lavage in rheumatoid arthritis and secondary sjögren’s syndrome. Lung 1990, 168, 221–231. [Google Scholar] [CrossRef]
- Travis, W.D.; Costabel, U.; Hansell, D.M.; King, T.E.; Lynch, D.A.; Nicholson, A.G.; Ryerson, C.J.; Ryu, J.H.; Selman, M.; Wells, A.U.; et al. An Official American Thoracic Society/European Respiratory Society Statement: Update of the International Multidisciplinary Classification of the Idiopathic Interstitial Pneumonias. Am. J. Respir. Crit. Care Med. 2013, 188, 733–748. [Google Scholar] [CrossRef]
- Tansey, D.; Wells, A.U.; Colby, T.V.; Ip, S.; Nikolakoupolou, A.; Bois, R.M.; Hansell, D.M.; Nicholson, A.G. Variations in histological patterns of interstitial pneumonia.pdf. Histopathology 2004, 44, 585–596. [Google Scholar] [CrossRef] [PubMed]
- Yoshinouchi, T.; Ohtsuki, Y.; Fujita, J.; Yamadori, I.; Bandoh, S.; Ishida, T.; Ueda, R. Nonspecific interstitial pneumonia pattern as pulmonary involvement of rheumatoid arthritis. Rheumatol. Int. 2005, 26, 121–125. [Google Scholar] [CrossRef] [PubMed]
- Yousem, S.A.; Colby, T.V.; Carrington, C.B. Lung biopsy in rheumatoid arthritis. Am. Rev. Respir. Dis. 1985, 131, 770–777. [Google Scholar]
- Lin, E.; Limper, A.H.; Moua, T. Obliterative bronchiolitis associated with rheumatoid arthritis: Analysis of a single-center case series. BMC Pulm. Med. 2018, 18, 105. [Google Scholar] [CrossRef] [PubMed]
- Kim, E.J.; Collard, H.R.; King, T.E. Rheumatoid arthritis-associated interstitial lung disease: The relevance of histopathologic and radiographic pattern. Chest 2009, 136, 1397–1405. [Google Scholar] [CrossRef] [PubMed]
- Iftikhar, I.H.; Alghothani, L.; Sardi, A.; Berkowitz, D.; Musani, A.I. Transbronchial Lung Cryobiopsy and Video-assisted Thoracoscopic Lung Biopsy in the Diagnosis of Diffuse Parenchymal Lung Disease. A Meta-analysis of Diagnostic Test Accuracy. Ann. Am. Thorac. Soc. 2017, 14, 1197–1211. [Google Scholar] [CrossRef]
- Kono, M.; Nakamura, Y.; Yoshimura, K.; Enomoto, Y.; Oyama, Y.; Hozumi, H.; Enomoto, N.; Fujisawa, T.; Inui, N.; Hamada, E.; et al. Nonspecific interstitial pneumonia preceding diagnosis of collagen vascular disease. Respir. Med. 2016, 117, 40–47. [Google Scholar] [CrossRef]
- Kalinova, D.; Kolarov, Z.; Rashkov, R. Organising pneumonia—The first manifestation of rheumatoid arthritis. Reumatologia 2017, 55, 314–317. [Google Scholar] [CrossRef]
- Roubille, C.; Haraoui, B. Interstitial lung diseases induced or exacerbated by DMARDS and biologic agents in rheumatoid arthritis: A systematic literature review. Semin. Arthritis Rheum. 2014, 43, 613–626. [Google Scholar] [CrossRef]
- Conway, R.; Low, C.; Coughlan, R.J.; O’Donnell, M.J.; Carey, J.J. Methotrexate and lung disease in rheumatoid arthritis: A meta-analysis of randomized controlled trials. Arthritis Rheumatol. 2014, 66, 803–812. [Google Scholar] [CrossRef]
- Salliot, C.; Van Der Heijde, D. Long-term safety of methotrexate monotherapy in patients with rheumatoid arthritis: A systematic literature research. Ann. Rheum. Dis. 2009, 68, 1100–1104. [Google Scholar] [CrossRef]
- Rojas-Serrano, J.; Herrera-Bringas, D.; Pérez-Román, D.I.; Pérez-Dorame, R.; Mateos-Toledo, H.; Mejía, M. Rheumatoid arthritis-related interstitial lung disease (RA-ILD): Methotrexate and the severity of lung disease are associated to prognosis. Clin. Rheumatol. 2017, 36, 1493–1500. [Google Scholar] [CrossRef]
- Perez-Alvarez, R.; Perez-de-Lis, M.; Diaz-Lagares, C.; Pego-Reigosa, J.M.; Retamozo, S.; Bove, A.; Brito-Zeron, P.; Bosch, X.; Ramos-Casals, M. Interstitial Lung Disease Induced or Exacerbated by TNF-Targeted Therapies: Analysis of 122 Cases. Semin. Arthritis Rheum. 2011, 41, 256–264. [Google Scholar] [CrossRef]
- Lahiri, M.; Dixon, W.G. Risk of infection with biologic antirheumatic therapies in patients with rheumatoid arthritis. Best Pract. Res. Clin. Rheumatol. 2015, 29, 290–305. [Google Scholar] [CrossRef]
- Singh, J.A.; Cameron, C.; Noorbaloochi, S.; Cullis, T.; Tucker, M.; Christensen, R.; Ghogomu, E.T.; Coyle, D.; Clifford, T.; Tugwell, P.; et al. Risk of serious infection in biological treatment of patients with rheumatoid arthritis: A systematic review and meta-analysis. Lancet 2015, 386, 258–265. [Google Scholar] [CrossRef]
- Zamora-Legoff, J.A.; Krause, M.L.; Crowson, C.S.; Ryu, J.H.; Matteson, E.L. Risk of serious infection in patients with rheumatoid arthritis-associated interstitial lung disease. Clin. Rheumatol. 2016, 35, 2585–2589. [Google Scholar] [CrossRef]
- Hozumi, H.; Nakamura, Y.; Johkoh, T.; Sumikawa, H.; Colby, T.V.; Kono, M.; Hashimoto, D.; Enomoto, N.; Fujisawa, T.; Inui, N.; et al. Acute exacerbation in rheumatoid arthritis-associated interstitial lung disease: A retrospective case control study. BMJ Open 2013, 3, e003132. [Google Scholar] [CrossRef]
- Hamblin, M.J.; Horton, M.R. Rheumatoid Arthritis-Associated Interstitial Lung Disease: Diagnostic Dilemma. Pulm. Med. 2011, 2011. [Google Scholar] [CrossRef]
- Løgstrup, B.B.; Ellingsen, T.; Pedersen, A.B.; Kjærsgaard, A.; Bøtker, H.E.; Maeng, M. Heart Failure and Ischemic Heart Disease in Patients with Rheumatoid Arthritis. J. Am. Coll. Cardiol. 2017, 70, 3069–3071. [Google Scholar] [CrossRef]
- Simon, T.A.; Thompson, A.; Gandhi, K.K.; Hochberg, M.C.; Suissa, S. Incidence of malignancy in adult patients with rheumatoid arthritis: A meta-analysis. Arthritis Res. Ther. 2015, 17, 212. [Google Scholar] [CrossRef]
- Hazlewood, G.S.; Barnabe, C.; Tomlinson, G.; Marshall, D.; Devoe, D.J.A.; Bombardier, C. Methotrexate monotherapy and methotrexate combination therapy with traditional and biologic disease modifying anti-rheumatic drugs for rheumatoid arthritis: A network meta-analysis. Cochrane Database Syst. Rev. 2016, 2016, 1–14. [Google Scholar] [CrossRef]
- Song, J.W.; Lee, H.-K.; Lee, C.K.; Chae, E.J.; Jang, S.J.; Colby, T.V.; Kim, D.S. Clinical course and outcome of rheumatoid arthritis-related usual interstitial pneumonia. Sarcoidosis Vasc. Diffus. lung Dis. Off. J. WASOG 2013, 30, 103–112. [Google Scholar]
- Wijsenbeek, M.; Kreuter, M.; Olson, A.; Fischer, A.; Bendstrup, E.; Wells, C.D.; Denton, C.P.; Mounir, B.; Zouad-Lejour, L.; Quaresma, M.; et al. Progressive fibrosing interstitial lung diseases: Current practice in diagnosis and management. Curr. Med. Res. Opin. 2019, 35, 2015–2024. [Google Scholar] [CrossRef]
- Wijsenbeek, M.; Bendstrup, E.; Valenzuela, C.; Henry, M.T.; Moor, C.; Bengus, M.; Perjesi, A.; Gilberg, F.; Kirchgaessler, K.-U.; Vancheri, C. Design of a Study Assessing Disease Behaviour During the Peri-Diagnostic Period in Patients with Interstitial Lung Disease: The STARLINER Study. Adv. Ther. 2019, 36, 232–243. [Google Scholar] [CrossRef]
- O’Dwyer, D.N.; Armstrong, M.E.; Cooke, G.; Dodd, J.D.; Veale, D.J.; Donnelly, S.C. Rheumatoid Arthritis (RA) associated interstitial lung disease (ILD). Eur. J. Intern. Med. 2013, 24, 597–603. [Google Scholar] [CrossRef]
- Fischer, A.; Brown, K.K.; Du Bois, R.M.; Frankel, S.K.; Cosgrove, G.P.; Fernandez-Perez, E.R.; Huie, T.J.; Krishnamoorthy, M.; Meehan, R.T.; Olson, A.L.; et al. Mycophenolate mofetil improves lung function in connective tissue disease-associated interstitial lung disease. J. Rheumatol. 2013, 40, 640–646. [Google Scholar] [CrossRef]
- Swigris, J.J.; Olson, A.L.; Fischer, A.; Lynch, D.A.; Cosgrove, G.P.; Frankel, S.K.; Meehan, R.T.; Brown, K.K. Mycophenolate mofetil is safe, well tolerated, and preserves lung function in patients with connective tissue disease-related interstitial lung disease. Chest 2006, 130, 30–36. [Google Scholar] [CrossRef]
- Tzouvelekis, A.; Galanopoulos, N.; Bouros, E.; Kolios, G.; Zacharis, G.; Ntolios, P.; Koulelidis, A.; Oikonomou, A.; Bouros, D. Effect and safety of mycophenolate mofetil or sodium in systemic sclerosis-associated interstitial lung disease: A meta-analysis. Pulm. Med. 2012, 2012. [Google Scholar] [CrossRef]
- Saketkoo, L.A.; Espinoza, L.R. Experience of mycophenolate mofetil in 10 patients with autoimmune-related interstitial lung disease demonstrates promising effects. Am. J. Med. Sci. 2009, 337, 329–335. [Google Scholar] [CrossRef]
- Oldham, J.M.; Lee, C.; Valenzi, E.; Witt, L.J.; Adegunsoye, A.; Hsu, S.; Chen, L.; Montner, S.; Chung, J.H.; Noth, I.; et al. Azathioprine response in patients with fibrotic connective tissue disease-associated interstitial lung disease. Respir. Med. 2016, 121, 117–122. [Google Scholar] [CrossRef]
- Rojas-Serrano, J.; González-Velásquez, E.; Mejía, M.; Sánchez-Rodríguez, A.; Carrillo, G. Interstitial lung disease related to rheumatoid arthritis: Evolution after treatment. Reumatol. Clin. 2012, 8, 68–71. [Google Scholar] [CrossRef]
- Ota, M.; Iwasaki, Y.; Harada, H.; Sasaki, O.; Nagafuchi, Y.; Nakachi, S.; Sumitomo, S.; Shoda, H.; Tohma, S.; Fujio, K.; et al. Efficacy of intensive immunosuppression in exacerbated rheumatoid arthritis-associated interstitial lung disease. Mod. Rheumatol. 2017, 27, 22–28. [Google Scholar] [CrossRef]
- Md Yusof, M.Y.; Kabia, A.; Darby, M.; Lettieri, G.; Beirne, P.; Vital, E.M.; Dass, S.; Emery, P. Effect of rituximab on the progression of rheumatoid arthritis-related interstitial lung disease: 10 years’ experience at a single centre. Rheumatology 2017, 56, 1348–1357. [Google Scholar] [CrossRef]
- Matteson, E.L.; Bongartz, T.; Ryu, J.H.; Crowson, C.S.; Hartman, T.E.; Dellaripa, P.F. Open-Label, Pilot Study of the Safety and Clinical Effects of Rituximab in Patients with Rheumatoid Arthritis-Associated Interstitial Pneumonia. Open J. Rheumatol. Autoimmun. Dis. 2012, 2, 53–58. [Google Scholar] [CrossRef]
- Keir, G.J.; Maher, T.M.; Ming, D.; Abdullah, R.; De Lauretis, A.; Wickremasinghe, M.; Nicholson, A.G.; Hansell, D.M.; Wells, A.U.; Renzoni, E.A. Rituximab in severe, treatment-refractory interstitial lung disease. Respirology 2014, 19, 353–359. [Google Scholar] [CrossRef]
- Antoniou, K.M.; Mamoulaki, M.; Malagari, K.; Kritikos, H.D.; Bouros, D.; Siafakas, N.M.; Boumpas, D.T. Infliximab therapy in pulmonary fibrosis associated with collagen vascular disease. Clin. Exp. Rheumatol. 2007, 25, 23–28. [Google Scholar]
- Akiyama, M.; Kaneko, Y.; Yamaoka, K.; Kondo, H.; Takeuchi, T. Association of disease activity with acute exacerbation of interstitial lung disease during tocilizumab treatment in patients with rheumatoid arthritis: A retrospective, case–control study. Rheumatol. Int. 2016, 36, 881–889. [Google Scholar] [CrossRef]
- Dowman, L.M.; McDonald, C.F.; Hill, C.J.; Lee, A.L.; Barker, K.; Boote, C.; Glaspole, I.; Goh, N.S.L.; Southcott, A.M.; Burge, A.T.; et al. The evidence of benefits of exercise training in interstitial lung disease: A randomised controlled trial. Thorax 2017, 72, 610–619. [Google Scholar] [CrossRef]
- Weill, D.; Benden, C.; Corris, P.A.; Dark, J.H.; Davis, R.D.; Keshavjee, S.; Lederer, D.J.; Mulligan, M.J.; Patterson, G.A.; Singer, L.G.; et al. A consensus document for the selection of lung transplant candidates: 2014—An update from the Pulmonary Transplantation Council of the International Society for Heart and Lung Transplantation. J. Heart Lung Transplant. 2015, 34, 1–15. [Google Scholar] [CrossRef]
- Yazdani, A.; Singer, L.G.; Strand, V.; Gelber, A.C.; Williams, L.; Mittoo, S. Survival and quality of life in rheumatoid arthritis-associated interstitial lung disease after lung transplantation. J. Heart Lung Transplant. 2014, 33, 514–520. [Google Scholar] [CrossRef]
- Courtwright, A.M.; El-Chemaly, S.; Dellaripa, P.F.; Goldberg, H.J. Survival and outcomes after lung transplantation for non-scleroderma connective tissue–related interstitial lung disease. J. Heart Lung Transplant. 2017, 36, 763–769. [Google Scholar] [CrossRef]
- Flaherty, K.R.; Wells, A.U.; Cottin, V.; Devaraj, A.; Walsh, S.L.F.; Inoue, Y.; Richeldi, L.; Kolb, M.; Tetzlaff, K.; Stowasser, S.; et al. Nintedanib in Progressive Fibrosing Interstitial Lung Diseases. N. Engl. J. Med. 2019, 381, 1718–1727. [Google Scholar] [CrossRef]
- Behr, J.; Neuser, P.; Prasse, A.; Kreuter, M.; Rabe, K.; Schade-Brittinger, C.; Wagner, J.; Günther, A. Exploring efficacy and safety of oral Pirfenidone for progressive, non-IPF lung fibrosis (RELIEF)—A randomized, double-blind, placebo-controlled, parallel group, multi-center, phase II trial. BMC Pulm. Med. 2017, 17, 122. [Google Scholar] [CrossRef]
- Phase ll Study of Pirfenidone in Patients with RAILD (TRAIL1)—ClinicalTrials.Gov Study ID NCT02808871. Available online: https://clinicaltrials.gov/ct2/show/NCT02808871 (accessed on 15 August 2019).
- Lacaille, D.; Avina-Zubieta, J.A.; Sayre, E.C.; Abrahamowicz, M. Improvement in 5-year mortality in incident rheumatoid arthritis compared with the general population-closing the mortality gap. Ann. Rheum. Dis. 2017, 76, 1057–1063. [Google Scholar] [CrossRef]
- Koduri, G.; Norton, S.; Young, A.; Cox, N.; Davies, P.; Devlin, J.; Dixey, J.; Gough, A.; Prouse, P.; Winfield, J.; et al. Interstitial lung disease has a poor prognosis in rheumatoid arthritis: Results from an inception cohort. Rheumatology 2010, 49, 1483–1489. [Google Scholar] [CrossRef]
- Solomon, J.J.; Chung, J.H.; Cosgrove, G.P.; Demoruelle, M.K.; Fernandez-Perez, E.R.; Fischer, A.; Frankel, S.K.; Hobbs, S.B.; Huie, T.J.; Ketzer, J.; et al. Predictors of mortality in rheumatoid arthritis-associated interstitial lung disease. Eur. Respir. J. 2016, 47, 588–596. [Google Scholar] [CrossRef]
- Solomon, J.J.; Ryu, J.H.; Tazelaar, H.D.; Myers, J.L.; Tuder, R.; Cool, C.D.; Curran-Everett, D.; Fischer, A.; Swigris, J.J.; Brown, K.K. Fibrosing interstitial pneumonia predicts survival in patients with rheumatoid arthritis-associated interstitial lung disease (RA-ILD). Respir. Med. 2013, 107, 1247–1252. [Google Scholar] [CrossRef]
- Nurmi, H.M.; Purokivi, M.K.; Kärkkäinen, M.S.; Kettunen, H.P.; Selander, T.A.; Kaarteenaho, R.L. Variable course of disease of rheumatoid arthritis-associated usual interstitial pneumonia compared to other subtypes. BMC Pulm. Med. 2016, 16, 107. [Google Scholar] [CrossRef]
- Nurmi, H.M.; Purokivi, M.K.; Kärkkäinen, M.S.; Kettunen, H.-P.; Selander, T.A.; Kaarteenaho, R.L. Are risk predicting models useful for estimating survival of patients with rheumatoid arthritis-associated interstitial lung disease? BMC Pulm. Med. 2017, 17, 16. [Google Scholar] [CrossRef]
- Sparks, J.A.; He, X.; Huang, J.; Fletcher, E.A.; Zaccardelli, A.; Friedlander, H.M.; Gill, R.R.; Hatabu, H.; Nishino, M.; Murphy, D.J.; et al. Rheumatoid arthritis disease activity predicting incident clinically-apparent RA-associated interstitial lung disease: A prospective cohort study. Arthritis Rheumatol. 2019. [Google Scholar] [CrossRef]
- Jacob, J.; Hirani, N.; Van Moorsel, C.H.M.; Rajagopalan, S.; Murchison, J.T.; Van Es, H.W.; Bartholmai, B.J.; Van Beek, F.T.; Struik, M.H.L.; Stewart, G.A.; et al. Predicting outcomes in rheumatoid arthritis related interstitial lung disease. Eur. Respir. J. 2019, 53. [Google Scholar] [CrossRef] [PubMed]
- Goh, N.S.L.; Desai, S.R.; Veeraraghavan, S.; Hansell, D.M.; Copley, S.J.; Maher, T.M.; Corte, T.J.; Sander, C.R.; Ratoff, J.; Devaraj, A.; et al. Interstitial lung disease in systemic sclerosis: A simple staging system. Am. J. Respir. Crit. Care Med. 2008, 177, 1248–1254. [Google Scholar] [CrossRef] [PubMed]
| Studies | Total Number of Subjects with ILD (n = 1138) | UIP (n) | Non-UIP (n) | Subtypes of Non-UIP (n) | HRCT Not Performed | ||||
|---|---|---|---|---|---|---|---|---|---|
| NSIP (n) | Bronchiolitis (n) | OP (n) | DAD (n) | Other (n) | |||||
| Tanaka et al. Radiology 2004 [39] | 63 | 26 | 37 | 19 | 11 | 5 | - | 2 | - |
| Mori et al. Journal of Rheumatology 2008 [41] | 25 | 2 | 23 | 11 | 10 | 2 | - | - | |
| Kim et al. European Respiratory Journal 2010 [42] | 84 | 20 | 64 | 19 | - | - | - | 45 | - |
| Tsuchiya et al. European Respiratory Journal 2011 [45] | 102 | 57 | 26 | 16 | - | 5 | 5 | - | 19 |
| Kelly et al. Annals of Rheumatology Dis 2014 [48] | 231 | 150 | 81 | 55 | - | 12 | - | 14 | - |
| Assayag et al. Radiology 2014 [46] | 69 | 38 * | 31 ** | - | - | - | - | - | - |
| Yunt et al. Respiratory Medicine 2017 [43] | 195 | 123 | 72 | 35 | 28 | 4 | - | 5 | - |
| Zhang et al. Annals of Rheumatic Diseases 2017 [49] | 237 | 44 | 193 | 137 | - | - | - | 56 | - |
| Juge et al. New England Journal of Medicine 2018 [8] | 620 | 207 * | 298 ** | - | - | - | - | - | - |
| Zamora-Legoff et al. Rheumatology 2017 [40] | 181 | 98 | 77 | 73 | - | 4 | - | - | - |
| Morisset et al. Respiratory Medicine 2017 [47] | 309 | 125 * | 184 ** | - | - | - | - | - | - |
| Nurmi et al. Respiratory Medicine 2018 [44] | 60 | 36 | 24 | 8 | - | 7 | 1 | 8 | - |
| % of total | 100% | 43% | 51% | 17% | 2% | 2% | 0% | 6% | 1% |
| Number | UIP | NSIP | Bronchiolitis | OP | DAD | Unclassifiable | |
|---|---|---|---|---|---|---|---|
| Tansey et al. Histopathology 2004 [61] | 16 | 2 | 7 | 7 | - | - | - |
| Lee et al. Chest 2005 [30] | 18 | 10 | 6 | 2 | - | - | - |
| Yoshinouchi et al. Rheumatology. International. 2005 [62] | 16 | 7 | 7 | - | - | - | 2 |
| Kim et al. Chest 2009 [65] | 14 | 10 | 3 | - | - | - | 1 |
| Nakamura et al. Respiratory Medicine 2012 [32] | 54 | 15 | 16 | 17 | 4 | - | 2 |
| Yousem et al. American Review of Respiratory Disease 1985 [63] | 19 | 5 | 5 | 1 | 6 | 2 | - |
| Tanaka et al. Radiology 2004 [39] | 17 | 2 | 10 | 2 | 2 | 1 | - |
| N (% of total) | 161 (100%) | 57 (35%) | 54 (34%) | 29 (18%) | 13 (8%) | 3 (2%) | 5 (3%) |
| Drug-induced lung toxicities * |
| Infections ** |
| Bronchiectasis |
| COPD |
| Congestive heart failure |
| Pleural effusions |
| Malignancy (lung cancer, lymphoma) |
| Acute exacerbations in RA-ILD |
| Smoking-related parenchymal lung diseases † |
| Rheumatoid nodules |
| Authors | Institution | Study Period | RA-ILD Cohort Size | HRCT Patterns | Survival |
|---|---|---|---|---|---|
| Bongartz et al. Arthritis & Rheumatology 2010 [11] | Population-based incidence cohort of RA patients in Rochester, USA | 1955–1995 | 46 | Not reported | Median survival 2.6 years |
| Kim et al. European Respiratory Journal 2010 [42] | Two referral centers, UCSF and Mayo Clinic, USA | 2001–2008 | 82 | 24% UIP 23% NSIP 51% indeterminate 2% other | Median survival UIP 3.2 years Not UIP 6.6 years |
| Koduri et al. Rheumatology 2010 [106] | ERAS Inception cohort of RA patients from nine rheumatology centers in the UK | 1986–1998 | 52 | Not reported | Median survival 3 years |
| Tsuchiya et al. European Respiratory Journal 2011 [45] | Respiratory center, Japan | 1996–2006 | 102 | 56% UIP 16% NSIP 5% OP 5% DAD 19% combined | Five-year survival UIP 36.6% NSIP 98.3% OP 60.0% DAD 20% |
| Nakamura et al. Respiratory Medicine 2012 [32] | Referral center, Japan | 1980–2009 | 54 | Not reported | Ten-year survival 76.6% (UIP 52.5%, NSIP 84.3%) |
| Solomon et al. Respiratory Medicine 2013 [108] | Two referral centers. National Jewish and Mayo Clinic, USA | 1977–1999 | 48 | Not reported | Median survival 45 months |
| Assayag et al. Radiology 2014 [46] | Three referral centers, USA and Korea | 1997–2011 | 69 | 29% definite UIP 26% possible UIP 45% inconsistent with UIP | Not reported |
| Kelly et al. Rheumatology 2014 [14] | BRILL network, Rheumatology centers in the UK | 1987–2012 | 230 | 65% UIP 24% NSIP OP 6% Overlap syndromes 6% | Not reported |
| Nurmi et al. BMC Pulmonary Medicine 2016 [109] | Referral center, Kuopio, Finland | 2000–2014 | 59 | 59% UIP 14% NSIP 12% OP 15% other | Median survival UIP 92 months Not UIP 137 months |
| Solomon et al. European Respiratory Journal 2016 [107] | Referral center, National Jewish, USA | 1995–2013 | 137 | 79% UIP 21% NSIP | Median survival 10.35 years UIP 10.2 years NSIP 13.6 years |
| Morisset et al. Respiratory Medicine 2017 [47] | Four referral centers, USA, Korea, Italy | NA | 309 | 24% UIP 16% possible UIP 60% inconsistent with UIP | Three-year mortality: GAP stage 1 1.9% GAP stage 2 17.6% GAP stage 3 50.3% |
| Nurmi et al. BMC Pulmonary Medicine 2017 [110] | Referral center, Kuopio, Finland | 2000–2014 | 59 | Reported in a previous study | Median survival GAP stage 1, 152 months, GAP stage 2, 61 months Three-year mortality GAP stage 1, 17.6% GAP stage 2, 27.3% |
| Rojas-Serrano et al. Clinical Rheumatology 2017 [72] | Mexico | 2004–2015 | 78 | 26% UIP 36% NSIP 19% LIP 6% OP 36% overlap | Median survival 5.8 years |
| Yunt et al. Respiratory Medicine 2017 [43] | Referral center, National Jewish, USA | 1995–2014 | 158 | 63% definite UIP 15% possible UIP 22% NSIP | Median survival Definite UIP 2.77 years Possible UIP 6.14 years NSIP incalculable |
| Zamora-Legoff et al. Rheumatology 2017 [40] | Referral center, Mayo Clinic, USA | 1998–2014 | 181 | 54% UIP 40% NSIP 6% OP | Five-year survival 59.7%, no difference among HRCT groups |
| Zhang et al. Clinical Rheumatology 2017 [38] | China | 2008–2013 | 237 | 18.6% UIP 57.8% NSIP | Not reported |
| Juge et al. New England Journal of Medicine 2018 [8] | France, China, Greece, Japan, Mexico, the Netherlands, and USA | NA | 620 | 41% UIP/possible UIP 59% inconsistent with UIP | Not reported |
| Nurmi et al. Respiratory Medicine 2018 [44] | Referral center, Kuopio, Finland | 2000–2014 | 60 | Reported in a previous study | Not reported |
| Sparks et al. Arthritis & Rheumatology 2019 [111] | BRASS cohort, USA | 2003–2016 | 85 | 49% fibrotic NSIP 32% cellular NSIP 19% UIP/AIP/DAD | 37.6% died during follow-up |
|
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bendstrup, E.; Møller, J.; Kronborg-White, S.; Prior, T.S.; Hyldgaard, C. Interstitial Lung Disease in Rheumatoid Arthritis Remains a Challenge for Clinicians. J. Clin. Med. 2019, 8, 2038. https://doi.org/10.3390/jcm8122038
Bendstrup E, Møller J, Kronborg-White S, Prior TS, Hyldgaard C. Interstitial Lung Disease in Rheumatoid Arthritis Remains a Challenge for Clinicians. Journal of Clinical Medicine. 2019; 8(12):2038. https://doi.org/10.3390/jcm8122038
Chicago/Turabian StyleBendstrup, Elisabeth, Janne Møller, Sissel Kronborg-White, Thomas Skovhus Prior, and Charlotte Hyldgaard. 2019. "Interstitial Lung Disease in Rheumatoid Arthritis Remains a Challenge for Clinicians" Journal of Clinical Medicine 8, no. 12: 2038. https://doi.org/10.3390/jcm8122038
APA StyleBendstrup, E., Møller, J., Kronborg-White, S., Prior, T. S., & Hyldgaard, C. (2019). Interstitial Lung Disease in Rheumatoid Arthritis Remains a Challenge for Clinicians. Journal of Clinical Medicine, 8(12), 2038. https://doi.org/10.3390/jcm8122038






