Non-Immersive Virtual Reality for Rehabilitation of the Older People: A Systematic Review into Efficacy and Effectiveness
Abstract
:1. Introduction
2. Methods
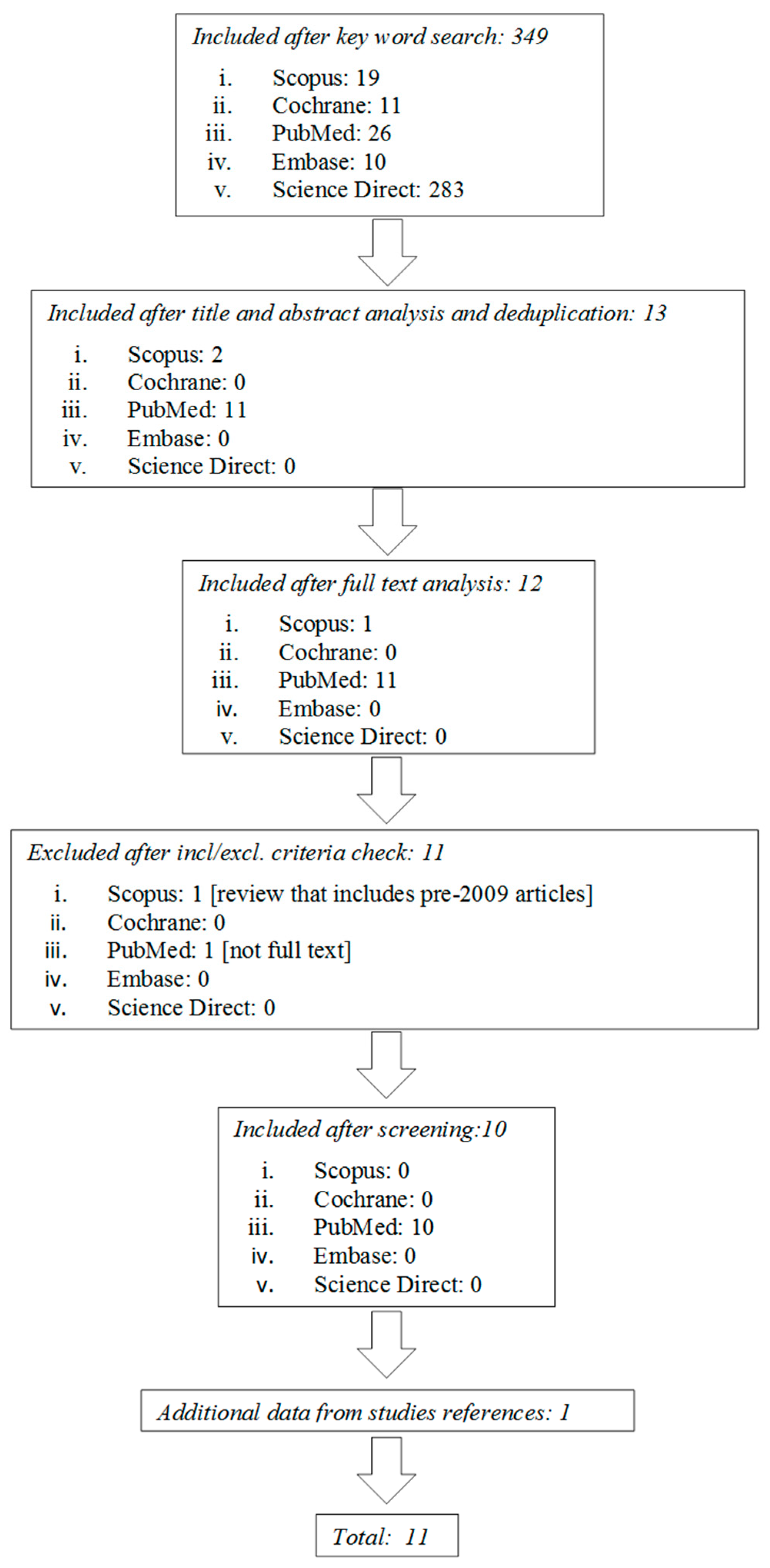
2.1. Literature Search and Study Selection
2.2. Study Selection
- Studies conducted on adult patients aged ≥65 years
- Studies devoted to use a non-immersive virtual reality in rehabilitation
- Studies including upper limb rehabilitation, lower limb rehabilitation, or cognitive rehabilitation
- Randomized clinical trials, with control group that received conventional rehabilitation therapy
- Before-after comparison of a single group
- Review articles
- Conference proceedings
- Studies for which the full text was not found
- Studies written in languages other than English
- Technical papers
- Qualitative studies
2.3. Data Collection
3. Results
3.1. Study Quality Evaluation
3.2. General Characteristics of the Study Population
3.3. Descriptive Analysis and Outcome Measures
3.4. Intervention Effects
4. Discussion
5. Conclusions
Author Contributions
Conflicts of Interest
Abbreviations
| VR | Virtual Reality |
| AD | Alzheimer disease |
| PD | Parkinson disease |
| MS | Multiple sclerosis |
| RCTs | Randomized Controlled Trials |
| EGs | experimental groups |
| CGs | control groups |
| BBS | Berg Balance Scale |
| IADL | Instrumental activities of daily living |
| MCI | Mild cognitive impairment |
References
- Torner, J.; Skouras, S.; Molinuevo, J.L.; Gispert, J.D.; Alpiste, F.; Molinuevo, J.L. Multipurpose Virtual Reality Environment for Biomedical and Health Applications. IEEE Trans. Neural Syst. Rehabil. Eng. 2019, 27, 1511–1520. [Google Scholar] [CrossRef] [PubMed]
- Sveistrup, H. Motor rehabilitation using virtual reality. J. Neuroeng. Rehabil. 2004, 1, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Henderson, A.; Korner-Bitensky, N.; Levin, M. Virtual reality in stroke rehabilitation: A systematic review of its effectiveness for upper limb motor recovery. Top. Stroke Rehabil. 2007, 14, 52–61. [Google Scholar] [CrossRef] [PubMed]
- Piron, L.; Turolla, A.; Agostini, M.; Zucconi, C.S.; Ventura, L.; Tonin, P.; Dam, M. Motor Learning Principles for Rehabilitation: A Pilot Randomized Controlled Study in Poststroke Patients. Neurorehabil. Neural Repair 2010, 6, 501–508. [Google Scholar] [CrossRef] [PubMed]
- Perrochon, A.; Borel, B.; Istrate, D.; Compagnat, M.; Daviet, J.C. Exercise-based games interventions at home in individuals with a neurological disease: A systematic review and meta-analysis. Ann. Phys. Rehabil. Med. 2019, 62, 366–378. [Google Scholar] [CrossRef] [PubMed]
- Mohammadi, R.; Semnani, A.V.; Mirmohammadkhani, M.; Grampurohit, N. Effects of Virtual Reality Compared to Conventional Therapy on Balance Poststroke: A Systematic Review and Meta-Analysis. J. Stroke Cerebrovasc. Dis. 2019, 28, 1787–1798. [Google Scholar] [CrossRef] [PubMed]
- Maggio, M.G.; De Cola, M.C.; Latella, D.; Maresca, G.; Finocchiaro, C.; La Rosa, G.; Cimino, V.; Sorbera, C.; Bramanti, P.; De Luca, R.; et al. What about the Role of Virtual Reality in Parkinson Disease’s Cognitive Rehabilitation? Preliminary Findings from a Randomized Clinical Trial. J. Geriatr. Psychiatry Neurol. 2018, 31, 312–318. [Google Scholar] [CrossRef]
- Alashram, A.R.; Annino, G.; Padua, E.; Romagnoli, C.; Mercuri, N.B. Cognitive rehabilitation post traumatic brain injury: A systematic review for emerging use of virtual reality technology. J. Clin. Neurosci. 2019, 66, 209–219. [Google Scholar] [CrossRef]
- Maggio, M.G.; Russo, M.; Cuzzola, M.F.; Destro, M.; La Rosa, G.; Molonia, F.; Bramanti, P.; Lombardo, G.; De Luca, R.; Calabrò, R.S. Virtual reality in multiple sclerosis rehabilitation: A review on cognitive and motor outcomes. J. Clin. Neurosci. 2019, 65, 106–111. [Google Scholar] [CrossRef]
- Freeman, D.; Reeve, S.; Robinson, A.; Ehlers, A.; Clark, D.; Spanlang, B.; Slater, M. Virtual reality in the assessment, understanding, and treatment of mental health disorders. Psychol. Med. 2017, 47, 2393–2400. [Google Scholar] [CrossRef]
- Ahn, S.; Hwang, S. Virtual rehabilitation of upper extremity function and independence for stoke: A meta-analysis. J. Exerc. Rehabil. 2019, 15, 358–369. [Google Scholar] [CrossRef] [PubMed]
- Van Schaik, P.; Blake, J.; Pernet, F.; Spears, I.; Fencott, C. Virtual augmented exercise gaming for older adults. Cyberpsychol. Behav. 2008, 11, 103–106. [Google Scholar] [CrossRef] [PubMed]
- Zelinski, E.M.; Reyes, R. Cognitive benefits of computer games for older adults. Gerontechnology 2009, 8, 220–235. [Google Scholar] [CrossRef] [PubMed]
- Patel, J.; Fluet, G.; Qiu, Q.; Yarossi, M.; Merians, A.; Tunik, E.; Adamovich, S. Intensive virtual reality and robotic based upper limb training compared to usual care, and associated cortical reorganization, in the acute and early sub-acute periods post-stroke: A feasibility study. J. Neuroeng. Rehabil. 2019, 16, 92. [Google Scholar] [CrossRef]
- Lee, S.H.; Jung, H.Y.; Yun, S.J.; Oh, B.M.; Seo, H.G. Upper Extremity Rehabilitation Using Fully Immersive Virtual Reality Games with a Head Mount Display: A Feasibility Study. PM R. 2019. [Google Scholar] [CrossRef]
- Dos Santos, L.F.; Christ, O.; Mate, K.; Schmidt, H.; Krüger, J.; Dohle, C. Movement visualisation in virtual reality rehabilitation of the lower limb: A systematic review. Biomed. Eng. Online 2016, 15 (Suppl. 3), 144. [Google Scholar]
- Ku, J.; Kim, Y.J.; Cho, S.; Lim, T.; Lee, H.S.; Kang, Y.J. Three-Dimensional Augmented Reality System for Balance and Mobility Rehabilitation in the Elderly: A Randomized Controlled Trial. Cyberpsychol. Behav. Soc. Netw. 2019, 22, 132–141. [Google Scholar] [CrossRef]
- Vogt, S.; Skjæret-Maroni, N.; Neuhaus, D.; Baumeister, J. Virtual reality interventions for balance prevention and rehabilitation after musculoskeletal lower limb impairments in young up to middle-aged adults: A comprehensive review on used technology, balance outcome measures and observed effects. Int. J. Med. Inform. 2019, 126, 46–58. [Google Scholar] [CrossRef]
- Törnbom, K.; Danielsson, A. Experiences of treadmill walking with non-immersive virtual reality after stroke or acquired brain injury—A qualitative study. PLoS ONE 2018, 13, e0209214. [Google Scholar] [CrossRef]
- An, C.M.; Park, Y.H. The effects of semi-immersive virtual reality therapy on standing balance and upright mobility function in individuals with chronic incomplete spinal cord injury: A preliminary study. J. Spinal. Cord Med. 2018, 41, 223–229. [Google Scholar] [CrossRef]
- Darekar, A.; McFadyen, B.J.; Lamontagne, A.; Fung, J. Efficacy of virtual reality-based intervention on balance and mobility disorders post-stroke: A scoping review. J. Neuroeng. Rehabil. 2015, 12, 46. [Google Scholar] [CrossRef] [PubMed]
- García-Betances, R.I.; Arredondo Waldmeyer, M.T.; Fico, G.; Cabrera-Umpiérrez, M.F. A succinct overview of virtual reality technology use in Alzheimer’s disease. Front. Aging Neurosci. 2015, 7, 80. [Google Scholar] [PubMed]
- Palacios-Navarro, G.; Albiol-Pérez, S.; García-Magariño García, I. Effects of sensory cueing in virtual motor rehabilitation. A review. J. Biomed. Inform. 2016, 60, 49–57. [Google Scholar] [CrossRef]
- Walker, M.L.; Ringleb, S.I.; Maihafer, G.C.; Walker, R.; Crouch, J.R.; Van Lunen, B.; Morrison, S. Virtual Reality–Enhanced Partial Body Weight–Supported Treadmill Training Poststroke: Feasibility and Effectiveness in 6 Subjects. Arch. Phys. Med. Rehabil. 2010, 91, 115–122. [Google Scholar] [CrossRef] [PubMed]
- Turolla, A.; Dam, M.; Ventura, L.; Tonin, P.; Agostini, M.; Zucconi, C.; Kiper, P.; Cagnin, A.; Piron, L. Virtual reality for the rehabilitation of the upper limb motor function after stroke: A prospective controlled trial. J. Neuroeng. Rehabil. 2013, 10, 85. [Google Scholar] [CrossRef]
- Allain, P.; Foloppe, D.A.; Besnard, J.; Yamaguchi, T.; Etcharry-Bouyx, F.; Le Gall, D.; Nolin, P.; Richard, P. Detecting everyday action deficits in Alzheimer’s disease using a nonimmersive virtual reality kitchen. J. Int. Neuropsychol. Soc. 2014, 20, 468–477. [Google Scholar] [CrossRef]
- Saposnik, G.; Cohen, L.G.; Mamdani, M.; Pooyania, S.; Ploughman, M.; Cheung, D.; Shaw, J.; Hall, J.; Nord, P.; Dukelow, S.; et al. Efficacy and safety of non-immersive virtual reality exercising in stroke rehabilitation (EVREST): A randomised, multicentre, single-blind, controlled trial. Lancet Neurol. 2016, 15, 1019–1027. [Google Scholar] [CrossRef]
- Mirelman, A.; Rochester, L.; Maidan, I.; Del Din, S.; Alcock, L.; Nieuwhof, F.; Rikkert, M.O.; Bloem, B.R.; Pelosin, E.; Avanzino, L.; et al. Addition of a non-immersive virtual reality component to treadmill training to reduce fall risk in older adults (V-TIME): A randomised controlled trial. Lancet 2016, 388, 1170–1182. [Google Scholar] [CrossRef]
- Segura-Ortí, E.; Pérez-Domínguez, B.; Ortega-Pérez de Villar, L.; Meléndez-Oliva, E.; Martínez-Gramage, J.; García-Maset, R.; Gil-Gómez, J.A. Virtual reality exercise intradialysis to improve physical function: A feasibility randomized trial. Scand. J. Med. Sci. Sports 2019, 29, 89–94. [Google Scholar] [CrossRef]
- Trevizan, I.L.; Silva, T.D.; Dawes, H.; Massetti, T.; Crocetta, T.B.; Favero, F.M.; Oliveira, A.S.B.; de Araújo, L.V.; Santos, A.C.C.; de Abreu, L.C.; et al. Efficacy of different interaction devices using non-immersive virtual tasks in individuals with Amyotrophic Lateral Sclerosis: A cross-sectional randomized trial. BMC Neurol. 2018, 18, 209. [Google Scholar] [CrossRef]
- Pelosin, E.; Cerulli, C.; Ogliastro, C.; Lagravinese, G.; Mori, L.; Bonassi, G.; Mirelman, A.; Hausdorff, J.M.; Abbruzzese, G.; Marchese, R.; et al. A multimodal training modulates short-afferent inhibition and improves complex walking in a cohort of faller older adults with an increased prevalence of Parkinson’s disease. J. Gerontol. A Biol. Sci. Med. Sci. 2019. [Google Scholar] [CrossRef] [PubMed]
- Maher, C.G.; Sherrington, C.; Herbert, R.D.; Moseley, A.M.; Elkins, M. Reliability of the PEDro scale for rating quality of randomized controlled trials. Phys. Ther. 2003, 83, 713–721. [Google Scholar] [PubMed]
- Doniger, G.M.; Beeri, M.S.; Bahar-Fuchs, A.; Gottlieb, A.; Tkachov, A.; Kenan, H.; Livny, A.; Bahat, Y.; Sharon, H.; Ben-Gal, O.; et al. Virtual reality-based cognitive-motor training for middle-aged adults at high Alzheimer’s disease risk: A randomized controlled trial. Alzh. Dement. (N. Y.) 2018, 4, 118–129. [Google Scholar] [CrossRef] [PubMed]
- Savović, J.; Weeks, L.; Sterne, J.A.C.; Turner, L.; Altman, D.G.; Moher, D.; Higgins, J.P.T. Evaluation of the Cochrane Collaboration’s tool for assessing the risk of bias in randomized trials: Focus groups, online survey, proposed recommendations and their implementation. Syst. Rev. 2014, 3, 37. [Google Scholar] [CrossRef] [PubMed]
- Weerdesteyn, V.; de Niet, M.; van Duijnhoven, H.J.R.; Geurts, A.C.H. Falls in individuals with stroke. JRRD 2008, 45, 1195–1214. [Google Scholar] [CrossRef]
- Botner, E.M.; Miller, W.C.; Eng, J.J. Measurement properties of the Activities-specific Balance Confidence Scale among individuals with stroke. Disabil. Rehabil. 2005, 27, 156–163. [Google Scholar] [CrossRef]
- Coyle, H.; Traynor, V.; Solowij, N. Computerized and virtual reality cognitive training for individuals at high risk of cognitive decline: Systematic review of the literature. Am. J. Geriatr. Psychiatry 2015, 23, 335–359. [Google Scholar] [CrossRef]
- Manera, V.; Chapoulie, E.; Bourgeois, J.; Guerchouche, R.; David, R.; Ondrej, J.; Drettakis, G.; Robert, P. A feasibility study with image-based rendered virtual reality in patients with mild cognitive impairment and dementia. PLoS ONE 2016, 11, e0151487. [Google Scholar] [CrossRef]
- Maggio, M.G.; Maresca, G.; Scarcella, I.; Latella, D.; De Domenico, C.; Destro, M.; De Luca, R.; Calabro, R.S. Virtual reality-based cognitive rehabilitation in progressive supranuclear palsy. Psychogeriatr. Soc. 2019. [Google Scholar] [CrossRef]
- Regulation (EU) No 282/2014 of the European Parliament and of the Councilof 11 March 2014 on the Establishment of a Third Programme for the Union’s Action in the Field of Health (2014–2020) and Repealing Decision No 1350/2007/EC. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX:32014R0282&from=EN (accessed on 23 October 2019).
| PEDro [32] | Walker et al., 2010 [24] RCT | Turolla et al., 2013 [25] RCT | Allain et al., 2014 [26] RCT | Saposnik et al., 2016 [27] RCT | Mirelman et al., 2016 [28] RCT | Seguera-Ortì et al., 2018 [29] RCT | Trevizan et al., 2018 [30] RCT | Pelosin et al., 2019 [31] RCT |
|---|---|---|---|---|---|---|---|---|
| Eligibility | Y | Y | Y | Y | Y | Y | Y | Y |
| Randomized allocation | N | N | N | Y | Y | Y | Y | Y |
| Concealed allocation | N | N | N | Y | Y | Y | Y | Y |
| Baseline comparability | Y | Y | N | Y | N | N | N | N |
| Blinded subject | N | N | N | N | N | N | N | N |
| Blinded therapists | N | N | N | N | N | N | N | N |
| Blinded raters | N | N | N | Y | Y | Y | Y | Y |
| Key outcomes | Y | Y | Y | Y | Y | Y | Y | Y |
| Intention to treat | N | N | N | Y | Y | Y | Y | N |
| Comparison between groups | N | Y | Y | Y | Y | Y | Y | Y |
| Precision and variability | Y | Y | Y | Y | Y | Y | Y | Y |
| 4/11 | 5/11 | 4/11 | 9/11 | 8/11 | 8/11 | 8/11 | 7/11 | |
| Cochrane’s Risk of bias tool [33] | ||||||||
| Sequence generation | N | N | N | Y | Y | Y | Y | Y |
| Allocation concealment | N | N | N | Y | Y | Y | Y | Y |
| Blinding of participants, personnel and outcome assessors. | N N N | N N N | N N N | N N Y | N N Y | N N Y | N N Y | N N Y |
| Incomplete outcome data. | N | N | N | N | N | N | N | N |
| Selective outcome reporting | Y | Y | Y | Y | Y | Y | Y | Y |
| Other sources of bias | N | Y | Y | Y | Y | N | N | Y |
| 1/8 | 2/8 | 2/8 | 5/8 | 5/8 | 4/8 | 4/8 | 5/8 |
| Population | Technological Devices | Intervention | Comparison | Outcome | |||
|---|---|---|---|---|---|---|---|
| Partecipants in Experimental Group | Partecipants in Control Group | Training Frequency | Intervention Group | Control Group | |||
| Walker et al., 2010 [24] | 6 adults within 1-year post-stroke N = 6, 3 F/3 M Age: 54.3 years (range 41–70 years) | - | A partial body weight-support treadmill in conjunction with a television mounted on a stand in front of the treadmill to display the VR walkthrough environment. | 2 or 3 training sessions per week with partial body weight-supported tredmill with virtual reality system (total 12 training sessions). Initial training duration is 10 minutes; duration was progressed as tolerated. | - | (1) FGA scores increased by 30% (2) BBS scores improved by 10% (3) Overground walking speed increased by 38% | |
| Turolla et al., 2013 [25] | n = 263 post-stroke patients, 105 F/158 M Age: 60.2 ± 14.3 years | n = 113 post-stroke patients, 41 F/72 M Age: 65.4 ±12.5 | The Virtual Reality Rehabilitation System (Khymeia group. Noventa padovana, Italy) includes a pc workstation connected to 3D motion-tracking system and a high-resolution LCD projector displaying the virtual scenarios on a large wall screen. | 40 sessions of daily therapy provided 5 days per week, for 4 weeks. | 40 sessions of daily therapy provided 5 days per week, for 4 weeks. 1 hour of conventional therapy and 1 hour of VR therapy | 2 hours of conventional treatment. | Within groups: F-M UE score improved by 4% in control group, and 10% in experimental group. Between groups: significantly greater motor improvement in experimental group. Within groups: FIM scores improved in both groups. Between groups: FIM scores improved by 5% in experimental group than in control group. |
| Allain et al., 2014 [26] | n = 24 AD patients, 14 F/10 M Age: 76.96 ± 6.05 years | n = 32 healthy older patients, 25 F/7 M Age: 74.13 ± 5.93 | The virtual environment simulated a fully texture, medium-size kitchen. In the foreground, there was a work plane with all the objects needed to prepare a cup of coffee with milk and sugar. Patients controlled the 2D cursor using a computer mouse. | 1. Virtual reality: 3 sessions: 2 of training and one test session to prepare a cup of coffee in virtual condition 2. Reality: to prepare a cup of coffee | Each training sessions lasts 15 minutes | Each training sessions lasts 15 minutes | Within groups: time to complete the virtual task and MMSE score are correlated in both groups |
| Saposnik et al., 2016 [27] | n = 71 stroke patients, 25 F/46 M Age: 62 ± 13 years | n = 70 stroke patients, 22 F/48 M Age: 62 ± 12 years | The Nintendo Wii gaming system or recreational activities (playing cards, bingo, jenga or ball game). | 10 sessions, 60 minutes each, over a 2 week period. | 30 minutes of traditional rehabilitation of the upper extremity + 30 minutes of virtual reality training | 60 minutes of traditional rehabilitation of the upper extremity | 1. Within groups: WMFT performance time improves from baseline to the end of treatment in both groups. 2. Between groups: no differences in WMFT at the end and at 4-weeks post-intervention 3. Between groups: better performance in BBT in control group at the end of treatment. |
| Mirelman et al., 2016 [28] | n = 146 older people at high risk of falls (more than 2 falls in 6 months), 48 F/98 M Age: 74.2 ± 6.9 years | n = 136 older people at high risk of falls (more than 2 falls in 6 months), 52 F/84 M Age: 73.3 ± 6.4 years | The treadmill plus VR intervention included a camera for motion capture and a computer generated simulation. The virtual environments included real-life challenges with obstacles, multiple pathway and distracters. | 3 times per week for 6 weeks, with each session lasting about 45 minutes | 45 minutes of treadmill training with virtual reality | 45 minutes of traditional treadmill training | In the 6 months after training, the incident rate was significantly lower in the treadmill training plus VR group. |
| Seguera-Ortì et al., 2018 [29] | n = 9 patients on hemodialysis, 4 F/5 M Age: 61.8 ± 13.0 | n = 9 patients on hemodialysis, 3 F/6 M Age: 68.3 ± 15.6 | The system is an adapted version of ACT (A la Caza del Tesoro), in which the subject tries to catch a series of targets by moving their leg. | 16 weeks of intra-dialysis exercise program. The program lasted 4 additional weeks. | 5 minutes warm-up; 30 minutes of virtual reality training. | 5 minutes warm-up and strengthening exercises; 30 minute of aerobic training; 5 minutes of stretching. | 1. Between groups: no significant differences in STS-60 2. Between groups: significant differences in gait speed 3. Within groups: significant improvements for STS-10, gait speed, 6 minute walking test between baseline-16 and 20 weeks, and 16–20 weeks. |
| Trevizan et al., 2018 [30] | n = 30 people with ALS, 12 F/18 M Age: 59 years (range 44–74 years) | n = 30 healthy people, equally matched for age and gender with experimental group | The VR environment is a 3D game in which the goal was to reach as many bubbles displayed on the computer monitor. The game was controlled by three different device system: motion tracking, finger motion and touch-screen. | Participants were randomly divided in 3 groups: motion tracking, finger motion control, touchscreen, to perform 3 task phases (acquisition, retention, transfer) | Both experimental and control group showed better performance whn using the touchscreen device in the transfer phase. | ||
| Pelosi net al., 2019 [31] | n = 10 PD + 7 OA 11 F/6 M Age: 73.2 ± 3.6 | n = 14 PD + 8 OA 15 F/7 M Age: 71.9 ± 4.1 | Treadmill with a non-immersive virtual reality that reacts to a virtual environment that included real-life challenges | 45 minutes/session, 3 times a week for 6 weeks | To walk on a treadmill with virtual reality that included obstacles, distracters. | To walk on a treadmill without virtual reality. | Experimental group increased SAI, reduced the number of falls, improved obstacle negotiation performance. |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bevilacqua, R.; Maranesi, E.; Riccardi, G.R.; Di Donna, V.; Pelliccioni, P.; Luzi, R.; Lattanzio, F.; Pelliccioni, G. Non-Immersive Virtual Reality for Rehabilitation of the Older People: A Systematic Review into Efficacy and Effectiveness. J. Clin. Med. 2019, 8, 1882. https://doi.org/10.3390/jcm8111882
Bevilacqua R, Maranesi E, Riccardi GR, Di Donna V, Pelliccioni P, Luzi R, Lattanzio F, Pelliccioni G. Non-Immersive Virtual Reality for Rehabilitation of the Older People: A Systematic Review into Efficacy and Effectiveness. Journal of Clinical Medicine. 2019; 8(11):1882. https://doi.org/10.3390/jcm8111882
Chicago/Turabian StyleBevilacqua, Roberta, Elvira Maranesi, Giovanni Renato Riccardi, Valentina Di Donna, Paolo Pelliccioni, Riccardo Luzi, Fabrizia Lattanzio, and Giuseppe Pelliccioni. 2019. "Non-Immersive Virtual Reality for Rehabilitation of the Older People: A Systematic Review into Efficacy and Effectiveness" Journal of Clinical Medicine 8, no. 11: 1882. https://doi.org/10.3390/jcm8111882
APA StyleBevilacqua, R., Maranesi, E., Riccardi, G. R., Di Donna, V., Pelliccioni, P., Luzi, R., Lattanzio, F., & Pelliccioni, G. (2019). Non-Immersive Virtual Reality for Rehabilitation of the Older People: A Systematic Review into Efficacy and Effectiveness. Journal of Clinical Medicine, 8(11), 1882. https://doi.org/10.3390/jcm8111882






