Usefulness of Liquid Biopsy Biomarkers from Aqueous Humor in Predicting Anti-VEGF Response in Diabetic Macular Edema: Results of a Pilot Study
Abstract
1. Introduction
2. Experimental Section
2.1. Patients
2.2. Definition of Response to Treatment
- (1)
- Rapid response: reduction >50% in CRT at three months (one month after the third injection);
- (2)
- Slow response: reduction between 10–50% in CRT at three months, but >50% at five months (one month after the fifth injection);
- (3)
- No response: <20% reduction in CRT at both three and five months [20].
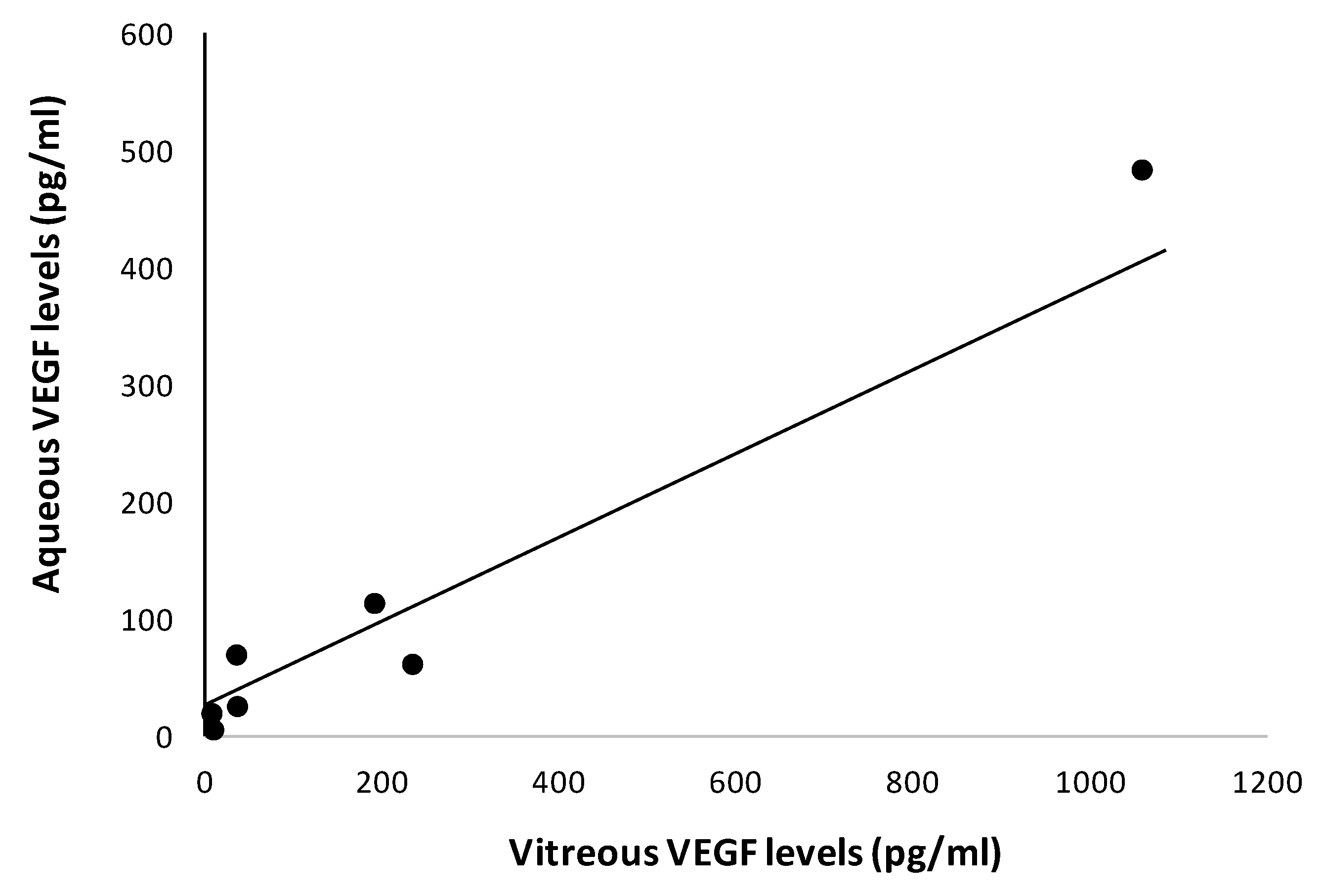
2.3. Aqueous and Vitreous Humor Measurements
2.4. Statistical Analyses
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Yau, J.W.; Rogers, S.L.; Kawasaki, R.; Lamoureux, E.L.; Kowalski, J.W.; Bek, T.; Chen, S.J.; Dekker, J.M.; Fletcher, A.; Grauslund, J.; et al. Global prevalence and major risk factors of diabetic retinopathy. Diabetes Care 2012, 35, 556–564. [Google Scholar] [CrossRef]
- Lightman, S.; Towler, H.M. Diabetic retinopathy. Clin. Cornerstone 2003, 5, 12–21. [Google Scholar] [CrossRef]
- Tong, L.; Vernon, S.A.; Kiel, W.; Sung, V.; Orr, G.M. Association of macular involvement with proliferative retinopathy in type 2 diabetes. Diabet. Med. 2001, 18, 388–394. [Google Scholar] [CrossRef] [PubMed]
- Simó, R.; Hernández, C. Novel approaches for treating diabetic retinopathy based on recent pathogenic evidence. Prog. Retin. Eye Res. 2015, 48, 160–180. [Google Scholar] [CrossRef] [PubMed]
- Tan, G.S.; Cheung, N.; Simó, R.; Cheung, G.C.; Wong, T.Y. Diabetic macular oedema. Lancet Diabetes Endocrinol. 2017, 5, 143–155. [Google Scholar] [CrossRef]
- Cheung, N.; Wong, I.Y.; Wong, T.Y. Ocular anti-VEGF therapy for diabetic retinopathy: Overview of clinical efficacy and evolving applications. Diabetes Care 2014, 37, 900–905. [Google Scholar] [CrossRef] [PubMed]
- Simó, R.; Sundstrom, J.M.; Antonetti, D.A. Ocular Anti-VEGF therapy for diabetic retinopathy: The role of VEGF in the pathogenesis of diabetic retinopathy. Diabetes Care 2014, 37, 893–899. [Google Scholar] [CrossRef]
- Virgili, G.; Parravano, M.; Menchini, F.; Evans, J.R. Anti-vascular endothelial growth factor for diabetic macular oedema. Cochrane Database Syst. Rev. 2014, 24, CD007419. [Google Scholar] [CrossRef]
- Gross, J.G.; Glassman, A.R.; Jampol, L.M.; Inusah, S.; Aiello, L.P.; Antoszyk, A.N.; Baker, C.W.; Berger, B.B.; Bressler, N.M.; Browning, D.; et al. Panretinal Photocoagulation vs Intravitreous Ranibizumab for Proliferative Diabetic Retinopathy: A Randomized Clinical Trial. JAMA 2015, 314, 2137–2146. [Google Scholar] [CrossRef]
- Aiello, L.P.; Avery, R.L.; Arrigg, P.G.; Keyt, B.A.; Jampel, H.D.; Shah, S.T.; Pasquale, L.R.; Thieme, H.; Iwamoto, M.A.; Park, J.E.; et al. Vascular endothelial growth factor in ocular fluid of patients with diabetic retinopathy and other retinal disorders. N. Engl. J. Med. 1994, 331, 1480–1487. [Google Scholar] [CrossRef]
- Simó-Servat, O.; Hernández, C.; Simó, R. Usefulness of the vitreous fluid analysis in the translational research of diabetic retinopathy. Mediat. Inflamm. 2012, 2012, 872978. [Google Scholar] [CrossRef] [PubMed]
- Funatsu, H.; Yamashita, H.; Noma, H.; Mimura, T.; Nakamura, S.; Sakata, K.; Hori, S. Aqueous humor levels of cytokines are related to vitreous levels and progression of diabetic retinopathy in diabetic patients. Graefe’s Arch. Clin. Exp. Opthalmol. 2005, 243, 3–8. [Google Scholar] [CrossRef] [PubMed]
- Noma, H.; Funatsu, H.; Yamasaki, M.; Tsukamoto, H.; Mimura, T.; Sone, T.; Hirayama, T.; Tamura, H.; Yamashita, H.; Minamoto, A.; et al. Aqueous humour levels of cytokines are correlated to vitreous levels and severity of macular oedema in branch retinal vein occlusion. Eye 2008, 22, 42–48. [Google Scholar] [CrossRef] [PubMed]
- Vujosevic, S.; Simó, R. Local and Systemic Inflammatory Biomarkers of Diabetic Retinopathy: An Integrative Approach. Investig. Ophthalmol. Vis. Sci. 2017, 58, BIO68–BIO75. [Google Scholar] [CrossRef] [PubMed]
- Shimura, M.; Yasuda, K.; Motohashi, R.; Kotake, O.; Noma, H. Aqueous cytokine and growth factor levels indicate response to ranibizumab for DME. Br. J. Ophthalmol. 2017, 101, 1518–1523. [Google Scholar] [CrossRef] [PubMed]
- Hillier, R.J.; Ojaimi, E.; Wong, D.T.; Mak, M.Y.K.; Berger, A.R.; Kohly, R.P.; Kertes, P.J.; Forooghian, F.; Boyd, S.R.; Eng, K.; et al. Aqueous Humor Cytokine Levels and Anatomic Response to Intravitreal Ranibizumab in Diabetic Macular Edema. JAMA Ophthalmol. 2018, 136, 382–388. [Google Scholar] [CrossRef]
- Nisic, F.; Jovanovic, N.; Mavija, M.; Alimanovic-Halilovic, E.; Nisic, A.; Lepara, O.; Cemerlic, A. Vitreous concentrations of vascular endothelial growth factor as a potential biomarker for postoperative complications following pars plana vitrectomy. Arch. Med. Sci. 2019, 15, 449–456. [Google Scholar] [CrossRef]
- Boyer, D.S.; Yoon, Y.H.; Belfort, R., Jr.; Bandello, F.; Maturi, R.K.; Augustin, A.J.; Li, X.Y.; Cui, H.; Hashad, Y.; Whitcup, S.M.; et al. Three-year, randomized, sham-controlled trial of dexamethasone intravitreal implant in patients with diabetic macular edema. Ophthalmology 2014, 121, 1904–1912. [Google Scholar] [CrossRef]
- Ankor, R.; Yonekawa, Y.; Todorich, B.; Van Laere, L.; Hussain, R.; Woodward, M.A.; Abbey, A.M.; Wolfe, J.D. Prediction of Anti-VEGF Response in Diabetic Macular Edema After 1 Injection. J. Vitreoretin. Dis. 2017, 1, 169–174. [Google Scholar] [CrossRef]
- Gonzalez, V.H.; Campbell, J.; Holekamp, N.M.; Kiss, S.; Loewenstein, A.; Augustin, A.J.; Ma, J.; Ho, A.C.; Patel, V.; Whitcup, S.M.; et al. Early and Long-Term Responses to Anti-Vascular Endothelial Growth Factor Therapy in Diabetic Macular Edema: Analysis of Protocol I Data. Am. J. Ophthalmol. 2016, 172, 72–79. [Google Scholar] [CrossRef]
- Thomas, B.J.; Shienbaum, G.; Boyer, D.S.; Flynn, H.W., Jr. Evolving strategies in the management of diabetic macular edema: Clinical trials and current management. Can. J. Ophthalmol. 2013, 48, 22–30. [Google Scholar] [CrossRef] [PubMed]
- Brown, D.M.; Nguyen, Q.D.; Marcus, D.M.; Boyer, D.S.; Patel, S.; Feiner, L.; Schlottmann, P.G.; Rundle, A.C.; Zhang, J.; Rubio, R.G.; et al. Long-term outcomes of ranibizumab therapy for diabetic macular edema: The 36-month results from two phase III trials: RISE and RIDE. Ophthalmology 2013, 120, 2013–2022. [Google Scholar] [CrossRef] [PubMed]
- Schmidt-Erfurth, U.; Lang, G.E.; Holz, F.G.; Schlingemann, R.O.; Lanzetta, P.; Massin, P.; Gerstner, O.; Bouazza, A.S.; Shen, H.; Osborne, A.; et al. Three-year outcomes of individualized ranibizumab treatment in patients with diabetic macular edema: The RESTORE extension study. Ophthalmology 2014, 121, 1045–1053. [Google Scholar] [CrossRef] [PubMed]
- Bressler, N.M.; Beaulieu, W.T.; Maguire, M.G.; Glassman, A.R.; Blinder, K.J.; Bressler, S.B.; Gonzalez, V.H.; Jampol, L.M.; Melia, M.; Sun, J.K.; et al. Diabetic Retinopathy Clinical Research Network. Early Response to Anti-Vascular Endothelial Growth Factor and Two-Year Outcomes among Eyes with Diabetic Macular Edema in Protocol T. Am. J. Ophthalmol. 2018, 195, 93–100. [Google Scholar] [CrossRef]
- Macky, T.A.; Mahgoub, M.M. The effect of glycemic control on visual and anatomic outcomes in response to therapy for diabetic macular edema. Eur. J. Ophthalmol. 2012. [Google Scholar] [CrossRef]
- Matsuda, S.; Tam, T.; Singh, R.P.; Kaiser, P.K.; Petkovsek, D.; Carneiro, G.; Zanella, M.T.; Ehlers, J.P. The impact of metabolic parameters on clinical response to VEGF inhibitors for diabetic macular edema. J. Diabetes Complicat. 2014, 28, 166–170. [Google Scholar] [CrossRef]
- Kim, T.K.; Shin, H.Y.; Kim, S.Y.; Lee, Y.C.; Lee, M.Y. Factors Influencing Intravitreal Bevacizumab and Triamcinolone Treatment in Patients with Diabetic Macular Edema. Eur. J. Ophthalmol. 2017, 27, 746–750. [Google Scholar] [CrossRef]
- Donath, M.Y.; Shoelson, S.E. Type 2 diabetes as an inflammatory disease. Nat. Rev. Immunol. 2011, 11, 98–107. [Google Scholar] [CrossRef]
- Simó, R.; Vidal, M.T.; García-Arumí, J.; Carrasco, E.; García-Ramírez, M.; Segura, R.M.; Hernández, C. Intravitreous hepatocyte growth factor in patients with proliferative diabetic retinopathy: A case-control study. Diabetes Res. Clin. Pract. 2006, 71, 36–44. [Google Scholar] [CrossRef]
- Hernández, C.; Segura, R.M.; Fonollosa, A.; Carrasco, E.; Francisco, G.; Simó, R. Interleukin-8, monocyte chemoattractant protein-1 and IL-10 in the vitreous fluid of patients with proliferative diabetic retinopathy. Diabet. Med. 2005, 22, 719–722. [Google Scholar] [CrossRef]
- Hillier, R.J.; Ojaimi, E.; Wong, D.T.; Mak, M.Y.; Berger, A.R.; Kohly, R.P.; Kertes, P.J.; Forooghian, F.; Boyd, S.R.; Eng, K.; et al. Aqueous humor cytokine levels as biomarkers of disease severity in diabetic macular edema. Retina 2017, 37, 761–769. [Google Scholar] [CrossRef]
- Chen, H.; Zhang, X.; Liao, N.; Wen, F. Assessment of biomarkers using multiplex assays in aqueous humor of patients with diabetic retinopathy. BMC Ophthalmol. 2017, 17, 176. [Google Scholar] [CrossRef]
- Kwon, J.W.; Jee, D. Aqueous humor cytokine levels in patients with diabetic macular edema refractory to anti-VEGF treatment. PLoS ONE 2018, 13, e0203408. [Google Scholar] [CrossRef]
- Sepah, Y.J.; Nguyen, Q.D.; Do, D.V.; Day, B.; Wakshull, E.; Stoilov, I. Trends for poorer vision outcomes in nAMD and DME patients with higher aqueous humor levels of IL-6. In Proceedings of the Association for Research in Vision and Ophthalmology’s (ARVO) Annual Meeting, Vancouver, BC, Canada, 2019. Abstract number 4938-A0259. [Google Scholar]
- Affridi, R.; Halim, M.S.; Sadiq, M.A.; Hassan, M.; Agarwal, A.; Do, D.V.; Nguyen, Q.D.; Sepah, J. Can the levels of inflammatory cytokines in the anterior chamber of eyes with diabetic macular edema predict response to therapy? In Proceedings of the Association for Research in Vision and Ophthalmology’s (ARVO) Annual Meeting, Baltimore, MD, USA, 2017. Abstract. [Google Scholar]
- Chalam, K.V.; Grover, S.; Sambhav, K.; Balaiya, S.; Murthy, R.K. Aqueous interleukin-6 levels are superior to vascular endothelial growth factor in predicting therapeutic response to bevacizumab in age-related macular degeneration. J. Ophthalmol. 2014, 2014, 502174. [Google Scholar] [CrossRef] [PubMed]
| CV Intra-Assay | Low Limit of Detection (pg/mL) | |
|---|---|---|
| VEGF | 0.57% | 2.76 |
| PlGF | 0.19% | 0.05 |
| HGF | 0.76% | 1.50 |
| EGF | 13.85% | 2.49 |
| Angiopoietin 2 | 0.56% | 2.41 |
| PDGF-AA | 0.65% | 0.88 |
| PDGF-AB/BB | 0.94% | 1.30 |
| MMP-9 | 0.17% | 1.10 |
| IL-1β | 1.16% | 0.24 |
| IL-6 | 1.15% | 0.78 |
| IL-8 | 0.51% | 1.20 |
| IL-10 | 0.71% | 1.75 |
| IL-12p70 | 0.59% | 1.69 |
| TNF-α | 1.02% | 1.41 |
| TNFR1 | 0.36% | 4.59 |
| TNFR2 | 0.55% | 9.00 |
| Rapid-Response to anti-VEGF (n = 11) | Slow-Response to anti-VEGF (n = 11) | Non-Response to anti-VEGF (n = 9) | p-Value | |
|---|---|---|---|---|
| Gender (M/F) | 6/5 | 6/5 | 7/2 | n.s. |
| Age (years) | 67.5 ± 9.7 | 71 ± 6.9 | 68.1 ± 11.6 | n.s. |
| Diabetes duration (years) | 12.7 ± 2.6 | 12.9 ± 2.6 | 11.6 ± 2.9 | n.s. |
| HbA1c (%) | 7.75 ± 1.34 | 7.54 ± 1.11 | 7.84 ± 1.3 | n.s. |
| Insulin treatment (%) | 70 | 81.8 | 33.3 | n.s. |
| Phakic status (%) | n.s. | |||
| No cataract | 4 | 2 | 5 | |
| Cataract | 0 | 1 | 0 | |
| Previous cataract surgery | 6 | 8 | 4 | |
| Glaucoma (%) | 10 | 27.3 | 33.3 | n.s. |
| NPDR (%) / PDR (%) | 91 / 9 | 82/18 | 89/11 | n.s. |
| Central Retinal Thickness (µm) | 443 ± 91 | 542 ± 179 | 449 ± 67 | n.s. |
| Macular volume (mm3) | 9.67 ± 1.83 | 11.55 ± 4.16 | 9.62 ± 1.37 | n.s. |
| Mean BCVA (ETDRS letters) | 65 (20/50) | 67.2 (20/50) | 62.5 (20/63) | n.s. |
| Rapid-Response to anti-VEGF (n = 11) | Delayed-Response to anti-VEGF (n = 11) | Non-Response to anti-VEGF (n = 9) | p-Value | |
|---|---|---|---|---|
| Group | 1 | 2 | 3 | |
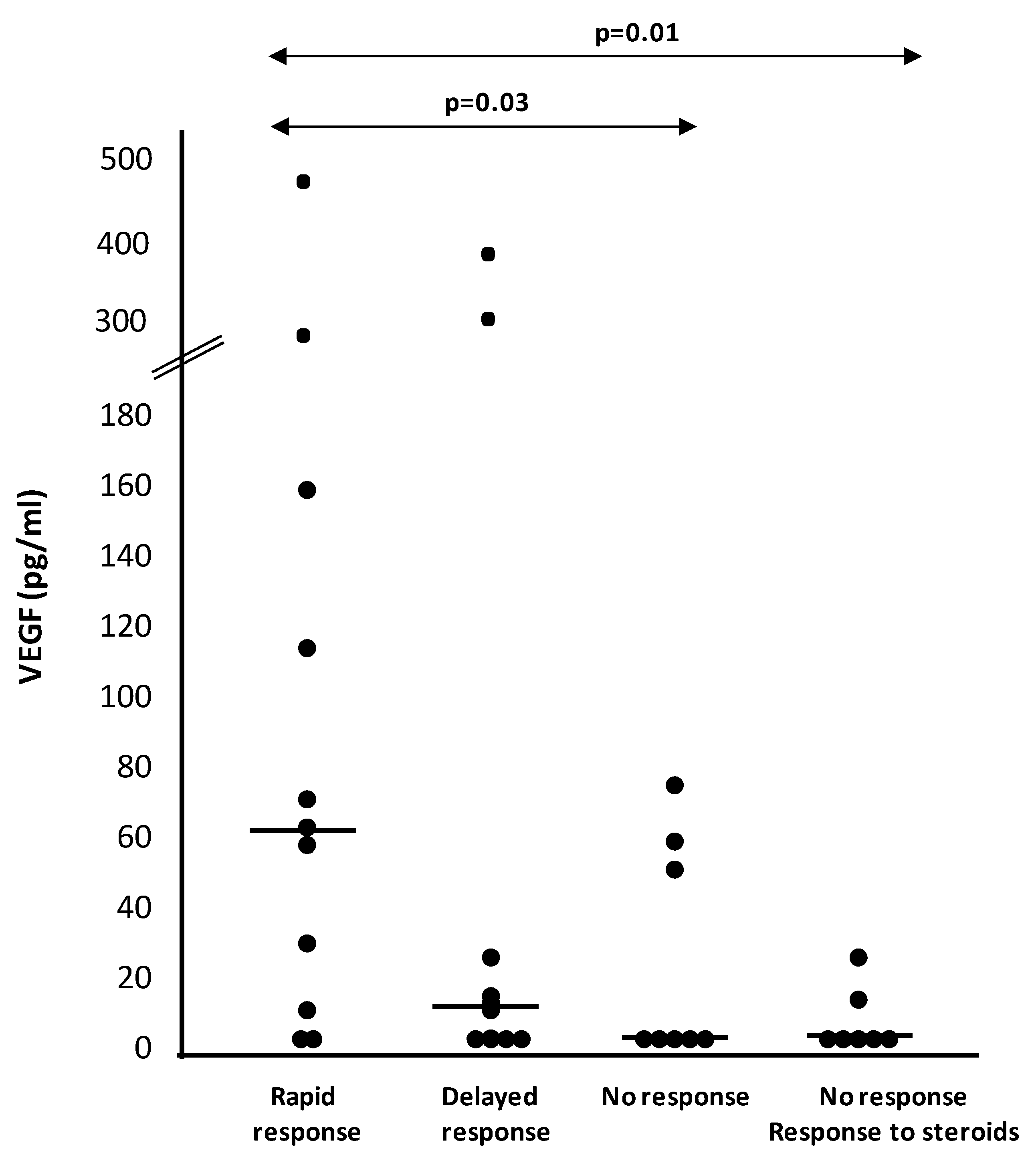
| VEGF (pg/mL) | 62.2 (10–160) | 10.1 (2.7–26.8) | 2.7 (2.7–55) | 0.08 * |
| 0.03 ** | ||||
| PIGF (pg/mL) | 266 (3.8–266) | 266 (8.9–266) | 266 (2.6–266) | 0.84 * |
| 0.99 ** | ||||
| HGF (pg/mL) | 343 (35.19–433) | 482 (198–2036) | 258 (177–759) | 0.15 * |
| 0.94 ** | ||||
| Angiopoietin-2 (pg/mL) | 5.6 (4.5–20.7) | 15.5 (5.6–30.62) | 6.8 (2–21.9) | 0.13 * |
| 0.88 ** | ||||
| PDGF-AA (pg/mL) | 18.9 (6.8–18.9) | 27.2 (22–27.3) | 13 (11.2–17.5) | 0.74 * |
| 0.37 ** | ||||
| MMP-9 (pg/mL) | 74.3 (28.4–174) | 237 (93.4–282) | 73 (2–182) | 0.07 * |
| 0.41 ** | ||||
| IL-6 (pg/mL) | 4 (0.7–17) | 6.7 (3–13.6) | 1.52 (0.7–7.7) | 0.51 * |
| 0.71 ** | ||||
| IL-8 (pg/mL) | 3.2 (1.6–10.6) | 11.5 (4.1–25.8) | 4.9 (2.3–12.4) | 0.07* |
| 0.60 ** | ||||
| TNFR1 (pg/mL) | 122 (32.5–168) | 171 (91.4–307) | 114 (46.5–219) | 0.10 * |
| 0.90 ** | ||||
| TNFR2 (pg/mL) | 215 (85.3–372) | 340 (210–755) | 234 (124–352) | 0.07 * |
| 0.76 ** |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Udaondo, P.; Hernández, C.; Briansó-Llort, L.; García-Delpech, S.; Simó-Servat, O.; Simó, R. Usefulness of Liquid Biopsy Biomarkers from Aqueous Humor in Predicting Anti-VEGF Response in Diabetic Macular Edema: Results of a Pilot Study. J. Clin. Med. 2019, 8, 1841. https://doi.org/10.3390/jcm8111841
Udaondo P, Hernández C, Briansó-Llort L, García-Delpech S, Simó-Servat O, Simó R. Usefulness of Liquid Biopsy Biomarkers from Aqueous Humor in Predicting Anti-VEGF Response in Diabetic Macular Edema: Results of a Pilot Study. Journal of Clinical Medicine. 2019; 8(11):1841. https://doi.org/10.3390/jcm8111841
Chicago/Turabian StyleUdaondo, Patricia, Cristina Hernández, Laura Briansó-Llort, Salvador García-Delpech, Olga Simó-Servat, and Rafael Simó. 2019. "Usefulness of Liquid Biopsy Biomarkers from Aqueous Humor in Predicting Anti-VEGF Response in Diabetic Macular Edema: Results of a Pilot Study" Journal of Clinical Medicine 8, no. 11: 1841. https://doi.org/10.3390/jcm8111841
APA StyleUdaondo, P., Hernández, C., Briansó-Llort, L., García-Delpech, S., Simó-Servat, O., & Simó, R. (2019). Usefulness of Liquid Biopsy Biomarkers from Aqueous Humor in Predicting Anti-VEGF Response in Diabetic Macular Edema: Results of a Pilot Study. Journal of Clinical Medicine, 8(11), 1841. https://doi.org/10.3390/jcm8111841






