The Association between Nutritional Markers and Heart Rate Variability Indices in Patients Undergoing Chronic Hemodialysis
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Population
2.2. Measurements of Heart Rate Variability
2.3. Nutritional Markers and Cut-Off Points
2.4. Statistical Analyses
3. Results
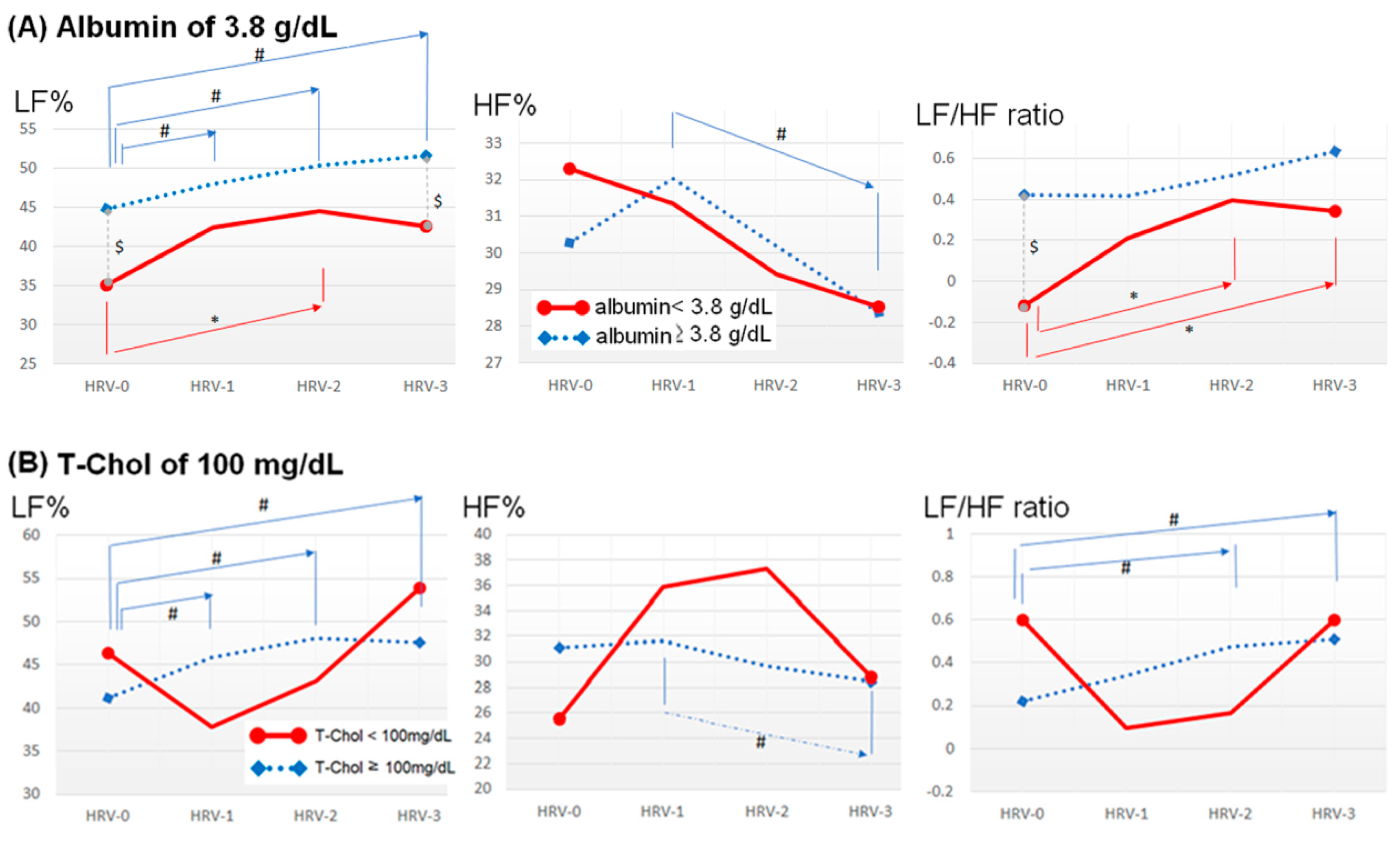
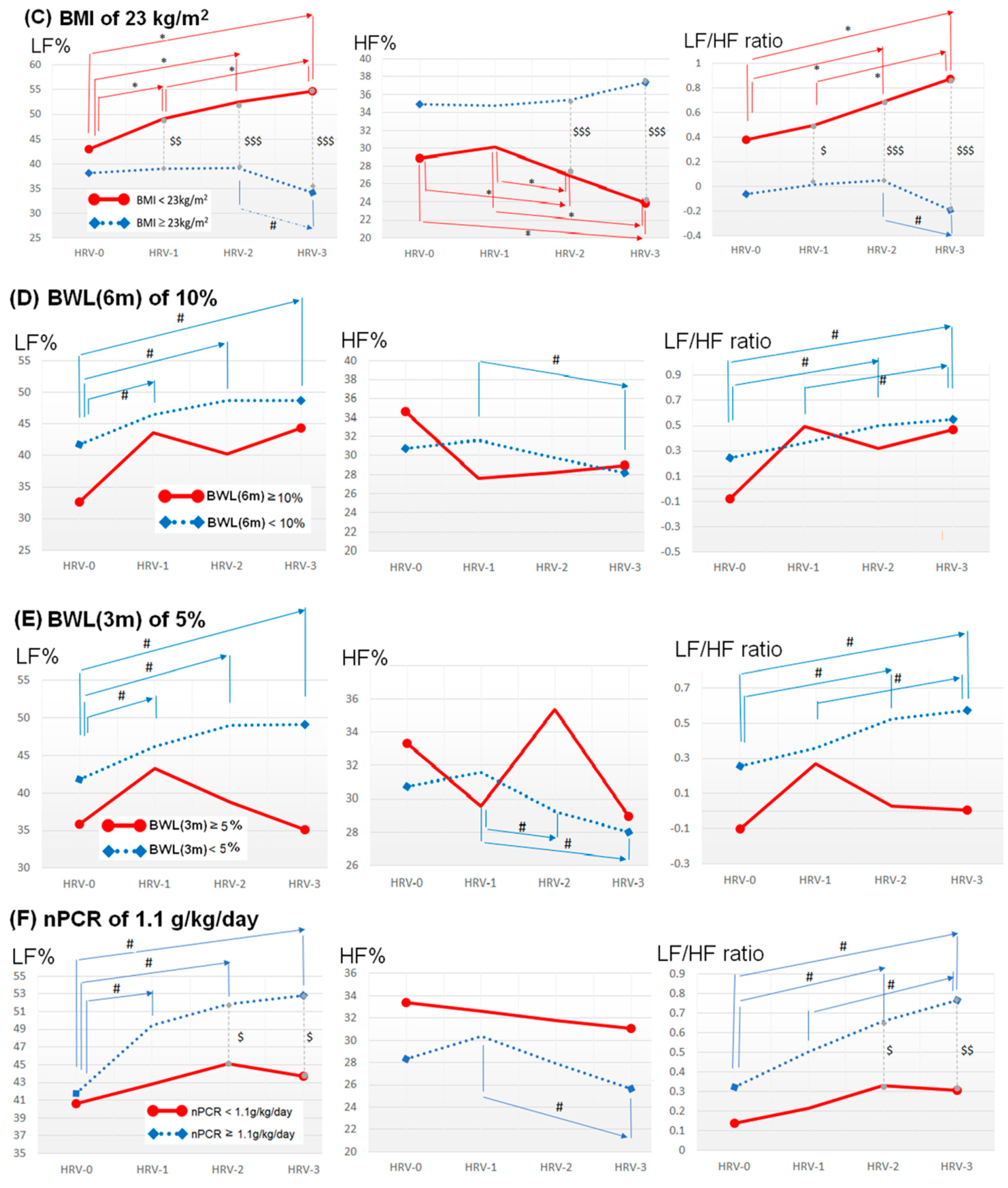
The Association between HRV Indices and Individual Nutritional Markers
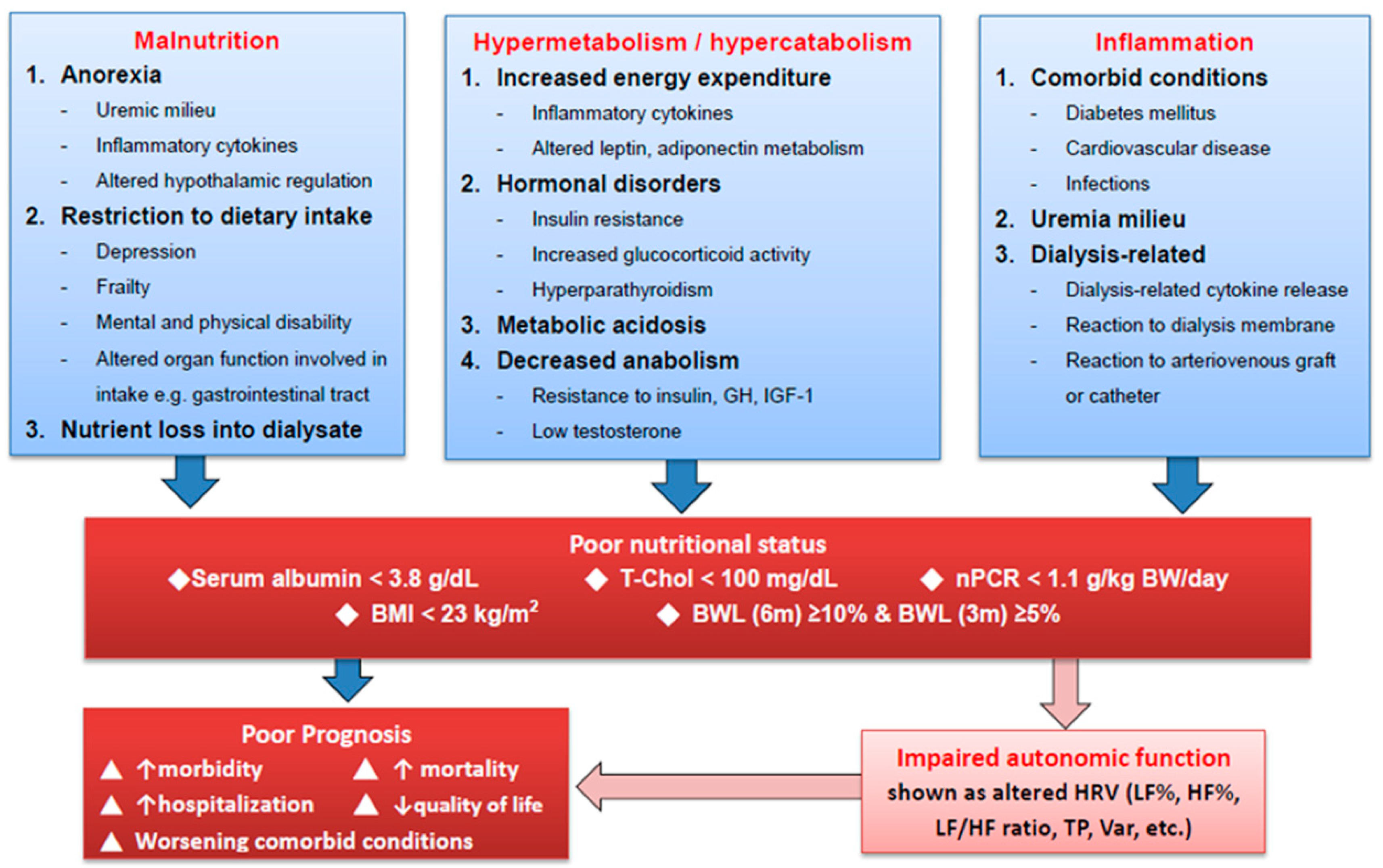
4. Discussion
4.1. Impact of Uremia on ANS
4.2. Impact of Nutritional Status on ANS
4.3. Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ikizler, T.A. A patient with CKD and poor nutritional status. Clin. J. Am. Soc. Nephrol. 2013, 8, 2174–2182. [Google Scholar] [CrossRef] [PubMed]
- Lodebo, B.T.; Shah, A.; Kopple, J.D. Is it Important to Prevent and Treat Protein-Energy Wasting in Chronic Kidney Disease and Chronic Dialysis Patients? J. Ren. Nutr. 2018, 28, 369–379. [Google Scholar] [CrossRef] [PubMed]
- Fouque, D.; Kalantar-Zadeh, K.; Kopple, J.; Cano, N.; Chauveau, P.; Cuppari, L.; Franch, H.; Guarnieri, G.; Ikizler, T.A.; Kaysen, G.; et al. A proposed nomenclature and diagnostic criteria for protein-energy wasting in acute and chronic kidney disease. Kidney Int. 2008, 73, 391–398. [Google Scholar] [CrossRef]
- Hyun, Y.Y.; Lee, K.B.; Han, S.H.; Kim, Y.H.; Kim, Y.S.; Lee, S.W.; Oh, Y.K.; Chae, D.W.; Ahn, C. Nutritional Status in Adults with Predialysis Chronic Kidney Disease: KNOW-CKD Study. J. Korean Med. Sci. 2017, 32, 257–263. [Google Scholar] [CrossRef]
- Araujo, I.C.; Kamimura, M.A.; Draibe, S.A.; Canziani, M.E.; Manfredi, S.R.; Avesani, C.M.; Sesso, R.; Cuppari, L. Nutritional parameters and mortality in incident hemodialysis patients. J. Ren. Nutr. 2006, 16, 27–35. [Google Scholar] [CrossRef]
- Ikizler, T.A.; Flakoll, P.J.; Parker, R.A.; Hakim, R.M. Amino acid and albumin losses during hemodialysis. Kidney Int. 1994, 46, 830–837. [Google Scholar] [CrossRef]
- Kang, S.S.; Chang, J.W.; Park, Y. Nutritional Status Predicts 10-Year Mortality in Patients with End-Stage Renal Disease on Hemodialysis. Nutrients 2017, 9, 399. [Google Scholar] [CrossRef]
- Bergstrom, J.; Lindholm, B. Malnutrition, cardiac disease, and mortality. Perit. Dial. Int. 1999, 19, 309–314. [Google Scholar]
- Laws, R.A.; Tapsell, L.C.; Kelly, J. Nutritional status and its relationship to quality of life in a sample of chronic hemodialysis patients. J. Ren. Nutr. 2000, 10, 139–147. [Google Scholar] [CrossRef]
- Young, H.A.; Benton, D. Heart-rate variability: A biomarker to study the influence of nutrition on physiological and psychological health? Behav. Pharmacol. 2018, 29, 140–151. [Google Scholar] [CrossRef]
- Malik, M. Heart rate variability: Standards of measurement, physiological interpretation and clinical use. Circulation 1996, 93, 1043–1065. [Google Scholar] [CrossRef]
- Elghozi, J.L.; Julien, C. Sympathetic control of short-term heart rate variability and its pharmacological modulation. Fundam. Clin. Pharmacol. 2007, 21, 337–347. [Google Scholar] [CrossRef] [PubMed]
- Ranpuria, R.; Hall, M.; Chan, C.T.; Unruh, M. Heart rate variability (HRV) in kidney failure: Measurement and consequences of reduced HRV. Nephrol. Dial. Transplant. 2008, 23, 444–449. [Google Scholar] [CrossRef] [PubMed]
- Karason, K.; Molgaard, H.; Wikstrand, J.; Sjostrom, L. Heart rate variability in obesity and the effect of weight loss. Am. J. Cardiol. 1999, 83, 1242–1247. [Google Scholar] [CrossRef]
- Chang, Y.M.; Shiao, C.C.; Huang, Y.T.; Chen, I.L.; Yang, C.L.; Leu, S.C.; Su, H.L.; Kao, J.L.; Tsai, S.C.; Jhen, R.N.; et al. Impact of metabolic syndrome and its components on heart rate variability during hemodialysis: A cross-sectional study. Cardiovasc. Diabetol. 2016, 15, 16. [Google Scholar] [CrossRef]
- Mani, A.R.; Montagnese, S.; Jackson, C.D.; Jenkins, C.W.; Head, I.M.; Stephens, R.C.; Moore, K.P.; Morgan, M.Y. Decreased heart rate variability in patients with cirrhosis relates to the presence and degree of hepatic encephalopathy. Am. J. Physiol. Gastrointest. Liver Physiol. 2009, 296, 330–338. [Google Scholar] [CrossRef]
- De Lima, D.C.; Ribeiro, H.S.; Cristina, R.; Oliveira, M.; Generoso Sde, V.; Lima, A.S.; Correia, M.I. Functional status and heart rate variability in end-stage liver disease patients: Association with nutritional status. Nutrition 2015, 31, 971–974. [Google Scholar] [CrossRef]
- Suzuki, M.; Hiroshi, T.; Aoyama, T.; Tanaka, M.; Ishii, H.; Kisohara, M.; Iizuka, N.; Murohara, T.; Hayano, J. Nonlinear measures of heart rate variability and mortality risk in hemodialysis patients. Clin. J. Am. Soc. Nephrol. 2012, 7, 1454–1460. [Google Scholar] [CrossRef]
- Huang, Y.T.; Chang, Y.M.; Chen, I.L.; Yang, C.L.; Leu, S.C.; Su, H.L.; Kao, J.L.; Tsai, S.C.; Jhen, R.N.; Tang, W.R.; et al. Heart rate variability during hemodialysis is an indicator for long-term vascular access survival in uremic patients. PLoS ONE 2017, 12, e0172212. [Google Scholar]
- Chang, Y.M.; Shiao, C.C.; Chang, K.C.; Chen, I.L.; Yang, C.L.; Leu, S.C.; Su, H.L.; Kao, J.L.; Tsai, S.C.; Jhen, R.N. Heart rate variability is an indicator for intradialytic hypotension among chronic hemodialysis patients. Clin. Exp. Nephrol. 2016, 20, 650–659. [Google Scholar] [CrossRef]
- Drawz, P.E.; Babineau, D.C.; Brecklin, C.; He, J.; Kallem, R.R.; Soliman, E.Z.; Xie, D.; Appleby, D.; Anderson, A.H.; Rahman, M.; et al. Heart rate variability is a predictor of mortality in chronic kidney disease: A report from the CRIC Study. Am. J. Nephrol. 2013, 38, 517–528. [Google Scholar] [CrossRef] [PubMed]
- Fleck, A.; Raines, G.; Hawker, F.; Trotter, J.; Wallace, P.I.; Ledingham, I.M.; Calman, K.C. Increased vascular permeability: A major cause of hypoalbuminaemia in disease and injury. Lancet 1985, 1, 781–784. [Google Scholar] [CrossRef]
- Fukuta, H.; Hayano, J.; Ishihara, S.; Sakata, S.; Mukai, S.; Ohte, N.; Ojika, K.; Yagi, K.; Matsumoto, H.; Sohmiya, S.; et al. Prognostic value of heart rate variability in patients with end-stage renal disease on chronic haemodialysis. Nephrol. Dial. Transplant. 2003, 18, 318–325. [Google Scholar] [CrossRef]
- Levey, A.S.; Coresh, J.; Bolton, K.; Culleton, B.; Harvey, K.S.; Ikizler, T.A.; Johnson, C.A.; Kausz, A.; Kimmel, P.L.; Kusek, J.; et al. K/DOQI clinical practice guidelines for cardiovascular disease in dialysis patients. Am. J. Kidney Dis. 2005, 45, 1–153. [Google Scholar]
- Chen, H.C.; Yang, C.C.; Kuo, T.B.; Su, T.P.; Chou, P. Cardiac vagal control and theoretical models of co-occurring depression and anxiety: A cross-sectional psychophysiological study of community elderly. BMC Psychiatry 2012, 12, 93. [Google Scholar] [CrossRef]
- Grassi, G.; Quarti-Trevano, F.; Seravalle, G.; Arenare, F.; Volpe, M.; Furiani, S.; Dell’Oro, R.; Mancia, G. Early sympathetic activation in the initial clinical stages of chronic renal failure. Hypertension 2011, 57, 846–851. [Google Scholar] [CrossRef]
- Rubinger, D.; Backenroth, R.; Sapoznikov, D. Sympathetic nervous system function and dysfunction in chronic hemodialysis patients. Semin. Dial. 2013, 26, 333–343. [Google Scholar] [CrossRef]
- Chandra, P.; Sands, R.L.; Gillespie, B.W.; Levin, N.W.; Kotanko, P.; Kiser, M.; Finkelstein, F.; Hinderliter, A.; Pop-Busui, R.; Rajagopalan, S.; et al. Predictors of heart rate variability and its prognostic significance in chronic kidney disease. Nephrol. Dial. Transplant. 2012, 27, 700–709. [Google Scholar] [CrossRef]
- Ewing, D.J.; Winney, R. Autonomic function in patients with chronic renal failure on intermittent haemodialysis. Nephron 1975, 15, 424–429. [Google Scholar] [CrossRef]
- Kim, Y.H.; Ahmed, M.W.; Kadish, A.H.; Goldberger, J.J. Characterization of the factors that determine the effect of sympathetic stimulation on heart rate variability. Pacing Clin. Electrophysiol. 1997, 20, 1936–1946. [Google Scholar] [CrossRef]
- Kuo, G.; Chen, S.W.; Huang, J.Y.; Wu, C.Y.; Fu, C.M.; Chang, C.H.; Liu, S.H.; Chan, Y.H.; Wu, I.W.; Yang, H.Y. Short-term heart rate variability as a predictor of long-term survival in patients with chronic hemodialysis: A prospective cohort study. J. Formos. Med. Assoc. 2018, 117, 1058–1064. [Google Scholar] [CrossRef] [PubMed]
- Kalantar-Zadeh, K.; Block, G.; Humphreys, M.H.; Kopple, J.D. Reverse epidemiology of cardiovascular risk factors in maintenance dialysis patients. Kidney Int. 2003, 63, 793–808. [Google Scholar] [CrossRef] [PubMed]
- Don, B.R.; Kaysen, G. Serum albumin: Relationship to inflammation and nutrition. Semin. Dial. 2004, 17, 432–437. [Google Scholar] [CrossRef] [PubMed]
- Kaysen, G.A.; Dubin, J.A.; Muller, H.G.; Rosales, L.; Levin, N.W.; Mitch, W.E.; Niddk, H.S.G. Inflammation and reduced albumin synthesis associated with stable decline in serum albumin in hemodialysis patients. Kidney Int. 2004, 65, 1408–1415. [Google Scholar] [CrossRef] [PubMed]
- Sabatino, A.; Regolisti, G.; Antonucci, E.; Cabassi, A.; Morabito, S.; Fiaccadori, E. Intradialytic parenteral nutrition in end-stage renal disease: Practical aspects, indications and limits. J. Nephrol. 2014, 27, 377–383. [Google Scholar] [CrossRef] [PubMed]
- Ballmer, P.E.; McNurlan, M.A.; Hulter, H.N.; Anderson, S.E.; Garlick, P.J.; Krapf, R. Chronic metabolic acidosis decreases albumin synthesis and induces negative nitrogen balance in humans. J. Clin. Investig. 1995, 95, 39–45. [Google Scholar] [CrossRef]
- Moshage, H.J.; Janssen, J.A.; Franssen, J.H.; Hafkenscheid, J.C.; Yap, S.H. Study of the molecular mechanism of decreased liver synthesis of albumin in inflammation. J. Clin. Investig. 1987, 79, 1635–1641. [Google Scholar] [CrossRef]
- Triggiani, A.I.; Valenzano, A.; Ciliberti, M.A.; Moscatelli, F.; Villani, S.; Monda, M.; Messina, G.; Federici, A.; Babiloni, C.; Cibelli, G. Heart rate variability is reduced in underweight and overweight healthy adult women. Clin. Physiol. Funct. Imaging 2017, 37, 162–167. [Google Scholar] [CrossRef]
- Kalantar-Zadeh, K.; Streja, E.; Kovesdy, C.P.; Oreopoulos, A.; Noori, N.; Jing, J.; Nissenson, A.R.; Krishnan, M.; Kopple, J.D.; Mehrotra, R.; et al. The obesity paradox and mortality associated with surrogates of body size and muscle mass in patients receiving hemodialysis. Mayo Clin. Proc. 2010, 85, 991–1001. [Google Scholar] [CrossRef]
- Port, F.K.; Ashby, V.B.; Dhingra, R.K.; Roys, E.C.; Wolfe, R.A. Dialysis dose and body mass index are strongly associated with survival in hemodialysis patients. J. Am. Soc. Nephrol. 2002, 13, 1061–1066. [Google Scholar]
- Campbell, K.L.; MacLaughlin, H.L. Unintentional weight loss is an independent predictor of mortality in a hemodialysis population. J. Ren. Nutr. 2010, 20, 414–418. [Google Scholar] [CrossRef] [PubMed]
- Wong, J.S.; Port, F.K.; Hulbert-Shearon, T.E.; Carroll, C.E.; Wolfe, R.A.; Agodoa, L.Y.; Daugirdas, J.T. Survival advantage in Asian American end-stage renal disease patients. Kidney Int. 1999, 55, 2515–2523. [Google Scholar] [CrossRef] [PubMed]
- Rastovic, M.; Srdic-Galic, B.; Barak, O.; Stokic, E. Association between anthropometric measures of regional fat mass and heart rate variability in obese women. Nutr. Diet. 2017, 74, 51–60. [Google Scholar] [CrossRef] [PubMed]
- Leal, V.O.; Moraes, C.; Stockler-Pinto, M.B.; Lobo, J.C.; Farage, N.E.; Velarde, L.G.; Fouque, D.; Mafra, D. Is a body mass index of 23 kg/m a reliable marker of protein-energy wasting in hemodialysis patients? Nutrition 2012, 28, 973–977. [Google Scholar] [CrossRef]
- Cooper, T.M.; McKinley, P.S.; Seeman, T.E.; Choo, T.H.; Lee, S.; Sloan, R.P. Heart rate variability predicts levels of inflammatory markers: Evidence for the vagal anti-inflammatory pathway. Brain Behav. Immun. 2015, 49, 94–100. [Google Scholar] [CrossRef]
- Lampert, R.; Bremner, J.D.; Su, S.; Miller, A.; Lee, F.; Cheema, F.; Goldberg, J.; Vaccarino, V. Decreased heart rate variability is associated with higher levels of inflammation in middle-aged men. Am. Heart J. 2008, 156, 759.e1–759.e7. [Google Scholar] [CrossRef]
- Ferrario, M.; Moissl, U.; Garzotto, F.; Cruz, D.N.; Clementi, A.; Brendolan, A.; Tetta, C.; Gatti, E.; Signorini, M.G.; Cerutti, S.; et al. Effects of fluid overload on heart rate variability in chronic kidney disease patients on hemodialysis. BMC Nephrol. 2014, 15, 26. [Google Scholar] [CrossRef]
- Tesfaye, S.; Chaturvedi, N.; Eaton, S.E.; Ward, J.D.; Manes, C.; Ionescu-Tirgoviste, C.; Witte, D.R.; Fuller, J.H.; Group EPCS. Vascular risk factors and diabetic neuropathy. N. Engl. J. Med. 2005, 352, 341–350. [Google Scholar] [CrossRef]
- Kilpatrick, R.D.; McAllister, C.J.; Kovesdy, C.P.; Derose, S.F.; Kopple, J.D.; Kalantar-Zadeh, K. Association between serum lipids and survival in hemodialysis patients and impact of race. J. Am. Soc. Nephrol. 2007, 18, 293–303. [Google Scholar] [CrossRef]
- Vincent, A.M.; Russell, J.W.; Low, P.; Feldman, E.L. Oxidative stress in the pathogenesis of diabetic neuropathy. Endocr. Rev. 2004, 25, 612–628. [Google Scholar] [CrossRef]
- Wanner, C.; Krane, V.; Marz, W.; Olschewski, M.; Mann, J.F.; Ruf, G.; Ritz, E.; German, D.; Dialysis Study, I. Atorvastatin in patients with type 2 diabetes mellitus undergoing hemodialysis. N. Engl. J. Med. 2005, 353, 238–248. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Coresh, J.; Eustace, J.A.; Longenecker, J.C.; Jaar, B.; Fink, N.E.; Tracy, R.P.; Powe, N.R.; Klag, M.J. Association between cholesterol level and mortality in dialysis patients: Role of inflammation and malnutrition. JAMA 2004, 291, 451–459. [Google Scholar] [CrossRef]
- Chen, W.R.; Liu, H.B.; Sha, Y.; Shi, Y.; Wang, H.; Yin, D.W.; Chen, Y.D.; Shi, X.M. Effects of Statin on Arrhythmia and Heart Rate Variability in Healthy Persons with 48-Hour Sleep Deprivation. J. Am. Heart Assoc. 2016, 5, e003833. [Google Scholar] [CrossRef] [PubMed]
- Noce, A.; Vidiri, M.F.; Marrone, G.; Moriconi, E.; Bocedi, A.; Capria, A.; Rovella, V.; Ricci, G.; De Lorenzo, A.; Di Daniele, N. Is low-protein diet a possible risk factor of malnutrition in chronic kidney disease patients? Cell Death Discov. 2016, 2, 16026. [Google Scholar] [CrossRef] [PubMed]
- Ravel, V.A.; Molnar, M.Z.; Streja, E.; Kim, J.C.; Victoroff, A.; Jing, J.; Benner, D.; Norris, K.C.; Kovesdy, C.P.; Kopple, J.D.; et al. Low protein nitrogen appearance as a surrogate of low dietary protein intake is associated with higher all-cause mortality in maintenance hemodialysis patients. J. Nutr. 2013, 143, 1084–1092. [Google Scholar] [CrossRef] [PubMed]
| Total Enrolled Patients (n = 175) | |
|---|---|
| Age (years) | 65.1 ± 12.9 |
| Woman | 100 (57.1%) |
| Comorbiditiesand drugs | |
| Diabetes mellitus | 55 (31.4%) |
| Hypertension | 128 (73.1%) |
| Taking beta-blockers or ACEi/ARB | 56 (32.0%) |
| Heart failure | 43 (24.6%) |
| Coronary artery disease | 43 (24.6%) |
| Cerebrovascular disease | 23 (13.1%) |
| Peripheral arterial disease | 13 (7.4%) |
| Causes of uremia | |
| Diabetic nephropathy | 55 (31.4%) |
| Hypertension | 2 (1.1%) |
| Chronic glomerulonephritis | 92 (52.6%) |
| Polycystic kidney disease | 11 (6.3%) |
| Chronic interstitial nephritis | 4 (2.3%) |
| Others | 11 (6.3%) |
| Baseline data | |
| Cardio-thoracic ratio (%) | 52.0 ± 5.0 |
| Blood urea nitrogen (mg/dL) | 74.7 ± 20.1 |
| Creatinine (mg/dL) | 10.5 ± 3.5 |
| Kt/V | 1.4 ± 0.2 |
| Urea reduction ratio (%) | 78.4 ± 54.8 |
| Calcium (mg/dL) | 9.1 ± 0.7 |
| Phosphate (mg/dL) | 4.9 ± 1.7 |
| Calcium × Phosphate ((mg/dL)2) | 44.6 ± 15.7 |
| Sodium (mg/dL) Potassium (mEq/L) | 137.8 ± 3.2 4.7 ± 0.8 |
| Intact parathyroid hormone (ug/L) | 285.5 ± 481.1 |
| Hemoglobin (g/dL) | 9.7 ± 1.4 |
| White blood cell count (×109/L) | 6.3 ± 2.1 |
| Triglyceride (mg/dL) | 157.8 ± 132.0 |
| Low-density lipoprotein (mg/dL) | 98.1 ± 30.3 |
| High-density lipoprotein (mg/dL) | 35.6 ± 18.7 |
| Sugar (non-fasting) (mg/dL) | 148.2 ± 54.9 |
| Glycated hemoglobin (%) | 7.1 ±1.5 |
| Nutritional markers | |
| Albumin (g/dL) | 3.8 ± 0.3 |
| Total cholesterol (mg/dL) | 163.0 ± 35.5 |
| Body mass index (kg/m2) | 22.1 ± 3.9 |
| BW loss within six months (kg) | 0.0 ± 5.8 |
| BW loss within three months (kg) | 0.3 ± 4.7 |
| nPCR (g/kg BW/day) | 1.1 ± 0.4 |
| Data at the index hemodialysis | |
| Dry weight (kg) | 57.6 ± 29.6 |
| Actual UF (kg) | 2.22 ± 0.94 |
| %UF (%) | 4.02 ± 1.65 |
| MAP at initial of hemodialysis | 90.5 ± 17.0 |
| sBP drop > 20 mmHg during hemodialysis | 45 (25.7%) |
| Serum chemistry | Body mass | Dietary intake | |||||
|---|---|---|---|---|---|---|---|
| Albumin < 3.8 versus ≥ 3.8 g/dL | T-Chol < 100 versus ≥ 100 mg/dL | BMI < 23 versus ≥ 23 kg/m2 | BWL (6 m) > 10% versus ≤ 10% | BWL (3 m) > 5% versus ≤ 5% | nPCR < 0.8 versus ≥ 0.8 g/kg BW/day | nPCR <1.1 versus ≥ 1.1 g/kg BW/day | |
| HRV-0 | |||||||
| VLF | ↓* | NS | NS | NS | NS | NS | NS |
| TP | NS | NS | NS | NS | NS | NS | ↓* |
| Var | NS | NS | NS | NS | NS | NS | ↓* |
| LF% | ↓* | NS | NS | NS | NS | NS | NS |
| HF% | NS | NS | NS | NS | NS | NS | NS |
| LF/HF | ↓* | NS | NS | NS | NS | NS | NS |
| HRV-1 | |||||||
| VLF | ↓* | NS | ↑* | NS | NS | NS | NS |
| TP | ↓* | NS | NS | NS | NS | NS | NS |
| Var | NS | NS | NS | NS | NS | NS | NS |
| LF% | NS | NS | ↑** | NS | NS | NS | ↓* |
| HF% | NS | NS | NS | NS | NS | NS | NS |
| LF/HF | NS | NS | ↑* | NS | NS | NS | NS |
| HRV-2 | |||||||
| VLF | NS | NS | NS | NS | NS | NS | NS |
| TP | NS | NS | NS | NS | NS | NS | NS |
| Var | NS | NS | NS | NS | NS | NS | NS |
| LF% | NS | NS | ↑*** | NS | NS | NS | ↓* |
| HF% | NS | NS | ↓*** | NS | NS | NS | NS |
| LF/HF | NS | NS | ↑*** | NS | NS | NS | NS |
| HRV-3 | |||||||
| VLF | NS | NS | NS | NS | NS | NS | NS |
| TP | NS | NS | NS | NS | NS | NS | NS |
| Var | NS | NS | NS | NS | NS | NS | NS |
| LF% | ↓* | NS | ↑*** | NS | NS | NS | ↓* |
| HF% | NS | NS | ↓*** | NS | NS | NS | ↑* |
| LF/HF | NS | NS | ↑*** | NS | NS | NS | ↓* |
| (A) | Serum albumin < 3.8 g/dL (Serum albumin ≥ 3.8 g/dL as reference) | ||||
| HRV indices a | B | p-value | aOR | 95% CI | |
| VLF | −1.13 | 0.003 | 0.32 | 0.15–0.68 | |
| TP | −1.15 | 0.015 | 0.32 | 0.12–0.80 | |
| Var | −0.99 | 0.028 | 0.37 | 0.15–0.60 | |
| LF% | −9.67 | 0.013 | 6.34 × 10-5 | 3.02 × 10−8–0.13 | |
| (B) | T-Chol < 100 mg/dL (T-Chol ≥ 100 mg/dL as reference) | ||||
| HRV indices a | B | p-value | aOR | 95% CI | |
| LF% | 47.26 | <0.001 | 3.35 × 1020 | 4.83 × 1010–2.32 × 1030 | |
| HF% | −14.57 | 0.034 | 4.71 × 10−7 | 6.70 x 10−13–0.33 | |
| LF/HF ratio | 2.22 | <0.001 | 9.17 | 2.80–30.05 | |
| (C) | BMI < 23 kg/m2 (BMI ≥ 23 kg/m2 as reference) | ||||
| HRV indices a | B | p-value | aOR | 95% CI | |
| HF | −1.03 | 0.031 | 0.36 | 0.14–0.91 | |
| LF% | 15.04 | <0.001 | 3.42 × 106 | 6.28 × 103–1.86 × 109 | |
| HF% | −12.40 | <0.001 | 4.14 × 10−6 | 1.27 × 10−7–1.35 × 10−4 | |
| LF/HF ratio | 0.86 | <0.001 | 2.34 | 1.72–3.27 | |
| (D) | BWL (6 m) ≥ 10% (BWL (6 m) < 10% as reference) | ||||
| HRV indices a | B | p-value | aOR | 95% CI | |
| LF% | −37.62 | <0.001 | 4.58 × 10−17 | 7.25 × 10−22–2.89 × 10−12 | |
| HF% | 13.51 | <0.001 | 7.38 × 105 | 9.21 × 102–5.92 × 108 | |
| LF/HF ratio | −1.77 | <0.001 | 0.17 | 0.09–0.32 | |
| (E) | BWL (3 m) ≥ 5% (BWL (3 m) < 5% as reference) | ||||
| HRV indices a | B | p-value | aOR | 95% CI | |
| LF% | −37.58 | <0.001 | 4.78 × 10−17 | 3.50 × 10−21–6.51 × 10−13 | |
| HF% | 18.84 | <0.001 | 1.53 × 108 | 3.63 × 105–6.42 × 1010 | |
| LF/HF ratio | −2.07 | <0.001 | 0.13 | 0.08–0.20 | |
| (F) | nPCR < 1.1 g/kg BW/day (nPCR ≥ 1.1 g/kg BW/day as reference) | ||||
| HRV indices a | B | p-value | aOR | 95% CI | |
| HF% | −5.21 | 0.047 | 0.01 | 3.23 × 105–0.93 | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wu, E.C.-H.; Huang, Y.-T.; Chang, Y.-M.; Chen, I.-L.; Yang, C.-L.; Leu, S.-C.; Su, H.-L.; Kao, J.-L.; Tsai, S.-C.; Jhen, R.-N.; et al. The Association between Nutritional Markers and Heart Rate Variability Indices in Patients Undergoing Chronic Hemodialysis. J. Clin. Med. 2019, 8, 1700. https://doi.org/10.3390/jcm8101700
Wu EC-H, Huang Y-T, Chang Y-M, Chen I-L, Yang C-L, Leu S-C, Su H-L, Kao J-L, Tsai S-C, Jhen R-N, et al. The Association between Nutritional Markers and Heart Rate Variability Indices in Patients Undergoing Chronic Hemodialysis. Journal of Clinical Medicine. 2019; 8(10):1700. https://doi.org/10.3390/jcm8101700
Chicago/Turabian StyleWu, Eric Chien-Hwa, Ya-Ting Huang, Yu-Ming Chang, I-Ling Chen, Chuan-Lan Yang, Show-Chin Leu, Hung-Li Su, Jsun-Liang Kao, Shih-Ching Tsai, Rong-Na Jhen, and et al. 2019. "The Association between Nutritional Markers and Heart Rate Variability Indices in Patients Undergoing Chronic Hemodialysis" Journal of Clinical Medicine 8, no. 10: 1700. https://doi.org/10.3390/jcm8101700
APA StyleWu, E. C.-H., Huang, Y.-T., Chang, Y.-M., Chen, I.-L., Yang, C.-L., Leu, S.-C., Su, H.-L., Kao, J.-L., Tsai, S.-C., Jhen, R.-N., Shiao, C.-C., & , on behalf of SMHGRG (Saint Mary’s Hospital Geriatric Research Group). (2019). The Association between Nutritional Markers and Heart Rate Variability Indices in Patients Undergoing Chronic Hemodialysis. Journal of Clinical Medicine, 8(10), 1700. https://doi.org/10.3390/jcm8101700






