Correlation between B7-H4 and Survival of Non-Small-Cell Lung Cancer Patients Treated with Nivolumab
Abstract
1. Introduction
2. Experimental Section
2.1. Nivolumab Cohort
2.2. Chemotherapy Cohort
2.3. Immunohistochemistry (IHC)
2.4. IHC Scoring
2.5. Statistical Analysis
3. Results
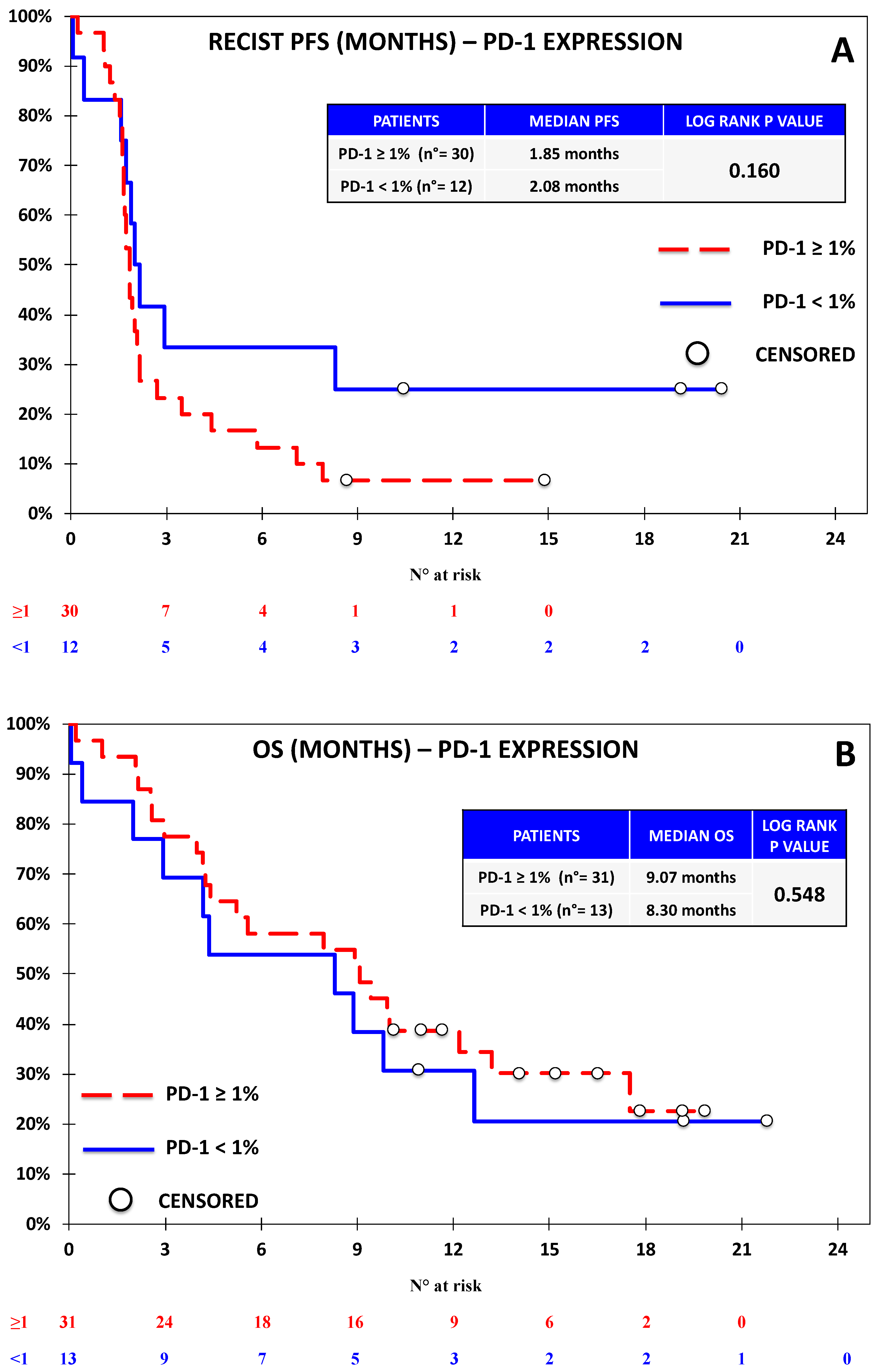
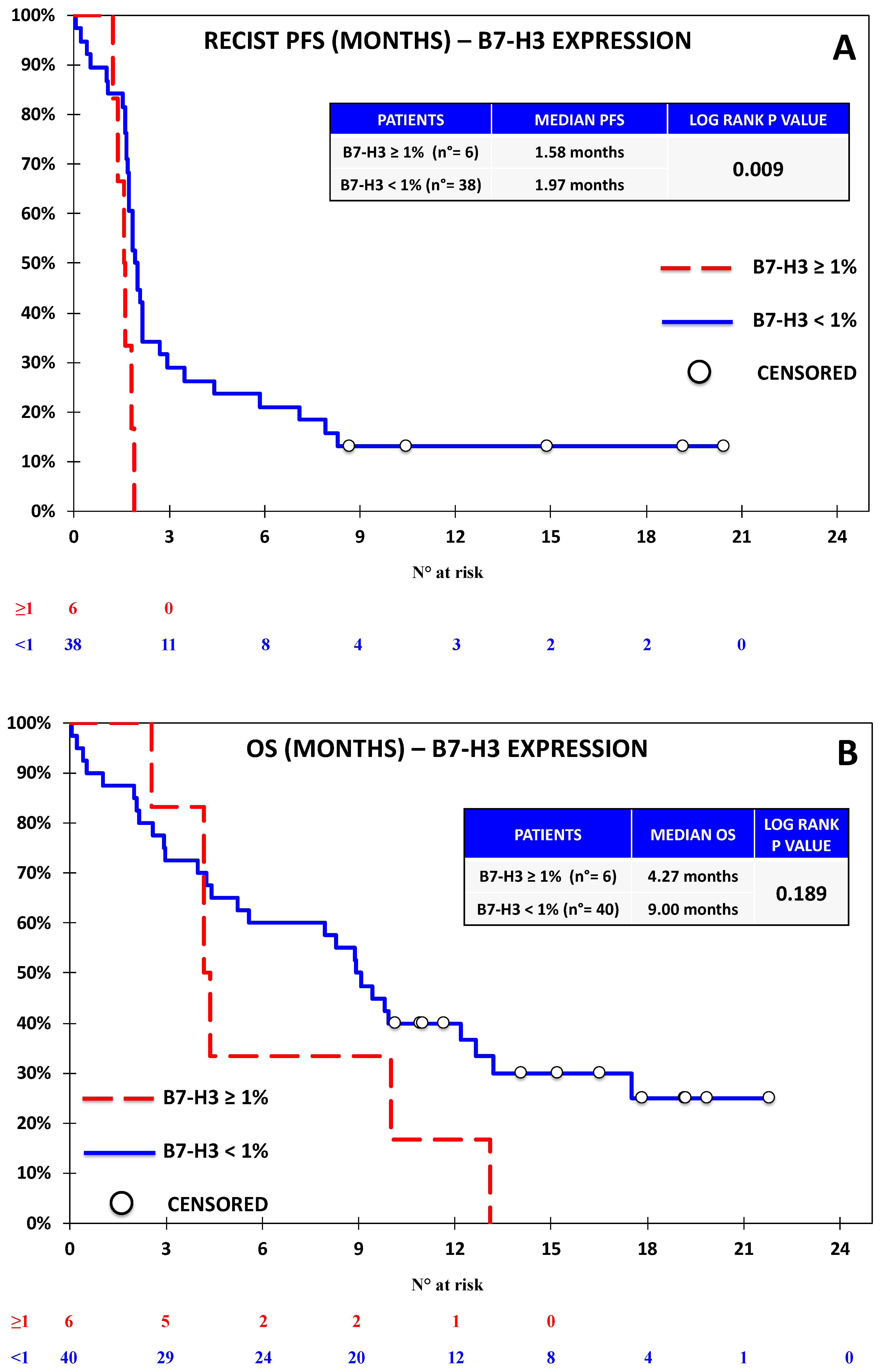
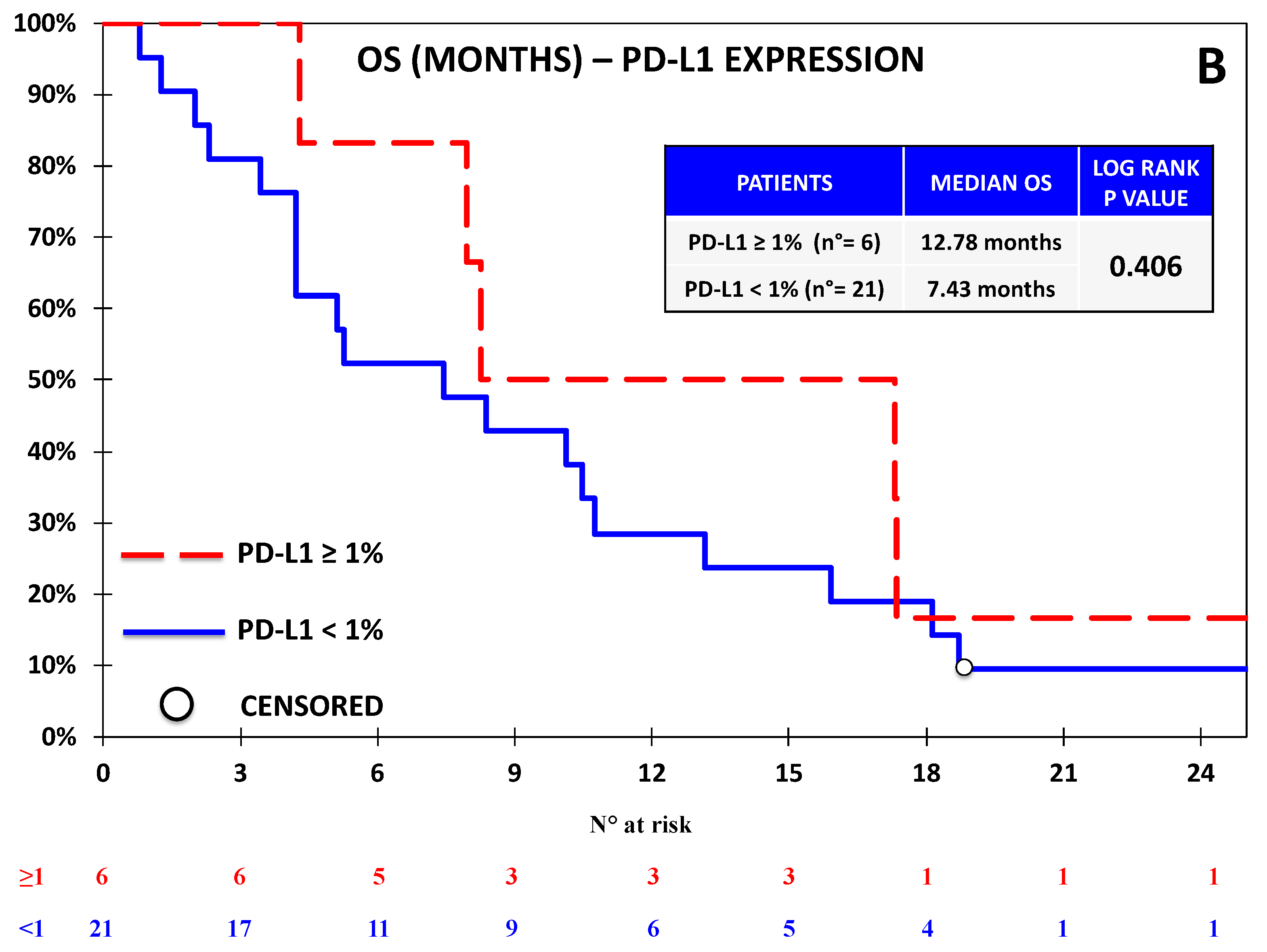
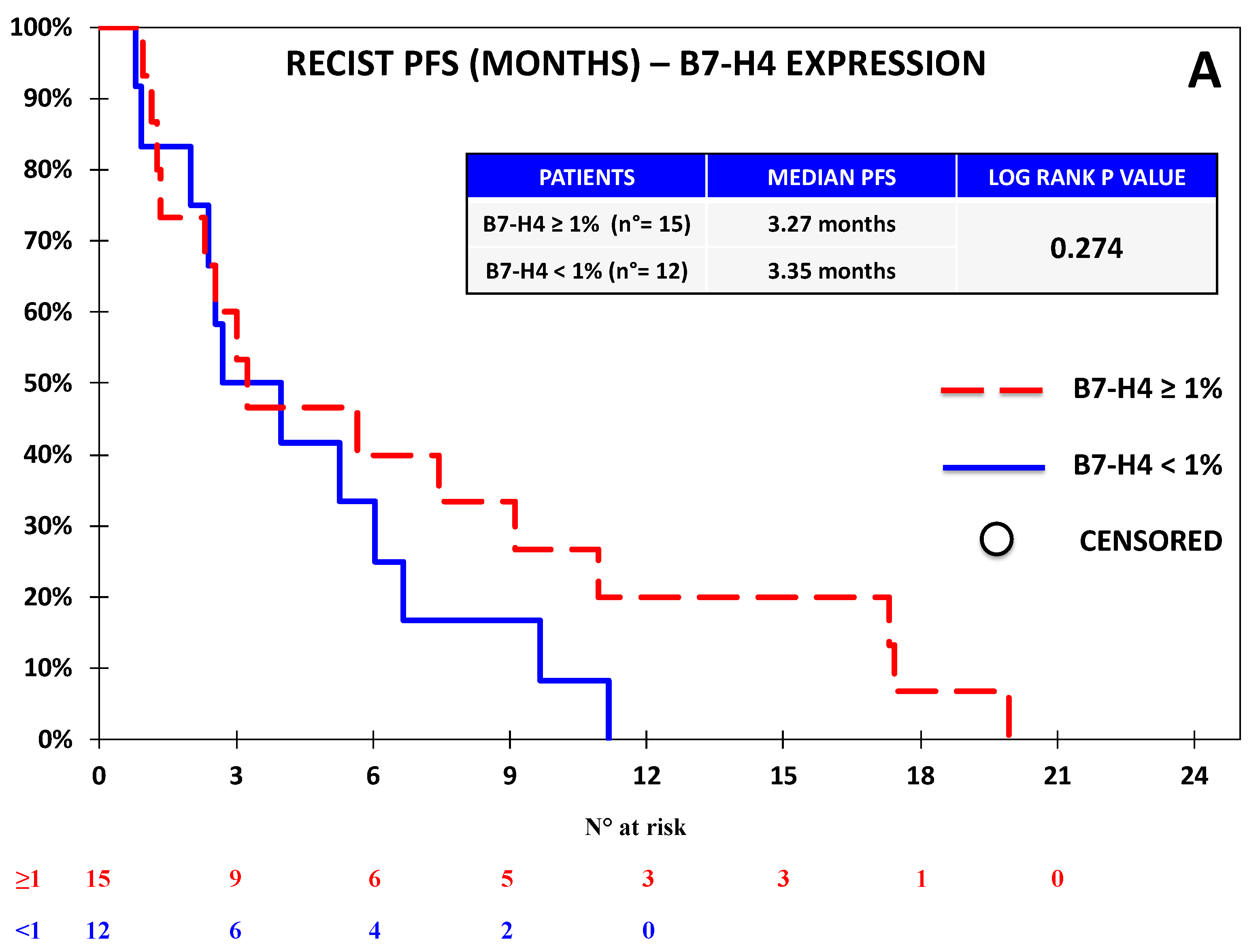
3.1. The Expression of B7-H4 Was Associated with Lower Progression-Free Survival in the Nivolumab Cohort
3.2. B7-H4 Expression Was Not Associated with Response or Survival in the Chemotherapy Cohort
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Iafolla, M.A.J.; Juergens, R.A. Update on Programmed Death-1 and Programmed Death-Ligand 1 Inhibition in the Treatment of Advanced or Metastatic Non-Small Cell Lung Cancer. Front. Oncol. 2017, 7, 67. [Google Scholar] [CrossRef] [PubMed]
- Dal Bello, M.G.; Alama, A.; Coco, S.; Vanni, I.; Grossi, F. Understanding the checkpoint blockade in lung cancer immunotherapy. Drug Discov. Today 2017, 22, 1266–1273. [Google Scholar] [CrossRef] [PubMed]
- Hirsch, F.R.; McElhinny, A.; Stanforth, D.; Ranger-Moore, J.; Jansson, M.; Kulangara, K.; Richardson, W.; Towne, P.; Hanks, D.; Vennapusa, B.; et al. PD-L1 Immunohistochemistry Assays for Lung Cancer: Results from Phase 1 of the Blueprint PD-L1 IHC Assay Comparison Project. J. Thorac. Oncol. 2017, 12, 208–222. [Google Scholar] [CrossRef] [PubMed]
- Ilie, M.; Long-Mira, E.; Bence, C.; Butori, C.; Lassalle, S.; Bouhlel, L.; Fazzalari, L.; Zahaf, K.; Lalvée, S.; Washetine, K.; et al. Comparative study of the PD-L1 status between surgically resected specimens and matched biopsies of NSCLC patients reveal major discordances: A potential issue for anti-PD-L1 therapeutic strategies. Ann. Oncol. 2016, 27, 147–153. [Google Scholar] [CrossRef]
- Grigg, C.; Rizvi, N.A. PD-L1 biomarker testing for non-small cell lung cancer: Truth or fiction? J. Immunother. Cancer 2016, 4, 48. [Google Scholar] [CrossRef]
- Zang, X.; Allison, J.P. The B7 family and cancer therapy: Costimulation and coinhibition. Clin. Cancer Res. 2007, 13 Pt 1, 5271–5279. [Google Scholar] [CrossRef]
- Kerr, K.M.; Hirsch, F.R. Programmed Death Ligand-1 Immunohistochemistry: Friend or Foe? Arch. Pathol. Lab. Med. 2016, 140, 326–331. [Google Scholar] [CrossRef] [PubMed]
- Munari, E.; Zamboni, G.; Marconi, M.; Sommaggio, M.; Brunelli, M.; Martignoni, G.; Netto, G.J.; Moretta, F.; Mingari, M.C.; Salgarello, M.; et al. PD-L1 expression heterogeneity in non-small cell lung cancer: Evaluation of small biopsies reliability. Oncotarget 2017, 8, 90123–90131. [Google Scholar] [CrossRef]
- Latchman, Y.; Wood, C.R.; Chernova, T.; Chaudhary, D.; Borde, M.; Chernova, I.; Iwai, Y.; Long, A.J.; Brown, J.A.; Nunes, R.; et al. PD-L2 is a second ligand for PD-1 and inhibits T cell activation. Nat. Immunol. 2001, 2, 261–268. [Google Scholar] [CrossRef]
- Xiao, Y.; Yu, S.; Zhu, B.; Bedoret, D.; Bu, X.; Francisco, L.M.; Hua, P.; Duke-Cohan, J.S.; Umetsu, D.T.; Sharpe, A.H.; et al. RGMb is a novel binding partner for PD-L2 and its engagement with PD-L2 promotes respiratory tolerance. J. Exp. Med. 2014, 211, 943–959. [Google Scholar] [CrossRef]
- Xu, H.; Chen, X.; Tao, M.; Chen, K.; Chen, C.; Xu, G.; Li, W.; Yuan, S.; Mao, Y. B7-H3 and B7-H4 are independent predictors of a poor prognosis in patients with pancreatic cancer. Oncol. Lett. 2016, 11, 1841–1846. [Google Scholar] [CrossRef] [PubMed]
- Chapoval, A.I.; Ni, J.; Lau, J.S.; Wilcox, R.A.; Flies, D.B.; Liu, D.; Dong, H.; Sica, G.L.; Zhu, G.; Tamada, K.; et al. B7-H3: A costimulatory molecule for T cell activation and IFN-gamma production. Nat. Immunol. 2001, 2, 269–274. [Google Scholar] [CrossRef] [PubMed]
- Leitner, J.; Klauser, C.; Pickl, W.F.; Stöckl, J.; Majdic, O.; Bardet, A.F.; Kreil, D.P.; Dong, C.; Yamazaki, T.; Zlabinger, G.; et al. B7-H3 is a potent inhibitor of human T-cell activation: No evidence for B7-H3 and TREML2 interaction. Eur. J. Immunol. 2009, 39, 1754–1764. [Google Scholar] [CrossRef] [PubMed]
- Prasad, D.V.; Nguyen, T.; Li, Z.; Yang, Y.; Duong, J.; Wang, Y.; Dong, C. Murine B7-H3 is a negative regulator of T cells. J. Immunol. 2004, 173, 2500–2506. [Google Scholar] [CrossRef] [PubMed]
- Yan, R.; Yang, S.; Gu, A.; Zhan, F.; He, C.; Qin, C.; Zhang, X.; Feng, P. Murine b7-h3 is a co-stimulatory molecule for T cell activation. Monoclon. Antibodies Immunodiagn. Immunother. 2013, 32, 395–398. [Google Scholar] [CrossRef] [PubMed]
- Inamura, K.; Yokouchi, Y.; Kobayashi, M.; Sakakibara, R.; Ninomiya, H.; Subat, S.; Nagano, H.; Nomura, K.; Okumura, S.; Shibutani, T.; et al. Tumor B7-H3 (CD276) expression and smoking history in relation to lung adenocarcinoma prognosis. Lung Cancer 2017, 103, 44–51. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.; Zhao, X.; Wu, S.; Du, R.; Zhu, Q.; Fang, H.; Zhang, X.; Zhang, C.; Zheng, W.; Yang, J.; et al. Overexpression of B7-H3 correlates with aggressive clinicopathological characteristics in non-small cell lung cancer. Oncotarget 2016, 7, 81750–81756. [Google Scholar] [CrossRef]
- He, C.; Qiao, H.; Jiang, H.; Sun, X. The inhibitory role of b7-h4 in antitumor immunity: Association with cancer progression and survival. Clin. Dev. Immunol. 2011, 2011, 695834. [Google Scholar] [CrossRef]
- Choi, I.H.; Zhu, G.; Sica, G.L.; Strome, S.E.; Cheville, J.C.; Lau, J.S.; Zhu, Y.; Flies, D.B.; Tamada, K.; Chen, L. Genomic organization and expression analysis of B7-H4, an immune inhibitory molecule of the B7 family. J. Immunol. 2003, 171, 4650–4654. [Google Scholar] [CrossRef]
- Qian, Y.; Sang, Y.; Wang, F.X.; Hong, B.; Wang, Q.; Zhou, X.; Weng, T.; Wu, Z.; Zheng, M.; Zhang, H.; et al. Prognostic significance of B7-H4 expression in matched primary pancreatic cancer and liver metastases. Oncotarget 2016, 7, 72242–72249. [Google Scholar] [CrossRef]
- Smith, J.B.; Stashwick, C.; Powell, D.J. B7-H4 as a potential target for immunotherapy for gynecologic cancers: A closer look. Gynecol. Oncol. 2014, 134, 181–189. [Google Scholar] [CrossRef] [PubMed]
- Tan, Z.; Shen, W. Prognostic role of B7-H4 in patients with non-small cell lung cancer: A meta-analysis. Oncotarget 2017, 8, 27137–27144. [Google Scholar] [CrossRef] [PubMed]
- Song, X.; Shao, Y.; Gu, W.; Xu, C.; Mao, H.; Pei, H.; Jiang, J. Prognostic role of high B7-H4 expression in patients with solid tumors: A meta-analysis. Oncotarget 2016, 7, 76523–76533. [Google Scholar] [CrossRef] [PubMed]
- Altan, M.; Pelekanou, V.; Schalper, K.A.; Toki, M.; Gaule, P.; Syrigos, K.; Herbst, R.S.; Rimm, D.L. B7-H3 expression in NSCLC and its association with B7-H4, PD-L1 and tumor-infiltrating lymphocytes. Clin. Cancer Res. 2017, 23, 5202–5209. [Google Scholar] [CrossRef] [PubMed]
- Available online: https://clinicaltrials.gov/ct2/show/NCT02475382 (accessed on 5 September 2019).
- Crinò, L.; Bidoli, P.; Delmonte, A.; Grossi, F.; De Marinis, F.; Sperandi, F.; Vitiello, F.; Vitali, M.; Soto Parra, H.J.; Scagnoli, S.; et al. Italian cohort of nivolumab Expanded Access Programme (EAP): Preliminary data from a real-world population. J. Clin. Oncol. 2016, 34, 3067. [Google Scholar] [CrossRef]
- Available online: https://clinicaltrials.gov/ct2/show/NCT02055144 (accessed on 5 September 2019).
- Grossi, F.; Rijavec, E.; Genova, C.; Barletta, G.; Biello, F.; Maggioni, C.; Burrafato, G.; Sini, C.; Dal Bello, M.G.; Meyer, K.; et al. Serum proteomic test in advanced non-squamous non-small cell lung cancer treated in first line with standard chemotherapy. Br. J. Cancer 2017, 116, 36–43. [Google Scholar] [CrossRef] [PubMed]
- Neuman, T.; London, M.; Kania-Almog, J.; Litvin, A.; Zohar, Y.; Fridel, L.; Sandbank, J.; Barshak, I.; Vainer, G.W. A Harmonization Study for the Use of 22C3 PD-L1 Immunohistochemical Staining on Ventana’s Platform. J. Thorac. Oncol. 2016, 11, 1863–1868. [Google Scholar] [CrossRef]
- Phillips, T.; Simmons, P.; Inzunza, H.D.; Cogswell, J.; Novotny, J., Jr.; Taylor, C.; Zhang, X. Development of an automated PD-L1 immunohistochemistry (IHC) assay for non-small cell lung cancer. Appl. Immunohistochem. Mol. Morphol. 2015, 23, 541–549. [Google Scholar] [CrossRef]
- Borghaei, H.; Paz-Ares, L.; Horn, L.; Spigel, D.R.; Steins, M.; Ready, N.E.; Chow, L.Q.; Vokes, E.E.; Felip, E.; Holgado, E.; et al. Nivolumab versus Docetaxel in Advanced Nonsquamous Non-Small-Cell Lung Cancer. N. Engl. J. Med. 2015, 373, 1627–1639. [Google Scholar] [CrossRef]
- Brahmer, J.; Reckamp, K.L.; Baas, P.; Crinò, L.; Eberhardt, W.E.; Poddubskaya, E.; Antonia, S.; Pluzanski, A.; Vokes, E.E.; Holgado, E.; et al. Nivolumab versus Docetaxel in Advanced Squamous-Cell Non-Small-Cell Lung Cancer. N. Engl. J. Med. 2015, 373, 123–135. [Google Scholar] [CrossRef]
- Herbst, R.S.; Baas, P.; Kim, D.W.; Felip, E.; Pérez-Gracia, J.L.; Han, J.Y.; Molina, J.; Kim, J.H.; Arvis, C.D.; Ahn, M.J.; et al. Pembrolizumab versus docetaxel for previously treated, PD-L1-positive, advanced non-small-cell lung cancer (KEYNOTE-010): A randomised controlled trial. Lancet 2016, 387, 1540–1550. [Google Scholar] [CrossRef]
- Dong, Y.; Sun, Q.; Zhang, X. PD-1 and its ligands are important immune checkpoints in cancer. Oncotarget 2017, 8, 2171–2186. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Wang, T.; Xu, M.; Xiao, L.; Luo, Y.; Huang, W.; Zhang, Y.; Geng, W. B7-H4 overexpression impairs the immune response of T cells in human cervical carcinomas. Hum. Immunol. 2014, 75, 1203–1209. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.W.; Li, C.; Zhang, R.L.; Xue, H.G.; Zhang, F.X.; Zhang, F.; Gai, X.D. B7-H1 and B7-H4 expression in colorectal carcinoma: Correlation with tumor FOXP3(+) regulatory T-cell infiltration. Acta Histochem. 2014, 116, 1163–1168. [Google Scholar] [CrossRef] [PubMed]
- Kryczek, I.; Wei, S.; Zhu, G.; Myers, L.; Mottram, P.; Cheng, P.; Chen, L.; Coukos, G.; Zou, W. Relationship between B7-H4, regulatory T cells, and patient outcome in human ovarian carcinoma. Cancer Res. 2007, 67, 8900–8905. [Google Scholar] [CrossRef] [PubMed]
- Ye, Z.; Zheng, Z.; Li, X.; Zhu, Y.; Zhong, Z.; Peng, L.; Wu, Y. B7-H3 Overexpression Predicts Poor Survival of Cancer Patients: A Meta-Analysis. Cell. Physiol. Biochem. 2016, 39, 1568–1580. [Google Scholar] [CrossRef] [PubMed]






| Clinical Characteristics | Nivolumab Cohort | Chemotherapy Cohort |
|---|---|---|
| n (%) | n (%) | |
| Patients | 46 | 27 |
| Gender | ||
| Male | 34 (73.9) | 20 (74.1) |
| Female | 12 (26.1) | 7 (25.9) |
| Age (year) | ||
| Range | 44–82 | 46–81 |
| Median | 70 | 69 |
| Smoking habit | ||
| Current | 13 (28.3) | 12 (44.4) 1 |
| Former | 24 (52.2) | 12 (44.4) 1 |
| Never | 9 (19.5) | 3 (11.1) 1 |
| Histology | ||
| Non-squamous | 35 (76.1) | 27 (100.0) |
| Squamous | 11 (23.9) | 0 (0.0) |
| Stage | ||
| IIIB | 2 (4.4) | 0 (0.0) |
| IV | 44 (95.6) | 27 (100.0) |
| ECOG PS | ||
| 0 | 17 (37.0) | 6 (22.2) |
| 1 | 26 (56.5) | 21 (77.8) |
| 2 | 3 (6.5) | 0 (0.0) |
| Previous lines of treatment | ||
| Range | 1–6 | - |
| Median | 2 | - |
| EGFR mutation | ||
| Yes | 3 (8.6) 2,4 | 1 (3.7) 3 |
| No | 32 (91.4) 4 | 26 (96.3) |
| ALK rearrangement | ||
| Yes | 0 (0.0) 4 | 0 (0.0) |
| No | 35 (100.0) 4 | 27 (100.0) |
| Nivolumab Cohort n (%) | |||||||
| Biomarkers | <1% | 1%–9% | 10%–49% | ≥50% | ND | Positive Total | Negative Total |
| PD-L1 | 38 (82.6) | 4 (8.7) | 2 (4.3) | 1 (2.2) | 1 (2.2) | 7 (15.6) | 38 (84.4) |
| PD-L2 | 38 (82.6) | 2 (4.3) | 5 (10.9) | 1 (2.2) | 0 (0.0) | 8 (17.4) | 38 (82.6) |
| PD-1 | 13 (28.3) | 8 (17.4) | 5 (10.9) | 18 (39.1) | 2 (4.3) | 31 (70.5) | 13 (29.5) |
| B7-H3 | 40 (87.0) | 2 (4.3) | 3 (6.5) | 1 (2.2) | 0 (0.0) | 6 (13.0) | 40 (87.0) |
| B7-H4 | 29 (63.0) | 5 (10.9) | 4 (8.7) | 8 (17.4) | 0 (0.0) | 17 (27.0) | 29 (63.0) |
| Chemotherapy Cohort n (%) | |||||||
| Biomarkers | <1% | 1%–9% | 10%–49% | ≥50% | ND | Positive Total | Negative Total |
| PD-L1 | 21 (77.8) | 5 (18.5) | 1 (3.7) | 0 (0.0) | 0 (0.0) | 6 (22.2) | 21 (77.8) |
| B7-H4 1 | 12 (44.4) | 3 (11.1) | 3 (11.1) | 9 (33.3%) | 0 (0.0%) | 15 (55.6) | 12 (44.4) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Genova, C.; Boccardo, S.; Mora, M.; Rijavec, E.; Biello, F.; Rossi, G.; Tagliamento, M.; Dal Bello, M.G.; Coco, S.; Alama, A.; et al. Correlation between B7-H4 and Survival of Non-Small-Cell Lung Cancer Patients Treated with Nivolumab. J. Clin. Med. 2019, 8, 1566. https://doi.org/10.3390/jcm8101566
Genova C, Boccardo S, Mora M, Rijavec E, Biello F, Rossi G, Tagliamento M, Dal Bello MG, Coco S, Alama A, et al. Correlation between B7-H4 and Survival of Non-Small-Cell Lung Cancer Patients Treated with Nivolumab. Journal of Clinical Medicine. 2019; 8(10):1566. https://doi.org/10.3390/jcm8101566
Chicago/Turabian StyleGenova, Carlo, Simona Boccardo, Marco Mora, Erika Rijavec, Federica Biello, Giovanni Rossi, Marco Tagliamento, Maria Giovanna Dal Bello, Simona Coco, Angela Alama, and et al. 2019. "Correlation between B7-H4 and Survival of Non-Small-Cell Lung Cancer Patients Treated with Nivolumab" Journal of Clinical Medicine 8, no. 10: 1566. https://doi.org/10.3390/jcm8101566
APA StyleGenova, C., Boccardo, S., Mora, M., Rijavec, E., Biello, F., Rossi, G., Tagliamento, M., Dal Bello, M. G., Coco, S., Alama, A., Vanni, I., Barletta, G., Bianchi, R., Maggioni, C., Bruzzi, P., & Grossi, F. (2019). Correlation between B7-H4 and Survival of Non-Small-Cell Lung Cancer Patients Treated with Nivolumab. Journal of Clinical Medicine, 8(10), 1566. https://doi.org/10.3390/jcm8101566







