Altered Adipose Tissue DNA Methylation Status in Metabolic Syndrome: Relationships Between Global DNA Methylation and Specific Methylation at Adipogenic, Lipid Metabolism and Inflammatory Candidate Genes and Metabolic Variables
Abstract
1. Introduction
2. Material and Methods
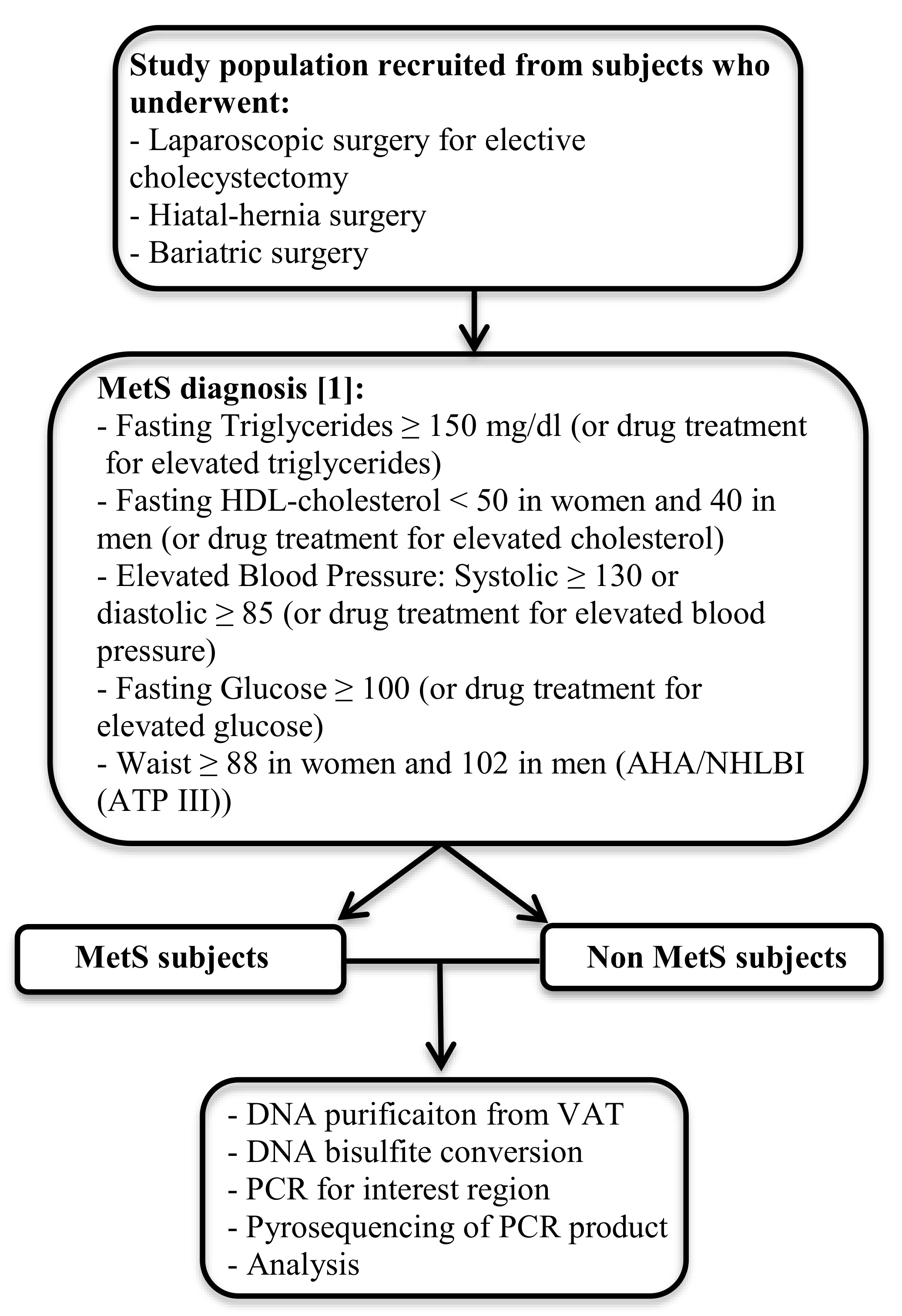
2.1. Study Population
2.2. Laboratory Measurements
2.3. Visceral Adipose Tissue DNA Isolation and Pyrosequencing
2.4. Statistical Analysis
3. Results
3.1. Patient Characterization and Global Methylation
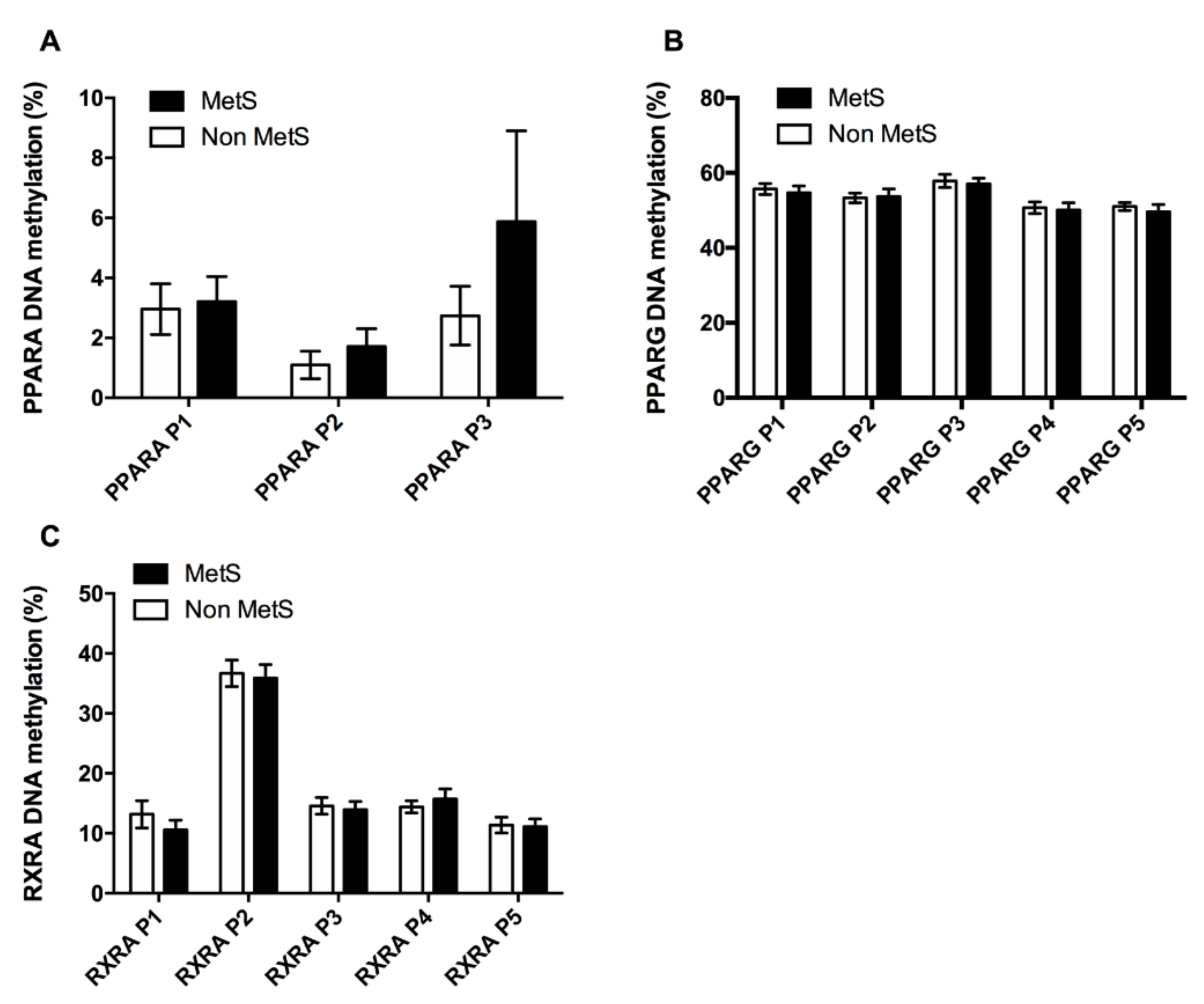
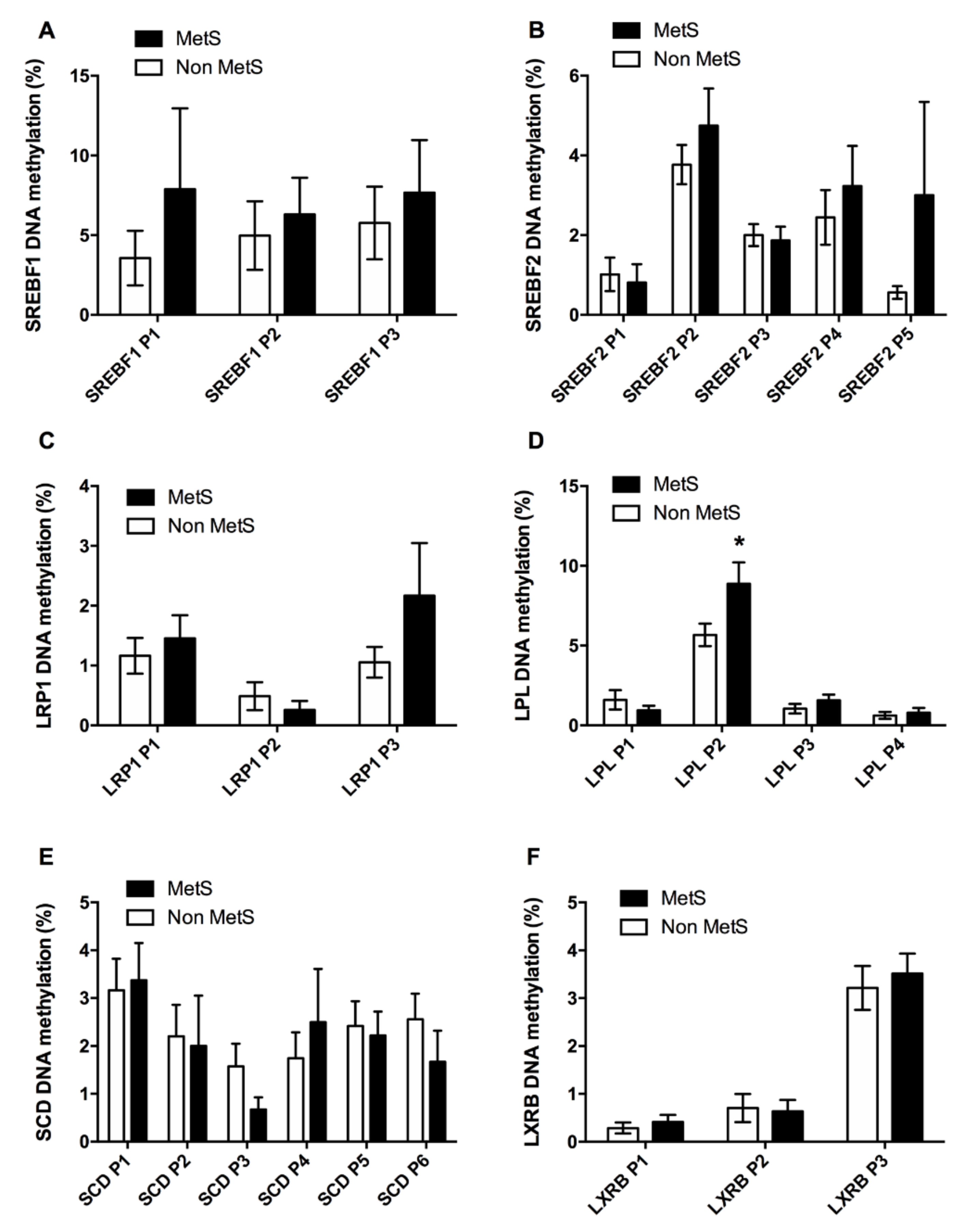
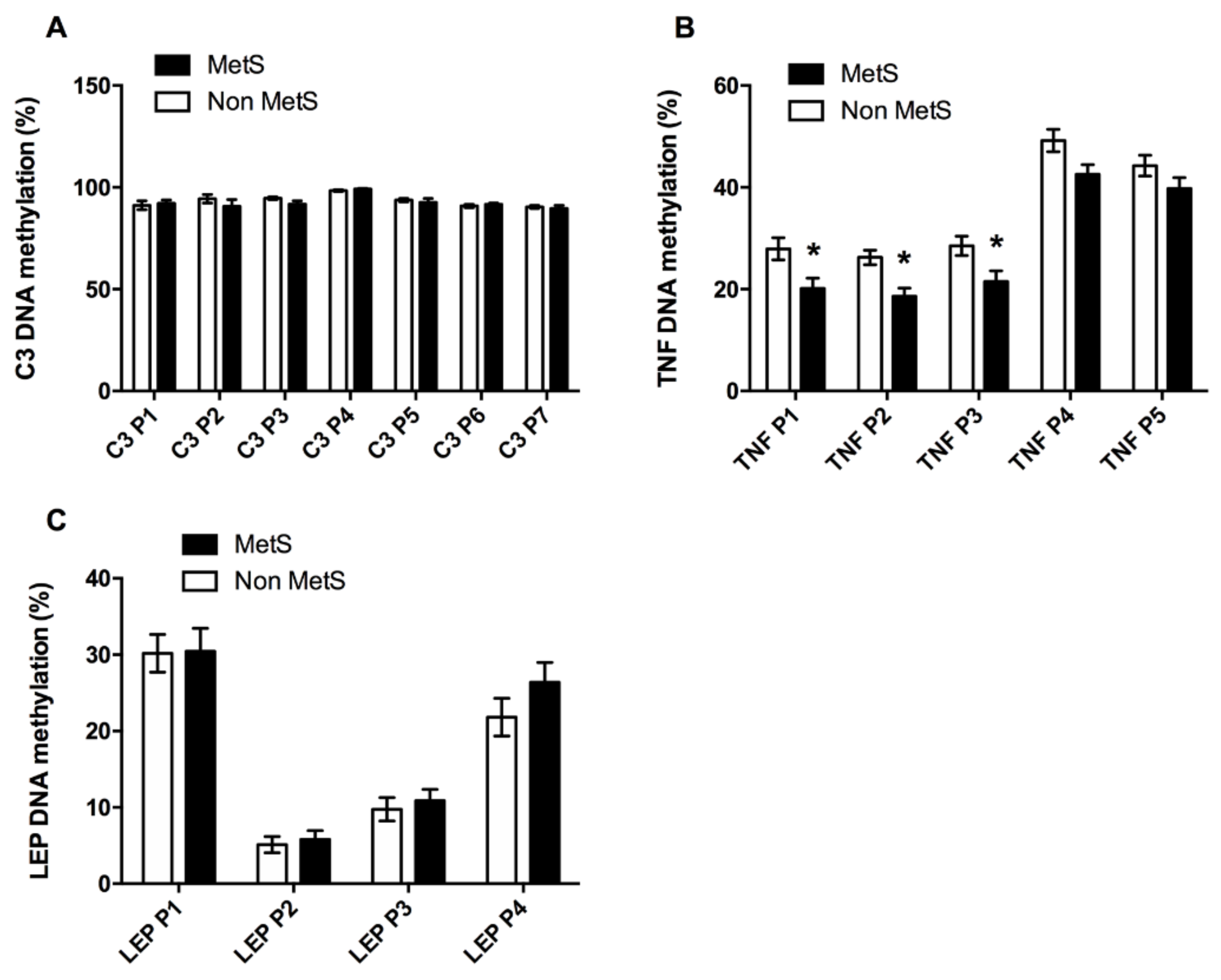
3.2. Gene Specific DNA Methylation in MetS versus Non MetS
3.2.1. Adipogenic and Lipid Metabolism Factors
3.2.2. Inflammation Factors
3.2.3. Regression Analyses
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| MetS | Metabolic syndrome |
| Non MetS | Non metabolic syndrome |
| CpGi | CpG island |
| VAT | Visceral adipose tissue |
| TG | Triglycerides |
| HOMA-IR | homeostasis model assessment of insulin resistance |
| BMI | Body mass index |
| LDL-c | LDL cholesterol |
| HDL-c | HDL cholesterol |
| SBP | Systolic blood pressure |
| DBP | Diastolic blood pressure |
| PUFAS | Monounsaturated fatty acids |
| PPARG | Peroxisome Proliferator Activated Receptor Gamma |
| PPARA | Peroxisome Proliferator Activated Receptor Alpha |
| RXRA | Retinoid X receptor alpha |
| SREBF1 | Sterol regulatory element-binding transcription factor 1 |
| SREBF2 | Sterol regulatory element-binding transcription factor 2 |
| SCD | Stearoyl-CoA desaturase |
| LXRB | Liver X receptor beta |
| LRP1 | Low density lipoprotein receptor-related protein 1 |
| C3 | Complement component 3 |
| TNF | Tumoral necrosis factor |
| LEP | Leptin |
References
- Alberti, K.G.M.M.; Eckel, R.H.; Grundy, S.M.; Zimmet, P.Z.; Cleeman, J.I.; Donato, K.A.; Fruchart, J.-C.; James, W.P.T.; Loria, C.M.; Smith, S.C. Harmonizing the Metabolic Syndrome: A Joint Interim Statement of the International Diabetes Federation Task Force on Epidemiology and Prevention; National Heart, Lung, and Blood Institute; American Heart Association; World Heart Federation; International Atherosclerosis Society; and International Association for the Study of Obesity. Circulation 2009, 120, 1640–1645. [Google Scholar] [CrossRef] [PubMed]
- Uzunlulu, M.; Telci Caklili, O.; Oguz, A. Association between Metabolic Syndrome and Cancer. Ann. Nutr. Metab. 2016, 178, 173–179. [Google Scholar] [CrossRef] [PubMed]
- Castellano-Castillo, D.; Moreno-Indias, I.; Fernandez-Garcia, J.C.; Alcaide-Torres, J.; Moreno-Santos, I.; Ocana, L.; Gluckman, E.; Tinahones, F.; Queipo-Ortuno, M.I.; Cardona, F. Adipose Tissue LPL Methylation Is Associated with Triglyceride Concentrations in the Metabolic Syndrome. Clin. Chem. 2017. [Google Scholar] [CrossRef]
- Suzuki, M.M.; Bird, A. DNA Methylation Landscapes: Provocative Insights from Epigenomics. Nat. Rev. Genet. 2008, 9, 465–476. [Google Scholar] [CrossRef] [PubMed]
- Deaton, A.; Bird, A. CpG Islands and the Regulation of Transcription. Genes Dev. 2011, 25, 1010–1022. [Google Scholar] [CrossRef]
- Jones, P.A. Functions of DNA Methylation: Islands, Start Sites, Gene Bodies and Beyond. Nat. Rev. Genet. 2012, 13, 484–492. [Google Scholar] [CrossRef]
- He, Z.; Zhang, R.; Jiang, F.; Hou, W.; Hu, C. Role of Genetic and Environmental Factors in DNA Methylation of Lipid Metabolism. Genes Dis. 2018, 5, 9–15. [Google Scholar] [CrossRef] [PubMed]
- Alegría-Torres, J.A.; Baccarelli, A.; Bollati, V. Epigenetics and Lifestyle. Epigenomics 2011, 3, 267–277. [Google Scholar] [CrossRef] [PubMed]
- Turcot, V.; Tchernof, A.; Deshaies, Y.; Pérusse, L.; Bélisle, A.; Marceau, S.; Biron, S.; Lescelleur, O.; Biertho, L.; Vohl, M.-C. LINE-1 Methylation in Visceral Adipose Tissue of Severely Obese Individuals Is Associated with Metabolic Syndrome Status and Related Phenotypes. Clin. Epigenet. 2012, 4, 10. [Google Scholar] [CrossRef] [PubMed]
- Ali, O.; Cerjak, D.; Kent, J.W.; James, R.; Blangero, J.; Carless, M.A.; Zhang, Y. Methylation of SOCS3 Is Inversely Associated with Metabolic Syndrome in an Epigenome-Wide Association Study of Obesity. Epigenetics 2016, 11, 699–707. [Google Scholar] [CrossRef] [PubMed]
- Hajer, G.R.; Van Haeften, T.W.; Visseren, F.L.J. Adipose Tissue Dysfunction in Obesity, Diabetes, and Vascular Diseases. Eur. Heart J. 2008, 29, 2959–2971. [Google Scholar] [CrossRef] [PubMed]
- Van Kruijsdijk, R.C.M.; van der Wall, E.; Visseren, F.L.J. Obesity and Cancer: The Role of Dysfunctional Adipose Tissue. Cancer Epidemiol. Biomark. Prev. 2009, 18, 2569–2578. [Google Scholar] [CrossRef] [PubMed]
- Clemente-Postigo, M.; Queipo-Ortuño, M.I.; Fernandez-Garcia, D.; Gomez-Huelgas, R.; Tinahones, F.J.; Cardona, F. Adipose Tissue Gene Expression of Factors Related to Lipid Processing in Obesity. PLoS ONE 2011, 6, e24783. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Z.G.; de Boer, I.H.; Mackey, R.H.; Jensen, M.K.; Lai, M.; Robson, S.C.; Tracy, R.; Kuller, L.H.; Mukamal, K.J. Associations of Insulin Resistance, Inflammation and Liver Synthetic Function with Very Low-density Lipoprotein: The Cardiovascular Health Study. Metabolism 2015, 65, 92–99. [Google Scholar] [CrossRef] [PubMed]
- Mao, H.; Lockyer, P.; Li, L.; Ballantyne, C.M.; Patterson, C.; Xie, L.; Pi, X. Endothelial LRP1 Regulates Metabolic Responses by Acting as a Co-Activator of PPARγ 3. Nat. Commun. 2017, 8, 14960. [Google Scholar] [CrossRef] [PubMed]
- Au, D.T.; Strickland, D.K.; Muratoglu, S.C. The LDL Receptor-Related Protein 1: At the Crossroads of Lipoprotein Metabolism and Insulin Signaling. J. Diabetes Res. 2017, 2017. [Google Scholar] [CrossRef] [PubMed]
- Goto, T.; Lee, J.-Y.; Teraminami, A.; Kim, Y.-I.; Hirai, S.; Uemura, T.; Inoue, H.; Takahashi, N.; Kawada, T. Activation of Peroxisome Proliferator-Activated Receptor-Alpha Stimulates Both Differentiation and Fatty Acid Oxidation in Adipocytes. J. Lipid Res. 2011, 52, 873–884. [Google Scholar] [CrossRef]
- Cawthorn, W.P.; Scheller, E.L.; MacDougald, O.A. Adipose Tissue Stem Cells Meet Preadipocyte Commitment: Going Back to the Future. J. Lipid Res. 2012, 53, 227–246. [Google Scholar] [CrossRef]
- Sarantopoulos, C.N.; Banyard, D.A.; Ziegler, M.E.; Sun, B.; Shaterian, A.; Widgerow, A.D. Elucidating the Preadipocyte and Its Role in Adipocyte Formation: A Comprehensive Review. Stem Cell Rev. Rep. 2018, 14, 27–42. [Google Scholar] [CrossRef]
- Landa, V.; Zidek, V.; Mlejnek, P.; Simakova, M.; Silhavy, J.; Trnovska, J.; Kazdova, L.; Pravenec, M. Sterol Regulatory Element Binding Protein 2 Overexpression Is Associated with Reduced Adipogenesis and Ectopic Fat Accumulation in Transgenic Spontaneously Hypertensive Rats. Physiol. Res. 2014, 63, 587–590. [Google Scholar]
- Madison, B.B. Srebp2: A Master Regulator of Sterol and Fatty Acid Synthesis. J. Lipid Res. 2016, 57, 333–335. [Google Scholar] [CrossRef] [PubMed]
- Yew Tan, C.; Virtue, S.; Murfitt, S.; Robert, L.D.; Phua, Y.H.; Dale, M.; Griffin, J.L.; Tinahones, F.; Scherer, P.E.; Vidal-Puig, A. Adipose Tissue Fatty Acid Chain Length and Mono-Unsaturation Increases with Obesity and Insulin Resistance. Sci. Rep. 2015, 5, 18366. [Google Scholar] [CrossRef] [PubMed]
- Ntambi, J.M.; Miyazaki, M.; Dobrzyn, A. Regulation of Stearoyl-CoA Desaturase Expression. Lipids 2004, 39, 1061–1065. [Google Scholar] [CrossRef] [PubMed]
- Hoang, M.H.; Jia, Y.; Mok, B.; Jun, H.J.; Hwang, K.Y.; Lee, S.J. Kaempferol Ameliorates Symptoms of Metabolic Syndrome by Regulating Activities of Liver X Receptor-β. J. Nutr. Biochem. 2015, 26, 868–875. [Google Scholar] [CrossRef] [PubMed]
- Faulds, M.H.; Zhao, C.; Dahlman-Wright, K. Molecular Biology and Functional Genomics of Liver X Receptors (LXR) in Relationship to Metabolic Diseases. Curr. Opin. Pharmacol. 2010, 10, 692–697. [Google Scholar] [CrossRef]
- Paz-Filho, G.; Mastronardi, C.A.; Licinio, J. Leptin Treatment: Facts and Expectations. Metabolism 2015, 64, 146–156. [Google Scholar] [CrossRef]
- La Cava, A. Leptin in Inflammation and Autoimmunity. Cytokine 2017, 98, 51–58. [Google Scholar] [CrossRef]
- Barbu, A.; Hamad, O.A.; Lind, L.; Ekdahl, K.N.; Nilsson, B. The Role of Complement Factor C3 in Lipid Metabolism. Mol. Immunol. 2015, 67, 101–107. [Google Scholar] [CrossRef]
- Castellano-Castillo, D.; Moreno-Indias, I.; Fernandez-Garcia, J.C.; Clemente-Postigo, M.; Castro-Cabezas, M.; Tinahones, F.J.; Queipo-Ortuño, M.I.; Cardona, F. Complement Factor C3 Methylation and MRNA Expression Is Associated to BMI and Insulin Resistance in Obesity. Genes 2018, 9, 410. [Google Scholar] [CrossRef]
- Palacios-Ortega, S.; Varela-Guruceaga, M.; Algarabel, M.; Milagro, F.I.; Martínez, J.A.; De Miguel, C. Effect of TNF-Alpha on Caveolin-1 Expression and Insulin Signaling during Adipocyte Differentiation and in Mature Adipocytes. Cell. Physiol. Biochem. 2015, 36, 1499–1516. [Google Scholar] [CrossRef]
- Srikanthan, K.; Feyh, A.; Visweshwar, H.; Shapiro, J.I.; Sodhi, K. Systematic Review of Metabolic Syndrome Biomarkers: A Panel for Early Detection, Management, and Risk Stratification in the West Virginian Population. Int. J. Med. Sci. 2016, 13, 25–38. [Google Scholar] [CrossRef] [PubMed]
- Perfilyev, A.; Dahlman, I.; Gillberg, L.; Rosqvist, F.; Iggman, D.; Volkov, P.; Nilsson, E.; Risérus, U.; Ling, C. Impact of Polyunsaturated and Saturated Fat Overfeeding on the DNA-Methylation Pattern in Human Adipose Tissue: A Randomized Controlled Trial. Am. J. Clin. Nutr. 2017, 105, 991–1000. [Google Scholar] [CrossRef] [PubMed]
- Matthews, D.R.; Hosker, J.P.; Rudenski, A.S.; Naylor, B.A.; Treacher, D.F.; Turner, R.C. Homeostasis Model Assessment: Insulin Resistance and Beta-Cell Function from Fasting Plasma Glucose and Insulin Concentrations in Man. Diabetologia 1985, 28, 412–419. [Google Scholar] [CrossRef]
- Daskalos, A.; Nikolaidis, G.; Xinarianos, G.; Savvari, P.; Cassidy, A.; Zakopoulou, R.; Kotsinas, A.; Gorgoulis, V.; Field, J.K.; Liloglou, T. Hypomethylation of Retrotransposable Elements Correlates with Genomic Instability in Non-Small Cell Lung Cancer. Int. J. Cancer 2009, 124, 81–87. [Google Scholar] [CrossRef] [PubMed]
- Li, L.-C.; Dahiya, R. MethPrimer: Designing Primers for Methylation PCRs. Bioinformatics 2002, 18, 1427–1431. [Google Scholar] [CrossRef] [PubMed]
- Panzeri, I.; Pospisilik, J.A. Epigenetic Control of Variation and Stochasticity in Metabolic Disease. Mol. Metab. 2018, 14, 26. [Google Scholar] [CrossRef] [PubMed]
- Davegårdh, C.; García-Calzón, S.; Bacos, K.; Ling, C. DNA Methylation in the Pathogenesis of Type 2 Diabetes in Humans. Mol. Metab. 2018, 14, 12–25. [Google Scholar] [CrossRef] [PubMed]
- Donkin, I.; Barrès, R. Sperm Epigenetics and Influence of Environmental Factors. Mol. Metab. 2018, 14, 1–11. [Google Scholar] [CrossRef]
- Yang, A.S. A Simple Method for Estimating Global DNA Methylation Using Bisulfite PCR of Repetitive DNA Elements. Nucleic Acids Res. 2004, 32, e38. [Google Scholar] [CrossRef]
- Martín-Núñez, G.M.; Rubio-Martín, E.; Cabrera-Mulero, R.; Rojo-Martínez, G.; Olveira, G.; Valdés, S.; Soriguer, F.; Castaño, L.; Morcillo, S. Type 2 Diabetes Mellitus in Relation to Global LINE-1 DNA Methylation in Peripheral Blood: A Cohort Study. Epigenetics 2014, 9, 1322–1328. [Google Scholar] [CrossRef]
- Ohashi, K.; Munetsuna, E.; Yamada, H.; Ando, Y.; Yamazaki, M.; Taromaru, N.; Nagura, A.; Ishikawa, H.; Suzuki, K.; Teradaira, R.; et al. High Fructose Consumption Induces DNA Methylation at PPARα and CPT1A Promoter Regions in the Rat Liver. Biochem. Biophys. Res. Commun. 2015, 468, 185–189. [Google Scholar] [CrossRef] [PubMed]
- Shiomi, Y.; Yamauchi, T.; Iwabu, M.; Okada-Iwabu, M.; Nakayama, R.; Orikawa, Y.; Yoshioka, Y.; Tanaka, K.; Ueki, K.; Kadowaki, T. A Novel Peroxisome Proliferator-Activated Receptor (PPAR)α Agonist and PPARy Antagonist, Z-551, Ameliorates High-Fat Diet-Induced Obesity and Metabolic Disorders in Mice. J. Biol. Chem. 2015, 290, 14567–14581. [Google Scholar] [CrossRef] [PubMed]
- Walczak, R.; Tontonoz, P. PPARadigms and PPARadoxes: Expanding Roles for PPARγ in the Control of Lipid Metabolism. J. Lipid Res. 2002, 43, 177–186. [Google Scholar] [PubMed]
- Soriguer, F.; Morcillo, S.; Cardona, F.; Rojo-martı, G. Pro12Ala Polymorphism of the PPARG2 Gene Is Associated with Type 2 Diabetes Mellitus and Peripheral Insulin Sensitivity in a Population with a High Intake of Oleic Acid. J. Nutr. 2006, 136, 2325–2330. [Google Scholar] [CrossRef] [PubMed]
- Fujiki, K.; Kano, F.; Shiota, K.; Murata, M. Expression of the Peroxisome Proliferator Activated Receptor Gamma Gene Is Repressed by DNA Methylation in Visceral Adipose Tissue of Mouse Models of Diabetes. BMC Biol. 2009, 7, 38. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Z.-H.; Fan, Y.-C.; Zhao, Q.; Dou, C.-Y.; Ji, X.-F.; Zhao, J.; Gao, S.; Li, X.-Y.; Wang, K. Promoter Methylation Status and Expression of Ppar-γ Gene Are Associated with Prognosis of Acute-on-Chronic Hepatitis B Liver Failure. Clin. Epigenet. 2015, 7, 115. [Google Scholar] [CrossRef] [PubMed]
- Sabatino, L.; Fucci, A.; Pancione, M.; Colantuoni, V. PPARG Epigenetic Deregulation and Its Role in Colorectal Tumorigenesis. PPAR Res. 2012, 2012, 687492. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Q.; Fan, Y.C.; Zhao, J.; Gao, S.; Zhao, Z.H.; Wang, K. DNA Methylation Patterns of Peroxisome Proliferator-Activated Receptor Gamma Gene Associated with Liver Fibrosis and Inflammation in Chronic Hepatitis B. J. Viral Hepat. 2013, 20, 430–437. [Google Scholar] [CrossRef]
- Wells, R.A.; Chan, L.S.A. Cross-Talk between PPARs and the Partners of RXR: A Molecular Perspective. PPAR Res. 2009, 2009. [Google Scholar] [CrossRef]
- Godfrey, K.M.; Sheppard, A.; Gluckman, P.D.; Lillycrop, K.A.; Burdge, G.C.; Mclean, C.; Rodford, J.; Slater-jefferies, J.L.; Garratt, E.; Crozier, S.R.; et al. Epigenetic Gene Promoter Methylation at Birth Is Associated with Child’s Later Adiposity. Diabetes 2011, 60, 1528–1534. [Google Scholar] [CrossRef]
- Rashid, S.; GENEST, J. Effect of Obesity on High-Density Lipoprotein Metabolism. Obesity 2007, 15, 2875–2888. [Google Scholar] [CrossRef] [PubMed]
- Masson, O.; Chavey, C.; Dray, C.; Meulle, A.; Daviaud, D.; Quilliot, D.; Muller, C.; Valet, P.; Liaudet-Coopman, E. LRP1 Receptor Controls Adipogenesis and Is Up-Regulated in Human and Mouse Obese Adipose Tissue. PLoS ONE 2009, 4, e7422. [Google Scholar] [CrossRef] [PubMed]
- Terrand, J.; Bruban, V.; Zhou, L.; Gong, W.; El Asmar, Z.; May, P.; Zurhove, K.; Haffner, P.; Philippe, C.; Woldt, E.; et al. LRP1 Controls Intracellular Cholesterol Storage and Fatty Acid Synthesis through Modulation of Wnt Signaling. J. Biol. Chem. 2009, 284, 381–388. [Google Scholar] [CrossRef] [PubMed]
- Macdonald, M.L.E.; Singaraja, R.R.; Bissada, N.; Ruddle, P.; Karasinska, J.M.; Gibson, W.T.; Fievet, C.; Vance, J.E.; Staels, B.; Hayden, M.R. Absence of Stearoyl-CoA Desaturase-1 Ameliorates Features of the Metabolic Syndrome in LDLR-Deficient Mice. J. Lipid Res. 2016, 49, 217–229. [Google Scholar] [CrossRef] [PubMed]
- Martín-Núñez, G.M.; Cabrera-Mulero, R.; Rubio-Martín, E.; Rojo-Martínez, G.; Olveira, G.; Valdés, S.; Soriguer, F.; Castaño, L.; Morcillo, S. Methylation Levels of the SCD1 Gene Promoter and LINE-1 Repeat Region Are Associated with Weight Change: An Intervention Study. Mol. Nutr. Food Res. 2014, 58, 1528–1536. [Google Scholar] [CrossRef] [PubMed]
- Morcillo, S.; Martín-Núñez, G.M.; García-Serrano, S.; Gutierrez-Repiso, C.; Rodriguez-Pacheco, F.; Valdes, S.; Gonzalo, M.; Rojo-Martinez, G.; Moreno-Ruiz, F.J.; Rodriguez-Cañete, A.; et al. Changes in SCD Gene DNA Methylation after Bariatric Surgery in Morbidly Obese Patients Are Associated with Free Fatty Acids. Sci. Rep. 2017, 7, 46292. [Google Scholar] [CrossRef] [PubMed]
- Reilly, S.M.; Saltiel, A.R. Adapting to Obesity with Adipose Tissue Inflammation. Nat. Rev. Endocrinol. 2017, 13, 633–643. [Google Scholar] [CrossRef]
- Zhang, J.; Wang, C.; Ha, X.; Li, W.; Xu, P.; Gu, Y.; Wang, T.; Wang, Y.; Xie, J. DNA Methylation of Tumor Necrosis Factor-α, Monocyte Chemoattractant Protein-1, and Adiponectin Genes in Visceral Adipose Tissue Is Related to Type 2 Diabetes in the Xinjiang Uygur Population. J. Diabetes 2017, 9, 699–706. [Google Scholar] [CrossRef]
- Cordero, P.; Campion, J.; Milagro, F.I.; Goyenechea, E.; Steemburgo, T.; Javierre, B.M.; Martinez, J.A. Leptin and TNF-Alpha Promoter Methylation Levels Measured by MSP Could Predict the Response to a Low-Calorie Diet. J. Physiol. Biochem. 2011, 67, 463–470. [Google Scholar] [CrossRef]
- Moreno-indias, I.; Oliva-olivera, W.; Omiste, A.; Castellano-castillo, D.; Lhamyani, S.; Camargo, A.; Tinahones, F.J. Adipose Tissue Infiltration in Normal-Weight Subjects and Its Impact on Metabolic Function. Transl. Res. 2016, 172, 6.e3–17.e3. [Google Scholar] [CrossRef]
- Wang, X.; Cao, Q.; Yu, L.; Shi, H.; Xue, B.; Shi, H. Epigenetic Regulation of Macrophage Polarization and Inflammation by DNA Methylation in Obesity. JCI Insight 2016, 1, e87748. [Google Scholar] [CrossRef] [PubMed]
- Cawthorn, W.P.; Sethi, J.K. TNF-α and Adipocyte Biology. FEBS Lett. 2008, 582, 117–131. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Wang, X.; Liu, D.; Yu, L.; Xue, B.; Shi, H. Epigenetic Regulation of Macrophage Polarization by DNA Methyltransferase 3b. Mol. Endocrinol. 2014, 28, 565–574. [Google Scholar] [CrossRef] [PubMed]
- Zhou, D.; Yang, K.; Chen, L.; Zhang, W.; Xu, Z.; Zuo, J.; Jiang, H.; Luan, J. Promising Landscape for Regulating Macrophage Polarization: Epigenetic Viewpoint. Oncotarget 2015, 8, 57693–57706. [Google Scholar] [CrossRef] [PubMed]
- Baardman, J.; Licht, I.; de Winther, M.P.; Van den, B.J. Metabolic–Epigenetic Crosstalk in Macrophage Activation. Epigenomics 2015, 7, 1155–1164. [Google Scholar] [CrossRef] [PubMed]
- Iikuni, N.; Lam, Q.L.K.; Lu, L.; Matarese, G.; La Cava, A. Leptin and Inflammation. Curr. Immunol. Rev. 2008, 4, 70–79. [Google Scholar] [CrossRef] [PubMed]
- Beltowski, J. Role of Leptin in Blood Pressure Regulation and Arterial Hypertension. J. Hypertens. 2006, 24, 789–801. [Google Scholar] [CrossRef]
- Simonds, S.E.; Pryor, J.T.; Ravussin, E.; Greenway, F.L.; Dileone, R.; Allen, A.M.; Bassi, J.; Elmquist, J.K.; Keogh, J.M.; Henning, E.; et al. Leptin Mediates the Increase in Blood Pressure Associated with Obesity. Cell 2014, 159, 1404–1416. [Google Scholar] [CrossRef]
- O’Rourke, L.; Yeaman, S.J.; Shepherd, P.R. Insulin and Leptin Acutely Regulate Cholesterol Ester Metabolism in Macrophages by Novel Signaling Pathways. Diabetes 2001, 50, 955–961. [Google Scholar] [CrossRef]
- Houde, A.A.; Légaré, C.; Biron, S.; Lescelleur, O.; Biertho, L.; Marceau, S.; Tchernof, A.; Vohl, M.C.; Hivert, M.F.; Bouchard, L. Leptin and Adiponectin DNA Methylation Levels in Adipose Tissues and Blood Cells Are Associated with BMI, Waist Girth and LDL-Cholesterol Levels in Severely Obese Men and Women. BMC Med. Genet. 2015, 16, 29. [Google Scholar] [CrossRef]
| Non MetS (n = 55) | MetS (n = 53) | |
|---|---|---|
| Age (years) | 48.4 ± 13.9 | 52.7 ± 14.6 |
| Male/female (%) | 52/48 | 44/56 |
| BMI (Kg/m2) ** | 29.8 ± 7.9 | 36.4 ± 10.9 |
| WC (cm) ** | 97.6 ± 14.8 | 112.6 ± 22.4 |
| Glucose (mg/dL) ** | 94.3 ± 11.6 | 118.4 ± 29.5 |
| Insulin (pmol/L) ** | 9.8 ± 7.4 | 16.2 ± 11.4 |
| HOMA-IR ** | 2.3 ± 1.9 | 4.7 ± 3.4 |
| TG (mg/dL) ** | 101.6 ± 38.1 | 164.2 ± 65.1 |
| Cholesterol (mg/dL) ** | 194.0 ± 32.5 | 214.5 ± 41.3 |
| HDL-c (mg/dL) ** | 55.0 ± 11.0 | 48.5 ± 14.2 |
| LDL-c (Friedwald) * | 119.0 ± 31.8 | 135.1 ± 30.2 |
| ApoA1 (mg/dL) | 171.6 ± 21.8 | 160.5 ± 29.5 |
| ApoB (mg/dL) ** | 91.9 ± 22.3 | 108.7 ± 22.3 |
| SBP (mm Hg) ** | 123.5 ± 17.8 | 139.8 ± 19.5 |
| DBP (mm Hg) ** | 76.1 ± 11.2 | 82.7 ± 10.3 |
| GOT (mg/dL) | 20.0 ± 13.1 | 19.3 ± 8.7 |
| GPT (mg/dL) | 40.3 ± 23.9 | 44.6 ± 21.5 |
| GGT (mg/dL) | 57.4 ± 203.4 | 42.1 ± 27.9 |
| Uric acid (mg/dL) ** | 4.6 ± 1.2 | 5.6 ± 1.2 |
| Leptin (ng/mL) ** | 18.9 ± 23.8 | 38.1 ± 30.5 |
| Adiponectin (μg/mL) * | 11.2 ± 5.3 | 8.2 ± 4.1 |
| Non MetS | MetS | |
|---|---|---|
| LINE-1 P1 (%) | 74.15 ± 0.39 | 74.37 ± 0.34 |
| LINE-1 P2 (%) | 65.84 ± 0.20 | 65.75 ± 0.33 |
| LINE-1 P3 (%) | 55.21 ± 0.29 | 55.29 ± 0.29 |
| LINE-1 P4 (%) | 61.37 ± 0.33 | 61.48 ± 0.24 |
| LINE-1 P5 (%) | 65.02 ± 0.17 | 65.24 ± 0.21 |
| LINE-1 P6 (%) | 65.05 ± 0.48 | 64.71 ± 0.23 |
| MetS Index | BMI | Waist | Glucose | Tg | HDL-c | LDL-c | SBP | DBP | HOMA-IR | |
|---|---|---|---|---|---|---|---|---|---|---|
| LINE-1 P1 | −0.167 | 0.057 | −0.031 | −0.246 * | −0.088 | 0.113 | 0.082 | 0.162 | 0.02 | −0.114 |
| LINE-1 P2 | −0.233 * | 0.025 | −0.068 | −0.334 ** | −0.208 | 0.074 | 0.028 | 0.171 | 0.010 | −0.199 |
| LINE-1 P3 | −0.136 | 0.018 | −0.011 | −0.168 | −0.072 | −0.115 | 0.093 | 0.220 | 0.155 | −0.101 |
| LINE-1 P4 | −0.068 | 0.042 | 0.012 | −0.158 | 0.039 | −0.112 | 0.077 | 0.168 | 0.010 | −0.041 |
| LINE-1 P5 | −0.137 | 0.093 | −0.037 | −0.238 * | 0.016 | −0.139 | 0.05 | 0.136 | 0.100 | −0.088 |
| LINE-1 P6 | −0.19 | −0.055 | −0.05 | −0.137 | −0.166 | 0.028 | 0.052 | 0.066 | 0.016 | −0.126 |
| MetS Index | BMI | Waist | Glucose | Tg | HDL-c | LDL-c | SBP | DBP | HOMA-IR | |
|---|---|---|---|---|---|---|---|---|---|---|
| PPARA P2 | 0.276 * | 0.076 | 0.165 | 0.166 | 0.392 ** | 0.061 | 0.08 | 0.066 | −0.025 | 0.229 * |
| PPARG P1 | −0.072 | 0.306 * | 0.169 | −0.224 | −0.194 | 0.015 | −0.197 | −0.2 | −0.293 * | −0.058 |
| PPARG P3 | −0.078 | 0.138 | 0.174 | 0.03 | −0.139 | 0.021 | −0.218 | 0.037 | −0.283 * | 0.112 |
| RXRA P1 | −0.102 | −0.298 ** | −0.229 * | −0.052 | 0.025 | −0.095 | 0.127 | −0.066 | −0.225 | −0.032 |
| SREBF2 P2 | 0.056 | 0.006 | 0.144 | 0.112 | 0.136 | −0.032 | 0.189 | −0.224 | −0.262 * | 0.121 |
| LRP1 P2 | 0.09 | −0.065 | −0.048 | 0.114 | −0.215 | 0.373 * | −0.055 | 0.192 | 0.180 | 0.251 |
| LPL P3 | 0.135 | 0.029 | 0.089 | 0.128 | 0.245 * | −0.102 | 0.085 | 0.126 | −0.111 | 0.149 |
| SCD P3 | −0.056 | −0.340 * | −0.283 | −0.096 | −0.018 | 0.108 | 0.22 | 0.087 | −0.117 | −0.03 |
| SCD P6 | −0.325 * | −0.116 | −0.17 | −0.141 | −0.134 | 0.121 | 0.102 | −0.275 | −0.232 | −0.172 |
| TNF P1 | −0.212 | 0.132 | 0.046 | −0.034 | −0.188 | 0.283 * | −0.02 | −0.010 | −0.115 | 0.029 |
| TNF P2 | −0.420 ** | 0.054 | −0.061 | −0.192 | −0.273 * | 0.304 * | −0.195 | −0.188 | −0.217 | −0.196 |
| TNF P3 | −0.320 * | 0.151 | −0.021 | −0.094 | −0.155 | 0.222 | −0.109 | −0.237 | −0.242 | −0.03 |
| TNF P4 | −0.330 * | −0.006 | −0.096 | −0.278 * | −0.203 | 0.098 | −0.295 * | −0.245 | −0.305 * | −0.133 |
| TNF P5 | −0.281 * | 0.132 | −0.100 | −0.153 | −0.281 * | 0.380 ** | −0.132 | −0.097 | −0.008 | −0.074 |
| LEP P1 | 0.088 | 0.081 | −0.159 | 0.061 | −0.071 | 0.015 | 0.229 * | 0.264 * | 0.230 * | 0.028 |
| Fasting triglycerides (R = 0.566; R2 = 0.320) | |||
|---|---|---|---|
| β | P | CI 95% | |
| Age | 0.111 | 0.425 | −0.582–1.358 |
| Gender | −0.268 | 0.047 | −48.377–(−0.312) |
| BMI | −0.101 | 0.446 | −1.825–0.816 |
| PPARA P2 | 0.332 | 0.012 | 1.32–10.012 |
| LPL P3 | 0.264 | 0.046 | 0.099–10.72 |
| TNF P2 | −0.117 | 0.347 | −1.867–0.669 |
| Non Mets/MetS (R2 = 0.506–0.686) | |||
|---|---|---|---|
| OR | p | CI 95% | |
| Gender | 5.813 | 0.094 | 0.739–45.699 |
| PPARA P2 | 1.630 | 0.246 | 0.714–3.719 |
| TNF P2 | 0.791 | 0.008 | 0.664–0.942 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Castellano-Castillo, D.; Moreno-Indias, I.; Sanchez-Alcoholado, L.; Ramos-Molina, B.; Alcaide-Torres, J.; Morcillo, S.; Ocaña-Wilhelmi, L.; Tinahones, F.; Queipo-Ortuño, M.I.; Cardona, F. Altered Adipose Tissue DNA Methylation Status in Metabolic Syndrome: Relationships Between Global DNA Methylation and Specific Methylation at Adipogenic, Lipid Metabolism and Inflammatory Candidate Genes and Metabolic Variables. J. Clin. Med. 2019, 8, 87. https://doi.org/10.3390/jcm8010087
Castellano-Castillo D, Moreno-Indias I, Sanchez-Alcoholado L, Ramos-Molina B, Alcaide-Torres J, Morcillo S, Ocaña-Wilhelmi L, Tinahones F, Queipo-Ortuño MI, Cardona F. Altered Adipose Tissue DNA Methylation Status in Metabolic Syndrome: Relationships Between Global DNA Methylation and Specific Methylation at Adipogenic, Lipid Metabolism and Inflammatory Candidate Genes and Metabolic Variables. Journal of Clinical Medicine. 2019; 8(1):87. https://doi.org/10.3390/jcm8010087
Chicago/Turabian StyleCastellano-Castillo, Daniel, Isabel Moreno-Indias, Lidia Sanchez-Alcoholado, Bruno Ramos-Molina, Juan Alcaide-Torres, Sonsoles Morcillo, Luis Ocaña-Wilhelmi, Francisco Tinahones, María Isabel Queipo-Ortuño, and Fernando Cardona. 2019. "Altered Adipose Tissue DNA Methylation Status in Metabolic Syndrome: Relationships Between Global DNA Methylation and Specific Methylation at Adipogenic, Lipid Metabolism and Inflammatory Candidate Genes and Metabolic Variables" Journal of Clinical Medicine 8, no. 1: 87. https://doi.org/10.3390/jcm8010087
APA StyleCastellano-Castillo, D., Moreno-Indias, I., Sanchez-Alcoholado, L., Ramos-Molina, B., Alcaide-Torres, J., Morcillo, S., Ocaña-Wilhelmi, L., Tinahones, F., Queipo-Ortuño, M. I., & Cardona, F. (2019). Altered Adipose Tissue DNA Methylation Status in Metabolic Syndrome: Relationships Between Global DNA Methylation and Specific Methylation at Adipogenic, Lipid Metabolism and Inflammatory Candidate Genes and Metabolic Variables. Journal of Clinical Medicine, 8(1), 87. https://doi.org/10.3390/jcm8010087







