Testosterone Therapy, Thrombophilia, Venous Thromboembolism, and Thrombotic Events
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Entry Criteria
2.2. Study Design, Cases, and Controls
2.3. Statistical Analysis
2.4. Laboratory Measures of Thrombophilia and Hypofibrinolysis
2.4.1. PCR Assays
2.4.2. Serologic Measures of Thrombophilia and Hypofibrinolysis
3. Results
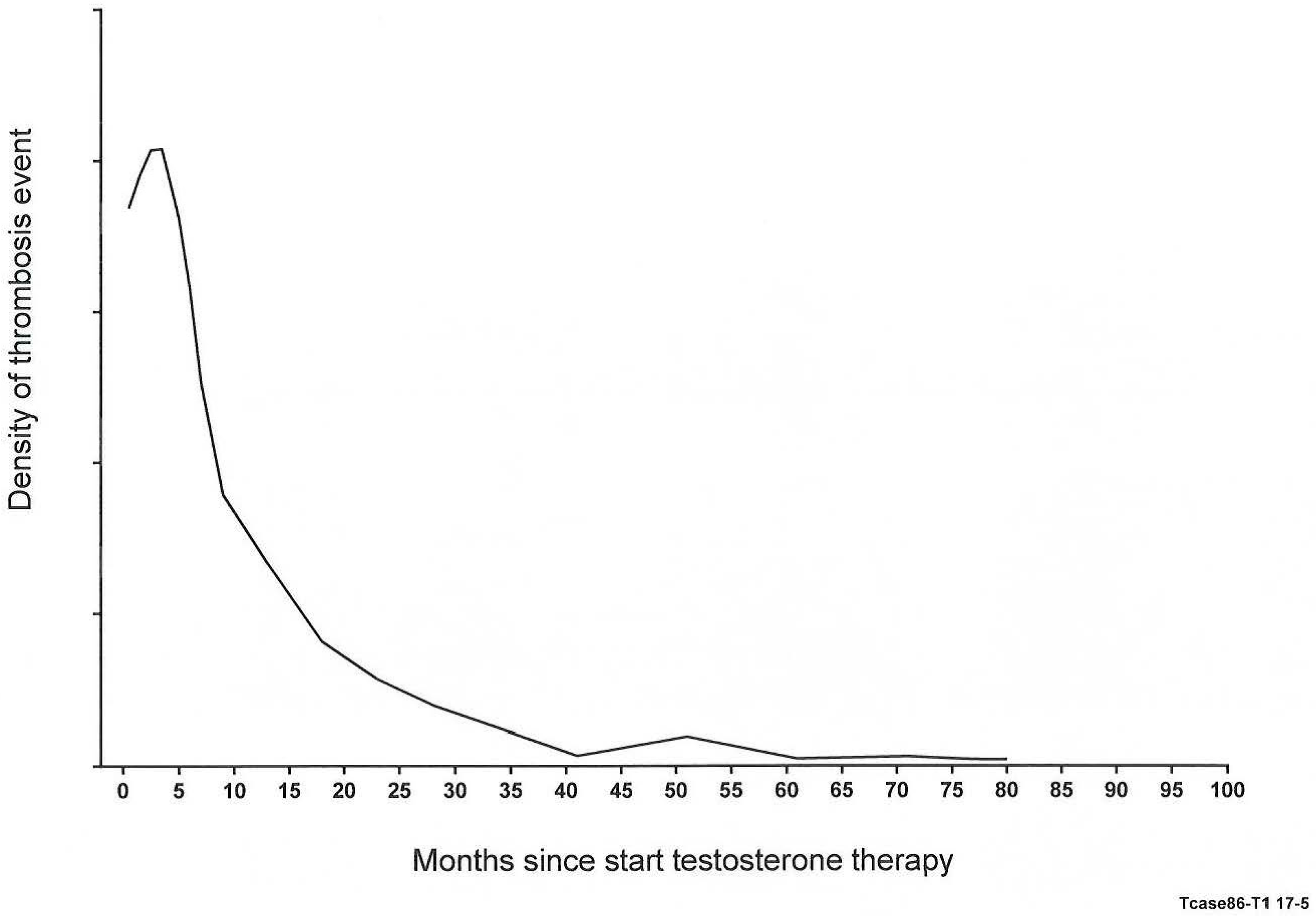
3.1. Sixty-Seven Patients with Thrombotic Events Six Months (Median) after Starting Testosterone Therapy
3.2. Twenty-One Men with Thrombotic Events Six Months (Median) after Starting Testosterone Therapy
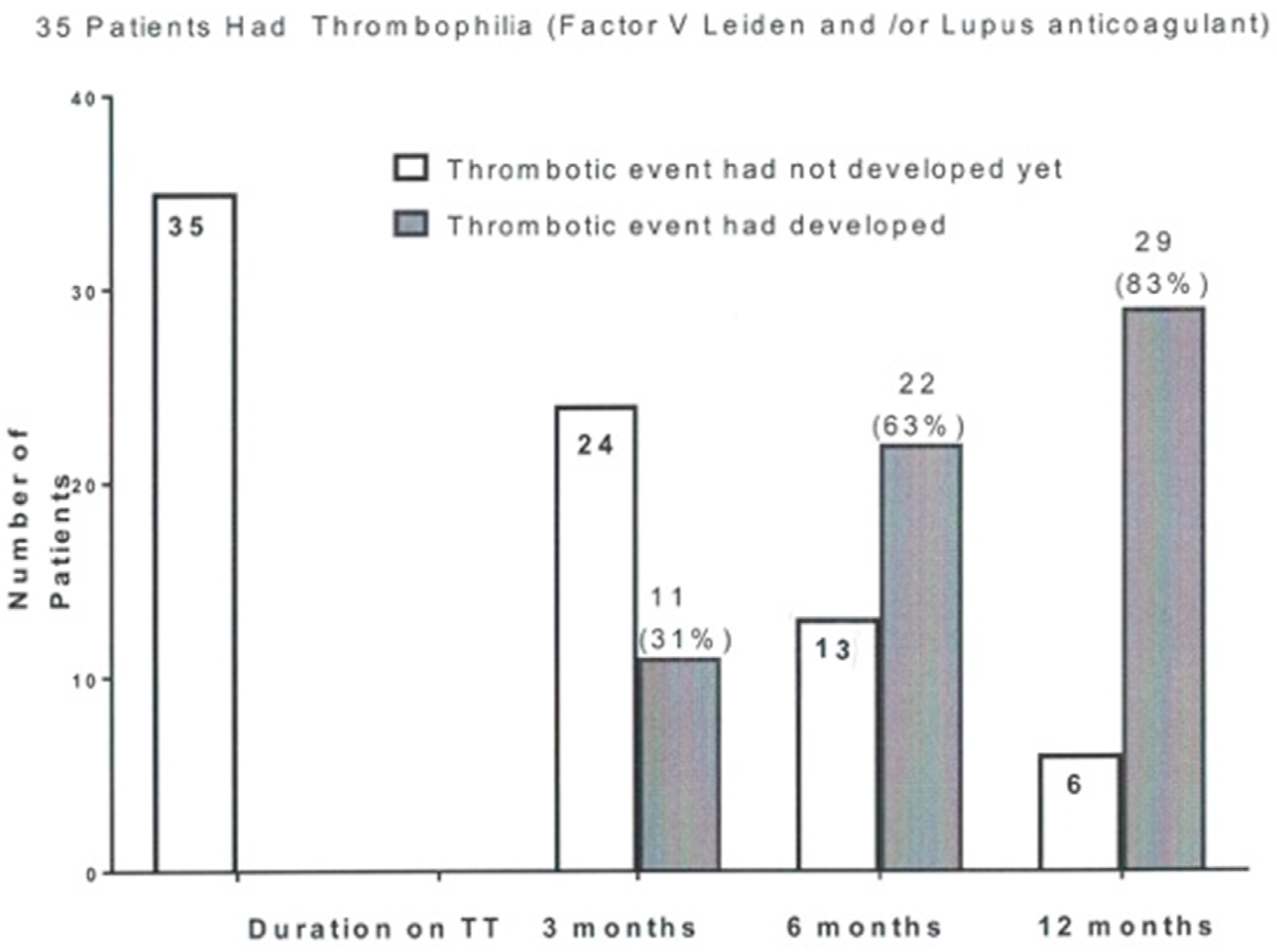
3.3. Pooled Analysis of 88 Cases
3.4. Recurrent Thrombotic Events when Testosterone Therapy Is Continued after a First Event, Despite Adequate Anticoagulation
3.5. Thrombotic Events in Nine Women at a Median of Three Months after Starting Testosterone Therapy
3.6. Osteonecrosis Developing at a Median of Six Months after Starting Testosterone Therapy in 16 Patients
3.7. Thrombotic Events in Six Patients with Klinefelter Syndrome
4. Discussion
5. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
Abbreviations
| TT | testosterone therapy |
| VTE | venous thromboembolism |
| FVL | Factor V Leiden |
| PE | pulmonary embolism |
References
- Glueck, C.J.; Goldenberg, N.; Budhani, S.; Lotner, D.; Abuchaibe, C.; Gowda, M.; Nayar, T.; Khan, N.; Wang, P. Thrombotic events after starting exogenous testosterone in men with previously undiagnosed familial thrombophilia. Transl. Res. 2011, 158, 225–234. [Google Scholar] [CrossRef]
- Food and Drug Administration UFaD. Testosterone Products: FDA/CDER Statement-Risk of Venous Blood Clots. 2014. Available online: http://www.safetyalertregistry.com/alerts/2558 (accessed on 20 December 2018).
- Health Canada. Information Update: POSSIBLE Cardiovascular Problems Associated with Testosterone Products. 2014. Available online: http://healthycanadians.gc.ca/recall-alert-rappel-avis/hc-sc/2014/40587a-eng.php/ (accessed on 20 December 2018).
- FDA. FDA DRUG SAFETY COMMUNICATION: FDA Cautions about Using Testosterone Products for Low Testosterone due to Aging; Required Labeling Change to Inform of Possible Increased Risk of Heart Attack and Stroke with Use; FDA: Silver Spring, MD, USA, 2016. [Google Scholar]
- Nguyen, C.P.; Hirsch, M.S.; Moeny, D.; Kaul, S.; Mohamoud, M.; Joffe, H.V. Testosterone and “Age-Related Hypogonadism”—FDA Concerns. N. Engl. J. Med. 2015, 373, 689–691. [Google Scholar] [CrossRef] [PubMed]
- Sogaard, K.K.; Schmidt, M.; Pedersen, L.; Horvath-Puho, E.; Sorensen, H.T. 30-year mortality after venous thromboembolism: A population-based cohort study. Circulation 2014, 130, 829–836. [Google Scholar] [CrossRef] [PubMed]
- Barco, S.; Woersching, A.L.; Spyropoulos, A.C.; Piovella, F.; Mahan, C.E. European Union-28: An annualised cost-of-illness model for venous thromboembolism. Thromb. Haemost. 2016, 115, 800–808. [Google Scholar] [CrossRef] [PubMed]
- Braekkan, S.K.; Grosse, S.D.; Okoroh, E.M.; Tsai, J.; Cannegieter, S.C.; Naess, I.A.; Krokstad, S.; Hansen, J.B.; Skjeldestad, F.E. Venous thromboembolism and subsequent permanent work-related disability. J. Thromb. Haemost. 2016, 14, 1978–1987. [Google Scholar] [CrossRef] [PubMed]
- Snyder, P.J.; Bhasin, S.; Cunningham, G.R.; Matsumoto, A.M.; Stephens-Shields, A.J.; Cauley, J.A.; Gill, T.M.; Barrett-Connor, E.; Swerdloff, R.S.; Wang, C.; et al. Effects of Testosterone Treatment in Older Men. N. Engl. J. Med. 2016, 374, 611–624. [Google Scholar] [CrossRef] [PubMed]
- Budoff, M.J.; Ellenberg, S.S.; Lewis, C.E.; Mohler, E.R., 3rd; Wenger, N.K.; Bhasin, S.; Barrett-Connor, E.; Swerdloff, R.S.; Stephens-Shields, A.; Cauley, J.A.; et al. Testosterone Treatment and Coronary Artery Plaque Volume in Older Men With Low Testosterone. JAMA 2017, 317, 708–716. [Google Scholar] [CrossRef]
- Resnick, S.M.; Matsumoto, A.M.; Stephens-Shields, A.J.; Ellenberg, S.S.; Gill, T.M.; Shumaker, S.A.; Pleasants, D.D.; Barrett-Connor, E.; Bhasin, S.; Cauley, J.A.; et al. Testosterone Treatment and Cognitive Function in Older Men With Low Testosterone and Age-Associated Memory Impairment. JAMA 2017, 317, 717–727. [Google Scholar] [CrossRef]
- Snyder, P.J.; Kopperdahl, D.L.; Stephens-Shields, A.J.; Ellenberg, S.S.; Cauley, J.A.; Ensrud, K.E.; Lewis, C.E.; Barrett-Connor, E.; Schwartz, A.V.; Lee, D.C.; et al. Effect of Testosterone Treatment on Volumetric Bone Density and Strength in Older Men With Low Testosterone: A Controlled Clinical Trial. JAMA Intern. Med. 2017, 177, 471–479. [Google Scholar] [CrossRef]
- Roy, C.N.; Snyder, P.J.; Stephens-Shields, A.J.; Artz, A.S.; Bhasin, S.; Cohen, H.J.; Farrar, J.T.; Gill, T.M.; Zeldow, B.; Cella, D.; et al. Association of Testosterone Levels with Anemia in Older Men: A Controlled Clinical Trial. JAMA Intern. Med. 2017, 177, 480–490. [Google Scholar] [CrossRef]
- Rossouw, J.E.; Anderson, G.L.; Prentice, R.L.; LaCroix, A.Z.; Kooperberg, C.; Stefanick, M.L.; Jackson, R.D.; Beresford, S.A.; Howard, B.V.; Johnson, K.C.; et al. Risks and benefits of estrogen plus progestin in healthy postmenopausal women: Principal results From the Women’s Health Initiative randomized controlled trial. JAMA 2002, 288, 321–333. [Google Scholar] [PubMed]
- Glueck, C.J.; Prince, M.; Patel, N.; Patel, J.; Shah, P.; Mehta, N.; Wang, P. Thrombophilia in 67 Patients With Thrombotic Events After Starting Testosterone Therapy. Clin. Appl. Thromb. Hemost. 2016, 22, 548–553. [Google Scholar] [CrossRef] [PubMed]
- Glueck, C.J.; Goldenberg, N.; Wang, P. Thromboembolism peaking 3 months after starting testosterone therapy: Testosterone-thrombophilia interactions. J. Investig. Med. 2018, 66, 733–738. [Google Scholar] [CrossRef] [PubMed]
- Glueck, C.J.; Jetty, V.; Goldenberg, N.; Shah, P.; Wang, P. Thrombophilia in Klinefelter Syndrome with Deep Venous Thrombosis, Pulmonary Embolism, and Mesenteric Artery Thrombosis on Testosterone Therapy: A Pilot Study. Clin. Appl. Thromb. Hemost. 2017, 23, 973–979. [Google Scholar] [CrossRef] [PubMed]
- Glueck, C.J.; Riaz, R.; Prince, M.; Freiberg, R.A.; Wang, P. Testosterone Therapy Can Interact with Thrombophilia, Leading to Osteonecrosis. Orthopedics 2015, 38, e1073–e1078. [Google Scholar] [CrossRef] [PubMed]
- Canada Health. Summary Safety Review-Testosterone Replacement Products and Cardiovascular Risk. Canada Health, 2014. Available online: https://regulatorydoctor.us/wp-content/uploads/2014/11/Testosterone-Replacement-Products-Cardiovascular-Risk.pdf (accessed on 20 December 2018).
- Valdiviezo, C.; Lawson, S.; Ouyang, P. An update on menopausal hormone replacement therapy in women and cardiovascular disease. Curr. Opin. Endocrinol. Diabetes Obes. 2013, 20, 148–155. [Google Scholar] [CrossRef] [PubMed]
- Glueck, C.J.; Freiberg, R.A.; Boriel, G.; Khan, Z.; Brar, A.; Padda, J.; Wang, P. The Role of the Factor V Leiden Mutation in Osteonecrosis of the Hip. Clin. Appl. Thromb. Hemost. 2012, 19, 499–503. [Google Scholar] [CrossRef] [PubMed]
- Glueck, C.J.; Freiberg, R.A.; Wang, P. Heritable thrombophilia–hypofibrinolysis and osteonecrosis of the femoral head. Clin. Orthop. Relat. Res. 2008, 466, 1034–1040. [Google Scholar] [CrossRef]
- den Heijer, M.; Koster, T.; Blom, H.J.; Bos, G.M.; Briet, E.; Reitsma, P.H.; Vandenbroucke, J.P.; Rosendaal, F.R. Hyperhomocysteinemia as a risk factor for deep-vein thrombosis. N. Engl. J. Med. 1996, 334, 759–762. [Google Scholar] [CrossRef]
- Brandt, J.T.; Triplett, D.A.; Alving, B.; Scharrer, I. Criteria for the diagnosis of lupus anticoagulants: An update. On behalf of the Subcommittee on Lupus Anticoagulant/Antiphospholipid Antibody of the Scientific and Standardisation Committee of the ISTH. Thromb. Haemost. 1995, 74, 1185–1190. [Google Scholar] [CrossRef]
- Bick, R.L. Antiphospholipid thrombosis syndromes. Clin. Appl. Thromb. Hemost. 2001, 7, 241–258. [Google Scholar] [CrossRef] [PubMed]
- Greco, T.P.; Amos, M.D.; Conti-Kelly, A.M.; Naranjo, J.D.; Ijdo, J.W. Testing for the antiphospholipid syndrome: Importance of IgA anti-beta 2-glycoprotein I. Lupus 2000, 9, 33–41. [Google Scholar] [CrossRef] [PubMed]
- Glueck, C.J.; Freiberg, R.A.; Boppana, S.; Wang, P. Thrombophilia, hypofibrinolysis, the eNOS T-786C polymorphism, and multifocal osteonecrosis. J. Bone Jt. Surg. Am. 2008, 90, 2220–2229. [Google Scholar] [CrossRef] [PubMed]
- Douketis, J.D.; Julian, J.A.; Crowther, M.A.; Kearon, C.; Bates, S.M.; Barone, M.; Piovella, F.; Middeldorp, S.; Prandoni, P.; Johnston, M.; et al. The effect of prothrombotic blood abnormalities on risk of deep vein thrombosis in users of hormone replacement therapy: A prospective case-control study. Clin. Appl. Thromb. Hemost. 2011, 17, E106–E113. [Google Scholar] [CrossRef] [PubMed]
- Gadelha, T.; Roldan, V.; Lecumberri, R.; Trujillo-Santos, J.; del Campo, R.; Poggio, R.; Monreal, M.; RIETE Investigators. Clinical characteristics of patients with factor V Leiden or prothrombin G20210A and a first episode of venous thromboembolism. Findings from the RIETE Registry. Thromb. Res. 2010, 126, 283–286. [Google Scholar] [CrossRef] [PubMed]
- Turan, O.; Undar, B.; Gunay, T.; Akkoclu, A. Investigation of inherited thrombophilias in patients with pulmonary embolism. Blood Coagul. Fibrinolysis. 2013, 24, 140–149. [Google Scholar] [CrossRef] [PubMed]
- Martinez, C.; Suissa, S.; Rietbrock, S.; Katholing, A.; Freedman, B.; Cohen, A.T.; Handelsman, D.J. Testosterone treatment and risk of venous thromboembolism: POPULATION based case-control study. BMJ 2016, 355, i5968. [Google Scholar] [CrossRef]
- Miettinen, O.S.; Caro, J.J. Principles of nonexperimental assessment of excess risk, with special reference to adverse drug reactions. J. Clin. Epidemiol. 1989, 42, 325–331. [Google Scholar] [CrossRef]
- Baillargeon, J.; Urban, R.J.; Morgentaler, A.; Glueck, C.J.; Baillargeon, G.; Sharma, G.; Kuo, Y.F. Risk of Venous Thromboembolism in Men Receiving Testosterone Therapy. Mayo Clin. Proc. 2015, 90, 1038–1045. [Google Scholar] [CrossRef] [PubMed]
- Finkle, W.D.; Greenland, S.; Ridgeway, G.K.; Adams, J.L.; Frasco, M.A.; Cook, M.B.; Fraumeni, J.F., Jr.; Hoover, R.N. Increased risk of non-fatal myocardial infarction following testosterone therapy prescription in men. PLoS ONE 2014, 9, e85805. [Google Scholar] [CrossRef]
- Basaria, S.; Coviello, A.D.; Travison, T.G.; Storer, T.W.; Farwell, W.R.; Jette, A.M.; Eder, R.; Tennstedt, S.; Ulloor, J.; Zhang, A.; et al. Adverse events associated with testosterone administration. N. Engl. J. Med. 2010, 363, 109–122. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Freeman, G.; Cowling, B.J.; Schooling, C.M. Testosterone therapy and cardiovascular events among men: A systematic review and meta-analysis of placebo-controlled randomized trials. BMC Med. 2013, 11, 108. [Google Scholar] [CrossRef] [PubMed]
- Vigen, R.; O’Donnell, C.I.; Baron, A.E.; Grunwald, G.K.; Maddox, T.M.; Bradley, S.M.; Barqawi, A.; Woning, G.; Wierman, M.E.; Plomondon, M.E.; et al. Association of testosterone therapy with mortality, myocardial infarction, and stroke in men with low testosterone levels. JAMA 2013, 310, 1829–1836. [Google Scholar] [CrossRef] [PubMed]
- Horwitz, H.; Andersen, J.T.; Dalhoff, K.P. Health consequences of androgenic anabolic steroid use. J. Intern. Med. 2018. [Google Scholar] [CrossRef] [PubMed]
- Colburn, S.; Childers, W.K.; Chacon, A.; Swailes, A.; Ahmed, F.M.; Sahi, R. The cost of seeking an edge: Recurrent renal infarction in setting of recreational use of anabolic steroids. Ann. Med. Surg. 2017, 14, 25–28. [Google Scholar] [CrossRef] [PubMed]
- Glueck, C.J.; Wang, P. Testosterone therapy, thrombosis, thrombophilia, cardiovascular events. Metabolism 2014, 63, 989–994. [Google Scholar] [CrossRef] [PubMed]
- Freedman, J.; Glueck, C.J.; Prince, M.; Riaz, R.; Wang, P. Testosterone, thrombophilia, thrombosis. Transl. Res. 2015, 165, 537–548. [Google Scholar] [CrossRef]
- Reed, B.G.; Bou Nemer, L.; Carr, B.R. Has testosterone passed the test in premenopausal women with low libido? A systematic review. Int. J. Womens Health 2016, 8, 599–607. [Google Scholar] [CrossRef]
- Meyer, P. [Testosterone therapy in female hypoactive sexual desire disorder]. Rev. Med. Suisse 2016, 12, 540–543. [Google Scholar]
- Cappelletti, M.; Wallen, K. Increasing women’s sexual desire: The comparative effectiveness of estrogens and androgens. Horm. Behav. 2016, 78, 178–193. [Google Scholar] [CrossRef]
- Davis, S.; Papalia, M.A.; Norman, R.J.; O’Neill, S.; Redelman, M.; Williamson, M.; Stuckey, B.G.; Wlodarczyk, J.; Gard’ner, K.; Humberstone, A. Safety and efficacy of a testosterone metered-dose transdermal spray for treating decreased sexual satisfaction in premenopausal women: A randomized trial. Ann. Intern. Med. 2008, 148, 569–577. [Google Scholar] [CrossRef] [PubMed]
- Davis, S.R.; Moreau, M.; Kroll, R.; Bouchard, C.; Panay, N.; Gass, M.; Braunstein, G.D.; Hirschberg, A.L.; Rodenberg, C.; Pack, S.; et al. Testosterone for low libido in postmenopausal women not taking estrogen. N. Engl. J. Med. 2008, 359, 2005–2017. [Google Scholar] [CrossRef] [PubMed]
- Reis, S.L.; Abdo, C.H. Benefits and risks of testosterone treatment for hypoactive sexual desire disorder in women: A critical review of studies published in the decades preceding and succeeding the advent of phosphodiesterase type 5 inhibitors. Clinics 2014, 69, 294–303. [Google Scholar] [CrossRef]
- Glueck, C.J.; Bowe, D.; Valdez, A.; Wang, P. Thrombosis in three postmenopausal women receiving testosterone therapy for low libido. Womens Health 2013, 9, 405–410. [Google Scholar] [CrossRef] [PubMed]
- Bjorkman, A.; Burtscher, I.M.; Svensson, P.J.; Hillarp, A.; Besjakov, J.; Benoni, G. Factor V Leiden and the prothrombin 20210A gene mutation and osteonecrosis of the knee. Arch. Orthop. Trauma Surg. 2005, 125, 51–55. [Google Scholar] [CrossRef]
- Jarman, M.I.; Lee, K.; Kanevsky, A.; Min, S.; Schlam, I.; Mahida, C.; Huda, A.; Milgrom, A.; Goldenberg, N.; Glueck, C.J.; et al. Case report: PRIMARY osteonecrosis associated with thrombophilia–hypofibrinolysis and worsened by testosterone therapy. BMC Hematol. 2017, 17, 5. [Google Scholar] [CrossRef] [PubMed]
- Glueck, C.J.; Freiberg, R.A.; Sieve, L.; Wang, P. Enoxaparin prevents progression of stages I and II osteonecrosis of the hip. Clin. Orthop. Relat. Res. 2005, 164–170. [Google Scholar] [CrossRef]
- Glueck, C.J.; Freiberg, R.A.; Wang, P. Medical treatment of osteonecrosis of the knee associated with thrombophilia–hypofibrinolysis. Orthopedics 2014, 37, e911–e916. [Google Scholar] [CrossRef]
- Glueck, C.J.; Freiberg, R.A.; Wissman, R.; Wang, P. Long term anticoagulation (4–16 years) stops progression of idiopathic hip osteonecrosis associated with familial thrombophilia. Adv. Orthop. 2015, 2015, 138382. [Google Scholar] [CrossRef]
- Zoller, B.; Ji, J.; Sundquist, J.; Sundquist, K. High Risk of Venous Thromboembolism in Klinefelter Syndrome. J. Am. Heart Assoc. 2016, 5. [Google Scholar] [CrossRef]
- Corona, G.; Dicuio, M.; Rastrelli, G.; Maseroli, E.; Lotti, F.; Sforza, A.; Maggi, M. Testosterone treatment and cardiovascular and venous thromboembolism risk: What is ‘new’? J. Investig. Med. 2017, 65, 964–973. [Google Scholar] [CrossRef] [PubMed]
- Blanco-Molina, A.; Monreal, M. Venous thromboembolism in women taking hormonal contraceptives. Expert Rev. Cardiovasc. Ther. 2010, 8, 211–215. [Google Scholar] [CrossRef] [PubMed]
- Wu, O.; Robertson, L.; Twaddle, S.; Lowe, G.; Clark, P.; Walker, I.; Langhorne, P.; Brenkel, I.; Regan, L.; Greer, I. Screening for thrombophilia in high-risk situations: A meta-analysis and cost-effectiveness analysis. Br. J. Haematol. 2005, 131, 80–90. [Google Scholar] [CrossRef] [PubMed]
- Wu, O.; Robertson, L.; Twaddle, S.; Lowe, G.D.; Clark, P.; Greaves, M.; Walker, I.D.; Langhorne, P.; Brenkel, I.; Regan, L.; et al. Screening for thrombophilia in high-risk situations: Systematic review and cost-effectiveness analysis. The Thrombosis: Risk and Economic Assessment of Thrombophilia Screening (TREATS) study. Health Technol. Assess. 2006, 10, 1–110. [Google Scholar] [CrossRef] [PubMed]
- Pezzini, A.; Grassi, M.; Iacoviello, L.; Del Zotto, E.; Archetti, S.; Giossi, A.; Padovani, A. Inherited thrombophilia and stratification of ischaemic stroke risk among users of oral contraceptives. J. Neurol. Neurosurg. Psychiatry 2007, 78, 271–276. [Google Scholar] [CrossRef]
- Grosse, S.D.; Nelson, R.E.; Nyarko, K.A.; Richardson, L.C.; Raskob, G.E. The economic burden of incident venous thromboembolism in the United States: A review of estimated attributable healthcare costs. Thromb. Res. 2016, 137, 3–10. [Google Scholar] [CrossRef]
- Kyrle, P.A.; Rosendaal, F.R.; Eichinger, S. Risk assessment for recurrent venous thrombosis. Lancet 2010, 376, 2032–2039. [Google Scholar] [CrossRef]
- Suissa, S.; Blais, L.; Spitzer, W.O.; Cusson, J.; Lewis, M.; Heinemann, L. First-time use of newer oral contraceptives and the risk of venous thromboembolism. Contraception 1997, 56, 141–146. [Google Scholar] [CrossRef]
- Li, H.; Benoit, K.; Wang, W.; Motsko, S. Association between Use of Exogenous Testosterone Therapy and Risk of Venous Thrombotic Events among Exogenous Testosterone Treated and Untreated Men with Hypogonadism. J. Urol. 2016, 195, 1065–1072. [Google Scholar] [CrossRef]
- Ramasamy, R.; Scovell, J.; Mederos, M.; Ren, R.; Jain, L.; Lipshultz, L. Association Between Testosterone Supplementation Therapy and Thrombotic Events in Elderly Men. Urology 2015, 86, 283–285. [Google Scholar] [CrossRef]
- Sharma, R.; Oni, O.A.; Chen, G.; Sharma, M.; Dawn, B.; Sharma, R.; Parashara, D.; Savin, V.J.; Barua, R.S.; Gupta, K. Association Between Testosterone Replacement Therapy and the Incidence of DVT and Pulmonary Embolism: A Retrospective Cohort Study of the Veterans Administration Database. Chest 2016, 150, 563–571. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Schooling, C.M. Differential risks in men and women for first and recurrent venous thrombosis: The role of genes and environment: Comment. J. Thromb. Haemost. 2015, 13, 884–886. [Google Scholar] [CrossRef] [PubMed]
- Houghton, D.E.; Alsawas, M.; Barrioneuvo, P.; Tello, M.; Farah, W.; Beuschel, B.; Prokop, L.J.; Layton, J.B.; Murad, M.H.; Moll, S. Testosterone therapy and venous thromboembolism: A systematic review and meta-analysis. Thromb. Res. 2018, 172, 94–103. [Google Scholar] [CrossRef] [PubMed]
- Onasanya, O.; Iyer, G.; Lucas, E.; Lin, D.; Singh, S.; Alexander, G.C. Association between exogenous testosterone and cardiovascular events: An overview of systematic reviews. Lancet Diabetes Endocrinol. 2016, 4, 943–956. [Google Scholar] [CrossRef]
| Normal Range | Mean ± SD | Low | Normal | High | |
|---|---|---|---|---|---|
| Before TT | |||||
| T (ng/dL) | 280–800 | 227 ± 124 | 66% | 34% | 0% |
| Free T (pg/mL) | 7.2–24 | 7.5 ± 7.6 | 67% | 30% | 4% |
| E2 (pg/mL) | ≤42.6 | 25.6 ± 11.9 | 87% | 13% | |
| On TT | |||||
| T (ng/dL) | 280–800 | 431 ± 301 | 28% | 62% | 10 |
| Free T (pg/mL) | 7.2–24 | 17.7 ±17.2 | 36% | 43% | 21% |
| E2 (pg/mL) | ≤42.6 | 30.3 ± 13.7 | 76% | 24% | |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Glueck, C.J.; Goldenberg, N.; Wang, P. Testosterone Therapy, Thrombophilia, Venous Thromboembolism, and Thrombotic Events. J. Clin. Med. 2019, 8, 11. https://doi.org/10.3390/jcm8010011
Glueck CJ, Goldenberg N, Wang P. Testosterone Therapy, Thrombophilia, Venous Thromboembolism, and Thrombotic Events. Journal of Clinical Medicine. 2019; 8(1):11. https://doi.org/10.3390/jcm8010011
Chicago/Turabian StyleGlueck, Charles J., Naila Goldenberg, and Ping Wang. 2019. "Testosterone Therapy, Thrombophilia, Venous Thromboembolism, and Thrombotic Events" Journal of Clinical Medicine 8, no. 1: 11. https://doi.org/10.3390/jcm8010011
APA StyleGlueck, C. J., Goldenberg, N., & Wang, P. (2019). Testosterone Therapy, Thrombophilia, Venous Thromboembolism, and Thrombotic Events. Journal of Clinical Medicine, 8(1), 11. https://doi.org/10.3390/jcm8010011





