Technical Developments and Clinical Use of Telemedicine in Sleep Medicine
Abstract
:1. Introduction
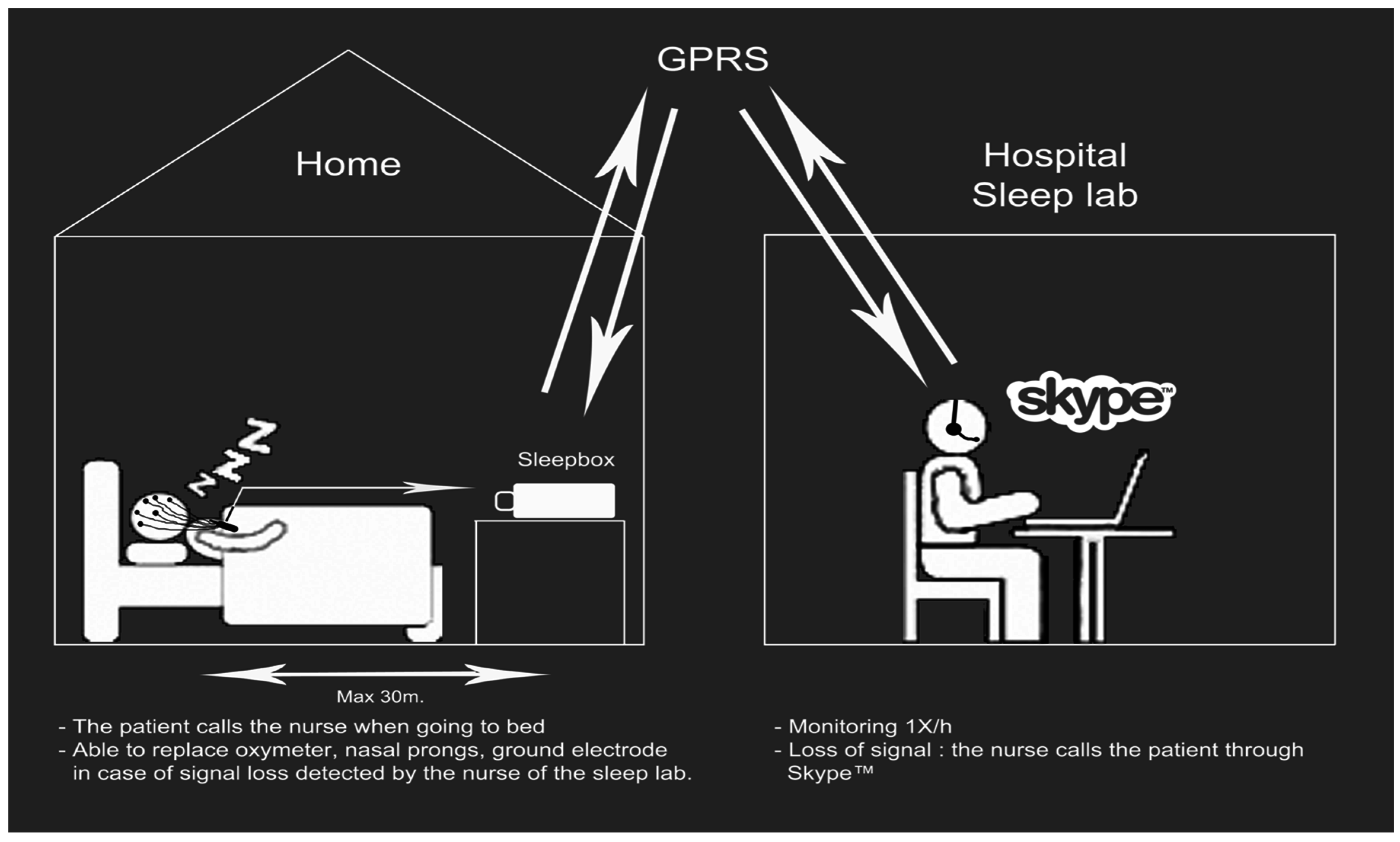
2. Telediagnostics for Obstructive Sleep Apnea Syndrome
3. Teleconsultation for Obstructive Sleep Apnea Syndrome
4. Teletherapy with CPAP for Obstructive Sleep Apnea Syndrome
5. Telemonitoring of CPAP-Treated Obstructive Sleep Apnea Syndrome Patients
6. Discussion
7. Conclusions
Acknowledgments
Conflicts of Interest
References
- Flodgren, G.; Rachas, A.; Farmer, A.J.; Inzitari, M.; Shepperd, S. Interactive telemedicine: Effects on professional practice and health care outcomes. Cochrane Library 2015. [Google Scholar]
- Zia, S.; Field, B.G. Sleep Telemedicine: An Emerging Field’s Latest Frontier. Chest 2016, 149, 1556–1565. [Google Scholar] [CrossRef] [PubMed]
- American Academy of Sleep Medicine. The International Classification of Sleep Disorders, 3rd ed.; American Academy of Sleep Medicine: Darien, IL, USA, 2014. [Google Scholar]
- Peppard, P.E.; Young, T.; Barnet, J.H.; Palta, M.; Hagen, E.W.; Hla, K.M. Increased prevalence of sleep-disordered breathing in adults. Am. J. Epidemiol. 2013, 177, 1006–1014. [Google Scholar] [CrossRef] [PubMed]
- Collop, N.A.; Anderson, W.M.; Boehlecke, B.; Claman, D.; Goldberg, R.; Gottlieb, D.J.; Hudgel, D.; Sateia, M.; Schwab, R. Clinical guidelines for the use of unattended portable monitors in the diagnosis of obstructive sleep apnea in adult patients. Portable Monitoring Task Force of the American Academy of Sleep Medicine. J. Clin. Sleep Med. 2007, 3, 737–747. [Google Scholar] [PubMed]
- Masa, J.F.; Corral, J.; Pereira, R.; Duran-Cantolla, J.; Cabello, M.; Hernandez-Blasco, L.; Monasterio, C.; Alonso, A.; Chiner, E.; Rubio, M.; et al. Effectiveness of home respiratory polygraphy for the diagnosis of sleep apnea and hypopnoea syndrome. Thorax 2011, 66, 567–573. [Google Scholar] [CrossRef] [PubMed]
- El Shayeb, M.; Topfer, L.A.; Stafinski, T.; Pawluk, L.; Menon, D. Diagnostic accuracy of level 3 portable sleep tests versus level 1 polysomnography for sleep-disordered breathing: A systematic review and meta-analysis. Can. Med. Association J. 2014, 186, E25–E51. [Google Scholar] [CrossRef] [PubMed]
- Bruyneel, M.; Ninane, V. Unattended home-based polysomnography for sleep disordered breathing: Current concepts and perspectives. Sleep Med. Rev. 2014, 18, 341–347. [Google Scholar] [CrossRef] [PubMed]
- Gagnadoux, F.; Pelletier-Fleury, N.; Philippe, C.; Rakotonanahary, D.; Fleury, B. Home unattended vs. hospital telemonitored polysomnography in suspected obstructive sleep apnea syndrome: A randomized crossover trial. Chest 2002, 121, 753–758. [Google Scholar] [CrossRef] [PubMed]
- Pelletier-Fleury, N.; Gagnadoux, F.; Philippe, C.; Rakotonanahary, D.; Lanoe, J.L.; Fleury, B. A cost-minimization study of telemedicine. The case of telemonitored polysomnography to diagnose obstructive sleep apnea syndrome. Int. J. Technol. Assess. Health Care 2001, 17, 604–611. [Google Scholar] [PubMed]
- Bruyneel, M.; Van den Broecke, S.; Libert, W.; Ninane, V. Real-time attended home-polysomnography with telematic data transmission. Int. J. Med. Inform. 2013, 82, 696–701. [Google Scholar] [CrossRef] [PubMed]
- Van den Broecke, S.; Jobard, O.; Montalescot, G.; Bruyneel, M.; Ninane, V.; Arnulf, I.; Similowski, T.; Attali, V. Very early screening for sleep-disordered breathing in acute coronary syndrome in patients without acute heart failure. Sleep Med. 2014, 15, 1539–1546. [Google Scholar] [CrossRef] [PubMed]
- Coma-Del-Corral, M.J.; Alonso-Alvarez, M.L.; Allende, M.; Cordero, J.; Ordax, E.; Masa, F.; Teran-Santos, J. Reliability of telemedicine in the diagnosis and treatment of sleep apnea syndrome. Telemed. J. Health 2013, 19, 7–12. [Google Scholar] [CrossRef] [PubMed]
- Isetta, V.; Leon, C.; Torres, M.; Embid, C.; Roca, J.; Navajas, D.; Farre, R.; Montserrat, J.M. Telemedicine-based approach for obstructive sleep apnea management: Building evidence. Interact. J. Med. Res. 2014, 3, e6. [Google Scholar] [CrossRef] [PubMed]
- Dellaca, R.; Montserrat, J.M.; Govoni, L.; Pedotti, A.; Navajas, D.; Farre, R. Telemetric CPAP titration at home in patients with sleep apnea-hypopnea syndrome. Sleep Med. 2011, 12, 153–157. [Google Scholar] [CrossRef] [PubMed]
- Kribbs, N.B.; Pack, A.I.; Kline, L.R.; Getsy, J.E.; Schuett, J.S.; Henry, J.N.; Maislin, G.; Dinges, D.F. Effects of one night without nasal CPAP treatment on sleep and sleepiness in patients with obstructive sleep apnea. Am. Rev. Respir. Dis. 1993, 147, 1162–1168. [Google Scholar] [CrossRef] [PubMed]
- Weaver, T.E.; Maislin, G.; Dinges, D.F.; Bloxham, T.; George, C.F.; Greenberg, H.; Kader, G.; Mahowald, M.; Younger, J.; Pack, A.I. Relationship between hours of CPAP use and achieving normal levels of sleepiness and daytime functioning. Sleep 2007, 30, 711–719. [Google Scholar] [PubMed]
- Barbe, F.; Duran-Cantolla, J.; Capote, F.; de la Pena, M.; Chiner, E.; Masa, J.F.; Gonzalez, M.; Marin, J.M.; Garcia-Rio, F.; de Atauri, J.D.; et al. Long-term effect of continuous positive airway pressure in hypertensive patients with sleep apnea. Am. J. Respir. Crit. Care Med. 2010, 181, 718–726. [Google Scholar] [CrossRef] [PubMed]
- Bouloukaki, I.; Giannadaki, K.; Mermigkis, C.; Tzanakis, N.; Mauroudi, E.; Moniaki, V.; Michelakis, S.; Siafakas, N.M.; Schiza, S.E. Intensive versus standard follow-up to improve continuous positive airway pressure compliance. Eur. Respir. J. 2014, 44, 1262–1274. [Google Scholar] [CrossRef] [PubMed]
- Aloia, M.S.; Arnedt, J.T.; Stanchina, M.; Millman, R.P. How early in treatment is PAP adherence established? Revisiting night-to-night variability. Behav. Sleep Med. 2007, 5, 229–240. [Google Scholar] [CrossRef] [PubMed]
- Reeves-Hoche, M.K.; Meck, R.; Zwillich, C.W. Nasal CPAP: An objective evaluation of patient compliance. Am. J. Respir. Crit. Care Med. 1994, 149, 149–154. [Google Scholar] [CrossRef] [PubMed]
- Rotenberg, B.W.; Murariu, D.; Pang, K.P. Trends in CPAP adherence over twenty years of data collection: A flattened curve. J. Otolaryngol. Head Neck Surg. 2016, 45, 43. [Google Scholar] [CrossRef] [PubMed]
- Gay, P.; Weaver, T.; Loube, D.; Iber, C. Evaluation of positive airway pressure treatment for sleep related breathing disorders in adults. Sleep 2006, 29, 381–401. [Google Scholar] [PubMed]
- Engleman, H.M.; Wild, M.R. Improving CPAP use by patients with the sleep apnea/hypopnoea syndrome (SAHS). Sleep Med. Rev. 2003, 7, 81–99. [Google Scholar] [CrossRef] [PubMed]
- Wozniak, D.R.; Lasserson, T.J.; Smith, I. Educational, supportive and behavioural interventions to improve usage of continuous positive airway pressure machines in adults with obstructive sleep apnea. Cochrane Library 2014. [Google Scholar]
- Taylor, Y.; Eliasson, A.; Andrada, T.; Kristo, D.; Howard, R. The role of telemedicine in CPAP compliance for patients with obstructive sleep apnea syndrome. Sleep Breath. 2006, 10, 132–138. [Google Scholar] [CrossRef] [PubMed]
- Stepnowsky, C.J.; Palau, J.J.; Marler, M.R.; Gifford, A.L. Pilot randomized trial of the effect of wireless telemonitoring on compliance and treatment efficacy in obstructive sleep apnea. J. Med. Internet. Res. 2007, 9, e14. [Google Scholar] [PubMed]
- Sparrow, D.; Aloia, M.; Demolles, D.A.; Gottlieb, D.J. A telemedicine intervention to improve adherence to continuous positive airway pressure: A randomised controlled trial. Thorax 2010, 65, 1061–1066. [Google Scholar] [CrossRef] [PubMed]
- Fox, N.; Hirsch-Allen, A.J.; Goodfellow, E.; Wenner, J.; Fleetham, J.; Ryan, C.F.; Kwiatkowska, M.; Ayas, N.T. The impact of a telemedicine monitoring system on positive airway pressure adherence in patients with obstructive sleep apnea. Sleep 2012, 35, 477–481. [Google Scholar] [PubMed]
- Isetta, V.; Negrin, M.A.; Monasterio, C.; Masa, J.F.; Feu, N.; Alvarez, A.; Campos-Rodriguez, F.; Ruiz, C.; Abad, J.; Vazquez-Polo, F.J.; et al. A Bayesian cost-effectiveness analysis of a telemedicine-based strategy for the management of sleep apnea: A multicentre randomised controlled trial. Thorax 2015, 70, 1054–1061. [Google Scholar] [CrossRef] [PubMed]
- Anttalainen, U.; Melkko, S.; Hakko, S.; Laitinen, T.; Saaresranta, T. Telemonitoring of CPAP therapy may save nursing time. Sleep Breath. 2016, 20, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Munafo, D.; Hevener, W.; Crocker, M.; Willes, L.; Sridasome, S.; Mushin, M. A telehealth program for CPAP adherence reduces labor and yields similar adherence and efficacity when compared to standard of care. Sleep Breath. 2016, 20, 777–785. [Google Scholar] [CrossRef] [PubMed]
- Frasnelli, M.; Baty, F.; Niederman, J.; Brutsche, M.H.; Schoch, O.D. Effect of telemetric monitoring in the first 30 days of continuous positive airway pressure adaptation for obstructive sleep apnoea syndrome—a controlled pilot study. J. Telemed. Telecare 2016, 22, 209–214. [Google Scholar] [CrossRef] [PubMed]
- Kuna, S.T.; Shuttleworth, D.; Chi, L.; Schutte-Rodin, S.; Friedman, E.; Guo, H.; Dhand, S.; Yang, L.; Zhu, J.; Bellamy, S.L.; et al. Web-Based Access to Positive Airway Pressure Usage with or without an Initial Financial Incentive Improves Treatment Use in Patients with Obstructive Sleep Apnea. Sleep 2015, 38, 1229–1236. [Google Scholar] [CrossRef] [PubMed]
- Rubin, M.N.; Wellik, K.E.; Channer, D.D.; Demaerschalk, B.M. Role of telemedicine in providing tertiary neurological care. Curr. Treat. Options Neurol. 2013, 15, 567–582. [Google Scholar] [CrossRef] [PubMed]
- Baig, M.M.; Antonescu-Turcu, A.; Ratarasarn, K. Impact of sleep telemedicine protocol in management of sleep apnea: A 5-year VA experience. Telemed. J. Health 2016, 22, 458–462. [Google Scholar] [CrossRef] [PubMed]
- Hirschkowitz, M.; Sharafkhaneh, A. A telemedicine program for diagnosis and management of sleep-disordered breathing: The fast-track for sleep apnea tele-sleep program. Semin. Respir. Crit. Care Med. 2014, 35, 560–570. [Google Scholar]
- Mulgrew, A.T.; Fox, N.; Ayas, N.T.; Ryan, C.F. Diagnosis and initial management of obstructive sleep apnea without polysomnography. Ann. Intern. Med. 2007, 146, 157–166. [Google Scholar] [CrossRef] [PubMed]
- Berry, R.B.; Hill, G.; Thompson, L.; McLaurin, V. Portable monitoring and autotitration versus polysomnography for the diagnosis and treatment of sleep apnea. Sleep 2008, 31, 1423–1431. [Google Scholar] [PubMed]
- Kuna, S.T.; Gurubhagavatula, I.; Maislin, G.; Hin, S.; Hartwig, K.C.; McCloskey, S.; Hachadoorian, R.; Hurley, S.; Gupta, R.; Staley, B.; Atwood, C.W. Noninferiority of functional outcome in ambulatory management of obstructive sleep apnea. Am. J. Respir. Crit. Care Med. 2011, 183, 1238–1244. [Google Scholar] [CrossRef] [PubMed]
- Rosen, C.L.; Auckley, D.; Benca, R.; Foldvary-Schaefer, N.; Iber, C.; Kapur, V.; Rueschman, M.; Zee, P.; Redline, S. A multisite randomized trial of portable sleep studies and positive airway pressure autotitration versus laboratory-based polysomnography for the diagnosis and treatment of obstructive sleep apnea: The HomePAP study. Sleep 2012, 35, 757–767. [Google Scholar] [CrossRef] [PubMed]
- Morgenthaler, T.I. Practice Parameters for the Use of Autotitrating Continuous Positive Airway Pressure Devices for Titrating Pressures and Treating Adult Patients with Obstructive Sleep Apnea Syndrome: An Update for 2007. Sleep 2008, 31, 141–147. [Google Scholar] [PubMed]
- Asteriou, C.; Lazopoulos, A.; Rallis, T.; Gogakos, A.S.; Paliouras, D.; Barbetakis, N. Fast-track rehabilitation following video-assisted pulmonary sublobar wedge resection: A prospective randomized study. J. Minim. Access Surg. 2016, 12, 209–213. [Google Scholar] [PubMed]
- Bahr, C. UNIversal solutions in TElemedicine Depolyment for European HEALTH care. Available online: http://united4health.eu/ (accessed on 11 Dec 2016).
- Chiron Health. State medical board regulations. Available online: http://chironhealth.com/telemedicine/regulations/ (accessed on 11 Dec 2016).
- Wechsler, L.R.; Tsao, J.W.; Levine, S.R.; Swain-Eng, R.J.; Adams, R.J.; Demaerschalk, B.M.; Hess, D.C.; Moro, E.; Schwamm, L.H.; Steffensen, S.; Stern, B.J.; et al. Teleneurology applications: Report of the Telemedicine Work Group of the American Academy of Neurology. Neurology 2013, 80, 670–676. [Google Scholar] [CrossRef] [PubMed]
| N = | AHI | TM Methods | Adherence TM vs. Usual Care | |
|---|---|---|---|---|
| Taylor et al. 2006 [26] | 114 | >5 | Internet support + feedback. | 1 month: 4.3 vs. 4.2 h/N (NS) |
| Stepnowsky et al. 2007 [27] | 45 | >15 | Interactive web-site with own CPAP data and guide for troubleshooting. | 2 months: 4.1 vs. 3.4 h/N (S) |
| Sparrow et al. 2010 [28] | 250 | >10 | Interactive voice response system (phone). | 6 months: 2.4 vs. 1.48 h/N (S) |
| Fox et al. 2012 [29] | 75 | >15, mean: 42 | Feedback by phone. | 3 months: 3.2 vs. 1.7 h/N (S) |
| Isetta et al. 2015 [30] | 139 | Mean: 49 | Feedback by web tools. | 6 months: 4.4 vs. 4.2 h/N (NS) |
| Anttalainen et al. 2016 [31] partially randomized | 111 | Mean: 34 | Nurse adjustment phone/visits. | 12 months: 6.4 vs. 6.1 h/N (NS) |
| Munafo et al. 2016 [32] | 132 | Mean: 34 (TM group), 27 (usual care) | Multimedia approach to contact patient about their CPAP use. | 1 month: 5.1 vs. 4.7 h/N (NS) |
| Frasnelli et al. 2016 [33] Patients selected at random | 223 | Median: 37 (TM group), 40 (usual care) | Feedback by phone. | 1 month: 5.3 vs. 4.6 h/N (S) |
© 2016 by the author; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bruyneel, M. Technical Developments and Clinical Use of Telemedicine in Sleep Medicine. J. Clin. Med. 2016, 5, 116. https://doi.org/10.3390/jcm5120116
Bruyneel M. Technical Developments and Clinical Use of Telemedicine in Sleep Medicine. Journal of Clinical Medicine. 2016; 5(12):116. https://doi.org/10.3390/jcm5120116
Chicago/Turabian StyleBruyneel, Marie. 2016. "Technical Developments and Clinical Use of Telemedicine in Sleep Medicine" Journal of Clinical Medicine 5, no. 12: 116. https://doi.org/10.3390/jcm5120116





