Proteinase-Activated Receptor 2 Is a Novel Regulator of TGF-β Signaling in Pancreatic Cancer
Abstract
:1. Pancreatic Ductal Adenocarcinoma and TGF-β Signaling
2. PAR2 and TGF-β Signaling Have Similar Functions: Circumstantial Evidence for a Functional Interaction
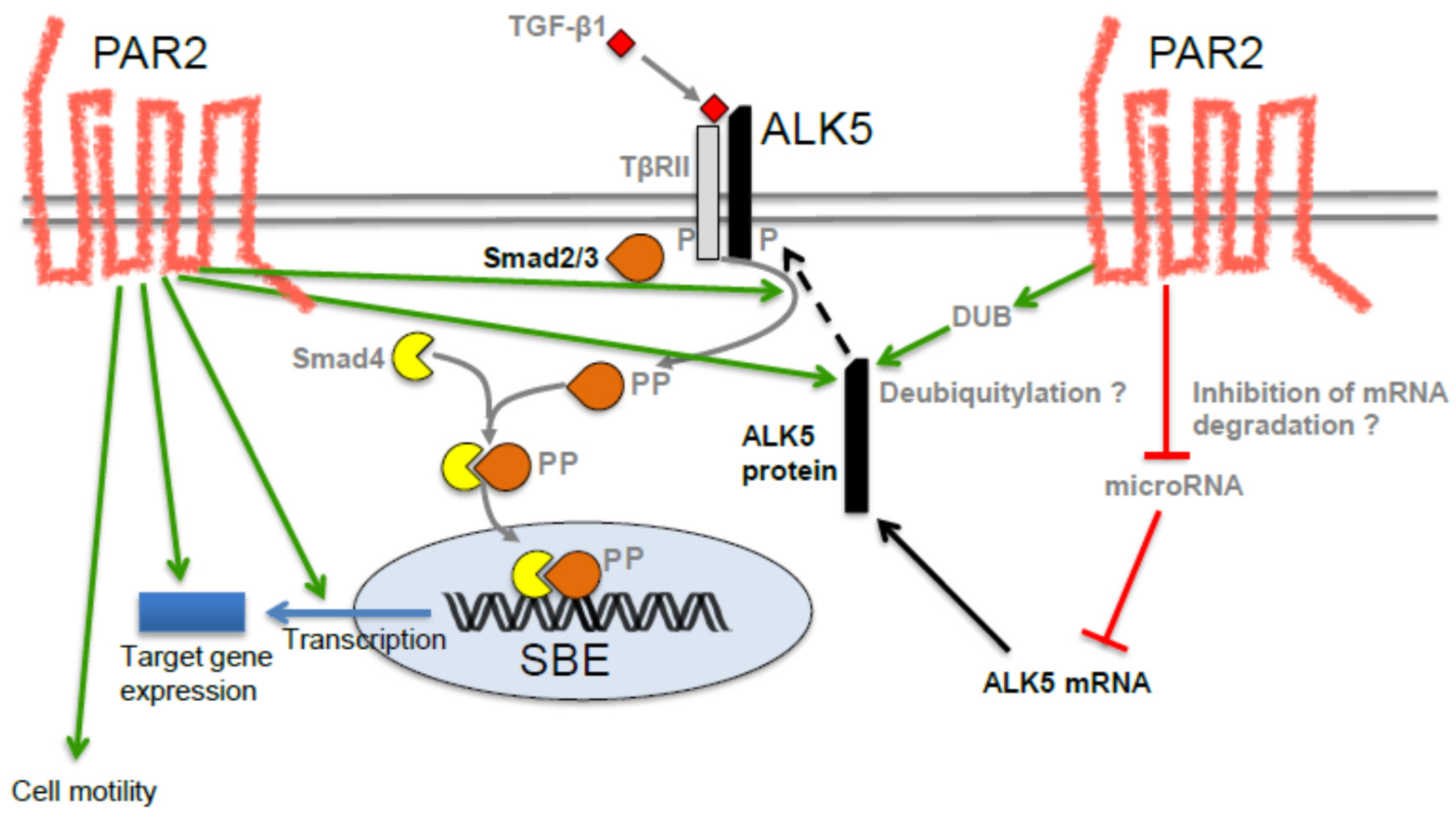
3. Possible Role of PAR2 in TGF-β-Induced EMT and EMT-Associated Alterations
4. The PAR2-TGF-β Crosstalk Also Operates in Non-Malignant Cells of Human and Murine Origin
5. Potential Therapeutic Implications of the TGF-β-PAR2 Interaction
6. Possible Involvement of PAR2 in Signal Transduction by Other TGF-β Family Members: Implications for a Role of PAR2 in Cancer-Related Cachexia
7. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Amundadottir, L.T. Pancreatic Cancer Genetics. Int. J. Biol. Sci. 2016, 12, 314–325. [Google Scholar] [CrossRef] [PubMed]
- Timp, J.F.; Braekkan, S.K.; Versteeg, H.H.; Cannegieter, S.C. Epidemiology of cancer-associated venous thrombosis. Blood 2013, 122, 1712–1723. [Google Scholar] [CrossRef] [PubMed]
- Mueller, T.C.; Burmeister, M.A.; Bachmann, J.; Martignoni, M.E. Cachexia and pancreatic cancer: Are there treatment options? World J. Gastroenterol. 2014, 20, 9361–9373. [Google Scholar] [PubMed]
- Yang, G.; Yang, X. Smad4-mediated TGF-beta signaling in tumorigenesis. Int. J. Biol. Sci. 2010, 6, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Jones, S.; Zhang, X.; Parsons, D.W.; Lin, J.C.; Leary, R.J.; Angenendt, P.; Mankoo, P.; Carter, H.; Kamiyama, H.; Jimeno, A.; et al. Core signaling pathways in human pancreatic cancers revealed by global genomic analyses. Science 2008, 321, 1801–1806. [Google Scholar] [CrossRef] [PubMed]
- Ijichi, H.; Chytil, A.; Gorska, A.E.; Aakre, M.E.; Fujitani, Y.; Fujitani, S.; Wright, C.V.; Moses, H.L. Aggressive pancreatic ductal adenocarcinoma in mice caused by pancreas-specific blockade of transforming growth factor-beta signaling in cooperation with active Kras expression. Genes Dev. 2006, 20, 3147–3160. [Google Scholar] [CrossRef] [PubMed]
- Hollenberg, M.D.; Mihara, K.; Polley, D.; Suen, J.Y.; Han, A.; Fairlie, D.P.; Ramachandran, R. Biased signalling and proteinase-activated receptors (PARs): Targeting inflammatory disease. Br. J. Pharmacol. 2014, 171, 1180–1194. [Google Scholar] [CrossRef] [PubMed]
- Gieseler, F.; Ungefroren, H.; Settmacher, U.; Hollenberg, M.D.; Kaufmann, R. Proteinase-activated receptors (PARs)—Focus on receptor-receptor-interactions and their physiological and pathophysiological impact. Cell Commun. Signal. 2013, 11, 86. [Google Scholar] [CrossRef] [PubMed]
- Nichols, H.L.; Saffeddine, M.; Theriot, B.S.; Hegde, A.; Polley, D.; El-Mays, T.; Vliagoftis, H.; Hollenberg, M.D.; Wilson, E.H.; Walker, J.K.; et al. β-Arrestin-2 mediates the proinflammatory effects of proteinase-activated receptor-2 in the airway. Proc. Natl. Acad. Sci. USA 2012, 109, 16660–16665. [Google Scholar] [CrossRef] [PubMed]
- Kawabata, A.; Matsunami, M.; Sekiguchi, F. Gastrointestinal roles for proteinase-activated receptors in health and disease. Br. J. Pharmacol. 2008, 153 (Suppl. 1), S230–S240. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Gao, J.; Zhao, T.; Wei, L.; Wu, W.; Bai, Y.; Zou, D.; Li, Z. Proteinase-activated receptor 2 mediates thermal hyperalgesia and is upregulated in a rat model of chronic pancreatitis. Pancreas 2011, 40, 300–307. [Google Scholar] [CrossRef] [PubMed]
- Knight, V.; Tchongue, J.; Lourensz, D.; Tipping, P.; Sievert, W. Protease-activated receptor 2 promotes experimental liver fibrosis in mice and activates human hepatic stellate cells. Hepatology 2012, 55, 879–887. [Google Scholar] [CrossRef] [PubMed]
- Timp, J.F.; Braekkan, S.K.; Versteeg, H.H.; Cannegieter, S.C. Epidemiology of cancer-associated venous thrombosis. Blood 2013, 122, 1712–1723. [Google Scholar] [CrossRef] [PubMed]
- Menapace, L.A.; Peterson, D.R.; Berry, A.; Sousou, T.; Khorana, A.A. Symptomatic and incidental thromboembolism are both associated with mortality in pancreatic cancer. Thromb. Haemost. 2011, 106, 371–378. [Google Scholar] [CrossRef] [PubMed]
- Saito, A.; Osuga, Y.; Yoshino, O.; Takamura, M.; Hirata, T.; Hirota, Y.; Koga, K.; Harada, M.; Takemura, Y.; Yano, T.; et al. TGF-β1 induces proteinase-activated receptor 2 (PAR2) expression in endometriotic stromal cells and stimulates PAR2 activation-induced secretion of IL-6. Hum. Reprod. 2011, 26, 1892–1898. [Google Scholar] [CrossRef] [PubMed]
- Kaufmann, R.; Oettel, C.; Horn, A.; Halbhuber, K.J.; Eitner, A.; Krieg, R.; Katenkamp, K.; Henklein, P.; Westermann, M.; Bohmer, F.D.; et al. Met receptor tyrosine kinase transactivation is involved in proteinase-activated receptor-2-mediated hepatocellular carcinoma cell invasion. Carcinogenesis 2009, 30, 1487–1496. [Google Scholar] [CrossRef] [PubMed]
- Shi, X.; Gangadharan, B.; Brass, L.F.; Ruf, W.; Mueller, B.M. Protease-activated receptors (PAR1 and PAR2) contribute to tumor cell motility and metastasis. Mol. Cancer Res. 2004, 2, 395–402. [Google Scholar] [PubMed]
- Versteeg, H.H.; Schaffner, F.; Kerver, M.; Ellies, L.G.; Andrade-Gordon, P.; Mueller, B.M.; Ruf, W. Protease-activated receptor (PAR) 2, but not PAR1, signaling promotes the development of mammary adenocarcinoma in polyoma middle T mice. Cancer Res. 2008, 68, 7219–7227. [Google Scholar] [CrossRef] [PubMed]
- Iwaki, K.; Shibata, K.; Ohta, M.; Endo, Y.; Uchida, H.; Tominaga, M.; Okunaga, R.; Kai, S.; Kitano, S. A small interfering RNA targeting proteinase-activated receptor-2 is effective in suppression of tumor growth in a Panc1 xenograft model. Int. J. Cancer 2008, 122, 658–663. [Google Scholar] [CrossRef] [PubMed]
- Segal, L.; Katz, L.S.; Lupu-Meiri, M.; Shapira, H.; Sandbank, J.; Gershengorn, M.C.; Oron, Y. Proteinase-activated receptors differentially modulate in vitro invasion of human pancreatic adenocarcinoma PANC1 cells in correlation with changes in the expression of CDC42 protein. Pancreas 2014, 43, 103–108. [Google Scholar] [CrossRef] [PubMed]
- Fujimoto, D.; Hirono, Y.; Goi, T.; Katayama, K.; Hirose, K.; Yamaguchi, A. Expression of protease activated receptor-2 (PAR-2) in gastric cancer. J. Surg. Oncol. 2006, 93, 139–144. [Google Scholar] [CrossRef] [PubMed]
- Olejar, T.; Vetvicka, D.; Zadinova, M.; Pouckova, P.; Kukal, J.; Jezek, P.; Matej, R. Dual role of host Par2 in a murine model of spontaneous metastatic B16 melanoma. Anticancer Res. 2014, 34, 3511–3515. [Google Scholar] [PubMed]
- Schniewind, B.; Groth, S.; Sebens Muerkoster, S.; Sipos, B.; Schafer, H.; Kalthoff, H.; Ungefroren, H. Dissecting the role of TGF-beta type I receptor/ALK5 in pancreatic ductal adenocarcinoma: Smad activation is crucial for both the tumor suppressive and prometastatic function. Oncogene 2007, 26, 4850–4862. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Yu, N.; Lee, C. Vicious cycle of TGF-β signaling in tumor progression and metastasis. Am. J. Clin. Exp. Urol. 2014, 2, 149–155. [Google Scholar] [PubMed]
- Ungefroren, H.; Sebens, S.; Seidl, D.; Lehnert, H.; Hass, R. Interaction of tumor cells with the microenvironment. Cell Commun. Signal. 2011, 9, 18. [Google Scholar] [CrossRef] [PubMed]
- Pickup, M.; Novitskiy, S.; Moses, H.L. The roles of TGFβ in the tumour microenvironment. Nat. Rev. Cancer 2013, 13, 788–799. [Google Scholar] [CrossRef] [PubMed]
- Chung, H.; Ramachandran, R.; Hollenberg, M.D.; Muruve, D.A. Proteinase-activated receptor-2 transactivation of epidermal growth factor receptor and transforming growth factor-β receptor signaling pathways contributes to renal fibrosis. J. Biol. Chem. 2013, 288, 37319–37331. [Google Scholar] [CrossRef] [PubMed]
- Zeeh, F.; Witte, D.; Gadeken, T.; Rauch, B.H.; Grage-Griebenow, E.; Leinung, N.; Fromm, S.J.; Stolting, S.; Mihara, K.; Kaufmann, R.; et al. Proteinase-activated receptor 2 promotes TGF-β-dependent cell motility in pancreatic cancer cells by sustaining expression of the TGF-β type I receptor ALK5. Oncotarget 2016, 7, 41095–41109. [Google Scholar] [CrossRef] [PubMed]
- Ungefroren, H.; UKSH, Lübeck, Germany. Unpublished work. 2016.
- Zheng, X.; Carstens, J.L.; Kim, J.; Scheible, M.; Kaye, J.; Sugimoto, H.; Wu, C.C.; LeBleu, V.S.; Kalluri, R. Epithelial-to-mesenchymal transition is dispensable for metastasis but induces chemoresistance in pancreatic cancer. Nature 2015, 527, 525–530. [Google Scholar] [CrossRef] [PubMed]
- Ungefroren, H.; UKSH, Lübeck, Germany. Unpublished work. 2015.
- Zhang, K.; Jiao, X.; Liu, X.; Zhang, B.; Wang, J.; Wang, Q.; Tao, Y.; Zhang, D. Knockdown of snail sensitizes pancreatic cancer cells to chemotherapeutic agents and irradiation. Int. J. Mol. Sci. 2010, 11, 4891–4892. [Google Scholar] [CrossRef] [PubMed]
- Radke, D.I.; Ungefroren, H.; Helm, O.; Voigt, S.; Alp, G.; Braun, H.; Hubner, S.; Dilchert, J.; Sebens, S.; Adam, D.; et al. Negative control of TRAIL-R1 signaling by transforming growth factor β1 in pancreatic tumor cells involves Smad-dependent down regulation of TRAIL-R1. Cell. Signal. 2016, 28, 1652–1662. [Google Scholar] [CrossRef] [PubMed]
- Du, S.; Barcellos-Hoff, M.H. Tumors as organs: Biologically augmenting radiation therapy by inhibiting transforming growth factor β activity in carcinomas. Semin. Radiat. Oncol. 2013, 23, 242–251. [Google Scholar] [CrossRef] [PubMed]
- Barcellos-Hoff, M.H.; Cucinotta, F.A. New tricks for an old fox: Impact of TGFβ on the DNA damage response and genomic stability. Sci. Signal. 2014, 7, re5. [Google Scholar] [CrossRef] [PubMed]
- Carl, C.; Flindt, A.; Hartmann, J.; Dahlke, M.; Rades, D.; Dunst, J.; Lehnert, H.; Gieseler, F.; Ungefroren, H. Ionizing radiation induces a motile phenotype in human carcinoma cells in vitro through hyperactivation of the TGF-beta signaling pathway. Cell. Mol. Life Sci. 2016, 73, 427–443. [Google Scholar] [CrossRef] [PubMed]
- Straub, J.M.; New, J.; Hamilton, C.D.; Lominska, C.; Shnayder, Y.; Thomas, S.M. Radiation-induced fibrosis: Mechanisms and implications for therapy. J. Cancer Res. Clin. Oncol. 2015, 141, 1985–1994. [Google Scholar] [CrossRef] [PubMed]
- Ungefroren, H.; Mubbach, F.; Gunther, B.; Katenkamp, K.; Henklein, P.; Westermann, M.; Settmacher, U.; Lenk, L.; Sebens, S.; Muller, J.P.; et al. Proteinase-activated receptor 2 (PAR2) in hepatic stellate cells—Evidence for a role in hepatocellular carcinoma growth in vivo. Mol. Cancer 2016, 15, 54. [Google Scholar]
- Lima, L.G.; Monteiro, R.Q. Activation of blood coagulation in cancer: Implications for tumour progression. Biosci. Rep. 2013, 33, e00064. [Google Scholar] [CrossRef] [PubMed]
- Tang, S.C.; Chen, Y.C. Novel therapeutic targets for pancreatic cancer. World J. Gastroenterol. 2014, 20, 10825–10844. [Google Scholar] [CrossRef] [PubMed]
- Sanchez-Elsner, T.; Botella, L.M.; Velasco, B.; Corbi, A.; Attisano, L.; Bernabeu, C. Synergistic cooperation between hypoxia and transforming growth factor-beta pathways on human vascular endothelial growth factor gene expression. J. Biol. Chem. 2001, 276, 38527–38535. [Google Scholar] [CrossRef] [PubMed]
- Calone, I.; Souchelnytskyi, S. Inhibition of TGFβ signaling and its implication in anticancer treatments. Exp. Oncol. 2012, 34, 9–16. [Google Scholar] [PubMed]
- Ramachandran, R.; Noorbakhsh, F.; Defea, K.; Hollenberg, M.D. Targeting proteinase-activated receptors: Therapeutic potential and challenges. Nat. Rev. Drug Discov. 2012, 11, 69–86. [Google Scholar] [CrossRef] [PubMed]
- Rebbapragada, A.; Benchabane, H.; Wrana, J.L.; Celeste, A.J.; Attisano, L. Myostatin signals through a transforming growth factor beta-like signaling pathway to block adipogenesis. Mol. Cell. Biol. 2003, 23, 7230–7242. [Google Scholar] [CrossRef] [PubMed]
- Unsicker, K.; Spittau, B.; Krieglstein, K. The multiple facets of the TGF-β family cytokine growth/differentiation factor-15/macrophage inhibitory cytokine-1. Cytokine Growth Factor Rev. 2013, 24, 373–384. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Wang, J.; Kong, J.; Tang, J.; Wu, Y.; Xu, E.; Zhang, H.; Lai, M. GDF15 promotes EMT and metastasis in colorectal cancer. Oncotarget 2016, 7, 860–872. [Google Scholar] [CrossRef] [PubMed]
- Elkina, Y.; von Haehling, S.; Anker, S.D.; Springer, J. The role of myostatin in muscle wasting: An overview. J. Cachexia Sarcopenia Muscle 2011, 2, 143–151. [Google Scholar] [CrossRef] [PubMed]
- Gorgens, S.W.; Eckardt, K.; Elsen, M.; Tennagels, N.; Eckel, J. Chitinase-3-like protein 1 protects skeletal muscle from TNFα-induced inflammation and insulin resistance. Biochem. J. 2014, 459, 479–488. [Google Scholar] [CrossRef] [PubMed]
- Antoniak, S.; Mackman, N. Multiple roles of the coagulation protease cascade during virus infection. Blood 2014, 123, 2605–2613. [Google Scholar] [CrossRef] [PubMed]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Witte, D.; Zeeh, F.; Gädeken, T.; Gieseler, F.; Rauch, B.H.; Settmacher, U.; Kaufmann, R.; Lehnert, H.; Ungefroren, H. Proteinase-Activated Receptor 2 Is a Novel Regulator of TGF-β Signaling in Pancreatic Cancer. J. Clin. Med. 2016, 5, 111. https://doi.org/10.3390/jcm5120111
Witte D, Zeeh F, Gädeken T, Gieseler F, Rauch BH, Settmacher U, Kaufmann R, Lehnert H, Ungefroren H. Proteinase-Activated Receptor 2 Is a Novel Regulator of TGF-β Signaling in Pancreatic Cancer. Journal of Clinical Medicine. 2016; 5(12):111. https://doi.org/10.3390/jcm5120111
Chicago/Turabian StyleWitte, David, Franziska Zeeh, Thomas Gädeken, Frank Gieseler, Bernhard H. Rauch, Utz Settmacher, Roland Kaufmann, Hendrik Lehnert, and Hendrik Ungefroren. 2016. "Proteinase-Activated Receptor 2 Is a Novel Regulator of TGF-β Signaling in Pancreatic Cancer" Journal of Clinical Medicine 5, no. 12: 111. https://doi.org/10.3390/jcm5120111
APA StyleWitte, D., Zeeh, F., Gädeken, T., Gieseler, F., Rauch, B. H., Settmacher, U., Kaufmann, R., Lehnert, H., & Ungefroren, H. (2016). Proteinase-Activated Receptor 2 Is a Novel Regulator of TGF-β Signaling in Pancreatic Cancer. Journal of Clinical Medicine, 5(12), 111. https://doi.org/10.3390/jcm5120111








