Reviewing Strategies and Our Approach to Mapping and Ablation of Left Ventricular Summit Arrhythmias
Abstract
1. Introduction
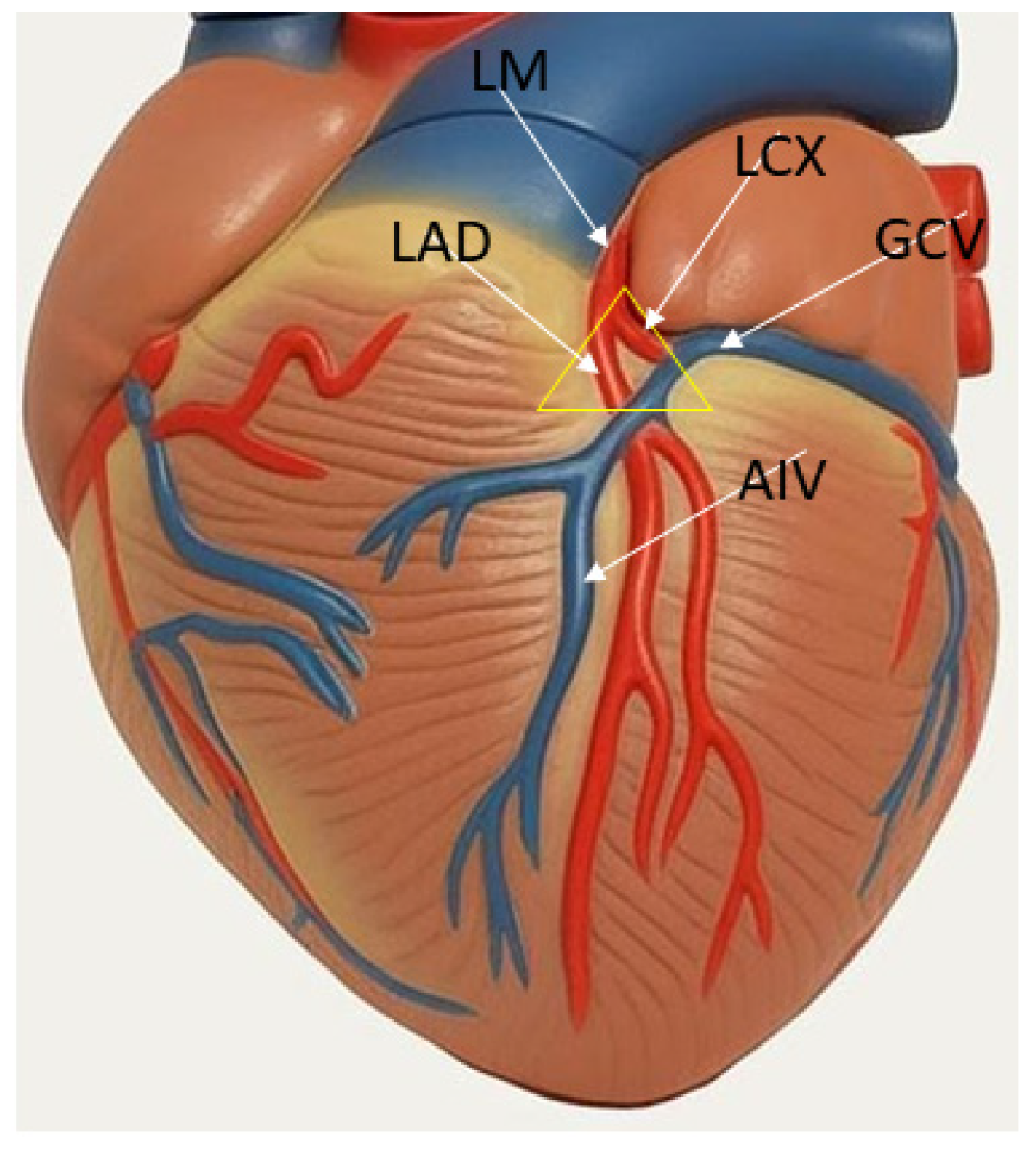
2. Anatomy of LV Summit
3. ECG Characteristics of LV Summit
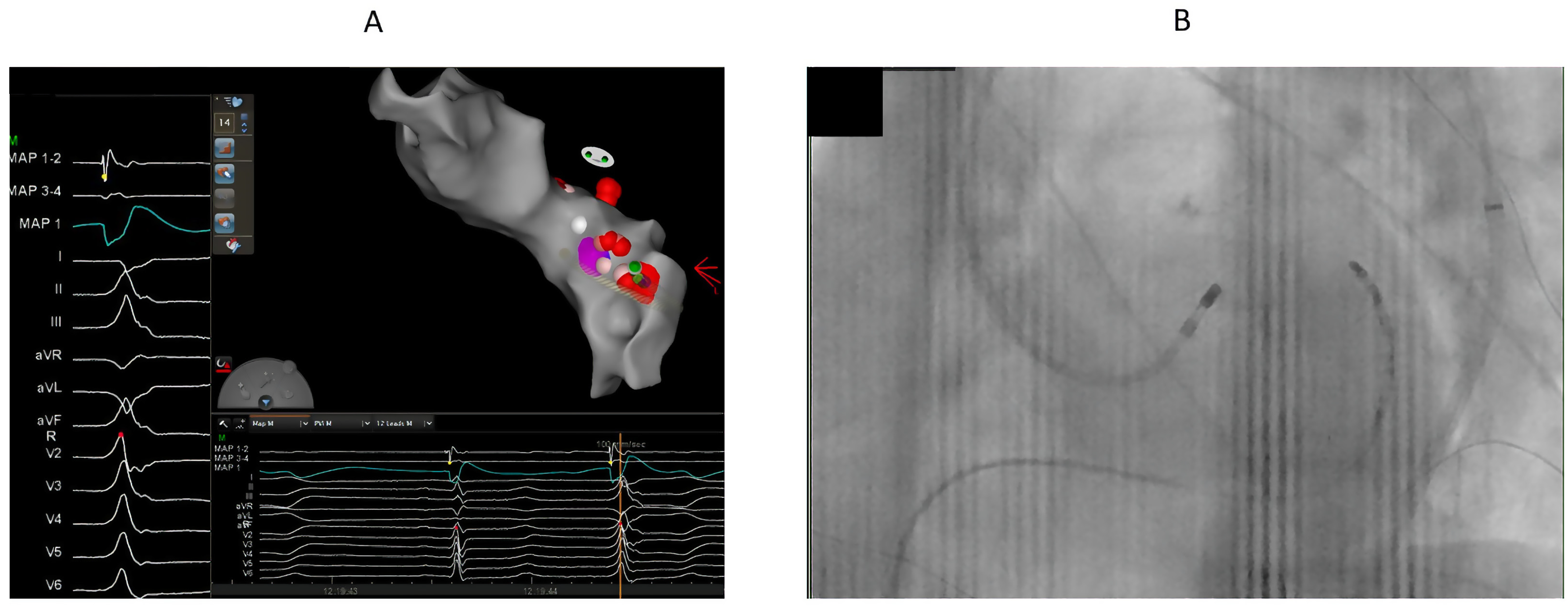
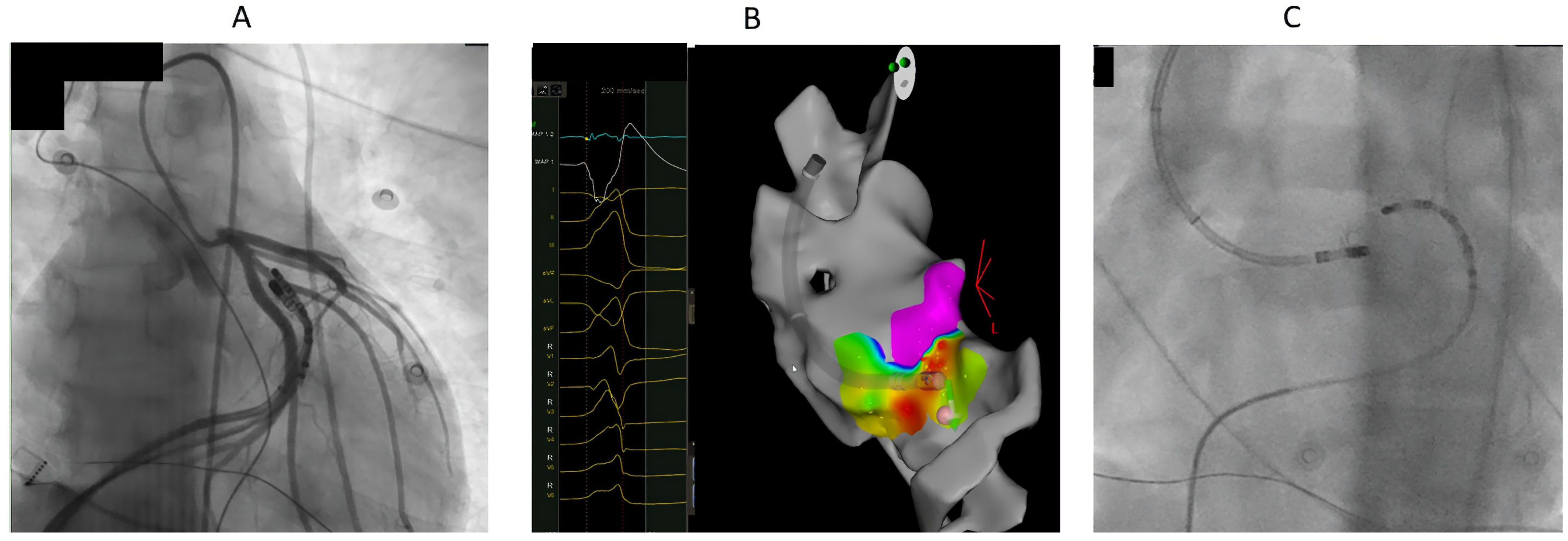
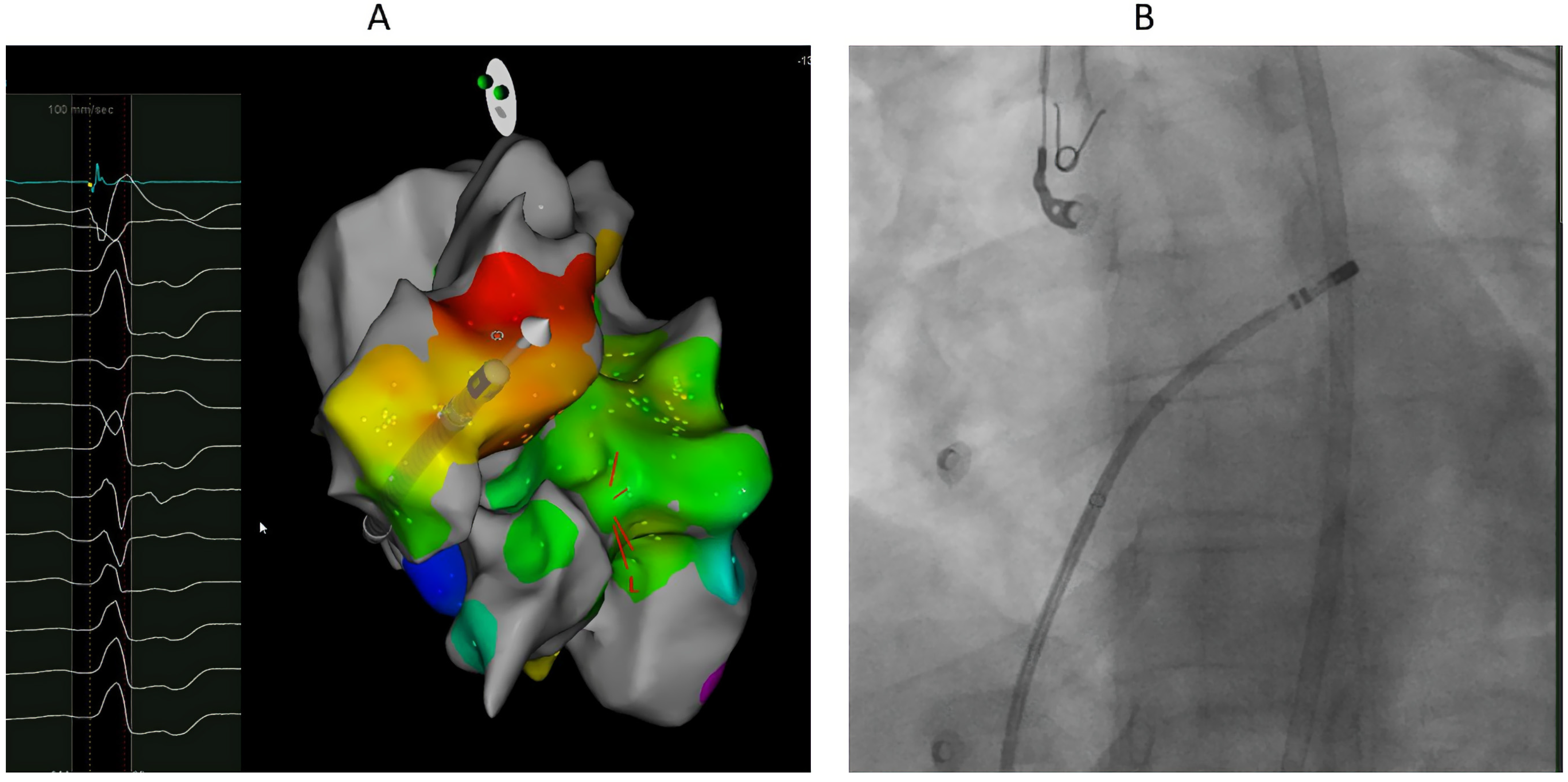
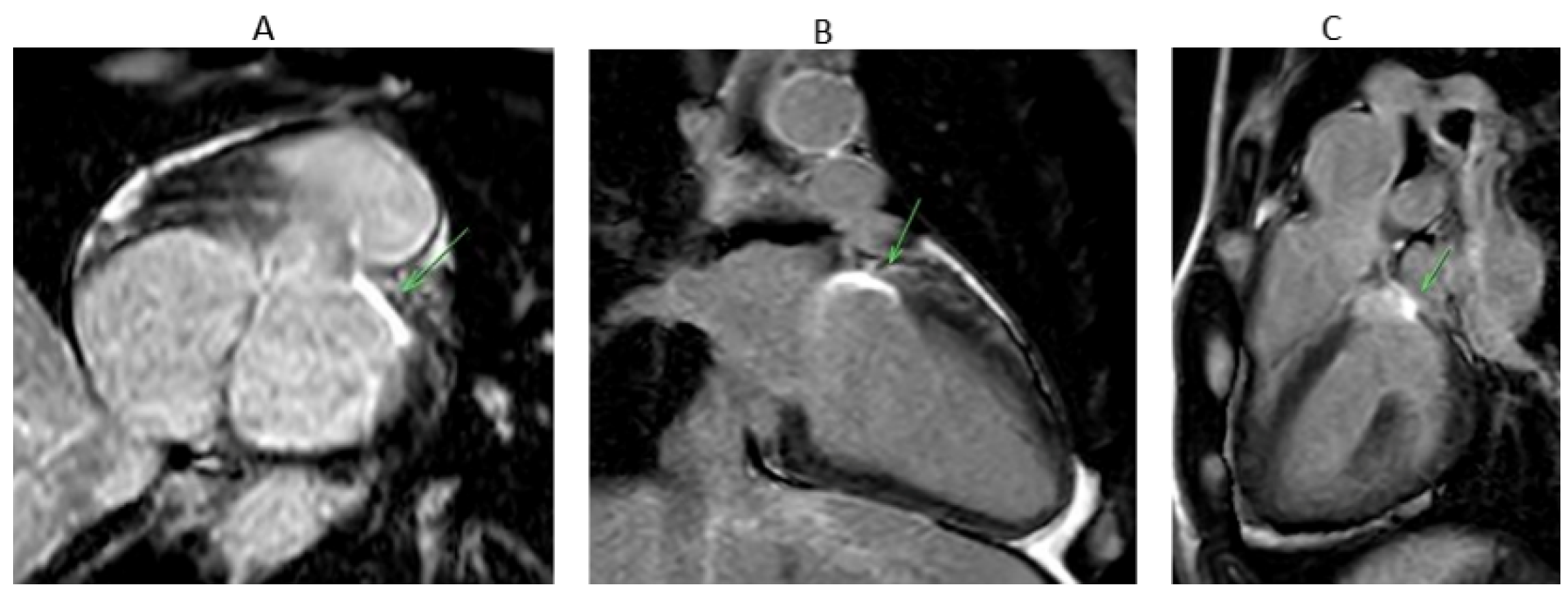
4. Mapping
5. Ablation
6. Special Ablation Techniques
6.1. Prolonged Duration Ablation
6.2. Bipolar Ablation
6.3. Ethanol Infusion
6.4. Wire Ablation
7. Our Approach
7.1. Procedure
7.2. Statistical Analysis
8. Results
8.1. Patients’ Characteristics
8.2. Ablation Sites
8.3. Clinical Follow Up
9. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Zeppenfeld, K.; Tfelt-Hansen, J.; De Riva, M.; Winkel, B.G.; Behr, E.R.; Blom, N.A.; Charron, P.; Corrado, D.; Dagres, N.; De Chillou, C.; et al. 2022 ESC guidelines for the management of patients with ventricular arrhythmias and the prevention of sudden cardiac death: Developed by the task force for the management of patients with ventricular arrhythmias and the prevention of sudden cardiac death of the European Society of Cardiology (ESC) Endorsed by the Association for European Paediatric and Congenital Cardiology (AEPC). Eur. Heart J. 2022, 43, 3997–4126. [Google Scholar]
- Yamada, T.; McElderry, H.T.; Doppalapudi, H.; Okada, T.; Murakami, Y.; Yoshida, Y.; Yoshida, N.; Inden, Y.; Murohara, T.; Plumb, V.J.; et al. Idiopathic Ventricular Arrhythmias Originating From the Left Ventricular Summit. Circ. Arrhythmia Electrophysiol. 2010, 3, 616–623. [Google Scholar] [CrossRef]
- Yamada, T.; Kumar, V.; Yoshida, N.; Doppalapudi, H. Eccentric Activation Patterns in the Left Ventricular Outflow Tract during Idiopathic Ventricular Arrhythmias Originating From the Left Ventricular Summit. Circ. Arrhythmia Electrophysiol. 2019, 12, e007419. [Google Scholar] [CrossRef] [PubMed]
- Motonaga, K.S.; Hsia, H.H. Road to the Summit May Follow an Eccentric Path. Circ. Arrhythmia Electrophysiol. 2019, 12, e007691. [Google Scholar] [CrossRef]
- Kuniewicz, M.; Baszko, A.; Ali, D.; Karkowski, G.; Loukas, M.; Walocha, J.A.; Hołda, M.K. Left Ventricular Summit—Concept, Anatomical Structure and Clinical Significance. Diagnostics 2021, 11, 1423. [Google Scholar] [CrossRef]
- Enriquez, A.; Malavassi, F.; Saenz, L.C.; Supple, G.; Santangeli, P.; Marchlinski, F.E.; Garcia, F.C. How to map and ablate left ventricular summit arrhythmias. Heart Rhythm 2017, 14, 141–148. [Google Scholar] [CrossRef]
- Romero, J.; Gamero, M.; Alviz, I.; Grushko, M.; Diaz, J.C.; Lorente, M.; Gabr, M.; Toquica, C.C.; Krishnan, S.; Velasco, A.; et al. Catheter Ablation of Left Ventricular Summit Arrhythmias from Adjacent Anatomic Vantage Points. Card. Electrophysiol. Clin. 2023, 15, 31–37. [Google Scholar] [CrossRef] [PubMed]
- Abularach, M.E.; Campos, B.; Park, K.M.; Tschabrunn, C.M.; Frankel, D.S.; Park, R.E.; Gerstenfeld, E.P.; Mountantonakis, S.; Garcia, F.C.; Dixit, S.; et al. Ablation of ventricular arrhythmias arising near the anterior epicardial veins from the left sinus of Valsalva region: ECG features, anatomic distance, and outcome. Heart Rhythm 2012, 9, 865–873. [Google Scholar] [CrossRef] [PubMed]
- Santangeli, P.; Marchlinski, F.E.; Zado, E.S.; Benhayon, D.; Hutchinson, M.D.; Lin, D.; Frankel, D.S.; Riley, M.P.; Supple, G.E.; Garcia, F.C.; et al. Percutaneous Epicardial Ablation of Ventricular Arrhythmias Arising from the Left Ventricular Summit. Circ. Arrhythmia Electrophysiol. 2015, 8, 337–343. [Google Scholar] [CrossRef]
- Bazan, V.; Gerstenfeld, E.P.; Garcia, F.C.; Bala, R.; Rivas, N.; Dixit, S.; Zado, E.; Callans, D.J.; Marchlinski, F.E. Site-specific twelve-lead ECG features to identify an epicardial origin for left ventricular tachycardia in the absence of myocardial infarction. Heart Rhythm 2007, 4, 1403–1410. [Google Scholar] [CrossRef]
- Hayashi, T.; Santangeli, P.; Pathak, R.K.; Muser, D.; Liang, J.J.; Castro, S.A.; Garcia, F.C.; Hutchinson, M.D.; Supple, G.E.; Frankel, D.S.; et al. Outcomes of Catheter Ablation of Idiopathic Outflow Tract Ventricular Arrhythmias With an R Wave Pattern Break in Lead V2: A Distinct Clinical Entity. J. Cardiovasc. Electrophysiol. 2017, 28, 504–514. [Google Scholar] [CrossRef]
- Liao, H.; Wei, W.; Tanager, K.S.; Miele, F.; Upadhyay, G.A.; Beaser, A.D.; Aziz, Z.; Nayak, H.M.; Ozcan, C.; Nishimura, T.; et al. Left ventricular summit arrhythmias with an abrupt V3 transition: Anatomy of the aortic interleaflet triangle vantage point. Heart Rhythm 2021, 18, 10–19. [Google Scholar] [CrossRef] [PubMed]
- Ouyang, F.; Mathew, S.; Wu, S.; Kamioka, M.; Metzner, A.; Xue, Y.; Ju, W.; Yang, B.; Zhan, X.; Rillig, A.; et al. Ventricular Arrhythmias Arising From the Left Ventricular Outflow Tract Below the Aortic Sinus Cusps. Circ. Arrhythmia Electrophysiol. 2014, 7, 445–455. [Google Scholar] [CrossRef]
- Benhayon, D.; Cogan, J.; Young, M. Left atrial appendage as a vantage point for mapping and ablating premature ventricular contractions originating in the epicardial left ventricular summit. Clin. Case Rep. 2018, 6, 1124–1127. [Google Scholar] [CrossRef] [PubMed]
- Marai, I.; Boulos, M.; Lessick, J.; Abadi, S.; Blich, M.; Suleiman, M. Outflow tract ventricular arrhythmia originating from the aortic cusps: Our approach for challenging ablation. J. Interv. Card. Electrophysiol. 2015, 45, 57–62. [Google Scholar] [CrossRef]
- De Sensi, F.; Miracapillo, G.; Cresti, A.; Paneni, F.; Limbruno, U. Image integration guided ablation of left outflow tract ventricular tachycardia: Is coronary angiography still necessary? Indian Pacing Electrophysiol. J. 2018, 18, 73–75. [Google Scholar] [CrossRef]
- Yamada, T. Predictors of Successful Endocardial Ablation of Epicardial Left Ventricular Summit Arrhythmias. Card. Electrophysiol. Clin. 2023, 15, 15–24. [Google Scholar] [CrossRef]
- Frankel, D.S.; Mountantonakis, S.E.; Dahu, M.I.; Marchlinski, F.E. Elimination of Ventricular Arrhythmias Originating From the Anterior Interventricular Vein With Ablation in the Right Ventricular Outflow Tract. Circ. Arrhythmia Electrophysiol. 2014, 7, 984–985. [Google Scholar] [CrossRef]
- Sosa, E.; Scanavacca, M.; d’Avila, A. Catheter Ablation of the Left Ventricular Outflow Tract Tachycardia from the Left Atrium. J. Interv. Card. Electrophysiol. 2002, 7, 61–65. [Google Scholar] [CrossRef] [PubMed]
- Enriquez, A.; Yogasundaram, H.; Neira, V.; Guandalini, G.; Markman, T.; Shivamurthy, P.; Hyman, M.; Hanumanthu, B.; Lin, D.; Schaller, R.; et al. Stepwise Anatomical Approach to Ablation of Intramural Outflow Tract Ventricular Arrhythmias Guided by Septal Coronary Venous Mapping. Circulation 2025, 151. [Google Scholar] [CrossRef]
- Hanson, M.; Futyma, P.; Bode, W.; Liang, J.J.; Tapia, C.; Adams, C.; Zarębski, Ł.; Wrzos, A.; Saenz, L.; Sadek, M.; et al. Catheter ablation of intramural outflow tract premature ventricular complexes: A multicentre study. EP Eur. 2023, 25, euad100. [Google Scholar] [CrossRef]
- Garg, L.; Daubert, T.; Lin, A.; Dhakal, B.; Santangeli, P.; Schaller, R.; Hyman, M.C.; Kumareswaran, R.; Arkles, J.; Nazarian, S.; et al. Utility of Prolonged Duration Endocardial Ablation for Ventricular Arrhythmias Originating From the Left Ventricular Summit. JACC Clin. Electrophysiol. 2022, 8, 465–476. [Google Scholar] [CrossRef]
- Enriquez, A.; Hanson, M.; Nazer, B.; Gibson, D.N.; Cano, O.; Tokioka, S.; Fukamizu, S.; Millan, P.S.; Hoyos, C.; Matos, C.; et al. Bipolar ablation involving coronary venous system for refractory left ventricular summit arrhythmias. Heart Rhythm O2 2024, 5, 24–33. [Google Scholar] [CrossRef]
- Futyma, P.; Sultan, A.; Zarębski, Ł.; Imnadze, G.; Maslova, V.; Bordignon, S.; Kousta, M.; Knecht, S.; Pavlović, N.; Peichl, P.; et al. Bipolar radiofrequency ablation of refractory ventricular arrhythmias: Results from a multicentre network. EP Eur. 2024, 26, euae248. [Google Scholar] [CrossRef]
- Nguyen, D.T.; Zheng, L.; Zipse, M.M.; Borne, R.T.; Tzou, W.S.; Fleeman, B.; Sauer, W.H. Bipolar radiofrequency ablation creates different lesion characteristics compared to simultaneous unipolar ablation. J. Cardiovasc. Electrophysiol. 2019, 30, 2960–2967. [Google Scholar] [CrossRef]
- Teh, A.W.; Reddy, V.Y.; Koruth, J.S.; Miller, M.A.; Choudry, S.; D’avila, A.N.; Dukkipati, S.R. Bipolar radiofrequency catheter ablation for refractory ventricular outflow tract arrhythmias. J. Cardiovasc. Electrophysiol. 2014, 25, 1093–1099. [Google Scholar] [CrossRef]
- Heeger, C.; Kuck, K.; Ouyang, F. Catheter ablation of pulmonary sinus cusp-derived ventricular arrhythmias by the reversed U-curve technique. J. Cardiovasc. Electrophysiol. 2017, 28, 776–777. [Google Scholar] [CrossRef]
- Zhang, J.; Tang, C.; Zhang, Y.; Su, X. Pulmonary sinus cusp mapping and ablation: A new concept and approach for idiopathic right ventricular outflow tract arrhythmias. Heart Rhythm 2018, 15, 38–45. [Google Scholar] [CrossRef] [PubMed]
- Futyma, P.; Santangeli, P.; Pürerfellner, H.; Pothineni, N.V.; Głuszczyk, R.; Ciąpała, K.; Moroka, K.; Martinek, M.; Futyma, M.; Marchlinski, F.E.; et al. Anatomic approach with bipolar ablation between the left pulmonic cusp and left ventricular outflow tract for left ventricular summit arrhythmias. Heart Rhythm 2020, 17, 1519–1527. [Google Scholar] [CrossRef] [PubMed]
- Futyma, P.; Sauer, W.H. Bipolar Radiofrequency Catheter Ablation of Left Ventricular Summit Arrhythmias. Card. Electrophysiol. Clin. 2023, 15, 57–62. [Google Scholar] [CrossRef] [PubMed]
- Futyma, P.; Sander, J.; Ciąpała, K.; Głuszczyk, R.; Wysokińska, A.; Futyma, M.; Kułakowski, P. Bipolar radiofrequency ablation delivered from coronary veins and adjacent endocardium for treatment of refractory left ventricular summit arrhythmias. J. Interv. Card. Electrophysiol. 2020, 58, 307–313. [Google Scholar] [CrossRef] [PubMed]
- Flautt, T.; Valderrábano, M. Retrograde Coronary Venous Ethanol Infusion for Ablation of Refractory Left Ventricular Summit Arrhythmias. Card. Electrophysiol. Clin. 2023, 15, 63–74. [Google Scholar] [CrossRef] [PubMed]
- Fuentes, S.; Valderrábano, M. Venous Ethanol Ablation Approaches for Radiofrequency-Refractory Cardiac Arrhythmias. Curr. Cardiol. Rep. 2023, 25, 917–924. [Google Scholar] [CrossRef]
- Tavares, L.; Valderrábano, M. Retrograde venous ethanol ablation for ventricular tachycardia. Heart Rhythm 2019, 16, 478–483. [Google Scholar] [CrossRef]
- Tavares, L.; Lador, A.; Fuentes, S.; Da-Wariboko, A.; Blaszyk, K.; Malaczynska-Rajpold, K.; Papiashvili, G.; Korolev, S.; Peichl, P.; Kautzner, J.; et al. Intramural Venous Ethanol Infusion for Refractory Ventricular Arrhythmias. JACC Clin. Electrophysiol. 2020, 6, 1420–1431. [Google Scholar] [CrossRef]
- Valderrábano, M.; Fuentes Rojas, S.C.; Lador, A.; Patel, A.; Schurmann, P.A.; Tapias, C.; Rodríguez, D.; Carlos Sáenz, L.; Malahfji, M.; Shah, D.J.; et al. Substrate Ablation by Multivein, Multiballoon Coronary Venous Ethanol for Refractory Ventricular Tachycardia in Structural Heart Disease. Circulation 2022, 146, 1644–1656. [Google Scholar] [CrossRef]
- Efremidis, M.; Vlachos, K.; Bazoukis, G.; Frontera, A.; Martin, C.A.; Dragasis, S.; Valkanas, K.; Letsas, K.P. Novel technique targeting left ventricular summit premature ventricular contractions using radiofrequency ablation through a guidewire. HeartRhythm Case Rep. 2021, 7, 134–138. [Google Scholar] [CrossRef]
- Xuan, F.; Liang, M.; Li, S.; Zuo, Z.; Han, Y.; Wang, Z. Guidewire ablation of epicardial ventricular arrhythmia within the coronary venous system: A case report. HeartRhythm Case Rep. 2022, 8, 195–199. [Google Scholar] [CrossRef]
- Okabe, T.; Rushing, G.D.; Kalbfleisch, S.J. Surgical Mapping and Ablation in the Left Ventricular Summit Guided by Presurgery Pericardial Mapping. J. Innov. Card. Rhythm Manag. 2019, 10, 3582–3587. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Rangaswamy, V.V.; Saggu, D.K.; Yalagudri, S.; Ramasubrahmanyam, G.; Narasimhan, C. Management of refractory ventricular tachycardia by direct intramyocardial injection of alcohol: A novel method. HeartRhythm Case Rep. 2020, 6, 618–621. [Google Scholar] [CrossRef]
- Mont, L.; Roca-Luque, I.; Althoff, T.F. Ablation Lesion Assessment with MRI. Arrhythmia Electrophysiol. Rev. 2022, 11, e02. [Google Scholar] [CrossRef]
- Dickfeld, T.; Tian, J.; Ahmad, G.; Jimenez, A.; Turgeman, A.; Kuk, R.; Peters, M.; Saliaris, A.; Saba, M.; Shorofsky, S.; et al. MRI-Guided Ventricular Tachycardia Ablation: Integration of late gadolinium-enhanced 3D scar in patients with implantable cardioverter-defibrillators. Circ. Arrhythmia Electrophysiol. 2011, 4, 172–184. [Google Scholar] [CrossRef]
- Reddy, V.Y.; Gerstenfeld, E.P.; Natale, A.; Whang, W.; Cuoco, F.A.; Patel, C.; Mountantonakis, S.E.; Gibson, D.N.; Harding, J.D.; Ellis, C.R.; et al. Pulsed Field or Conventional Thermal Ablation for Paroxysmal Atrial Fibrillation. N. Engl. J. Med. 2023, 389, 1660–1671. [Google Scholar] [CrossRef]
- Ruwald, M.H.; Johannessen, A.; Hansen, M.L.; Haugdal, M.; Worck, R.; Hansen, J. Focal pulsed field ablation and ultrahigh-density mapping—Versatile tools for all atrial arrhythmias? Initial procedural experiences. J. Interv. Card. Electrophysiol. 2023, 67, 99–109. [Google Scholar] [CrossRef]
- Reddy, V.Y.; Peichl, P.; Anter, E.; Rackauskas, G.; Petru, J.; Funasako, M.; Minami, K.; Koruth, J.S.; Natale, A.; Jais, P.; et al. A Focal Ablation Catheter Toggling Between Radiofrequency and Pulsed Field Energy to Treat Atrial Fibrillation. JACC Clin. Electrophysiol. 2023, 9, 1786–1801. [Google Scholar] [CrossRef] [PubMed]
- Jiang, S.; Qian, F.; Ji, S.; Li, L.; Liu, Q.; Zhou, S.; Xiao, Y. Pulsed Field Ablation for Atrial Fibrillation: Mechanisms, Advantages, and Limitations. Rev. Cardiovasc. Med. 2024, 25, 138. [Google Scholar] [CrossRef] [PubMed]
- du Pre, B.C.; van Driel, V.J.; van Wessel, H.; Loh, P.; Doevendans, P.A.; Goldschmeding, R.; Wittkampf, F.H.; Vink, A. Minimal coronary artery damage by myocardial electroporation ablation. EP Eur. 2012, 15, 144–149. [Google Scholar] [CrossRef] [PubMed]
- Iyengar, S.K.; Iyengar, S.; Srivathsan, K. The promise of pulsed field ablation and the challenges ahead. Front Cardiovasc Med. 2023, 10, 1235317. [Google Scholar] [CrossRef]
- Benali, K.; Yokoyama, M.; Vlachos, K.; Kneizeh, K.; Monaco, C.; Sava, R.; Duchateau, J.; Ploux, S.; Da Costa, A.; Sacher, F.; et al. Targeting the Left Ventricular Summit: Pulsed Field Ablation for Refractory Premature Ventricular Complexes. JACC Clin. Electrophysiol. 2025, 11, 1080–1086. [Google Scholar] [CrossRef]
- Spenkelink, D.; van Wessel, H.; van Driel, V.J.; Ramanna, H.; van der Heijden, J.F. Pulsed field ablation as a feasible option for the treatment of epicardial left ventricular summit premature complex foci near the coronary arteries: A case report. Eur. Heart J.—Case Rep. 2024, 8, ytae478. [Google Scholar] [CrossRef]
| N = 38 | |
|---|---|
| Age: years | 57 ± 12.3 |
| Males (%) | 25 (65.8) |
| Hypertension (%) | 19 (50) |
| Diabetes mellitus (%) | 15 (39.5) |
| Coronary artery disease (%) | 10 (26.3) |
| Ejection fraction (%) | 50.34 ± 11.63 |
| Burden of ventricular premature contractions (%) | 21.4 ± 3 |
| Total = 38 | V Transition by ECG | Failure = 3 | Comments | |
|---|---|---|---|---|
| LVOT below LCC | 17 | V1 = 9 | 0 | |
| V2 = 3 | 0 | |||
| V3 = 5 | 0 | |||
| LCC+ LVOT | 9 | V1 = 2 | 0 | |
| V2 = 3 | 1 | |||
| V3 = 4 | 0 | |||
| LVOT/LCC+RVOT/LPC | 7 | V1 = 2 | 0 | Ablation at LPC was performed in two patients |
| V2 = 4 | 0 | |||
| V3 = 1 | 0 | |||
| LVOT+ GCV-AIV | 2 | V1 = 1 | 1 | |
| V2 = 1 | 0 | |||
| RVOT only | 1 | V3 = 1 | 0 | |
| Percutaneous epicardial ablation after failure of endocardial ablation | 2 | V1 = 1 | 1 | Endocardial ablation included LCC/LVOT |
| V2 = 1 | 0 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abuiznait, Z.; Ghanayem, M.; Andria, N.; Sakhnini, A.; Birati, E.; Marai, I. Reviewing Strategies and Our Approach to Mapping and Ablation of Left Ventricular Summit Arrhythmias. J. Clin. Med. 2025, 14, 6120. https://doi.org/10.3390/jcm14176120
Abuiznait Z, Ghanayem M, Andria N, Sakhnini A, Birati E, Marai I. Reviewing Strategies and Our Approach to Mapping and Ablation of Left Ventricular Summit Arrhythmias. Journal of Clinical Medicine. 2025; 14(17):6120. https://doi.org/10.3390/jcm14176120
Chicago/Turabian StyleAbuiznait, Ziad, Mohamad Ghanayem, Nizar Andria, Ali Sakhnini, Edo Birati, and Ibrahim Marai. 2025. "Reviewing Strategies and Our Approach to Mapping and Ablation of Left Ventricular Summit Arrhythmias" Journal of Clinical Medicine 14, no. 17: 6120. https://doi.org/10.3390/jcm14176120
APA StyleAbuiznait, Z., Ghanayem, M., Andria, N., Sakhnini, A., Birati, E., & Marai, I. (2025). Reviewing Strategies and Our Approach to Mapping and Ablation of Left Ventricular Summit Arrhythmias. Journal of Clinical Medicine, 14(17), 6120. https://doi.org/10.3390/jcm14176120







