Portal Vein Thrombosis, Management and Approaches in Liver Transplantation: A Narrative Review
Abstract
1. Introduction
2. Incidence and Mechanisms
3. Portal Vein Thrombosis Classification
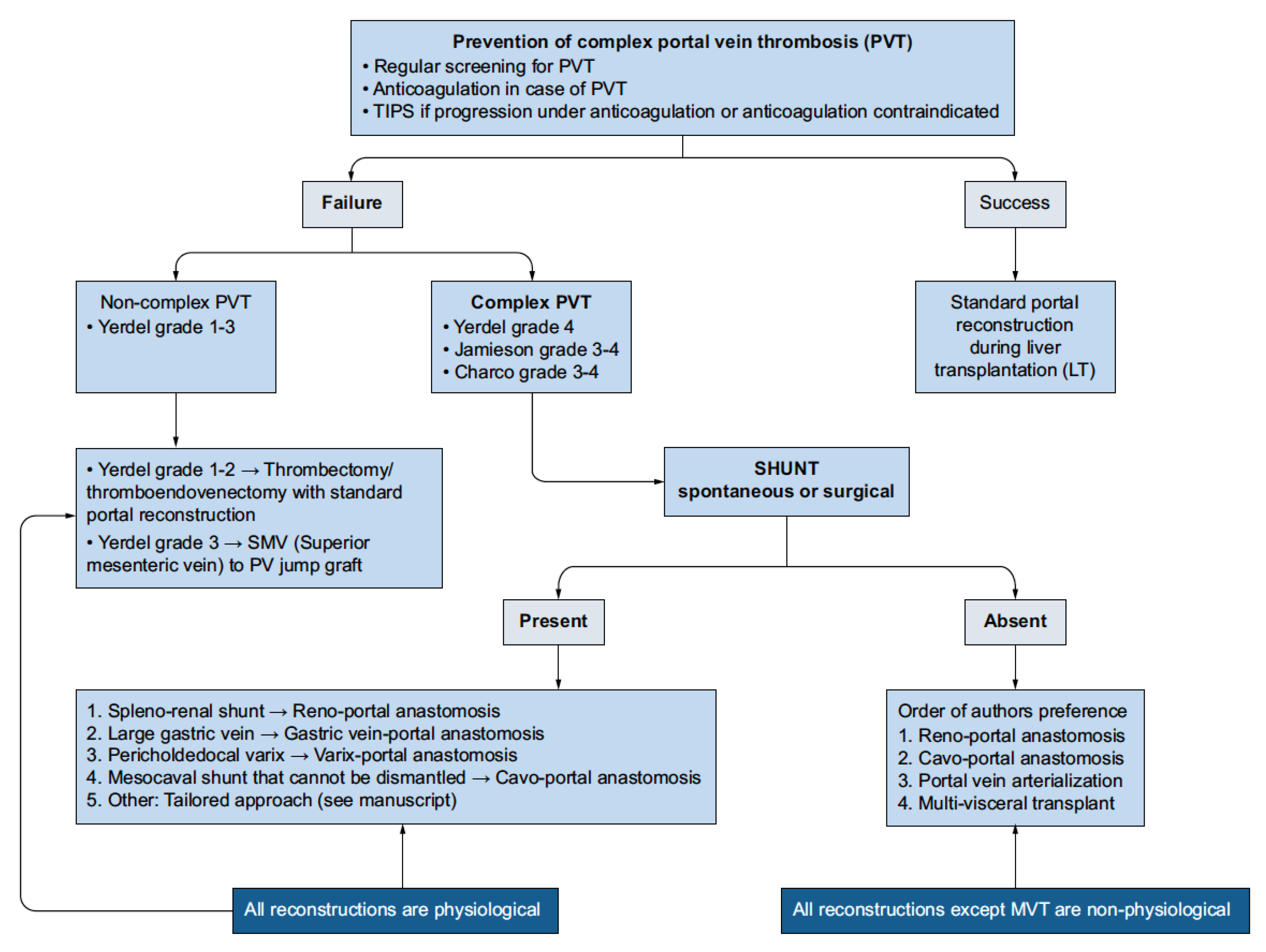
4. Management of Portal Vein Thrombosis in Candidates for Transplantation
4.1. Anticoagulation in Pre-Transplant Candidates
4.2. Transjugular Intrahepatic Portosystemic Shunt (TIPS)
5. Strategies During Liver Transplantation
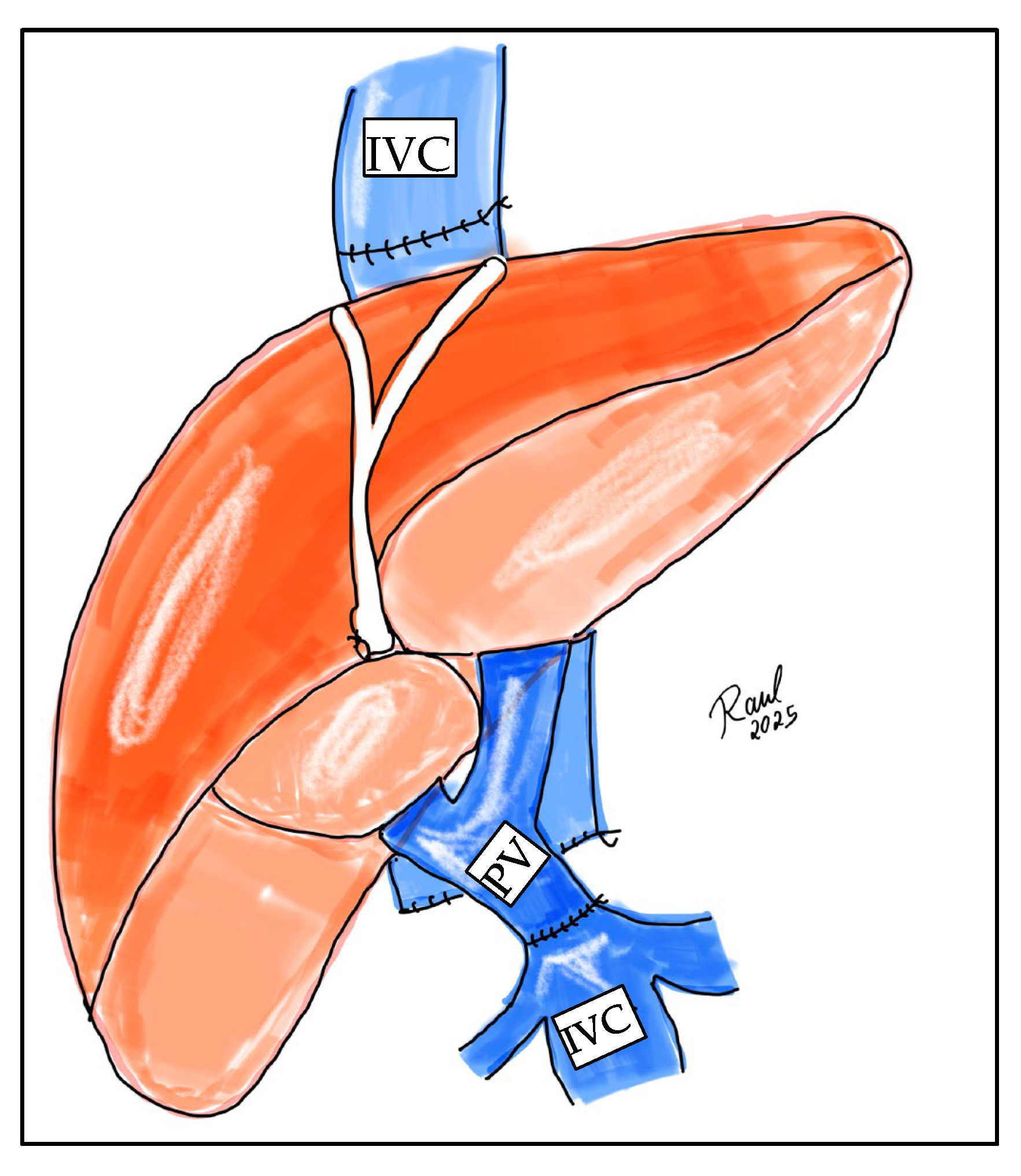
5.1. Eversion Thrombectomy
5.2. Jump Graft from SMV
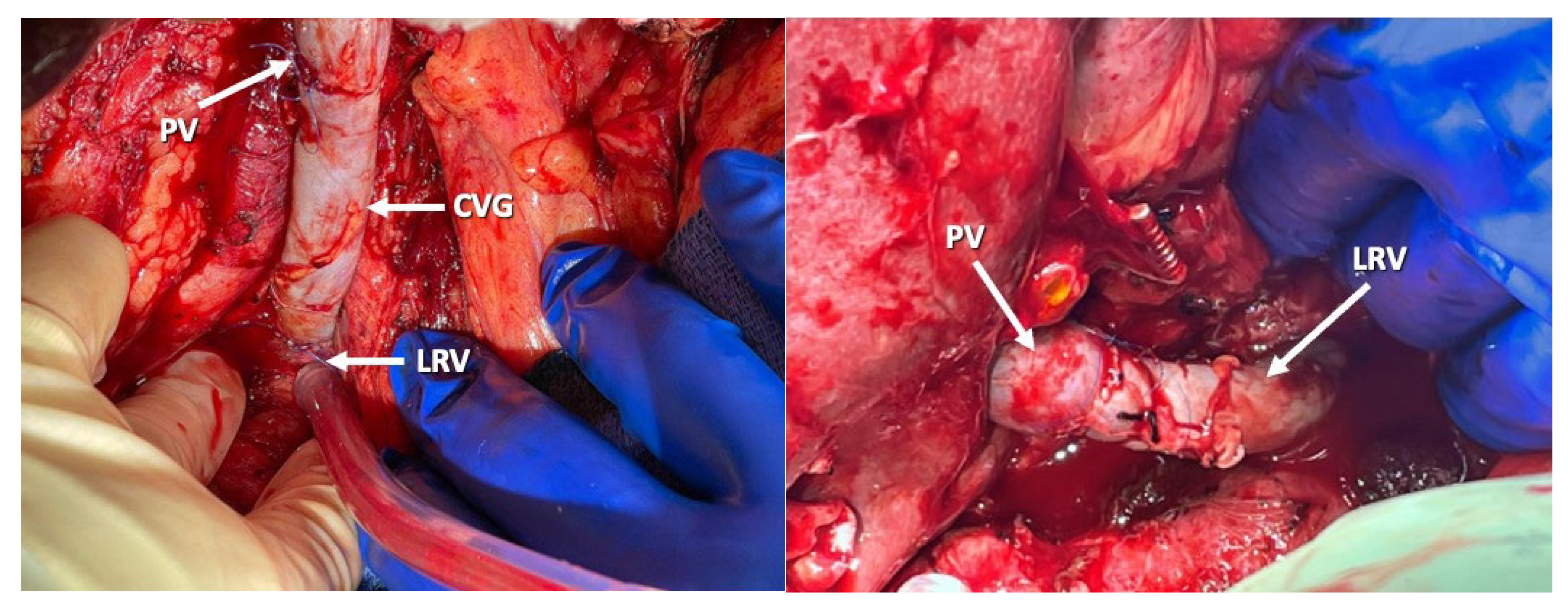
5.3. Other Strategies
5.4. Multivisceral Transplantation
6. Post-Transplant Management
6.1. Anticoagulation in Post-Transplant Patients
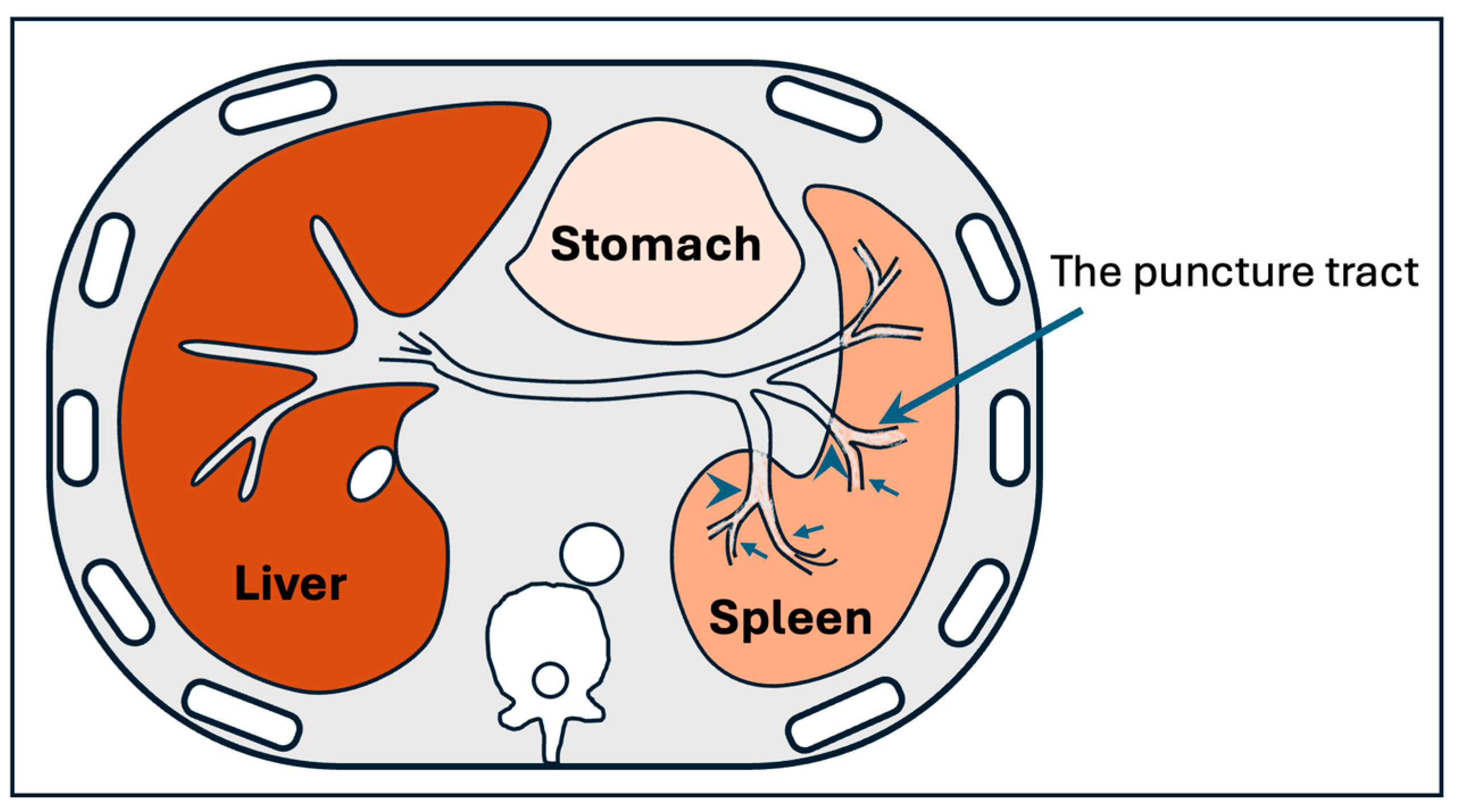
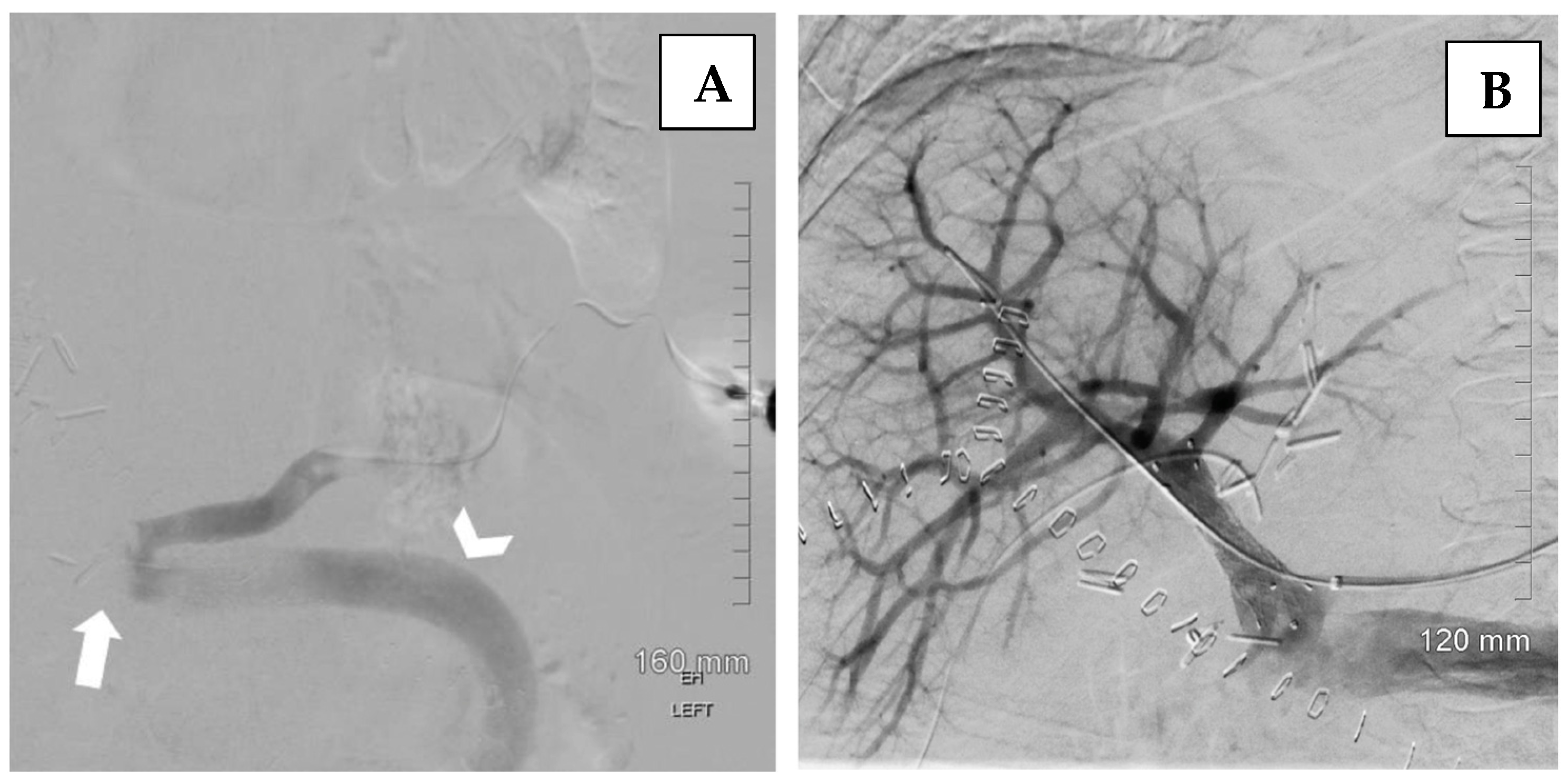
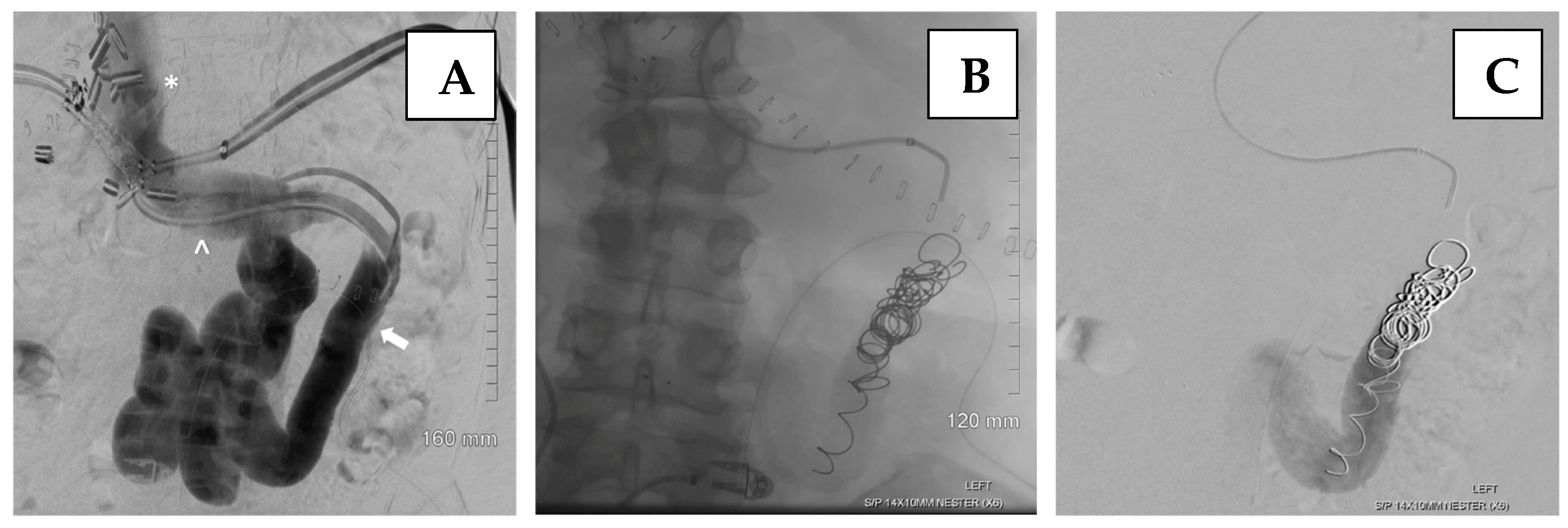
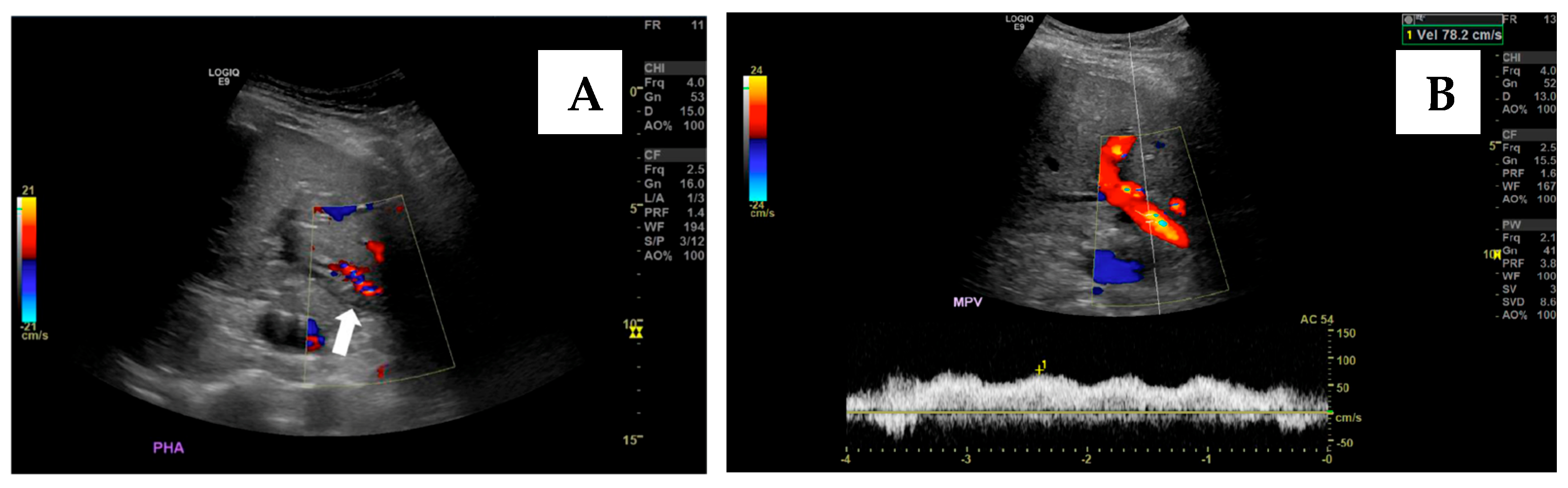
6.2. Endovascular Management of Portal Vein Thrombosis
6.3. Late Portal Vein Thrombosis After Liver Transplantation
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
Abbreviations
| PVT | Portal Vein Thrombosis |
| LT | Liver Transplantation |
| TIPS | Transjugular Intrahepatic Portosystemic Shunts |
| MVT | Multivisceral Transplantation |
References
- Rizzari, M.D.; Safwan, M.; Sobolic, M.; Kitajima, T.; Collins, K.; Yoshida, A.; Abouljoud, M.; Nagai, S. The Impact of Portal Vein Thrombosis on Liver Transplant Outcomes: Does Grade or Flow Rate Matter? Transplantation 2021, 105, 363–371. [Google Scholar] [CrossRef]
- Garcia-Pagan, J.C.; Valla, D.C. Portal vein thrombosis: A predictable milestone in cirrhosis? J. Hepatol. 2009, 51, 632–634. [Google Scholar] [CrossRef]
- Ghabril, M.; Agarwal, S.; Lacerda, M.; Chalasani, N.; Kwo, P.; Tector, A.J. Portal Vein Thrombosis is a Risk Factor for Poor Early Outcomes After Liver Transplantation: Analysis of Risk Factors and Outcomes for Portal Vein Thrombosis in Waitlisted Patients. Transplantation 2016, 100, 126–133. [Google Scholar] [CrossRef] [PubMed]
- Arabpour, E.; Hatami, B.; Pasharavavesh, L.; Rabbani, A.H.; Zarean Shahraki, S.; Amiri, M.; Zali, M.R. Clinical characteristics and predictors of benign portal vein thrombosis in patients with liver cirrhosis: A retrospective single-center study. Medicine 2024, 103, e39823. [Google Scholar] [CrossRef] [PubMed]
- Northup, P.G.; Sundaram, V.; Fallon, M.B.; Reddy, K.R.; Balogun, R.A.; Sanyal, A.J.; Anstee, Q.M.; Hoffman, M.R.; Ikura, Y.; Caldwell, S.H. Coagulation in Liver Disease Group. Hypercoagulation and thrombophilia in liver disease. J. Thromb. Haemost. 2008, 6, 2–9. [Google Scholar] [CrossRef] [PubMed]
- Amitrano, L.; Guardascione, M.A.; Brancaccio, V.; Margaglione, M.; Manguso, F.; Iannaccone, L.; Grandone, E.; Balzano, A. Risk factors and clinical presentation of portal vein thrombosis in patients with liver cirrhosis. J. Hepatol. 2004, 40, 736–741. [Google Scholar] [CrossRef]
- Amitrano, L.; Brancaccio, V.; Guardascione, M.A.; Margaglione, M.; Iannaccone, L.; D’Andrea, G.; Marmo, R.; Ames, P.R.; Balzano, A. Inherited coagulation disorders in cirrhotic patients with portal vein thrombosis. Hepatology 2000, 31, 345–348. [Google Scholar] [CrossRef]
- Zocco, M.A.; Di Stasio, E.; De Cristofaro, R.; Novi, M.; Ainora, M.E.; Ponziani, F.; Riccardi, L.; Lancellotti, S.; Santoliquido, A.; Flore, R.; et al. Thrombotic risk factors in patients with liver cirrhosis: Correlation with MELD scoring system and portal vein thrombosis development. J. Hepatol. 2009, 51, 682–689. [Google Scholar] [CrossRef]
- Jamieson Neville, V. Changing perspectives in portal vein thrombosis and liver transplantation. Transplantation 2000, 69, 1772–1774. [Google Scholar] [CrossRef]
- Yerdel, M.A.; Gunson, B.; Mirza, D.; Karayalçin, K.; Olliff, S.; Buckels, J.; Mayer, D.; McMaster, P.; Pirenne, J. Portal vein thrombosis in adults undergoing liver transplantation: Risk factors, screening, management, and outcome. Transplantation 2000, 69, 1873–1881. [Google Scholar] [CrossRef]
- Hibi, T.; Nishida, S.; Levi, D.M.; Selvaggi, G.; Tekin, A.; Fan, J.; Ruiz, P.; Tzakis, A.G. When and why portal vein thrombosis matters in liver transplantation: A critical audit of 174 cases. Ann. Surg. 2014, 259, 760–766. [Google Scholar] [CrossRef]
- Di Benedetto, F.; Magistri, P.; Di Sandro, S.; Boetto, R.; Tandoi, F.; Camagni, S.; Lauterio, A.; Pagano, D.; Nicolini, D.; Violi, P.; et al. Portal vein thrombosis and liver transplantation: Management, matching, and outcomes. A retrospective multicenter cohort study. Int. J. Surg. 2024, 110, 2874–2882. [Google Scholar] [CrossRef] [PubMed]
- de Franchis, R.; Baveno VI Faculty. Expanding consensus in portal hypertension: Report of the Baveno VI Consensus Workshop: Stratifying risk and individualizing care for portal hypertension. J. Hepatol. 2015, 63, 743–752. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Zhao, Y.; Han, B.; Zhu, Z.; Sun, L.; Cui, X. The Efficacy and Safety of Anticoagulants in the Treatment of Cirrhotic Portal Vein Thrombosis: A Systematic Review and Meta-Analysis. Clin. Appl. Thromb. Hemost. 2022, 28, 10760296221104797. [Google Scholar] [CrossRef]
- Han, G.; Qi, X.; He, C.; Yin, Z.; Wang, J.; Xia, J.; Yang, Z.; Bai, M.; Meng, X.; Niu, J.; et al. Transjugular intrahepatic portosystemic shunt for portal vein thrombosis with symptomatic portal hypertension in liver cirrhosis. J. Hepatol. 2011, 54, 78–88. [Google Scholar] [CrossRef] [PubMed]
- Luca, A.; Miraglia, R.; Caruso, S.; Milazzo, M.; Sapere, C.; Maruzzelli, L.; Vizzini, G.; Tuzzolino, F.; Gridelli, B.; Bosch, J. Short- and long-term effects of the transjugular intrahepatic portosystemic shunt on portal vein thrombosis in patients with cirrhosis. Gut 2011, 60, 846–852. [Google Scholar] [CrossRef]
- Valentin, N.; Korrapati, P.; Constantino, J.; Young, A.; Weisberg, I. The role of transjugular intrahepatic portosystemic shunt in the management of portal vein thrombosis: A systematic review and meta-analysis. Eur. J. Gastroenterol. Hepatol. 2018, 30, 1187–1193. [Google Scholar] [CrossRef]
- Salem, R.; Vouche, M.; Baker, T.; Herrero, J.I.; Caicedo, J.C.; Fryer, J.; Hickey, R.; Habib, A.; Abecassis, M.; Koller, F.; et al. Pretransplant Portal Vein Recanalization-Transjugular Intrahepatic Portosystemic Shunt in Patients with Complete Obliterative Portal Vein Thrombosis. Transplantation 2015, 99, 2347–2355. [Google Scholar] [CrossRef]
- Wang, Z.; Jiang, M.S.; Zhang, H.L.; Weng, N.N.; Luo, X.F.; Li, X.; Yang, L. Is Post-TIPS Anticoagulation Therapy Necessary in Patients with Cirrhosis and Portal Vein Thrombosis? A Randomized Controlled Trial. Radiology 2016, 279, 943–951. [Google Scholar] [CrossRef]
- Clavien, P.A.; Selzner, M.; Tuttle-Newhall, J.E.; Harland, R.C.; Suhocki, P. Liver transplantation complicated by misplaced TIPS in the portal vein. Ann. Surg. 1998, 227, 440–445. [Google Scholar] [CrossRef]
- Swersky, A.; Borja-Cacho, D.; Deitch, Z.; Thornburg, B.; Salem, R. Portal Vein Recanalization-Transjugular Intrahepatic Portosystemic Shunt (PVR-TIPS) Facilitates Liver Transplantation in Cirrhotic Patients with Occlusive Portal Vein Thrombosis. Semin. Intervent Radiol. 2023, 40, 38–43. [Google Scholar] [CrossRef]
- Santibanes, M.; Ardiles, V.; Uño, J.W.; Mattera, J.; de Santibanes, E.; Pekolj, J. Surgical strategies to treat portal vein thrombosis during adult liver transplantation. Langenbecks Arch. Surg. 2023, 408, 399. [Google Scholar] [CrossRef] [PubMed]
- Tublin, M.E.; Dodd, G.D., 3rd; Baron, R.L. Benign and malig- nant portal vein thrombosis: Differentiation by CT characteristics. AJR Am. J. Roentgenol. 1997, 168, 719–723. [Google Scholar] [CrossRef] [PubMed]
- Zerwonko, M.E.; Pekolj, J.; Mattera, J.; Peralta, O.A.; García-Mónaco, R.D.; de Santibañes, E.; de Santibañes, M. Intraoperative stent placement for the treatment of acute portal vein complications in pediatric living donor liver transplantation. Langenbecks Arch. Surg. 2019, 404, 123–128. [Google Scholar] [CrossRef] [PubMed]
- Marini, M.; Castro-Lopez, E.; Manteiga, D.F.; Vazquez, M.A.; Soidán, D.G.; Polo, J.I.R.; Gómez-Gutiérrez, M. Endovascular Treatment of Spleno-Mesenteric-Portal Vein Thrombosis During Orthotopic Liver Transplant: 20 Years Later. Transplant. Proc. 2020, 52, 1459–1463. [Google Scholar] [CrossRef]
- Maillot, B.; Bouzille, G.; Mabrut, J.Y.; Girard, E.; Laurent, A.; Navarro, F.; Chebaro, A.; Chiche, L.; Faitot, F.; Sulpice, L.; et al. The Role of Cavoportal and Renoportal Hemitransposition in Liver Transplantation. Ann. Transplant. 2022, 27, e935892. [Google Scholar] [CrossRef]
- Nazzal, M.; Sun, Y.; Okoye, O.; Diggs, L.; Evans, N.; Osborn, T.; Etesami, K.; Varma, C. Reno-portal shunt for liver transplant, an alternative inflow for recipients with grade III-IV portal vein thrombosis: Tips for a better outcome. Int. J. Surg. Case Rep. 2017, 41, 251–254. [Google Scholar] [CrossRef]
- Tekin, A.; Beduschi, T.; Vianna, R.; Mangus, R.S. Multivisceral transplant as an option to transplant cirrhotic patients with severe portal vein thrombosis. Int. J. Surg. 2020, 82S, 115–121. [Google Scholar] [CrossRef]
- Bhangui, P.; Lim, C.; Levesque, E.; Salloum, C.; Lahat, E.; Feray, C.; Azoulay, D. Novel classification of non-malignant portal vein thrombosis: A guide to surgical decision-making during liver transplantation. J. Hepatol. 2019, 71, 1038–1050. [Google Scholar] [CrossRef]
- Nicolau-Raducu, R.; Livingstone, J.; Salsamendi, J.; Beduschi, T.; Vianna, R.; Tekin, A.; Selvaggi, G.; Raveh, Y. Visceral arterial embolization prior to multivisceral transplantation in recipient with cirrhosis, extensive portomesenteric thrombosis, and hostile abdomen: Performance and outcome analysis. Clin. Transplant. 2019, 33, e13645. [Google Scholar] [CrossRef]
- Xie, K.; Yang, H.; Wang, S.; Xiao, C.; Lan, T.; Jiang, H.; Li, S.; Tu, H.; Yang, J.; Lyv, T.; et al. Comparing the efficacy and safety of thromboprophylaxis with enoxaparin versus normal saline after liver transplantation: A randomized clinical trial. Br. J. Surg. 2025, 112, znae325. [Google Scholar] [CrossRef]
- De Pietri, L.; Montalti, R.; Nicolini, D.; Troisi, R.I.; Moccheggiani, F.; Vivarelli, M. Perioperative thromboprophylaxis in liver transplant patients. World J. Gastroenterol. 2018, 24, 2931–2948. [Google Scholar] [CrossRef]
- Bos, I.; Blondeau, M.; Wouters, D.; Camus, C.; Houssel-Debry, P.; van Der Plas, W.S.; Nieuwenhuis, L.M.; Bardou-Jacquet, E.; Lisman, T.; de Meijer, V.E.; et al. Therapeutic anticoagulation after liver transplantation is not useful among patients with pre-transplant Yerdel-grade I/II portal vein thrombosis: A two-center retrospective study. J. Thromb. Haemost. 2021, 19, 2760–2771. [Google Scholar] [CrossRef]
- Yeo, J.W.; Law, M.S.N.; Lim, J.C.L.; Ng, C.H.; Tan, D.J.H.; Tay, P.W.L.; Syn, N.; Tham, H.Y.; Huang, D.Q.; Siddiqui, M.S.; et al. Meta-analysis and systematic review: Prevalence, graft failure, mortality, and post-operative thrombosis in liver transplant recipients with pre-operative portal vein thrombosis. Clin. Transplant. 2022, 36, e14520. [Google Scholar] [CrossRef]
- Brown, M.A.; Donahue, L.; Gueyikian, S.; Hu, J.; Huffman, S. Endovascular transsplenic recanalization with angioplasty and stenting of an occluded main portal vein in an adult liver transplant recipient. Radiol. Case Rep. 2020, 15, 615–623. [Google Scholar] [CrossRef] [PubMed]
- Zhu, K.; Meng, X.; Zhou, B.; Qian, J.; Huang, W.; Deng, M.; Shan, H. Percutaneous transsplenic portal vein catheterization: Technical procedures, safety, and clinical applications. J. Vasc. Interv. Radiol. 2013, 24, 518–527. [Google Scholar] [CrossRef]
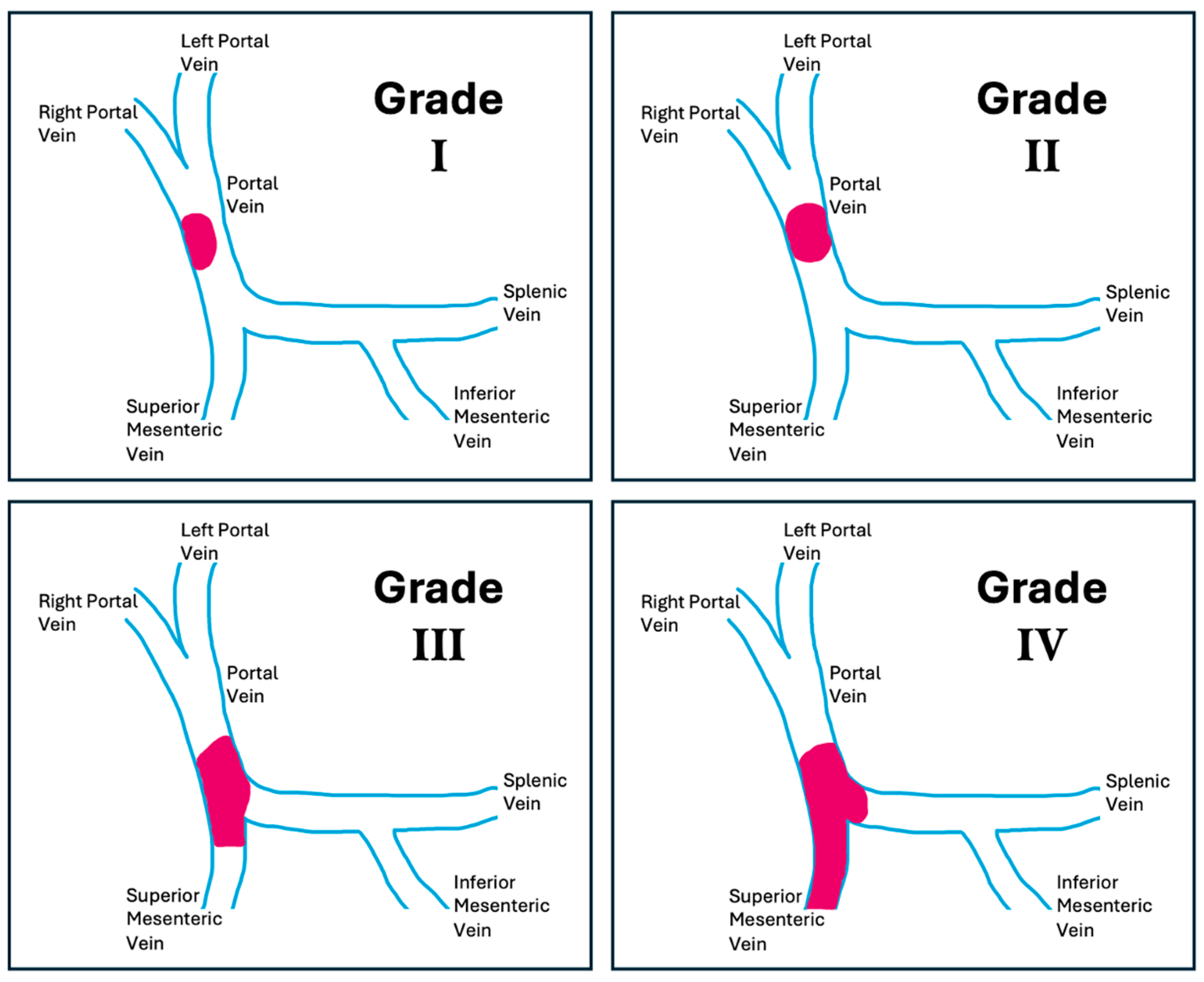


| Grade | Description |
|---|---|
| Grade I | <50% of vessel lumen thrombosis of PV, with or without minimal extension to the SMV |
| Grade II | >50% occlusion of the PV, including total occlusions, with or without minimal extension to the SMV |
| Grade III | Complete thrombosis of both PV and proximal SMV (distal SMV is open) |
| Grade IV | Complete thrombosis of the PV as well as proximal and distal SMV |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sancio, J.B.; Ponte, R.V.; Beduschi, T.; Soma, D. Portal Vein Thrombosis, Management and Approaches in Liver Transplantation: A Narrative Review. J. Clin. Med. 2025, 14, 6100. https://doi.org/10.3390/jcm14176100
Sancio JB, Ponte RV, Beduschi T, Soma D. Portal Vein Thrombosis, Management and Approaches in Liver Transplantation: A Narrative Review. Journal of Clinical Medicine. 2025; 14(17):6100. https://doi.org/10.3390/jcm14176100
Chicago/Turabian StyleSancio, João Bernardo, Raul Valério Ponte, Thiago Beduschi, and Daiki Soma. 2025. "Portal Vein Thrombosis, Management and Approaches in Liver Transplantation: A Narrative Review" Journal of Clinical Medicine 14, no. 17: 6100. https://doi.org/10.3390/jcm14176100
APA StyleSancio, J. B., Ponte, R. V., Beduschi, T., & Soma, D. (2025). Portal Vein Thrombosis, Management and Approaches in Liver Transplantation: A Narrative Review. Journal of Clinical Medicine, 14(17), 6100. https://doi.org/10.3390/jcm14176100








