Risk Factors for Perioperative Urinary Tract Infection After Living Donor Kidney Transplantation Characterized by High Prevalence of Desensitization Therapy: A Single-Center Analysis
Abstract
1. Introduction
2. Materials and Methods
2.1. Patients (Recipients)
2.2. Perioperative UTI
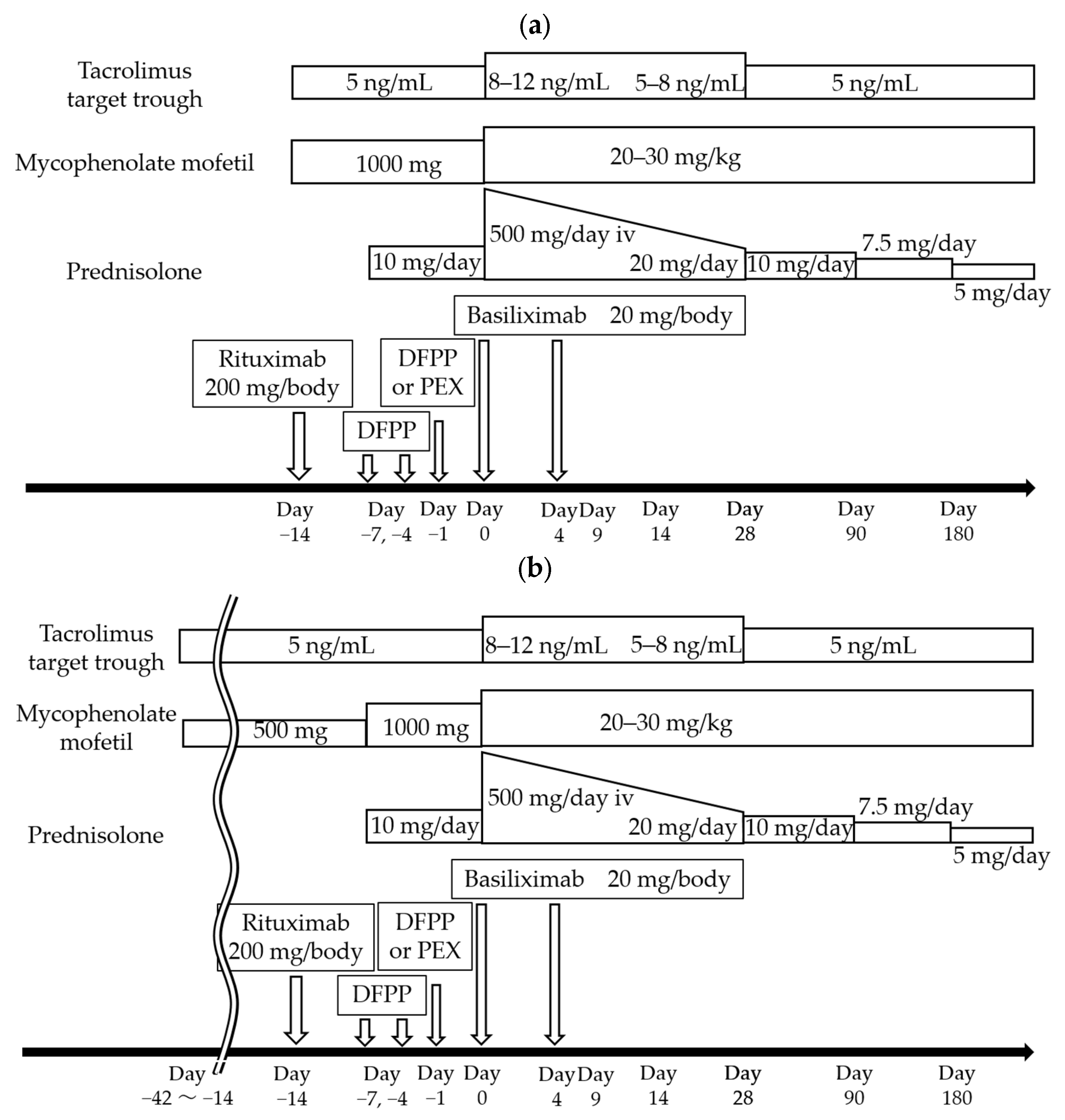
2.3. Immunosuppressive Therapy Regimens
2.4. Standard Procedures of LDKT
2.5. Bacteriological Examination and Prophylactic Antimicrobial Agents
2.6. Statistical Methods and Analyses
2.7. Ethics
3. Results
3.1. Recipient Characteristics
3.2. Antimicrobial Prophylaxis and Results of the Procedure
3.3. Perioperative UTIs and Outcomes
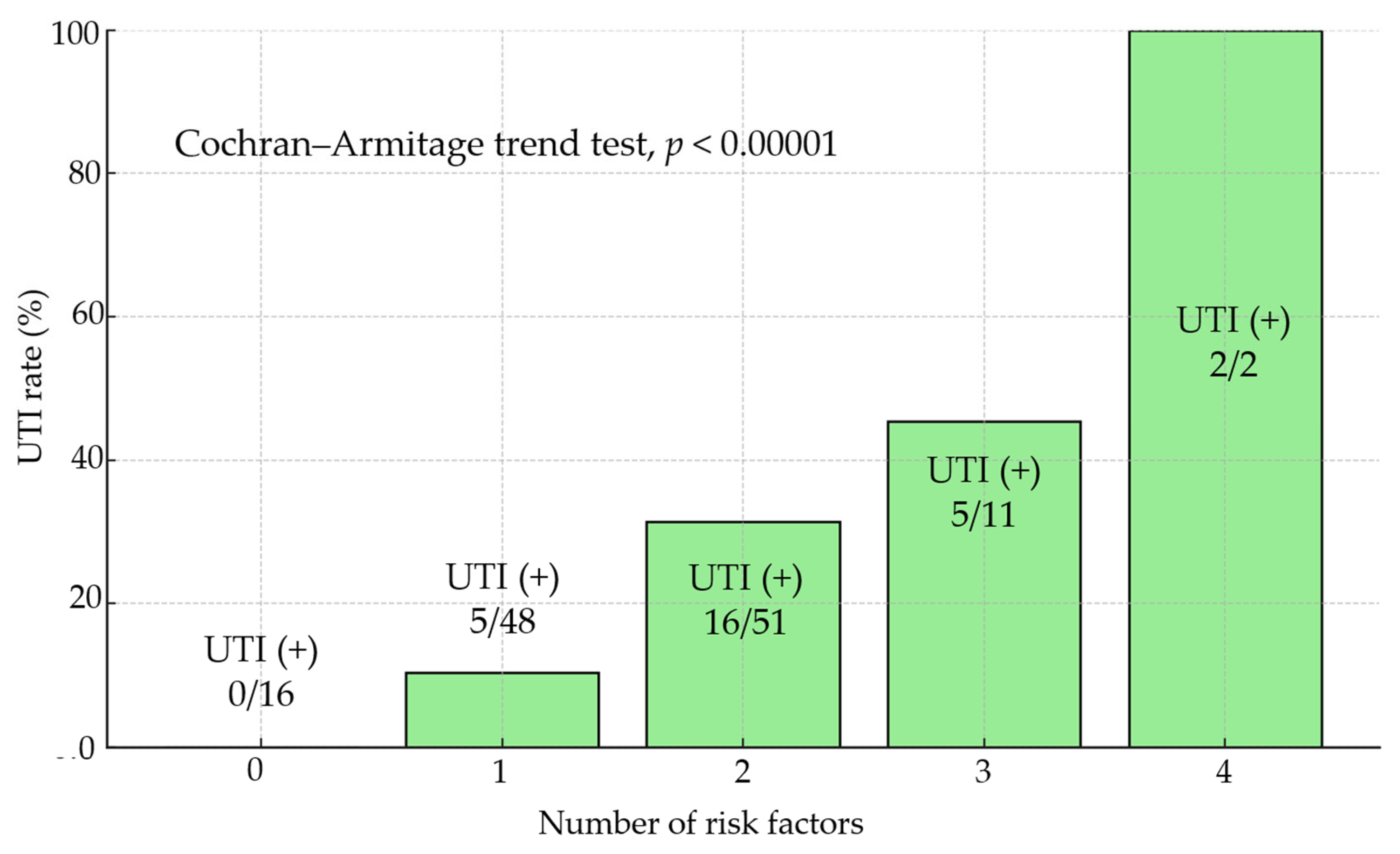
3.4. Risk Factors for Perioperative UTIs
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| UTI | Urinary Tract Infection |
| LDKT | living donor kidney transplantation |
| DSA | donor-specific antibody |
| NLR | neutrophil-to-lymphocyte ratio |
| WIT | warm ischemic time |
| BMI | body mass index |
| HLA | human leukocyte antigen |
| CNI | calcineurin inhibitor |
| MMF | mycophenolate mofetil |
| DFPP | double filtration plasmapheresis |
| PEX | plasma exchange |
| FSGS | focal segmental glomerulosclerosis |
| IVIG | intravenous immunoglobulin |
| DM | diabetes mellitus |
| ESBL | extended-spectrum beta-lactamase |
| AUC | area under the curve |
| ASA | American Society of Anesthesiologists |
| ECD | expanded criteria donors |
| DGF | delayed graft function |
| SSI | Surgical Site Infection |
| CKD | Chronic Kidney Disease |
| VUR | vesicoureteral reflux |
| DIC | disseminated intravascular coagulation |
| RI | Remote Infection |
| AMR | antimicrobial resistance |
| POD | postoperative day |
| TMP-SMX | trimethoprim-sulfamethoxazole |
References
- Lee, J.R.; Bang, H.; Dadhania, D.; Hartono, C.; Aull, M.J.; Satlin, M.; August, P.; Suthanthiran, M.; Muthukumar, T. Independent risk factors for urinary tract infection and for subsequent bacteremia or acute cellular rejection: A single-center report of 1166 kidney allograft recipients. Transplantation 2013, 96, 732–738. [Google Scholar] [CrossRef]
- Takahashi, K. Recent findings in ABO-incompatible kidney transplantation: Classification and therapeutic strategy for acute antibody-mediated rejection due to ABO-blood-group-related antigens during the critical period preceding the establishment of accommodation. Clin. Exp. Nephrol. 2007, 11, 128–141. [Google Scholar] [CrossRef] [PubMed]
- de Weerd, A.; Vonk, A.; van der Hoek, H.; van Groningen, M.; Weimar, W.; Betjes, M.; van Agteren, M. Late antibody-mediated rejection after ABO-incompatible kidney transplantation during Gram-negative sepsis. BMC Nephrol. 2014, 15, 31. [Google Scholar] [CrossRef]
- Pellé, G.; Vimont, S.; Levy, P.P.; Hertig, A.; Ouali, N.; Chassin, C.; Arlet, G.; Rondeau, E.; Vandewalle, A. Acute pyelonephritis represents a risk factor impairing long-term kidney graft function. Am. J. Transpl. 2007, 7, 899–907. [Google Scholar] [CrossRef]
- Nishi, S.P.; Valentine, V.G.; Duncan, S. Emerging bacterial, fungal, and viral respiratory infections in transplantation. Infect. Dis. Clin. N. Am. 2010, 24, 541–555. [Google Scholar] [CrossRef]
- Adamska, Z.; Karczewski, M.; Cichańska, L.; Więckowska, B.; Małkiewicz, T.; Mahadea, D.; Stronka, M. Bacterial Infections in Renal Transplant Recipients. Transpl. Proc. 2015, 47, 1808–1812. [Google Scholar] [CrossRef]
- Rabkin, D.G.; Stifelman, M.D.; Birkhoff, J.; Richardson, K.A.; Cohen, D.; Nowygrod, R.; Benvenisty, A.; Hardy, M. Early catheter removal decreases incidence of urinary tract infections in renal transplant recipients. Transpl. Proc. 1998, 30, 4314–4316. [Google Scholar] [CrossRef]
- Capocasale, E.; De Vecchi, E.; Mazzoni, M.P.; Dalla Valle, R.; Pellegrino, C.; Ferretti, S.; Sianesi, M.; Iaria, M. Surgical site and early urinary tract infections in 1000 kidney transplants with antimicrobial perioperative prophylaxis. Transpl. Proc. 2014, 46, 3455–3458. [Google Scholar] [CrossRef]
- Orlando, G.; Manzia, T.M.; Sorge, R.; Iaria, G.; Angelico, R.; Sforza, D.; Toti, L.; Peloso, A.; Patel, T.; Katari, R.; et al. One-shot versus multidose perioperative antibiotic prophylaxis after kidney transplantation: A randomized, controlled clinical trial. Surgery 2015, 157, 104–110. [Google Scholar] [CrossRef]
- The Japan Society for Transplantation. Transplantation Fact Book 2023. Available online: http://www.asas.or.jp/jst/pdf/factbook/factbook2023.pdf (accessed on 31 July 2025). (In Japanese).
- Japanese Society for Clinical Renal Transplantation, The Japan Society for Transplantation. Annual Progress Report from the Japanese Renal Transplant Registry: Number of Renal Transplantations in 2023 and Follow-Up Survey. Available online: https://www.jstage.jst.go.jp/article/jst/59/3/59_217/_article/-char/ja/ (accessed on 31 July 2025).
- Takahashi, K.; Saito, K.; Takahara, S.; Fuchinoue, S.; Yagisawa, T.; Aikawa, A.; Watarai, Y.; Yoshimura, N.; Tanabe, K.; Morozumi, K.; et al. Results of a multicenter prospective clinical study in Japan for evaluating efficacy and safety of desensitization protocol based on rituximab in ABO-incompatible kidney transplantation. Clin. Exp. Nephrol. 2017, 21, 705–713. [Google Scholar] [CrossRef]
- Morath, C.; Zeier, M.; Döhler, B.; Opelz, G.; Süsal, C. ABO-Incompatible Kidney Transplantation. Front. Immunol. 2017, 8, 234. [Google Scholar] [CrossRef]
- Yamamoto, S.; Shigemura, K.; Kiyota, H.; Wada, K.; Hayami, H.; Yasuda, M.; Takahashi, S.; Ishikawa, K.; Hamasuna, R.; Arakawa, S.; et al. Essential Japanese guidelines for the prevention of perioperative infections in the urological field: 2015 edition. Int. J. Urol. 2016, 23, 814–824. [Google Scholar] [CrossRef]
- Kanda, Y. Investigation of the freely available easy-to-use software ‘EZR’ for medical statistics. Bone Marrow Transpl. 2013, 48, 452–458. [Google Scholar] [CrossRef] [PubMed]
- Meena, P.; Bhargava, V.; Rana, D.S.; Bhalla, A.K. Urinary tract infection in renal transplant recipient: A clinical comprehensive review. Saudi J. Kidney Dis. Transpl. 2021, 32, 307–317. [Google Scholar] [CrossRef] [PubMed]
- Bratzler, D.W.; Dellinger, E.P.; Olsen, K.M.; Perl, T.M.; Auwaerter, P.G.; Bolon, M.K.; Fish, D.N.; Napolitano, L.M.; Sawyer, R.G.; Slain, D.; et al. Clinical practice guidelines for antimicrobial prophylaxis in surgery. Am. J. Health Syst. Pharm. 2013, 70, 195–283. [Google Scholar] [CrossRef]
- Kälble, T.; Lucan, M.; Nicita, G.; Sells, R.; Burgos Revilla, F.J.; Wiesel, M. EAU guidelines on renal transplantation. Eur. Urol. 2005, 47, 156–166. [Google Scholar] [CrossRef] [PubMed]
- Røine, E.; Bjørk, I.T.; Oyen, O. Targeting risk factors for impaired wound healing and wound complications after kidney transplantation. Transpl. Proc. 2010, 42, 2542–2546. [Google Scholar] [CrossRef]
- Trivin, C.; Tran, A.; Moulin, B.; Choukroun, G.; Gatault, P.; Courivaud, C.; Augusto, J.-F.; Ficheux, M.; Vigneau, C.; Thervet, E.; et al. Infectious complications of a rituximab-based immunosuppressive regimen in patients with glomerular disease. Clin. Kidney J. 2017, 10, 461–469. [Google Scholar] [CrossRef]
- Schrezenmeier, E.; Budde, K.; Staeck, O.; Lehner, L.; Duerr, M.; Khadzhynov, D.; Dörner, T.; Halleck, F. Incidence of Infectious Disease and Malignancies After Rituximab Therapy in Kidney Transplant Recipients: Results From a Cohort in Germany. Transpl. Proc. 2017, 49, 2269–2273. [Google Scholar] [CrossRef]
- Kamar, N.; Milioto, O.; Puissant-Lubrano, B.; Esposito, L.; Pierre, M.C.; Mohamed, A.O.; Lavayssière, L.; Cointault, O.; Ribes, D.; Cardeau, I.; et al. Incidence and predictive factors for infectious disease after rituximab therapy in kidney-transplant patients. Am. J. Transpl. 2010, 10, 89–98. [Google Scholar] [CrossRef]
- Tydén, G.; Kumlien, G.; Efvergren, M. Present techniques for antibody removal. Transplantation 2007, 84 (Suppl. S12), S27–S29. [Google Scholar] [CrossRef] [PubMed]
- Melexopoulou, C.; Filiopoulos, V.; Marinaki, S. Therapeutic apheresis in renal transplantation: An update. Transfus. Apher. Sci. 2024, 63, 103844. [Google Scholar] [CrossRef] [PubMed]
- Chung, B.H.; Yun, J.T.; Ha, S.E.; Kim, J.I.; Moon, I.S.; Choi, B.S.; Park, C.; Kim, Y.; Yang, C. Combined use of rituximab and plasmapheresis pre-transplant increases post-transplant infections in renal transplant recipients with basiliximab induction therapy. Transpl. Infect. Dis. 2013, 15, 559–568. [Google Scholar] [CrossRef] [PubMed]
- Lynch, R.J.; Ranney, D.N.; Shijie, C.; Lee, D.S.; Samala, N.; Englesbe, M.J. Obesity, surgical site infection, and outcome following renal transplantation. Ann. Surg. 2009, 250, 1014–1020. [Google Scholar] [CrossRef]
- Semins, M.J.; Shore, A.D.; Makary, M.A.; Weiner, J.; Matlaga, B.R. The impact of obesity on urinary tract infection risk. Urology 2012, 79, 266–269. [Google Scholar] [CrossRef]
- Lu, J.L.; Kalantar-Zadeh, K.; Ma, J.Z.; Quarles, L.D.; Kovesdy, C.P. Association of body mass index with outcomes in patients with CKD. J. Am. Soc. Nephrol. 2014, 25, 2088–2096. [Google Scholar] [CrossRef]
- Yamamoto, T.; Nakayama, M.; Miyazaki, M.; Sato, H.; Matsushima, M.; Sato, T.; Ito, S. Impact of lower body mass index on risk of all-cause mortality and infection-related death in Japanese chronic kidney disease patients. BMC Nephrol. 2020, 21, 244. [Google Scholar] [CrossRef]
- Ozato, N.; Saito, S.; Yamaguchi, T.; Katashima, M.; Tokuda, I.; Sawada, K.; Katsuragi, Y.; Imoto, S.; Ihara, K.; Nakaji, S. Association between Nutrients and Visceral Fat in Healthy Japanese Adults: A 2-Year Longitudinal Study Brief Title: Micronutrients Associated with Visceral Fat Accumulation. Nutrients 2019, 11, 2698. [Google Scholar] [CrossRef]
- Ogawa, W.; Hirota, Y.; Miyazaki, S.; Nakamura, T.; Ogawa, Y.; Shimomura, I.; Yamauchi, T.; Yokote, K. Definition, criteria, and core concepts of guidelines for the management of obesity disease in Japan. Endocr. J. 2024, 71, 223–231. [Google Scholar] [CrossRef]
- World Health Organization. Obesity and Overweight. Available online: https://www.who.int/news-room/fact-sheets/detail/obesity-and-overweight (accessed on 31 July 2025).
- Sheetz, K.H.; Zhao, L.; Holcombe, S.A.; Wang, S.C.; Reddy, R.M.; Lin, J.; Orringer, M.B.; Chang, A.C. Decreased core muscle size is associated with worse patient survival following esophagectomy for cancer. Dis. Esophagus 2013, 26, 716–722. [Google Scholar] [CrossRef]
- Lutz, C.T.; Quinn, L.S. Sarcopenia, obesity, and natural killer cell immune senescence in aging: Altered cytokine levels as a common mechanism. Aging 2012, 4, 535–546. [Google Scholar] [CrossRef]
- Chen, J.L.; Lee, M.C.; Kuo, H.C. Reduction of cystometric bladder capacity and bladder compliance with time in patients with end-stage renal disease. J. Formos. Med. Assoc. 2012, 111, 209–213. [Google Scholar] [CrossRef] [PubMed]
- Hotta, K.; Miura, M.; Wada, Y.; Fukuzawa, N.; Iwami, D.; Sasaki, H.; Seki, T.; Harada, H. Atrophic bladder in long-term dialysis patients increases the risk for urological complications after kidney transplantation. Int. J. Urol. 2017, 24, 314–319. [Google Scholar] [CrossRef] [PubMed]
- Cheng, S.F.; Jiang, Y.H.; Kuo, H.C. Urothelial Dysfunction and Chronic Inflammation are Associated With Increased Bladder Sensation in Patients With Chronic Renal Insufficiency. Int. Neurourol. J. 2018, 22 (Suppl. S1), S46–S54. [Google Scholar] [CrossRef] [PubMed]
- Hosseinpour, M.; Pezeshgi, A.; Mahdiabadi, M.Z.; Sabzghabaei, F.; Hajishah, H.; Mahdavynia, S. Prevalence and risk factors of urinary tract infection in kidney recipients: A meta-analysis study. BMC Nephrol. 2023, 24, 284. [Google Scholar] [CrossRef]
- Pergialiotis, V.; Papalios, T.; Haidopoulos, D.; Papapanagiotou, A.; Vlachos, A.; Rodolakis, A.; Thomakos, N. Pre-Operative Neutrophil-to-Lymphocyte Ratio as a Predictor of Post-Operative Infectious Morbidity in Gynecologic Oncology Patients. Surg. Infect. 2023, 24, 390–396. [Google Scholar] [CrossRef]
- Mohri, Y.; Tanaka, K.; Toiyama, Y.; Ohi, M.; Yasuda, H.; Inoue, Y.; Kusunoki, M. Impact of Preoperative Neutrophil to Lymphocyte Ratio and Postoperative Infectious Complications on Survival After Curative Gastrectomy for Gastric Cancer: A Single Institutional Cohort Study. Medicine 2016, 95, e3125. [Google Scholar] [CrossRef]
- Lu, H.; Yu, C.; Maimaiti, M.; Li, G. The predictive value of perioperative circulating markers on surgical complications in patients undergoing robotic-assisted radical prostatectomy. World J. Surg. Oncol. 2023, 21, 179. [Google Scholar] [CrossRef]
- Foley, M.E.; Vinson, A.J.; Skinner, T.A.A.; Kiberd, B.A.; Tennankore, K.K. The Impact of Combined Warm and Cold Ischemia Time on Post-transplant Outcomes. Can. J. Kidney Health Dis. 2023, 10, 20543581231178960. [Google Scholar] [CrossRef]
- Alshaikh, E.A.; Astor, B.C.; Muth, B.; Jorgenson, M.; Swanson, K.; Garg, N.; Aziz, F.; Mohamed, M.; Mandelbrot, D.; Parajuli, S. Delayed Graft Function Among Kidney Transplant Recipients Is Associated With an Increased Risk of Urinary Tract Infection and BK Viremia. Transpl. Direct. 2023, 9, e1526. [Google Scholar] [CrossRef]
- Pontrelli, P.; Grandaliano, G.; Van Kooten, C. Editorial: Kidney Transplantation and Innate Immunity. Front. Immunol. 2020, 11, 603982. [Google Scholar] [CrossRef]
- Faba, O.R.; Boissier, R.; Budde, K.; Figueiredo, A.; Hevia, V.; García, E.L.; Regele, H.; Zakri, R.H.; Olsburgh, J.; Bezuidenhout, C.; et al. European Association of Urology Guidelines on Renal Transplantation: Update 2024. Eur. Urol. Focus. 2024; in press. [Google Scholar] [CrossRef]
- Lightner, D.J.; Wymer, K.; Sanchez, J.; Kavoussi, L. Best Practice Statement on Urologic Procedures and Antimicrobial Prophylaxis. J. Urol. 2020, 203, 351–356. [Google Scholar] [CrossRef]
- Mitsui, M.; Sadahira, T.; Nagasaki, N.; Maruyama, Y.; Sekito, T.; Iwata, T.; Katayama, S.; Bekku, K.; Araki, M. Postoperative infections after robotic-assisted radical prostatectomy in a single large institution: Effect of type and duration of prophylactic antibiotic administration. Int. J. Urol. 2025, 32, 258–263. [Google Scholar] [CrossRef] [PubMed]
- Rosado-Canto, R.; Parra-Avila, I.; Tejeda-Maldonado, J.; Kauffman-Ortega, C.; Rodriguez-Covarrubias, F.T.; Trujeque-Matos, M.; Cruz-Martínez, R.; Maravilla-Franco, E.; Criollo-Mora, E.; Arreola-Guerra, J.M.; et al. Perioperative fosfomycin disodium prophylaxis against urinary tract infection in renal transplant recipients: A randomized clinical trial. Nephrol. Dial. Transpl. 2020, 35, 1996–2003. [Google Scholar] [CrossRef]
- Velioglu, A.; Guneri, G.; Arikan, H.; Asicioglu, E.; Tigen, E.T.; Tanidir, Y.; Tinay, I.; Yegen, C.; Tuglular, S.; Stepkowski, S. Incidence and risk factors for urinary tract infections in the first year after renal transplantation. PLoS ONE 2021, 16, e0251036. [Google Scholar] [CrossRef] [PubMed]
- Sakaeda, K.; Sadahira, T.; Maruyama, Y.; Iwata, T.; Watanabe, M.; Wada, K.; Araki, M. The Genotypic and Phenotypic Characteristics Contributing to Flomoxef Sensitivity in Clinical Isolates of ESBL-Producing, E. coli Strains from Urinary Tract Infections. Antibiotics 2023, 12, 522. [Google Scholar] [CrossRef] [PubMed]
- Moller, J.K.; Sørensen, M.; Hardahl, C.; Pappalardo, F. Prediction of risk of acquiring urinary tract infection during hospital stay based on machine-learning: A retrospective cohort study. PLoS ONE 2021, 16, e0248636. [Google Scholar] [CrossRef]
- Alizadeh, N.; Vahdat, K.; Shashaani, S.; Swann, J.L.; Özaltιn, O.Y.; Villavicencio, G.P. Risk score models for urinary tract infection hospitalization. PLoS ONE 2024, 19, e0290215. [Google Scholar] [CrossRef]
- Trang, V.A.V.; Truyen, T.T.T.T.; Nguyen, M.T.; Mai, H.P.; Phan, T.C.; Phan, S.H.; Le Nguyen, H.M.; Nguyen, H.-D.T.; Le, N.H.D.; Tu, M.N.; et al. Development of a novel risk score for diagnosing urinary tract infections: Integrating Sysmex UF-5000i urine fluorescence flow cytometry with urinalysis. PLoS ONE 2025, 20, e0323664. [Google Scholar] [CrossRef]

| N | 128 | |
|---|---|---|
| Median age (IQR) a | 43 | (32.8–57) |
| Median BMI b, kg/m2 (IQR) | 21.7 | (19.2–24.4) |
| Median duration of dialysis before LDKT c (n = 91), year (IQR) | 1.3 | (0.5–3) |
| Pre-LDKT s-albumin, d g/dL (IQR) | 3.9 | (3.5–4.1) |
| Pre-LDKT Neutrophil-to-lymphocyte ratio (NLR), (IQR) | 2.7 | (2–3.8) |
| Sex, n (%) | ||
| male | 86 | (67) |
| female | 42 | (33) |
| ASA e physical status classification, n (%) | ||
| 2 | 5 | (4) |
| 3 | 119 | (93) |
| 4 | 4 | (3) |
| Diabetes Mellitus, n (%) | 27 | (21) |
| Diabetic nephropathy, n (%) | 24 | (19) |
| ABO-incompatible, n (%) | 49 | (38) |
| Donor-specific antibody, n (%) | 25 | (20) |
| Administration of rituximab, n (%) | 90 | (70) |
| Plasmapheresis + rituximab before LDKT, n (%) | 74 | (58) |
| Pre-emptive kidney transplantation, n (%) | 37 | (29) |
| Positive urine culture before surgery, n (%) | 20 | (16) |
| Donor | ||
| Median age (IQR) | 60 | (53–67) |
| Expanded criteria donor, n (%) | 69 | (54) |
| N | 128 | ||
|---|---|---|---|
| Type of prophylactic antimicrobials, cases (%) | |||
| Ampicillin/sulbactam | 9 | (7) | |
| Cefazolin | 111 | (87) | |
| Cefotiam | 1 | (1) | |
| Cefmetazole | 1 | (1) | |
| Flomoxef | 3 | (2) | |
| Levofloxacin | 3 | (2) | |
| Duration of prophylactic antimicrobials, cases (%) | |||
| Single-dose | 86 | (67) | |
| Within 72 h (up to POD a 2) | 42 | (33) | |
| Median operative time, min (IQR) | 490 | (427–559) | |
| Median EBL b, mL (IQR) | 178 | (100–318) | |
| Median WIT c, min (IQR) | 5 | (3.3–6.7) | |
| With retroperitoneal drain, cases (%) | 128 | (100) | |
| Median duration of placement, days (IQR) | 5 | (4–7) | |
| Urethral catheter, cases (%) | 128 | (100) | |
| Median duration of placement, days (IQR) | 7 | (7–8) | |
| Subcutaneous drain placement, cases (%) | 37 | (29) | |
| Median duration of placement, days (IQR) | 6 | (4–9) | |
| Surgical site infection, n (%) | 0 | (0) | |
| Urinary tract infection, n (%) | 28 | (22) | |
| Lymphocele, n (%) | 5 | (4) | |
| Viral infection, n (%) | 2 | (2) | |
| Fungal infection, n (%) | 1 | (1) | |
| Delayed graft function, n (%) | 1 | (1) | |
| Acute rejection within 1-month, n (%) | 2 | (2) | |
| Graft loss within 1-month, n (%) | 0 | (0) | |
| 1-month postoperative serum-Creatinine, mg/dL (IQR) | 1.3 | (1.1–1.7) | |
| UTI (−) | UTI (+) | |||
|---|---|---|---|---|
| N | 100 | 28 | ||
| Median age (IQR) | 47 | (31–57) | 44.5 | (36.8–57.3) |
| Median age of donor (IQR) | 59.5 | (53–66.3) | 62.5 | (54.3–68) |
| Median BMI, kg/m2 (IRQ) | 21.7 | (19.5–24.3) | 21.1 | (18.2–25.6) |
| Median duration of dialysis before LDKT (n = 91), years (IQR) | 0.5 | (0–1.7) n = 68 | 1.3 | (0.2–3) n = 23 |
| Median pre-LDKT s-albumin, mg/dL (IQR) | 3.9 | (3.5–4.2) | 3.8 | (3.5–4.1) |
| Median pre-LDKT NLR, (IQR) | 2.5 | (2–3.6) | 3.3 | (2.1–4) |
| Sex, n (%) | ||||
| male | 68 | (68) | 18 | (64) |
| female | 32 | (32) | 10 | (36) |
| ASA risk, n (%) | ||||
| 2 | 5 | (5) | 0 | (0) |
| 3 | 93 | (93) | 26 | (93) |
| 4 | 2 | (2) | 2 | (7) |
| Diabetic Mellitus, n (%) | 19 | (19) | 8 | (29) |
| ABO incompatible LDKT, n (%) | 35 | (35) | 14 | (50) |
| Donor-specific antibody, n (%) | 15 | (15) | 9 | (32) |
| Administration of rituximab, n (%) | 67 | (67) | 23 | (82) |
| Plasmapheresis + rituximab before LDKT, n (%) | 52 | (52) | 22 | (79) |
| Pre-emptive kidney transplantation, n (%) | 32 | (32) | 5 | (18) |
| Positive urine culture before surgery, n (%) | 17 | (17) | 3 | (11) |
| Donor | ||||
| Median age (IQR) | 59.5 | (53–66.3) | 62.5 | (54.3–68) |
| Expanded criteria donor, n (%) | 51 | (51) | 18 | (64) |
| Type of antimicrobials, n (%) | ||||
| cefazolin | 84 | (84) | 27 | (96) |
| ampicillin/sulbactam, FMOX, LVFX, CTM, CMZ | 16 | (16) | 1 | (4) |
| Duration of prophylactic antimicrobials, n (%) | ||||
| Single-dose | 65 | (65) | 21 | (75) |
| Within 72 h (up to POD2) | 35 | (35) | 7 | (25) |
| Median operative time, min (IQR) | 493 | (442–577) | 467 | (404–555) |
| Median EBL, mL (IQR) | 190 | (100–333) | 145 | (100–224) |
| Median WIT, min (IQR) | 5 | (3.3–6.5) | 5.1 | (3–7.9) |
| Median duration of pelvic drain placement, days (IQR) | 5 | (4–7) | 5 | (4–8) |
| Median duration of urethral catheter placement, days (IQR) | 7 | (7–7) | 7 | (7–13) |
| Median duration of subcutaneous drain placement (n = 37), days (IQR) | 5.5 | (4–8.3) n = 29 | 7.5 | (4.5–9.3) n = 8 |
| Urine Samples | Number of Isolates |
|---|---|
| Escherichia coli | 10 |
| Escherichia coli (QREC a) | 4 |
| Escherichia coli (ESBL b) | 5 |
| Klebsiella pneumoniae | 3 |
| Enterobactor cloacae | 3 |
| Proteus mirabilis | 2 |
| Pseudomonas aeruginosa | 1 |
| Citrobactor speies | 1 |
| CNS c | 2 |
| Univariate Analysis | |||
|---|---|---|---|
| Variable | OR a | 95% CI b | p |
| Age (<45) | 0.85 | 0.37–1.97 | 0.708 |
| Sex (Female) | 1.18 | 0.49–2.85 | 0.712 |
| BMI (<20, or 25<, kg/m2) | 3.14 | 1.32–7.46 | 0.01 |
| Duration of dialysis before LDKT (2.5<, years) | 2.71 | 1.07–6.89 | 0.036 |
| Serum-albumin (<3.9, g/dL) | 3.9 | 0.35–1.85 | 0.603 |
| ASA (3<) | 3.77 | 0.51–28.1 | 0.195 |
| Diabetes Mellitus | 1.71 | 0.65–4.45 | 0.276 |
| Pre-LDKT NLR (3≤) | 2.63 | 1.11–6.22 | 0.028 |
| Administration of rituximab | 2.27 | 0.79–6.49 | 0.128 |
| Desensitization (plasmapheresis + rituximab) | 3.38 | 1.26–9.06 | 0.015 |
| Positive urine culture before LDKT | 0.59 | 0.16–2.16 | 0.422 |
| Administration of cefazolin | 5.14 | 0.65–40.6 | 0.12 |
| Duration of prophylactic antimicrobials (single-dose) | 0.62 | 0.24–1.6 | 0.322 |
| Donor Age (60<) | 2.11 | 0.87–5.11 | 0.098 |
| Expanded criteria donor | 1.73 | 0.73–4.11 | 0.215 |
| WIT (7.8≤, min) | 2.93 | 1.06–8.12 | 0.038 |
| Duration of retroperitoneal drain placement (10<, days) | 1.45 | 0.47–4.5 | 0.515 |
| Duration of urethral catheter placement (7<, days) | 0.86 | 0.31–2.38 | 0.777 |
| Duration of double-J ureter stent placement (18<, days) | 0.59 | 0.23–1.53 | 0.279 |
| Multivariate Analysis | ||||
|---|---|---|---|---|
| Variable | OR | 95% CI | p | |
| Model I | BMI (<20, or 25<, kg/m2 ) | 3 | 1.18–7.63 | 0.021 |
| Duration of dialysis before LDKT (2.5<, years) | 2.88 | 1.04–8 | 0.042 | |
| Desensitization (plasmapheresis + rituximab) | 3.54 | 1.24–10.1 | 0.018 | |
| Pre-LDRT NLR (3≤) | 2.06 | 0.82–5.21 | 0.126 | |
| AUC = 0.759 | 0.665–0.854 | |||
| Model II | BMI (<20, or 25<, kg/m2 ) | 3.35 | 1.3–8.6 | 0.012 |
| Duration of dialysis before LDKT (2.5<, years) | 3.15 | 1.1–9 | 0.031 | |
| Desensitization (plasmapheresis + rituximab) | 4.31 | 1.46–12.8 | 0.008 | |
| WIT (7.8≤, min) | 3.53 | 1.1–11.3 | 0.033 | |
| AUC = 0.764 | 0.672–0.856 | |||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nishimura, S.; Inoue, S.; Sekito, T.; Tsuboi, I.; Tokunaga, M.; Yoshinaga, K.; Maruyama, Y.; Mitsui, Y.; Yamanoi, T.; Kawada, T.; et al. Risk Factors for Perioperative Urinary Tract Infection After Living Donor Kidney Transplantation Characterized by High Prevalence of Desensitization Therapy: A Single-Center Analysis. J. Clin. Med. 2025, 14, 6102. https://doi.org/10.3390/jcm14176102
Nishimura S, Inoue S, Sekito T, Tsuboi I, Tokunaga M, Yoshinaga K, Maruyama Y, Mitsui Y, Yamanoi T, Kawada T, et al. Risk Factors for Perioperative Urinary Tract Infection After Living Donor Kidney Transplantation Characterized by High Prevalence of Desensitization Therapy: A Single-Center Analysis. Journal of Clinical Medicine. 2025; 14(17):6102. https://doi.org/10.3390/jcm14176102
Chicago/Turabian StyleNishimura, Shingo, Shota Inoue, Takanori Sekito, Ichiro Tsuboi, Moto Tokunaga, Kasumi Yoshinaga, Yuki Maruyama, Yosuke Mitsui, Tomoaki Yamanoi, Tatsushi Kawada, and et al. 2025. "Risk Factors for Perioperative Urinary Tract Infection After Living Donor Kidney Transplantation Characterized by High Prevalence of Desensitization Therapy: A Single-Center Analysis" Journal of Clinical Medicine 14, no. 17: 6102. https://doi.org/10.3390/jcm14176102
APA StyleNishimura, S., Inoue, S., Sekito, T., Tsuboi, I., Tokunaga, M., Yoshinaga, K., Maruyama, Y., Mitsui, Y., Yamanoi, T., Kawada, T., Kubota, R., Sadahira, T., Tominaga, Y., Iwata, T., Katayama, S., Bekku, K., Edamura, K., Wada, K., Kobayashi, Y., & Araki, M. (2025). Risk Factors for Perioperative Urinary Tract Infection After Living Donor Kidney Transplantation Characterized by High Prevalence of Desensitization Therapy: A Single-Center Analysis. Journal of Clinical Medicine, 14(17), 6102. https://doi.org/10.3390/jcm14176102







